Abstract
The association of hepatic steatosis with hepatic insulin resistance and type 2 diabetes has prompted investigators to elucidate the underlying mechanism. In this review we focus on pathways of lipid metabolism, and we review recent data, primarily from mouse models, that link lipid intermediates with insulin resistance. Most of the studies that implicate acyl-CoA, lysophosphatidic acid, phosphatidic acid, diacylglycerol, or ceramide rely on indirect associations. Convincing data to support the hypothesis that specific lipid intermediates initiate pathways that alter insulin signaling will require studies in which the concentration of each purported signaling molecule can be manipulated independently.
Keywords: Hepatic steatosis, diacylglycerol, phosphatidic acid, lysophosphatidic acid, ceramide
Nonalcoholic fatty liver disease (NAFLD) is characterized by excessive lipid accumulation in the liver in the absence of ‘significant’ alcohol consumption and may progress to nonalcoholic steatohepatitis, fibrosis, cirrhosis, and hepatocellular carcinoma. In the United States, the prevalence of NAFLD is estimated to be 20% (1); however, the true prevalence of NAFLD is unknown because many individuals in earlier stages of the disease are asymptomatic and remain undiagnosed. Although NAFLD was previously thought to be a benign condition, its development parallels the development of the insulin resistance (IR) syndrome and is associated with type 2 diabetes mellitus. Patients with the IR syndrome have a 4- to 11-fold increased risk of developing NAFLD.
With overnutrition and lack of exercise, liver and other tissues store excess energy as triacylglycerol (TAG). Shunting carbon-energy into a storage form is likely protective against cytotoxic FA accumulation. Hepatic IR is associated with the accumulation of TAG and FA metabolites (fatty acyl-CoA, diacylglycerol (DAG), ceramide, and glycosphingolipid). The short-term protection achieved from sequestering FA may lead to long-term morbidity with the development of further IR leading to type 2 diabetes mellitus and the deterioration of hepatic function.
Although fatty liver correlates with hepatic IR, it remains unclear whether IR or excess TAG stores develop first and whether fatty liver invariably leads to IR. Peripheral IR may cause fatty liver by elevating plasma FA, glucose, and insulin, which stimulate hepatic lipid synthesis and impair hepatic β-oxidation. However, high-fat feeding may cause hepatic IR before systemic IR develops. We will focus on the metabolic pathways involved in the development of NAFLD, the role of altered hepatic TAG metabolism, and the role of lipid metabolites in the development of IR.
METABOLIC PATHWAYS INVOLVED IN FATTY LIVER DEVELOPMENT
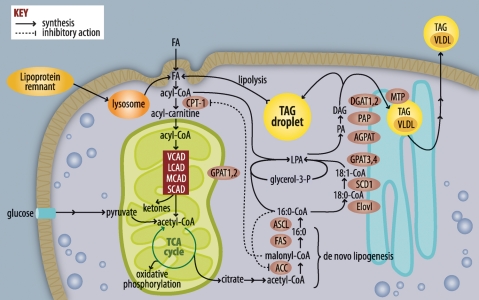
Hepatic lipid accumulation can be caused by four different metabolic perturbations: increased FA delivery to hepatocytes from lipolyzed adipose TAG, dietary lipids, or hepatic de novo lipogenesis (DNL); increased TAG synthesis; decreased hepatic FA oxidation; and inadequate TAG secretion in VLDL (Fig. 1).
Fig. 1.
Pathways of FA and triacylglycerol metabolism. FAs used for triacylglycerol (TAG) synthesis or β-oxidation are activated to form acyl-CoAs. If targeted toward β-oxidation, acyl-CoAs are converted to acyl-carnitines, enter the mitochondria, and are metabolized by a succession of dehydrogenases that are relatively specific for very-long (VLAD), long (LCAD), medium (MCAD), and short (SCAD) chain acyl-CoAs. The end-product acetyl-CoA enters the tricarboxylic acid cycle (TCA), producing NADH and FADH, which contribute to ATP formation by oxidative phosphorylation. When excess calories from glucose are available, acetyl-CoA entering the TCA cycle is converted to citrate, which leaves the mitochondria and is used for FA synthesis via acetyl-CoA carboxylase (ACC) and FAS. FA elongases (Elovl) and desaturases (SCD) modify acyl-CoAs, which are used to esterify glycerol-3-phosphate by glycerol-3-P acyltransferases (GPAT). The lysophosphatidic acid (LPA) product is esterified to form phosphatidic acid (PA) by an acyl-glycerol-3-P acyltransferase, the PA is hydrolyzed by phosphatidic acid phosphohydrolase (PAP), and the final esterification step catalyzed by diacylglycerol acyltransferase (DGAT) produces TAG. Liver TAG can be stored in lipid droplets or transferred to VLDL by the mitochondrial TAG transport protein.
Fatty acid delivery and synthesis
After consuming a meal containing fat, chylomicron-TAG is delivered to the liver and is lipolyzed in lysosomes with the release of FA. FA can also arise from DNL in response to a high carbohydrate meal, as excess glucose is metabolized to acetyl-CoA, the major substrate for FA synthesis. Malonyl-CoA, the product of acetyl-CoA carboxylase (ACC), is not only a substrate for FAS , but also inhibits carnitine-palmitoyltransferase-1 (CPT-1), which regulates long-chain FA entry into mitochondria for β-oxidation. FAS sequentially adds two carbons from malonyl-CoA to synthesize 16 and 18 carbon FA. These saturated FAs must be activated by long-chain acyl-CoA synthetase (ACSL) before they can be elongated by an elongase (ELOVL6), desaturated by stearoyl-CoA desaturase (SCD), or used for the synthesis of glycerolipids or cholesterol esters.
TAG synthesis
Hepatic FAs are derived from entering chylomicron remnants, TAG lipolysis in lipid droplets, or DNL. After activation, the acyl-CoAs enter the glycerolipid synthetic pathway. In the liver, TAG may either be stored in cytoplasmic lipid droplets or incorporated into VLDL particles and secreted into the blood. The four-step synthesis of TAG, first identified by Eugene Kennedy and his colleagues (2), occurs on the cytosolic surface of the mitochondrial outer membrane and the endoplasmic reticulum. Four glycerol-3-phosphate acyltransferases (GPAT), potentially six 1-acylglycerol-3-phosphate acyltransferases (AGPAT),1,2 three phosphatidic acid phosphatases (PAP), and two DAG acyltransferases (DGAT) have been cloned3.
Hepatic TAG lipolysis and β-oxidation
During fasting, insulin levels fall, adipose TAG is hydrolyzed, and FAs are released and travel to the liver. It is not clear whether exogenously derived FAs must first be reesterified and stored in lipid droplets before they can be oxidized, or whether acyl-CoAs can be directly converted to acyl-carnitines and enter the oxidative pathway. Within the mitochondrial matrix, acyl-carnitines are converted to acyl-CoAs by CPT-2. Very long chain acyl-CoA dehydrogenase (VLCAD) acts on C12-C24 carbon acyl-CoAs, long chain acyl-CoA dehydrogenase (LCAD) acts on C8-C20 acyl-CoAs, medium chain acyl-CoA dehydrogenase (MCAD) acts on C4-C12 acyl-CoAs, and short-chain acyl-CoA dehydrogenase (SCAD) acts on C4-C6 acyl-CoAs. When the tricarboxylic acid (TCA) cycle cannot metabolize all the acetyl-CoA generated from β-oxidation, excess acetyl-CoA is converted to ketone bodies.
VLDL secretion
The origin of VLDL-TAG remains poorly understood. A precursor VLDL particle that contains apoB and a small amount of TAG is formed in the endoplasmic reticulum (ER). This small VLDL precursor may fuse within the ER with a larger droplet of TAG to form a TAG-rich particle. The TAG that fuses is thought to arise from cytosolic TAG derived primarily from plasma FA uptake and less from DNL (7). Incorporation of cytosolic TAG into VLDL may require lipolysis and reesterification steps at the ER (3).
TRANSCRIPTIONAL REGULATION OF DE NOVO LIPOGENESIS AND FA OXIDATION
An excess of glucose, FA, and insulin ultimately lead to hepatic steatosis and worsening hepatic IR via a network of transcription factors (Fig. 2). Activation of hepatic liver-X-receptor (LXR) by endogenous oxysterol ligands results in up-regulation of genes involved in cholesterol, lipid, and bile acid metabolism. In mice, oral LXR agonists result in enhanced hepatic FA synthesis, hepatic steatosis, and hypertriglyceridemia, all mediated by increased expression of SREBP-1c (4). Transgenic hepatic over-expression of SREBP-1c produces a fatty liver and a 4-fold increase in the rate of hepatic FA synthesis, with increases in lipogenic genes like FAS, ACC and SCD-1 (5), and GPAT1 (5). Insulin increases LXR's ability to activate the SREBP-1c promoter and increase hepatic lipogenesis (6). Thus, hyperinsulinemia can activate LXR and SREBP-1c and result in increased lipogenesis, liver TAG accumulation, and worsening hepatic IR.
Fig. 2.
Altered insulin signaling by lipid intermediates. Increased nutrient influx raises intracellular FA concentrations. With nutrient surfeit, elevated malonyl-CoA inhibits CPT-1 and β-oxidation and results in increased availability of acyl-CoAs for TAG synthesis and storage or secretion. Intermediates in the TAG biosynthetic pathway activate inhibitors of insulin signaling, including protein kinase C (PKC), mTOR, and S6K, which suppress IRS-1 activation of PIP3. Ceramide inhibits Akt-mediated insulin signaling, derepressing FOXO and leading to increases in transcription of phosphoenolpyruvate (PEPCK) and glucose-6-phosphatase (G-6-Pase). These enhance gluconeogenesis and hepatic glucose output. Nutrients act on transcription factors (LXR, ChREBP, SREBP-1c and PPAR γ), which increase transcription of genes involved in FA and TAG synthesis (ACC, FAS, GPAT1). These enhance steatosis and hepatic insulin resistance. DHAP, dihydroxyacetone-P; G3P, glycerol-3-P; GSK, glycogen synthase kinase.
ChREBP regulates hepatic lipid synthesis through transcriptional control of the lipogenic genes ACC and FAS in response to glucose (7). Liver-specific ChREBP inhibition results in decreased hepatic lipogenesis and ameliorated hepatic steatosis in ob/ob mice (7).
DO INTERMEDIATES IN THE KENNEDY PATHWAY OF GLYCEROLIPID BIOSYNTHESIS INITIATE SIGNALING PATHWAYS?
Although IR correlates highly with TAG content in skeletal muscle and liver, TAG itself is believed to be a surrogate marker for the true disruptor of the insulin signal, variously hypothesized to be a) a cytokine released from macrophages or adipocytes, b) activation of the NFκB pathway (8), or c) a signaling pathway initiated by a FA-derived lipid. Lysophosphatidic acid (LPA), phosphatidic acid (PA), and DAG are well-established initiators of signaling pathways but have been studied primarily after their hydrolysis from membrane phospholipids. In liver, however, the major production of LPA, PA, and DAG occurs via de novo glycerolipid synthesis; lipid intermediates derived from this pathway and related lipids [acyl-CoAs (9, 10), ceramide (11) and acyl-carnitines] may also act as signaling molecules. Because incubating cells with FA or infusing FA into mice causes IR within a few hours, it appears that a direct FA metabolite can promote IR.
BLOCK OF INSULIN SIGNALING BY LIPID METABOLITES
With hepatic IR, insulin cannot effectively suppress hepatic glucose output in a hyperinsulinemic-euglycemic clamp study. IR can also be inferred by impaired tyrosine phosphorylation of the insulin receptor and aberrant phosphorylation of serine residues of insulin receptor substrate-1 (IRS-1) and downstream members of the insulin signaling pathway.
Our studies implicate GPAT1-produced DAG as a cause of IR (12, 13); when excess DAG is formed, protein kinase C (PKC)ɛ is activated, and IRS-1 is phosphorylated on Ser307, which suppresses IRS-1 tyrosine phosphorylation and diminishes phosphatidylinositol 3-kinase activation. These data suggest that the DAG formed during glycerolipid synthesis can interact with and activate PKC at distant membranes and show that GPAT1 modulates the cell content of lipid intermediates. This is not entirely surprising because lipogenic diets normally increase SREBP-1c-mediated GPAT1 mRNA expression (2). In addition to DAG, the glycerolipid intermediates LPA and PA can also initiate signaling pathways. LPA is a ligand for peroxisome proliferator-activated receptor (PPAR) γ (14) and might be responsible for the up-regulation of PPARγ target genes observed in NAFLD, whereas intracellular PA activates mammalian target of rapamycin (15), which down-regulates the insulin signal by promoting serine phosphorylation of IRS when nutrients are present in excess.
THE ROLE OF LIPID METABOLITES IN THE DEVELOPMENT OF HEPATIC INSULIN RESISTANCE
Although fatty liver is associated with IR, several rodent models with steatosis remain insulin sensitive (16–21). Factors that may explain the variable relationship between fatty liver and IR include the specific pathways that contribute to hepatic fat accumulation, the type of lipid metabolites that accumulate, the cellular location and FA composition of those metabolites, the presence or absence of hepatic or systemic inflammation, and the type of rodent diet used.
Altered de novo lipogenesis and glycerolipid synthesis
In humans with NAFLD, the estimated contribution of adipose lipolysis, dietary FA, and hepatic DNL to hepatic TAG and VLDL-TAG are 60%, 15%, and 24%, respectively (22). It is not known, however, whether the lipid intermediates derived from these pathways have similar effects on the development of hepatic IR, or whether one pathway predominates. When over-expression of hepatic LPL increases FA flux into the liver from lipoproteins, hepatic IR is associated with an increased hepatic content of TAG and acyl-CoA (23). Augmented DNL also appears to be important, because ob/ob mice deficient in either SREBP-1c or ChREBP are protected from hepatic steatosis and IR (24, 25). Further, rodents with liver-specific decreases in the synthesis of malonyl-CoA, ACC null mice, and rats that over-express malonyl-CoA decarboxylase in liver have improved hepatic insulin sensitivity due to lower DNL and increased β-oxidation (26, 27). In contrast, mice with a liver-specific knockout of FAS develop a fatty liver despite suppressed DNL because PPARα is not activated (28). Despite the fatty liver that results from decreased β-oxidation, FAS null mice have impaired gluconeogenesis and are hypoglycemic and insulin sensitive.
Increased SREBP-1c activity in ob/ob mouse liver increases GPAT1 mRNA expression and hepatic de novo glycerolipid synthesis (29), whereas hepatic GPAT1 knockdown in ob/ob mice lowers hepatic TAG and DAG content and plasma glucose concentrations (30). Studies in GPAT1 knock out (KO) mice and in rats with hepatic over-expression of GPAT1 suggest that the lipid accumulation from de novo glycerolipid synthesis contributes to the development of hepatic IR in the absence of obesity or a high fat diet (12, 13). Unlike lipid accumulation from DNL, hepatic steatosis caused by a block in VLDL-TAG secretion does not cause hepatic IR, perhaps because lipid intermediates do not accumulate, suggesting that lipids destined for VLDL secretion are in a separate cellular pool that cannot affect inhibitors of insulin signaling (17).
Impaired β-oxidation
Obese mouse models largely demonstrate that activating β-oxidation decreases hepatic lipid accumulation and improves insulin sensitivity. For example, activating PPARα with Wy-14,643 increases β-oxidation, reduces hepatic TAG accumulation, and improves insulin signaling in ob/ob mice and in lipoatrophic A-ZIP/F-1 mice (31, 32). However, the role of suppressed β-oxidation in the development of IR is less clear. Both prolonged fasting and short-term pharmacological inhibition of CPT-1 cause a fatty liver without IR (18, 21). Unfortunately, DAG and other lipid intermediates were not measured in this study.
Fasting-induced steatosis
When hepatic TAG accumulates in lean fasted animals, it does not cause hepatic IR. Perhaps signaling lipids are sequestered in pools that cannot interfere with insulin signaling. Similar to pharmacological inhibition of β-oxidation, genetic deficiency of PPARα, VLCAD, or MCAD causes a fatty liver and mild to severe hypoglycemia under fasting conditions (33–35). PPARα KO mice are protected from hepatic IR when fed a high fat (HF)-coconut oil diet (20), but not when fed HF-palm oil or HF-lard diets (36). The insulin sensitivity of VLCAD and MCAD mice has not been studied, but LCAD KO mice develop hepatic steatosis and reduced FA oxidation without hypoglycemia (37). Although fasting hepatic DAG and ceramide content do not differ between wild-type (WT) and LCAD KO mice, the hepatic DAG content increases 3-fold in the LCAD KO livers after insulin stimulation during a hyperinsulinemic-euglycemic clamp, and is associated with hepatic IR (43). Thus, inhibiting β-oxidation in fasted lean mice causes hepatic steatosis without IR, whereas inhibiting β-oxidation in previously high-fat-fed animals interferes with insulin signaling. Perhaps impaired β-oxidation is most detrimental to insulin sensitivity when DNL is activated in response to feeding, thereby resulting in increased synthesis of FA and glycerolipid intermediates. It is not clear what makes one kind of lipid accumulation differ from another, but differences in cellular location or in lipid species may determine whether insulin signaling is impaired. Additionally, none of these models describes a liver-specific inhibition of β-oxidation. Inhibiting β-oxidation in adipose tissue or muscle may alter plasma cytokine levels that secondarily improve or inhibit hepatic insulin signaling.
Diet effects
Both DAG and ceramide can diminish hepatic insulin sensitivity, and feeding rats a HF safflower oil diet (HF-SD) for three days increases hepatic DAG and PKCɛ activity and hepatic IR (10). Conversely, a liver-specific knock-down of PKCɛ prevents HF-SD-induced hepatic IR, strengthening the hypothesis that PKCɛ and DAG mediate hepatic IR in this model (38). Other mouse models that support this hypothesis include hepatic over-expression of GPAT1 (13), LCAD KO mice (37), Elovl6 KO mice fed a high fat-high sucrose diet (19), and mouse models fed with the HF-SD including: ACC2 KO (19), ACC1 and 2 knockdown (26), GPAT1 KO (12), and DGAT2 knockdown mice (16). Although these studies provide strong evidence that DAG and PKCɛ are important mediators of hepatic IR in HF-SD fed animals, several studies question the importance of DAG. For example, GPAT1 KO mice are protected from hepatic IR when fed the HF-SD, but not when fed a diet high in saturated fat (39, 40). Similarly, mice treated with DGAT2 ASO are protected from HF-SD IR, but not from IR induced by a HF saturated diet (26, 41). These discrepancies suggest that different fatty acid species affect mechanisms of hepatic IR differently. For example, a HF-lard diet causes a ceramide-associated hepatic IR that is reversed by inhibiting sphingolipid synthesis, whereas a HF-soy oil diet causes a DAG-associated IR that is not affected by sphingolipid inhibition (11). Perhaps high-saturated, but not high-unsaturated fat diets induce a low-grade systemic inflammation. Increased macrophage production of TNFα activates NFκB in hepatocytes and increases ceramide and hepatic IR (8, 42). However, none of the studies using a HF-SD to induce IR reported increases in inflammation, so this diet may not be as proinflammatory as diets high in saturated fat. Human diets consist of both saturated and unsaturated fats, and DAG and ceramide may both be relevant lipid mediators of IR in human NAFLD.
Elevated lipid intermediates without impaired insulin sensitivity
Mice can have elevated hepatic TAG, DAG, and ceramide without hepatic IR (16, 17). This discrepancy might be explained if different pools of DAG and ceramide exist within cells, with only certain pools able to regulate inhibitors of insulin signaling. Thus, measuring hepatic DAG and ceramide total content instead of the content in specific membrane fractions may obscure the relevant mechanism. Additionally, some DAG and sphingolipid species may be poor inhibitors of insulin signaling. The fact that diets high in different FA species have remarkably different effects on hepatic insulin sensitivity and gene expression (11, 43) suggests that DAG FA composition could be relevant. Further, ceramide-induced IR may be caused by ganglioside metabolites of ceramide (44). Future researchers should measure not only gross lipid content, but also the intracellular locations and FA composition of lipids, as well as inflammatory markers that are associated with IR.
SUMMARY AND FUTURE DIRECTIONS
A major difficulty in assessing the importance or the significance of any lipid intermediate to impaired insulin signaling is the lack of direct experiments. Most of the studies that implicate acyl-CoA, LPA, PA, DAG, or ceramide rely on indirect associations. The effects of different diets are not well documented or explained mechanistically, and the inter-relationships between different putative signaling pathways have been inadequately delineated. Convincing data to support the hypothesis that specific lipid intermediates initiate signaling pathways that alter insulin signaling may require additional studies in cultured hepatocytes in which the concentration of each purported signaling molecule can be manipulated independently.
Abbreviations
ACC, acetyl-CoA carboxylase
ACSL, acyl-CoA synthetase
AGPAT, 1-acylglycerol-3-phosphate acyltransferase
ChREBP, carbohydrate-responsive element-binding protein
CPT, carnitine-palmitoyltransferase
DAG, diacylglycerol
DGAT, DAG acyltransferase
GPAT, glycerol-3-phosphate acyltransferase
HF, high fat
HF-SD, HF safflower oil diet
IR, insulin resistance
IRS, insulin receptor substrate
KO, knock out
LCAD, long chain acyl-CoA dehydrogenase
LPA, lysophosphatidic acid
LXR, liver-X-receptor
MCAD, medium chain acyl-CoA dehydrogenase
NAFLD, nonalcoholic fatty liver disease
DNL, de novo lipogenesis
PA, phosphatidic acid
PAP, phosphatidic acid phosphatase
PKC, protein kinase C
PPAR, peroxisome proliferator-activated receptor
TAG, triacylglycerol
SCAD, short-chain acyl-CoA dehydrogenase
SCD, stearoyl-CoA desaturase
SREBP, sterol regulatory element binding protein
TCA, tricarboxylic acid
WT, wild type
VLCAD, long chain acyl-CoA dehydrogenase
This work was supported by National Institutes of Health Grants DK56598, DK59935, and DK56350.
Published, JLR Papers in Press, November 6, 2008.
Footnotes
AGPAT8 and AGPAT6 are correctly termed GPAT3 and GPAT4, respectively.
CGI-58, and endophilin both have AGPAT activity.
Several additional enzymes exhibit some DGAT activity, including MGAT2.
References
- 1.Ruhl C. E., and J. E. Everhart. 2004. Epidemiology of nonalcoholic fatty liver. Clin. Liver Dis. 8 501–519. [DOI] [PubMed] [Google Scholar]
- 2.Coleman R. A., and D. P. Lee. 2004. Enzymes of triacylglycerol synthesis and their regulation. Prog. Lipid Res. 43 134–176. [DOI] [PubMed] [Google Scholar]
- 3.Gibbons G. F., D. Wiggins, A. M. Brown, and A. M. Hebbachi. 2004. Synthesis and function of hepatic very-low-density lipoprotein. Biochem. Soc. Trans. 32 59–64. [DOI] [PubMed] [Google Scholar]
- 4.Grefhorst A., B. M. Elzinga, P. J. Voshol, T. Plösch, T. Kok, V. W. Bloks, F. H. van der Sluijs, L. M. Havekes, J. A. Romijn, H. J. Verkade, et al. 2002. Stimulation of lipogenesis by pharmacological activation of the liver X receptor leads to production of large, triglyceride-rich very low density lipoprotein particles. J. Biol. Chem. 277 34182–34190. [DOI] [PubMed] [Google Scholar]
- 5.Shimano H., J. D. Horton, I. Shimomura, R. E. Hammer, M. S. Brown, and J. L. Goldstein. 1997. Isoform 1c of sterol regulatory element binding protein is less active than isoform 1a in livers of transgenic mice and in cultured cells. J. Clin. Invest. 99 846–854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen G., G. Liang, J. Ou, J. L. Goldstein, and M. S. Brown. 2004. Central role for liver X receptor in insulin-mediated activation of Srebp-1c transcription and stimulation of fatty acid synthesis in liver. Proc. Natl. Acad. Sci. USA. 101 11245–11250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Uyeda K., and J. J. Repa. 2006. Carbohydrate response element binding protein, ChREBP, a transcription factor coupling hepatic glucose utilization and lipid synthesis. Cell Metab. 4 107–110. [DOI] [PubMed] [Google Scholar]
- 8.Cai D., M. Yuan, D. F. Frantz, P. A. Melendez, L. Hansen, J. Lee, and S. E. Shoelson. 2005. Local and systemic insulin resistance resulting from hepatic activation of IKK-and NF-B. Nat. Med. 11 183–190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen M. T., L. N. Kaufman, T. Spennetta, and E. Shrago. 1992. Effects of high fat-feeding to rats on the interrelationship of body weight, plasma insulin, and fatty acyl-coenzyme A esters in liver and skeletal muscle. Metabolism. 41 564–569. [DOI] [PubMed] [Google Scholar]
- 10.Samuel V. T., Z-X. Liu, X. Qu, B. D. Elder, S. Bliz, D. Befroy, A. J. Romanelli, and G. I. Shulman. 2004. Mechanism of hepatic insulin resistance in non-alcoholic fatty liver disease. J. Biol. Chem. 279 32345–32353. [DOI] [PubMed] [Google Scholar]
- 11.Holland W. L., J. T. Brozinick, L. P. Wang, E. D. Hawkins, K. M. Sargent, Y. Liu, K. Narra, K. L. Hoehn, T. A. Knotts, A. Siesky, et al. 2007. Inhibition of ceramide synthesis ameliorates glucocorticoid-, saturated-fat-, and obesity-induced insulin resistance. Cell Metab. 5 167–179. [DOI] [PubMed] [Google Scholar]
- 12.Neschen S., K. Morino, L. E. Hammond, D. Zhang, Z. X. Liu, A. J. Romanelli, G. W. Cline, R. L. Pongratz, X. M. Zhang, C. S. Choi, et al. 2005. Prevention of hepatic steatosis and hepatic insulin resistance in mitochondrial acyl-CoA:glycerol-sn-3-phosphate acyltransferase 1 knock out mice. Cell Metab. 2 55–65. [DOI] [PubMed] [Google Scholar]
- 13.Nagle C. A., J. An, M. Shiota, T. P. Torres, G. W. Cline, Z-X. Liu, S. Wang, R. L. Catlin, G. I. Shulman, C. B. Newgard, et al. 2007. Hepatic overexpression of glycerol-sn-3-phosphate acyltransferase 1 in rats causes insulin resistance. J. Biol. Chem. 282 14807–14815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McIntyre T. M., A. V. Pontsler, A. R. Silva, A. St. Hilaire, Y. Xu, J. C. Hinshaw, G. A. Zimmerman, K. Hama, J. Aoki, H. Arai, et al. 2003. Identification of an intracellular receptor for lysophosphatidic acid (LPA): LPA is a transcellular PPAR agonist. Proc. Natl. Acad. Sci. USA. 100 131–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Foster D. A. 2007. Regulation of mTOR by phosphatidic acid? Cancer Res. 67 1–4. [DOI] [PubMed] [Google Scholar]
- 16.Monetti M., M. C. Levin, M. J. Watt, M. P. Sajan, S. Marmor, B. K. Hubbard, R. D. Stevens, J. R. Bain, C. B. Newgard, R. V. Farese, Sr., et al. 2007. Dissociation of hepatic steatosis and insulin resistance in mice overexpressing DGAT in the liver. Cell Metab. 6 69–78. [DOI] [PubMed] [Google Scholar]
- 17.Minehira K., S. G. Young, C. J. Villanueva, L. Yetukuri, M. Oresic, M. K. Hellerstein, R. V. Farese, Jr., J. D. Horton, F. Preitner, B. Thorens, et al. 2008. Blocking VLDL secretion causes hepatic steatosis but does not affect peripheral lipid stores or insulin sensitivity in mice. J. Lipid Res. 49 2038–2044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Grefhorst A., J. Hoekstra, T. G. Derks, D. M. Ouwens, J. F. Baller, R. Havinga, L. M. Havekes, J. A. Romijn, and F. Kuipers. 2005. Acute hepatic steatosis in mice by blocking beta-oxidation does not reduce insulin sensitivity of very-low-density lipoprotein production. Am. J. Physiol. Gastrointest. Liver Physiol. 289 G592–G598. [DOI] [PubMed] [Google Scholar]
- 19.Matsuzaka T., H. Shimano, N. Yahagi, T. Kato, A. Atsumi, T. Yamamoto, N. Inoue, M. Ishikawa, S. Okada, N. Ishigaki, et al. 2007. Crucial role of a long-chain fatty acid elongase, Elovl6, in obesity-induced insulin resistance. Nat. Med. 13 1193–1202. [DOI] [PubMed] [Google Scholar]
- 20.Guerre-Millo M., C. Rouault, P. Poulain, J. André, V. Poitout, J. M. Peters, F. J. Gonzalez, J. C. Fruchart, G. Reach, and B. Staels. 2001. PPAR-alpha-null mice are protected from high-fat diet-induced insulin resistance. Diabetes. 50 2809–2814. [DOI] [PubMed] [Google Scholar]
- 21.Heijboer A. C., E. Donga, P. J. Voshol, Z. C. Dang, L. M. Havekes, J. A. Romijn, and E. P. Corssmit. 2005. Sixteen hours of fasting differentially affects hepatic and muscle insulin sensitivity in mice. J. Lipid Res. 46 582–588. [DOI] [PubMed] [Google Scholar]
- 22.Donnelly K. L., C. I. Smith, S. J. Schwarzenberg, J. Jessurun, M. D. Boldt, and E. J. Parks. 2005. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J. Clin. Invest. 115 1343–1351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kim J. K., J. J. Fillmore, Y. Chen, C. Yu, I. K. Moore, M. Pypaert, E. P. Lutz, Y. Kako, W. Velez-Carrasco, I. J. Goldberg, et al. 2001. Tissue-specific overexpression of lipoprotein lipase causes tissue-specific insulin resistance. Proc. Natl. Acad. Sci. USA. 98 7522–7527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dentin R., F. Benhamed, I. Hainault, V. Fauveau, F. Foufelle, J. R. Dyck, J. Girard, and C. Postic. 2006. Liver-specific inhibition of ChREBP improves hepatic steatosis and insulin resistance in ob/ob mice. Diabetes. 55 2159–2170. [DOI] [PubMed] [Google Scholar]
- 25.Yahagi N., H. Shimano, A. H. Hasty, T. Matsuzaka, T. Ide, T. Yoshikawa, M. Amemiya-Kudo, S. Tomita, H. Okazaki, Y. Tamura, et al. 2002. Absence of sterol regulatory element-binding protein-1 (SREBP-1) ameliorates fatty livers but not obesity or insulin resistance in Lep(ob)/Lep(ob) mice. J. Biol. Chem. 277 19353–19357. [DOI] [PubMed] [Google Scholar]
- 26.Savage D. B., C. S. Choi, V. T. Samuel, Z. X. Liu, D. Zhang, A. Wang, X. M. Zhang, G. W. Cline, X. X. Yu, J. G. Geisler, et al. 2006. Reversal of diet-induced hepatic steatosis and insulin resistance by antisense oligonucleotide inhibitors of acetyl-CoA carboxylases 1 and 2. J. Clin. Invest. 116 817–824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.An J., D. M. Muoio, M. Shiota, Y. Fujimoto, G. W. Cline, G. I. Shulman, T. R. Koves, R. Stevens, D. S. Millington, and C. B. Newgard. 2004. Hepatic expression of malonyl-CoA decarboxylase reverses muscle, liver and whole-animal insulin resistance. Nat. Med. 10 268–274. [DOI] [PubMed] [Google Scholar]
- 28.Chakravarthy M. V., Z. Pan, Y. Zhu, K. Tordjman, J. G. Schneider, T. Coleman, J. Turk, and C. F. Semenkovich. 2005. “New” hepatic fat activates PPARalpha to maintain glucose, lipid, and cholesterol homeostasis. Cell Metab. 1 309–322. [DOI] [PubMed] [Google Scholar]
- 29.Lindén D., L. William-Olsson, M. Rhedin, A-K. Asztély, J. C. Clapham, and S. Schreyer. 2004. Overexpression of mitochondrial glycerol-3-phosphate acyltransferase in rat hepatocytes leads to decreased fatty acid oxidation and increased glycerolipid biosynthesis. J. Lipid Res. 45 1279–1288. [DOI] [PubMed] [Google Scholar]
- 30.Xu H., D. Wilcox, P. Nguyen, M. Voorbach, T. Suhar, S. J. Morgan, W. F. An, L. Ge, J. Green, Z. Wu, et al. 2006. Hepatic knockdown of mitochondrial GPAT1 in ob/ob mice improves metabolic profile. Biochem. Biophys. Res. Commun. 349 439–448. [DOI] [PubMed] [Google Scholar]
- 31.Ide T., M. Tsunoda, T. Mochizuki, and K. Murakami. 2004. Enhancement of insulin signaling through inhibition of tissue lipid accumulation by activation of peroxisome proliferators-activated receptor (PPAR) alpha in obese mice. Med. Sci. Monit. 10 BR388–BR395. [PubMed] [Google Scholar]
- 32.Chou C. J., M. Haluzik, C. Gregory, K. R. Dietz, C. Vinson, O. Garvrilova, and M. L. Reitman. 2002. WY14,643, a peroxisome proliferator-activated receptor alpha (PPARalpha) agonist, improves hepatic and muscle steatosis and reverses insulin resistance in lipoatrophic A-ZIP/F-1 mice. J. Biol. Chem. 277 24484–24489. [DOI] [PubMed] [Google Scholar]
- 33.Leone T. C., C. J. Winheimer, and D. P. Kelly. 1999. A critical role for the peroxisome proliferator-activated receptor alpha in the cellular fasting response: the PPARalpha-null mouse as a model of fatty acid oxidation disorders. Proc. Natl. Acad. Sci. USA. 96 7473–7478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cox K. B., D. A. Hamm, D. S. Millington, D. Matern, J. Vockley, P. Rinaldo, C. A. Pinkert, W. J. Rhead, J. R. Lindsey, and P. A. Wood. 2001. Gestational, pathologic and biochemical differences between very long-chain acyl-CoA dehydrogenase deficiency and long-chain acyl-CoA dehydrogenase deficiency in the mouse. Hum. Mol. Genet. 10 2069–2077. [DOI] [PubMed] [Google Scholar]
- 35.Tolwani R., D. Hamm, L. Tian, J. Sharer, J. Vockley, P. Rinaldo, D. Matern, T. Schoeb, and P. Wood. 2005. Medium-chain acyl-CoA dehydrogenase deficiency in gene-targeted mice. PLOS Genet. 1 e23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Patsouris D., J. K. Reddy, M. Müller, and S. Kersten. 2006. Peroxisome proliferator-activated receptor alpha mediates the effects of high-fat diet on hepatic gene expression. Endocrinology. 147 1508–1516. [DOI] [PubMed] [Google Scholar]
- 37.Zhang D., Z. X. Liu, C. S. Choi, L. Tian, R. Kibbey, J. Dong, G. W. Cline, P. A. Wood, and G. I. Shulman. 2007. Mitochondrial dysfunction due to long-chain Acyl-CoA dehydrogenase deficiency causes hepatic steatosis and hepatic insulin resistance. Proc. Natl. Acad. Sci. USA. 104 17075–17080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Samuel V. T., Z. X. Liu, A. Wang, S. A. Beddow, J. G. Geisler, M. Kahn, X. M. Zhang, B. P. Monia, S. Bhanot, and G. I. Shulman. 2007. Inhibition of protein kinase Cepsilon prevents hepatic insulin resistance in nonalcoholic fatty liver disease. J. Clin. Invest. 117 739–745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hammond L. E., S. Neschen, A. J. Romanelli, G. W. Cline, O. R. Ilkayeva, G. I. Shulman, D. M. Muoio, and R. A. Coleman. 2005. Mitochondrial glycerol-3-phosphate acyltransferase-1 is essential in liver for the metabolism of excess acyl-CoAs. J. Biol. Chem. 280 25629–25636. [DOI] [PubMed] [Google Scholar]
- 40.Yazdi M., A. Ahnmark, L. William-Olsson, M. Snaith, N. Turner, F. Osla, M. Wedin, A. K. Asztély, A. Elmgren, M. Bohlooly-Y., S. Schreyer, and D. Lindén. 2008. The role of mitochondrial glycerol-3-phosphate acyltransferase-1 in regulating lipid and glucose homeostasis in high-fat diet fed mice. Biochem. Biophys. Res. Commun. 369 1065–1070. [DOI] [PubMed] [Google Scholar]
- 41.Yu X. X., S. F. Murray, S. K. Pandey, S. L. Booten, D. Bao, X. Z. Song, S. Kelly, S. Chen, R. McKay, B. P. Monia, et al. 2005. Antisense oligonucleotide reduction of DGAT2 expression improves hepatic steatosis and hyperlipidemia in obese mice. Hepatology. 42 362–371. [DOI] [PubMed] [Google Scholar]
- 42.Nieto-Vazquez I., S. Fernández-Veledo, D. K. Krämer, R. Vila-Bedmar, L. Garcia-Guerra, and M. Lorenzo. 2008. Insulin resistance associated to obesity: the link TNF-alpha. Arch. Physiol. Biochem. 114 183–194. [DOI] [PubMed] [Google Scholar]
- 43.Buettner R., K. G. Parhofer, M. Woenckhaus, C. E. Wrede, L. A. Kunz-Schughart, J. Schölmerich, and L. C. Bollheimer. 2006. Defining high-fat-diet rat models: metabolic and molecular effects of different fat types. J. Mol. Endocrinol. 36 485–501. [DOI] [PubMed] [Google Scholar]
- 44.Aerts J. M., R. Ottenhoff, A. S. Powlson, A. Grefhorst, M. van Eijk, P. F. Dubbelhuis, J. Aten, F. Kuipers, M. J. Serlie, T. Wennekes, et al. 2007. Pharmacological inhibition of glucosylceramide synthase enhances insulin sensitivity. Diabetes. 56 1341–1349. [DOI] [PMC free article] [PubMed] [Google Scholar]