The presentations on Pneumocystis discussed here represent 10 international meetings that span over 20 years of research. These workshops were initiated by a nascent community of researchers investigating a poorly understood organism, Pneumocystis, which at that time was gaining prominence because of its importance to immunosuppressed patients, especially those infected with human immunodeficiency virus (HIV). Pneumocystis defied phylogenetic classification, which at the time was based primarily on morphological characteristics. Because of this uncertainty, Pneumocystis lay outside the mainstream research of both mycology and parasitology and did not have a forum for this new group of investigators. The first “Workshop on Pneumocystis carinii” filled that gap in 1988, and subsequent workshops were platforms for the cutting-edge science that was performed in this microbial arena. The first reports of its identity as a member of the fungal kingdom were presented in the workshop series, and discussions of importance to the community were held under its auspices, such as the initiation of the genome project and nomenclatural changes. The 10th workshop, conducted in 2008, continued this tradition, and the scientific achievements of the community are summarized here.
THE ORGANISM AND ITS HISTORY
As a context for discussion of the workshop presentations, it is imperative that critical basic biological and historical principles are summarized. Pneumocystis organisms occupy a fungal genus whose members are harbored by a wide variety of mammalian hosts. They are thought to exist in a commensal-like state and cause an asymptomatic or subclinical infection when first confronted by neonates or children. A more lethal type of infection, Pneumocystis pneumonia (PCP), occurs in immunosuppressed hosts, especially in individuals infected with HIV or undergoing chemotherapy. The pneumonia in humans had been one of the most common infections associated with immunosuppressed, HIV-infected humans, but its incidence in developed countries has declined due to effective prophylaxis and therapy. It remains a serious clinical problem in developing countries, where it continues its role as an AIDS-defining illness (100).
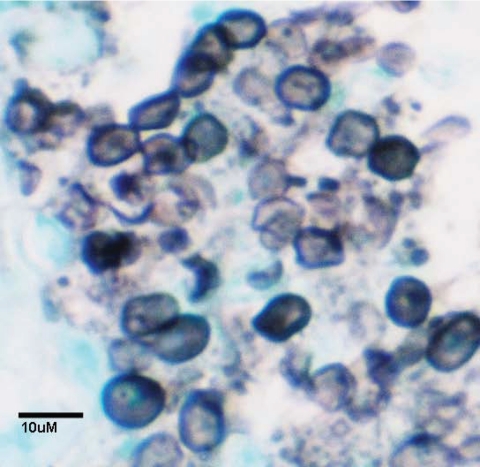
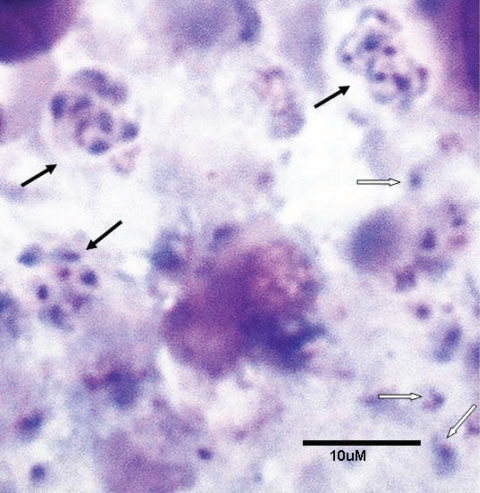
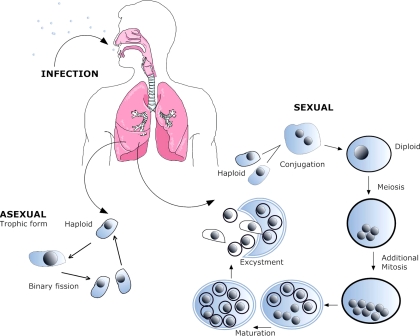
Pneumocystis spp. are extracellular, obligate, host-specific, yeast-like parasitic fungi virtually restricted to lung tissues. The morphological forms range in size from 1 to 10 μm. The cyst, the most characteristic form, is defined as an eight-spore ascus and serves as the primary diagnostic form identified by staining with the fungal stain methenamine silver (Fig. 1). There is a spectrum of developmental forms that range from the apparent vegetative unicellular trophic form through intermediate stages that are predecessors to the mature ascus (Fig. 2). A presumptive life cycle is shown in Fig. 3.
FIG. 1.
P. carinii asci/cyst forms stained with methenamine silver. Black objects are P. murina asci/cysts. Host tissue is counterstained green.
FIG. 2.
Pneumocystis carinii trophic and cyst forms. Touch preparation of an infected rat lung stained with a rapid variant of the Wright-Giemsa stain. Black arrows, asci/cysts; white arrows, trophic forms.
FIG. 3.
Putative life cycle of Pneumocystis. (i) Infection. The agent of infection is suspected to be airborne spores, but these have not been identified. (ii) Asexual phase. Haploid trophic forms replicate asexually by binary fission. (iii) Sexual phase. Two presumptive mating types conjugate, undergo karyogamy, and produce a diploid zygote, which progresses through meiosis and then an additional mitosis to produce eight nuclei. The nuclei are packaged into spores by invagination of the ascus cell membranes. After completion, excystment occurs via protunicate release by unknown mechanisms. The released spores become the vegetative forms that can then undergo asexual or sexual replication.
The early history of Pneumocystis investigation arose from the study of trypanosomal infections in a variety of mammals, including black-tufted-ear marmosets (Callithrix penicillata) and guinea pigs. In 1909, Carlos Chagas described an eight-spore cyst in the lung tissue which he called “schizogonicas” (14) and considered to be part of the Trypanosoma cruzi life cycle. He focused on this unique eight-celled form and then coined a new genus for this commingled infection, Schizotrypanum cruzi (13), complete with a detailed description of a life cycle involving both T. cruzi and the “schizogonicas” in the lung, which were undoubtedly Pneumocystis cysts. In 1910, Carini was studying Norway rats infected with another trypanosome, Trypanosoma lewisi, and he too noted the presence of the “eschizogonicas,” thus separating the species link to only T. cruzi (8). In 1912, Delanoë and Delanoë (29) noted the presence of “schizogonie” (cysts) in the absence of any trypanosomal infection and suggested that these cysts represented a new parasitic species in rats which were related to “des Coccidies,” and they coined the new genus and species, Pneumocystis carinii.
NOMENCLATURE AND SYSTEMATICS
Under the St. Louis Code (International Code of Botanical Nomenclature [ICBN]) (41), the names Pneumocystis and P. carinii would have been invalid, but they are valid under the more forgiving International Code of Zoological Nomenclature. Because of the changes adopted in the ICBN in the Vienna Code, these names are now acceptable and valid under revised article 45.4 (91). The species P. carinii was later typified (neotype) in the same publication in which Pneumocystis jirovecii was typified using a micrograph as a lectotype (113). To date, the following three other Pneumocystis species have been formally described and typified according to the ICBN: Pneumocystis wakefieldiae in the rat host (19, 20), Pneumocystis murina in the mouse host (56), and Pneumocystis oryctolagi in the rabbit host (28). A synopsis of the higher-order systematics for Pneumocystis is shown below, based on the work of D. S. Hibbett et al. (46).
MOLECULAR BIOLOGY OF THE ORGANISM
Investigators attending the first Workshop on Pneumocystis carinii in 1988 reported on methods for purification of the organism from host lung tissue for a variety of purposes, including studies of the nucleic acids (129a). Prior to this meeting, there were few articles reporting on the characteristics of the organism's RNA or DNA (e.g., melting curves), and those studies were often compromised by contaminating host nucleic acids. During the first meeting, various characterizations and isolations of the nucleic acids and cloning of mitochondrial and nuclear ribosomal DNA genes were described. The work was slow and painstaking, requiring sequencing gels. Primers pAZ101 and pAZ102, directed to the large-subunit mitochondrial ribosomal DNA, were described by Wakefield et al. (135). These primers were adopted almost universally by investigators interested in diagnosis and detection of the organisms in a variety of host species and were also used for phylogenetic inferences, and they remain in use. Sequencing of the nuclear small rRNA subunit from Pneumocystis revealed the fungal identity of the genus Pneumocystis (32, 126), which was validated over the ensuing years by comparative genetic analyses and by the Pneumocystis genome project (21). Although no member of the genus can be cultured continuously outside the mammalian host even today, there has been an explosion of information about the genes and genomes of the members of this genus. In 1997, consensus for a genome project (using P. carinii) was obtained from the Pneumocystis community at the 5th International Workshop on Opportunistic Protists (2, 18). Prior to that, only about 40 gene accessions for any species of Pneumocystis were listed. A transcriptional profile (22) and gene inventory (http://pgp.cchmc.org) provided the community at large with sufficient genetic information to begin functional characterizations, predict metabolic pathways, and conduct phylogenetic analyses.
Heterologous complementation and interrogation of the life cycle and metabolism.
Because there is no species of Pneumocystis than can be cultured continuously, the molecular tools available to the research community are limited. The use of heterologous systems to demonstrate the potential function of a Pneumocystis gene translated to protein became a necessity. The organism of choice has been Saccharomyces cerevisiae due to extensive deletions and selection choices available. Genes from the species P. carinii (as opposed to those of other Pneumocystis species) have been the most frequently expressed genes in heterologous hosts. Schizosaccharomyces pombe, though phylogenetically closer to Pneumocystis than S. cerevisiae, has been used at a lower frequency due to the paucity of deletions. However, S. pombe was used to characterize the P. carinii ortholog of S. pombe brl1, PcBrl1, which encodes an integral nuclear envelope protein essential for RNA export from the nucleus (44, 79). The P. carinii ortholog was also able to complement the S. cerevisiae brlq, brr6, and brr6/brrl1 null mutants by use of a centromere expression vector, p416 (79, 80). One important consideration for expression of Pneumocystis genes appears to be the choice of the promoter. The same investigators compared the efficiencies of cloning orthologs from Candida glabrata, Pneumocystis carinii, and S. cerevisiae into yeast deletion mutants (80). Whereas the native promoter was necessary for cloning S. cerevisiae genes into the deletion strains, both C. glabrata and P. carinii genes required strong constitutive promoters from S. cerevisiae. The investigators also noted that at least 500 bp of sequence including the promoter was also necessary for efficient complementation. These observations are important for future attempts for functional characterization of Pneumocystis genes, as no means are yet available for a reverse genetic approach in these organisms.
Investigators dissecting the life cycle of P. carinii characterized the meiotic inhibitor PcRan1 kinase (7). Ran1p kinases must be inactivated for meiosis to occur. Using an S. cerevisiae overexpression system, PcRan1p was purified by immunoprecipitation or produced by an in vitro rapid translation system (Roche) and evaluated in kinase assays. These assays revealed that PcRan1p could phosphorylate at a wide range of pHs and that its activity was greatly reduced at temperatures above 25°C. Circular dichroism and fluorescence spectroscopy corroborated these findings and showed PcRan1p to be a strongly temperature-sensitive protein with induced conformational changes at subphysiological temperatures beginning at 25°C. These data show that the enzyme is a pH refractory protein and suggest that this temperature regulation may coincide with inactivation of the enzyme within the mammalian lung, permitting meiosis to occur. This finding is intriguing and should be followed by studies identifying other regulatory factors that contribute to the switching of vegetative growth to meiosis within the mammalian lung.
In previous studies investigating the life cycle of P. carinii, PcSte20 was shown to be upregulated during binding of the trophic forms to alveolar epithelial cells (59). In fungi, Ste20 proteins are commonly activated by small G proteins belonging to the Cdc42-like family. The PcCdc42 gene was cloned from a cDNA library, using a portion of the sequence obtained from the Pneumocystis Genome Project database (60). The cDNA was 576 bp long, and the predicted molecular mass was about 38 kDa. The gene was shown to be a single copy by Southern blotting. Cyst and trophic forms expressed similar levels of the mRNA, as detected by Northern blot analysis. Heterologous expression of PcCdc42 in a Δcdc42 mutant S. cerevisiae strain could restore growth. The protein demonstrated GTP binding activity and GTPase activity with kinetics similar to those for previously described Cdc42 proteins. Identification of the functional PcCdc42p protein adds to our understanding of the life cycle and signaling processes of P. carinii.
The Pneumocystis cyst wall contains β-1,3 glucan, which contributes in large part to the rigidity of this developmental stage. It is assumed that after replication within the ascus, the eight daughter forms or spores are released to continue the vegetative or meiotic process (Fig. 3). The mechanism for such a release is considered a protunicate one in Pneumocystis, where there is no active shooting of spores but rather a dissolution of the ascus (78). The action of an endo-β-1,3 glucanase could digest the rigid layer of the cyst, permitting excystation. To investigate the potential role of glucanase in the process, investigators first showed that glucanase activity was present in lung homogenates from a P. carinii-infected lung (134). The sequence of a homolog of the endo-β-1,3 glucanase genes in S. cerevisiae and S. pombe was identified in the Pneumocystis Genome Project database (http://pgp.cchmc.org). A 2.2-kb open reading frame containing four introns was cloned using rapid amplification of cDNA ends and PCR. Expression in an S. cerevisiae Eng1 deletion mutant showed partial recovery from the cell separation defect, suggesting that it may exert a similar function in the native organism.
Transcriptional analysis and genomics.
The transcriptome of P. carinii during fulminant infection was recently examined using microarray analysis (22). Custom slides were comprised of 70-mer signature sequences of the cDNA unigene set and of predicted fungal gene homologs identified in the assembled genomic sequences (http://pgp.cchmc.org). These custom arrays of 3,067 putative open reading frames were used in two applications at the present meeting. In the first, P. carinii organisms in a carriage state were compared to those in a fulminant infection as a means to better understand the shift from colonization to the disease state (12). Genes that were differentially regulated included those that were involved in the cell cycle and the gene for the glucan synthase enzyme involved in cell wall biosynthesis. Since trophic forms are considered to be the primary developmental forms in the vegetative phase, mitotic cell control signals are in keeping with the increased growth expected in the disease state. The regulation of a key enzyme in cell wall synthesis suggests that the trophic forms may be the primary stage in a cryptic colonization state which changes to include cyst formation as the infection increases in number in the immunosuppressed host.
Mitochondrial genome.
Sequences for the mitochondrial genome of P. carinii became available as a result of the Pneumocystis Genome Project. Sequences gleaned from the project database were assembled into a contiguous sequence of 22 kb by use of gap4 (http://www.molgen.mpg.de/∼service/scisoft/staden/gap4_unix_2.html). Mapping of the contig by Gene Ontology (GO) molecular function annotation revealed the presence of four open reading frames and 19 tRNAs in addition to 17 other genes that were typical of fungal mitochondrial genomes (119). A unique feature was the presence of a 24-bp unit that was repeated one to five times, depending on the organism isolate. Migration on a contour-clamped homogeneous electric field gel, digestion with BAL 31, and failure to close the ends to form a circle were highly suggestive of a linear genome. Identification of telomere-like repeats at each end of the sequence, similar to those found in other linear fungal mitochondrial genomes, further supported the linear nature of the P. carinii mitochondrial genome. The significance of a linear genome in Pneumocystis or in any other member of the fungal kingdom is not yet understood, but it has been suggested that it may convey a survival advantage (106).
MSG family.
A family of genes encoding surface glycoproteins is multiply repeated at the telomeric ends of all Pneumocystis species examined to date. The number of genes appears to vary for each individual Pneumocystis species, with approximately 80/genome for P. carinii and approximately half that for P. murina (125). The number of major surface glycoprotein (MSG) genes in the P. jirovecii genome is not known. It is believed that the organism uses these surface antigens to evade immune surveillance and also for adhesion to host cells and to organisms in other life cycle stages that grow as tightly adherent clusters within the alveolar lumen.
To better understand the heterogeneity within the MSG family of genes, 696 MSG sequences were analyzed from two populations of P. carinii (57). Pairwise comparison showed that the average number of nucleotide differences between reads was 57 ± 15 (19%). Since some of these could be due to sequencing errors, the 696 reads were assembled into contigs under conditions that permitted a 5% variance to assess the depth of such errors. Analysis of these sequences showed an error rate of 0.0013, or one error per 769 bp. These results suggested that sequence reads that are at least 99% identical should be grouped together because such sequences likely originated from templates that were identical. Genetic strains are expected to exhibit allelic variation. Since the probability of error was 0.0013 per site, the probability that an error would occur twice at a given site was 1.7 × 10−6. Therefore, observation of a variant nucleotide at a given site in at least two reads in a contig would likely be due to single nucleotide polymorphisms rather than error. Based on these criteria, the distributions of single nucleotide polymorphisms in the two populations suggest that allelic variation occurs in MSG genes. These data should be considered in studies attempting to genotype human isolates that target MSG genes.
IN VITRO STUDIES
Biofilms are three-dimensional structures used by prokaryotes and eukaryotes alike to provide a variety of survival advantages. The morphology of the stratification of Pneumocystis cells in the mammalian lung coupled with the ability to survive under conditions of reduced oxygen levels (52) led investigators to probe the ability of Pneumocystis to form biofilms in vitro (24). Using an insert well system, they showed that a morphological transformation occurred over a 21-day period, was microscopically reproducible, and possessed characteristics of other fungal biofilms. The investigators demonstrated the ability of the biofilms to infect immunosuppressed rats. Moreover, the phase from planktonic (nonadherent) cells to sessile (biofilm) cells was assessed by microarray analysis and shown to involve specific genes. The establishment of such a system should facilitate studies of the life cycle and potential survival mechanisms and should be considered in future drug screening for use against these organisms.
Although long-term culture of any Pneumocystis species remains elusive, incremental improvements could serve to improve existing short-term culture systems, perhaps leading to a more sustainable method. One such in vitro study investigated the effects of vitamins and antioxidants on P. carinii and P. murina in short-term culture under minimal conditions (71). Real-time PCR was used to determine whether alteration of oxidant levels affected Pneumocystis rRNA internal transcribed spacer (ITS) transcription rates. Vitamin E caused an increase in organism number and transcriptional activity in this short-time culture. Several vitamin and nonvitamin antioxidants mediated increases in organism number and transcriptional activity, whereas treatment with nonantioxidant vitamins did not show these effects. These findings suggest that administration of antioxidants could be used to jump start the infection, reducing the length of time to severe disease. Such a finding would create a useful tool for investigators reliant upon organisms from animal models.
In vitro study of the organism remains complicated by inherent difficulties in purifying the life cycle stages for further study. High-speed cell sorting could provide a useful technique, using combinations of monoclonal and polyclonal antibodies to purify cystic and trophic forms and also to remove host cell debris (86).
HOST DEFENSE
Antibody responses to Pneumocystis in humans.
Although the clinical and experimental Pneumocystis literature has focused extensively on the components of cellular immunity that are important for defense against infection, humoral responses are also of considerable importance (26, 116). Most studies have focused on reactivities to the MSG family of surface proteins or to peptides of the entire protein. It is currently held that these proteins are important in several biological processes, including adhesion and escape from the host immune response (125). Whether antibody responses predict the outcome of infection is an area of ongoing interest. Sera obtained from patients enrolled in the Multicenter AIDS Cohort Study were examined to determine whether antibody responses predicted clinical outcome (31). Antibody levels directed against four recombinant fragments of the carboxyl terminus of the MSG were measured, and death from Pneumocystis pneumonia was associated with higher antibody levels directed against two of the fragments (MsgC1 and MsgC3) after adjusting for age and race. These data suggest that antibody responses could be important predictors of outcome.
Serum antibody responses to the carboxyl-terminal fragment of MSG were also compared for HIV-infected patients hospitalized with first episodes of Pneumocystis pneumonia and HIV-infected patients hospitalized with first episodes of pneumonia caused by other organisms (48). Responses to different recombinant fragments of MsgC varied, with changes in responses to MsgC1 being the most useful to discriminate the etiology of the pneumonia. Since reactivities to MSG were shown to vary among patients depending upon their exposure to Pneumocystis pneumonia, HIV-infected and uninfected populations in Cameroon were examined for serum antibody responses directed against MsgA, MsgB, and MsgC (104). There were no differences in antibody responses between HIV-infected and uninfected individuals. However, significant differences were present in the HIV-infected populations studied, depending on the use of sulfa prophylaxis at the location of their care. The data suggest that the differences in antibody responses are due to a lower incidence of Pneumocystis infection in the patients receiving sulfa prophylaxis.
Although much of the previous work in this field has focused on the carboxyl-terminal fragment of MSG, the amino terminus of the MSG molecule (MsgA) could also be an important antigenic determinant for the host response, as this is a more variable region than the conserved carboxy terminus. To explore this concept, a panel of six MsgA molecules was screened by enzyme-linked immunosorbent assay for reactivity with sera obtained from HIV-infected and uninfected individuals (27). The mean level of recognition of the antigen clones was higher for sera from HIV-infected individuals for most of the clones, suggesting that further investigation of responses to MsgA is warranted. Among health care workers, antibody responses to Msg have been used to evaluate exposure to and carriage of the organism (72). In a study of health care workers and controls, no differences in MsgA or MsgB were observed, but titers of MsgC were higher in the health care workers (130). There were no associations with age, gender, department of clinical activity, or duration of patient exposure. Whether the intensity of patient exposure or of exposure to respiratory secretions affects the antibody response remains to be investigated.
Antibody responses to Pneumocystis in animal models.
Because of the limitations of measuring antibody responses in humans, animal models have contributed important information to this field. As a nonhuman primate model, macaques with simian-human immunodeficiency virus (SHIV) infection were evaluated for immunoglobulin responses to the Pneumocystis protease kexin (58). About 95% of macaques had detectable baseline antibody titers against Pneumocystis. After SHIV infection and exposure to Pneumocystis-infected macaques, colonization was determined by antibody responses and PCR detection. Those macaques with higher baseline immunoglobulin G (IgG) titers and IgA titers were less likely to become colonized with Pneumocystis, suggesting that humoral responses prior to immunosuppression prevent or delay colonization.
Mouse models have been used extensively to study humoral immunity against infection, and B-cell-deficient mice are unable to clear Pneumocystis infection (82, 83). B cells in this model produce tumor necrosis factor (TNF), corresponding to increased B-cell proliferation in the lung and in draining lymph nodes. In adoptive transfer experiments using SCID mice, B cells were shown to be necessary for T-cell-mediated clearance of organisms (109). Further studies will address which aspects of B-cell-T-cell interaction are essential for clearance of infection. Natural antibodies are produced in the absence of external stimulatory signals and provide rapid and broad protection against pathogens. They play a critical but nonredundant role in the host humoral response. These antibodies are produced by a subset of B cells (B-1) that are long-lived and self-replenishing (3). The role of natural antibodies has not previously been studied in the context of Pneumocystis. However, natural antibodies could be important in responses to conserved fungal cell wall carbohydrates. Investigators showed that sera from specific-pathogen-free and germfree mice showed high titers of IgM directed against β-glucan and chitosan/chitin, essential components of fungal cell walls (112). Additionally, transfer of sera containing natural antibodies was more protective against Pneumocystis infection than transfer of sera without natural antibodies. Such studies should provide further insights into the complex host responses to these fungal organisms and perhaps suggest alternative therapeutic or prophylactic approaches.
Traditional vaccination is problematic in CD4-depleted hosts, such as individuals with untreated HIV infection. Accordingly, DNA vaccination provides an attractive alternative. Previously, vaccination with kexin, a Pneumocystis protease, in association with CD40 ligand has been shown to protect mice against challenge with Pneumocystis (144). To determine whether such a strategy could work across mammalian hosts, a region of kexin with high homology across species of Pneumocystis (miniKexin) was investigated (145). Intramuscular DNA vaccination elicited robust antibody responses in mice and protected them against Pneumocystis infection when the mice were subsequently depleted of CD4+ T cells. These results hold promise for future immunology-based treatment strategies. In another exploration of potential vaccine candidates, investigators screened a P. murina cDNA library by using a monoclonal antibody (4F11) that has been shown to cross-hybridize to the surface antigens of Pneumocystis organisms from various mammalian hosts, including humans (40, 140). A resultant clone, named A12, was fully sequenced and characterized. A12 was shown to have a region of similarity to PcKex1 but no homology to other known proteins. In previous studies, a partial construct of A12 conferred some immunological protection against P. murina infection (139) and, by inference, could be an important target against P. jirovecii as well.
Inflammatory signals in pathogenesis of pneumonia.
Development of lung inflammation is an important pathogenic determinant of immune reconstitution disease, but the inflammatory signals that are responsible for this syndrome require further investigation. The chemokine receptor CCR2 and its ligand, monocyte chemoattractant protein 1 (MCP-1), have been implicated in this hyperinflammatory syndrome (84). By crossing CCR2 knockout mice with SCID mice and then providing immune reconstitution with donor splenocytes, it was apparent that CCR2 contributed to inflammatory cell recruitment and lung injury (6). Furthermore, CCR2 expression on lymphocytes is required for recruitment of these cells to the lung.
Syndecan-1 is an abundant cell surface heparan sulfate proteoglycan and serves as a receptor for extracellular ligands and microorganisms. In a previous study, syndecan-1 knockout mice were found to have decreased susceptibility to infection with Pseudomonas aeruginosa (45). To evaluate the role of syndecan-1 in the progression of P. murina pneumonia, syndecan-1 null mice and wild-type controls were immunosuppressed and assessed for organism burden over 7 to 8 weeks. These studies demonstrated significantly higher organism burdens at late stages of infection than those in wild-type mice (85). In contrast to the role of syndecan-1 in other infections, this proteoglycan may help to control the growth of Pneumocystis at later stages of infection.
There are clear differences in immune responses to Pneumocystis in adults and neonates, and a mouse model was used to investigate these developmental differences (47). Specifically, proinflammatory cytokine production and T-lymphocyte recruitment were impaired in the lungs of neonatal mice. This delay in inflammation was associated with elevated transforming growth factor β1 in the lungs. Additionally, alveolar macrophages from neonatal mice failed to activate NF-κB in response to Pneumocystis. Therefore, differences in the lung environment and immune cells both contributed to delayed responses to Pneumocystis in neonates.
Immune responses to Pneumocystis contribute to inflammatory injury in several animal models of infection. Mice with SCID clear Pneumocystis after immunologic reconstitution but can develop fatal hyperinflammatory responses (90). The use of sulfasalazine to inhibit NF-κB, which is essential in signaling for many immune responses, significantly decreased lung inflammation in response to Pneumocystis, as well as decreasing the organism burden (136). This result was T cell dependent, and one mechanism that was suggested to explain these observations is increased apoptosis and turnover of alveolar macrophages.
Factors present in bronchoalveolar lavage fluid are known to induce lung inflammation in response to Pneumocystis (62). To further investigate and identify these factors, bronchoalveolar lavage samples were obtained from Pneumocystis-infected rats (68). These lavage samples, with or without Pneumocystis organisms, were used to inoculate subsequent groups of rats. The rats receiving lavage fluid plus organisms demonstrated accelerated weight loss and decreased survival. This effect was dependent upon the cellular fraction of the lavage samples and was present in both the adherent and the nonadherent cell fractions. Therefore, it was concluded that both phagocytes and lymphocytes contributed to this effect.
Cell wall components of Pneumocystis, including β-glucans, interact with alveolar macrophages and alveolar epithelial cells to stimulate the release of inflammatory mediators, contributing to lung inflammation and damage (75, 108, 133). An additional interaction is mediated through host cell membrane lactosylceramide (43). Using mice with established Pneumocystis infection, investigators showed that treatment with glycosphingolipid synthesis inhibitors (which reduce lactosylceramide levels) decreased both neutrophilic inflammation and organism burden (76). Furthermore, Pneumocystis contains a glucosylceramide synthesis gene, which is necessary for organism viability. Glycosphingolipid synthesis inhibitors could be beneficial to decrease both organism burden and host inflammation during infection and offer promise as supplemental therapeutic agents.
Nicotine has previously been shown to inhibit the development of Pneumocystis infection by reducing the supply of S-adenosylmethionine in the host lung (123). One mechanism of this effect may be upregulation of polyamine metabolism, a pathway that consumes S-adenosylmethionine. In a study presented at the workshop, investigators demonstrated that an inhibitor of ornithine decarboxylase, the polyamine anabolic rate-controlling enzyme difluormethylornithine, increased S-adenosylmethionine levels about 10% (96). Treatment with difluormethylornithine combined with nicotine resulted in reversal of S-adenosylmethionine depletion. Laser capture microdissection was then used to localize ornithine decarboxylase activity in the lung after nicotine treatment. Most of the increase in activity occurred in alveolar regions, with a minimal increase in airway epithelium, mirroring the anatomic distribution of Pneumocystis infection. Therefore, the proximate cause of S-adenosylmethionine depletion caused by nicotine occurs through upregulation of polyamine metabolism.
While clinical Pneumocystis infections are almost always confined to the lungs, with rare evidence of dissemination, animal models demonstrate that lung infection can modulate function in other organs (93). By examining a model in which mice are deficient in both lymphocytes and the type I interferon receptor, Pneumocystis infection was found to contribute to profound bone marrow suppression without evidence of organism dissemination beyond the lung (94). Although the mechanisms for this suppression are under active investigation, possibilities include a primary bone marrow effect or a lung-derived signal that occurs during Pneumocystis infection. These findings are intriguing, as they may help to explain some of the downstream pathologies in chronic disease states where Pneumocystis is present.
Alveolar macrophages.
Pneumocystis induces apoptosis and increases reactive oxygen species in alveolar macrophages (64). Previous investigations have determined that inhibition of this apoptosis by inhibition of caspase-9 improves survival (67). Because peptide inhibitors of apoptosis are difficult to use in vivo, the use of antioxidants provides another mechanism to inhibit apoptosis. N-Acetylcysteine in the rat model and Trolox (a water-soluble vitamin E derivative) in the mouse model both increased numbers of alveolar macrophages and decreased apoptosis (69). Furthermore, both compounds decreased organism burdens and improved survival, suggesting that antioxidant suppression of macrophage apoptosis could be a potential therapy for infection.
Another pathway important in alveolar macrophage responses to Pneumocystis depends on calmodulin expression (63). Calmodulin expression was decreased in alveolar macrophages obtained from animals with Pneumocystis infection, and this effect occurred in both dexamethasone-treated rats and CD4-depleted mice (70). The downregulation of calmodulin has important downstream effects, including decreased production of nitric oxide and granulocyte macrophage colony-stimulating factor. In contrast, calmodulin downregulation increases the production of reactive oxygen species, leading to increased alveolar macrophage apoptosis.
Phagocytosis of Pneumocystis by alveolar macrophages involves recognition using various cell surface receptors, including mannose and dectin-1 (38, 111). Alveolar macrophages obtained from CD4-depleted mice with and without Pneumocystis infection were analyzed for mRNA expression for the mannose receptor, dectin-1, and scavenger receptors (121). The expression of mannose receptor and dectin-1 was severely decreased in the macrophages obtained from infected mice, while expression of the scavenger receptor CD36 was only mildly decreased. These data help explain, in part, the defective phagocytosis of alveolar macrophages observed during Pneumocystis pneumonia.
In another investigation, the ability of polyamines to induce apoptosis in alveolar macrophages was examined. In alveolar macrophages obtained from Pneumocystis-infected rats, levels of polyamine oxidase were increased, but levels of spermine oxidase were unchanged. These data demonstrate that overexpression of polyamine oxidase in alveolar macrophages during infection increases the production of hydrogen peroxide and induces apoptosis (73). Using labeled spermidine, polyamine uptake assays were performed to determine whether extracellular polyamines contribute to apoptosis of alveolar macrophages during Pneumocystis infection (74). Macrophages from infected rats showed significant increases in polyamine uptake compared with macrophages from uninfected rats, demonstrating that increased polyamine levels in alveolar macrophages during Pneumocystis infection are at least partially caused by increased uptake.
GATA-2, a zinc finger transcription factor, is downregulated in alveolar macrophages during Pneumocystis infection, contributing to defective macrophage phagocytosis (65, 66, 129). To further investigate the mechanisms of defective phagocytosis, alveolar macrophages were obtained from healthy mice, and mannose receptor and dectin-1 expression was evaluated (143). Silencing RNA directed against GATA-2 decreased alveolar macrophage mRNA expression of mannose receptor and dectin-1, suggesting that GATA-2's effects are mediated by downregulation of receptors on macrophages. An additional transcription factor, PU.1 (purine-rich box 1), was also studied in alveolar macrophages during Pneumocystis infection (137). This transcription factor is usually expressed in hematopoietic cells and has important effects on macrophage surface molecules. In comparing alveolar macrophages from infected and uninfected rats, the expression of PU.1 was significantly inhibited by Pneumocystis infection. Furthermore, expression of PU.1 is controlled at least partially by GATA-2, as demonstrated by silencing RNA experiments. To investigate events further downstream, the ability of PU.1 expression to modulate dectin-1 expression was examined (138). Using a silencing RNA approach, inhibition of PU.1 expression caused significant decreases in dectin-1 expression at both the RNA and protein levels.
Lung pathology caused by Pneumocystis.
Accelerated emphysema has been documented for HIV-infected patients, particularly those who smoke (105). Additionally, Pneumocystis colonization has been documented for many groups of patients, including those with chronic obstructive pulmonary disease (COPD) (101). Whether Pneumocystis colonization could contribute to the pathogenesis of emphysema in these individuals is not known. A model using immunocompetent mice examined the interaction of cigarette smoke exposure and P. murina colonization, acquired by cohousing uninfected mice with infected mice (5). The combination of cigarette smoke exposure plus P. murina colonization resulted in airspace enlargement, indicative of emphysema, and pulmonary inflammation. This model provides an important research tool that will be useful in dissecting the immune responses and physiological changes outside the context of frank, fulminant infection.
In primates, Pneumocystis colonization produces significant changes in lung function (105). After infection with SHIV, macaques colonized with Pneumocystis demonstrated airway obstruction that was not reversible with bronchodilators (122). Quantitative computed tomography of the lungs demonstrated decreased tissue density consistent with the development of emphysema. Preliminary investigations of cytokine responses in the colonized macaques demonstrated increased inflammatory cytokines, which likely contribute to lung inflammation and the development of emphysematous changes.
Pulmonary hypertension is a known complication of HIV infection (127), but the mechanisms underlying the development of pulmonary hypertension are unclear. Using a mouse model, development of pulmonary hypertension was shown to coincide with a resurgence of CD4+ T cells in previously CD4-depleted mice (128). Using several knockout mice, experiments demonstrated that Th2 cytokines may have contributed to the development of perivascular fibrosis but were not absolutely required. In mice that underwent short-term depletion of CD4+ T cells, alveolar macrophages and dendritic cells showed elevated expression of scavenger receptor A (CD204), which has been implicated in development of fibrosis in other models. Ongoing work will address whether macrophages and dendritic cells expressing high levels of CD204 promote a profibrotic phenotype in pulmonary fibroblasts.
CARRIAGE AND COLONIZATION
Prevalence of Pneumocystis in animals.
All available published data confirm that Pneumocystis species are strictly specific for their mammalian hosts (28). For example, P. carinii infects rats but cannot infect closely related rodents, such as the mouse. A survey of the prevalence of Pneumocystis in bats from Central America, South America, and Europe determined that Pneumocystis was present in 33% of samples, as measured by PCR (1). Wild bats had a 46% prevalence rate, while captive bats had a 10% rate. DNA analysis of the Pneumocystis organisms revealed that the sequences were unique for each species of bats, in keeping with strict host specificity. Pneumocystis was also identified in species of bats previously not known to be infected. A study of bats in Brazil demonstrated the presence of Pneumocystis by PCR in several species, including Nyctinomops laticaudatus, Desmodus rotundas, and others (49). Thus, the range of mammalian host species susceptible to Pneumocystis continues to expand. The presence of Pneumocystis was also assessed in swine populations located in Brazil (9). Genetically distinct Pneumocystis genotypes existed in these swine, again by PCR, and these genotypes occurred in at least two separate clusters which were not separated by geography.
Prevalence of P. jirovecii in populations of humans.
Much of the recent PCR data documenting colonization has focused on specific patient populations, but the prevalence of colonization in general populations is unknown. In a prospective series of unselected individuals presenting for care at a local outpatient clinic in southern Spain, P. jirovecii DNA was present in about 10% of the individuals (92). Colonization was not associated with sex or smoking status but was associated with increased age. These studies indicate that immunocompetent persons could serve as potential reservoirs or sources of infection.
The prevalence of colonization in young HIV-infected patients has not been well documented. In a cohort of 20 individuals in Spain who were monitored since childhood, 10% had a history of P. jirovecii pneumonia and 40% were colonized (42). This relatively high rate of colonization was not associated with clinical factors, and none of the colonized individuals developed pneumonia during a 1-year follow-up period. However, such a high rate of prevalence indicates that the organism can commonly be found in this population, and this could facilitate the transmission of organisms with mutations in the dihydropteroate gene, as previously reported in the United States (4).
Colonization with P. jirovecii was previously reported for cystic fibrosis patients in Seville, Spain, with a prevalence of 22%, but whether this finding generalizes to other populations of cystic fibrosis patients is unknown (114). In Brittany, the French region with the highest prevalence of cystic fibrosis, only 1.3% of cystic fibrosis patients were colonized (103). These differences may be due to climatic factors but also could be due to differences in population density and in the incidence of HIV infection and P. jirovecii pneumonia in these two communities. Another study examined colonization in a cohort of cystic fibrosis patients in Brazil (142). About 38% of this patient group was colonized by PCR criteria. Colonization was associated with the presence of Pseudomonas spp. and with the absence of anti-Pneumocystis therapy, but these trends did not reach statistical significance.
Idiopathic pulmonary fibrosis is a chronic, progressive, interstitial lung disease with an unknown cause. The average survival from time of diagnosis varies from 2 to 4 years. A study of patients with idiopathic pulmonary fibrosis in Spain examined the modulation of host inflammation in response to Pneumocystis (39). About 40% of patients were colonized with P. jirovecii at the time of bronchoalveolar lavage, as detected by nested PCR targeting the mitochondrial large-subunit (mtLSU) rRNA. Lavage samples from colonized patients contained lower concentrations of TNF, interleukin-6, and surfactant protein D than did lavage samples from patients who were not colonized, but concentrations of interleukin-8 and surfactant protein A did not differ. This modulation of the inflammatory response could provide a mechanism by which organisms escape host recognition and removal.
COPD patients have a higher prevalence of Pneumocystis colonization than the general population (102), but the importance of cigarette smoking in the likelihood of colonization is controversial. In animal models, it has been demonstrated that nicotine reduces the rate of P. carinii infection due to depletion of S-adenosylmethionine (123). In a cohort of 238 COPD patients, about 15% were colonized and 76% were smokers (87). Smokers were more likely to be colonized (19%) than nonsmokers (5%), and multivariate analysis showed smoking to be an independent risk factor. These results are in contrast to those for animal models and may be due to other detrimental influences by nicotine, such as mucociliary dysfunction.
In contrast, P. jirovecii may induce systemic inflammatory responses in patients with COPD. Using a case-control design, P. jirovecii-colonized COPD patients were each matched with two noncolonized COPD patient controls (99). Serum concentrations of interleukin-8, TNF, interleukin-6, and MCP-1 were all significantly elevated in the colonized patients compared with those in the noncolonized controls. Since high levels of airway and systemic inflammatory markers are associated with a faster decline in lung function, the presence of P. jirovecii in these patients could contribute to the pathology of the disease syndrome.
A series of Brazilian children with hematologic malignancies but without clinical evidence of P. jirovecii pneumonia were examined for colonization by performing PCR on bronchoalveolar lavage specimens (141). About 19% of these children were colonized, suggesting that prophylaxis with anti-Pneumocystis drugs should be considered prior to an immunosuppressive treatment.
Much of the colonization literature examines bronchoalveolar lavage fluid or oropharyngeal washes, but there are data from animal models suggesting that oral swabs may be effective in documenting colonization (77). By collecting serial oral swabs from a cohort of HIV-infected individuals and then performing PCR, colonization was detected in 28% of the individuals studied over a period of 2 to 3 years (120). There were no clinical factors associated with swab positivity, but a trend toward lower CD4 numbers was observed. These findings are within the variation of results reported using other detection methods, and oral swabs could provide an alternative method of sample collection for patients who are unable to undergo bronchoscopy or to gargle.
A study designed to address whether health care workers could be carriers for Pneumocystis was conducted in an intensive care unit in France over a 1-year period (89). Two hundred forty-three volunteer workers underwent repeated oropharyngeal washings. P. jirovecii was detected by real-time PCR targeting the mtLSU gene. About 30% of the 487 samples were positive for P. jirovecii, comprising about 36% of the individuals studied. Of these individuals, about one-half had more than one positive sample. The percentage of individuals colonized increased when a patient with P. jirovecii pneumonia was hospitalized in the intensive care unit. These studies indicate that nurses and medical staff could be carriers and represent a potential source of infection for patients.
Use of real-time PCR for detection.
There have been concerns about using double-nested PCR to determine whether populations of individuals are colonized with P. jirovecii (50). Real-time PCR could advance this field by allowing quantitation, distinguishing colonization from infection. For HIV-infected individuals undergoing bronchoalveolar lavage or sputum induction, nested and real-time PCRs were compared (61). Nested PCR was directed to the mtLSU gene, and real-time PCR was performed using the same primers with a probe targeting the sequence. Among patients with clinical pneumonia, all were positive by nested PCR, but only 67% were positive by real-time PCR. Among patients without pneumonia, 23% were deemed colonized by nested PCR and 15% were positive by real-time PCR. Although real-time PCR was less sensitive than nested PCR, it demonstrated a high specificity and negative predictive value. Another study examined the utility of real-time PCR in detecting infection in AIDS patients with confirmed P. jirovecii pneumonia (117). Using oropharyngeal washings and induced sputa from patients with clinically confirmed pneumocystosis cases, the investigators used real-time PCR directed to the mtLSU rRNA gene. All samples were positive at the time of diagnosis, again suggesting that real-time PCR can be developed into a powerful diagnostic modality.
CLINICAL OUTCOME AND THERAPY
Pneumocystis genetic diversity, outcome, and transmission.
Previous literature reported mutations in DNA sequences within the P. jirovecii genome, with the most extensively studied mutations occurring at the dihydropteroate synthase (DHPS) locus (4). The DHPS enzyme is a target of sulfa-based prophylactic and therapeutic regimens, and the frequency of mutations increases with the duration of sulfa exposure (124). Whether mutations confer resistance or adversely affect clinical outcomes remains controversial. Similarly, whether clinical outcomes differ in infections caused by mixed genotypes is unclear. A prospective study of patients with proven infection (diagnosed by sputum induction or bronchoscopy) identified mixed infections in about 20% of samples (15). Although there was a trend toward worse outcomes for patients with mixed genotypes (or mutant genotypes) than for those with wild-type genotypes, the differences did not reach statistical significance.
In a retrospective analysis, the clinical outcome was correlated with an analysis of four genomic regions of Pneumocystis (ITS1, 26S, mt26S, and β-tubulin) and with DHPS mutations (132). The series of patients included individuals with HIV infection and with other causes of immunodeficiency. The overall mortality was 20% at 1 month. Predictors of death in this series were infection with the type 7 genotype, hematologic malignancy, older age, and a need for mechanical ventilation. In contrast, infection with the M2 DHPS mutant was associated with sulfa treatment success. Further large studies are needed to corroborate these results, which could have implications for treatment and management strategies.
A multilocus PCR method was developed to examine sequence diversity in thioredoxin reductase, thymidylate synthase, and β-tubulin gene sequences in HIV-infected individuals (36, 37). Genotyping demonstrated three nucleotide sequences for the β-tubulin locus, but only the wild-type sequence was observed for the other two loci. Such multiplex PCR may yield important information in correlating genetic diversity with clinical outcomes.
A study of P. jirovecii isolates from infants examined the genetic diversity of the 5.8S rRNA gene, using primers specific for the ITS1 and ITS2 loci (131). Specific mutations were observed, and the diversity in this gene appears to be greater than was previously thought (81). More investigation will be needed to determine whether the genetic diversity observed is associated with specific geographic or clinical factors.
Respiratory transmission of Pneumocystis spp. is now accepted, but whether other routes of acquisition are important requires further study. Transplacental transmission was suggested in an earlier report using histological methods to detect P. jirovecii in stillborn infants (110). To determine whether transplacental transmission of P. jirovecii might occur, tissues from human fetuses and placentas were evaluated using the exquisitely sensitive detection method of PCR targeting the mtLSU and DHPS genes (97). P. jirovecii DNA was identified in 35% of fetal tissues and in one placenta, suggesting that transplacental transmission can occur in humans and that it is not a rare event. Transplacental transmission has been shown to occur in rabbits (11) but not in mice or rats (51), suggesting that there are species-specific differences among transmission routes.
Restriction fragment length polymorphism (RFLP) analysis was employed as a means to determine if variability in the repertoire of the MSG gene family of P. jirovecii could be used as a typing method to distinguish isolates from infected human beings (115). After amplification of an ∼1,300-bp region of Msg, RFLP analysis followed by Southern blot assay and computer-assisted analysis of fingerprints revealed that differences could be identified among the isolates. However, reproducibility was shown to be highest for samples with higher organism loads. RFLP patterns were conserved in paired samples collected from the same patients at close time points, but distinct profiles were observed among samples from different individuals. Such a system may be helpful in studying clusters of patients to determine whether there was a common source of infection.
Diagnostic and therapeutic developments.
Current diagnostics for Pneumocystis pneumonia depend upon morphological or DNA detection via PCR with respiratory specimens. However, β-glucans are components of fungal cell walls and can be detected in sera. There are anecdotal reports of the utility of commercial β-glucan test kits for diagnosis of Pneumocystis jirovecii infections in patients (55, 88). Sera from patients with confirmed Pneumocystis pneumonia were compared with those from negative controls (30). Using a commercially available assay (Fungitell; Cape Cod Associates), sera were positive for β-glucan in the patients with pneumonia, at significantly higher concentrations than those in sera from negative controls. Such a noninvasive test could augment current methods for diagnosis, but caution must be used in interpretation, since other fungal infections will also test positive with this assay.
Current drugs to treat Pneumocystis infection have limitations caused by their toxicity and sometimes by a lack of efficacy. Few new drug targets have emerged, despite concerted efforts of the scientific community. The echinocandins, antifungal drugs that inhibit β-(1,3)-d-glucan synthesis, have shown promise in several models of Pneumocystis infection (118). Using a mouse model, three echinocandins (caspofungin, anidulafungin, and micafungin) were shown to be highly efficacious in decreasing cyst burdens and had no observable toxicity (25). Micafungin was shown to lose efficacy at 1 mg/kg of body weight or less, whereas anidulafungin and caspofungin remained efficacious at concentrations as low as 0.1 mg/kg given once per week. The reductions were comparable to those achieved with trimethoprim-sulfamethoxazole, suggesting that this drug class may have promise for treating clinical infections.
Interestingly, in vitro studies from the same laboratory that evaluated the echinocandins in the mouse model of pneumocystosis showed little to no effect at concentrations of 100 μg/ml or below in an established short-term drug screening assay based on ATP levels (23). Consideration of recent reports of the effects of sera on MICs of echinocandins in assays of Candida and Aspergillus (107) resulted in a serum concentration study with anidulafungin, caspofungin, and micafungin against P. carinii planktonic and biofilm cultures (16). Planktonic-phase organisms were refractory to anidulafungin and caspofungin at 10 and 20% concentrations of calf serum, but this was reversed at 1% and 5% concentrations. Micafungin had little effect at any serum concentration. In established biofilms, organisms were resistant to caspofungin at all serum concentrations, while micafungin and anidulafungin had increased activity with decreasing serum concentrations. These data strongly suggest that investigators evaluate in vitro efficacy in the context of serum concentration and the physical phase of the in vitro system.
Potential drug targets.
Although Pneumocystis spp. are resistant to standard antifungals that target sterol biosynthesis, e.g., azoles, previous in vitro studies demonstrated the efficacy of some proprietary inhibitors of the sterol pathway (54). More recently, gene inventories from the Pneumocystis genome project showed the presence of many genes in the sterol biosynthetic pathway, many of which were expressed during fulminant infection (22). Since the bulk sterol of Pneumocystis is cholesterol rather than ergosterol, it has been assumed that uptake or salvage of cholesterol from the host was the primary means to obtain this essential compound. The expression and in vitro inhibition studies suggest that a sterol biosynthetic pathway is at least partially functional. It was recently reported that blockade of Hsp90 in several yeast erg3 mutants reduced their resistance to azoles. Because P. carinii appears to lack the Erg3 gene, actively transcribes Hsp90, and has a functional Erg11 gene (the target of azoles), the effects of the combination of geldanamycin (GDA), an Hsp90 inhibitor, and various sterol pathway inhibitors were evaluated in an ATP assay system (17, 23). A range of results were observed. The IC50 for berberine, a sterol C24 methyltransferase inhibitor, was decreased 95% by 10 ng/ml GDA; the triazole hexaconazole IC50 was reduced 32%; and that of simvastatin, an HMG coenzyme A reductase, was reduced 25%. Hsp90 inhibitors structurally related to GDA are currently in phase I/II clinical trials as anticancer drugs, and concentrations needed to overcome fungal drug resistance are clinically well tolerated. These inhibitors could be used in combination with lower doses of anti-Pneumocystis drugs or in combination with novel therapeutic agents.
Another potential drug target in the P. carinii sterol biosynthetic pathway, PcErg7, was functionally characterized and localized to lipid particles in a heterologous yeast system. The PcErg7 gene, which encodes lanosterol synthase, was chosen because its product is the first sterol intermediate in the sterol biosynthetic pathway (53). The gene contains seven introns, and the cDNA was able to functionally complement an erg7 mutation of S. cerevisiae, which is a lethal mutation in yeast. It was also shown that lanosterol, the product of the enzymatic reactions of Erg7p, was produced by the PcErg7 in yeast. Localization to lipid particles in yeast and in P. carinii was demonstrated using a polyclonal antibody to PcErg7p. The latter finding was in contrast to an earlier study that reported it did not traffic to these organelles in yeast (95). Localization to lipid particles is important for enzyme functionality, and these studies support the activity of this enzyme in P. carinii. Although the sterol biosynthetic pathway was not previously considered a viable target for Pneumocystis drug development, the presence and activities of many of these gene products open another potential avenue to pursue.
SUMMARY
The contrast between the depth and quality of presentations during the 1st workshop, in 1988, and those at the 10th workshop, in 2008, is a testament to the progress that has been made during the ensuing 20 years. The genome and transcriptome of P. carinii have been sequenced, the key players in metabolic functions and pathways are being dissected using heterologous systems, and the interplay between host responses to the MSGs and the genetic nature of antigenic variation are being exploited for diverse purposes, including diagnostic modalities and a better understanding of these organisms' survival strategies. The host responses to the organisms are being delineated more finely and appear to involve an entire cadre of cytokines, immune cells, the humoral arm, and other host-related factors. Host responses are also dependent on the physiological state of the particular host and differ across a spectrum of intact defense to frank immunosuppression. In some cases, it appears that the organism itself can downregulate certain immune responses, such as phagocytosis. The role of β-glucan continues to evolve as an injurious inflammatory factor but also as a key signature molecule for diagnosis. Entirely new populations of carriers, reservoirs, and those under colonization are being defined. Many such populations have underlying chronic diseases, such as COPD, to which it is suspected that Pneumocystis contributes in a harmful manner. Although a long-term in vitro culture system remains elusive, antioxidants and vitamins appear to increase replication, and the report of biofilm formation by P. murina and P. carinii holds promise for a more tractable system. As research continues to progress in this area, these once enigmatic fungi are becoming more approachable and understandable and now provide insights into the diversity of microbial survival strategies and biological processes.
Acknowledgments
This work was supported in part by NIH grant R01 HL083482 (J.M.B.), by merit review funds from the Department of Veterans Affairs (J.M.B. and M.T.C.), and by NIH grants R01 AI050450 and N01-A1-25647 (M.T.C.).
Footnotes
Published ahead of print on 23 January 2009.
REFERENCES
- 1.Akbar, H., C. M. Aliouat, S. Derouiche, M. L. Taylor, M. Chabe, L. E. Carreto-Binaghi, A. Courpon, E. M. Aliouat, E. Dei-Cas, and C. Demanche. 2008. Prevalence of Pneumocystis in bats, abstr. PL6. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 2.Arnold, J., and M. T. Cushion. 1997. Constructing a physical map of the Pneumocystis genome. J. Eukaryot. Microbiol. 448S. [DOI] [PubMed] [Google Scholar]
- 3.Baumgarth, N., J. W. Tung, and L. A. Herzenberg. 2005. Inherent specificities in natural antibodies: a key to immune defense against pathogen invasion. Springer Semin. Immunopathol. 26347-362. [DOI] [PubMed] [Google Scholar]
- 4.Beard, C. B., J. L. Carter, S. P. Keely, L. Huang, N. J. Pieniazek, I. N. Moura, J. M. Roberts, A. W. Hightower, M. S. Bens, A. R. Freeman, S. Lee, J. R. Stringer, J. S. Duchin, C. del Rio, D. Rimland, R. P. Baughman, D. A. Levy, V. J. Dietz, P. Simon, and T. R. Navin. 2000. Genetic variation in Pneumocystis carinii isolates from different geographic regions: implications for transmission. Emerg. Infect. Dis. 6265-272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beck, J. M., A. M. Preston, T. Ling, M. Du, W. B. Fields, J. L. Curtis, and P. J. Christensen. 2008. Pneumocystis infection and cigarette smoke exposure interact to cause delayed clearance of organisms, development of airspace enlargement, and pulmonary inflammation in mice, abstr. PL32. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 6.Bhagwat, S. P., F. Gigliotti, and T. W. Wright. 2008. Role of CCR2 in inflammatory cell recruitment and kinetics of inflammatory lung injury in the Pneumocystis-mediated immune restitution disease (IRD), abstr. PO36. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 7.Burgess, J. W., T. J. Kottom, J. D. Lamont, E. M. Baden, M. Ramirez-Alvarado, and A. H. Limper. 2008. The meiotic inhibitor PcRan1 kinase from Pneumocystis carinii demonstrates a temperature-sensitive, pH refractory enzymatic profile, abstr. PL7. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 8.Carini, A. 1910. Fòrmas de eschizogonia do Trypanosoma lewisi. Commun. Soc. Med. Sao Paulo 1910204. [Google Scholar]
- 9.Cavallini Sanches, E. M., M. Borba, A. Spanamberg, A. P. Ravazzolo, J. M. Santurio, D. E. S. N. Barcellos, D. Driemieir, M. Berthelemy, J. Guillot, and L. Ferreiro. 2008. Phylogeny of Pneumocystis obtained from thirty-four lungs of Brazilian swine, abstr. PO2. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 10.Cavalier-Smith, T. 1998. A revised six-kingdom system of life. Biol. Rev. 73203-266. [DOI] [PubMed] [Google Scholar]
- 11.Cere, N., F. Drouet-Viard, E. Dei-Cas, N. Chanteloup, and P. Coudert. 1997. In utero transmission of Pneumocystis carinii sp. f. oryctolagi. Parasite 4325-330. [DOI] [PubMed] [Google Scholar]
- 12.Chabe, M., A. G. Smulian, M. S. Collins, E. M. Aliouat, C. M. Aliouat, E. Dei-Cas, and M. Cushion. 2008. Comparison of Pneumocystis murina gene expression profiles between carriage and disease states in mice: preliminary results, abstr. PL41. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 13.Chagas, C. 1909. Nova tripanozomiase humana: estudos sobre a morfolojia e o ciclo evolutivo do Schizotrypanum cruzi n.gen., n.sp., ajente etiolojico de nova entidade morbida do homem. Mem. Inst. Oswaldo Cruz 1159-218. [Google Scholar]
- 14.Chagas, C. 1909. Trabalho do Instituto Manguinhos sobre uma nova trypanosomiase humana, pelo dr. Carlos Chagas, assistente do Instituto. Ann. Acad. Med. Rio de Janeiro 75188-190. [Google Scholar]
- 15.Chi, A., C. B. Beard, K. Crothers, J. L. Jones, G. G. Lawrence, M. F. Avery, S. Eiser, M. R. Bensley, and L. Huang. 2008. HIV-associated Pneumocystis pneumonia (PCP) with mixed Pneumocystis dihydropteroate synthase (DHPS) genotype infection: predictors and outcomes, abstr. PL38. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 16.Collins, M. S., and M. T. Cushion. 2008. Effects of serum on in vitro susceptibility of Pneumocystis carinii to echinocandins, abstr. PO6. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 17.Collins, M. S., and M. T. Cushion. 2008. The anti-Pneumocystis carinii activities of sterol biosynthesis inhibitors are potentiated by the addition of geldanamycin, an HSP90 inhibitor, abstr. PO11. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 18.Cushion, M. T., and J. Arnold. 1997. Proposal for a Pneumocystis genome project. J. Eukaryot. Microbiol. 447S. [DOI] [PubMed] [Google Scholar]
- 19.Cushion, M. T., S. P. Keely, and J. R. Stringer. 2004. Molecular and phenotypic description of Pneumocystis wakefieldiae sp. nov., a new species in rats. Mycologia 96429-438. [PubMed] [Google Scholar]
- 20.Cushion, M. T., S. P. Keely, and J. R. Stringer. 2005. Validation of the name Pneumocystis wakefieldiae. Mycologia 97268. [PubMed] [Google Scholar]
- 21.Cushion, M. T., and A. G. Smulian. 2001. The Pneumocystis genome project: update and issues. J. Eukaryot. Microbiol. 2001(Suppl.)182S-183S. [DOI] [PubMed] [Google Scholar]
- 22.Cushion, M. T., A. G. Smulian, B. E. Slaven, T. Sesterhenn, J. Arnold, C. Staben, A. Porollo, R. Adamczak, and J. Meller. 2007. Transcriptome of Pneumocystis carinii during fulminate infection: carbohydrate metabolism and the concept of a compatible parasite. PLoS One 2e423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cushion, M. T., P. D. Walzer, A. Ashbaugh, S. Rebholz, R. Brubaker, J. J. Vanden Eynde, A. Mayence, and T. L. Huang. 2006. In vitro selection and in vivo efficacy of piperazine- and alkanediamide-linked bisbenzamidines against Pneumocystis pneumonia in mice. Antimicrob. Agents Chemother. 502337-2343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cushion, M. T., M. S. Collins, T. Sesterhenn, and A. G. Smulian. 2008. Biofilm formation by Pneumocystis spp., abstr. PL54. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 25.Cushion, M. T., A. D. Ashbaugh, M. S. Collins, and P. D. Walzer. 2008. In vivo efficacy of echinocandins in a mouse model of Pneumocystis murina pneumonia, abstr. PO51. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 26.Daly, K. R., L. Huang, A. Morris, J. Koch, K. Crothers, L. Levin, S. Eiser, S. Satwah, P. Zucchi, and P. D. Walzer. 2006. Antibody response to Pneumocystis jirovecii major surface glycoprotein. Emerg. Infect. Dis. 121231-1237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Daly, K. R., J. V. Koch, and P. D. Walzer. 2008. Antigenic complexity at the amino terminus of the major surface glycoprotein of Pneumocystis jirovecii, abstr. PO4. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 28.Dei-Cas, E., M. Chabe, R. Moukhlis, I. Durand-Joly, E. M. Aliouat, J. R. Stringer, M. Cushion, C. Noel, G. S. de Hoog, J. Guillot, and E. Viscogliosi. 2006. Pneumocystis oryctolagi sp. nov., an uncultured fungus causing pneumonia in rabbits at weaning: review of current knowledge, and description of a new taxon on genotypic, phylogenetic and phenotypic bases. FEMS Microbiol. Rev. 30853-871. [DOI] [PubMed] [Google Scholar]
- 29.Delanoë, P., and M. Delanoë. 1912. Sur les rapports des kystes de Carini du poumon des rats avec le Trypanosoma lewisi. C. R. Acad. Sci. 155658-660. [Google Scholar]
- 30.Divaret, G., D. Magne, A. Angoulvant, and P. Roux. 2008. Usefulness of serum (1-3)β-d-glucan assay for Pneumocystis jirovecii pneumoniae diagnosis, abstr. PO17. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 31.Djawe, K., L. Levin, K. R. Daly, J. Koch, A. Morris, and P. D. Walzer. 2008. Identification of specific host factors associated with high antibody levels to Pneumocystis antigens in HIV patients, abstr. PO37. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 32.Edman, J. C., J. A. Kovacs, H. Masur, D. V. Santi, H. J. Elwood, and M. L. Sogin. 1989. Ribosomal RNA genes of Pneumocystis carinii. J. Protozool. 3618S-20S. [DOI] [PubMed] [Google Scholar]
- 33.Eriksson, O. E., and K. Winka. 1997. Supraordinal taxa of Ascomycota. Myconet 11-16. [Google Scholar]
- 34.Reference deleted.
- 35.Eriksson, O. E. 1994. Pneumocystis carinii, a parasite in lungs of mammals, referred to a new family and order (Pneumocystidaceae, Pneumocystidales, Ascomycota). Systema Ascomycetum 13165-180. [Google Scholar]
- 36.Esteves, F., M. C. Costa, M. I. Moser, A. Tavares, J. Gaspar, T. Marques, R. Leite, F. Antunes, K. Mansinho, and O. Matos. 2008. Pneumocystis jirovecii multilocus genotyping: relationship with clinical data—preliminary study, abstr. PL43. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 37.Esteves, F., J. Gaspar, F. Antunes, K. Mansinho, and O. Matos. 2008. Molecular analysis of three Pneumocystis jirovecii gene loci in HIV-positive Portuguese patients, abstr. PO8. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 38.Ezekowitz, R. A., D. J. Williams, H. Koziel, M. Y. Armstrong, A. Warner, F. F. Richards, and R. M. Rose. 1991. Uptake of Pneumocystis carinii mediated by the macrophage mannose receptor. Nature 351155-158. [DOI] [PubMed] [Google Scholar]
- 39.Friaza, V., S. Gutierrez, C. De La Horra, J. Martin-Juan, M. A. Montes-Cano, R. Morilla, L. Rivero, I. Martin-Garrido, F. J. Medrano, J. M. Varela, E. J. Calderón, and N. Respaldiza. 2008. Modulation of the host inflammatory response in idiopathic pulmonary fibrosis patients colonized by Pneumocystis jirovecii, abstr. PO14. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 40.Gigliotti, F., D. C. Stokes, A. B. Cheatham, D. S. Davis, and W. T. Hughes. 1986. Development of murine monoclonal antibodies to Pneumocystis carinii. J. Infect. Dis. 154315-322. [DOI] [PubMed] [Google Scholar]
- 41.Greuter, W., J. McNeill, F. R. Barrie, H. M. Burdet, V. Demoulin, T. S. Filgueiras, D. H. Nicolson, P. C. Silva, J. E. T. P. Skog, N. J. Turland, and D. L. Hawksworth (ed.). 2000. International code of botanical nomenclature (St. Louis code). Adopted by the Sixteenth International Botanical Congress, St. Louis, Missouri, July-August 1999. Regnum Veg. 1381-474. [Google Scholar]
- 42.Gutiérrez, S., I. Martin-Garrido, J. A. León, R. Morilla, L. Rivero, V. Friaza, N. Respaldiza, J. M. Varela, M. A. Montes Cano, F. J. Medrano, C. De La Horra, and E. Calderón. 2008. High prevalence of Pneumocystis jirovecii colonization among young HIV-infected patients, abstr. PO45. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 43.Hahn, P. Y., S. E. Evans, T. J. Kottom, J. E. Standing, R. E. Pagano, and A. H. Limper. 2003. Pneumocystis carinii cell wall beta-glucan induces release of macrophage inflammatory protein-2 from alveolar epithelial cells via a lactosylceramide-mediated mechanism. J. Biol. Chem. 2782043-2050. [DOI] [PubMed] [Google Scholar]
- 44.Hauser, P. M., L. Lo Presti, M. Cockell, L. Cerutti, and V. Simanis. 2008. Functional gene analysis of Pneumocystis carinii by complementation of yeast mutants, abstr. PL42. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 45.Haynes, A., III, F. Ruda, J. Oliver, A. N. Hamood, J. A. Griswold, P. W. Park, and K. P. Rumbaugh. 2005. Syndecan 1 shedding contributes to Pseudomonas aeruginosa sepsis. Infect. Immun. 737914-7921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hibbett, D. S., M. Binder, J. F. Bischoff, M. Blackwell, P. F. Cannon, O. E. Eriksson, S. Huhndorf, T. James, P. M. Kirk, R. Lucking, L. H. Thorsten, F. Lutzoni, P. B. Matheny, D. J. McLaughlin, M. J. Powell, S. Redhead, C. L. Schoch, J. W. Spatafora, J. A. Stalpers, R. Vilgalys, M. C. Aime, A. Aptroot, R. Bauer, D. Begerow, G. L. Benny, L. A. Castlebury, P. W. Crous, Y. C. Dai, W. Gams, D. M. Geiser, G. W. Griffith, C. Gueidan, D. L. Hawksworth, G. Hestmark, K. Hosaka, R. A. Humber, K. D. Hyde, J. E. Ironside, U. Koljalg, C. P. Kurtzman, K. H. Larsson, R. Lichtwardt, J. Longcore, J. Miadlikowska, A. Miller, J. M. Moncalvo, S. Mozley-Standridge, F. Oberwinkler, E. Parmasto, V. Reeb, J. D. Rogers, C. Roux, L. Ryvarden, J. P. Sampaio, A. Schussler, J. Sugiyama, R. G. Thorn, L. Tibell, W. A. Untereiner, C. Walker, Z. Wang, A. Weir, M. Weiss, M. M. White, K. Winka, Y. J. Yao, and N. Zhang. 2007. A higher-level phylogenetic classification of the fungi. Mycol. Res. 111509-547. [DOI] [PubMed] [Google Scholar]
- 47.Hollifield, M. L., A. Rogosky, K. M. Empey, and B. A. Garvy. 2008. Unresponsiveness to Pneumocystis in neonatal mice is due to developmental control of anti-inflammatory cytokines and intrinsic unresponsiveness in alveolar macrophages, abstr. PO38. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 48.Huang, L., K. R. Daly, A. Swartzman, M. R. Bensley, B. Roth, J. Koch, K. Djawe, L. Levin, and P. D. Walzer. 2008. Serum antibody responses in HIV patients with pneumonia due to Pneumocystis jirovecii and other causes, abstr. PO5. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 49.Hummel, J., E. M. Cavallini Sanches, S. M. Pacheco, A. S. Cericatto, R. M. Melo, A. Spanamberg, E. M. Colodel, J. M. Santurio, and L. Ferreiro. 2008. Detection of Pneumocystis in lungs of bats from Brazil by PCR amplification, abstr. PO1. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 50.Huggett, J. F., R. F. Miller, M. S. Taylor, A. M. Costello, and A. Zumla. 2006. Problems of developing molecular diagnostic tests for opportunistic pathogens: the example of Pneumocystis jirovecii. J. Eukaryot. Microbiol. 53(Suppl. 1)S85-S86. [DOI] [PubMed] [Google Scholar]
- 51.Icenhour, C. R., S. L. Rebholz, M. S. Collins, and M. T. Cushion. 2001. Early acquisition of Pneumocystis carinii in neonatal rats using targeted PCR and oral swabs. J. Eukaryot. Microbiol. 2001(Suppl.)135S-136S. [DOI] [PubMed] [Google Scholar]
- 52.Joffrion, T. M., M. S. Collins, and M. T. Cushion. 2006. Microaerophilic conditions increase viability and affect responses of Pneumocystis carinii to drugs in vitro. J. Eukaryot. Microbiol. 53(Suppl. 1)S117-S118. [DOI] [PubMed] [Google Scholar]
- 53.Joffrion, T., T. Sesterhenn, and M. T. Cushion. 2008. Functional characterization and localization of P. carinii lanosterol synthase, abstr. PL24. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 54.Kaneshiro, E. S., M. S. Collins, and M. T. Cushion. 2000. Inhibitors of sterol biosynthesis and amphotericin B reduce the viability of Pneumocystis carinii f. sp. carinii. Antimicrob. Agents Chemother. 441630-1638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kawagishi, N., S. Miyagi, K. Satoh, Y. Akamatsu, S. Sekiguchi, and S. Satomi. 2007. Usefulness of beta-d glucan in diagnosing Pneumocystis carinii pneumonia and monitoring its treatment in a living-donor liver-transplant recipient. J. Hepatobiliary Pancreat. Surg. 14308-311. [DOI] [PubMed] [Google Scholar]
- 56.Keely, S. P., J. M. Fischer, M. T. Cushion, and J. R. Stringer. 2004. Phylogenetic identification of Pneumocystis murina sp. nov., a new species in laboratory mice. Microbiology 1501153-1165. [DOI] [PubMed] [Google Scholar]
- 57.Keely, S. P., and J. R. Stringer. 2008. Genetic variation in major surface glycoprotein genes, abstr. PL45. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 58.Kling, H. M., T. Shipley, S. Patil, A. Morris, and K. A. Norris. 2008. Pneumocystis-specific antibodies protect SHIV-immunosuppressed macaques from colonization, abstr. PL35. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 59.Kottom, T. J., J. R. Kohler, C. F. Thomas, Jr., G. R. Fink, and A. H. Limper. 2003. Lung epithelial cells and extracellular matrix components induce expression of Pneumocystis carinii STE20, a gene complementing the mating and pseudohyphal growth defects of STE20 mutant yeast. Infect. Immun. 716463-6471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Krajicek, B. J., T. J. Kottom, and A. H. Limper. 2008. Characterization of the Cdc42 small G-protein from Pneumocystis carinii, abstr. PL9. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 61.Kristoff, J., S. Radhi, R. Srivastava, T. Alexander, T. Johnson, J. Kolls, and A. Morris. 2008. Real time PCR detection of PCP and Pneumocystis colonization in HIV-positive subjects, abstr. PO16. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 62.Lasbury, M. E., P. J. Durant, and C. H. Lee. 2001. Bronchoalveolar lavage fluid from Pneumocystis carinii-infected rats inhibits phagocytosis in normal alveolar macrophages. J. Eukaryot. Microbiol. 2001(Suppl.)163S-164S. [DOI] [PubMed] [Google Scholar]
- 63.Lasbury, M. E., P. J. Durant, S. H. Wang, C. Zhang, C. P. Liao, D. Tschang, and C. H. Lee. 2006. GM-CSF expression in the lung during Pneumocystis pneumonia. J. Eukaryot. Microbiol. 53(Suppl. 1)S124-S126. [DOI] [PubMed] [Google Scholar]
- 64.Lasbury, M. E., S. Merali, P. J. Durant, D. Tschang, C. A. Ray, and C. H. Lee. 2007. Polyamine-mediated apoptosis of alveolar macrophages during Pneumocystis pneumonia. J. Biol. Chem. 28211009-11020. [DOI] [PubMed] [Google Scholar]
- 65.Lasbury, M. E., X. Tang, P. J. Durant, and C. H. Lee. 2001. Effect of the transcription factor GATA-2 on phagocytic activity of alveolar macrophages from Pneumocystis carinii-infected hosts. J. Eukaryot. Microbiol. 2001(Suppl.)158S-159S. [DOI] [PubMed] [Google Scholar]
- 66.Lasbury, M. E., X. Tang, P. J. Durant, and C. H. Lee. 2003. Effect of transcription factor GATA-2 on phagocytic activity of alveolar macrophages from Pneumocystis carinii-infected hosts. Infect. Immun. 714943-4952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lasbury, M. E., S. H. Wang, C. Zhang, C. P. Liao, P. J. Durant, D. Tschang, and C. H. Lee. 2006. Caspase-9 as a target for Pneumocystis pneumonia therapy. J. Eukaryot. Microbiol. 53(Suppl. 1)S138-S139. [DOI] [PubMed] [Google Scholar]
- 68.Lasbury, M. E., P. J. Durant, C. Zhang, C. Liao, and C. Lee. 2008. Bronchoalveolar lavage components contribute to inflammation during Pneumocystis pneumonia, abstr. PL31. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 69.Lasbury, M. E. 2008. Potential of antioxidant therapy in established Pneumocystis pneumonia, abstr. PL39. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 70.Lasbury, M. E., and P. J. Durant. 2008. Reductions in alveolar macrophage calmodulin and its functions during Pneumocystis pneumonia, abstr. PL48. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 71.Lasbury, M. E., P. J. Durant, C. Zhang, C. Liao, and C. Lee. 2008. In vitro effects of vitamins and antioxidants on Pneumocystis proliferation and rRNA transcription as assessed by real-time PCR, abstr. PO47. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 72.Leigh, T. R., M. J. Millett, B. Jameson, and J. V. Collins. 1993. Serum titres of Pneumocystis carinii antibody in health care workers caring for patients with AIDS. Thorax 48619-621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Liao, C., M. E. Lasbury, S. Wang, C. Zhang, S. Shao, P. J. Durant, and C. Lee. 2008. Overexpression of polyamine oxidase in alveolar macrophage during Pneumocystis pneumonia, abstr. PL50. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 74.Liao, C., M. E. Lasbury, S. Wang, C. Zhang, S. Shao, P. J. Durant, and C. Lee. 2008. Increased uptake of polyamines by alveolar macrophages during Pneumocystis pneumonia, abstr. PO48. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 75.Limper, A. H., F. Lebron, S. E. Evans, and R. Y. Hahn. 2003. Pneumocystis carinii: cell wall beta-glucan-mediated pulmonary inflammation. J. Eukaryot. Microbiol. 50(Suppl.)646. [DOI] [PubMed] [Google Scholar]
- 76.Limper, A. H., Z. Vuk-Pavlovic, T. J. Kottom, and R. E. Pagano. 2008. A novel approach to treat lung inflammation and organism burden in Pneumocystis pneumonia, abstr. PL34. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 77.Linke, M. J., S. Rebholz, M. Collins, R. Tanaka, and M. T. Cushion. 2003. Noninvasive method for monitoring Pneumocystis carinii pneumonia. Emerg. Infect. Dis. 91613-1616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Liu, Y. J., and B. D. Hall. 2004. Body plan evolution of ascomycetes, as inferred from an RNA polymerase II phylogeny. Proc. Natl. Acad. Sci. USA 1014507-4512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lo, P. L., M. Cockell, L. Cerutti, V. Simanis, and P. M. Hauser. 2007. Functional characterization of Pneumocystis carinii brl1 by transspecies complementation analysis. Eukaryot. Cell 62448-2452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lo Presti, L., L. Cerutti, M. Monod, and P. M. Hauser. 2008. Functional complementation in Saccharomyces cerevisiae with Pneumocystis carinii genes: which promoter to use?, abstr. PO12. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 81.Lu, J. J., M. S. Bartlett, M. M. Shaw, S. F. Queener, J. W. Smith, M. Ortiz-Rivera, M. J. Leibowitz, and C. H. Lee. 1994. Typing of Pneumocystis carinii strains that infect humans based on nucleotide sequence variations of internal transcribed spacers of rRNA genes. J. Clin. Microbiol. 322904-2912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lund, F. E., M. Hollifield, K. Schuer, J. L. Lines, T. D. Randall, and B. A. Garvy. 2006. B cells are required for generation of protective effector and memory CD4 cells in response to Pneumocystis lung infection. J. Immunol. 1766147-6154. [DOI] [PubMed] [Google Scholar]
- 83.Lund, F. E., K. Schuer, M. Hollifield, T. D. Randall, and B. A. Garvy. 2003. Clearance of Pneumocystis carinii in mice is dependent on B cells but not on P. carinii-specific antibody. J. Immunol. 1711423-1430. [DOI] [PubMed] [Google Scholar]
- 84.Lundien, M. C., K. A. Mohammed, N. Nasreen, R. S. Tepper, J. A. Hardwick, K. L. Sanders, R. D. Van Horn, and V. B. Antony. 2002. Induction of MCP-1 expression in airway epithelial cells: role of CCR2 receptor in airway epithelial injury. J. Clin. Immunol. 22144-152. [DOI] [PubMed] [Google Scholar]
- 85.Lynch, K., P. W. Park, and M. T. Cushion. 2008. Syndecan 1 null mice are susceptible to infection by Pneumocystis murina and develop higher organism burdens than wild type mice, abstr. PO50. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 86.Martinez, A., E. M. Aliouat, M. Pottier, N. Gantois, A. Vitse, C. Pinçon, E. Dei-Cas, and C. M. Aliouat-Denis. 2008. Purification of infectious trophic and cystic forms of Pneumocystis carinii using high speed cell sorter, abstr. PL55. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 87.Martin-Garrido, I., C. De La Horra, N. Respaldiza, V. Friaza, R. Morilla, L. Rivero, S. Gutierrez, R. Garcia-Contreras, F. J. Medrano, J. M. Varela, E. J. Calderón, and M. A. Montes-Cano. 2008. Cigarette smoking is a major risk factor for Pneumocystis jirovecii colonization in chronic obstructive pulmonary disease, abstr. PO40. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 88.Marty, F. M., S. Koo, J. Bryar, and L. R. Baden. 2007. (1→3)beta-d-glucan assay positivity in patients with Pneumocystis (carinii) jirovecii pneumonia. Ann. Intern. Med. 14770-72. [DOI] [PubMed] [Google Scholar]
- 89.Maury, E., P. Lacube, J. L. Baudel, L. Chalumeau-Lemoine, T. Qureshi, S. Chapelain, G. Offenstadt, and P. Roux. 2008. Are there Pneumocystis jirovecii carriers among the medical staff in an intensive care unit? Preliminary results of a longitudinal prospective study, abstr. PO18. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 90.McKinley, L., A. J. Logar, F. McAllister, M. Zheng, C. Steele, and J. K. Kolls. 2006. Regulatory T cells dampen pulmonary inflammation and lung injury in an animal model of Pneumocystis pneumonia. J. Immunol. 1776215-6226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.McNeill, J., F. R. Barrie, H. M. Burdet, V. Demoulin, D. L. Hawksworth, K. Marhold, D. H. Nicolson, J. Prado, P. C. Silva, J. E. Skog, J. H. Wiersema, and N. J. Turland. 2006. International Code of Botanical Nomenclature (Vienna Code). Regnum Vegetabile 146, article 45.4, http://ibot.sav.sk/icbn/main.htm.
- 92.Medrano, F. J., R. Morilla, J. R. Garcia-Fabero, N. Respaldiza, S. Pinazo, V. Friaza, J. Cabrera, C. De La Horra, R. Yang, L. Rivero, S. Gutierrez, I. Martin-Garrido, M. A. Montes-Cano, J. M. Varela, and E. Calderon. 2008. High prevalence of Pneumocystis jirovecii colonization in the general population of southern Spain, abstr. PO13. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 93.Meissner, N., M. Rutkowski, A. L. Harmsen, S. Han, and A. G. Harmsen. 2007. Type I interferon signaling and B cells maintain hemopoiesis during Pneumocystis infection of the lung. J. Immunol. 1786604-6615. [DOI] [PubMed] [Google Scholar]
- 94.Meissner, N., S. Han, Y. Ran, and A. Harmsen. 2008. Bone marrow failure and immune response to Pneumocystis pneumonia (PcP): a mouse model to examine bone marrow failure in AIDS?, abstr. PL29. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 95.Milla, P., F. Viola, B. S. Oliaro, F. Rocco, L. Cattel, B. M. Joubert, R. J. LeClair, S. P. Matsuda, and G. Balliano. 2002. Subcellular localization of oxidosqualene cyclases from Arabidopsis thaliana, Trypanosoma cruzi, and Pneumocystis carinii expressed in yeast. Lipids 371171-1176. [DOI] [PubMed] [Google Scholar]
- 96.Moncada, C. A., A. B. Clarkson, O. Perez-Leal, and S. Merali. 2008. Nicotine, polyamine cycling and resistance to PcP, abstr. PL33. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 97.Montes-Cano, M. A., M. Chabe, M. Fontillon-Alberdi, C. De La Horra, N. Respaldiza, V. Friaza, R. Morilla, N. Gantois, F. J. Medrano, J. M. Varela, J. Fernández-Alonso, E. Dei-Cas, and E. J. Calderón. 2008. Molecular evidence of Pneumocystis jirovecii transplacental transmission in humans, abstr. PO41. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 98.Moore, R. T. 1980. Taxonomic proposals for the classification of marine yeasts and other yeast-like fungi including the smuts. Bot. Mar. 23361-373. [Google Scholar]
- 99.Morilla, R., L. Rivero, V. Friaza, S. Gutierrez, N. Respaldiza, J. Martin-Juan, M. A. Montes-Cano, I. Martin-Garrido, F. J. Medrano, J. M. Varela, E. J. Calderón, and C. De La Horra. 2008. Pneumocystis jirovecii colonization induces systemic inflammatory response in chronic obstructive pulmonary disease patients, abstr. PO15. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 100.Morris, A. 2008. Is there anything new in Pneumocystis jirovecii pneumonia? Changes in P. jirovecii pneumonia over the course of the AIDS epidemic. Clin. Infect. Dis. 46634-636. [DOI] [PubMed] [Google Scholar]
- 101.Morris, A., F. C. Sciurba, I. P. Lebedeva, A. Githaiga, W. M. Elliott, J. C. Hogg, L. Huang, and K. A. Norris. 2004. Association of chronic obstructive pulmonary disease severity and Pneumocystis colonization. Am. J. Respir. Crit. Care Med. 170408-413. [DOI] [PubMed] [Google Scholar]
- 102.Morris, A., F. C. Sciurba, and K. A. Norris. 2008. Pneumocystis: a novel pathogen in chronic obstructive pulmonary disease? COPD 543-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Nevez, G., S. Le Gal, G. Hery-Arnaud, S. Ramel, M. Virmaux, C. Da Costa, and A. Totet. 2008. Pneumocystis jirovecii in cystic fibrosis patients from Brittany, France, abstr. PO10. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 104.Nkinin, S. W., E. Ndzi, T. Asonganyi, K. Daly, J. Koch, P. D. Walzer, and E. S. Kaneshiro. Detection of Pneumocystis jirovecii antibodies in sera from Cameroonians by ELISA using recombinant MSG fragments, abstr. PO3. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 105.Norris, K. A., A. Morris, S. Patil, and E. Fernandes. 2006. Pneumocystis colonization, airway inflammation, and pulmonary function decline in acquired immunodeficiency syndrome. Immunol. Res. 36175-187. [DOI] [PubMed] [Google Scholar]
- 106.Nosek, J., L. Tomaska, H. Fukuhara, Y. Suyama, and L. Kovac. 1998. Linear mitochondrial genomes: 30 years down the line. Trends Genet. 14184-188. [DOI] [PubMed] [Google Scholar]
- 107.Odabasi, Z., V. Paetznick, J. H. Rex, and L. Ostrosky-Zeichner. 2007. Effects of serum on in vitro susceptibility testing of echinocandins. Antimicrob. Agents Chemother. 514214-4216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Olson, E. J., J. E. Standing, N. Griego-Harper, O. A. Hoffman, and A. H. Limper. 1996. Fungal beta-glucan interacts with vitronectin and stimulates tumor necrosis factor alpha release from macrophages. Infect. Immun. 643548-3554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Opata, M., M. Hollifield, and B. Garvy. 2008. Adoptive transfer of B-lymphocytes facilitates efficient clearance of Pneumocystis infection in SCID mice, abstr. PO20. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 110.Pavlica, F. 1962. The first observation of congenital pneumocystic pneumonia in a fully developed stillborn child. Ann. Paediatr. 198184. [PubMed] [Google Scholar]
- 111.Rapaka, R. R., E. S. Goetzman, M. Zheng, J. Vockley, L. McKinley, J. K. Kolls, and C. Steele. 2007. Enhanced defense against Pneumocystis carinii mediated by a novel dectin-1 receptor Fc fusion protein. J. Immunol. 1783702-3712. [DOI] [PubMed] [Google Scholar]
- 112.Rapaka, R. R., M. Zheng, and J. K. Kolls. 2008. Pneumocystis conserved fungal cell wall carbohydrates are targeted by natural antibodies and direct host adaptive antibody responses, abstr. PL30. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 113.Redhead, S. A., M. T. Cushion, J. K. Frenkel, and J. R. Stringer. 2006. Pneumocystis and Trypanosoma cruzi: nomenclature and typifications. J. Eukaryot. Microbiol. 532-11. [DOI] [PubMed] [Google Scholar]
- 114.Respaldiza, N., M. A. Montes-Cano, F. J. Dapena, C. de la Horra, I. Mateos, F. J. Medrano, E. Calderon, and J. M. Varela. 2005. Prevalence of colonisation and genotypic characterisation of Pneumocystis jirovecii among cystic fibrosis patients in Spain. Clin. Microbiol. Infect. 111012-1015. [DOI] [PubMed] [Google Scholar]
- 115.Ripamonti, C., A. Orenstein, G. Kutty, L. Huang, C. Vinton, P. Conville, and J. Kovacs. 2008. RFLP analysis demonstrates substantial variation in the major surface glycoprotein gene repertoire among Pneumocystis jirovecii isolates, abstr. PL44. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 116.Roths, J. B., and C. L. Sidman. 1993. Single and combined humoral and cell-mediated immunotherapy of Pneumocystis carinii pneumonia in immunodeficient SCID mice. Infect. Immun. 611641-1649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Roux, P., A. Angoulvant, M. G. Lebrette, P. M. Girard, E. Maury, and D. Magne. 2008. Real time quantitative PCR for noninvasive pneumocystosis diagnosis and followup, abstr. PO44. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 118.Schmatz, D. M., M. A. Powles, D. McFadden, K. Nollstadt, F. A. Bouffard, J. F. Dropinski, P. Liberator, and J. Andersen. 1995. New semisynthetic pneumocandins with improved efficacies against Pneumocystis carinii in the rat. Antimicrob. Agents Chemother. 391320-1323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Sesterhenn, T. M., B. E. Slaven, S. P. Keely, B. F. Lang, A. G. Smulian, and M. T. Cushion. 2008. Sequence of the mitochondrial genome of Pneumocystis carinii: gene mapping and evidence of linearity, abstr. PL40. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 120.Sesterhenn, T. M., J. C. Peterson, S. M. Kralovic, F. J. Gomez, P. D. Walzer, D. Moore, A. G. Smulian, and M. T. Cushion. 2008. Pneumocystis jirovecii colonization in human subpopulations: validation of oral swabs for detection and identification of potential risk factors, abstr. PO39. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 121.Shao, S., C. Zhang, M. E. Lasbury, C. Liao, P. J. Durant, and C. Lee. 2008. Defective phagocytosis of alveolar macrophages from Pneumocystis-infected host correlates with down-regulation of phagocytic receptors, abstr. PO46. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 122.Shipley, T. W., H. M. Kling, S. P. Patil, A. Morris, P. Thompson, R. Rogers, F. Sciurba, and K. A. Norris. 2008. Pneumocystis colonization contributes to the development of COPD in a nonhuman primate model of AIDS, abstr. PL47. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 123.Shivji, M., S. Burger, C. A. Moncada, A. B. Clarkson, Jr., and S. Merali. 2005. Effect of nicotine on lung S-adenosylmethionine and development of Pneumocystis pneumonia. J. Biol. Chem. 28015219-15228. [DOI] [PubMed] [Google Scholar]
- 124.Stein, C. R., C. Poole, P. Kazanjian, and S. R. Meshnick. 2004. Sulfa use, dihydropteroate synthase mutations, and Pneumocystis jirovecii pneumonia. Emerg. Infect. Dis. 101760-1765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Stringer, J. R. 2007. Antigenic variation in Pneumocystis. J. Eukaryot. Microbiol. 548-13. [DOI] [PubMed] [Google Scholar]
- 126.Stringer, S. L., J. R. Stringer, M. A. Blase, P. D. Walzer, and M. T. Cushion. 1989. Pneumocystis carinii: sequence from ribosomal RNA implies a close relationship with fungi. Exp. Parasitol. 68450-461. [DOI] [PubMed] [Google Scholar]
- 127.Swain, S. D., S. Han, A. Harmsen, K. Shampeny, and A. G. Harmsen. 2007. Pulmonary hypertension can be a sequela of prior Pneumocystis pneumonia. Am. J. Pathol. 171790-799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Swain, S. D., D. Pauli, and A. G. Harmsen. 2008. Pulmonary hypertension as a sequela of Pneumocystis pneumonia: possible role of macrophage-fibroblast interactions, abstr. PO21. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 129.Tang, X., M. E. Lasbury, D. D. Davidson, M. S. Bartlett, J. W. Smith, and C. H. Lee. 2000. Down-regulation of GATA-2 transcription during Pneumocystis carinii infection. Infect. Immun. 684720-4724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129a.The Society of Protozoologists and the National Institute of Allergy and Infectious Diseases. 1988. Workshop on Pneumocystis carinii, Bristol, England, United Kingdom, 19 to 22 July 1988. J. Protozool. 361S-80S. [PubMed] [Google Scholar]
- 130.Tipirneni, R., K. R. Daly, L. G. Jarlsberg, J. V. Koch, A. Swartzman, B. Roth, P. Walzer, and L. Huang. 2008. Health care worker occupation is associated with immune response to Pneumocystis, abstr. PL5. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 131.Totet, A., N. Brahimi, C. Raccurt, and G. Nevez. Pneumocystis jirovecii 5.8S rRNA gene polymorphism in infants with primary Pneumocystis infection, abstr. PO35. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 132.Vaillant, L., P. M. Hauser, P. Vanhems, and M. Rabodonirina. 2008. Outcome of Pneumocystis jirovecii pneumonia: impact of P. jirovecii genotype and DHPS mutations, abstr. PO7. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 133.Vassallo, R., J. Standing, and A. H. Limper. 1999. Beta-glucan from Pneumocystis carinii stimulates TNF alpha release from alveolar macrophages. J. Eukaryot. Microbiol. 46145S. [PubMed] [Google Scholar]
- 134.Villegas, L. R., T. J. Kottom, and A. H. Limper. 2008. Characterization of a Pneumocystis carinii endo-β-1,3-glucanase, abstr. PL8. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 135.Wakefield, A. E., J. M. Hopkin, J. Burns, J. B. Hipkiss, T. J. Stewart, and E. R. Moxon. 1989. Cloning of DNA from Pneumocystis carinii. J. Protozool. 365S-7S. [DOI] [PubMed] [Google Scholar]
- 136.Wang, J., F. Gigliotti, S. P. Bhagwat, and T. W. Wright. 2008. Sulfasalazine increases the turnover of alveolar macrophages during Pneumocystis pneumonia, abstr. PL49. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 137.Wang, S., C. Zhang, S. Shao, M. E. Lasbury, C. Liao, P. J. Durant, and C. Lee. 2008. PU.1 down-regulation in alveolar macrophages during Pneumocystis pneumonia, abstr. PL51. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 138.Wang, S., C. Zhang, M. E. Lasbury, C. Liao, P. J. Durant, and C. Lee. PU.1 regulates dectin-1 gene expression in alveolar macrophages, abstr. PO49. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 139.Wells, J., C. G. Haidaris, T. W. Wright, and F. Gigliotti. 2006. Active immunization against Pneumocystis carinii with a recombinant P. carinii antigen. Infect. Immun. 742446-2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Wilkinson, M. L., J. E. Malone, C. G. Haidaris, T. W. Wright, and F. Gigliotti. 2008. Cloning and characterization of a novel Pneumocystis antigen, abstr. PO9. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 141.Wissmann, G., J. C. Prolla, L. Z. Goldani, I. Martin-Garrido, V. Friaza, R. Morilla, C. De La Horra, M. Montes-Cano, N. Respaldiza, F. J. Medrano, J. M. Varela, and E. Calderón. 2008. Colonization by Pneumocystis jirovecii in Brazilian children patients with hematologic malignancies, abstr. PO42. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 142.Wissmann, G., J. C. Prolla, L. Z. Goldani, I. Martin-Garrido, V. Friaza, R. Morilla, C. De La Horra, M. Montes-Cano, N. Respaldiza, F. J. Medrano, J. M. Varela, and E. Calderón. 2008. Colonization by Pneumocystis jirovecii in Brazilian cystic fibrosis patients, abstr. PO43. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 143.Zhang, C., S. Shao, M. E. Lasbury, C. Liao, P. J. Durant, and C. Lee. 2008. GATA-2 down-regulation leads to decreased expression of phagocytic receptors in alveolar macrophages during Pneumocystis pneumonia, abstr. PL46. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.
- 144.Zheng, M., J. E. Shellito, L. Marrero, Q. Zhong, S. Julian, P. Ye, V. Wallace, P. Schwarzenberger, and J. K. Kolls. 2001. CD4+ T cell-independent vaccination against Pneumocystis carinii in mice. J. Clin. Investig. 1081469-1474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Zheng, M., J. Bindas, R. Rapaka, and J. K. Kolls. 2008. Antibody responses in homologous kexin DNA vaccination against Pneumocystis murina, abstr. PO19. 10th Int. Workshops Opportunistic Protists, Boston, MA, 28 to 31 May 2008.