Abstract
Purpose
The aim of this study was to investigate the utility of apparent diffusion coefficient (ADC) for prediction and early detection of treatment response in head and neck squamous cell carcinomas (HNSCC).
Experimental Design
Diffusion weighted MRI studies were performed on 40 patients with newly diagnosed HNSCC before, during, and after the end of chemoradiation therapy. Analysis was performed on data from 33 patients after exclusion of 7 patients that had incomplete data.
Results
Pre-treatment ADC value of complete responders (CR, 1.04 ± 0.19 × 10−3 mm2/s) was significantly lower (p < 0.05) than that from partial responders (PR, 1.35 ± 0.30 × 10−3 mm2/s). A significant increase in ADC was observed in CR patients within one week of treatment (p < 0.01), which remained high until the end of the treatment. The CR patients also showed significantly higher increase in ADC than the PR patients by the first week of chemoradiation (p < 0.01). When pre-treatment ADC value was used for predicting treatment response, the area under the receiver operating characteristics curve (AUC) was 0.80 with a sensitivity of 65% and a specificity of 86%. However, change in ADC within the first week of chemoradiation therapy resulted in an AUC of 0.88 with 86% sensitivity and 83% specificity for prediction of treatment response.
Conclusions
These results suggest that ADC can be used as a marker for prediction and early detection of response to concurrent chemoradiation therapy in HNSCC.
Keywords: Head and neck, squamous cell carcinoma, diffusion weighted imaging, apparent diffusion coefficient, radiation therapy, treatment response
INTRODUCTION
Head and neck cancer represents approximately 5% of cancers diagnosed annually in the United States (1), and is more prevalent in developing countries to rank it as the sixth most common cancer in the world (2). These cancers predominately originate from mutations in the mucosal squamous cells and usually present as locoregional disease (3, 4). Treatment of head and neck squamous cell carcinoma (HNSCC) is challenging as the quality of life of the patient can be severely affected by possible functional losses (impaired swallowing and eating, speech deficit) as well as social losses due to cosmetic deformity from surgery.
Organ preserving definitive radiation therapy, typically with concurrent chemotherapy, has been accepted as a standard management option for patients with metastatic cervical nodes (1–6). Despite these rigorous treatment methods, the overall survival rate of these patients has not improved significantly as the 5 year survival rate of these patients remains below 50% (1, 5, 6). The treatment outcome may be improved by using an optimized treatment strategy tailor fitted to an individual patient based on imaging biomarkers (7). If the outcome can be predicted before or at an early stage of treatment, the patient could also be spared from ineffective and unnecessary toxicity. Magnetic resonance techniques, including proton spectroscopy (8), diffusion weighted imaging (DWI) (9–12), and dynamic contrast enhanced imaging (13) have been proposed as such non-invasive imaging biomarkers for prediction and early detection of response to cancer therapy.
DWI has been suggested as the modality of choice for early detection of treatment response in tumors (9, 10, 14–16). In a recent study, it was reported that in comparison to spin echo MRI or positron emission tomography (PET), ADC values resulted in lower false positives for lesions at the primary site and persistent nodal disease in the post radiation therapy period (17). However, the efficacy of pre-treatment ADC values in prediction or for detection of early treatment response (within one to two weeks of chemo-radiotherapy) in HNSCC has not been reported. Accurate and timely detection of treatment response or presence of non-responsive tumor can be critical in disease management since the optimal time window for successful surgery or alternative treatment methods may be limited. This study was therefore conducted to investigate the efficacy of ADC in prediction and early detection of treatment response of HNSCC. ADC from the metastatic node was measured three times (prior to, during and at the end of chemoradiation therapy).
MATERIALS AND METHODS
Patient Population and Treatment
The institutional review board approved this study, and written informed consent was obtained from all 40 subjects before the MRI studies (9 females and 31 males, age =59.8 ± 10.8 years, recruited between January 2005 and October 2007) who were newly diagnosed with HNSCC with no prior treatment and referred for pre-operative chemoradiation therapy. All patients were assessed by a radiation oncologist by clinical reports and a physical exam and had palpable metastatic cervical lymph node masses. Patients received accelerated radiation treatment with 220 cGy per fraction for a total dose of 7040 cGy to the gross tumor volume in 32 fractions over a total of 44 days, with concurrent chemotherapy (cisplatin, 100 mg/m2 on days 1, 22 and 43 of radiation treatment, n=33) or immunotherapy (cetuximab, 400 mg/m2 3–7 days prior to radiation therapy, and then 250 mg/m2 on days 1, 8, 15, 22, 29, 36 and 43 of radiation treatment, n=7). Seven patients were excluded from the study because of death unrelated to the treatment (n = 4), claustrophobia (n = 1), withdrawal by patient (n = 1), and severe dental artifacts (n = 1). Hence, analysis of MRI data was performed for 33 patients (7 females and 26 males, age =61.0 ± 10.8 year old).
The median follow up time for the patients was 12 months (range: 6–24 months). Although a post-treatment follow up of six months or longer may impact the long term or overall survival, the current study was focused on assessing local control of the metastatic node. Additional therapeutic strategies performed after the end of chemoradiation therapy for potential partial/non-responders including surgery and chemotherapy make it difficult to assess the role of neo-adjuvant chemoradiation therapy. Thus, the status at the end of chemoradiation therapy was used as the clinical end point in this study. The criterion for a complete responder (CR) was absence of viable tumor on pathology or determination of complete response based on clinical/radiological assessment if surgery was not performed. All partial responders (PR) were confirmed by the presence of viable tumor on pathology. Individual patient data is shown in Table 1.
Table 1.
Summary of patient information
| Patient No. |
Sex | Age | Primary Site | Tumor Staging |
Concurrent systemic therapy |
Post-Tx Surgery |
Pathology | 6-month follow-up |
|---|---|---|---|---|---|---|---|---|
| 1 | M | 51 | vallecula | T2N2cM0 | chemo | Y | positive | DM* |
| 2 | M | 76 | tonsil | TxN2bM0 | chemo | Y | positive | DM |
| 3 | M | 75 | base of tongue | T4N2cM0 | chemo | Y | negative | NED** |
| 4 | M | 42 | tonsil | T2N2bM0 | chemo | Y | negative | NED |
| 5 | F | 53 | tonsil | TxN2bM0 | chemo | Y | negative | NED |
| 6 | M | 50 | base of tongue | T2N2aM0 | chemo | Y | negative | NED |
| 7 | M | 58 | base of tongue | T3N2cM0 | chemo | Y | negative | NED |
| 8 | M | 76 | larynx | T2N2bM0 | chemo | Y | positive | NED |
| 9 | M | 56 | larynx | T2N3M0 | chemo | Y | positive | DM |
| 10 | M | 31 | larynx | T4aN2bM0 | chemo | N | NED | |
| 11 | M | 72 | unknown primary | TxN2bM0 | chemo | N | NED | |
| 12 | F | 78 | unknown primary | T4aN2bM0 | immuno | N | NED | |
| 13 | F | 68 | base of tongue | T4N2bM0 | chemo | N | DM | |
| 14 | M | 49 | tonsil | T3N2aM0 | immuno | Y | negative | NED |
| 15 | M | 61 | base of tongue | T3N2aM0 | immuno | Y | negative | NED |
| 16 | M | 51 | base of tongue | TxN2bM0 | chemo | Y | negative | NED |
| 17 | M | 64 | unknown primary | TxN3M0 | chemo | Y | positive | DM |
| 18 | M | 55 | tonsil | T3N2bM0 | chemo | Y | negative | NED |
| 19 | M | 60 | tonsil | T4N1M0 | chemo | Y | positve | NED |
| 20 | F | 55 | larynx | T3N2cM0 | chemo | Y | negative | NED |
| 21 | F | 62 | tonsil | T2N2cM0 | chemo | Y | negative | NED |
| 22 | M | 65 | unknown primary | TxN2bM0 | chemo | Y | negative | NED |
| 23 | M | 63 | larynx | T2N2bM0 | chemo | Y | positve | NED |
| 24 | F | 48 | tonsil | T4bN2bM0 | immuno | N | Relapse | |
| 25 | M | 72 | base of tongue | T4N2cM0 | immuno | N | NED | |
| 26 | F | 61 | larynx | TxN2aM0 | chemo | Y | negative | NED |
| 27 | M | 67 | unknown primary | T0N2bM0 | immuno | N | NED | |
| 28 | M | 52 | base of tongue | T1N2abM0 | chemo | Y | negative | NED |
| 29 | M | 67 | tonsil | T2N2bM0 | chemo | N | NED | |
| 30 | M | 70 | tonsil | T4N1aM0 | chemo | N | NED | |
| 31 | M | 67 | base of tongue | T4aN2cM0 | chemo | N | NED | |
| 32 | M | 59 | base of tongue | T4N2bM0 | chemo | Y | negative | NED |
| 33 | M | 72 | base of tongue | T2N2bM0 | immuno | N | NED |
DM: Distant Metastasis
NED: No Evidence of Disease
All patients underwent three MRI studies, before treatment (Pre-Tx), one week after radiation therapy (Wk1-Tx), and about two weeks after the completion of the treatment (Post-Tx). While it was difficult to keep the exact timing of serial MRI scans for all patients, all efforts were made to minimize this variability. The Wk1-Tx scans were conducted at 11.8 ± 3.7 and 11.4 ± 2.9 days after initiation of treatment for CR and PR groups with no significant difference (p=0.81, two-tailed t-test with unequal variance). Post-Tx scans were performed at 18.0 ± 8.3 and 10.4 ± 10.6 days after completion of treatment for CR and PR groups, respectively, also with no significant difference (p=0.19). The difference in Post-Tx scan time points was probably due to the large variability in clinical condition of the patient by the end of the treatment. It was assumed that the ADC of the residual nodal masses did not have any substantial change within the range of our Post-Tx scan time points.
Data Acquisition
The MRI study was performed using a 1.5T Siemens Sonata scanner (n = 24) or a 3T Siemens Trio scanner (n = 9) (Siemens Medical Systems, Iselin, NJ). A neck array coil or a neurovascular coil was used for 1.5T or 3T scanners, respectively. T2 weighted (T2w) and T1 weighted (T1w) axial images were acquired using a spin echo sequence (TR/TE = 4 s/120 ms for T2w, and TR/TE = 600 ms/10 ms for T1w). The metastatic nodal masses were identified by a head and neck radiologist on cross sectional imaging at each MRI scan, and were used as primary imaging targets. The metastatic nodes were used instead of the primary tumor because they are less sensitive to artifacts induced by continuous physiological motion, such as breathing and swallowing, as well as susceptibility artifacts.
Eight axial slices with an FOV = 26 cm and slice thickness = 5 mm were selected to cover the metastatic cervical lymph node for T2 and ADC measurement. Quantitative T2 measurement was performed by acquiring a series of T2 weighted images using a spin echo imaging sequence with four different echo times; 13, 53, 80, and 110 ms (TR = 2 s). Diffusion weighted images were acquired using a pulsed gradient spin echo (PGSE) / echo planar imaging (EPI) sequence (TR/TE = 4 s/89 ms, 4 averages) with three b-values; 0, 500, and 1000 s/mm2. Huisman et al has earlier reported that ADC values from the brain are field dependent (18), however, no such studies have been performed on the head and neck. Thus, we performed ADC experiments on the neck region of three healthy controls at both 1.5T and 3T scanners within one hour and the ADC values from the sub-mandibular glands were compared within each individual from both scanners. For the patient studies, all three scans from each patient were performed on the same scanner to reduce any differences due to the magnetic field used. As part of the clinical protocol, contrast enhanced imaging was performed using a single dose of Gd-DTPA (Omniscan; Nycomed) at a concentration of 0.1 mM/kg body weight. The contrast agent was injected at 1 mL/s into an antecubital vein, followed by saline flush with a power injector (Medrad, Idianola, PA). After 10 minutes, another set of T1 weighted (T1w-Gd) axial images were acquired using a gradient echo sequence (TR/TE = 300 ms/4 ms, flip angle = 90°).
Data Analysis
Regions of interest (ROI) for metastatic nodal mass were drawn by a neuroradiologist (LL) based on multi-slice T1w, T2w, and T1w-Gd images. These ROIs were used to measure total tumor volume, T2 and ADC values of the node. Since the images of the head and neck region are subject to voluntary as well as involuntary motion, the images were not aligned well with each other in most cases. Thus, prior to data analysis, all images were co-registered to the spin echo images with a TE=80 ms using a two step non-rigid image registration technique. The first step involved a 3D registration with affine transformation to minimize global misalignment. Subsequently, each slice was co-registered using a 2D non-rigid registration with 2nd order discrete sine bases (19); for both x- and y-axes. Mutual information (20) was used as the cost function for both co-registration steps. T2 and ADC values were estimated on a pixel-by-pixel basis by fitting a mono-exponential function to T2 weighted and diffusion weighted data, respectively. The median value of the selected ROI was calculated for each parameter. In addition to the volume, T2, and ADC at each time point, change in each parameter by Wk1-Tx or Post-Tx was measured by normalizing each parameter to the corresponding Pre-Tx value for individual patient. The difference between the two treatment response groups (CR and PR) was assessed using Mann-Whitney U Test (21). The receiver operating characteristic (ROC) analysis (22) was used to evaluate the efficacy of ADC as a predictive marker for response to chemoradiation therapy. Data analysis tools were implemented using IDL routines (ITT Visual Information Solutions, Boulder, CO).
RESULTS
Response to treatment was determined at the end of chemoradiotherapy, based on clinical (n=11) or pathological (n=22) assessment, as shown in Table 1. The patients were categorized as complete responders (CR, with no evidence of disease, n=26), or partial responders (PR, with evidence of residual disease, n=7). All PR cases were confirmed by pathology for the presence of a viable tumor from the surgically removed nodes. 57 % (n=4) of the PR group presented with metastasis within six months after complete dissection of the remaining nodes. In contrast, only one patient from the CR group developed distant metastasis and one exhibited local relapse at the six month follow-up (8%, n=2). This observation indicates that the treatment response assessed at the end of neoadjuvant chemoradiation therapy strongly correlates with the results of six month follow up.
Three healthy volunteers were scanned at both 1.5T and 3T scanners to test the magnetic field dependency of ADC. ROI’s were drawn manually on two slices from the left and right submandibular glands. The measured ADC values are provided in Supplemental Table 1. The ADCs values measured at 3T were only 4.6±2.7% higher than those measured at 1.5T without statistical significance (p > 0.05 in all cases). As these results showed a field independent role of ADC values, we combined the data from patients scanned at the two magnets for group comparisons.
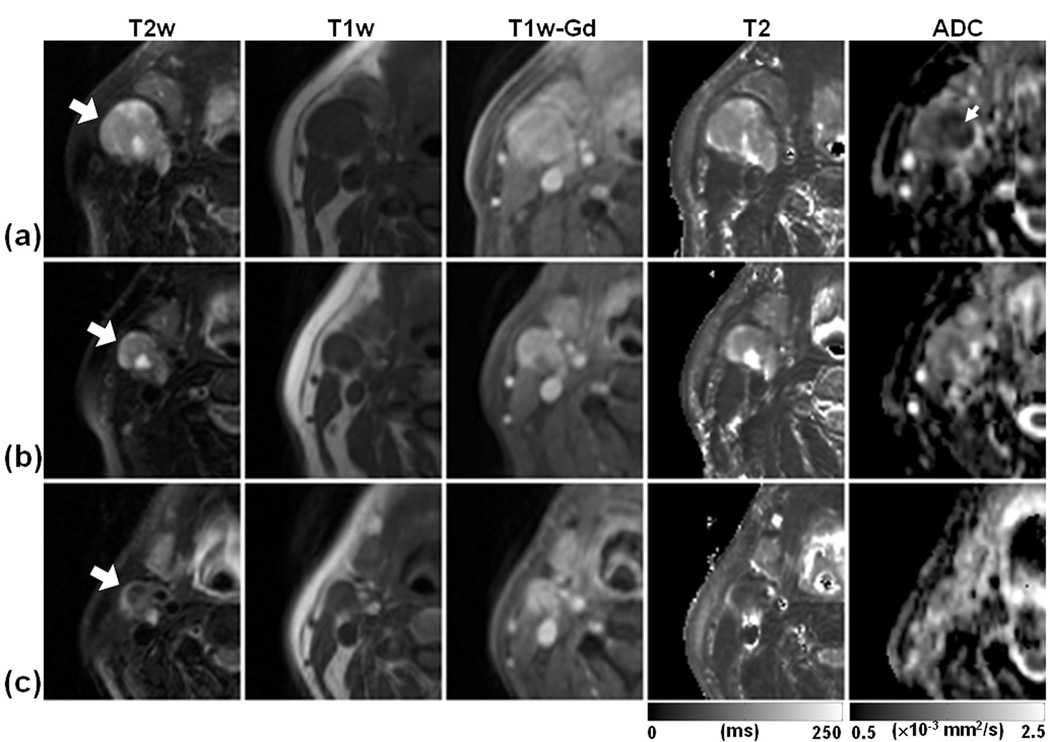
Figure 1 shows representative images from the central section of a metastatic node of a CR patient (primary at base of tongue) at three imaging time points; Pre-Tx (Fig. 1a), Wk1-Tx (Fig. 1b), and Post-Tx (Fig. 1c). The nodal mass (indicated by large arrows) was hyper-intense on T2w image, hypo-intense on T1w image and exhibited enhancement on Gd-DTPA enhanced T1w image. T2 values of the node were 152.6 ± 56.6 ms, 149.7 ± 191.2 ms, and 97.6 ± 26.3 ms for Pre-Tx, Wk1-Tx, and Post-Tx time points, respectively. The central part of the tumor (indicated by a small arrow) had a lower ADC value than the outer region. ADC values of the node were 1.06 ± 0.28 ×10−3 mm2/s, 1.34 ± 0.28 ×10−3 mm2/s, and 1.49 ± 0.40 ×10−3 mm2/s for Pre-Tx, Wk1-Tx, and Post-Tx, respectively. The residual mass was negative for the presence of viable tumor on histopathology performed after surgery. There has been no evidence of a recurrence of disease in this patient as of the writing of this manuscript (17 months after the end of chemoradiation therapy).
Figure 1.
Representative images of a patient who exhibited complete response to treatment as no viable tumor was found at surgery. The images in each row are from three measurement time points; Pre-Tx (a), Wk1-Tx (b), and Post-Tx (c). The T2w, T1w, and T1w-Gd images were windowed to have similar image contrast, whereas T2 and ADC images were scaled based on the gray scale bars shown at the bottom of the corresponding images. Large arrows indicate the same nodal metastatic mass that was followed through the treatment course. Small arrow indicates the central region of the mass with lower ADC values than the peripheral region.
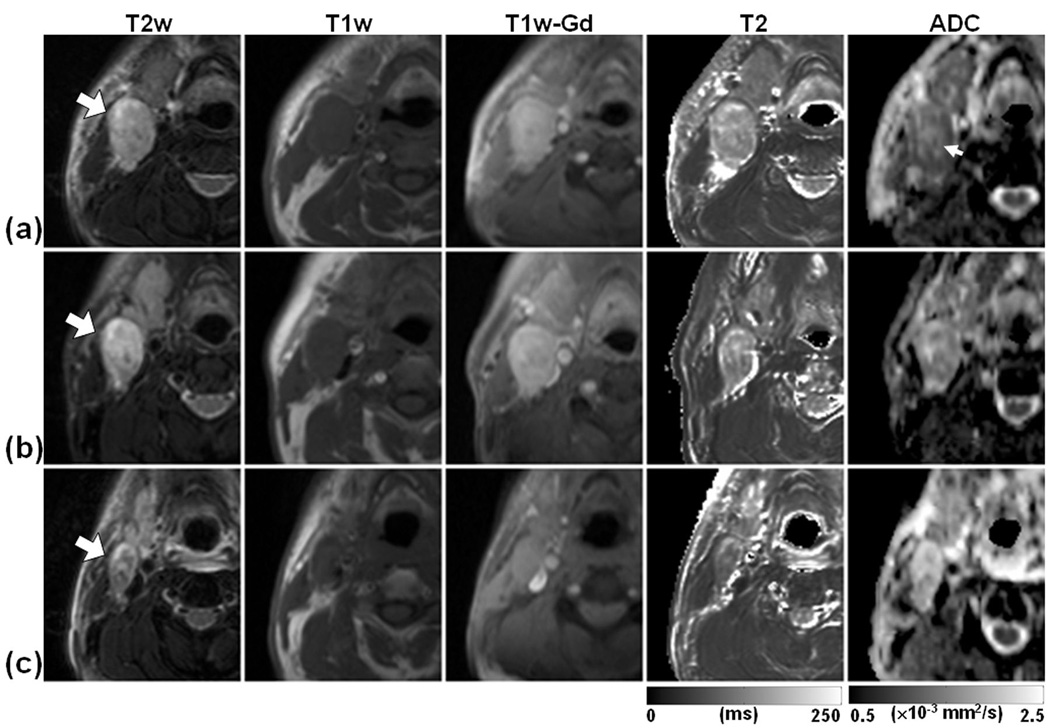
Figure 2 demonstrates a case with PR (primary at epiglottic vallecula) in which residual viable tumor was confirmed histopathologically. The node had a similar image contrast on T2w, T1w, and T1w-Gd images as in the case depicted in Figure 1, indicating the limited sensitivity of these techniques in differentiating the two cases. T2 values were 150.0 ± 30.6 ms, 141.5 ± 57.4 ms, and 109.8 ± 26.1 ms at Pre-Tx, Wk1-Tx, and Post-Tx, respectively. ADC values were 1.22 ± 0.23 ×10−3 mm2/s, 1.47 ± 0.21 ×10−3 mm2/s, and 1.79 ± 0.26× 10−3 mm2/s at Pre-Tx, Wk1-Tx, and Post-Tx, respectively. In contrast to the ADC map of the first case shown in Figure 1, the central part of the mass (indicated by a small arrow) has higher ADC values than the outer region. This patient died with disease (metastasis to lung) nine months after the completion of the chemoradiation therapy.
Figure 2.
Representative images of a patient who exhibited partial response to treatment as evident by the presence of viable tumor at surgery. The images in each row are from three measurement time points; Pre-Tx (a), Wk1-Tx (b), and Post-Tx (c). The T2w, T1w, and T1w-Gd images were windowed to have similar image contrast, whereas T2 and ADC images were scaled based on the gray scale bars shown at the bottom of the corresponding images. The large arrows indicate the same metastatic nodal mass that was followed through the treatment course. The small arrow indicates the central region of the mass with ADC higher than the rim.
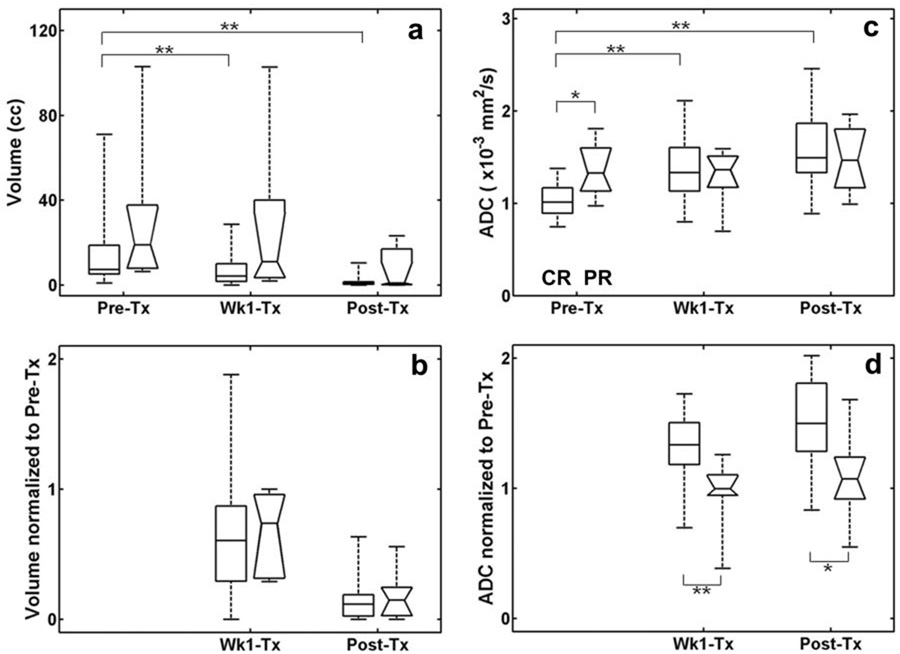
A summary of data from all patients is shown by the box-whisker plots in Fig. 3 as a comparison between CR and PR groups in terms of tumor volume and ADC values. For some patients, the tumor shrank dramatically during the treatment such that it was not possible to reliably draw an ROI to measure any MRI parameter. Thus, as indicated in the figure caption, the number of available patient data became smaller at Wk1-Tx and Post-Tx compared with Pre-Tx. In addition to ADC, the nodal volumes of the CR and PR groups were compared at three time points to evaluate the efficacy of nodal volume as a predictive marker for treatment response. Fig. 3a indicates that the median nodal volume of the PR group is higher than those of the CR group. However, no significant differences were found between CR and PR groups at any time point (p > 0.05). The median volumes decreased significantly in the CR group at Wk1-Tx and Post-Tx (p < 0.01) as compared to the volumes measured Pre-Tx. To assess the treatment effect on each patient individually, post-treatment data (Wk1-Tx and Post-Tx) were normalized to the respective pre-treatment values. There was no significant difference in the normalized nodal volumes between the CR and PR groups (Fig. 3b). Figure 3c shows that there was a significant difference (p < 0.05) between median ADC values of CR (1.04 ± 0.19 ×10−3 mm2/s) and PR (1.35 ± 0.30 ×10−3 mm2/s) patients at Pre-Tx time point. The median ADC values of CR at Wk1-Tx and Post-Tx were also significantly higher (p < 0.01) than the Pre-Tx values, whereas there was no significant change in the median ADC values of PR during treatment. In terms of the relative change in ADC from each patient, CR patients exhibited a significantly higher (p < 0.01 for Wk1-Tx, p < 0.05 for Post-Tx) change in ADC than PR (Fig. 3d).
Figure 3.
Comparison of CR (rectangular boxes) and PR (boxes with notches) groups from all patients scanned using the 1.5T and 3T scanner, in terms of volume (a), normalized volume (b), ADC (c), and normalized ADC (d). The edges represent the 25th and 75th percentiles and the middle lines in the boxes are the median values. The whisker lines indicate the minimum and maximum values observed. The number of patients for CR group was 26, 22, and 17 for Pre-Tx, Wk1-Tx, and Post-Tx, respectively. The number of patients for PR group was 7, 6, and 5 for Pre-Tx, Wk1-Tx, and Post-Tx, respectively. * represents significant difference with p < 0.05 and ** with p < 0.01.
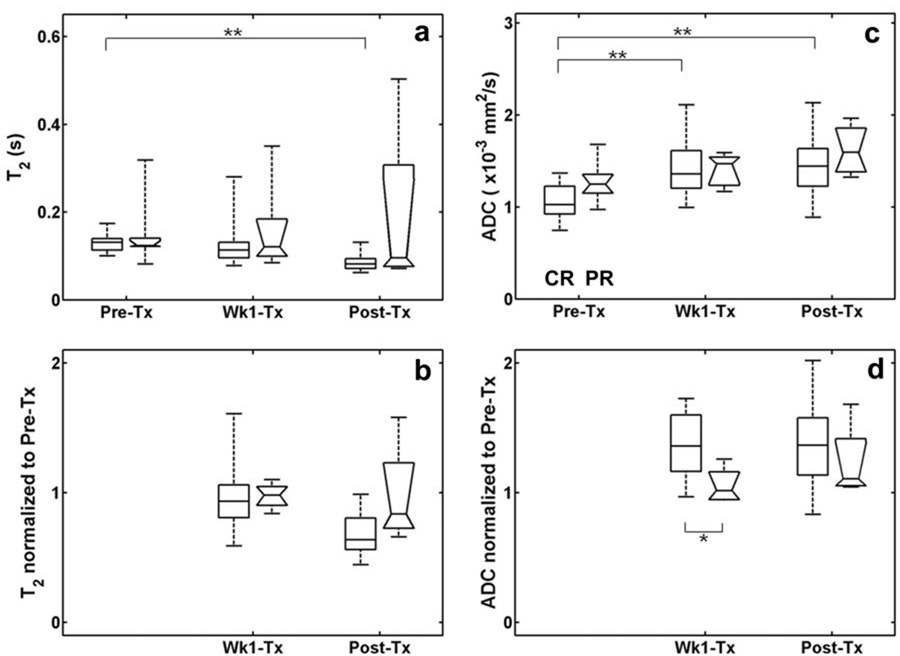
Since T2 values are dependent on the magnetic field strength used, the analysis of T2 values from CR and PR groups was performed separately for each magnet (1.5T and 3T). For comparison, a similar analysis was also performed for ADC values. Figure 4 shows a comparison of T2 and ADC values between the CR and PR groups measured at 1.5T. No significant difference between the CR and PR groups was observed at any time point without (Fig. 4a) or with (Fig. 4b) normalization to Pre-Tx value. For T2 values, a significant difference (p < 0.01) was found only in the CR group between Pre-Tx and Post-Tx time points. For the same patient population, the median ADC of the CR group increased significantly (p < 0.01) at Wk1-Tx and Post-Tx (Fig. 4c). The normalized Wk1-Tx ADC values were also significantly higher (p < 0.05) in CR than in PR. These results were similar to the observation when data from both magnets were combined, indicating the field independence of ADC (Fig. 4d). The CR patients scanned at 3T also showed similar results; however, since only one PR patient was scanned at 3T, no statistical comparison was attempted between the CR and PR groups.
Figure 4.
Comparison of CR (rectangular boxes) and PR (boxes with notches) groups from the patients scanned using the 1.5T scanner, in terms of T2 (a), normalized T2 (b), ADC (c), and normalized ADC (d). The edges represent the 25th and 75th percentiles and the middle lines in the boxes are the median values. The whisker lines indicate the minimum and maximum values. The number of patients for CR group was 18, 16 and 12 for Pre-Tx, Wk1-Tx, and Post-Tx, respectively. The number of patients for PR group was 6, 5, and 4 for Pre-Tx, Wk1-Tx, and Post-Tx, respectively. * represents significant difference with p < 0.05 and ** with p < 0.01.
As shown in Table 1, seven patients received EGFR targeted immunotherapy and they all showed complete response at the end of therapy. Since only a small number of patients received immunotherapy, it is difficult to assess the prognostic value of diffusion for this particular type of therapy. Within the complete responders, there was no significant (p>0.05) difference in ADC between patients receiving chemotherapy or immunotherapy at all three imaging time points. Within the patients who received conventional chemotherapy a similar trend was observed in that there was a significant difference in Pre-Tx ADC values between complete and partial responders (p<0.05) and a significant (p<0.01) increase of ADC in CR (Supplemental Figure 1).
The accuracy of ADC values in differentiating CR from PR patients was tested using binary classification and receiver operating characteristic (ROC) analyses. The results are summarized in Table 2 with a cutoff value for maximizing the Youden’s index (= sensitivity + specificity - 1). As shown in Table 2, the best test accuracy was achieved by the normalized ADC value at Wk1-Tx with a sensitivity of 86.4% and specificity of 83.3%.
Table 2.
Diagnostic characteristic of ADC for differentiating treatment response. The cutoff value was selected to maximize the sum of sensitivity and specificity. The numbers in the parentheses are the lower and upper limits of 95% confidence interval.
| Parameter | Cutoff value | Sensitivity (%) | Specificity (%) | AUC* |
|---|---|---|---|---|
| Pre-Tx | 1.11×10−3 mm2/s | 65 (44, 82) | 86 (42, 99) | 0.80 (0.62, 0.99) |
| Wk1-Tx/Pre-Tx | 1.11 | 86 (64, 96) | 83 (36, 99) | 0.88 (0.74, 1.00) |
| Post-Tx/Pre-Tx | 1.11 | 82 (56, 95) | 80 (30, 99) | 0.80 (0.57, 1.00) |
AUC: Area under receiver operating characteristic curve
DISCUSSION
In this study we investigated the efficacy of ADC for prediction and early detection of treatment response to chemoradiation therapy in HNSCC. The patients who responded favorably to chemoradiation therapy had significantly lower pre-treatment ADC than partial/non-responders. In addition, the change in ADC, as compared to the pre-treatment value, after the first week of chemoradiation therapy, showed the highest test accuracy along with a high sensitivity and specificity of separating complete responders from partial responders. These results suggest that ADC can be used as a predictive biomarker for therapeutic response in HNSCC and can thus aid in guiding therapeutic options for patients with HNSCC.
The potential of ADC in diagnosis of head and neck cancers has been reported earlier (23–25). Wang et al (23) reported that the mean ADC from malignant lesions was significantly smaller than that of benign solid or cystic lesions. It has also been reported that DWI is useful in discriminating metastatic lymph nodes from benign lymphadenopathy or nodal lymphomas (23–25), while T1 and T2 weighted images were not specific in differentiating the two pathologies (26). Furthermore, it has been demonstrated that ADC could differentiate radiation response from recurrent tumors (17, 27). In a recent study, it was reported that in comparison to spin echo MRI or positron emission tomography (PET), ADC values resulted in lower false positives for lesions at the primary site and persistent nodal disease in the post radiation therapy period (17). However, the efficacy of ADC in predicting or detecting treatment response in the head and neck cancer has not been reported earlier.
Pre-clinical studies in brain tumor models have demonstrated the sensitivity of DWI in detecting early changes induced by chemotherapy (9) or retrovirus-mediated gene therapy (10). Clinical studies on the efficacy of ADC for prediction or early detection of treatment response have been reported for brain tumor (16), breast cancer (12), and cervical cancer (28). As a biomarker for treatment response, Moffat et al (16) proposed the use of the fractional volume of significantly increased ADC within the tumor after the first 3 weeks of treatment. This method of analysis was termed as functional diffusion mapping (fDM). It is important to emphasize that in comparison to these published studies, we observed a predictive power of ADC even before initiation of treatment and that the sensitivity of early response assessment was highest at one week after chemoradiation treatment. Recent studies on a preclinical model of metastatic prostate cancer to the bone (11) and breast cancer (12) also demonstrated significantly increased ADC after 1 week into chemotherapy. Taken together, these studies and our results suggest that the sensitivity of ADC in detecting early therapeutic response depends on the type of cancer, treatment method, and imaging protocol. Nevertheless, the sensitivity of ADC over T2 in detecting response has been reported consistently (10) as observed in this study.
While the fDM analysis has been used to assess treatment response in brain tumors (16), it is difficult to implement it for organs outside the brain where co-registration of MR images acquired at different time points may be problematic due to differences in orientation of the images and artifacts induced by cardiac and respiratory motion. Thus, we used the median value of the whole tumor and changes therein during treatment. McVeigh et al (28) used a similar approach and reported that the pre-treatment ADC values (90th percentile) were significantly lower in responders than non-responders in patients with cervical cancer. This is consistent with our observation as differences between the two groups remained significant when tested with the 75th percentiles instead of the median values (data not shown).
Since ADC values from the extracellular and intracellular spaces are similar in magnitude (29), an increase in ADC after treatment indicates a substantial decrease in restriction of water diffusion within the extracellular space, intercellular space, or both. Gupta et al (30) reported a lower ADC in malignant brain tumors than benign tumors which correlated with histological measures of higher cell density in malignant tumors. Chinnaiyan et al (31) also reported that a change in ADC was associated with increased number of apoptotic cells and loss of cellularity during apoptosis-induced cancer therapy. Although the mechanism underlying increased water diffusion following cytotoxic chemotherapy in experimental and human tumors is not fully understood, it has been reported that this phenomenon coincides with reduced cell density and enlarged extracellular space due to apoptosis or necrosis (32). Thus, the observed increase in ADC in our study appears to be in line with the expected effect of successful treatment.
Our results with HNSCC are in agreement with a study in cervical cancer (28) in that the pre-treatment ADC of complete responders was lower than the partial responders. Since a negative correlation between ADC and cell density has been reported (30), it appears that viable cells in the highly proliferating solid tumor (lower ADC) have a better outcome of response to chemoradiation than tumors that have higher ADC (possibly including areas of necrosis). This may be related to a better perfusion of the actively proliferating solid tumor which helps delivery of cytotoxic drugs as well as oxygen during radiation therapy. This hypothesis is supported by recent diffusion and perfusion studies (33, 34) in which it was reported that the contrast enhancing regions of high grade gliomas have higher blood volume and lower ADC than the low grade gliomas (33). Recent CT perfusion studies from patients with esophageal squamous cell carcinomas also indicate higher pre-treatment blood flow and blood volume in complete responders than partial responders (34, 35).
Current methods for predicting or detecting tumor response include fluorodeoxyglucose (FDG) PET (36), PET/CT (37), proton (38) or phosphorous (39) MR spectroscopy, and dynamic contrast enhanced (DCE) MRI (40). Since these methods have been reported on various tumor types using different treatment regimens, it is difficult to make a direct comparison of the results of our study with these methods in terms of its efficacy for prediction and detection of treatment response. However, we believe that the advantage of ADC over PET or DCE-MRI is that it does not require injection of an isotope or any contrast agent. The acquisition time for ADC values is about 2 to 3 minutes, which is much shorter than PET, DCE-MRI or MR spectroscopy. In addition to its simplicity in estimating ADC from the DWI data, the ADC value is quantitative and magnetic field independent such that it is one of the most suitable metrics for multi-center and longitudinal studies.
While we have demonstrated the potential of ADC in clinical studies of HNSCC, it is challenging to routinely perform DWI in the head and neck due to susceptibility and motion artifacts. We have used a PGSE / single shot-EPI sequence as used in previously reported studies (16, 17, 24). The quality of the DWI data for the head and neck can be improved further by imaging sequences that are less sensitive to susceptibility artifacts, such as line scan DWI (25) or multi-shot sequence (41), or by using a special anti-susceptibility device on the neck (23). In order to perform a pixel-wise comparison of imaging parameters, images from different sequences (T1, T2, ADC etc) need to be co-registered with each other. Image misalignment between different modalities was minimized by using an image co-registration method in this study. However, we feel that this coregistration step could benefit further from improvement in image quality using ways to reduce susceptibility artifacts by methods discussed above as well as methods to account for artifacts induced by voluntary and involuntary motion. This study demonstrates the feasibility of ADC in predicting treatment response of HNSCC using a small cohort of patients recruited between January 2005 and October 2007. In this particular patient population, we had a smaller number of partial responders than complete responders, the effect of this difference on our finding is not known at the present time. Thus, further evaluation of the efficacy of the ADC for clinical application is warranted on a larger patient population.
The current study was performed to measure ADC in the metastatic lymph nodes. A similar analysis on tumors at the primary site would be of great interest. However, in comparison to the metastatic node, ADC measurements on the primary site are challenging as these tumors are generally located in areas prone to artifacts induced by continuous physiological motion such as breathing and swallowing. In addition, squamous cell carcinomas are located at the air-tissue interface, which increases the susceptibility artifacts on diffusion weighted images. Although in the present study, treatment response of only the metastatic node was assessed, it would be interesting to know as to whether the ADC values from the metastatic node can also predict overall treatment response. A preliminary assessment of this data can be made from Table 1, which shows the status of each patient at the six month follow up. While response assessment at the end of treatment was used as an end-point in the present study, it is interesting to note that for most patients the disease status remained unchanged at six-month follow up. Future studies would be required to assess the utility of ADC in predicting overall survival or long term disease free survival.
There is emerging evidence that suggests that squamous cell carcinomas may have biological differences due to differences in smoking and alcohol use as well as due to molecular alterations, such as EGFR expression and HPV infection status (1, 2). Due to the small sample size, the present study was not performed to address the role of ADC in separating these biological subtypes. However, it is interesting to note that despite these potential biological differences, our results show that ADC was sensitive in differentiating complete responders from partial/non-responders, indicating the robustness of diffusion imaging as a potential biomarker for prediction and early detection of treatment response.
It is currently difficult to predict which combination of treatment modalities will be best suited for any particular individual. Methods of assessing response to chemo-radiotherapy would be useful, as it would permit the oncologist to change therapies, either in type or degree, in cases when the subject does not respond to the initial therapy regimen. Development of noninvasive imaging biomarkers, such as ADC, will aid in accurately predicting and/or assessing treatment response and help in increasing the chance of successful treatment.
Supplementary Material
Comparison of CR (rectangular boxes) and PR (boxes with notches) groups from the patients who received concurrent chemotherapy, in terms of ADC (a) and normalized ADC (b). The edges represent the 25th and 75th percentiles and the middle lines in the boxes are the median values. The whisker lines indicate the minimum and maximum values. The number of patients for CR group was 19, 15 and 14 for Pre-Tx, Wk1-Tx, and Post-Tx, respectively. The number of patients for PR group was 7, 6, and 5 for Pre-Tx, Wk1-Tx, and Post-Tx, respectively. * represents significant difference with p < 0.05 and ** with p < 0.01.
Comparison of ADC values of the submandibular glands from normal healthy control subjects at 1.5T and 3T scanners.
Acknowledgments
Grant support: NIH Grant RO1-CA102756
Footnotes
Statement of Clinical Relevance
Organ preserving definitive radiation therapy, typically with concurrent chemotherapy, without a neck dissection has become an accepted standard management option for locally advanced head and neck squamous cell carcinoma (HNSCC) especially when patients present with metastatic cervical nodes. However, the survival rate of these patients has not improved significantly, raising a question if all patients with locally advanced HNSCC benefit from this treatment paradigm. With the development of organ preserving surgical techniques, the need to identify radiosensitive HNSCC is even more critical to better triage patients with resectable HNSCC. Apparent diffusion coefficient (ADC) measured by diffusion weighted MRI has been proposed as a marker for early response to treatment in brain tumor and breast cancers. However, its efficacy for prediction or detection of early treatment response in HNSCC has not been reported to date. This study was therefore conducted to investigate the efficacy of ADC for prediction and early detection of response to concurrent chemoradiation therapy in locally advanced HNSCC. Our results indicate that ADC can be effectively used as a noninvasive imaging marker for prediction and early detection of response to concurrent chemoradiation therapy in HNSCC.
REFERENCES
- 1.American Cancer Society. Cancer Facts & Figures 2007. Atlanta: American Cancer Society; 2007. [Google Scholar]
- 2.Nagpal JK, Das BR. Oral cancer: reviewing the present understanding of its molecular mechanism and exploring the future directions for its effective management. Oral oncology. 2003;39:213–221. doi: 10.1016/s1368-8375(02)00162-8. [DOI] [PubMed] [Google Scholar]
- 3.El-Deiry M, Funk GF, Nalwa S, et al. Long-term quality of life for surgical and nonsurgical treatment of head and neck cancer. Archives of otolaryngology--head & neck surgery. 2005;131:879–885. doi: 10.1001/archotol.131.10.879. [DOI] [PubMed] [Google Scholar]
- 4.Ang KK, Harris J, Garden AS, et al. Concomitant boost radiation plus concurrent cisplatin for advanced head and neck carcinomas: radiation therapy oncology group phase II trial 99-14. J Clin Oncol. 2005;23:3008–3015. doi: 10.1200/JCO.2005.12.060. [DOI] [PubMed] [Google Scholar]
- 5.Zorat PL, Paccagnella A, Cavaniglia G, et al. Randomized phase III trial of neoadjuvant chemotherapy in head and neck cancer: 10-year follow-up. Journal of the National Cancer Institute. 2004;96:1714–1717. doi: 10.1093/jnci/djh306. [DOI] [PubMed] [Google Scholar]
- 6.Bonner JA, Harari PM, Giralt J, et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. The New England journal of medicine. 2006;354:567–578. doi: 10.1056/NEJMoa053422. [DOI] [PubMed] [Google Scholar]
- 7.Ludwig JA, Weinstein JN. Biomarkers in cancer staging, prognosis and treatment selection. Nature reviews. 2005;5:845–856. doi: 10.1038/nrc1739. [DOI] [PubMed] [Google Scholar]
- 8.Tzika AA. Proton magnetic resonance spectroscopic imaging as a cancer biomarker for pediatric brain tumors (Review) International journal of oncology. 2008;32:517–526. doi: 10.3892/ijo_32_3_517. [DOI] [PubMed] [Google Scholar]
- 9.Chenevert TL, McKeever PE, Ross BD. Monitoring early response of experimental brain tumors to therapy using diffusion magnetic resonance imaging. Clin Cancer Res. 1997;3:1457–1466. [PubMed] [Google Scholar]
- 10.Poptani H, Puumalainen A-M, Grohn OH, Loimas S, Kainulainen R, Yla-Herttuala S, Kauppinen RA. Monitoring thymidine kinase and ganciclovir-induced changes in rat malignant glioma in vivo by nuclear magnetic resonance imaging. Cancer Gene Therapy. 1998;5:101–109. [PubMed] [Google Scholar]
- 11.Lee KC, Sud S, Meyer CR, et al. An imaging biomarker of early treatment response in prostate cancer that has metastasized to the bone. Cancer research. 2007;67:3524–3528. doi: 10.1158/0008-5472.CAN-06-4236. [DOI] [PubMed] [Google Scholar]
- 12.Lee KC, Moffat BA, Schott AF, et al. Prospective early response imaging biomarker for neoadjuvant breast cancer chemotherapy. Clin Cancer Res. 2007;13:443–450. doi: 10.1158/1078-0432.CCR-06-1888. [DOI] [PubMed] [Google Scholar]
- 13.Raatschen HJ, Simon GH, Fu Y, et al. Vascular Permeability during Antiangiogenesis Treatment: MR Imaging Assay Results as Biomarker for Subsequent Tumor Growth in Rats. Radiology. 2008 doi: 10.1148/radiol.2472070363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Galons JP, Altbach MI, Paine-Murrieta GD, Taylor CW, Gillies RJ. Early increases in breast tumor xenograft water mobility in response to paclitaxel therapy detected by non-invasive diffusion magnetic resonance imaging. Neoplasia (New York) 1999;1:113–117. doi: 10.1038/sj.neo.7900009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chenevert TL, Stegman LD, Taylor JM, et al. Diffusion magnetic resonance imaging: an early surrogate marker of therapeutic efficacy in brain tumors. Journal of the National Cancer Institute. 2000;92:2029–2036. doi: 10.1093/jnci/92.24.2029. [DOI] [PubMed] [Google Scholar]
- 16.Moffat BA, Chenevert TL, Lawrence TS, et al. Functional diffusion map: a noninvasive MRI biomarker for early stratification of clinical brain tumor response. Proc Natl Acad Sci U S A. 2005;102:5524–5529. doi: 10.1073/pnas.0501532102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vandecaveye V, De Keyzer F, Nuyts S, et al. Detection of head and neck squamous cell carcinoma with diffusion weighted MRI after (chemo)radiotherapy: correlation between radiologic and histopathologic findings. Int J Radiat Oncol Biol Phys. 2007;67:960–971. doi: 10.1016/j.ijrobp.2006.09.020. [DOI] [PubMed] [Google Scholar]
- 18.Huisman TA, Loenneker T, Barta G, et al. Quantitative diffusion tensor MR imaging of the brain: field strength related variance of apparent diffusion coefficient (ADC) and fractional anisotropy (FA) scalars. Eur Radiol. 2006;16:1651–1658. doi: 10.1007/s00330-006-0175-8. [DOI] [PubMed] [Google Scholar]
- 19.Ashburner J, Friston KJ. Nonlinear spatial normalization using basis functions. Hum Brain Mapp. 1999;7:254–266. doi: 10.1002/(SICI)1097-0193(1999)7:4<254::AID-HBM4>3.0.CO;2-G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Maes F, Collignon A, Vandermeulen D, Marchal G, Suetens P. Multimodality image registration by maximization of mutual information. IEEE Trans Med Imaging. 1997;16:187–198. doi: 10.1109/42.563664. [DOI] [PubMed] [Google Scholar]
- 21.Mann HB, Wihitney DR. On a test of whether one of two random variables is stochastically larger than the other. Annals of Mathematical Statistics. 1947;18:50–60. [Google Scholar]
- 22.Obuchowski NA. Receiver operating characteristic curves and their use in radiology. Radiology. 2003;229:3–8. doi: 10.1148/radiol.2291010898. [DOI] [PubMed] [Google Scholar]
- 23.Wang J, Takashima S, Takayama F, et al. Head and neck lesions: characterization with diffusion-weighted echo-planar MR imaging. Radiology. 2001;220:621–630. doi: 10.1148/radiol.2202010063. [DOI] [PubMed] [Google Scholar]
- 24.Sumi M, Sakihama N, Sumi T, et al. Discrimination of metastatic cervical lymph nodes with diffusion-weighted MR imaging in patients with head and neck cancer. AJNR Am J Neuroradiol. 2003;24:1627–1634. [PMC free article] [PubMed] [Google Scholar]
- 25.Maeda M, Kato H, Sakuma H, Maier SE, Takeda K. Usefulness of the apparent diffusion coefficient in line scan diffusion-weighted imaging for distinguishing between squamous cell carcinomas and malignant lymphomas of the head and neck. AJNR Am J Neuroradiol. 2005;26:1186–1192. [PMC free article] [PubMed] [Google Scholar]
- 26.King AD, Yuen EH, Lei KI, Ahuja AT, Van Hasselt A. Non-Hodgkin lymphoma of the larynx: CT and MR imaging findings. AJNR Am J Neuroradiol. 2004;25:12–15. [PMC free article] [PubMed] [Google Scholar]
- 27.Abdel Razek AA, Kandeel AY, Soliman N, et al. Role of diffusion-weighted echo-planar MR imaging in differentiation of residual or recurrent head and neck tumors and posttreatment changes. AJNR Am J Neuroradiol. 2007;28:1146–1152. doi: 10.3174/ajnr.A0491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McVeigh PZ, Syed AM, Milosevic M, Fyles A, Haider MA. Diffusion-weighted MRI in cervical cancer. Eur Radiol. 2008 doi: 10.1007/s00330-007-0843-3. [DOI] [PubMed] [Google Scholar]
- 29.Duong TQ, Ackerman JJ, Ying HS, Neil JJ. Evaluation of extra- and intracellular apparent diffusion in normal and globally ischemic rat brain via 19F NMR. Magn Reson Med. 1998;40:1–13. doi: 10.1002/mrm.1910400102. [DOI] [PubMed] [Google Scholar]
- 30.Gupta RK, Cloughesy TF, Sinha U, et al. Relationships between choline magnetic resonance spectroscopy, apparent diffusion coefficient and quantitative histopathology in human glioma. Journal of Neuro-Oncology. 2000;50:215–226. doi: 10.1023/a:1006431120031. [DOI] [PubMed] [Google Scholar]
- 31.Chinnaiyan AM, Prasad U, Shankar S, et al. Combined effect of tumor necrosis factor-related apoptosis-inducing ligand and ionizing radiation in breast cancer therapy. Proceedings of the National Academy of Sciences of the United States of America. 2000;97:1754–1759. doi: 10.1073/pnas.030545097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kauppinen RA. Monitoring cytotoxic tumour treatment response by diffusion magnetic resonance imaging and proton spectroscopy. NMR in Biomedicine. 2002;15:6–17. doi: 10.1002/nbm.742. [DOI] [PubMed] [Google Scholar]
- 33.Zonari P, Baraldi P, Crisi G. Multimodal MRI in the characterization of glial neoplasms: the combined role of single-voxel MR spectroscopy, diffusion imaging and echo-planar perfusion imaging. Neuroradiology. 2007;49:795–803. doi: 10.1007/s00234-007-0253-x. [DOI] [PubMed] [Google Scholar]
- 34.Hayano K, Okazumi S, Shuto K, et al. Perfusion CT can predict the response to chemoradiation therapy and survival in esophageal squamous cell carcinoma: initial clinical results. Oncol Rep. 2007;18:901–908. [PubMed] [Google Scholar]
- 35.Zima A, Carlos R, Gandhi D, Case I, Teknos T, Mukherji SK. Can pretreatment CT perfusion predict response of advanced squamous cell carcinoma of the upper aerodigestive tract treated with induction chemotherapy? AJNR Am J Neuroradiol. 2007;28:328–334. [PMC free article] [PubMed] [Google Scholar]
- 36.Weber WA. Positron emission tomography as an imaging biomarker. J Clin Oncol. 2006;24:3282–3292. doi: 10.1200/JCO.2006.06.6068. [DOI] [PubMed] [Google Scholar]
- 37.Weber WA, Figlin R. Monitoring cancer treatment with PET/CT: does it make a difference? J Nucl Med. 2007;48 Suppl 1:36S–44S. [PubMed] [Google Scholar]
- 38.Mukherji SK, Schiro S, Castillo M, Kwock L, Muller KE, Blackstock W. Proton MR spectroscopy of squamous cell carcinoma of the extracranial head and neck: in vitro and in vivo studies. AJNR Am J Neuroradiol. 1997;18:1057–1072. [PMC free article] [PubMed] [Google Scholar]
- 39.Shukla-Dave A, Poptani H, Loevner LA, et al. Prediction of treatment response of head and neck cancers with P-31 MR spectroscopy from pretreatment relative phosphomonoester levels. Academic radiology. 2002;9:688–694. doi: 10.1016/s1076-6332(03)80314-8. [DOI] [PubMed] [Google Scholar]
- 40.Thukral A, Thomasson DM, Chow CK, et al. Inflammatory breast cancer: dynamic contrast-enhanced MR in patients receiving bevacizumab--initial experience. Radiology. 2007;244:727–735. doi: 10.1148/radiol.2443060926. [DOI] [PubMed] [Google Scholar]
- 41.Deng J, Miller FH, Salem R, Omary RA, Larson AC. Multishot diffusion-weighted PROPELLER magnetic resonance imaging of the abdomen. Invest Radiol. 2006;41:769–775. doi: 10.1097/01.rli.0000236808.84746.95. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Comparison of CR (rectangular boxes) and PR (boxes with notches) groups from the patients who received concurrent chemotherapy, in terms of ADC (a) and normalized ADC (b). The edges represent the 25th and 75th percentiles and the middle lines in the boxes are the median values. The whisker lines indicate the minimum and maximum values. The number of patients for CR group was 19, 15 and 14 for Pre-Tx, Wk1-Tx, and Post-Tx, respectively. The number of patients for PR group was 7, 6, and 5 for Pre-Tx, Wk1-Tx, and Post-Tx, respectively. * represents significant difference with p < 0.05 and ** with p < 0.01.
Comparison of ADC values of the submandibular glands from normal healthy control subjects at 1.5T and 3T scanners.