Abstract
Sphingolipids (SLs) are essential constituents of eukaryotic cells. Besides playing structural roles in cellular membranes, some metabolites, including ceramide, sphingosine, and sphingosine-1-phosphate, have drawn attention as bioactive signaling molecules involved in the regulation of cell growth, differentiation, senescence, and apoptosis. Understanding the many cell regulatory functions of SL metabolites requires an advanced knowledge of how and where in the cell they are generated, converted, or degraded. This review will provide a short overview of the metabolism, localization, and compartmentalization of SLs. Also, a discussion on bioactive members of the SL family and inducers of SL enzymes that lead to ceramide generation will be presented.
Keywords: ceramide, bioactive lipids, compartmentalization
Sphingolipids (SLs) represent a major class of lipids that are ubiquitous constituents of membranes in eukaryotes. First discovered by J. L. W. Thudichum in 1876, for a long time SLs were considered to play primarily structural roles in membrane formation. However, intensive research on SL metabolism and function has revealed members of the SL family, including ceramide (Cer), sphingosine (Sph), Sph-1-phosphate (S1P), and Cer-1-phosphate (C1P), as bioactive molecules playing roles from regulation of signal transduction pathways, through direction of protein sorting to the mediation of cell-to-cell interactions and recognition. SLs have also been reported to dynamically cluster with sterols to form lipid microdomains or rafts, which function as hubs for effective signal transduction and protein sorting (1).
Various stimuli such as tumor necrosis factor-α (TNF-α), interleukin (IL)-1, Fas ligand, ionizing radiation (2), phorbol esters (3), heat stress (4), oxidative stress (5), and chemotherapeutics (6) induce the formation of Cer.
Understanding the complex regulatory mechanisms of bioactive SLs requires a broad knowledge of how these molecules are generated, degraded, or converted, and how and where they are located or metabolized. Recent molecular advances in identifying enzymes involved in the SL metabolism as well as technical advances in analytical measurements by mass spectrometry have shed light on their roles and regulation and involvement in cellular processes.
In this review, we will briefly summarize the current knowledge of these aspects. For more detailed insights, the reader is referred to recent references and reviews (7–9).
METABOLISM OF BIOACTIVE SLS
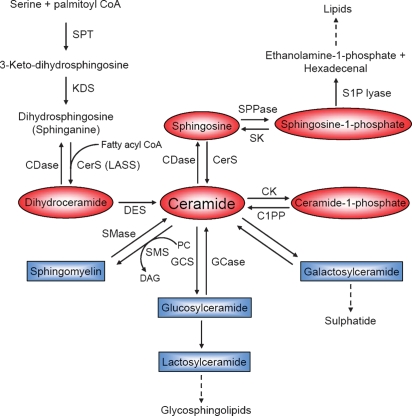
The SL metabolic pathway displays an intricate network of reactions resulting in the formation of a multitude of SLs, with Cer [and dihydroceramide (dhCer)] as the center of SL biosynthesis, catabolism, and as precursors of complex SLs. Cer can be produced in at least two distinct ways. First, it can be synthesized through the de novo pathway, and second, through the hydrolysis of complex lipids, especially sphingomyelin (SM) (Fig. 1). The de novo pathway commences with the condensation of serine and palmitoyl-CoA catalyzed by serine palmitoyl transferase to generate 3-keto-dihydrosphingosine. 3-Keto-dihydrosphingosine is subsequently reduced to form dihydrosphingosine (sphinganine), which is then N-acylated by (dh)Cer synthases to produce dhCer or Cer (10). Six mammalian genes that encode (dh)Cer synthase have recently been cloned. Each individual so-called longevity-assurance homolog (LASS1-6)/Cer synthase (CerS1-6) isoform shows substrate preference for specific chain length fatty acyl CoAs. CerS1, for example, shows significant preference for C18:0 FA CoA, whereas CerS5 and CerS6 preferably catalyze the acylation of (dh)Sph with myristoyl-, palmitoyl-, and stearoyl-CoA compared with very long-chain FA CoAs (11). dhCer is then desaturated by dhCer desaturase, generating a 4,5-trans-double bond to form Cer (12).
Fig. 1.
Scheme of SL metabolism. Pathways are shown as described in the text. Bioactive SL metabolites are highlighted in red. SPT, serine palmitoyl transferase; KDS, 3-keto-dihydrosphingosine reductase; DES, dihydroceramide desaturase; SPPase, Sph phosphate phosphatase; CK, Cer kinase; C1PP, C1P phosphatase; SMS, SM synthase; PC, phosphatidylcholine; DAG, diacylglycerol; GCS, glucosylceramide synthase; GCase, glucosyl CDase.
In the following SL biosynthetic reactions, Cer is primarily used for the synthesis of SM by transferring a phosphocholine headgroup from phosphatidylcholine through the action of SM synthases, thereby also generating diacylglycerol (13).
Cer can also be phosphorylated by Cer kinase, which in turn can be recycled by a C1P phosphatase (14) or glycosylated by glucosyl or galactosyl Cer synthases (15).
In the hydrolytic pathway, SM is cleaved by one of several sphingomyelinases (SMases), releasing phosphocholine and Cer. The SMases can be distinguished according to their pH optima and subcellular localization. Several SMases have been characterized: lysosomal acid SMase (aSMase), zinc-dependent secretory SMase, neutral magnesium-dependent SMase (nSMase), and alkaline SMase (16).
Another important source of Cer is provided by the breakdown of glycosphingolipids through sequential removal of their terminal hydrophilic portions catalyzed by a series of specific hydrolases. The products glucosylceramide (GlcCer) and galactosylceramide are subsequently hydrolyzed by specific β-glucosidases and galactosidases to release Cer (17).
Cer is metabolized by ceramidases (CDases), removing the amide-linked FA to form Sph. Three types of CDases have been classified by their pH optimum and subcellular localization: acid CDase, neutral/alkaline CDase, and alkaline CDase (18). Sph is then available for recycling into SL pathways or for phosphorylation by one of two Sph kinases, SK1 and SK2, to form S1P (19). In return, S1P phosphatases dephosphorylate S1P regenerating Sph (20). Finally, S1P lyase can metabolize S1P to release ethanolamine phosphate and hexadecenal (21).
COMPARTMENTALIZATION AND REGULATION OF BIOACTIVE SLS
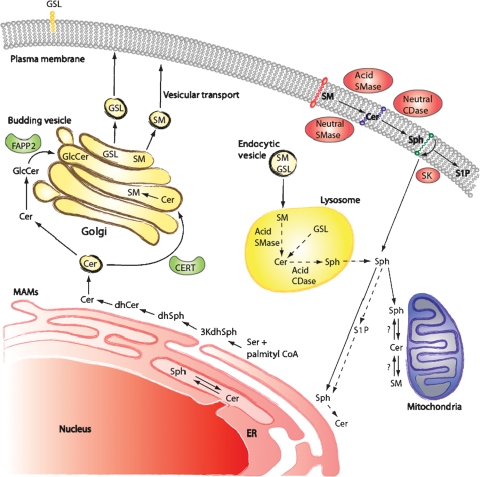
Enzymatic reactions in SL metabolism are distributed throughout different cellular compartments (Fig. 2). Understanding the functions of SLs requires insight into the specific subcellular localization of the enzymes involved in the SL pathway. The initial steps of SL de novo synthesis leading to Cer formation take place on the cytosolic surface of the endoplasmic reticulum (ER) and potentially in ER-associated membranes, such as the perinuclear membrane and mitochondria-associated membranes (12). Synthesis of more complex SL metabolites like SM and GluCer occurs in the Golgi apparatus. Two specific pathways are known for the transport of Cer from the ER to the Golgi. First, SM is formed of Cer provided by the Cer transfer protein, CERT. Second, the synthesis of GluCer is based on Cer deriving from vesicular transport. Subsequently, the transport protein FAPP2 delivers GluCer as precursor for glycosphingolipid (GSL) synthesis (22). The synthesis of complex GSLs (e.g., gangliosides) occurs in the luminal side of the Golgi. Therefore, GluCer needs to flip from the cytosolic surface to the inside of the Golgi. This mechanism is supported by the ABC transporter, P-glycoprotein (also known as MDR1) (23).
Fig. 2.
Compartmentalization of metabolites and enzymes of the SL pathway. 3KdhSph, 3-keto-dihydrosphingosine; dhSph, dihydrosphingosine; CERT, Cer transfer protein; MAMs, mitochondria associated membranes.
Subsequently, SM and complex GSLs are transported to the plasma membrane via vesicular trafficking. There, SM can be metabolized to Cer, and accordingly other bioactive lipids, either by aSMase on the outer leaflet of the membrane or by nSMase, which resides on the inner leaflet of the bilayer.
From the plasma membrane, SLs may be recirculated through the endosomal pathway. Thereby, SM and GluCer are metabolized to Cer in the lysosomal compartment by SMases and glucosidases and Cer is then degraded by acid CDase to form Sph. Due to its ionizable positive charge, the salvaged Sph is able to leave the lysosome and shows adequate solubility in the cytosol to move between membranes, including the ER, where it would be available for recycling (24).
It is also important to realize that the subcellular localization of enzymes of SL metabolism is a key determinant of site of action of bioactive SLs. SLs are either hydrophobic or amphipathic molecules that have hydrophobic as well as hydrophilic attributes. Hence, it is not surprising that these molecules are mostly integral components of biological membranes and show little movement between membranes, unless acted upon by specific transport mechanisms (such as Cer transfer protein or FAPP2).
SLS AS BIOACTIVE MOLECULES
Bioactive SLs are induced by several agonists, and, in turn, they regulate several downstream targets that mediate their various effects on cell function. It should be noted that the cellular levels of the various bioactive SLs exhibit great differences. Concentrations of Cer, Sph, and S1P differ approximately by an order of magnitude, with Cer presenting the highest and S1P the lowest level. A small change in Cer can therefore drastically increase the levels of Sph or S1P.
Within the family of SLs, Cer and S1P are the best explored bioactive components. Cer and Sph are reported to act as tumor-suppressor lipids involved not only in intracellular but also in extracellular processes.
Cer signaling has been intimately involved in the regulation of cell growth, differentiation (25), senescence (26), necrosis (27), proliferation (28), and apoptosis (29). Cer functions, at least in part, by activating protein phosphatases PP1A and PP2A, which exhibit specificity for the D-erythro stereoisomer in vitro (30). It has also been shown that Cer may regulate protein kinase C (PKC) ζ (31), raf-1 (32), and the kinase-suppressor of Ras (33), significantly changing the level of phosphorylation of various key substrates. Another binding target for Cer is the cellular protease cathepsin D, which may mediate the actions of lysosomally generated Cer (34).
Sph has been connected with cellular processes such as inducing cell cycle arrest and apoptosis by modulation of protein kinases and other signaling pathways. It has roles in regulating the actin cytoskeleton and endocytosis and has been shown to inhibit PKC (35). Kinase targets for sphingoid bases including pkh1p and pkh2p have been found in yeast, indicating functions in regulating endocytosis, cell cycle arrest, and protein synthesis (36).
S1P can be seen as a tumor-promoting lipid involved in the regulation of proliferation, cell growth, cell survival, cell migration, inflammation, angiogenesis, vasculogenesis, and resistance to apoptotic cell death, and therefore shows antagonizing effects to those of Cer (37). S1P binds to G-protein-coupled receptors (S1P receptors) on the cell surface, thus regulating diverse G proteins and subsequently various intracellular signaling pathways (38).
More recent studies have broadened the attention to additional bioactive SLs such as C1P, GlcCer, lyso-SM, and dhCer. Metabolism and biological function of S1P and C1P have recently been reviewed (39).
C1P has been implicated in playing roles in inflammation and vesicular trafficking (40). It mediates the activation of phospholipase A2 and the release of arachidonic acid in response to interleukin-β (41).
GlcCer has be shown to be involved in post-Golgi trafficking and in drug resistance (42).
INDUCERS OF SL ENZYMES
In recent years, substantial evidence has accumulated demonstrating that Cer generation pathways such as the SMase and de novo pathway are activated by inducers of apoptosis or growth arrest. Thus, endogenous Cer is produced in response to various stimuli such as cytokines, heat stress (4), UV radiation, hypoxia/reperfusion (43), lipopolysaccharides (44), chemotherapeutic agents, and other miscellaneous agents (6). TNF-α and IL-1 have been shown to act on the de novo pathway as well as on nSMase (2). Furthermore, Cer has recently been linked to Alzheimer's disease and neurodegeneration, possibly mediating the cytotoxic response to amyloid-β peptide (45).
aSMase, on the other hand, is induced by various stress stimuli, such as UV and ionizing radiation, ligation of death receptors, and chemotherapeutic agents such as platinum, paclitaxel, and histone deacetylase inhibitors. Recent studies also implicate an activation of aSMase by reactive oxygen species and nitrosative stress (46). Interestingly, activation of aSMase in response to phorbol esters and to UV radiation has been shown to lead to an increase in Cer formation through the salvage (or recycling) pathway, i.e., by inducing formation of Cer, then Sph, which is recycled back to Cer (3).
SK is activated by various growth factors such as epidermal growth factor, platelet-derived growth factor, and proinflammatory cytokines such as TNF-α and IL-1. However, this reaction is part of a complex signaling cascade dependent on upstream regulators such as PKC (47), phospholipase D (48), and the extracellular signal-regulated kinase mitogen-activated protein kinases. The change in the level of the product S1P subsequently regulates cell viability and inflammatory responses (19).
For more detailed insights into SL pathway activators, the reader is referred to recent reviews (49, 50).
CONCLUSIONS
In recent years, there is more and more evidence that SLs function as key components in modulating cell responses and act as signaling/regulatory molecules. To begin to understand and integrate SL function and regulation, one has to look not only into individual regulated pathways (such as SK1 or aSMase) but also into the complex network of SL metabolism, as one bioactive SL may be metabolically transformed into another bioactive molecule, often with counter functions (e.g., Cer and S1P). Importantly, one should also consider the place of action of bioactive SLs by determining the compartmentalization of both the involved enzymes and their substrates and products. Additional challenges are the experimental difficulties in studying lipid metabolism and function. The last years yielded many useful analytical and molecular tools that now enable extensive research in metabolic, topologic, structural, and functional aspects of Cer generations. More and more enzymes of SL metabolism have now been cloned and characterized, and several SL specific antibodies have been generated, which provides the possibility and the tools to study the topology of SL enzyme actions. Increasingly, animal models and knockouts in various enzymes of SL metabolism are revealing important and critical functions for SLs. The application of mass spectrometry now allows specific qualitative and quantitative analysis of endogenous SL metabolites in experimental systems and in response to agonist stimulation.
Although many significant findings regarding function and regulation of SL metabolism have been made in the last years, there are still multiple problems to be solved regarding lipid-protein interactions, intracellular transport, and mechanisms of regulation. By pursuing the puzzle pieces of the whole spectrum of SLs and their metabolizing enzymes, research over the next few years will pursue to disclose the complexity and relevance of this metabolic pathway in cellular responses and the function of biologically active SLs, and, consequently, downstream signaling pathways.
Abbreviations
aSMase, lysosomal acid sphingomyelinase
CDase, ceramidase
Cer, ceramide
CerS, ceramide synthase
C1P, ceramide-1-phosphate
dhCer, dihydroceramide
ER, endoplasmic reticulum
GlcCer, glucosylceramide
GSL, glycosphingolipid
IL, interleukin
nSMase, neutral magnesium-dependent sphingomyelinase
PKC, protein kinase C
S1P, sphingosine-1-phosphate
SK, sphingosine kinase
SL, sphingolipid
SM, sphingomyelin
SMase, sphingomyelinase
Sph, sphingosine
TNF, tumor necrosis factor
This work was supported by National Institutes of Health Grants GM-43825 and CA-87584.
Published, JLR Papers in Press, November 17, 2008.
References
- 1.Simons K., and E. Ikonen. 1997. Functional rafts in cell membranes. Nature. 387 569–572. [DOI] [PubMed] [Google Scholar]
- 2.Kolesnick R. N., A. Haimovitz-Friedman, and Z. Fuks. 1994. The sphingomyelin signal transduction pathway mediates apoptosis for tumor necrosis factor, Fas, and ionizing radiation. Biochem. Cell Biol. 72 471–474. [DOI] [PubMed] [Google Scholar]
- 3.Becker K. P., K. Kitatani, J. Idkowiak-Baldys, J. Bielawski, and Y. A. Hannun. 2005. Selective inhibition of juxtanuclear translocation of protein kinase C betaII by a negative feedback mechanism involving ceramide formed from the salvage pathway. J. Biol. Chem. 280 2606–2612. [DOI] [PubMed] [Google Scholar]
- 4.Jenkins G. M., L. A. Cowart, P. Signorelli, B. J. Pettus, C. E. Chalfant, and Y. A. Hannun. 2002. Acute activation of de novo sphingolipid biosynthesis upon heat shock causes an accumulation of ceramide and subsequent dephosphorylation of SR proteins. J. Biol. Chem. 277 42572–42578. [DOI] [PubMed] [Google Scholar]
- 5.Goldkorn T., N. Balaban, M. Shannon, V. Chea, K. Matsukuma, D. Gilchrist, H. Wang, and C. Chan. 1998. H2O2 acts on cellular membranes to generate ceramide signaling and initiate apoptosis in tracheobronchial epithelial cells. J. Cell Sci. 111 3209–3220. [DOI] [PubMed] [Google Scholar]
- 6.Bose R., M. Verheij, A. Haimovitz-Friedman, K. Scotto, Z. Fuks, and R. Kolesnick. 1995. Ceramide synthase mediates daunorubicin-induced apoptosis: an alternative mechanism for generating death signals. Cell. 82 405–414. [DOI] [PubMed] [Google Scholar]
- 7.Hannun Y. A., and L. M. Obeid. 2008. Principles of bioactive lipid signalling: lessons from sphingolipids. Nat. Rev. Mol. Cell Biol. 9 139–150. [DOI] [PubMed] [Google Scholar]
- 8.Ogretmen B., and Y. A. Hannun. 2004. Biologically active sphingolipids in cancer pathogenesis and treatment. Nat. Rev. 4 604–616. [DOI] [PubMed] [Google Scholar]
- 9.Zheng W., J. Kollmeyer, H. Symolon, A. Momin, E. Munter, E. Wang, S. Kelly, J. C. Allegood, Y. Liu, Q. Peng, et al. 2006. Ceramides and other bioactive sphingolipid backbones in health and disease: lipidomic analysis, metabolism and roles in membrane structure, dynamics, signaling and autophagy. Biochim. Biophys. Acta. 1758 1864–1884. [DOI] [PubMed] [Google Scholar]
- 10.Mandon E. C., I. Ehses, J. Rother, G. van Echten, and K. Sandhoff. 1992. Subcellular localization and membrane topology of serine palmitoyltransferase, 3-dehydrosphinganine reductase, and sphinganine N-acyltransferase in mouse liver. J. Biol. Chem. 267 11144–11148. [PubMed] [Google Scholar]
- 11.Pewzner-Jung Y., S. Ben-Dor, and A. H. Futerman. 2006. When do Lasses (longevity assurance genes) become CerS (ceramide synthases)?: insights into the regulation of ceramide synthesis. J. Biol. Chem. 281 25001–25005. [DOI] [PubMed] [Google Scholar]
- 12.Michel C., and G. van Echten-Deckert. 1997. Conversion of dihydroceramide to ceramide occurs at the cytosolic face of the endoplasmic reticulum. FEBS Lett. 416 153–155. [DOI] [PubMed] [Google Scholar]
- 13.Tafesse F. G., P. Ternes, and J. C. Holthuis. 2006. The multigenic sphingomyelin synthase family. J. Biol. Chem. 281 29421–29425. [DOI] [PubMed] [Google Scholar]
- 14.Shinghal R., R. H. Scheller, and S. M. Bajjalieh. 1993. Ceramide 1-phosphate phosphatase activity in brain. J. Neurochem. 61 2279–2285. [DOI] [PubMed] [Google Scholar]
- 15.Ichikawa S., and Y. Hirabayashi. 1998. Glucosylceramide synthase and glycosphingolipid synthesis. Trends Cell Biol. 8 198–202. [DOI] [PubMed] [Google Scholar]
- 16.Marchesini N., and Y. A. Hannun. 2004. Acid and neutral sphingomyelinases: roles and mechanisms of regulation. Biochem. Cell Biol. 82 27–44. [DOI] [PubMed] [Google Scholar]
- 17.Tettamanti G. 2004. Ganglioside/glycosphingolipid turnover: new concepts. Glycoconj. J. 20 301–317. [DOI] [PubMed] [Google Scholar]
- 18.Mao C., and L. M. Obeid. 2008. Ceramidases: regulators of cellular responses mediated by ceramide, sphingosine, and sphingosine-1-phosphate. Biochim. Biophys. Acta. 1781 424–434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hait N. C., C. A. Oskeritzian, S. W. Paugh, S. Milstien, and S. Spiegel. 2006. Sphingosine kinases, sphingosine 1-phosphate, apoptosis and diseases. Biochim. Biophys. Acta. 1758 2016–2026. [DOI] [PubMed] [Google Scholar]
- 20.Johnson K. R., K. Y. Johnson, K. P. Becker, J. Bielawski, C. Mao, and L. M. Obeid. 2003. Role of human sphingosine-1-phosphate phosphatase 1 in the regulation of intra- and extracellular sphingosine-1-phosphate levels and cell viability. J. Biol. Chem. 278 34541–34547. [DOI] [PubMed] [Google Scholar]
- 21.Bandhuvula P., and J. D. Saba. 2007. Sphingosine-1-phosphate lyase in immunity and cancer: silencing the siren. Trends Mol. Med. 13 210–217. [DOI] [PubMed] [Google Scholar]
- 22.Yamaji T., K. Kumagai, N. Tomishige, and K. Hanada. 2008. Two sphingolipid transfer proteins, CERT and FAPP2: their roles in sphingolipid metabolism. IUBMB Life. 60 511–518. [DOI] [PubMed] [Google Scholar]
- 23.Lannert H., K. Gorgas, I. Meissner, F. T. Wieland, and D. Jeckel. 1998. Functional organization of the Golgi apparatus in glycosphingolipid biosynthesis. Lactosylceramide and subsequent glycosphingolipids are formed in the lumen of the late Golgi. J. Biol. Chem. 273 2939–2946. [DOI] [PubMed] [Google Scholar]
- 24.Riboni L., R. Bassi, A. Caminiti, A. Prinetti, P. Viani, and G. Tettamanti. 1998. Metabolic fate of exogenous sphingosine in neuroblastoma neuro2A cells. Dose-dependence and biological effects. Ann. N. Y. Acad. Sci. 845 46–56. [DOI] [PubMed] [Google Scholar]
- 25.Okazaki T., R. M. Bell, and Y. A. Hannun. 1989. Sphingomyelin turnover induced by vitamin D3 in HL-60 cells. Role in cell differentiation. J. Biol. Chem. 264 19076–19080. [PubMed] [Google Scholar]
- 26.Venable M. E., J. Y. Lee, M. J. Smyth, A. Bielawska, and L. M. Obeid. 1995. Role of ceramide in cellular senescence. J. Biol. Chem. 270 30701–30708. [DOI] [PubMed] [Google Scholar]
- 27.Hetz C. A., M. Hunn, P. Rojas, V. Torres, L. Leyton, and A. F. Quest. 2002. Caspase-dependent initiation of apoptosis and necrosis by the Fas receptor in lymphoid cells: onset of necrosis is associated with delayed ceramide increase. J. Cell Sci. 115 4671–4683. [DOI] [PubMed] [Google Scholar]
- 28.Adam D., M. Heinrich, D. Kabelitz, and S. Schutze. 2002. Ceramide: does it matter for T cells? Trends Immunol. 23 1–4. [DOI] [PubMed] [Google Scholar]
- 29.Obeid L. M., C. M. Linardic, L. A. Karolak, and Y. A. Hannun. 1993. Programmed cell death induced by ceramide. Science. 259 1769–1771. [DOI] [PubMed] [Google Scholar]
- 30.Chalfant C. E., K. Kishikawa, M. C. Mumby, C. Kamibayashi, A. Bielawska, and Y. A. Hannun. 1999. Long chain ceramides activate protein phosphatase-1 and protein phosphatase-2A. Activation is stereospecific and regulated by phosphatidic acid. J. Biol. Chem. 274 20313–20317. [DOI] [PubMed] [Google Scholar]
- 31.Wang G., J. Silva, K. Krishnamurthy, E. Tran, B. G. Condie, and E. Bieberich. 2005. Direct binding to ceramide activates protein kinase Czeta before the formation of a pro-apoptotic complex with PAR-4 in differentiating stem cells. J. Biol. Chem. 280 26415–26424. [DOI] [PubMed] [Google Scholar]
- 32.Blazquez C., I. Galve-Roperh, and M. Guzman. 2000. De novo-synthesized ceramide signals apoptosis in astrocytes via extracellular signal-regulated kinase. FASEB J. 14 2315–2322. [DOI] [PubMed] [Google Scholar]
- 33.Ruvolo P. P. 2003. Intracellular signal transduction pathways activated by ceramide and its metabolites. Pharmacol. Res. 47 383–392. [DOI] [PubMed] [Google Scholar]
- 34.Heinrich M., M. Wickel, W. Schneider-Brachert, C. Sandberg, J. Gahr, R. Schwandner, T. Weber, P. Saftig, C. Peters, J. Brunner, et al. 1999. Cathepsin D targeted by acid sphingomyelinase-derived ceramide. EMBO J. 18 5252–5263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Smith E. R., A. H. Merrill, L. M. Obeid, and Y. A. Hannun. 2000. Effects of sphingosine and other sphingolipids on protein kinase C. Methods Enzymol. 312 361–373. [DOI] [PubMed] [Google Scholar]
- 36.Cowart L. A., and Y. A. Hannun. 2007. Selective substrate supply in the regulation of yeast de novo sphingolipid synthesis. J. Biol. Chem. 282 12330–12340. [DOI] [PubMed] [Google Scholar]
- 37.Spiegel S., and S. Milstien. 2003. Sphingosine-1-phosphate: an enigmatic signalling lipid. Nat. Rev. Mol. Cell Biol. 4 397–407. [DOI] [PubMed] [Google Scholar]
- 38.Taha T. A., K. M. Argraves, and L. M. Obeid. 2004. Sphingosine-1-phosphate receptors: receptor specificity versus functional redundancy. Biochim. Biophys. Acta. 1682 48–55. [DOI] [PubMed] [Google Scholar]
- 39.Kihara A., S. Mitsutake, Y. Mizutani, and Y. Igarashi. 2007. Metabolism and biological functions of two phosphorylated sphingolipids, sphingosine 1-phosphate and ceramide 1-phosphate. Prog. Lipid Res. 46 126–144. [DOI] [PubMed] [Google Scholar]
- 40.Chalfant C. E., and S. Spiegel. 2005. Sphingosine 1-phosphate and ceramide 1-phosphate: expanding roles in cell signaling. J. Cell Sci. 118 4605–4612. [DOI] [PubMed] [Google Scholar]
- 41.Pettus B. J., A. Bielawska, P. Subramanian, D. S. Wijesinghe, M. Maceyka, C. C. Leslie, J. H. Evans, J. Freiberg, P. Roddy, Y. A. Hannun, et al. 2004. Ceramide 1-phosphate is a direct activator of cytosolic phospholipase A2. J. Biol. Chem. 279 11320–11326. [DOI] [PubMed] [Google Scholar]
- 42.Radin N. S., J. A. Shayman, and J. Inokuchi. 1993. Metabolic effects of inhibiting glucosylceramide synthesis with PDMP and other substances. Adv. Lipid Res. 26 183–213. [PubMed] [Google Scholar]
- 43.Hernandez O. M., D. J. Discher, N. H. Bishopric, and K. A. Webster. 2000. Rapid activation of neutral sphingomyelinase by hypoxia-reoxygenation of cardiac myocytes. Circ. Res. 86 198–204. [DOI] [PubMed] [Google Scholar]
- 44.Haimovitz-Friedman A., C. Cordon-Cardo, S. Bayoumy, M. Garzotto, M. McLoughlin, R. Gallily, C. K. Edwards III, E. H. Schuchman, Z. Fuks, and R. Kolesnick. 1997. Lipopolysaccharide induces disseminated endothelial apoptosis requiring ceramide generation. J. Exp. Med. 186 1831–1841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lee J. T., J. Xu, J. M. Lee, G. Ku, X. Han, D. I. Yang, S. Chen, and C. Y. Hsu. 2004. Amyloid-beta peptide induces oligodendrocyte death by activating the neutral sphingomyelinase-ceramide pathway. J. Cell Biol. 164 123–131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Castillo S. S., M. Levy, J. V. Thaikoottathil, and T. Goldkorn. 2007. Reactive nitrogen and oxygen species activate different sphingomyelinases to induce apoptosis in airway epithelial cells. Exp. Cell Res. 313 2680–2686. [DOI] [PubMed] [Google Scholar]
- 47.Johnson K. R., K. P. Becker, M. M. Facchinetti, Y. A. Hannun, and L. M. Obeid. 2002. PKC-dependent activation of sphingosine kinase 1 and translocation to the plasma membrane. Extracellular release of sphingosine-1-phosphate induced by phorbol 12-myristate 13-acetate (PMA). J. Biol. Chem. 277 35257–35262. [DOI] [PubMed] [Google Scholar]
- 48.Melendez A., R. A. Floto, D. J. Gillooly, M. M. Harnett, and J. M. Allen. 1998. FcgammaRI coupling to phospholipase D initiates sphingosine kinase-mediated calcium mobilization and vesicular trafficking. J. Biol. Chem. 273 9393–9402. [DOI] [PubMed] [Google Scholar]
- 49.El Alwani M., B. X. Wu, L. M. Obeid, and Y. A. Hannun. 2006. Bioactive sphingolipids in the modulation of the inflammatory response. Pharmacol. Ther. 112 171–183. [DOI] [PubMed] [Google Scholar]
- 50.Levade T., N. Auge, R. J. Veldman, O. Cuvillier, A. Negre-Salvayre, and R. Salvayre. 2001. Sphingolipid mediators in cardiovascular cell biology and pathology. Circ. Res. 89 957–968. [DOI] [PubMed] [Google Scholar]