Abstract
We investigated the role of Lef1, one of the four transcription factors that transmit Wnt signaling to the genome, in the regulation of bone mass. Microcomputed tomographic analysis of 13- and 17-week-old mice revealed significantly reduced trabecular bone mass in Lef1+/− females compared to littermate wild-type females. This was attributable to decreased osteoblast activity and bone formation as indicated by histomorphometric analysis of bone remodeling. In contrast to females, bone mass was unaffected by Lef1 haploinsufficiency in males. Similarly, females were substantially more responsive than males to haploinsufficiency in Gsk3β, a negative regulator of the Wnt pathway, displaying in this case a high bone mass phenotype. Lef1 haploinsufficiency also led to low bone mass in males lacking functional androgen receptor (AR) (tfm mutants). The protective skeletal effect of AR against Wnt-related low bone mass is not necessarily a result of direct interaction between the AR and Wnt signaling pathways, because Lef1+/− female mice had normal bone mass at the age of 34 weeks. Thus, our results indicate an age- and gender-dependent role for Lef1 in regulating bone formation and bone mass in vivo. The resistance to Lef1 haploinsufficiency in males with active AR and in old females could be due to the reduced bone turnover in these mice.
Introduction
The wingless/Wnt family of secreted glycoproteins has critical roles in cell growth and differentiation, and is highly conserved among vertebrates, flies, and primitive multicellular organisms [1], [2]. In mammals, the canonical Wnt pathway is pivotal to embryogenesis and tumorigenesis as well as in the maintenance and regeneration of tissues such as skin, intestine, liver, cardiac muscle, and the nervous system [1]–[7]. Wnt ligands bind to membrane complexes consisting of a seven transmembrane domain receptor of the frizzled family [3]–[5] and a ‘single-pass’ co-receptor, Lipoprotein Receptor-Related Protein 6 (Lrp6) and possibly Lrp5 as well [6]–[8]. In the absence of stimulation, the Wnt pathway is under the negative control of a pair of protein-serine kinases, Glycogen Synthase Kinase 3α and 3β (Gsk3α, Gsk3β), which phosphorylate β-catenin, a Wnt transducer, resulting in its ubiquitination and subsequent proteasomal degradation [9], [10]. Upon Wnt stimulation, the Gsk3-mediated phosphorylation of β-catenin is attenuated through disruption of the β-catenin destruction complex [11]. Subsequently, β-catenin accumulates and translocates to the nucleus, where it activates Wnt target genes by associating with the DNA-binding HMG box transcription factors lymphoid enhancer factor 1 (Lef1), T-cell factor 7 (Tcf7), Tcf7L1, and/or Tcf7L2 [12]–[15]. Tcf7, Tcf7L1 and Tcf7L2 are commonly known as Tcf1, Tcf3, and Tcf4, respectively, and these common names are used in this paper.
Osteoporosis, the most prevalent degenerative disease in western societies, is characterized by decreased bone mass and structural integrity. The mammalian skeleton undergoes continuous turnover, where overall bone gain or loss is determined by the difference between bone resorption and formation. After birth, bone mass increases until it reaches “peak bone mass”, which is then maintained at a constant level during young adulthood, followed by an age-related bone loss [16]. Bone mass accrual is subject to sexual dimorphism, with males having higher trabecular bone volume density and lower bone turnover than females [16]–[19]. The age-related bone loss is associated with reduction in turnover rate especially in females [20], [21].
In humans and mice alike, bone mass is strongly dependent on the Wnt signaling pathway [22], [23]. Mutations and polymorphism in Wnt10b, Dkk1, Dkk2, Sfrp1, Sost, Lrp6, and Gsk3β affect osteoblast function and therefore bone formation and bone mass [24]–[30]. The Wnt pathway has also been implicated in mediating positive and negative control of bone formation and bone mass by environmental factors such as pharmacological glucocorticoids [31]–[34] and mechanical stimulation [35]. Interestingly, Wnt signaling in osteoblasts also controls osteoclast activity, as demonstrated by increased resorption and a low bone mass (LBM) phenotype in mice whose osteoblasts lack β-catenin or Tcf1 [23]. The role of Lef1 in postnatal bone metabolism has not been studied, in part because Lef1 deficient mice die within the first week of life [36].
During early embryogenesis, Tcf1 and Lef1 are redundant. Mice lacking both genes have multiple defects, including duplicated neural tubes and malformed limb buds, whereas mice lacking either gene alone do not exhibit these defects [37], [38]. Non-redundant functions of these transcription factors are suggested by the distinct phenotypes observed in mice lacking either Tcf1 or Lef1. Whereas Tcf1-null mice display attenuated T cell differentiation [39], Lef1-null mice exhibit developmental defects in teeth, hair follicles, mammary glands and the brain [36], [40]. In terms of skeletal metabolism, analysis of Tcf1-null mice disclosed accelerated bone resorption [23], but similar analysis of Lef1-null mice is hampered by their perinatal lethality. In this study, we employed the viable Lef1 haploinsufficient mice, which do not display the abnormalities observed in the null mice, to assess the role of Lef1 in postnatal bone metabolism. We demonstrate a low bone mass phenotype in Lef1+/− mice, which appears to manifest specifically in the context of high bone turnover rate.
Results
Low bone mass (LBM) phenotype in Lef1 heterozygous female mice
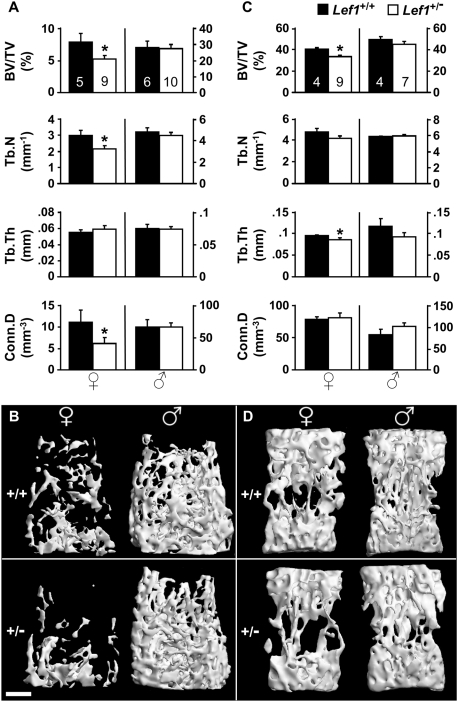
Micro-computed tomographic (μCT) analysis of 13-week old Lef1 haploinsufficient female mice revealed a LBM phenotype compared to littermate controls (Figure 1). The trabecular bone volume density (BV/TV) measured in the distal femur (Figure 1A) and the vertebral body (Figure 1C) of Lef1 haploinsufficient mice was 34% and 17% lower than the wild-type (WT) controls, respectively. In contrast to females, male mice showed no difference between Lef1+/− and WT animals (Figure 1). The decrease in BV/TV due to Lef1 haploinsufficiency resembles the magnitude of trabecular bone loss due to Lrp6 haploinsufficiency [24].
Figure 1. Low trabecular bone mass in Lef1+/− female mice.
(A,C) μCT analysis of the distal femoral (A) and the vertebral (C) trabecular bone of Lef1+/+ (black) and Lef1+/− (white) female (left) and male (right) 13-week old mice. BV/TV – trabecular bone volume density, Tb.N – trabecular number, Tb.Th – trabecular thickness, Conn.D – connectivity density (Mean±SEM of 4–10 specimens as indicated within the bars at the top, * = p<0.05). (B,D) μCT images of distal femoral (B) and vertebral (D) trabecular bone of female (left) and male (right) 13-week old mice with median BV/TV. Scale bar = 0.5 mm.
Detailed analysis of the trabecular bone parameters in the 13-week old female mice revealed interesting site-specific responses to Lef1 haploinsufficiency. While the decreased BV/TV at the vertebral bodies was attributable to thinning of trabeculae, the LBM at the distal femoral metaphysis was due to decreased trabecular number (Tb.N) and was also associated with decreased connectivity density (Conn.D, Figure 1). Skeletal site-specific control of bone mass, both related and unrelated to the Wnt signaling pathway, has been previously observed [25], [41]–[44], although the underlying mechanisms remain to be elucidated. A similar trabecular bone phenotype was observed in 17-week old Lef1+/− females, and again males were unaffected. At 13 weeks of age, the Lef1+/− females, but not males, also exhibited reduced cortical bone thickness (0.162 mm vs. 0.188 mm in wild-type females, p = 0.021), but the femoral length and mid-diaphyseal diameter were unaffected by the Lef1 gene dosage in either gender (data not shown). Additionally, we did not detect any skeletal abnormalities in Lef1 knockout newborns, as indicated by whole mount staining and histological analysis (Figure S1). The total body weight was similar in Lef1 heterozygous and gender/age-matched WT controls (data not shown). Thus, Lef1 haploinsufficiency leads to a LBM phenotype specifically in females, demonstrating for the first time a role for Lef1 in bone metabolism.
Decreased bone formation in Lef1+/− female mice
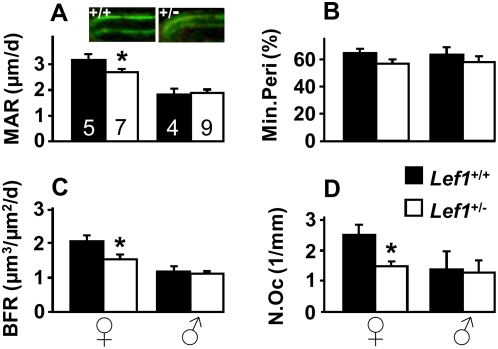
Wnt signaling has been implicated in both promoting osteoblast [45] and attenuating osteoclast function [23]. Accordingly, Lef1 haploinsufficiency could lead to a LBM phenotype by either inhibiting bone formation or stimulating bone resorption. We therefore assessed trabecular bone formation and resorption in distal femoral metaphyses of 17-week old Lef1+/− mice and littermate controls using vital calcein labeling and TRAP staining, respectively. As shown in Figure 2C, Lef1+/− female mice exhibited a 25% lower bone formation rate (BFR) as compared to WT controls (Figure 2C), attributable mainly to decreased mineral apposition rate (MAR; Figure 2A), which represents the activity of the average osteoblast. Thus, the female Lef1+/− LBM phenotype is attributable to reduced osteoblast function. In contrast, there was no indication for increased bone resorption in Lef1+/− females because they had less, not more TRAP-positive cells compared to controls (Figure 2D). Importantly, the skeletal remodeling analysis in male mice revealed no difference between Lef1+/− and Lef1 +/+ animals (Figure 2). On a side note, our findings demonstrate lower bone turnover in WT male compared to WT female mice (−42.1% MAR; −51.4% BFR and −45.6% osteoclast number, p<0.05 for each parameter), which is consistent with previous reports [46], [47]. The higher bone turnover in females compared to males may predispose the formers to Lef1 haploinsufficiency-induced LBM (see below).
Figure 2. Low bone formation in Lef1+/− female mice.
Histomorphometric analysis of the distal femoral metaphysis from Lef1 +/+ (black) and Lef1+/− (white) female (left) and male (right) 17-week old mice. (A) mineral apposition rate (MAR), a surrogate for osteoblast activity; images show representative histological sections from Lef1 +/+ (left) and Lef1+/− (right) females; (B) mineralizing perimeter (Min.Peri), a surrogate for osteoblast number; (C) bone formation rate (BFR); (D) trabecular bone osteoclast number (N.Oc/BS). Data represent mean±SEM of 4–9 specimens as indicated within the bars in A; * = p<0.05.
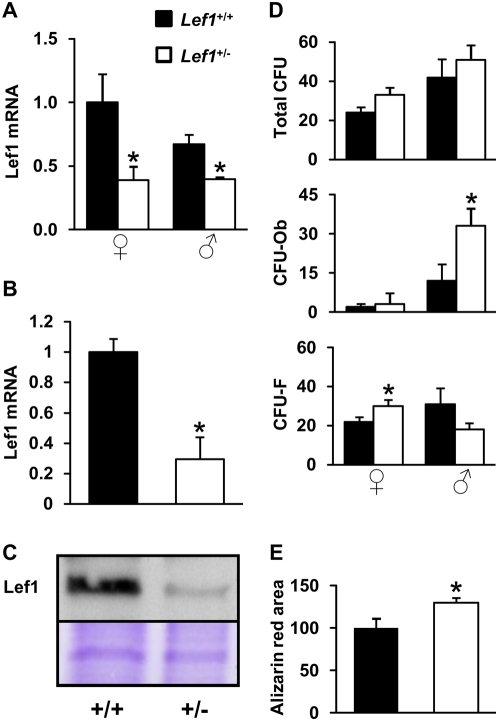
We next assessed the effects of Lef1 heterozygocity on Lef1 expression and on osteoblast differentiation in vitro. First, we confirmed that Lef1 expression was significantly reduced in bones and in newborn mouse calvarial osteoblast (NeMCO) cultures derived from Lef1 heterozygous compared to control mice (Figure 3A–3C). In mesenchymal stem cell (MSC) cultures derived from bone marrow of female mice, Lef1 haploinsufficiency increased the number of CFU-F, whereas the number of CFU-Ob was unchanged (Figure 3D). Interestingly, Lef1 haploinsufficiency in male mice, which did not reduce bone mass in vivo (Figure 1), was associated with an increase in bone marrow-derived CFU-Ob (Figure 3D). In vitro osteoblast differentiation as defined by mineralization in NeMCO cultures was accelerated by Lef1 haploinsufficiency (Figure 3E).
Figure 3. Reduced Lef1 expression is not associated with impaired osteoblast differentiation in vitro.
(A,B) Lef1 mRNA in tibia of Lef1 +/− and control male and female mice (A) and in NeMCO cultures (B) was assessed by RT-qPCR and corrected for the expression of GAPDH and rpL10A, respectively. Bars represent relative expression levels (Mean±SEM, n = 3). (C) Western blot analysis of Lef1 expression in NeMCO cultures. Equal loading was demonstrated by Coomassie blue staining (bottom panel shows a ∼60 KDa Coomassie blue-stained band). (D) Bone marrow mesenchymal cells from WT and Lef1+/− mice of each gender were cultured for 28 days, fixed and stained with Alizarin red. Colonies were manually counted in each of 6–11 independent cultures for each condition. Alizarin red-positive colonies were counted as osteoblastic colony-forming units (CFU-Ob), and the rest were considered fibroblastic CFU (CFU-F). (E) Day-20 NeMCO cultures from WT and Lef1+/− mice were fixed and stained with Alizarin red. Data represent mean mineralized area relative to WT±SEM in at least 3 cultures per condition. * = p<0.05 vs. WT.
High bone mass (HBM) in Gsk3β haploinsufficient female mice
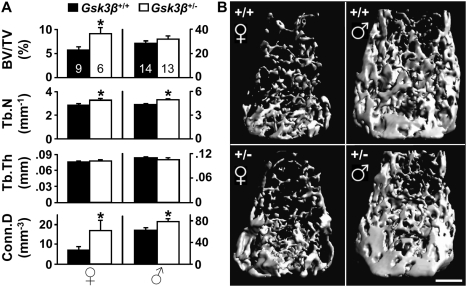
Alterations in components of the Wnt pathway other than Lef1 may also have stronger skeletal effects in females as compared to males. To address this notion, we compared the role of Gsk3β, a negative regulator of the Wnt pathway, in female versus male bone mass. Because the Gsk3ß-null mice die in utero [48], we analyzed the trabecular bone in the distal femoral metaphysis of mice haploinsufficient for Gsk3ß. Indeed, female, but not male Gsk3β +/− mice exhibited a high bone mass (HBM) phenotype compared to WT littermates (Figure 4A). The elevated BV/TV was attributable to increased trabecular number, and was associated with increased connectivity density (Figure 4), a mirror image of the respective Lef1+/− LBM phenotype. Jointly, the gender-preferential effects of both Lef1 and Gsk3β haploinsufficiency suggest that the skeleton is more sensitive to variations in Wnt signaling in females compared to males.
Figure 4. High trabecular bone mass in Gsk3β +/− female mice.
(A) μCT analysis of the distal femoral trabecular bone of 16 week old Gsk3β +/+ (black) and Gsk3β +/− (white) female (left) and male (right) mice. Data represent mean±SEM of 6–14 specimens as indicated within the bars at the top, * = p<0.05. (B) μCT images from female (left) and male (right) mice with median BV/TV. Scale bar = 0.5 mm.
Androgen signaling protects against Lef1 haploinsufficiency
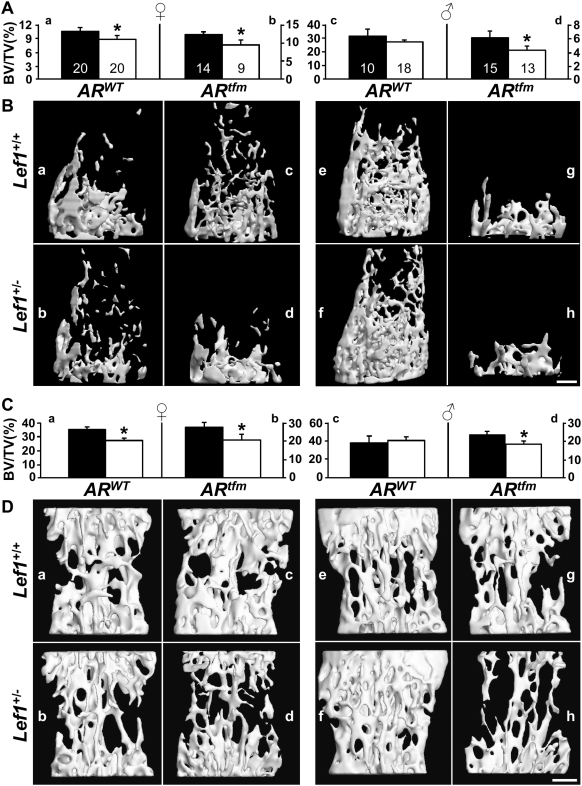
The female-preferential skeletal phenotype of the Lef1+/− mice could be explained by a compensatory gene(s) on the Y chromosome, hypersensitization by estrogens, or protection by androgens. In support of the latter possibility, androgens can augment Wnt signaling [49], [50], and even stimulate Lef1 expression in osteoblasts (Figure S2) similar to what has been observed in adipocytes [51]. To test the hypothesis that androgen signaling protects against Lef1 haploinsufficiency-induced LBM in vivo, we employed tfm male mice, in which androgen signaling is absent due to a naturally occurring mutation in the androgen receptor (AR) [52]. To generate Lef1 +/−;ARtfm mice, Lef1+/− males were bred with female tfm carriers. Because the ARtfm allele is embedded in a tabby genomic sequence [53], we first analyzed the distal femora in offspring with wild type AR, but which are no longer on a pure C57BL/6 background. Similar to the original results (Figure 1), female Lef1+/− mice that partially carry the tabby genome exhibited a LBM phenotype (Figure 5Aa, and compare Figure 5Ba to 5Bb), while male mice on the same genetic background were protected (Figure 5Ac, and compare Figure 5Be to 5Bf). Remarkably, however, male tfm mice were vulnerable to Lef1 haploinsufficiency, similar to females (Figure 5). Specifically, Lef1 +/−;ARtfm mice had a 22% lower BV/TV as compared to their Lef1 +/+;ARtfm counterparts (Figure 5Ad, and compare Figure 5Bg to 5Bh). Similar results were observed in the vertebral bodies (Figure 5C and 5D). Because estrogen levels in tfm males do not approach those of females [54], and because these mice still carry an intact Y chromosome, these results demonstrate that androgen signaling protects against Lef1 haploinsufficiency-induced LBM.
Figure 5. Androgen signaling protects against Lef1 haploinsufficiency-induced low bone mass.
μCT analysis of the trabecular bone compartment in distal femora (A,B) and vertebral bodies (C,D) of 13-week old female (left) and male (right) Lef1 +/+ (black) and Lef1+/− (white) mice. ARtfm males have no functional AR, while ARtfm females are carriers for the defective AR allele. Data represent mean±SEM of 9–20 specimens as indicated within the bars at the top, * = p<0.05. (B) and (D) show respective μCT images of female (left) and male (right) mice with median BV/TV. Mice in a, b, e and f carry the wild-type AR (ARWT); mice in c, d, g and h carry the ARtfm allele; mice in a, c, e and g are Lef1 +/+; mice in b, d, f and h are Lef1 +/−. Scale bar = 0.5 mm.
Aged females are resistant to Lef1 haploinsufficiency-induced LBM
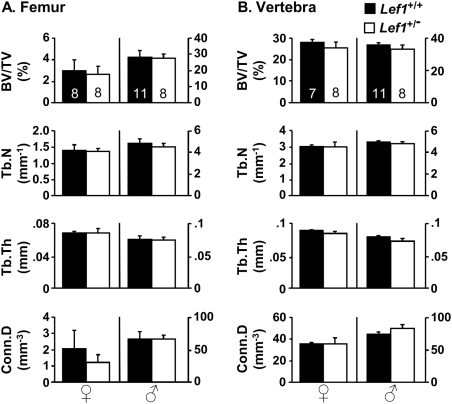
It has been previously suggested that androgen signaling can augment Wnt signaling in bone cells [49], [50]. This could explain how males are protected against Lef1 haploinsufficiency-induced LBM. Alternatively, androgens could also indirectly protect males by restraining bone turnover [54], [55]. If low bone turnover protects male mice from Lef1 haploinsufficiency, then Lef1+/− females may no longer display a low bone mass phenotype compared to WT females at ages older than 17 weeks, when bone turnover decreases [20], [21]. Indeed, μCT analysis of 34-week old female mice revealed no difference between Lef1+/− and WT females (Figure 6). At this age, the male skeleton was again unaffected by Lef1 haploinsufficiency (Figure 6). In summary, Lef1 haploinsufficiency induces LBM in a gender- and age-specific manner.
Figure 6. Thirty four-week old female mice are insensitive to Lef1 haploinsufficiency.
(A,B) μCT analysis of the distal femoral (A) and the vertebral (B) trabecular bone of Lef1+/+ (black) and Lef1+/− (white) female (left) and male (right) 34-week old mice (Data represent mean±SEM of 7–11 specimens as indicated within the bars at the top).
Discussion
The present work demonstrates low bone mass in mice, in which one Lef1 gene copy is disrupted. Interestingly, bone mass was reduced in 13- and 17-week old females, but not in males of any age or in 34-week old females. This gender-specificity reflected neither protection by a Y chromosome-associated gene nor sensitization by estrogens, since Lef1+/− male mice harboring a testicular feminization mutation (tfm) also displayed a LBM phenotype despite their having an intact Y chromosome and not having feminine estrogen levels [54]. Gsk3β+/− mice displayed a mirror image of the Lef1+/− phenotype, namely increased bone mass in female mice only. In line with our findings, disruption of Sfrp1, a Wnt antagonist, resulted in a female-preferential bone phenotype [29]. Thus, genetic alterations in components of the Wnt pathway appear to affect female more than male mice. That Lrp5 deficiency reduces bone mass equally in males and females [56] is not inconsistent with our conclusion in light of the recent work of Yadav et al. [57], who disputed the paradigm that places Lrp5 upstream of Gsk3β and Lef1 in osteoblasts. Plausibly, a genetic lesion at the level of Lef1 itself compromises canonical Wnt signaling and is therefore more consequential in females than in males.
All mice that were sensitive to Lef1 haploinsufficiency in our study, namely young females and tfm males, are also characterized by a relatively high rate of bone turnover (Figure 2 and ref. [54]). In contrast, mice resistant to Lef1 haploinsufficiency—AR WT males and aged females (Figure 1, Figure 5, Figure 6)—have a lower rate of bone turnover (Figure 2 and [20], [21]). Thus, a unifying explanation for our observations is that bone turnover rate determines the skeletal response to genetic alterations in canonical Wnt signaling. Our favored interpretation of the gender-specific sensitivity to Lef1 haploinsufficiency is that androgens protect the skeleton from the potential deleterious effect of reduced Lef1 by restraining bone turnover [54], [55].
We cannot rule out an alternative explanation whereby AR activity compensates for Lef1 haploinsufficiency via molecular interaction with the canonical Wnt pathway. In fact, it has been shown that DHT stimulates Lef/Tcf-mediated transcription in osteoblasts [49], [50]. This could occur via physical interaction of liganded AR with β-catenin [51] or with membrane residents such as Src [58], which could then impinge on the Wnt pathway through activation of the PI3 kinase/Akt/Gsk3β axis [50], [59], [60]. Alternatively, androgens could regulate the expression of either Lef1 itself (Figure S2 and [51]) or Wnt agonists and/or antagonists [61]. However, the normal bone phenotype observed in 34-week old Lef1+/− female mice favors the hypothesis that androgen signaling, much like aging in females, overrides the skeletal sensitivity to Lef1 haploinsufficiency by restraining bone turnover. Obviously, the two explanations for AR-mediated protection against Lef1 haploinsufficiency – molecular interaction with the Wnt pathway and attenuation of bone turnover – are not mutually exclusive.
It remains to be examined to what extent the effect of Lef1 on bone formation is cell autonomous. In favor of cell autonomy is the reduced Lef1 expression in Lef1 +/− osteoblasts and the observed gender-dependent changes in the numbers of CFU-F and CFU-Ob in bone marrow-derived MSCs cultures (Figure 3D). Possibly, Lef1 haploinsufficiency promotes premature osteoblast differentiation ([62], [63] and Figure 3E). However, much like the effect of duodenal Lrp5 [57], the role of Lef1 in regulating bone formation may reside in cells other than osteoblasts. Cell type-specific knockout studies will be necessary to clarify this issue.
An intriguing, albeit speculative, extrapolation from our findings is that females reach lower peak bone mass than males because, in the absence of androgens, higher rate of bone turnover renders the young female skeleton more vulnerable to sub-optimal activity of canonical Wnt signaling and possibly other pathways. Other investigators reported on age- and gender-dependent bone phenotypes in mice with genetic alterations in different pathways. For example, osteoblast-specific disruption of BMP type-IA receptor leads to LBM in young mice but HBM in old mice [64]. Very similar age-dependent effects were reported for Runx2 haploinsufficiency in mice [20]. In addition, the strong anabolic effect of estrogen in young Runx2 +/− mice was almost completely abolished in aged mice [20]. With regard to gender specificity, and in addition to the female-preferential response to genetic manipulation of Lef1, GSK3ß and Sfrp1, ablation of Cathepsin K results in a 3-fold stronger effect in female compared to male mice [47]. Age- and gender-related variations in bone turnover may explain the differential skeletal responses to some of these and other genetic aberrations. Furthermore, hormonal and age-related variation in bone turnover may contribute to gender- and age-related susceptibility to osteoporosis and response to therapies.
Materials and Methods
Animals
Lef1+/− and Gsk3β +/− mice and their controls, all on a C57BL/6 background, were generated by breeding Lef1+/− [36] or Gsk3β +/− [26] mice with C57BL/6 mice from either Harlan Laboratories (Indianapolis, Indiana, USA) or the Ontario Cancer Institute (Toronto, Canada), respectively. Mice carrying the testicular feminization mutation (Tfm) (Jackson Laboratories, Bar Harbor, Maine, USA) on a C57BL/6J-A-Ta<6J> background were bred with the Lef1+/− mice and F1 litters were examined. To measure the percentage of bone surface undergoing mineralization and the mineralization rate, mice were injected intraperitoneally with 15 mg/kg of the fluorochrome calcein (Sigma-Aldrich, St. Louis, MO, USA) four days and again one day prior to sacrifice. One femur and the fifth lumbar vertebra (L5) from each mouse were dissected and fixed in 10% phosphate-buffered formalin (pH = 7.2) for 24 hours, and then stored in 70% ethanol. All experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Southern California and of the University of Toronto.
Micro-computed tomography
Femora (one per mouse) and fifth lumbar vertebrae (L5) were examined as reported previously [65], [66] using either Scanco μCT 40 (Scanco Medical AG, Brüttisellen, Switzerland), or Siemens MicroCAT II (Siemens Medical Solutions, Knoxville, TN, USA). Briefly, scans were performed at a 20-µm resolution in all three spatial dimensions. The mineralized tissues were differentially segmented by a global thresholding procedure [67]. Trabecular parameters in the secondary spongiosa of the distal femoral metaphysis included trabecular bone volume density (BV/TV), trabecular thickness (Tb.Th), trabecular number (Tb.N) and connectivity density (Conn.D). Cortical thickness, diaphyseal diameter, and medullary cavity diameter were determined in the mid-diaphyseal region. In L5 bodies, the entire trabecular bone compartment was analyzed. All morphometric parameters were determined by using a direct 3D approach [68]. Differences between groups were analyzed by student's t-test (two-tailed) and were considered significant when p<0.05.
Histomorphometry
After μCT image acquisition, femora were embedded undecalcified in polymethylmethacrylate (Technovit 9100, Heraeus Kulzer, Germany). Undeplasticized longitudinal 5-µm sections from the center of each bone were left unstained for dynamic histomorphometric measurements. To identify osteoclasts, consecutive sections were deplasticized and stained with tartrate-resistant acid phosphatase (TRAP; Sigma-Aldrich, St. Louis, MO, USA) and counterstained with Mayer's hematoxylin [69]. The morphometric analysis was performed using the Image-Pro Discovery software (Media Cybernetics, Silver Spring, MD, USA). The following parameters were determined: mineral apposition rate (MAR), mineralizing perimeter (Min.Peri), bone formation rate (BFR) and osteoclast number (N.Oc/BS). The terminology and units used for these measurements were according to the convention of standardized nomenclature [70]. Statistical analysis was performed as above.
Tissue culture
NeMCO cultures were prepared from one day-old pups as described previously [71]. Cells were cultured in 6-well plates for Western blot analysis and in 12-well plates for RT-qPCR and mineralization assays. For the latter, osteogenic medium containing ascorbic acid (50 µg/mL) and β-glycerophosphate (10 mM) was initiated at confluence and alizarin red staining was performed at day 20. For MSC cultures, the cellular content of the bone marrow cavity from two femurs and two tibiae from each mouse was flushed using αMEM and passed through needles with decreasing diameters (down to 25G) to obtain a single cell suspension. Cells were then plated at 3×106 per well in 6-well plates and incubated for 3 days in αMEM (Invitrogen) supplemented with 15% FBS (Gemini Bio-Products, West Sacramento, CA). Starting at day 3, the MSC were cultured in osteogenic medium and stained with Alizarin red after 28 days.
Lef1 expression
Total RNA was extracted from freshly isolated tibiae of 10 week-old mice. Upon harvesting, one tibia per animal was stabilized in RNALater (Ambion, Austin, TX), homogenized in Trizol (Invitrogen), purified using 1-Bromo-3-Chloropropane and isopropanol, then rinsed in 70% ethanol. RNA from cells was extracted using Aurum Total RNA Mini Kit (Biorad, Hercules, CA). cDNA was produced using Superscript III First Strand cDNA synthesis kit (Invitrogen) and Real-Time PCR was performed using iQ SYBR green supermix (Biorad) and an Opticon 2 real time PCR machine (Biorad). Lef1 mRNA levels in tibiae and NeMCO cultures were corrected for GAPDH and ribosomal protein L10A (rpL10A) mRNA, respectively. Primers used for PCR are listed in Table 1. Western blot analysis of Lef1 in NeMCO cultures was performed essentially as previously described [71] using anti-Lef1 antibody from Cell Signaling (Danvers, MA) and secondary antibodies from Santa Cruz Biotechnology (Santa Cruz, CA).
Table 1. Primers for genotyping and RT–qPCR.
| Genotyping | ||
| Lef1 | LPP2.2 | 5′TGTCTCTCTTTCCGTGCTAGTTC3′ |
| D8 | 5′CCGTTTCAGTGGCACGCCCTCTCC3′ | |
| Neo | 5′ATGGCGATGCCTGCTTGCCGAATA3′ | |
| Sry | Fwd | 5′TCATGAGACTGCCAACCACAG3′ |
| Rev | 5′CATGACCACCACCACCACCAA3′ | |
| Tfm* | Fwd | 5′GTGAAGCAGGTAGCTCTGGG3′ |
| Rev | 5′GTTCTCCAGCTTGATACGGG3′ | |
| RT-qPCR | ||
| GAPDH | Fwd | 5′CCAGAACATCATCCCTGCAT3′ |
| Rev | 5′CTTGCCCACAGCCTTGGCAGC3′ | |
| rpL10A | Fwd | 5′CGCCGCAAGTTTCTGGAGAC3′ |
| Rev | 5′CTTGCCAGCCTTGTTTAGGC3′ | |
| Lef1 | Fwd | 5′TGAGTGCACGCTAAAGGAGA3′ |
| Rev | 5′ATAATTGTCTCGCGCTGACC3′ | |
PCR product was digested with MwoI resulting in either a 137 bp (WT) or 182 bp (Tfm) band.
Supporting Information
Lef1 −/− mice have normal bone development. Histological evaluation of wild type (a–e) versus Lef1 −/− (f–j) newborn mice. (a,b,f,g) Alizarin red/Alcian blue staining of craniofacial bones. (c,h) Alizarin red/Alcian blue staining of hind limb and vertebrae. (d,i) H-E staining of longitudinal femoral sections. (e,j) Toluidine blue staining of distal femoral growth plates. Representative images are shown. No abnormality was detected in the Lef1 −/− skeletons, except for the previously reported lack of teeth (f) (van Genderen et al. 1994, Genes Dev 8, 2691-703)
(3.47 MB TIF)
DHT stimulates Lef1 expression in vitro. MC3T3-E1 osteoblast cultures maintained in phenol-red free αMEM supplemented with 10% charcoal-stripped serum were treated with 30 nM DHT or 100 nM estradiol for 48 hours. Expression of the four members of the Lef/Tcf gene family was assessed by RT-qPCR and corrected for the expression of rpL10A. Bars represent expression levels in the presence of hormone relative to the ethanol vehicle, defined for each gene as 1.
(0.88 MB TIF)
Acknowledgments
We thank Rudolf Grosschedl (University of Munich and Max-Planck-Institute, Germany) for the Lef1-targeted mice.
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: BF's group was supported by the Arthritis Foundation (Atlanta, GA), by the NIH (AR047052), and by the J. Harold and Edna L. LaBriola Chair in Genetic Orthopaedic Research at the University of Southern California. IB's team was supported by the NIH (AR047052). JW's group was supported by the Canadian Institutes of Health Research (MOP#74711). TK and RM were supported by the Swiss National Science Foundation (Grants FP 620-58097.99 and PP-104317/1). YG was partly supported by a Meyer Young Investigator Fellowship from the Arthritis Foundation Southern California Chapter. The use of the microCAT II image system at the USC Molecular Imaging Center was made possible by a 2005 NCRR shared instrumentation award. The experiments were conducted in part in a facility constructed with support from Research Facilities Improvement Program Grant Number C06 (RR10600-01, CA62528-01, RR14514-01) from the NIH/NCRR. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Miller JR, Moon RT. Signal transduction through beta-catenin and specification of cell fate during embryogenesis. Genes Dev. 1996;10:2527–2539. doi: 10.1101/gad.10.20.2527. [DOI] [PubMed] [Google Scholar]
- 2.Cadigan KM, Nusse R. Wnt signaling: a common theme in animal development. Genes Dev. 1997;11:3286–3305. doi: 10.1101/gad.11.24.3286. [DOI] [PubMed] [Google Scholar]
- 3.Bhanot P, Brink M, Samos CH, Hsieh JC, Wang Y, et al. A new member of the frizzled family from Drosophila functions as a Wingless receptor. Nature. 1996;382:225–230. doi: 10.1038/382225a0. [DOI] [PubMed] [Google Scholar]
- 4.He X, Saint-Jeannet JP, Wang Y, Nathans J, Dawid I, et al. A member of the Frizzled protein family mediating axis induction by Wnt-5A. Science. 1997;275:1652–1654. doi: 10.1126/science.275.5306.1652. [DOI] [PubMed] [Google Scholar]
- 5.Yang-Snyder J, Miller JR, Brown JD, Lai CJ, Moon RT. A frizzled homolog functions in a vertebrate Wnt signaling pathway. Curr Biol. 1996;6:1302–1306. doi: 10.1016/s0960-9822(02)70716-1. [DOI] [PubMed] [Google Scholar]
- 6.Pinson KI, Brennan J, Monkley S, Avery BJ, Skarnes WC. An LDL-receptor-related protein mediates Wnt signalling in mice. Nature. 2000;407:535–538. doi: 10.1038/35035124. [DOI] [PubMed] [Google Scholar]
- 7.Tamai K, Semenov M, Kato Y, Spokony R, Liu C, et al. LDL-receptor-related proteins in Wnt signal transduction. Nature. 2000;407:530–535. doi: 10.1038/35035117. [DOI] [PubMed] [Google Scholar]
- 8.Wehrli M, Dougan ST, Caldwell K, O'Keefe L, Schwartz S, et al. arrow encodes an LDL-receptor-related protein essential for Wingless signalling. Nature. 2000;407:527–530. doi: 10.1038/35035110. [DOI] [PubMed] [Google Scholar]
- 9.Hart MJ, de los Santos R, Albert IN, Rubinfeld B, Polakis P. Downregulation of beta-catenin by human Axin and its association with the APC tumor suppressor, beta-catenin and GSK3 beta. Curr Biol. 1998;8:573–581. doi: 10.1016/s0960-9822(98)70226-x. [DOI] [PubMed] [Google Scholar]
- 10.Itoh K, Krupnik VE, Sokol SY. Axis determination in Xenopus involves biochemical interactions of axin, glycogen synthase kinase 3 and beta-catenin. Curr Biol. 1998;8:591–594. doi: 10.1016/s0960-9822(98)70229-5. [DOI] [PubMed] [Google Scholar]
- 11.Ikeda S, Kishida S, Yamamoto H, Murai H, Koyama S, et al. Axin, a negative regulator of the Wnt signaling pathway, forms a complex with GSK-3beta and beta-catenin and promotes GSK-3beta-dependent phosphorylation of beta-catenin. Embo J. 1998;17:1371–1384. doi: 10.1093/emboj/17.5.1371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Travis A, Amsterdam A, Belanger C, Grosschedl R. LEF-1, a gene encoding a lymphoid-specific protein with an HMG domain, regulates T-cell receptor alpha enhancer function [corrected]. Genes Dev. 1991;5:880–894. doi: 10.1101/gad.5.5.880. [DOI] [PubMed] [Google Scholar]
- 13.Waterman ML, Fischer WH, Jones KA. A thymus-specific member of the HMG protein family regulates the human T cell receptor C alpha enhancer. Genes Dev. 1991;5:656–669. doi: 10.1101/gad.5.4.656. [DOI] [PubMed] [Google Scholar]
- 14.van de Wetering M, Oosterwegel M, Dooijes D, Clevers H. Identification and cloning of TCF-1, a T lymphocyte-specific transcription factor containing a sequence-specific HMG box. Embo J. 1991;10:123–132. doi: 10.1002/j.1460-2075.1991.tb07928.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Korinek V, Barker N, Willert K, Molenaar M, Roose J, et al. Two members of the Tcf family implicated in Wnt/beta-catenin signaling during embryogenesis in the mouse. Mol Cell Biol. 1998;18:1248–1256. doi: 10.1128/mcb.18.3.1248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bab I, Müller R, Hajbi-Yonissi C, Gabet Y. Micro-Tomographic Atlas of the Mouse Skeleton. New York: Springer; 2007. [Google Scholar]
- 17.Neu CM, Manz F, Rauch F, Merkel A, Schoenau E. Bone densities and bone size at the distal radius in healthy children and adolescents: a study using peripheral quantitative computed tomography. Bone. 2001;28:227–232. doi: 10.1016/s8756-3282(00)00429-4. [DOI] [PubMed] [Google Scholar]
- 18.Macdonald H, Kontulainen S, Petit M, Janssen P, McKay H. Bone strength and its determinants in pre- and early pubertal boys and girls. Bone. 2006;39:598–608. doi: 10.1016/j.bone.2006.02.057. [DOI] [PubMed] [Google Scholar]
- 19.Glatt V, Canalis E, Stadmeyer L, Bouxsein ML. Age-related changes in trabecular architecture differ in female and male C57BL/6J mice. J Bone Miner Res. 2007;22:1197–1207. doi: 10.1359/jbmr.070507. [DOI] [PubMed] [Google Scholar]
- 20.Juttner KV, Perry MJ. High-dose estrogen-induced osteogenesis is decreased in aged RUNX2(+/−) mice. Bone. 2007;41:25–32. doi: 10.1016/j.bone.2007.03.012. [DOI] [PubMed] [Google Scholar]
- 21.Pantschenko AG, Zhang W, Nahounou M, McCarthy MB, Stover ML, et al. Effect of osteoblast-targeted expression of bcl-2 in bone: differential response in male and female mice. J Bone Miner Res. 2005;20:1414–1429. doi: 10.1359/JBMR.050315. [DOI] [PubMed] [Google Scholar]
- 22.Glass DA, 2nd, Karsenty G. Molecular bases of the regulation of bone remodeling by the canonical Wnt signaling pathway. Curr Top Dev Biol. 2006;73:43–84. doi: 10.1016/S0070-2153(05)73002-7. [DOI] [PubMed] [Google Scholar]
- 23.Glass DA, 2nd, Bialek P, Ahn JD, Starbuck M, Patel MS, et al. Canonical Wnt signaling in differentiated osteoblasts controls osteoclast differentiation. Dev Cell. 2005;8:751–764. doi: 10.1016/j.devcel.2005.02.017. [DOI] [PubMed] [Google Scholar]
- 24.Holmen SL, Giambernardi TA, Zylstra CR, Buckner-Berghuis BD, Resau JH, et al. Decreased BMD and limb deformities in mice carrying mutations in both Lrp5 and Lrp6. J Bone Miner Res. 2004;19:2033–2040. doi: 10.1359/JBMR.040907. [DOI] [PubMed] [Google Scholar]
- 25.Bennett CN, Longo KA, Wright WS, Suva LJ, Lane TF, et al. Regulation of osteoblastogenesis and bone mass by Wnt10b. Proc Natl Acad Sci U S A. 2005;102:3324–3329. doi: 10.1073/pnas.0408742102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kugimiya F, Kawaguchi H, Ohba S, Kawamura N, Hirata M, et al. GSK-3beta controls osteogenesis through regulating Runx2 activity. PLoS ONE. 2007;2:e837. doi: 10.1371/journal.pone.0000837. doi:10.1371/journal.pone.0000837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Morvan F, Boulukos K, Clement-Lacroix P, Roman Roman S, Suc-Royer I, et al. Deletion of a single allele of the Dkk1 gene leads to an increase in bone formation and bone mass. J Bone Miner Res. 2006;21:934–945. doi: 10.1359/jbmr.060311. [DOI] [PubMed] [Google Scholar]
- 28.Li X, Liu P, Liu W, Maye P, Zhang J, et al. Dkk2 has a role in terminal osteoblast differentiation and mineralized matrix formation. Nat Genet. 2005;37:945–952. doi: 10.1038/ng1614. [DOI] [PubMed] [Google Scholar]
- 29.Bodine PV, Zhao W, Kharode YP, Bex FJ, Lambert AJ, et al. The Wnt antagonist secreted frizzled-related protein-1 is a negative regulator of trabecular bone formation in adult mice. Mol Endocrinol. 2004;18:1222–1237. doi: 10.1210/me.2003-0498. [DOI] [PubMed] [Google Scholar]
- 30.Kamiya N, Ye L, Kobayashi T, Mochida Y, Yamauchi M, et al. BMP signaling negatively regulates bone mass through sclerostin by inhibiting the canonical Wnt pathway. Development. 2008;135:3801–3811. doi: 10.1242/dev.025825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Smith E, Coetzee GA, Frenkel B. Glucocorticoids inhibit cell cycle progression in differentiating osteoblasts via glycogen synthase kinase-3beta. J Biol Chem. 2002;277:18191–18197. doi: 10.1074/jbc.M109708200. [DOI] [PubMed] [Google Scholar]
- 32.Ohnaka K, Taniguchi H, Kawate H, Nawata H, Takayanagi R. Glucocorticoid enhances the expression of dickkopf-1 in human osteoblasts: novel mechanism of glucocorticoid-induced osteoporosis. Biochem Biophys Res Commun. 2004;318:259–264. doi: 10.1016/j.bbrc.2004.04.025. [DOI] [PubMed] [Google Scholar]
- 33.Leclerc N, Noh T, Cogan J, Samarawickrama DB, Smith E, et al. Opposing effects of glucocorticoids and Wnt signaling on Krox20 and mineral deposition in osteoblast cultures. J Cell Biochem. 2007 doi: 10.1002/jcb.21587. [DOI] [PubMed] [Google Scholar]
- 34.Wang FS, Lin CL, Chen YJ, Wang CJ, Yang KD, et al. Secreted frizzled-related protein 1 modulates glucocorticoid attenuation of osteogenic activities and bone mass. Endocrinology. 2005;146:2415–2423. doi: 10.1210/en.2004-1050. [DOI] [PubMed] [Google Scholar]
- 35.Armstrong VJ, Muzylak M, Sunters A, Zaman G, Saxon LK, et al. Wnt/beta-catenin signaling is a component of osteoblastic bone cell early responses to load-bearing and requires estrogen receptor alpha. J Biol Chem. 2007;282:20715–20727. doi: 10.1074/jbc.M703224200. [DOI] [PubMed] [Google Scholar]
- 36.van Genderen C, Okamura RM, Farinas I, Quo RG, Parslow TG, et al. Development of several organs that require inductive epithelial-mesenchymal interactions is impaired in LEF-1-deficient mice. Genes Dev. 1994;8:2691–2703. doi: 10.1101/gad.8.22.2691. [DOI] [PubMed] [Google Scholar]
- 37.Galceran J, Farinas I, Depew MJ, Clevers H, Grosschedl R. Wnt3a−/−like phenotype and limb deficiency in Lef1(−/−)Tcf1(−/−) mice. Genes Dev. 1999;13:709–717. doi: 10.1101/gad.13.6.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Okamura RM, Sigvardsson M, Galceran J, Verbeek S, Clevers H, et al. Redundant regulation of T cell differentiation and TCRalpha gene expression by the transcription factors LEF-1 and TCF-1. Immunity. 1998;8:11–20. doi: 10.1016/s1074-7613(00)80454-9. [DOI] [PubMed] [Google Scholar]
- 39.Staal FJ, Meeldijk J, Moerer P, Jay P, van de Weerdt BC, et al. Wnt signaling is required for thymocyte development and activates Tcf-1 mediated transcription. Eur J Immunol. 2001;31:285–293. doi: 10.1002/1521-4141(200101)31:1<285::AID-IMMU285>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 40.Galceran J, Miyashita-Lin EM, Devaney E, Rubenstein JL, Grosschedl R. Hippocampus development and generation of dentate gyrus granule cells is regulated by LEF1. Development. 2000;127:469–482. doi: 10.1242/dev.127.3.469. [DOI] [PubMed] [Google Scholar]
- 41.Babij P, Zhao W, Small C, Kharode Y, Yaworsky PJ, et al. High bone mass in mice expressing a mutant LRP5 gene. J Bone Miner Res. 2003;18:960–974. doi: 10.1359/jbmr.2003.18.6.960. [DOI] [PubMed] [Google Scholar]
- 42.Akhter MP, Wells DJ, Short SJ, Cullen DM, Johnson ML, et al. Bone biomechanical properties in LRP5 mutant mice. Bone. 2004;35:162–169. doi: 10.1016/j.bone.2004.02.018. [DOI] [PubMed] [Google Scholar]
- 43.Sawakami K, Robling AG, Ai M, Pitner ND, Liu D, et al. The Wnt co-receptor LRP5 is essential for skeletal mechanotransduction but not for the anabolic bone response to parathyroid hormone treatment. J Biol Chem. 2006;281:23698–23711. doi: 10.1074/jbc.M601000200. [DOI] [PubMed] [Google Scholar]
- 44.Wu Y, Torchia J, Yao W, Lane NE, Lanier LL, et al. Bone microenvironment specific roles of ITAM adapter signaling during bone remodeling induced by acute estrogen-deficiency. PLoS ONE. 2007;2:e586. doi: 10.1371/journal.pone.0000586. doi:10.1371/journal.pone.0000586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hill TP, Spater D, Taketo MM, Birchmeier W, Hartmann C. Canonical Wnt/beta-catenin signaling prevents osteoblasts from differentiating into chondrocytes. Dev Cell. 2005;8:727–738. doi: 10.1016/j.devcel.2005.02.013. [DOI] [PubMed] [Google Scholar]
- 46.DeMambro VE, Clemmons DR, Horton LG, Bouxsein ML, Wood TL, et al. Gender-specific changes in bone turnover and skeletal architecture in igfbp-2-null mice. Endocrinology. 2008;149:2051–2061. doi: 10.1210/en.2007-1068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pennypacker B, Shea M, Liu Q, Masarachia P, Saftig P, et al. Bone density, strength, and formation in adult cathepsin K (−/−) mice. Bone. 2008 doi: 10.1016/j.bone.2008.08.130. [DOI] [PubMed] [Google Scholar]
- 48.Hoeflich KP, Luo J, Rubie EA, Tsao MS, Jin O, et al. Requirement for glycogen synthase kinase-3beta in cell survival and NF-kappaB activation. Nature. 2000;406:86–90. doi: 10.1038/35017574. [DOI] [PubMed] [Google Scholar]
- 49.Liu XH, Kirschenbaum A, Yao S, Liu G, Aaronson SA, et al. Androgen-induced Wnt signaling in preosteoblasts promotes the growth of MDA-PCa-2b human prostate cancer cells. Cancer Res. 2007;67:5747–5753. doi: 10.1158/0008-5472.CAN-07-0478. [DOI] [PubMed] [Google Scholar]
- 50.Liu XH, Kirschenbaum A, Yao S, Levine AC. Androgens Promote Preosteoblast Differentiation via Activation of the Canonical Wnt Signaling Pathway. Ann N Y Acad Sci. 2007;1116:423–431. doi: 10.1196/annals.1402.017. [DOI] [PubMed] [Google Scholar]
- 51.Singh R, Artaza JN, Taylor WE, Braga M, Yuan X, et al. Testosterone inhibits adipogenic differentiation in 3T3-L1 cells: nuclear translocation of androgen receptor complex with beta-catenin and T-cell factor 4 may bypass canonical Wnt signaling to down-regulate adipogenic transcription factors. Endocrinology. 2006;147:141–154. doi: 10.1210/en.2004-1649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lyon MF, Hawkes SG. X-linked gene for testicular feminization in the mouse. Nature. 1970;227:1217–1219. doi: 10.1038/2271217a0. [DOI] [PubMed] [Google Scholar]
- 53.Lyon MF. Gene action in the X-chromosome of the mouse (Mus musculus L.). Nature. 1961;190:372–373. doi: 10.1038/190372a0. [DOI] [PubMed] [Google Scholar]
- 54.Vandenput L, Swinnen JV, Boonen S, Van Herck E, Erben RG, et al. Role of the androgen receptor in skeletal homeostasis: the androgen-resistant testicular feminized male mouse model. J Bone Miner Res. 2004;19:1462–1470. doi: 10.1359/JBMR.040505. [DOI] [PubMed] [Google Scholar]
- 55.Matsumoto C, Inada M, Toda K, Miyaura C. Estrogen and androgen play distinct roles in bone turnover in male mice before and after reaching sexual maturity. Bone. 2006;38:220–226. doi: 10.1016/j.bone.2005.08.019. [DOI] [PubMed] [Google Scholar]
- 56.Dubrow SA, Hruby PM, Akhter MP. Gender specific LRP5 influences on trabecular bone structure and strength. J Musculoskelet Neuronal Interact. 2007;7:166–173. [PubMed] [Google Scholar]
- 57.Yadav VK, Ryu JH, Suda N, Tanaka KF, Gingrich JA, et al. Lrp5 controls bone formation by inhibiting serotonin synthesis in the duodenum. Cell. 2008;135:825–837. doi: 10.1016/j.cell.2008.09.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Migliaccio A, Varricchio L, De Falco A, Castoria G, Arra C, et al. Inhibition of the SH3 domain-mediated binding of Src to the androgen receptor and its effect on tumor growth. Oncogene. 2007;26:6619–6629. doi: 10.1038/sj.onc.1210487. [DOI] [PubMed] [Google Scholar]
- 59.Kang HY, Cho CL, Huang KL, Wang JC, Hu YC, et al. Nongenomic androgen activation of phosphatidylinositol 3-kinase/Akt signaling pathway in MC3T3-E1 osteoblasts. J Bone Miner Res. 2004;19:1181–1190. doi: 10.1359/JBMR.040306. [DOI] [PubMed] [Google Scholar]
- 60.Smith E, Frenkel B. Glucocorticoids inhibit the transcriptional activity of LEF/TCF in differentiating osteoblasts in a glycogen synthase kinase-3beta-dependent and -independent manner. J Biol Chem. 2005;280:2388–2394. doi: 10.1074/jbc.M406294200. [DOI] [PubMed] [Google Scholar]
- 61.Nantermet PV, Xu J, Yu Y, Hodor P, Holder D, et al. Identification of genetic pathways activated by the androgen receptor during the induction of proliferation in the ventral prostate gland. J Biol Chem. 2004;279:1310–1322. doi: 10.1074/jbc.M310206200. [DOI] [PubMed] [Google Scholar]
- 62.Kahler RA, Galindo M, Lian J, Stein GS, van Wijnen AJ, et al. Lymphocyte enhancer-binding factor 1 (Lef1) inhibits terminal differentiation of osteoblasts. J Cell Biochem. 2006;97:969–983. doi: 10.1002/jcb.20702. [DOI] [PubMed] [Google Scholar]
- 63.Kahler RA, Westendorf JJ. Lymphoid enhancer factor-1 and beta-catenin inhibit Runx2-dependent transcriptional activation of the osteocalcin promoter. J Biol Chem. 2003;278:11937–11944. doi: 10.1074/jbc.M211443200. [DOI] [PubMed] [Google Scholar]
- 64.Mishina Y, Starbuck MW, Gentile MA, Fukuda T, Kasparcova V, et al. Bone morphogenetic protein type IA receptor signaling regulates postnatal osteoblast function and bone remodeling. J Biol Chem. 2004;279:27560–27566. doi: 10.1074/jbc.M404222200. [DOI] [PubMed] [Google Scholar]
- 65.Bajayo A, Goshen I, Feldman S, Csernus V, Iverfeldt K, et al. Central IL-1 receptor signaling regulates bone growth and mass. Proc Natl Acad Sci U S A. 2005;102:12956–12961. doi: 10.1073/pnas.0502562102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yirmiya R, Goshen I, Bajayo A, Kreisel T, Feldman S, et al. Depression induces bone loss through stimulation of the sympathetic nervous system. Proc Natl Acad Sci U S A. 2006;103:16876–16881. doi: 10.1073/pnas.0604234103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ruegsegger P, Koller B, Muller R. A microtomographic system for the nondestructive evaluation of bone architecture. Calcif Tissue Int. 1996;58:24–29. doi: 10.1007/BF02509542. [DOI] [PubMed] [Google Scholar]
- 68.Hildebrand T, Laib A, Muller R, Dequeker J, Ruegsegger P. Direct three-dimensional morphometric analysis of human cancellous bone: microstructural data from spine, femur, iliac crest, and calcaneus. J Bone Miner Res. 1999;14:1167–1174. doi: 10.1359/jbmr.1999.14.7.1167. [DOI] [PubMed] [Google Scholar]
- 69.Erlebacher A, Derynck R. Increased expression of TGF-beta 2 in osteoblasts results in an osteoporosis-like phenotype. J Cell Biol. 1996;132:195–210. doi: 10.1083/jcb.132.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, et al. Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res. 1987;2:595–610. doi: 10.1002/jbmr.5650020617. [DOI] [PubMed] [Google Scholar]
- 71.Leclerc N, Noh T, Cogan J, Samarawickrama DB, Smith E, et al. Opposing effects of glucocorticoids and Wnt signaling on Krox20 and mineral deposition in osteoblast cultures. J Cell Biochem. 2008;103:1938–1951. doi: 10.1002/jcb.21587. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Lef1 −/− mice have normal bone development. Histological evaluation of wild type (a–e) versus Lef1 −/− (f–j) newborn mice. (a,b,f,g) Alizarin red/Alcian blue staining of craniofacial bones. (c,h) Alizarin red/Alcian blue staining of hind limb and vertebrae. (d,i) H-E staining of longitudinal femoral sections. (e,j) Toluidine blue staining of distal femoral growth plates. Representative images are shown. No abnormality was detected in the Lef1 −/− skeletons, except for the previously reported lack of teeth (f) (van Genderen et al. 1994, Genes Dev 8, 2691-703)
(3.47 MB TIF)
DHT stimulates Lef1 expression in vitro. MC3T3-E1 osteoblast cultures maintained in phenol-red free αMEM supplemented with 10% charcoal-stripped serum were treated with 30 nM DHT or 100 nM estradiol for 48 hours. Expression of the four members of the Lef/Tcf gene family was assessed by RT-qPCR and corrected for the expression of rpL10A. Bars represent expression levels in the presence of hormone relative to the ethanol vehicle, defined for each gene as 1.
(0.88 MB TIF)