Abstract
Background
Some previous prospective studies showed that dietary intake of ω-3 polyunsaturated fatty acids was associated with lower risk of heart failure (HF), but no study has examined the association between plasma fatty acids and HF.
Methods
We included 3,592 white participants from the Minneapolis field center of the Atherosclerosis Risk in Communities (ARIC) Study, aged 45-64 at baseline (1987-89), initially free of coronary heart disease, stroke and HF, and who had cholesterol ester and phospholipid plasma fatty acids measured. Participants were followed through 2003, and incident HF was defined by a hospital discharge or death including a HF ICD code.
Results
During the 14.3-year follow-up, we identified 197 cases of HF (110 for men and 87 for women). After adjustment for age and other confounders, higher saturated fatty acids, especially myristic (14:0) acid, were associated positively with incident HF in both men and women. Higher arachidonic (20:3,ω-6) and long-chain ω-3 polyunsaturated fatty acids, especially docosahexaenoic (22:6,ω-3), were associated inversely with HF in women, but not in men. Neither plasma α-linolenic nor eicosapentaenoic acid was associated with incident HF.
Conclusions
In both men and women, greater levels of saturated fatty acids may increase risk of HF. In women, arachidonic acid and long-chain ω-3 polyunsaturated fatty acids may decrease risk of HF. (211 words/ 250 limits)
Keywords: longitudinal study, epidemiology, plasma fatty acids, docosahexaenoic acid, eicosapentaenoic acid, cardiovascular disease
Introduction
Since the Seven Countries Study (1), it has been broadly believed that dietary intakes of fatty acids have a substantial impact on coronary heart disease (CHD); ie, saturated fatty acids (SFA) increase and polyunsaturated fatty acids (PUFA) decrease serum cholesterol levels and risk of CHD (2). Based on this hypothesis, several epidemiological studies have examined associations between several fatty acids and CHD stroke, arrhythmia and coronary disease risk factors (3-5), but evidence on the association between fatty acid composition and heart failure (HF) is limited. The Cardiovascular Health Study (CHS) reported that dietary intakes of fish and/or ω-3 PUFA estimated by food frequency questionnaire (FFQ) were associated with reduced risk of incident HF (6). A recent report from the Atherosclerosis Risk in Communities (ARIC) Study (7) showed that intake of high-fat dairy by FFQ was positively, and intake of whole grains was inversely, associated with incident HF. Similarly, whole grain cereal consumption by FFQ was inversely associated with HF risk in the Physicians’ Health Study (8). Greater egg consumption has also been reported to be associated with increased risk of incident HF (7,9). However, to date, few studies have examined associations between individual fatty acids and HF. Furthermore, data are quite limited on plasma fatty acids, biomarkers of fatty acid intake, in relation to HF.
In this study, we examined the association between plasma fatty acid composition and incident HF in participants recruited from one ARIC field center. In the Minneapolis ARIC field center, fractions of cholesterol ester (CE) and phospholipid (PL) plasma fatty acids were measured. Because CE and PL are found in cell membrane, each is considered to be a good biomarker of dietary intake over recent weeks (10). We have previously reported their associations with carotid artery intima-media thickness (11), hypertension (12), diabetes (13), CHD (14) and cognitive decline (15). Our a priori hypothesis was that a higher proportion of plasma SFA, and a lower proportion of long-chain ω-3 PUFA, would be associated with higher risk of HF among white men and women.
Methods
Study cohort
The ARIC Study comprised a community-based sample of 15,792 persons from 4 research field centers: Forsyth County, North Carolina; the city of Jackson, Mississippi; north-western suburbs of Minneapolis, Minnesota, and Washington County, Maryland. Participants were 45-64 years of age during the baseline period (1987-1989). The ARIC study protocol was approved by each field center’s institutional review board. After written informed consent was obtained, participants underwent a baseline clinical examination. The details of the ARIC study protocol were described elsewhere (16). In the Minneapolis field center only (n=4,009), plasma was saved at baseline and analyzed for fatty acids. We excluded persons who reported a history of CHD, stroke or HF at the baseline survey, or those without plasma fatty acid data. Prevalent HF was defined as reported current medication use for HF, and/or having manifest HF as defined by Gothenburg criteria (17) stage 3, which requires the presence of specific cardiac and pulmonary symptoms as well as medical treatment for HF. We also excluded non-white subjects due to their small number. Our final sample included 1,667 men and 1,908 women.
Plasma fatty acids measurement
Twelve-hour fasting blood was collected into 10-ml vacuum tubes containing EDTA. Plasma was separated and dispensed into two 1.5 ml aliquots and frozen at −70°C for approximately 2 years before analysis for fatty acid content by a single technician.
Details of plasma fatty acid measurement have been described elsewhere (18). After separation of PL and CE fractions, the identity of 28 fatty acid peaks was ascertained by comparison of the peak’s retention time shown by the gas-liquid chromatogram relative to the retention times of fatty acids in synthetic standards of known fatty acid composition. The percentage of each fatty acid among total fatty acids (% total fatty acids) was quantified by the total area for all fatty acids. Absolute values of each fatty acid were not estimated. The data were electrically transferred from the gas chromatogram to a computer. Short-term test-retest reliability coefficients have been reported previously (19); the values for SFA were r=0.70 for CE, 0.57 for PL; for monounsaturated fatty acids (MUFA) were r=0.71 for CE, 0.46 for PL; and for PUFA were r=0.71 for CE, 0.32 for PL. That report also provided long-term reliabilities (19), which were somewhat lower than short-term reliabilities. The correlations between plasma and dietary fatty acids measured by FFQ also have been reported (18) and were: SFA (r=0.23 for CE, 0.15 for PL), PUFA (r=0.31 for CE, 0.25 for PL), and MUFA (r=0.01 for CE, 0.05 for PL).
We examined several individual fatty acids: myristic (14:0), pentadecanoic (15:0), palmitic (16:0), margaric (17:0), stearic (18:0), palmitoleic (16:1,ω7), oleic (18:1,ω9), linoleic (18:2,ω6), γ-linoleic (18:3,ω6), dihomo-γ-linoleic (20:3,ω6), arachidonic (20:4,ω6), α-linoleic (18:3,ω3), eicosapentaenoic (20:5,ω3) and docosahexaenoic (22:6,ω3) acids. For grouped fatty acids, we defined SFA as the sum of (12:0), (14:0), (15:0), (16:0), (17:0), (18:0), (20:0), (22:0), (23:0) and (24:0); MUFA as the sum of (14:1 ,ω5), (16:1,ω7), (18:1,ω9), (20:1,ω9), (22:1,ω9) and (24:1,ω9); ω-6 PUFA as the sum of (18:2,ω6), (18:3,ω6), (20:2,ω6), (20:3,ω6), (20:4,ω6), (22:4,ω6) and (22:5,ω6); ω-3 PUFA as the sum of (18:3,ω3), (20:3,ω3), (20:5,ω3), (22:5,ω3) and (22:6,ω3); and long-chain ω-3 PUFA as the sum of (20:5,ω3), (22:5,ω3) and (22:6,ω3).
Ascertainment of incident HF events
Cases of HF were identified through annual telephone calls to participants to ascertain HF hospitalizations, review of local hospital discharge lists, and via death certificates. Incident HF was defined by the first HF hospitalization (ICD-9 code 428 in any position), or any deaths where the death certificate included a HF code (code 428, ICD-9 or I50, ICD-10, in any position). Non-hospitalized, non-fatal HF was not captured. All cohort hospitalizations and deaths that occurred before January 1, 2004, were included. More details of the methods for ascertainment of incident HF in ARIC were given elsewhere (20).
Statistical Analysis
For each participant, we calculated the person-years of follow-up from baseline in 1987-1989 to the first HF endpoint, death, loss to follow-up, or the end of 2003. Hazard ratios (HRs) with 95% confidence intervals (CIs) across the quintiles of each fatty acid, measured as the percentage of total fatty acids, were calculated after adjustment for age, sex and the following baseline cardiovascular disease (CVD) risk factors with Cox proportional hazard models: body mass index, systolic blood pressure, antihypertensive medication use (yes or no), plasma total and HDL-cholesterols, prevalent diabetes (yes or no), smoking status (never, ex- or current smoker), pack-years, alcohol intake, sports index (<2.0, 2.0-2.5, 2.5-3.0 or ≥3.0) and education level (>high school graduate or ≤high school graduate). The linear trend of HRs across the quintiles was tested by using a variable with −2, −1, 0, 1, 2 assigned to successive quintiles. Interactions between sex and each fatty acid in relation to incident HF were tested using cross-product terms. If an interaction was statistically significant for either the CE or PL fraction, sex-specific analyses were performed for both fractions. The proportional hazards assumption was tested using time by fatty acid interaction terms and was not violated except for arachidonic acid in men.
In secondary analyses, we defined desaturase and elongase activity indices (21,22) to examine the impact of each emzyme activity on incident HF: the ratios of (16:1,ω7)/(16:0) as Δ9-desaturase index; (18:3,ω6)/(18:2,ω6) as Δ-6 desaturase index for CE fraction; (20:3,ω6)/(18:2,ω6) as Δ-6 desaturase index for PL fraction; (20:4,ω6)/(20:3,ω6) as Δ-5 desaturase index; and (18:0)/(16:0) as elongase index. Quintiles of each index were used in the analyses.
We used SAS version 9.1.3 Service Pack 4 (SAS Institute Inc., Cary, NC) for the analyses. All probability values for statistical tests were two-tailed and values of p<0.05 were regarded as statistically significant.
Results
During 51,960 person-years of follow-up of 3,575 persons (1,667 men and 1,908 women), we documented 195 HF cases (109 men and 86 women).
Mean age at baseline was 54.2 in men and 53.3 in women (Table 1). Body mass index, blood pressure, triglyceride, diabetes, sports index, education level, cigarette amount, alcohol, and energy intake were higher, and plasma total and HDL-cholesterol levels were lower, in men than in women. Since mean fatty acid composition did not differ much by sex, means and standard deviations of fatty acids were calculated for the sex-combined sample (Table 2).
Table 1.
Sex-specific unadjusted baseline characteristics, Minneapolis Field Center, ARIC Study, 1987-89.
| Men |
Women |
|||||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||
| Number | 1,667 | 1,908 | ||||
| Age | 54.2 | ± | 5.6 | 53.3 | ± | 5.5 |
| Body mass index (kg/m2) | 27.7 | ± | 3.7 | 26.2 | ± | 5.0 |
| Systolic blood pressure (mmHg) | 120.5 | ± | 14.8 | 116.9 | ± | 17.0 |
| Diastolic blood pressure (mmHg) | 75.5 | ± | 9.2 | 72.1 | ± | 9.1 |
| Anti-hypertensive medication (%) | 17 | 20 | ||||
| Plasma total cholesterol (mg/dl) | 212 | ± | 39 | 216 | ± | 42 |
| Plasma HDL-cholesterol (mg/dl) | 44 | ± | 12 | 60 | ± | 17 |
| Plasma triglyceride (mg/dl) | 139 | ± | 94 | 116 | ± | 73 |
| Diabetes (%) | 8 | 6 | ||||
| Sports index (range 1-5) | 2.7 | ± | 0.8 | 2.5 | ± | 0.8 |
| Education (>high school graduate, %) |
69 | 51 | ||||
| Current smoking (%) | 21 | 23 | ||||
| Cigarette-years | 405 | ± | 440 | 234 | ± | 334 |
| Alcohol intake (g/wk) | 88.0 | ± | 128.3 | 33.4 | ± | 56.0 |
| Total energy intake (Kcal/d) | 1818 | ± | 622 | 1492 | ± | 499 |
| Fish-oil supplement use (%) | 3 | 3 | ||||
| Hormone therapy use (%) | - | 22 | ||||
Table 2.
Unadjusted baseline fatty acid composition, Minneapolis Field Center, ARIC Study, 1987-89.
| Men and Women | ||||
|---|---|---|---|---|
| CE fraction |
PL fraction |
|||
| Mean, % | SD | Mean, % | SD | |
| Saturated fatty acids | 11.64 | 0.97 | 40.63 | 1.39 |
| Myristic (14:0) | 0.56 | 0.18 | 0.26 | 0.08 |
| Pentadecanoic (15:0) | 0.15 | 0.03 | 0.17 | 0.04 |
| Palmitic (16:0) | 10.02 | 0.81 | 25.41 | 1.64 |
| Margaric (17:0) | 6.47 | 1.87 | 8.92 | 2.80 |
| Stearic (18:0) | 0.89 | 0.21 | 13.27 | 1.19 |
| Monounsaturated fatty acids | 18.60 | 3.02 | 9.96 | 1.27 |
| Palmitoleic (16:1,ω7) | 2.56 | 1.24 | 0.64 | 0.18 |
| Oleic (18:1,ω9) | 15.95 | 2.07 | 8.59 | 1.17 |
| ω-6 Polyunsaturated fatty acids | 64.24 | 4.01 | 38.19 | 1.84 |
| Linoleic (18:2,ω6) | 54.17 | 4.84 | 21.99 | 2.69 |
| γ-Linolenic (18:3,ω6) | 1.02 | 0.37 | 0.11 | 0.06 |
| Dihomo γ-linolenic (20:3,ω6) | 0.76 | 0.17 | 3.33 | 0.77 |
| Arachidonic (20:4,ω6) | 8.23 | 1.69 | 11.45 | 1.96 |
| ω-3 Polyunsaturated fatty acids | 1.41 | 0.42 | 4.43 | 1.11 |
| α-linolenic (18:3,ω3) | 0.41 | 0.11 | 0.14 | 0.05 |
| Long chain ω-3 polyunsaturated fatty acids | 1.00 | 0.39 | 4.28 | 1.11 |
| Eicosapentaenoic (20:5,ω3) | 0.55 | 0.28 | 0.56 | 0.30 |
| Docosahexaenoic (22:6,ω3) | 0.44 | 0.16 | 2.81 | 0.89 |
Table 3 presents age-adjusted hazard ratios of HF for the highest vs lowest quintiles of each fatty acid. Greater plasma SFAs, in particular palmitic acid, were generally associated with higher risk of HF. An increased risk was also observed for MUFA, particularly for palmitoleic acid. Higher plasma levels of ω-6 PUFA were associated with reduced risks of HF. Of these, linoleic acid was associated inversely, and dihomo γ-linolenic acid was associated positively, with risk of HF. Arachidonic acid was associated approximately 60% reduced risk of HF in the highest vs lowest quintiles, but only among women (p for sex-interactions=0.004 for CE and 0.003 for PL). Reduced risks of HF were also observed for greater long chain ω-3 PUFA for women (p for sex-interaction=0.05 for CE and 0.002 for PL), but not for men. A strong inverse association was observed for docosahexaenoic acid for women. No such association was observed for α-linolenic or eicosapentaenoic acid. We also tested associations of Δ9, Δ6, Δ5-desaturase and elongase indices with risk of HF. Delta-9 and Δ6-desaturase indices were associated positively with HF incidence. On the other hand, Δ5-desaturase index was associated inversely with risk of HF, primarily among women. The inverse association among men was less clear and did not reach statistical significance (p for sex-interaction=0.02 for both CE and PL). Elongase index was associated inversely with HF for PL, but not for CE.
Table 3.
Age and sex-adjusted hazard ratios and 95% confidence intervals of incident heart failure for the highest and lowest quintiles of each fatty acid, ARIC, 1987-2003.
| Heart Failure |
||||
|---|---|---|---|---|
| CE fraction |
PL fraction |
|||
| HR (95%CI) | p for trend |
HR (95%CI) | p for trend |
|
| Saturated fatty acids* | 3.68(2.11-6.40) | <0.001 | 2.71(1.64-4.45) | <0.001 |
| Myristic (14:0)* | 1.70(1.06-2.71) | 0.005 | 1.29(0.80-2.08) | 0.42 |
| Pentadecanoic (15:0)* | 0.80(0.50-1.28) | 0.39 | 0.62(0.38-1.02) | 0.04 |
| Palmitic (16:0)* | 4.02(2.24-7.21) | <0.001 | 2.16(1.36-3.43) | <0.001 |
| Margaric (17:0)* | 0.81(0.52-1.26) | 0.38 | 0.55(0.35-0.85) | 0.008 |
| Stearic (18:0)* | 1.63(1.01-2.62) | 0.05 | 1.01(0.64-1.58) | 0.90 |
| Monounsaturated fatty acids* | 2.37(1.47-3.82) | 0.001 | 1.36(0.88-2.11) | 0.32 |
| Palmitoleic (16:1,ω7)* | 2.26(1.39-3.68) | <0.001 | 1.67(1.10-2.52) | 0.01 |
| Oleic (18:1,ω9)* | 1.80(1.13-2.85) | 0.004 | 1.38(0.90-2.11) | 0.17 |
| ω-6 Polyunsaturated fatty acids* | 0.34(0.20-0.57) | <0.001 | 0.69(0.43-1.09) | 0.16 |
| Linoleic (18:2,ω6)* | 0.54(0.34-0.88) | 0.001 | 0.57(0.36-0.92) | 0.009 |
| γ-Linolenic (18:3,ω6)* | 1.48(0.92-2.37) | 0.09 | 1.39(0.87-2.23) | 0.51 |
| Dihomo γ-linolenic (20:3,ω6)* | 2.44(1.48-4.00) | <0.001 | 2.26(1.38-3.70) | <0.001 |
| Arachidonic (20:4,ω6) | ||||
| Men | 1.37(0.77-2.42) | 0.28 | 1.34(0.79-2.27) | 0.21 |
| Women | 0.43(0.19-0.95) | 0.02 | 0.38(0.16-0.91) | 0.004 |
| ω-3 Polyunsaturated fatty acids | ||||
| α-linolenic (18:3,ω3)* | 0.99(0.63-1.53) | 0.81 | 0.97(0.61-1.54) | 0.88 |
| Long chain ω-3 polyunsaturated fatty acids | ||||
| Men | 1.49(0.84-2.63) | 0.40 | 0.99(0.55-1.77) | 0.43 |
| Women | 0.42(0.19-0.92) | 0.09 | 0.24(0.11-0.54) | <0.001 |
| Eicosapentaenoic (20:5,ω3)* | 1.37(0.85-2.20) | 0.26 | 1.61(0.98-2.64) | 0.06 |
| Docosahexaenoic (22:6,ω3) | ||||
| Men | 1.30(0.73-2.32) | 0.47 | 1.17(0.66-2.07) | 0.51 |
| Women | 0.21(0.10-0.44) | <0.001 | 0.16(0.07-0.40) | <0.001 |
| Δ9-desaturase index (16:1,ω7/16:0)* | 1.68(1.08-2.63) | 0.004 | 1.68(1.08-2.61) | 0.07 |
| Δ6-desaturase index*† | 1.55(0.96-2.52) | 0.02 | 2.31(1.42-3.77) | <0.001 |
| Δ5-desaturase index (20:4,ω6/20:3,ω6) | ||||
| Men | 0.86(0.47-1.57) | 0.24 | 0.67(0.36-1.26) | 0.11 |
| Women | 0.21(0.09-0.49) | <0.001 | 0.27(0.12-0.59) | <0.001 |
| Elongase index (18:0/16:0)* | 1.24(0.79-1.94) | 0.99 | 0.54(0.34-0.84) | 0.02 |
Men and women were combined because of non-significant sex-interactions for both CE and PL.
Δ6-desaturase indices were defined as 18:3,ω6/18:2,ω6 for CE and as 20:3,ω6/18:2,ω6 for PL.
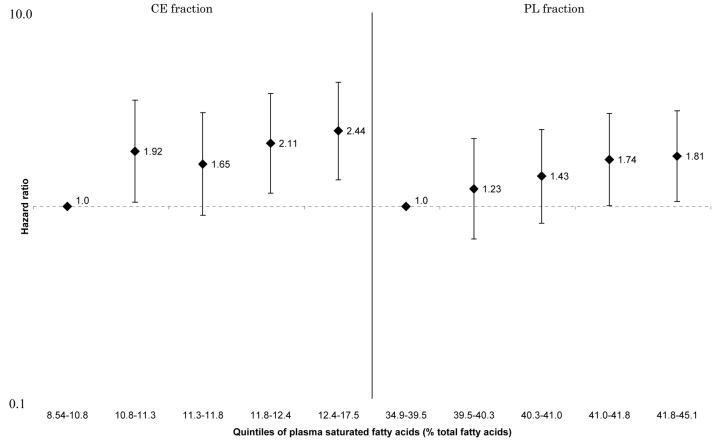
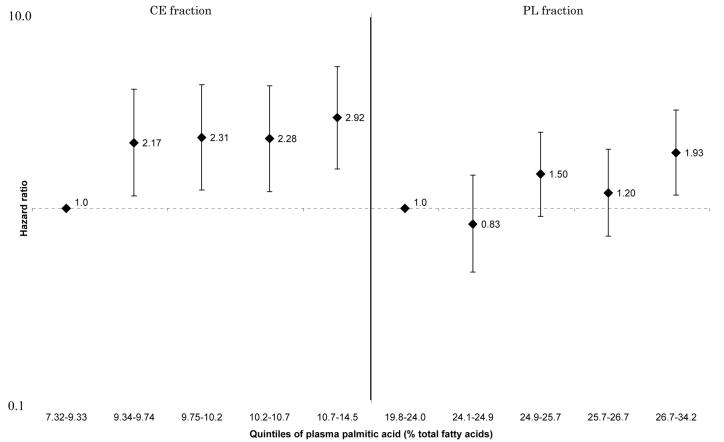
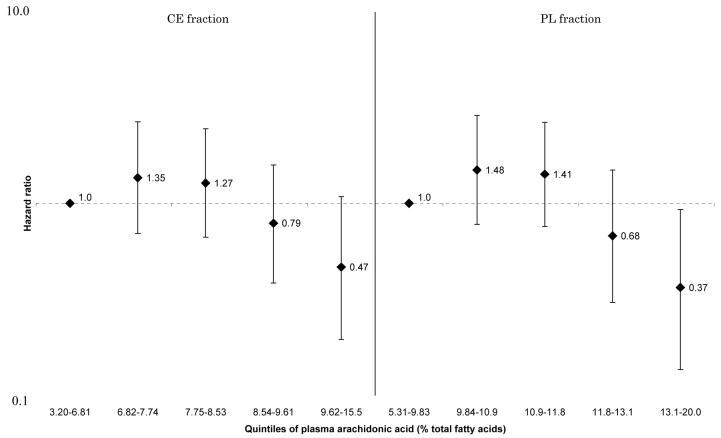
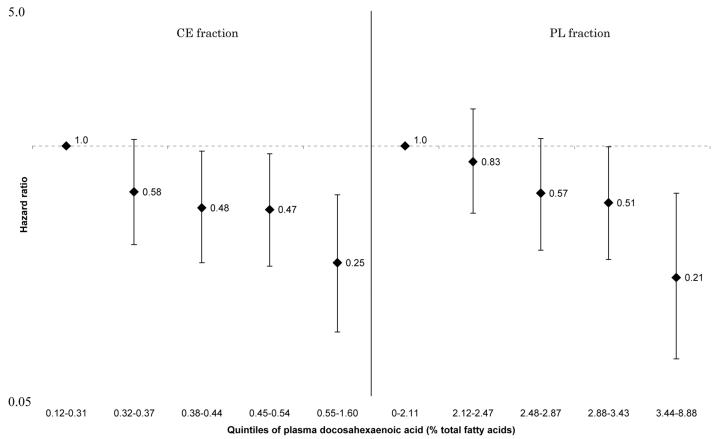
These associations were generally attenuated by further adjustment for selected CVD risk factors. However, the positive linear associations between HF and total SFA and palmitic acid in all participants (Figure 1 and 2), and the inverse linear associations between HF and arachidonic acid (Figure 3) and docosahexaenoic acid (Figure 4) in women remained statistically significant for both CE and PL fractions. No such associations were observed in men (p for sex-interactions<0.01 for CE and PL, arachidonic and docosahexaenoic acids). For enzyme indices, the associations were attenuated and no longer statistically significant except for PL elongase index. There were no interactions between sex and Δ5-desaturase index in terms of incident HF (p for interaction=0.15 for both CE and PL), although the association with HF in women remained, whereas it was attenuated to nonsignificance in men: the HR for the highest vs lowest quintiles=0.39[0.16-0.94], p for trend=0.04 for CE, and HR=0.47[0.20-1.10], p for trend=0.03 for PL, data not shown). Detailed results showing multivariable-adjusted HRs for each fatty acid are provided in the Supplement.
Figure 1.
Multivariate hazard ratios and 95% confidence interval of heart failure according to quintiles of plasma saturated fatty acids (men and women combined). Left: cholesterol ester fraction (p for trend across the quintiles=0.003), right: phospholipid fraction (p for trend across the quintiles=0.01).
Figure 2.
Multivariate hazard ratios and 95% confidence interval of heart failure according to quintiles of plasma palmitic acid (men and women combined). Left: cholesterol ester fraction (p for trend across the quintiles=0.001), right: phospholipid fraction (p for trend across the quintiles=0.004).
Figure 3.
Multivariate hazard ratios and 95% confidence interval of heart failure according to quintiles of plasma arachidonic acid (women only). Left: cholesterol ester fraction (p for trend across the quintiles=0.03), right: phospholipid fraction (p for trend across the quintiles=0.007).
Figure 4.
Multivariate hazard ratios and 95% confidence interval of heart failure according to quintiles of plasma docosahexaenoic acid (women only). Left: cholesterol ester fraction (p for trend across the quintiles=0.001), right: phospholipid fraction (p for trend across the quintiles<0.001).
Since incident CHD may partly mediate the association between plasma fatty acid composition and HF, we repeated the analysis censoring at the event date of the 372 incident CHD events that occurred before developing HF. The associations for SFA, palmitic, arachidonic, and docosahexaenoic acids were not materially altered. Furthermore, exclusion of prevalent diabetes (n=235 excluded) or women using hormone therapy at baseline (n=424 excluded) did not change the results substantially. Very few cases of incident HF occurred among fish oil supplement users (n=4 for men and 0 for women; data not shown).
Discussion
We observed positive associations between incident HF and SFA, especially palmitic acid, among men and women, and inverse associations between incident HF and arachidonic acid and long-chain PUFA, especially docosahexaenoic acid, among women. These associations remained after adjustment for baseline levels of select CVD risk factors. To our knowledge, the present study is the first report of associations between plasma fatty acid composition and incident HF.
Few epidemiological studies have examined associations between HF and intakes of dietary fatty acids measured by FFQ. The CHS (6) showed an inverse association between baked or broiled fish, but not fried fish, and incident HF. Similar results were observed for dietary intake of long-chain ω-3 PUFA. Our observation in women was in line with these prior reports. On the other hand, a previous study involving the whole ARIC sample found that there was no association between fish intake measured by FFQ and incident HF (7). When we analyzed the FFQ using the present sample (Minnesota only), there was still no association between fish intake and incident HF in either men or women. A possible reason for this discrepancy is that a protective effect of fish or long-chain ω-3 PUFA intake on HF may be masked by intake of fried fish; however, information on fried fish intake was not separately available in the ARIC Study. The CHS showed a positive association between fried fish intake and incident HF (6). We believe the use of plasma values of long-chain ω-3 PUFA may reflect the intake of long-chain ω-3 PUFA more objectively than estimated by questionnaire, although plasma fatty acids are not likely to reflect long-term intakes.
The positive association between SFA and HF is consistent with the FFQ results in ARIC, in which there was a positive association between high-fat dairy intake, but not low-fat dairy intake, and HF (7). According to the United States Department of Agriculture (23), whole fat milk contains 0.83g of palmitic acid, whereas low fat milk contains 0.26-0.31g and non-fat milk contains 0.03-0.07g. Investigators from the Nurses’ Health Study recently reported that pentadecanoic acid (15:0) and trans-(16:1,ω7) are useful biomakers of dietary dairy intake (24). In the present study, we did not observe any associations between pentadecanoic acid and incident HF. We unfortunately could not separate plasma cis- and trans-fatty acids, and plasma palmitoleic acid (16:1,ω7) did not show any association with risk of HF.
We also found inverse associations of arachidonic acid and Δ-5 desaturase index (a ratio of arachidonic to dihomo γ-linolenic acids, which is considered to be an estimate of Δ-5 desaturase activity) with HF among women. Several studies (14,25,26), including ARIC (14), have shown an inverse association between plasma arachidonic acid and risk of CHD. Higher Δ-5 desaturase activity has been reported to be associated with higher insulin sensitivity (27-29), and lower incidence of the metabolic syndrome (21) or CHD (30), which may partly explain the inverse association between arachidonic acid and HF observed here.
Several studies have shown SFA to be positively associated, and arachidonic acid and/or long-chain ω-3 PUFA to be inversely associated, with incident CHD using plasma or other tissue (14,25,26,31,32). The results of present study were generally consistent with these CHD studies. However, in the present study, when we censored for incident cases of CHD in addition to excluding prevalent CHD cases (baseline status), our results did not change. This may suggest that any protective effect of long-chain ω-3 PUFA on HF is not mediated solely by prevention of CHD. Several mechanisms, including arrhythmia suppression, lowered blood pressure, lowered systemic vascular resistance, lowered heart rate, or preserved diastolic function, have been suggested for the cardioprotective effects of long-chain ω-3 PUFAs (33,34).
A strength of the present study was the usage of a biomarker, which is a more objective indicator of dietary intake than a questionnaire. On the other hand, plasma fatty acids do not fully reflect their intake from diet, since they are also influenced by preferential incorporation, endogenous synthesis, or lifestyle. In this population, plasma ω-3 PUFA, especially docosahexaenoic acid, correlated well with dietary intake (r=0.42 for CE and 0.42 for PL docosahexaenoic acid), and SFA correlated moderately with diet, whereas plasma MUFA did not (18).
Other limitations warrant discussion. First, the short- and long-term repeatability of some plasma fatty acids was modest, especially those composing low proportions of total fatty acids, including docosahexaenoic acid (r=0.53-0.58 for short-term, 0.46-0.48 for long-term). However, the repeatability of palmitic (r=0.74 for short-term and 0.67-0.72 for long-term) and arachidonic (r =0.86-0.88 for short-term and 0.81-0.83 for long-term) acids were fair (19). Therefore, some fatty acids in the present study may have been misclassified, which could have obscured associations. Second, the ascertainment of HF relied on unvalidated hospital discharge records and death certificates with HF ICD codes, and thus out-patient HF cases were not included in this study. However, since out-patient HF cases are less likely to be severe cases, it resulted in high specificity of the HF definition. Previous evidence suggests most heart failure cases are eventually hospitalized and the validity of heart failure ICD codes is high (35). Third, we do not have any explanation for the sex difference in associations of arachidonic and docosahexaenoic acids with HF. Sex differences in the synthesis of docosahexaenoic acid has been suggested presumably due to differences in sex hormone concentrations that may regulate the activity of the desaturation/elongation pathway (36).
In conclusion, we found a significant positive association between SFA and HF among men and women, and inverse associations of arachidonic acid and long-chain ω-3 PUFA with incident HF among women. These results suggest dietary intakes of SFA may have a harmful effect, and long-chain ω-3 PUFA may have a beneficial effect, on prevention of HF.
Supplementary Material
Acknowledgment
The Atherosclerosis Risk in Communities Study is carried out as a collaborative study supported by National Heart, Lung, and Blood Institute contracts N01-HC-55015, N01-HC-55016, N01-HC-55018, N01-HC-55019, N01-HC-55020, N01-HC-55021, and N01-HC-55022. The authors thank the staff and participants of the ARIC study for their important contributions. The authors also appreciate Linda Lewis for analyzing the plasma fatty acids, and Dr. Lyn M. Steffen for valuable comments. Kazumasa Yamagishi was supported by the Kanae Foundation for the Promotion of Medical Science, Tokyo, Japan.
References
- 1.Keys A, Mienotti A, Karvonen MJ, et al. The diet and 15-year death rate in the Seven Counties Study. Am J Epidemiol. 1986;124:903–915. doi: 10.1093/oxfordjournals.aje.a114480. [DOI] [PubMed] [Google Scholar]
- 2.Erkkilä A, de Mello VDF, Risérus U, Laaksonen DE. Dietary fatty acids and cardiovascular disease: An epidemiological approach. Prog Lipid Res. 2008;47:172–187. doi: 10.1016/j.plipres.2008.01.004. [DOI] [PubMed] [Google Scholar]
- 3.Psota TL, Gebauer SK, Kris-Etherton P. Dietary omega-3 fatty acid intake and cardiovascular risk. Am J Cardiol. 2006;98(4A):3i–18i. doi: 10.1016/j.amjcard.2005.12.022. [DOI] [PubMed] [Google Scholar]
- 4.Leaf A. Omega-3 fatty acids and prevention of arrythmias. Curr Opin Lipidol. 2007;18:31–34. doi: 10.1097/MOL.0b013e328012d61b. [DOI] [PubMed] [Google Scholar]
- 5.Balk EM, Lichtenstein AH, Chung M, et al. Effects of omega-3 fatty acids on serum markers of cardiovascular disease risk: a systematic review. Atherosclerosis. 2006;189:19–30. doi: 10.1016/j.atherosclerosis.2006.02.012. [DOI] [PubMed] [Google Scholar]
- 6.Mozaffarian D, Bryson CL, Lemaitre RN, et al. Fish intake and risk of incident heart failure. J Am Coll Cardiol. 2005;45:2015–2021. doi: 10.1016/j.jacc.2005.03.038. [DOI] [PubMed] [Google Scholar]
- 7.Nettleton JA, Steffen LM, Loehr LR, et al. Food intake and risk of incident heart failure in the Atherosclerosis Risk in Communities (ARIC) Study Am J Epidemiol 2007165S130 (Abstract) [Google Scholar]
- 8.Djoussé L, Gaziano JM. Breakfast cereals and risk of heart failure in the Physicians’ Health Study I. Arch Intern Med. 2007;167:2080–2085. doi: 10.1001/archinte.167.19.2080. [DOI] [PubMed] [Google Scholar]
- 9.Djoussé L, Gaziano JM. Egg consumption and risk of heart failure in the Physicians’ Health Study. Circulation. 2008;117:512–516. doi: 10.1161/CIRCULATIONAHA.107.734210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Riboli E, Rönnholm H, Saracci R. Biological markers of diet. Cancer Surv. 1987;6:685–718. [PubMed] [Google Scholar]
- 11.Ma J, Folsom AR, Lewis L, et al. Relation of plasma phospholipid and cholesterol ester fatty acid composition to carotid artery intima-media thickness: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Clin Nutr. 1997;65:551–559. doi: 10.1093/ajcn/65.2.551. [DOI] [PubMed] [Google Scholar]
- 12.Zheng ZJ, Folsom AR, Ma J, et al. Plasma fatty acid composition and 6-year incidence of hypertension in middle-aged adults: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Epidmiol. 1999;150:492–500. doi: 10.1093/oxfordjournals.aje.a010038. [DOI] [PubMed] [Google Scholar]
- 13.Wang L, Folsom AR, Zheng ZJ, et al. Plasma fatty acid composition and incidence of diabetes in middle-aged adults: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Clin Nutr. 2003;78:91–98. doi: 10.1093/ajcn/78.1.91. [DOI] [PubMed] [Google Scholar]
- 14.Wang L, Folsom AR, Eckfeldt JH. Plasma fatty acid composition and incidence of coronary heart disease in middle aged adults: the Atherosclerosis Risk in Communities (ARIC) Study. Nutr Metab Cardiovasc Dis. 2003;13:256–266. doi: 10.1016/s0939-4753(03)80029-7. [DOI] [PubMed] [Google Scholar]
- 15.Beydoun MA, Kaufman JS, Satia JA, et al. Plasma n-3 fatty acids and the risk of cognitive decline in older adults: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Clin Nutr. 2007;85:1103–1111. doi: 10.1093/ajcn/85.4.1103. [DOI] [PubMed] [Google Scholar]
- 16.The ARIC Investigators The Atherosclerosis Risk in Communities (ARIC) Study: Design and objectives. Am J Epidemiol. 1989;129:687–702. [PubMed] [Google Scholar]
- 17.Eriksson H, Caidhal K, Larsson B, et al. Cardiac and pulmonary causes of dyspnoea: validation of a scoring test for clinical-epidemiological use: the Study of Men Born in 1913. Eur Heart J. 1987;8:1007–1014. doi: 10.1093/oxfordjournals.eurheartj.a062365. [DOI] [PubMed] [Google Scholar]
- 18.Ma J, Folsom AR, Shahar E, et al. Plasma fatty acid composition as an indicator of habitual dietary fat intake in middle-aged adults. Am J Clin Nutr. 1995;62:564–571. doi: 10.1093/ajcn/62.3.564. [DOI] [PubMed] [Google Scholar]
- 19.Ma J, Folsom AR, Eckfeldt JH, et al. Short- and long-term repeatability of fatty acid composition of human plasma phospholipids and cholesterol esters. Am J Clin Nutr. 1995;62:572–578. doi: 10.1093/ajcn/62.3.572. [DOI] [PubMed] [Google Scholar]
- 20.Loehr LR, Rosamond WD, Chang PP, et al. Heart failure incidence and survival (from the Atherosclerosis Risk in Communities study) Am J Cardiol. 2008;101:1016–1022. doi: 10.1016/j.amjcard.2007.11.061. [DOI] [PubMed] [Google Scholar]
- 21.Warensjö E, Risérus U, Vessby B. Fatty acid composition of serum lipids predicts the development of the metabolic syndrome in men. Diabetologia. 2005;48:1999–2005. doi: 10.1007/s00125-005-1897-x. [DOI] [PubMed] [Google Scholar]
- 22.Okada T, Furuhashi N, Kuromori Y, et al. Plasma palmitoleic acid content and obesity in children. Am J Clin Nutr. 2005;82:747–750. doi: 10.1093/ajcn/82.4.747. [DOI] [PubMed] [Google Scholar]
- 23.U.S. Department of Agriculture, Agricultural Research Service USDA National Nutrient Database for Standard Reference, Release 20. Nutrient Data Laboratory Home Page. 2007 http://www.ars.usda.gov/ba/bhnrc/ndl
- 24.Sun Q, Ma J, Campos H, et al. Plasma and erythrocyte biomarkers of dairy fat intake and risk of ischemic heart disease. Am J Clin Nutr. 2007;86:929–937. doi: 10.1093/ajcn/86.4.929. [DOI] [PubMed] [Google Scholar]
- 25.Salonen JT, Salonen R, Penttilä I, et al. Serum fatty acids, apolipoproteins, selenium, and vitamin antioxidants and the risk of death from coronary artery disease. Am J Cardiol. 1985;56:226–231. doi: 10.1016/0002-9149(85)90839-2. [DOI] [PubMed] [Google Scholar]
- 26.Miettinen TA, Naukkarinen V, Huttunen JK, et al. Fatty-acid composition of serum lipids predicts myocardial infarction. Br Med J. 1982;285:993–996. doi: 10.1136/bmj.285.6347.993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Borkman M, Storlien LH, Pan DA, et al. The relation between insulin sensitivity and the fatty-acid composition of skeletal-muscle phospholipids. N Engl J Med. 1993;328:238–244. doi: 10.1056/NEJM199301283280404. [DOI] [PubMed] [Google Scholar]
- 28.Vessby B, Tengblad S, Lithell H. Insulin sensitivity is related to the fatty acid composition of serum lipids and skeletal muscle phospholipids in 70-year-old men. Diabetologia. 1994;37:1044–1050. doi: 10.1007/BF00400468. [DOI] [PubMed] [Google Scholar]
- 29.Pan DA, Lillioja S, Milner MR, et al. Skeletal muscle membrane lipid composition is related to adiposity and insulin action. J Clin Invest. 1995;96:2802–2808. doi: 10.1172/JCI118350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Öhrvall M, Berglund L, Salminen I, Lithell H, Aro A, Vessby B. The serum cholesterol ester fatty acid composition but not the serum concentration of alpha tocopherol predicts the development of myocardial infarction in 50-year-old men: 19 years follow-up. Atherosclerosis. 1996;127:65–71. doi: 10.1016/s0021-9150(96)05936-9. [DOI] [PubMed] [Google Scholar]
- 31.Simon JA, Hodgkins ML, Browner WS, et al. Serum fatty acids and the risk of coronary heart disease. Am J Epidemiol. 1995;142:469–476. doi: 10.1093/oxfordjournals.aje.a117662. [DOI] [PubMed] [Google Scholar]
- 32.Pedersen JI, Ringstad J, Almendingen K, et al. Adipose tissue fatty acids and risk of myocardial infarction: a case-control study. Eur J Clin Nutr. 2000;54:618–625. doi: 10.1038/sj.ejcn.1601064. [DOI] [PubMed] [Google Scholar]
- 33.Stanley WC, Recchia FA, Okere IC. Metabolic therapies for heart disease: fish for prevention and treatment of cardiac failure? Cardiovasc Res. 2005;68:175–177. doi: 10.1016/j.cardiores.2005.09.002. [DOI] [PubMed] [Google Scholar]
- 34.Mozaffarian D. Fish, n-3 fatty acids, and cardiovascular haemodynamics. J Cardiovasc Med. 2007;8(Suppl 1):S23–26. doi: 10.2459/01.JCM.0000289279.95427.e2. [DOI] [PubMed] [Google Scholar]
- 35.Roger VL, Weston SA, Redfield MM, et al. Trends in heart failure incidence and survival in a community-based population. JAMA. 2004;292:344–350. doi: 10.1001/jama.292.3.344. [DOI] [PubMed] [Google Scholar]
- 36.Childs CE, Romeu-Nadal M, Burdge GC, et al. Gender difference in the n-3 fatty acid content of tissues. Proc Nutr Soc. 2008;67:19–27. doi: 10.1017/S0029665108005983. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.