Abstract
c-Myc is a powerful trigger of β-cell apoptosis, proliferation, and dedifferentiation in rodent islets in vivo. In a transgenic mouse model, c-Myc induction causes rapid β-cell apoptosis and overt diabetes. When suppression of apoptosis is achieved by overexpression of Bcl-xL in an inducible model of c-Myc activation, a full spectrum of tumor development, including distant metastasis, occurs. Caspase-3 is a key pro-apoptotic protein involved in the execution phase of multiple apoptotic pathways. To test whether caspase-3 is an essential mediator of apoptosis in this model of tumorigenesis, we generated caspase-3 knock-out mice containing the inducible c-myc transgene (c-Myc+Casp3-/-). In contrast to Bcl-xL-overexpressing c-Myc+ mice, c-Myc+Casp3-/- mice remained euglycemic for up to 30 days of c-Myc activation, and there was no evidence of tumor formation. Interestingly, caspase-3 deletion also led to the suppression of proliferation, perhaps through regulation of the cell cycle inhibitory protein p27, suggesting a possible mechanism for maintaining a balance between suppression of apoptosis and excessive proliferation in the context of c-Myc activation. Additionally, c-Myc-activated Casp3-/- mice were protected from streptozotocin-induced diabetes. Our studies demonstrate that caspase-3 deletion confers protection from c-Myc-induced apoptosis and diabetes development without unwanted tumorigenic effects. These results may lead to further elucidation of the mechanisms of c-Myc biology relevant to β-cells, which may result in novel therapeutic strategies for diabetes.
Progressive β-cell insufficiency is a hallmark of both type 1 and 2 diabetes. Although the instigating factors that lead to β-cell failure in these two types of diabetes may differ, the apoptotic machinery that results in β-cell apoptosis is likely common. In type 2 diabetes, the decrease in functional β-cell mass involves both β-cell loss due to increased β-cell apoptosis (1) and a secretory/glucose-sensing defect in surviving β-cells (2). In animal models, chronic hyperglycemia leads to β-cell hypertrophy, loss of β-cell differentiation markers, as well as increased expression of the transcription factor c-Myc (3, 4).
c-Myc is a basic helix-loop-helix transcription factor that has been extensively studied as a proto-oncogene, but it also has a fundamental physiological role during development and in cell cycle progression in adulthood, particularly in tissues with high proliferative capacity. It is a potent inducer of both cell proliferation and apoptosis and can prevent cells from exiting the cell cycle (5). c-Myc appears to sensitize cells to apoptotic triggers by augmenting the death receptor pathway and priming the mitochondria to release cytochrome c (6, 7). The ultimate role of c-Myc is highly cell type- and context-specific (8, 9); however, in pancreatic β-cells, c-Myc appears to have a predominantly pro-apoptotic role. In in vitro models, prolonged culture in either low or high glucose induces expression of c-Myc and leads to caspase-dependent apoptosis (10, 11). In vivo, overexpression of c-Myc in the β-cells of transgenic mice leads to rapid proliferation, followed by apoptosis culminating in neonatal diabetes and death (12). However, this model induces c-Myc overexpression embryologically, as the transgene is under the control of the rat insulin promoter, which is expressed at day 9.5 of embryogenesis (13).
To study the role of c-Myc in adult β-cells, a transgenic model has been developed that allows for inducible c-Myc activation in tissues of interest (14). In this model, the c-myc gene under the control of a specific promoter is fused with the hormone-binding domain of the 4-hydroxytamoxifen-responsive mutant murine estrogen receptor, allowing for inducible c-Myc expression in the presence of tamoxifen. Pelengaris et al. (15) used this transgene under the direction of the rat insulin promoter to allow for the specific activation of c-Myc only in β-cells. Their study showed that c-Myc induction led to rapid β-cell proliferation, followed by massive apoptosis leading to diabetes development in 6-10 days. Furthermore, suppression of mitochondrial apoptosis by overexpression of the anti-apoptotic bcl-xL gene in β-cells revealed the oncogenic potential of c-Myc, and a full spectrum of tumor progression including distant metastasis was observed.
Caspases are evolutionarily conserved cysteine aspartate-specific proteases that play a crucial role in apoptosis. There are two main pathways of apoptosis: the extrinsic pathway, which involves the tumor necrosis factor family of receptors such as TNFR1 and Fas, and the intrinsic or mitochondrial pathway, which involves Bcl protein family members such as Bax and Bad (16, 17). Caspase-3 is one of the critical effector caspases involved in both pathways, and its activation leads to disablement of key structural proteins and important signaling, homeostatic, and repair enzymes, resulting in cell death (18). Several in vitro studies have suggested that caspase-3 activation is essential for β-cell apoptosis. Cultured islets were shown to undergo caspase-3-dependent apoptosis in response to activation of Fas, a receptor that is up-regulated in human islets in response to elevated glucose concentrations (19, 20).
Other studies have indicated that chronic hyperglycemia increases cell death through the intrinsic apoptotic pathway by activating Bax and caspase-3 (21). In vivo, loss of caspase-3 has been shown to protect mice from diabetes in a multiple low dose streptozotocin (MLDS)3 autoimmune diabetes model, implicating caspase-3-mediated β-cell apoptosis as a key initiating event in type 1 diabetes (22).
To examine the essential role of caspase-3 in the context of c-Myc activation, we generated and analyzed caspase-3 knock-out mice containing the inducible c-myc transgene (c-Myc+Casp3-/-). In contrast to the Bcl-xL overexpression model, these mice were protected from c-Myc-induced apoptosis with no evidence of diabetes or islet tumor development.
EXPERIMENTAL PROCEDURES
Mouse Protocol—Generation of Casp3-/- mice and Myc-ER(Tam) (Myc+) transgenic mice has been described previously (23). Myc+ mice were bred with Casp3-/- mice to generate c-Myc+Casp3+/-, and these mice were intercrossed to generate c-Myc+Casp3+/+, c-Myc+Casp3+/-, and c-Myc+Casp3-/- mice. A small number of Myc- mice were also produced using this breeding strategy. Control mice for all experiments were c-Myc+Casp3+/+ or c-Myc+Casp3+/- littermates. Mice were maintained on a C57BL/6 background. In this background, approximately half of the Casp3-/- mice survived to adulthood and appeared generally healthy. Genotyping was performed by PCR using ear clip DNA as described previously (23). The activity of the mice was not restricted, and they were maintained on a 12-h light/dark schedule. All mice were fed regular chow (Harlan Teklad). All protocols were approved by the Ontario Cancer Institute, Animal Resource Colony.
Activation of c-Myc and Blood Glucose Determination—The Myc-ER(Tam) transgene was activated by a daily injection of 1 mg of tamoxifen (Sigma) suspended in peanut oil (Sigma) at a final concentration of 1 mg/ml in adult mice (2-10 months old). Tamoxifen was injected intraperitoneally in all experimental mice daily for 1, 2, 6, and 30 days. Base-line (day 0) blood glucose was measured by the One Touch Ultra automated glucose meter (LifeScan) prior to each injection.
Metabolic Studies—Mice were fasted overnight for 14-16 h in duration prior to experiments. Blood glucose levels were determined from tail venous blood. Glucose tolerance tests were performed on fasted mice using a glucose dose of 1 g/kg of body weight injected intraperitoneally. Blood glucose measurements were obtained at 0, 15, 30, 45, 60, and 120 min after glucose injection.
Islet Isolation—Pancreatic islets were isolated as described previously (22). Briefly, 3 ml of collagenase (3 mg/ml; Sigma) was injected into the common bile duct, and pancreatic tissue was gently removed and digested in collagenase solution at 37 °C with shaking for 30 min. The digestion was stopped by ice-cold Hanks' balanced salt solution and passed through a filter. Islets were then handpicked under a dissecting microscope.
mRNA Isolation and Reverse Transcription (RT)-PCR—mRNA was extracted from isolated islets using TRIzol (Invitrogen) following the manufacturer's protocol and treated with RNase-free DNase (Invitrogen). Semiquantitative RT-PCR amplification was performed with a one-step RT-PCR kit (Invitrogen). p27, interleukin-1β (IL-1β), and β-actin cDNA were amplified by PCR using specific primers. Densitometric analysis was performed using ImageJ Version 1.37. To correct for differences in loading, we corrected the densitometric values of p27 and IL-1β cDNAs with the corresponding values of β-actin cDNA and calculated the p27:β-actin and IL-1β:β-actin ratios.
Western Blotting—Islets from mice at days 1 and 6 of tamoxifen injection as well as from noninjected littermates were isolated, and protein lysates were obtained as described previously (24). Lysates were separated by SDS-10% polyacrylamide gel electrophoresis and immunoblotted with antibodies for c-Myc (9E10; a gift of L. Penn); cleaved caspase-3 and caspase-3 (Cell Signaling); cyclin D1, Cdk-4, and p27 (Santa Cruz Biotechnology); and GLUT2 (Chemicon). Densitometric quantification of protein bands was performed using ImageJ Version 1.37. Samples were normalized to β-tubulin.
Immunohistochemistry, Immunofluorescence Staining, and Islet Morphometry—Pancreases were isolated from mice at days 0, 1, 2, 6, and 30 after tamoxifen injection. Pancreatic tissue was fixed for 24 h in 4% paraformaldehyde in 0.1 m phosphate-buffered saline (PBS), pH 7.4, at 4 °C. Samples were dehydrated and prepared as paraffin blocks. Seven-μm-thick sections were obtained at 100-150-μm intervals on at least three levels and stained with hematoxylin and eosin, synaptophysin (Roche Applied Science), and Ki67 (DAKO). Total islet area and total pancreatic area were determined from synaptophysin-stained sections by automated area counting of synaptophysin-stained area versus total pancreatic area and expressed as total islet area divided by total pancreatic area. The islet number was calculated by visualizing synaptophysin-stained sections by light microscopy and counting the number of islets present per section. Additionally, immunofluorescent staining was performed to detect insulin (DAKO) and glucagon (Sigma), which was visualized using a Zeiss inverted fluorescence microscope. To examine apoptotic cells, terminal deoxynucleotidyltransferase-mediated dUTP nick end labeling (TUNEL; Roche Applied Science) was performed as described previously (22).
Senescence-associated β-Galactosidase Activity Staining—The senescence-associated β-galactosidase assay was performed as described previously (25). Frozen tissue sections and cells were fixed using fresh 4% paraformaldehyde in PBS for 10 min at room temperature. After three washes with PBS, the tissue sections and cells were incubated at 37 °C overnight in a humidified chamber with freshly prepared senescence-associated β-galactosidase staining solution (1 mg/ml X-gal in dimethyl sulfoxide, 40 mmol/liter citric acid and phosphate buffer, pH 6.0, 5 mmol/liter potassium ferrocyanide, 5 mmol/liter potassium ferricyanide, 150 mmol/liter sodium chloride, 2 mmol/liter magnesium chloride). Cells were rinsed with PBS and overlaid with 70% ethanol prior to visualization on a Leica stereomicroscope. Images were analyzed using Image ProPlus Version 4.5.1 (Media Cybernetics, Bethesda, MD).
After staining, frozen sections to be costained with insulin were incubated for 1 h with the anti-insulin antibody (1:500 dilution; DAKO), followed by biotin-conjugated anti-rabbit IgG (Vector Laboratories) and streptavidin-horseradish peroxidase (ID Lab) as secondary antibodies and NovaRed (Vector Laboratories) as substrate. Slides were counterstained in Nuclear Fast Red for nuclei and mounted in Permount (Fisher).
Propidium Iodide Staining and Flow Cytometry—Isolated islets were trypsinized with 0.05% trypsin/EDTA (Invitrogen) for 10 min in a 37 °C CO2 incubator. RPMI 1640 medium with 10% fetal calf serum was added immediately afterward. Cells were centrifuged (1,200 rpm, 4 °C, 7 min), washed with cold PBS with 5% bovine serum albumin, and fixed with cold 100% ethanol. Cells were resuspended in PBS containing 50 μl/ml propidium iodide (Sigma), 0.1% Triton X-100, and 100 μg/ml DNase-free RNase A (Sigma). Samples were incubated in the dark for 1 h at 4 °C and quantified by flow cytometry (FACSCalibur). Cell cycle was evaluated with FlowJo Version 7.2.5.
Streptozotocin Protocol—Mice were injected intraperitoneally with streptozotocin (STZ; 40 mg of STZ/kg of body weight) for 5 consecutive days as described previously (22). Tail vein glucose was monitored weekly using a One Touch Ultra automated glucose meter. To activate c-Myc, mice were also injected daily with tamoxifen for 6 days prior to the first STZ injection, as well as throughout the 5-day STZ injection period and the following 4 weeks to maintain c-Myc activation throughout the duration of the experiment.
Statistical Analysis—Data are presented as means ± S.E. and were analyzed by one-sample t tests and independent sample t tests where appropriate. Data were analyzed using the statistical package SPSS for PC Version 14.0.
RESULTS
Generation of c-Myc+Casp3-/- Mice and c-myc Transgene Activation—Caspase-3 knock-out mice containing the inducible c-myc transgene in β-cells (c-Myc+Casp3-/-) were generated by breeding Casp3-/- mice with c-Myc+ animals. We observed efficient caspase-3 deletion in caspase-3 knock-out mice (Fig. 1A). We also observed an increase in c-Myc-ER protein expression upon 1- and 6-day injection with tamoxifen in control and c-Myc+Casp3-/- mice, respectively, indicating effective and sustained activation of the transgene with tamoxifen (Fig. 1B). Expression of the c-Myc transgenic protein was equal in control and c-Myc+Casp3-/- mice, indicating that the c-myc transgene was active in these animals at a similar level (Fig. 1B). After 1 day of c-Myc activation, we observed activated (cleaved) caspase-3 expression only in the isolated islets of the c-Myc+Casp3+/+ mice (Fig. 1C), further confirming the lack of functional caspase-3 in the pancreatic β-cells of the c-Myc+Casp3-/- mice.
FIGURE 1.
Effective caspase-3 deletion and c-Myc activation in c-Myc+Casp3-/- mice. A, Western blot and analysis showing caspase-3 deletion in isolated islets from c-Myc+Casp3-/- mice. ***, p < 0.001. B, Western blot showing inducible and sustained c-Myc activation after 1 day of tamoxifen (Tam) injection in c-Myc+Casp3+/+ islets and after 6 days of tamoxifen injection in c-Myc+Casp3-/- islets. An analysis of Western blots confirms similar levels of c-Myc expression in both genotypes. **, p < 0.005 (n = 3). C, immunoblot detecting expression of cleaved caspase-3 only in wild-type mice and confirming presence of activated caspase-3 after 1 day of tamoxifen treatment. *, p < 0.001 (n = 3). Results represent means ± S.E.
Loss of Caspase-3 Confers Protection from Diabetes Induced by c-Myc Activation without Leading to Tumor Formation—Mice were injected daily for 30 days with tamoxifen to activate the c-myc transgene as described previously (15). Control c-Myc+Casp3+/+ and c-Myc+Casp3+/- mice developed diabetes with blood glucose reaching above 20 mmol/liter within 3 days of c-Myc activation, and they remained hyperglycemic for the duration of the experiment (Fig. 2A). We observed complete islet ablation with a significant reduction in islet area and number in these mice by day 30 of c-Myc activation (Fig. 2, B-D). There was no difference in total pancreatic area between c-Myc+Casp3+/+ and c-Myc+Casp3-/- mice (Fig. 2E), indicating that the reduction in islet mass in c-Myc+Casp3+/+ mice was not due to decreased pancreas size. What little islet mass was present in the c-Myc+Casp3+/+ mice can be attributed to islet remnants rather than functional β-cells, as there was no evidence of insulin-staining cells, and the characteristic islet architecture was lost (Fig. 2B, middle panels). In fact, consistent with previous reports (15), complete islet ablation in c-Myc+Casp3+/+ was evident as early as 6 days following initial c-Myc activation (see Fig. 5A, middle panel). In contrast, c-Myc+Casp3-/- mice were protected from diabetes and remained euglycemic despite a sustained 30-day c-Myc activation. Islet mass was maintained (Fig. 2, C and D), and the islets retained the characteristic architecture, as α- and β-cell distribution remained intact (Fig. 2B, right panels). Furthermore, despite the absence of caspase-3 and sustained c-Myc activation, examination of sections immunostained with hematoxylin and eosin (data not shown), synaptophysin (Fig. 2B, upper panels), and glucagon and insulin (Fig. 2B, lower panels) revealed no evidence of the profound tumorigenesis seen in the Bcl-xL overexpression model (15). Furthermore, these mice with Bcl-xL overexpression and concomitant c-Myc activation have been shown to have increased inflammatory cytokine IL-1β mRNA transcript levels in isolated islets compared with control mice, which have been reported as a contributing factor to the tumorigenesis observed in this tumor model (26). Upon further examination of our mouse model, we found that there was no induction in IL-1β transcript levels upon c-Myc activation, and there was no difference between c-Myc+Casp3+/+ and c-Myc+-Casp3-/- islets with 2 days of c-Myc activation (Fig. 2F), which is in keeping with the lack of tumor development in these mice. Thus, in the context of c-Myc activation, caspase-3 absence confers protection from β-cell apoptosis and diabetes without causing unwanted tumorigenic effects associated with activation of this oncogene.
FIGURE 2.
Absence of caspase-3 protects against diabetes induced by c-Myc activation without leading to islet dedifferentiation or tumor formation. Mice were injected daily with 1 mg of tamoxifen for 30 days. A, blood glucose profile of mice receiving daily tamoxifen injections (n = 3 per genotype, except n = 5 for c-Myc+Casp3-/-; p < 0.001 for all time points above and including day 2). B, synaptophysin, glucagon, and insulin immunostaining of islets following a 30-day c-Myc activation. C and D, decreased total islet area (C)(n = 4 per genotype; *, p = 0.019) and decreased islet number (D)(n = 4 per genotype; **, p = 0.01) in c-Myc+Casp3+/+ mice after prolonged c-Myc activation. E, total pancreatic area quantitation indicates no difference in pancreas size between genotypes. F, RT-PCR quantification of IL-1β transcript levels in islets after 2 days of c-Myc activation. -, +/+, c-Myc-; +, +/+, c-Myc+Casp3+/+ and c-Myc+Casp3+/-; +, -/-, c-Myc+Casp3-/-. Results represent means ± S.E.
FIGURE 5.
Proliferation observed in the initial stages of c-Myc activation ceases after prolonged c-Myc induction. A, mice were injected with tamoxifen (TAM) for 6 days. Pancreatic sections immunostained for glucagon and insulin indicate return to normal islet architecture in c-Myc+Casp3-/- mice. B, shown is Ki67 islet staining after 6 days of tamoxifen injection. C, shown is quantification of Ki67 staining (n = 3 per genotype). Results represent means ± S.E. D, shown are representative images of senescence-associated β-galactosidase activity staining on pancreatic sections costained with insulin. -, +/+, c-Myc-; +, +/+, c-Myc+Casp3+/+; +, -/-, c-Myc+Casp3-/-.
c-Myc Activation Leads to Initial Rapid Proliferation and Protection from Apoptosis in c-Myc+Casp3-/- Islets—To assess the early effects of c-Myc activation, we examined islet morphology as well as evaluated the extent of proliferation and apoptosis of β-cells after 2 days of tamoxifen injection. Activation of c-Myc in β-cells has been shown to lead to an early increase in proliferation (15). Indeed, at day 2 after tamoxifen injection, there was a rapid appearance of increased β-cell proliferation, as evidenced by markedly increased Ki67-positive staining (Fig. 3A, upper panels). Costaining with insulin confirmed that the proliferating cells were β-cells (Fig. 3A, lower panels), and the increase in β-cell proliferation was similar between c-Myc+Casp3+/+ and c-Myc+Casp3-/- mice (Fig. 3B). c-Myc in β-cells has been shown to have dual roles in both proliferation and apoptosis, with apoptosis predominating upon sustained c-Myc activation (15). Therefore, we examined for apoptosis by TUNEL staining of pancreases. After 2 days of c-Myc induction, evidence of apoptosis was observed in c-Myc+Casp3+/+ mice, whereas no apoptosis was observed in the β-cells of c-Myc+Casp3-/- mice (Fig. 3, C, middle and lower panels, and D), consistent with the absence of a key proapoptotic mediator in these mice.
FIGURE 3.
c-Myc activation initially leads to β-cell proliferation in c-Myc+ and apoptosis in c-Myc+ Casp3+/+ mice. A, representative Ki67 staining and Ki67/insulin double staining of pancreatic sections from mice injected for 2 days with tamoxifen. C, representative TUNEL staining and TUNEL/insulin double staining indicating the presence of TUNEL-positive nuclei in c-Myc+Casp3+/+ pancreatic sections from mice injected for 2 days with tamoxifen. 4′,6-Diamidino-2-phenylindole (DAPI) staining is shown as a control. B and D, quantification of Ki67 (B) and TUNEL (D) staining (n = 3 per genotype). Increased proliferation was seen in both c-Myc+Casp3+/+ and c-Myc+Casp3-/- mice after 2 days of c-Myc activation (B), whereas apoptosis was observed solely in c-Myc+Casp3+/+ animals (D). **, p < 0.01. -, +/+, c-Myc-; +, +/+, c-Myc+Casp3+/+; +, -/-, c-Myc+Casp3-/-. Results represent means ± S.E.
At this time, there was no difference in islet area or islet number between c-Myc+Casp3+/+ and c-Myc+Casp3-/- mice (Fig. 4, B and C); however, insulin and glucagon immunostaining revealed disrupted islet morphology in both genotypes (Fig. 4A). The characteristic α- and β-cell distribution was preserved in the c-Myc- control, indicating that the disruption of islet architecture in c-Myc+ mice was a consequence of c-Myc activation. Interestingly, at this time, blood glucose levels were high only in c-Myc+Casp3+/+ mice (Fig. 2A), despite the similar islet area and the disruption of islet morphology in both genotypes. The high glucose levels in c-Myc+Casp3+/+ mice suggest that c-Myc-mediated effects lead to the impairment of β-cell function at this stage, whereas the absence of caspase-3 appears to protect islets from this c-Myc-mediated impairment. Glucose tolerance tests further confirmed a defect in glucose homeostasis in c-Myc+Casp3+/+ mice in the early days of c-Myc activation (Fig. 4D).
FIGURE 4.
Islet morphology and function in the early phase of c-Myc activation. A, glucagon and insulin immunostaining of islets after 2 days of c-Myc activation show disrupted islet architecture. B and C, no difference was observed in total islet area (B) and islet number (C) in c-Myc+Casp3 mice after 2 days of c-Myc activation (n = 7). -, +/+, c-Myc-; +, +/+, c-Myc+Casp3+/+; +, -/-, c-Myc+Casp3-/-. D, glucose tolerance tests indicate impaired glucose tolerance in c-Myc+Casp3+/+ mice upon 2-day c-Myc activation. *, p < 0.05 (n = 3-7). Results represent means ± S.E.
Proliferation Does Not Persist in c-Myc+Casp3-/- Mice upon Continual c-Myc Activation—To evaluate the morphology and proliferation status of β-cells at a later phase of c-Myc induction, we injected mice with tamoxifen for 6 days. Interestingly, the disruption in islet morphology seen in the early phase of c-Myc induction was no longer evident in the c-Myc+Casp3-/- mice after 6 days of c-Myc activation (Fig. 5A, right panel), and the morphology continued to remain normal for up to 30 days of c-Myc activation in these mice (Fig. 2B, right panels). In contrast, the remnant islets were already apparent after 6 days of c-Myc activation in c-Myc+-Casp3+/+ mice (Fig. 5A, middle panel). In keeping with the quiescent appearing islets, proliferation was also absent in these mice (Fig. 5, B and C), suggesting that the disrupted architecture observed in the early stages of c-Myc activation is likely linked to the rapid onset of proliferation. Given that the c-Myc+Casp3-/- islets appeared to have stopped proliferating at this time, we wanted to assess whether these cells were undergoing senescence or whether these cells were simply quiescent and not proliferating but remained functional, as was suggested by the blood glucose data. We assessed the presence of senescent cells by X-gal staining of pancreases. X-Gal staining has been used histochemically to detect β-galactosidase activity, a known characteristic of senescent cells not found in presenescent, immortal, or quiescent cells (27, 28). Our results show that 2 days of c-Myc activation led to senescence in the c-Myc+Casp3+/+ islets, which is in keeping with previous reports showing c-Myc-induced senescence and apoptosis in mouse embryonic fibroblasts (27). On the other hand, the c-Myc+Casp3-/- islets stained for X-gal showed little, if any, senescence-associated staining (Fig. 5D). These data support that caspase-3 is essential in mediating c-Myc-induced apoptosis and senescence, and in its absence, the islets remain protected not only from apoptosis but also from c-Myc-induced senescence. Importantly, the state of quiescence achieved in the tamoxifen-injected day 6 c-Myc+Casp3-/- islets is in the presence of sustained c-Myc activation as shown in Fig. 1B.
Together, these results indicate that with the absence of caspase-3 and concomitant c-Myc activation, β-cells undergo early proliferation. However, the absence of caspase-3 protects β-cells not only from c-Myc-induced apoptosis but also from persistent proliferation and senescence, leading to the recovery of islet morphology and maintained function even after prolonged c-Myc activation.
Cell Cycle Inhibition in c-Myc+Casp3-/- Mice upon Sustained c-Myc Activation—In an attempt to elucidate the mechanism for maintaining a balance between the suppression of β-cell apoptosis and the absence of excessive proliferation in the context of c-Myc activation in c-Myc+Casp3-/- mice, we examined the protein expression of selected cell cycle regulatory proteins. Cell cycle progression is governed by cyclins and cyclin-dependent kinases. In particular, the cyclin D·Cdk-4 complex has been shown to play a major role in the maintenance of β-cell homeostasis. In addition, p27 is a major cell cycle inhibitor for β-cell proliferation (29, 30). c-Myc promotes cell cycle progression by regulation of a number of genes, including induction of cyclins and suppression of cell cycle inhibitors (31-33). In addition to being a c-Myc target, p27 is also a caspase-3 cleavage substrate (34, 35). Consistent with this, upon 1 day of c-Myc activation, we observed an increase in cyclin D and Cdk-4 expression and a decrease in p27 expression in c-Myc+Casp3+/+ mice (Fig. 6, A and B). On the other hand, 1 day of c-Myc induction in c-Myc+Casp3-/- mice led to persistence of p27 expression as well as an increase in cyclin D expression levels compared with control mice (Fig. 6B). We performed RT-PCR on isolated islets to assess whether there were any transcriptional changes to p27 as a consequence of caspase-3 deletion. Interestingly, p27 transcription was not altered in the c-Myc+Casp3-/- mice upon 1 day of c-Myc induction (Fig. 6E). These data further support that the modulation in p27 observed in our c-Myc+Casp3-/- islets occurs at the post-translational state.
FIGURE 6.
Effect of caspase-3 deletion and c-Myc activation on selected cell cycle regulatory proteins. A-D, Western blots and quantification of protein expression upon 1 day (A and B) and 6 days (C and D) of c-Myc activation (n = 3 per genotype). GLUT2 is shown to represent comparable proportion of β-cells. Results represent means ± S.E. and are shown as -fold change from noninjected (TAM-) mice for both genotypes. E, RT-PCR quantification of p27 transcript levels in isolated islets after 1 day of c-Myc induction. Results represent means ± S.E. and are shown as -fold change from noninjected (TAM-) c-Myc+Casp3+/+ mice. F, propidium iodide staining of islets showing increased cell cycle progression in the islet cells of c-Myc+Casp3-/- mice treated with tamoxifen for 1 day. +, +/+, c-Myc+Casp3+/+; +, -/-, c-Myc+Casp3-/-; Tam-, no tamoxifen injection; Tam+, tamoxifen injection. *, p < 0.05.
To evaluate the expression of cell cycle markers at a time when c-Myc+Casp3-/- β-cells switched from increased proliferation to quiescence, we tested islets from mice that had been injected with tamoxifen for 6 consecutive days. In keeping with our day 1 observations, we observed that p27 expression was maintained at an increased level upon sustained c-Myc activation (Fig. 6, C and D). Although the level of cyclin D1 was still increased at this time, this does not necessarily reflect the activation of these cell cycle components. In fact, the corresponding Cdk protein, which is the catalytic subunit of the cyclin·Cdk complex, was decreased, suggesting that the continued elevated presence of p27 may explain why, in this model, c-Myc+Casp3-/- islets are protected from excessive proliferation, in contrast to the Bcl-xL overexpression model, where carcinogenic progression ensues.
To further examine cell cycle progression, we performed cell cycle analyses by flow cytometry on isolated islets from the mice. We observed an increased percentage of c-Myc+Casp3-/- islet cells treated with tamoxifen for 1 day in the S and G2-M phases of the cell cycle compared with their controls (Fig. 6F). These results further demonstrate that caspase-3 deletion and concomitant c-Myc activation in the β-cells lead to enhanced cell cycle progression, which is consistent with the increased Ki67-positive islet cells in these mice after 1 day of tamoxifen treatment (Fig. 3A). Interestingly, the c-Myc-activated c-Myc+Casp3+/+ islet cells did not show a significant increase in cell cycle progression. This may be due to the net effect of the 1-day c-Myc activation on proliferation as well as apoptosis in the wild-type islets, in contrast to caspase-3-deficient β-cells, which did not undergo apoptosis upon 1 day of tamoxifen treatment.
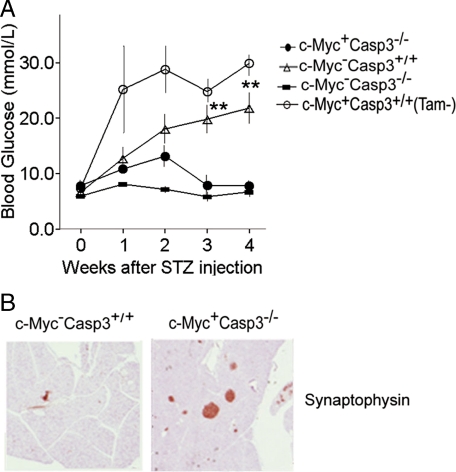
c-Myc+Casp3-/- Mice Are Protected from Streptozotocin-induced Diabetes—The absence of caspase-3 has been shown to confer protection from diabetes in the MLDS model of type 1 diabetes (22). To examine whether caspase-3 absence continues to provide protection against β-cell apoptosis in an experimental model of diabetes in the presence of continued c-Myc activation, we injected the mice with streptozotocin for 5 days. Mice were also injected daily with tamoxifen to activate c-Myc throughout the duration of the experiment. The c-Myc+Casp3-/- mice were protected from diabetes development after MLDS injections (Fig. 7, A and B), indicating that despite the presence of two different apoptotic stimuli, caspase-3 absence provides a sufficient block of the apoptotic machinery to allow for protection from diabetes. Collectively, our results show that an absence of caspase-3 protects β-cells from diabetes induced by c-Myc activation as well as from c-Myc apoptosis in the context of an experimental diabetes model.
FIGURE 7.
Caspase-3 deletion protects β-cells from c-Myc-induced apoptosis in the MLDS model of diabetes. A, blood glucose levels in MLDS-treated c-Myc+Casp3 mice in the context of c-Myc activation. Mice were injected intraperitoneally with STZ for 5 consecutive days. Mice were also injected with tamoxifen 6 days prior to STZ injection and daily throughout the duration of the experiment to maintain c-Myc activation. **, p < 0.01 (n = 3 except n = 5 for c-Myc+Casp3-/-). B, representative synaptophysin-stained pancreatic sections taken 4 weeks post-STZ injection showing β-cell destruction in control animals and β-cell protection in caspase-3-deficient animals. Results represent means ± S.E.
DISCUSSION
Several in vitro and in vivo studies have pointed to the possible role of c-Myc in regulation of β-cell mass. Normal adult islets have low c-Myc expression (3); however, c-Myc expression is increased in islets of diabetic rats and in isolated islets cultured under high glucose conditions (3, 10). In vivo, in β-cells, c-Myc is an inducer of both proliferation and apoptosis; however, its predominant effect is pro-apoptotic (36). Overexpression of the c-myc gene in β-cells in mice leads to diabetes and mortality 3 days after birth due to profound β-cell apoptosis, which overwhelms the initial proliferative effects of c-Myc (12). In an inducible tamoxifen model of c-Myc activation in β-cells, c-Myc induction leads to proliferation accompanied by rapid apoptosis and complete β-cell ablation after 6 days of c-Myc induction (15). Overexpression of the anti-apoptotic protein Bcl-xL in this model leads to a full spectrum of tumor progression by blocking c-Myc-induced apoptosis. Loss of the tumor suppressor p19ARF in the inducible c-Myc model also leads to carcinogenesis, however, via a different mechanism. Although inactivation of p19ARF enhances c-Myc apoptosis, it also exerts a profound proliferative effect that leads to tumor formation (37).
To further dissect the biology of c-Myc-induced apoptosis in β-cells, we generated caspase-3 knock-out mice containing the inducible c-myc transgene (c-Myc+Casp3-/-). In contrast to the studies of Pelengaris et al. (15), in which the anti-apoptotic protein Bcl-xL was overexpressed, we eliminated the key pro-apoptotic mediator caspase-3 with the aim of identifying which physiological molecules important in apoptosis are relevant and essential in the c-Myc model. We have shown that the absence of caspase-3 confers protection from c-Myc-induced apoptosis, and in contrast to the phenotypes observed with the Bcl-xL gain of function or the p19ARF loss of function, no tumor formation was observed. c-Myc+Casp3-/- mice were protected from diabetes, and β-cell function remained normal under conditions of sustained c-Myc activation.
c-Myc+Casp3-/- mice were also protected from STZ-induced diabetes in the context of c-Myc activation. Although one in vivo study has indicated a role for caspase-3 in the initiation of type 1 diabetes (22), the specific role of caspase-3 in β-cell apoptosis and the progression to diabetes has not been well characterized. The results of our STZ experiments in c-Myc+ mice lend support to the notion that caspase-3 is a critical mediator of apoptosis in β-cells, and in its absence, even the synergistic effects of streptozotocin treatment combined with the effects of the profound apoptotic inducer c-Myc are not sufficient to trigger apoptosis in these cells.
There exist two other known caspase-3-like proteases (caspase-6 and -7) with an analogous in vitro substrate preference for caspase-3 (38). However, individual caspase functions have been shown to be highly tissue- and context-specific (23, 35, 39-44), and functional redundancy does not appear to play a significant role in the context of c-Myc activation, as caspase-6 or -7 does not appear to be able to substitute for caspase-3 to induce apoptosis in caspase-3-deficient animals.
The dramatic anti-apoptotic influence of Bcl-xL overexpression seen in the inducible c-Myc model suggests that c-Myc-induced apoptosis in β-cells proceeds predominantly through the intrinsic mitochondrial pathway (15). In addition, further evidence implicating the intrinsic apoptotic pathway comes from studies indicating a specific requirement for Bax in c-Myc-induced apoptosis. Mice lacking the pro-apoptotic protein Bax rapidly develop invasive, angiogenic tumors upon c-Myc activation (45). Thus, it appears that in the context of c-Myc-induced apoptosis, caspase-3 activation takes place through the intrinsic pathway. However, given the central role of caspase-3 in both the intrinsic and extrinsic apoptotic pathways, our findings are surprising in that the absence of this critical apoptotic mediator is able to uncouple and mask the oncogenic properties of c-Myc while obliterating its pro-apoptotic function. Collectively, these results would suggest that Bcl-2 family members modulate additional oncogenic properties beyond that of apoptosis regulation per se.
In addition to its function in promoting apoptosis, caspase-3 has been shown to play a role in cell cycle regulation. Caspase-3 cleaves the cyclin-dependent kinase inhibitors p21 and p27 (34, 35), which inhibit the cell cycle, and consistent with this role, we have shown increased or persistent expression of p27 in c-Myc+Casp3-/- mice relative to their wild-type controls upon c-Myc induction. Recently, others have shown that in nonproliferating skeletal muscle, fibroblast, and embryonic kidney cells, temporary and permanent growth arrest must be actively maintained by constant expression of Cdk inhibitors such as p21 and p27, and nonproliferating cells can be mitotically reactivated solely by suppression of these inhibitors (46). In the caspase-3 knock-out mice, absence of caspase-3 may preclude p27 cleavage, and as a result, these proteins accumulate, leading to cell cycle inhibition. In addition, transgenic mouse studies have demonstrated that overexpression of p27 in β-cells leads to reduced β-cell proliferation (30). In accordance with this, in our model, elevated p27 expression continues upon sustained c-Myc activation and is accompanied by decreases in Cdk-4, which suggests that p27 may be the pivotal protein through which caspase-3 can maintain the balance between the suppression of β-cell apoptosis and the absence of excessive proliferation and tumor progression in the inducible c-Myc model. We acknowledge, however, that it would be naïve to think that the profound oncogenic properties of c-Myc can be held in check simply by increased expression of a cell cycle inhibitor, and numerous other genes are likely to be involved in this process.
In addition to being a caspase-3 cleavage target, p27 is also repressed by c-Myc (31, 33). c-Myc activates or represses the transcription of a multitude of genes, and efforts to identify new direct and indirect targets are still ongoing. Targets identified thus far include genes involved in the cell cycle, cell growth, metabolism, differentiation, and apoptosis (47, 48). Furthermore, c-Myc action is cell- and context-specific, meaning that genes that may have been identified as putative c-Myc targets in other cells may not be regulated in the same fashion in β-cells. Much information about the mechanism behind repression of c-Myc-induced apoptosis in c-Myc+Casp3-/- animals could be gleaned from microarray studies comparing levels of gene expression in c-Myc-activated and -nonactivated c-Myc+Casp3-/- mice. This approach has already been successfully applied by others to compare the in vivo c-Myc targets that are activated or repressed upon c-Myc induction and tumor formation in c-Myc+ mice overexpressing Bcl-xL (49). 233 candidate tumor maintenance genes were identified in this manner, 10 of which share the correct dynamics to be putative direct c-Myc transcriptional targets. It would be interesting to apply a similar approach to the c-Myc+Casp3-/- model, as it may lead to a more comprehensive explanation for the protection from diabetes and tumorigenesis conferred by the absence of caspase-3.
Our in vivo work shows that caspase-3 appears to control the c-Myc-mediated cell death and cell cycle in an integrative manner to reach a state of homeostasis. This role of caspase-3 that is uncovered in the context of c-Myc activation is highly novel and is not similar to any of the models using any of the knockouts of genes that are important in apoptosis. For example, overexpression of Bcl-xL concomitantly with activation of c-Myc leads to massive tumor formation, whereas deletion of caspase-8 and ablation of the mitochondrial arm of the apoptotic signaling pathway through inhibition of cytochrome c release both lead to apoptosis and the complete destruction of islets.4 Therefore, the unique effect of caspase-3 deletion is not just a simple reflection of abolishment of apoptosis per se. In contrast, we believe that this study shows the essential role of caspase-3 in controlling other aspects of cellular processes in addition to apoptosis. We have indeed shown this notion of the role of caspase-3 in differentiation and the cell cycle control in other cell types (35, 41, 50). Given the increasing evidence for a potential role of c-Myc in type 2 diabetes (12, 15), targeting c-Myc-induced apoptosis would represent an attractive therapeutic option in diabetes (34). Our finding that the absence of caspase-3 masks the oncogenic properties of c-Myc while protecting β-cells from apoptosis is encouraging in that regard. Although deregulated c-Myc is a potent oncogene and thus any strategies involving manipulation of this gene must be carefully approached, results from these and other experiments may lead to further identification of mechanisms of c-Myc biology relevant to β-cells, which in turn may lead to novel therapeutic strategies for the maintenance of β-cell mass and function.
Acknowledgments
We thank Saswati Sen, Sung Ah Jun, Xiaohong Wu, and Xin Liang Mao for technical assistance.
This work was supported in part by grants from the Canadian Institutes for Health Research and the Canadian Diabetes Association (to M. W.).
Footnotes
The abbreviations used are: MLDS, multiple low dose streptozotocin; RT, reverse transcription; PBS, phosphate-buffered saline; X-gal, 5-bromo-4-chloro-3-indolyl-β-d-galactopyranoside; STZ, streptozotocin; TUNEL, terminal deoxynucleotidyltransferase-mediated dUTP nick end labeling; IL-1β, interleukin-1β.
A. Radziszewska, S. A. Schroer, and M. Woo, unpublished data.
References
- 1.Butler, A. E., Janson, J., Bonner-Weir, S., Ritzel, R., Rizza, R. A., and Butler P. C. (2003) Diabetes 52 102-110 [DOI] [PubMed] [Google Scholar]
- 2.Rhodes, C. J. (2005) Science 307 380-384 [DOI] [PubMed] [Google Scholar]
- 3.Jonas, J. C., Sharma, A., Hasenkamp, W., Ilkova, H., Patane, G., Laybutt, R., Bonner-Weir, S., and Weir, G. C. (1999) J. Biol. Chem. 274 14112-14121 [DOI] [PubMed] [Google Scholar]
- 4.Laybutt, D. R., Glandt, M., Xu, G., Bai Ahn, Y., Trivedi, N., Bonner-Weir, S., and Weir, G. C. (2003) J. Biol. Chem. 278 2997-3005 [DOI] [PubMed] [Google Scholar]
- 5.Pelengaris, S., and Khan, M. (2003) Arch. Biochem. Biophys. 416 129-136 [DOI] [PubMed] [Google Scholar]
- 6.Juin, P., Hueber, A.-O., Littlewood, T. D., and Evan, G. I. (1999) Genes Dev. 13 1367-1381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Klefstrom, J., Verschuren, E. W., and Evan, G. I. (2002) J. Biol. Chem. 277 43224-43232 [DOI] [PubMed] [Google Scholar]
- 8.Mougneau, E., Lemieux, L., Rassoulzadegan, M., and Cuzin, F. (1984) Proc. Natl. Acad. Sci. U. S. A. 81 5758-5762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pelengaris, S., Littlewood, T. D., Khan, M., Elia, G., and Evan, G. I. (1999) Mol. Cell 3 565-577 [DOI] [PubMed] [Google Scholar]
- 10.Jonas, J. C., Laybutt, D. R., Steil, G. M., Trivedi, N., Pertusa, J. A., Van de Casteele, M., Weir, G. C., and Henquin, J. C. (2001) J. Biol. Chem. 276 35375-35381 [DOI] [PubMed] [Google Scholar]
- 11.Van de Casteele, M., Kefas, B. A., Cai, Y., Heimberg, H., Scott, D. K., Henquin, J. C., Pipeleers, D., and Jonas, J. C. (2003) Biochem. Biophys. Res. Commun. 312 937-944 [DOI] [PubMed] [Google Scholar]
- 12.Laybutt, D. R., Weir, G. C., Kaneto, H., Lebet, J., Palmiter, R. D., Sharma, A., and Bonner-Weir, S. (2002) Diabetes 51 1793-1804 [DOI] [PubMed] [Google Scholar]
- 13.Gittes, G. K., and Rutter, W. J. (1992) Proc. Natl. Acad. Sci. U. S. A. 89 1128-1132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Littlewood, T. D., Hancock, D. C., Danielian, P. S., Parker, M. G., and Evan, G. I. (1995) Nucleic Acids Res. 23 1686-1690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pelengaris, S., Khan, M., and Evan, G. I. (2002) Cell 109 321-334 [DOI] [PubMed] [Google Scholar]
- 16.Thornberry, N. A., and Lazebnik, Y. (1998) Science 281 1312-1316 [DOI] [PubMed] [Google Scholar]
- 17.Woo, M., Hakem, R., and Mak, T. W. (2000) Cell Res. 10 267-278 [DOI] [PubMed] [Google Scholar]
- 18.Porter, A. G., and Jänicke, R. U. (1999) Cell Death Differ. 6 99-104 [DOI] [PubMed] [Google Scholar]
- 19.Maedler, K., Spinas, G. A., Lehmann, R., Sergeev, P., Weber, M., Fontana, A., Kasier, N., and Donath, M. Y. (2001) Diabetes 50 1683-1690 [DOI] [PubMed] [Google Scholar]
- 20.Yamada, K., Ichikawa, F., Ishiyama-Shigemoto, S., Yuan, X., and Nonaka, K. (1999) Diabetes 48 478-483 [DOI] [PubMed] [Google Scholar]
- 21.Kim, W. H., Lee, J. W., Suh, Y. H., Hong, S. H., Choi, J. S., Lim, J. H., Song, J. H., Gao, B., and Jung, M. H. (2005) Diabetes 54 2602-2611 [DOI] [PubMed] [Google Scholar]
- 22.Liadis, N., Murakami, K., Eweida, M., Elford, A. R., Sheu, L., Gaisano, H. Y., Hakem, R., Ohashi, P. S., and Woo, M. (2005) Mol. Cell. Biol. 25 3620-3629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Woo, M., Hakem, R., Soengas, M. S., Duncan, G. S., Shahinian, A., Kagi, D., Hakem, A., McCurrach, M., Khoo, W., Kaufman, S. A., Senaldi, G., Howard, T., Lowe, S. W., and Mak, T. W. (1998) Genes Dev. 12 806-819 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wijesekara, N., Konrad, D., Eweida, M., Jefferies, C., Liadis, N., Giacca, A., Crackover, M., Suzuki, A., Mak, T. W., Kahn, C. R., Klip, A., and Woo, M. (2005) Mol. Cell. Biol. 25 1135-1145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dimri, G. P., Lee, X., Basile, G., Acosta, M., Scott, G., Roskelley, C., Medrano, E. E., Linskens, M., Rubelj, I., and Pereira-Smith, O. (1995) Proc. Natl. Acad. Sci. U. S. A. 92 9363-9367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shchors, K., Shchors, E., Rostker, F., Lawlor, E. R., Brown-Swigart, L., and Evan, G. I. (2006) Genes Dev. 20 2527-2538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li, J., Sejas, D. P., Burma, S., Chen, D. J., and Pang, Q. (2007) Carcinogenesis 28 1163-1170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Serrano, M., Lin, A. W., McCurrach, M. E., Beach, D., and Lowe, S. W. (1997) Cell 88 593-602 [DOI] [PubMed] [Google Scholar]
- 29.Rane, S. G., Dubus, P., Mettus, R. V., Galbreath, E. J., Boden, G., Reddy, E. P., and Barbacid, M. (1999) Nat. Genet. 22 44-52 [DOI] [PubMed] [Google Scholar]
- 30.Uchida, T., Nakamura, T., Hashimodo, N., Matsuda, T., Kotani, K., Sakaue, H., Kido, Y., Hayashi, Y., Nakayama, K. I., White, M. F., and Kasuga, M. (2005) Nat. Med. 11 175-182 [DOI] [PubMed] [Google Scholar]
- 31.Daksis, J. I., Lu, R. Y., Facchini, L. M., Marhin, W., and Penn, L. Z. (1994) Oncogene 9 3635-3645 [PubMed] [Google Scholar]
- 32.O'Connell, B. C., Cheung, A. F., Simkevich, C. P., Tam, W., Ren, X., Mateyak, M. K., and Sedivy, J. (2003) J. Biol. Chem. 278 12563-12573 [DOI] [PubMed] [Google Scholar]
- 33.Yang, W., Shen, J., Wu, M., Arsura, M., FitzGerald, M., Suldan, Z., Kim, D. W., Hofmann, C. S., Pianetti, S., Romieu-Mourez, R., Freedman, L. P., and Sonenshein, G. E. (2001) Oncogene 20 1688-1702 [DOI] [PubMed] [Google Scholar]
- 34.Levkau, B., Koyama, H., Raines, E. W., Clurman, B. E., Herren, B., Orth, K., Roberts, J. M., and Ross, R. (1998) Mol. Cell 1 553-563 [DOI] [PubMed] [Google Scholar]
- 35.Woo, M., Hakem, R., Furlonger, C., Hakem, A., Duncan, G. S., Sasaki, T., Bouchard, D., Lu, L., Wu, G. E., Paige, C. J., and Mak, T. W. (2003) Nat. Immunol. 4 1016-1022 [DOI] [PubMed] [Google Scholar]
- 36.Pelengaris, S., Rudolph, B., and Littlewood, T. D. (2000) Curr. Opin. Genet. Dev. 10 100-105 [DOI] [PubMed] [Google Scholar]
- 37.Finch, A., Prescott, J., Shchors, K., Hunt, A., Soucek, L., Dansen, T. B., Swigart, L. B., and Evan, G. I. (2006) Cancer Cell 10 113-120 [DOI] [PubMed] [Google Scholar]
- 38.Telanian, R. V., Quinian, C., Trautz, S., Hackett, M. C., Mankovich, J. A., Banach, D., Ghayur, T., Brady, K. D., and Wong, W. W. (1997) J. Biol. Chem. 272 9677-9682 [DOI] [PubMed] [Google Scholar]
- 39.Hakem, R., Hakem, A., Duncan, G. S., Henderson, J. T., Woo, M., Soengas, M. S., Elia, A. J., de la Pompa, J. L., Kagi, D., Khoo, W., Potter, J., Yoshida, R., Kaufman, S. A., Lowe, S. W., Penninger, J. M., and Mak, T. W. (1998) Cell 94 339-352 [DOI] [PubMed] [Google Scholar]
- 40.Kuida, K., Zheng, T. S., Na, S., Kuan, C., Yang, D., Karasuyama, H., Rakic, P., and Flavell, R. A. (1996) Nature 384 368-372 [DOI] [PubMed] [Google Scholar]
- 41.Okuyama, R., Nguyen, B. C., Talora, C., Ogawa, E., Tommasi di Vignano, A., Lioumi, M., Chiorino, G., Tagami, H., Woo, M., and Dotto, G. P. (2004) Dev. Cell 6 551-562 [DOI] [PubMed] [Google Scholar]
- 42.Sakamaki, K., Inoue, T., Asano, M., Sudo, K., Kazama, H., Sakagami, S., Sakata, M., Ozaki, S., Nakamura, S., Toyokuni, S., Osumi, N., Iwakura, Y., and Yonehara, S. (2002) Cell Death Differ. 9 1196-1206 [DOI] [PubMed] [Google Scholar]
- 43.Salmena, L., Lemmers, B., Hakem, A., Matysiak-Zablocki, E., Murakami, K., Au, P. Y., Berry, D. M., Tamblyn, L., Shehabeldin, A., Migon, E., Wakeham, A., Bouchard, D., Yeh, W. C., McGlade, J. C., Ohashi, P. S., and Hakem, R. (2003) Genes Dev. 17 883-895 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Woo, M., Hakem, A., Elia, A. J., Hakem, R., Duncan, G. S., Patterson, B. J., and Mak, T. W. (1999) J. Immunol. 163 4909-4916 [PubMed] [Google Scholar]
- 45.Dansen, T. B., Whitfield, J., Rostker, F., Swigart, L. B., and Evan, G. I. (2006) J. Biol. Chem. 281 10890-10895 [DOI] [PubMed] [Google Scholar]
- 46.Pajalunga, D., Mazzola, A., Salzano, A. M., Biferi, M. G., De Luca, G., and Crescenzi, M. (2007) J. Cell Biol. 176 807-818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Dang, C. V. (1999) Mol. Cell. Biol. 19 1-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Dang, C. V., O'Donnell, K. A., Zeller, K. I., Nguyen, T., Osthus, R. C., and Li, F. (2006) Semin. Cancer Biol. 16 253-264 [DOI] [PubMed] [Google Scholar]
- 49.Lawlor, E. R., Soucek, L., Swigart L. B., Shchors, K., Bialucha, C. U., and Evan, G. I. (2006) Cancer Res. 66 4591-4601 [DOI] [PubMed] [Google Scholar]
- 50.Janzen, V., Fleming, H. E., Riedt, T., Karlsson, G., Riese, M. J., Celso, C. L., Reynolds, G., Milne, C. D., Paige, C. J., Karlsson, S., Woo, M., and Scadden, D. T. (2008) Cell Stem Cell 2 584-594 [DOI] [PMC free article] [PubMed] [Google Scholar]