Abstract
Background: Low-fat vegetarian and vegan diets are associated with weight loss, increased insulin sensitivity, and improved cardiovascular health.
Objective: We compared the effects of a low-fat vegan diet and conventional diabetes diet recommendations on glycemia, weight, and plasma lipids.
Design: Free-living individuals with type 2 diabetes were randomly assigned to a low-fat vegan diet (n = 49) or a diet following 2003 American Diabetes Association guidelines (conventional, n = 50) for 74 wk. Glycated hemoglobin (Hb A1c) and plasma lipids were assessed at weeks 0, 11, 22, 35, 48, 61, and 74. Weight was measured at weeks 0, 22, and 74.
Results: Weight loss was significant within each diet group but not significantly different between groups (−4.4 kg in the vegan group and −3.0 kg in the conventional diet group, P = 0.25) and related significantly to Hb A1c changes (r = 0.50, P = 0.001). Hb A1c changes from baseline to 74 wk or last available values were −0.34 and −0.14 for vegan and conventional diets, respectively (P = 0.43). Hb A1c changes from baseline to last available value or last value before any medication adjustment were −0.40 and 0.01 for vegan and conventional diets, respectively (P = 0.03). In analyses before alterations in lipid-lowering medications, total cholesterol decreased by 20.4 and 6.8 mg/dL in the vegan and conventional diet groups, respectively (P = 0.01); LDL cholesterol decreased by 13.5 and 3.4 mg/dL in the vegan and conventional groups, respectively (P = 0.03).
Conclusions: Both diets were associated with sustained reductions in weight and plasma lipid concentrations. In an analysis controlling for medication changes, a low-fat vegan diet appeared to improve glycemia and plasma lipids more than did conventional diabetes diet recommendations. Whether the observed differences provide clinical benefit for the macro- or microvascular complications of diabetes remains to be established. This trial was registered at clinicaltrials.gov as NCT00276939.
INTRODUCTION
Low-fat vegetarian and vegan diets are associated with reduced body weight, increased insulin sensitivity, and reductions in cardiovascular risk factors (1–3). The potential cardiovascular benefits of vegetarian and vegan diets may be especially important for individuals with diabetes, for whom cardiovascular disease is a main cause of premature mortality; the effects of such diets on cardiovascular risk factors appear to be similar in individuals with and without diabetes (3).
Prior studies have shown that near-vegetarian diets reduce the need for insulin and oral medications in individuals with type 2 diabetes (4, 5). We previously reported that in individuals with type 2 diabetes, a low-fat, vegan diet was associated with improved glycemic control, weight loss, and improved plasma lipid control during a 22-wk study period (6). What is particularly critical in diabetes management is long-term improvement in clinical measures, particularly glycemia and cardiovascular risk factors. Well-planned low-fat vegan diets are nutritionally adequate (7) and, in research studies, have shown acceptability comparable with that of other therapeutic diets (8–10), suggesting they are suitable for long-term use. We therefore followed these study participants for an additional year and now report the effects of a low-fat vegan diet and a diet based on 2003 American Diabetes Association (ADA) guidelines after 74 wk of study.
SUBJECTS AND METHODS
Participants
The study methods have been previously described (6). Briefly, individuals with type 2 diabetes, defined by a fasting plasma glucose concentration >125 mg/dL on 2 occasions or a prior diagnosis of type 2 diabetes treated with medications for blood glucose control for ≥6 mo, were recruited through newspaper advertisements in the Washington, DC, area on 2 occasions (October–December 2003 and October–December 2004) to complete the study from January 2004 through June 2005 and January 2005 through June 2006, respectively. Exclusionary criteria were a glycated hemoglobin (Hb A1c) <6.5% or >10.5%; use of insulin for >5 y; smoking, alcohol, or drug abuse; pregnancy; unstable medical status; and current use of a low-fat vegetarian diet. The protocol was approved by the George Washington University Institutional Review Board. All participants gave written informed consent.
Hb A1c was assayed with the use of affinity chromatography on an Abbott IMx analyzer (Abbott Laboratories, Abbott Park, IL) (11). Volunteers were ranked in order of Hb A1c concentrations and randomly assigned in sequential pairs, using a random-number table, to a low-fat vegan diet or a diet following 2003 ADA guidelines (12) (conventional diet group).
Intervention
The prescribed vegan diet (≈10% of energy from fat, 15% protein, 75% carbohydrate) consisted of vegetables, fruit, grains, and legumes. Participants were asked to 1) avoid animal products (ie, meats, dairy products, eggs); 2) avoid fatty foods, such as added oils, fried products, avocados, nuts, and seeds; and 3) favor low–glycemic index foods, such as beans and green vegetables. These diet changes increase dietary fiber and complex carbohydrate at the expense of total and saturated fat, cholesterol, and animal protein. Portion sizes, energy intake, and carbohydrate intake were unrestricted.
The conventional diet (15–20% protein, <7% saturated fat, 60–70% carbohydrate and monounsaturated fats; cholesterol ≤200 mg/d) was individualized, based on body weight and plasma lipid concentrations, following 2003 ADA guidelines (12). Participants in the conventional group with a body mass index (in kg/m2) >25 (all but 3 participants) were prescribed energy intake deficits of 500–1000 kcal.
Participants were free living, and no meals were provided. To meet the vitamin B-12 needs of the vegan group while maintaining the same intervention in the conventional diet group, all participants were provided a vitamin B-12 supplement (100 μg) to be taken every other day. For both groups, alcoholic beverages were limited to 1/d for women and 2/d for men. Participants were asked not to alter exercise habits during the first 22 wk of the study, but they were free to alter their exercise regimens thereafter.
Each participant met for 1 h with a registered dietitian experienced in the use of the assigned diet to establish an appropriate diet plan. Thereafter, participants attended weekly 1-h meetings of their assigned groups for nutrition and cooking instruction conducted by a physician and a registered dietitian or a cooking instructor for 22 wk, followed by optional biweekly sessions for an additional 52 wk. Sessions for the 2 groups followed established curricula that were similar in duration and content, except for dietary details.
At 7 points during the trial (weeks 4, 8, 13, 20, 33, 45, and 60), a registered dietitian made unannounced telephone calls to each participant to administer a 24-h diet recall, using a multipass approach (Food and Nutrient Database 35, released May 2004, software version 5.0; Nutrition Coordinating Center, University of Minnesota, Minneapolis, MN) (13) and reported instances of poor dietary adherence to the dietitians responsible for the initial dietary instruction for additional dietary counseling as needed.
In addition, a 3-d dietary record was completed by each participant at weeks 0, 11, 22, and 74 on 2 weekdays and 1 weekend day, using a food scale, after participants had completed a 3-d practice record. With the use of the NUTRITION DATA SYSTEM FOR RESEARCH software (13), a registered dietitian certified by the Nutrition Coordinating Center analyzed all 3-d dietary records and diet recalls. For statistical purposes, dietary adherence for the vegan group was defined by 3 criteria: 1) the absence of meat, poultry, fish, dairy, or egg intake in any 24-h recalls, 3-d dietary records, or incidentally at any point; 2) saturated fat ≤5% and total fat ≤25% of energy on 3-d dietary records at 22 and 74 wk; and 3) mean daily cholesterol intake ≤50 mg on 3-d dietary records at 22 and 74 wk. Adherence for the conventional diet group was defined, based on the 22- and 74-wk 3-d dietary records, as 1) mean daily energy intake ≤200 kcal in excess of the prescribed intake and 2) saturated fat ≤10% of energy. Individuals who attended <10 of the first 22 weekly sessions were also considered nonadherent on either diet. These adherence criteria were used for descriptive purposes (not for participant teaching) and were not used in the primary outcome analysis.
Participants were asked to continue their preexisting medication regimens, except when fasting plasma glucose determinations were <80 mg/dL (4.4 mmol/L) or hypoglycemic symptoms were accompanied by a capillary glucose reading <65 mg/dL (3.6 mmol/L). In such cases, medications were reduced for participant safety by a study endocrinologist who remained blind to group assignment. Dosage adjustments followed an established protocol, which generally called for 20% dosage reductions for participants treated with insulin and 50% dosage reductions for participants on oral agents. For those individuals for whom Hb A1c at 22 wk was >8%, a single medication adjustment was made, based on an established protocol, as follows: 1) participants on insulin were prescribed a 10–20% increased dose, 2) participants treated with oral agents could be raised to the maximal recommended dosages, and 3) participants on no medications were prescribed 2 mg/d glimepiride. Because medication changes confound the interpretation of the effect of diet on glycemia, analyses were designed to account for this factor to the extent possible, as described below.
Outcome measures
Laboratory measurements were made after a 12-h fast by technicians blind to group assignment. Hb A1c, plasma glucose, and plasma lipids were assayed at weeks 0, 11, 22, 35, 48, 61, and 74. All other measures were assessed at weeks 0, 22, and 74, except as noted. Plasma glucose, cholesterol, triglyceride, and urinary albumin were measured with the use of laboratory methods previously described (6).
Physical activity was assessed over 3-d periods at weeks 0, 22, and 74 by pedometer (Omron HJ-112; Omron Healthcare Inc, Bannockburn, IL) and with the Bouchard 3-Day Physical Activity Record (14). At weeks 1, 11, 22, and 74, body weight, waist and hip circumference, and blood pressure were measured as previously described (6).
Statistical analyses
To have an 80% chance of detecting as significant (at the 2-sided 5% level) a 1.5-point between-group Hb A1c difference, with an assumed SD of 1.9 and a loss to follow-up of 26%, 34 participants were required per group. An interim analysis indicated group differences of 0.8 with SD of 1.3, so a revised power analysis was conducted. To have an 80% chance of detecting a 0.8 difference as significant with an SD of 1.3 and loss to follow-up of 33%, an additional 15 participants were required per group. Data for the 2 cohorts were combined, such that the 74-wk observations were collected 74 wk after baseline determinations for all participants.
The primary analysis of Hb A1c included all participants based on intention to treat. Repeated-measures analysis of variance was performed to see whether there were effects associated with time (within-person variable), diet group (between-group variable), or the interaction of time and diet group. The dependent variable for the repeated-measures analysis was the Hb A1c value at baseline and weeks 11, 22, 35, 48, 61, and 74.
Between-subjects t tests were calculated for Hb A1c, and all other dependent measures to determine whether changes associated with the intervention diet were greater than those associated with the control diet. Within each diet group, paired comparison t tests were calculated to test whether the change from baseline to 74 wk, or last available value, was significantly different from zero. In cases of missing Hb A1c values, the last available values were brought forward. Because medication changes influence the dependent measures, additional analyses were done by carrying forward the last values before any medication changes for individuals whose medications for glucose or lipid control had changed, and by limiting the analysis to individuals with no medication changes. For body weight, additional analyses were conducted with the use of baseline weights for missing values. An α of 0.05 was used for all statistical tests, with no adjustment for multiple comparisons.
Regression analyses assessed whether the diet group effects on Hb A1c and body weight were significant while controlling for baseline values, and whether the diet group effect on Hb A1c was significant while controlling for baseline Hb A1c and changes in body weight. Pearson's correlation coefficients were calculated for the relation between HB A1c change and weight change. Between-group differences in diet adherence were analyzed with the use of the chi-square test. The software used was SAS for Windows, version 8.2 (SAS Institute Inc, Cary, NC).
RESULTS
Of 1049 individuals screened by telephone, 99 met participation criteria and were randomly assigned to the vegan (n = 49) or conventional diet (n = 50) groups. Reasons for exclusion were Hb A1c values outside the required range (n = 201), failure to meet other participation criteria (n = 279), inability to attend scheduled meetings (n = 187), failure to keep interview appointment (n = 153), reluctance to change diet (n = 72), and other or unspecified (n = 58). Demographic characteristics reflected those of the Washington, DC, area (Table 1). Seven vegan and 5 conventional diet participants failed to complete 74-wk laboratory assessments. Nine vegan and 7 conventional diet participants failed to complete 74-wk dietary records. No significant baseline clinical or demographic differences were observed between these individuals and study completers.
TABLE 1.
Selected baseline demographic and clinical characteristics of study participants1
| Vegan group (n = 49) | Conventional diet group (n = 50) | P2 | |
| Age (y) | 56.7 ± 9.8 (35–82)3 | 54.6 ± 10.2 (27–80) | 0.29 |
| Sex [n (%)] | 0.26 | ||
| Male | 22 (45) | 17 (34) | |
| Female | 27 (55) | 33 (66) | |
| Race, ethnicity [n (%)] | 0.714 | ||
| Black, non-Hispanic | 22 (45) | 22 (44) | |
| White, non-Hispanic | 21 (47) | 22 (44) | |
| White, Hispanic | 4 (8) | 2 (4) | |
| Asian, non-Hispanic | 2 (4) | 4 (8) | |
| Education [n (%)] | 0.69 | ||
| High school, partial or graduate | 6 (12) | 3 (6) | |
| College, partial or graduate | 26 (53) | 25 (50) | |
| Graduate degree | 17 (35) | 22 (44) | |
| Years since diabetes diagnosis | 8.6 ± 6.85 | 8.5 ± 6.1 | 0.96 |
| On insulin [n (%)] | 11 (22) | 5 (10) | 0.09 |
| Glargine | 6 (12) | 3 (6) | |
| Premixed insulin | 3 (6) | 1 (2) | |
| NPH | 1 (2) | 1 (2) | |
| Glargine + rapid-acting analog | 1 (2) | 0 (0) | |
| On metformin [n (%)] | 34 (69) | 39 (78) | 0.33 |
| On sulfonylurea [n (%)] | 25 (51) | 29 (58) | 0.49 |
| On thiazolidinedione [n (%)] | 16 (33) | 15 (30) | 0.78 |
| On other diabetes medications [n (%)] | 1 (2) | 2 (4) | 0.57 |
| On blood pressure medications [n (%)] | 31 (63) | 38 (76) | 0.17 |
| On lipid-lowering medications [n (%)] | 27 (55) | 27 (54) | 0.88 |
| History of eye involvement [n (%)] | 9 (18) | 10 (20) | 0.82 |
| History of renal involvement [n (%)] | 6 (12) | 4 (8) | 0.48 |
| History of neuropathy [n (%)] | 18 (37) | 24 (48) | 0.25 |
| BMI (kg/m2) | 33.9 ± 7.8 | 35.9 ± 7.0 | 0.18 |
| BMI [n (%)] | |||
| <25 kg/m2 | 5 (10) | 2 (4) | |
| 25–29.9 kg/m2 | 14 (29) | 5 (10) | |
| ≥30 kg/m2 | 30 (61) | 43 (86) |
NPH, neutral protamine Hagedorn (an intermediate-acting insulin).
Refers to t tests for continuous variables and chi-square test for categorical variables.
Mean ± SD; range in parentheses (all such values).
Calculated for race distribution; for ethnicity (Hispanic compared with non-Hispanic), P = 0.39.
Mean ± SD (all such values).
Dietary intake, adherence, and medication changes
Both groups reduced energy intake (Table 2). The decreases in total, saturated, monounsaturated, and trans fat intake and in cholesterol intake, expressed in relation to energy intake, were significantly greater in the vegan group. Carbohydrate and fiber intakes increased in the vegan group, with smaller changes in these variables in the conventional diet group. For micronutrients, compared with the conventional diet group, the vegan group reported greater increases in vitamin C, folate, magnesium, and iron intakes and greater reductions in vitamin D, calcium, and zinc intakes. Increases in fruit and vegetable intake were greater in the vegan group. Pedometer readings and self-reported energy expenditure showed no significant between-group differences. At 22 wk, all group-specific dietary adherence criteria were met by 67% (33 of 49) of participants in the vegan group and 44% (22 of 50) of participants in the conventional diet group. At 74 wk, all dietary adherence criteria were met by 51% (25 of 49) of participants in the vegan group and 48% (24 of 50) of participants in the conventional diet group.
TABLE 2.
Nutrient, fruit, and vegetable intakes for individuals completing 74 wk of study1
| Vegan group (n = 40) |
Conventional diet group (n = 43) |
|||||||
| Baseline | Final | Change | Baseline | Final | Change | Effect size2 | P3 | |
| Energy (kcal) | 1798 ± 724 | 1366 ± 81 | −432 ± 815 | 1840 ± 91 | 1422 ± 65 | −418 ± 795 | −14 (−239 to 211) | 0.90 |
| Fat (% of energy) | 36.3 ± 1.3 | 22.3 ± 1.4 | −14.0 ± 1.85 | 34.7 ± 1.2 | 33.7 ± 1.3 | −1.0 ± 1.7 | −13.0 (−18.0 to −8.1) | <0.0001 |
| Saturated fat (% of energy) | 11.9 ± 0.5 | 5.1 ± 0.5 | −6.7 ± 0.75 | 10.8 ± 0.5 | 9.9 ± 0.5 | −0.9 ± 0.6 | −5.9 (−7.6 to −4.1) | <0.0001 |
| Monounsaturated fat (% of energy) | 14.1 ± 0.70 | 8.2 ± 0.7 | −5.9 ± 0.9 5 | 13.7 ± 0.6 | 13.1 ± 0.7 | −0.6 ± 0.8 | −5.3 (−7.7 to −3.0) | <0.0001 |
| Polyunsaturated fat (% of energy) | 7.4 ± 0.4 | 7.0 ± 0.4 | −0.4 ± 0.5 | 7.1 ± 0.3 | 7.7 ± 0.5 | 0.5 ± 0.5 | −0.9 (−2.4 to 0.6) | 0.25 |
| trans Fat (% of energy) | 2.3 ± 0.2 | 1.1 ± 0.1 | −1.2 ± 0.25 | 2.0 ± 0.1 | 1.7 ± 0.2 | −0.3 ± 0.2 | −0.9 (−1.4 to −0.3) | 0.002 |
| Cholesterol (mg/1000 kcal) | 159.0 ± 15.9 | 36.3 ± 8.8 | −122.7 ± 16.95 | 168.2 ± 10.8 | 170.3 ± 13.4 | +2.2 ± 12.5 | −124.9 (−166.2 to −83.6) | <0.0001 |
| Carbohydrate (% of energy) | 47.7 ± 1.7 | 66.3 ± 1.8 | +18.6 ± 2.45 | 46.3 ± 1.4 | 46.5 ± 1.6 | +0.2 ± 1.9 | 18.5 (12.5 to 24.4) | <0.0001 |
| Protein (% of energy) | 17.0 ± 0.6 | 14.8 ± 0.5 | −2.2 ± 0.76 | 19.0 ± 0.6 | 21.1 ± 0.7 | +2.1 ± 0.97 | −4.3 (−6.6 to −2.0) | 0.0003 |
| Total fiber (g/1000 kcal) | 10.8 ± 0.7 | 21.7 ± 1.2 | +10.8 ± 1.15 | 11.0 ± 0.7 | 13.4 ± 0.8 | +2.4 ± 0.96 | 8.5 (5.8 to 11.2) | <0.0001 |
| Soluble fiber (g/1000 kcal) | 2.9 ± 0.2 | 5.5 ± 0.3 | 2.5 ± 0.35 | 2.9 ± 0.2 | 3.3 ± 0.2 | 0.4 ± 0.3 | 2.1 (1.3 to 2.9) | <0.0001 |
| Insoluble fiber (g/1000 kcal) | 7.8 ± 0.5 | 16.0 ± 1.0 | 8.3 ± 0.85 | 8.0 ± 0.5 | 10.0 ± 0.6 | 2.0 ± 0.66 | 6.3 (4.2 to 8.4) | <0.0001 |
| Total vitamin A activity (IU/1000 kcal) | 5129 ± 659 | 8188 ± 1360 | 3059 ± 14017 | 5653 ± 803 | 7276 ± 711 | 1624 ± 1132 | 1435 (−2127 to 4997) | 0.43 |
| Vitamin D (μg/1000 kcal) | 2.4 ± 0.2 | 1.5 ± 0.2 | −0.9 ± 0.36 | 2.9 ± 0.3 | 3.2 ± 0.3 | 0.3 ± 0.5 | −1.2 (−2.3 to −0.03) | 0.04 |
| Vitamin E (α-tocopherol) (mg/1000 kcal) | 5.6 ± 1.7 | 5.1 ± 0.3 | −0.5 ± 1.7 | 4.0 ± 0.3 | 4.9 ± 0.4 | 0.9 ± 0.5 | −1.4 (−4.8 to 2.0) | 0.44 |
| Vitamin K (μg/1000 kcal) | 71.3 ± 9.8 | 154.9 ± 25.6 | 83.7 ± 27.06 | 94.5 ± 12.6 | 148.4 ± 18.4 | 53.9 ± 19.66 | 29.7 (−36.0 to 95.4) | 0.37 |
| Vitamin C (mg/1000 kcal) | 44.3 ± 4.7 | 88.6 ± 8.8 | 44.3 ± 8.25 | 50.3 ± 5.4 | 69.0 ± 6.1 | 18.7 ± 7.67 | 25.6 (3.3 to 47.9) | 0.03 |
| Vitamin B-6 (mg/1000 kcal) | 1.0 ± 0.1 | 1.2 ± 0.1 | 0.2 ± 0.16 | 1.0 ± 0.1 | 1.2 ± 0.1 | 0.2 ± 0.17 | −0.04 (−0.3 to 0.2) | 0.74 |
| Folate (μg/1000 kcal) | 260 ± 18 | 363 ± 18 | 103 ± 248 | 249 ± 14 | 265 ± 14 | 16 ± 20 | 87 (25 to 149) | 0.007 |
| Vitamin B-12 (μg/1000 kcal) | 3.2 ± 0.4 | 1.9 ± 0.4 | −1.2 ± 0.67 | 3.8 ± 0.7 | 3.9 ± 0.5 | 0.1 ± 0.9 | −1.4 (−3.6 to 0.9) | 0.21 |
| Calcium (mg/1000 kcal) | 432 ± 29 | 412 ± 19 | −20 ± 33 | 380 ± 21 | 478 ± 32 | 97 ± 326 | −118 (−209 to −26) | 0.01 |
| Magnesium (mg/1000 kcal) | 159 ± 10 | 243 ± 11 | 84 ± 115 | 161 ± 7 | 195 ± 10 | 34 ± 116 | 50 (20 to 80) | 0.001 |
| Iron (mg/1000 kcal) | 8.7 ± 0.6 | 11.5 ± 0.5 | 2.8 ± 0.78 | 8.4 ± 0.4 | 8.4 ± 0.4 | 0.0 ± 0.6 | 2.8 (1.1 to 4.6) | 0.002 |
| Zinc (mg/1000 kcal) | 6.0 ± 0.4 | 5.2 ± 0.2 | −0.8 ± 0.4 | 6.1 ± 0.3 | 6.7 ± 0.4 | 0.6 ± 0.5 | −1.5 (−2.7 to −0.2) | 0.02 |
| Sodium (mg/1000 kcal) | 1824 ± 63 | 1840 ± 100 | 16 ± 119 | 1989 ± 88 | 1854 ± 62 | −134 ± 97 | 151 (−152 to 453) | 0.32 |
| Potassium (mg/1000 kcal) | 1374 ± 55 | 1973 ± 87 | 599 ± 815 | 1414 ± 61 | 1780 ± 77 | 365 ± 908 | 234 (−8 to 475) | 0.06 |
| Fruit (servings/d)9 | 1.3 ± 0.2 | 2.8 ± 0.3 | 1.6 ± 0.35 | 1.4 ± 0.1 | 2.0 ± 0.2 | 0.5 ± 0.27 | 1.0 (0.3 to 1.8) | 0.005 |
| Vegetables (servings/d)10 | 2.5 ± 0.2 | 4.7 ± 0.5 | 2.2 ± 0.55 | 3.2 ± 0.2 | 3.6 ± 0.4 | 0.4 ± 0.4 | 1.9 (0.6 to 3.1) | 0.004 |
Data are for all participants who completed dietary records at both baseline and 74 wk and do not include dietary supplements.
Values in this column represent differences between the mean change scores of the vegan group and those of the conventional diet group; 95% CIs in parentheses.
Refers to t tests for between-group (vegan compared with conventional) comparisons of changes from baseline to final values.
Mean ± SE (all such values).
Significantly different from baseline (within-group t test comparison): 5P < 0.0001, 6P < 0.01, 7P < 0.05, 8P < 0.001.
Fruit servings were defined as one medium whole fruit; 1/2 cup chopped, cooked, or canned fruit; 1/4 cup dried fruit; or 1/2 cup fruit juice. Fruit-based savory snacks were not included.
Vegetable servings were defined as 1 cup raw leafy vegetables, 1/2 cup other cooked or raw vegetables or legumes, or 1/2 cup vegetable juice. White potatoes, fried potatoes, and vegetable-based savory snacks were not included.
During the 74-wk study period, 71% (35 of 49) of participants in the vegan group and 58% (29 of 50) of participants in the conventional diet group altered their diabetes medications, either as prescribed by the study protocol or without investigators' authorization. Net 74-wk dosages were reduced in 17 (35%) participants in the vegan group and 10 (20%) participants in the conventional diet group, were increased in 7 (14%) participants in the vegan group and 12 (24%) participants in the conventional diet group, and were unchanged or mixed (changes in opposite directions in ≥2 medications) in the remainder.
Body weight
When data from all participants from baseline to 74 wk or last available value were included, both diets were associated with significant sustained weight reduction (−4.4 kg in the vegan group and −3.0 kg in the conventional diet group), with no significant difference between groups (P = 0.25) (Table 3). Using baseline values for missing values did not substantially change the outcome (vegan group: −3.7 kg; conventional diet group: −2.6 kg; between-group P = 0.36). Limiting the analysis to participants who met dietary adherence criteria at 22 and 74 wk (21 vegan, 16 conventional), weight loss was 6.8 kg in the vegan group and 4.9 kg in the conventional diet group (P = 0.45).
TABLE 3.
Clinical outcomes, based on intention-to-treat analyses1
| Vegan group (n = 49, except as noted) |
Conventional diet group (n = 50, except as noted) |
|||||||
| Baseline | Final2 | Change | Baseline | Final2 | Change | Effect size3 | P4 | |
| Weight (kg) | 97.0 ± 3.35 | 92.6 ± 3.5 | −4.4 ± 0.96 | 99.3 ± 3.0 | 96.3 ± 3.2 | −3.0 ± 0.87 | −1.4 (−3.8 to 1.0) | 0.25 |
| BMI (kg/m2) | 33.9 ± 1.1 | 32.3 ± 1.2 | −1.6 ± 0.36 | 35.9 ± 1.0 | 34.8 ± 1.1 | −1.1 ± 0.37 | −0.5 (−1.4 to 0.4) | 0.25 |
| Waist (cm) | 110.8 ± 2.6 | 106.6 ± 2.8 | −4.2 ± 1.07 | 112.3 ± 2.1 | 110.5 ± 2.1 | −1.8 ± 0.88 | −2.4 (−4.9 to 0.1) | 0.06 |
| Hip (cm) | 118.4 ± 2.5 | 115.0 ± 2.6 | −3.4 ± 0.76 | 121.3 ± 1.8 | 119.0 ± 1.8 | −2.3 ± 0.79 | −1.1 (−3.0 to 0.8) | 0.24 |
| Waist-hip ratio | 0.94 ± 0.01 | 0.93 ± 0.01 | −0.01 ± 0.01 | 0.93 ± 0.01 | 0.93 ± 0.01 | 0.00 ± 0.01 | −0.01 (−0.03 to 0.00) | 0.15 |
| Hb A1c last available value (%) | 8.05 ± 0.16 | 7.71 ± 0.19 | −0.34 ± 0.19 | 7.93 ± 0.14 | 7.79 ± 0.18 | −0.14 ± 0.17 | −0.20 (−0.71 to 0.30) | 0.43 |
| Fasting plasma glucose (mg/dL) | 163.5 ± (7.6 | 144.0 ± 7.7) | −19.5 ± 7.19 | 160.4 ± 5.8 | 146.4 ± 8.03 | −14.0 ± 8.2 | −5.5 (−27.1 to 16.0) | 0.61 |
| Total cholesterol (mg/dL) | 187.0 ± 5.3 | 165.5 ± 4.5 | −21.6 ± 4.26 | 198.9 ± 6.2 | 184.1 ± 5.5 | −14.8 ± 5.19 | −6.8 (−19.9 to 6.4) | 0.31 |
| Non-HDL cholesterol (mg/dL) | 134.7 ± 5.6 | 113.1 ± 4.5 | −21.6 ± 4.26 | 149.0 ± 6.2 | 135.5 ± 5.5 | −13.5 ± 5.18 | −8.1 (−21.3 to 5.1) | 0.23 |
| LDL cholesterol (mg/dL; n = 49 conventional) | 104.4 ± 4.7 | 90.9 ± 4.4 | −13.5 ± 4.39 | 117.7 ± 5.9 | 108.3 ± 5.4 | −9.4 ± 4.58 | −4.1 (−16.4 to 8.2) | 0.51 |
| HDL cholesterol (mg/dL) | 52.3 ± 2.8 | 51.3 ± 2.6 | −1.0 ± 1.0 | 49.8 ± 2.1 | 48.6 ± 2.1 | −1.3 ± 1.7 | 0.3 (−3.7 to 4.2) | 0.89 |
| Total cholesterol-to-HDL ratio | 4.0 ± 0.2 | 3.6 ± 0.2 | −0.4 ± 0.17 | 4.3 ± 0.2 | 4.1 ± 0.2 | −0.2 ± 0.2 | −0.2 (−0.6 to 0.2) | 0.31 |
| VLDL cholesterol (mg/dL; n = 47 vegan, n = 47 conventional) | 26.2 ± 2.1 | 21.6 ± 1.6 | −4.5 ± 1.39 | 26.8 ± 2.0 | 22.9 ± 1.4 | −3.9 ± 1.9 | −0.7 (−5.3 to 0.1) | 0.77 |
| Triglycerides (mg/dL) | 148.1 ± 16.1 | 114.2 ± 9.7 | −33.9 ± 12.78 | 158.1 ± 18.8 | 150.3 ± 28.8 | −7.8 ± 28.9 | −26.1 (−89.2 to 37.0) | 0.41 |
| Log triglycerides | 2.08 ± 0.04 | 1.99 ± 0.03 | −0.09 ± 0.039 | 2.12 ± 0.03 | 2.07 ± 0.04 | −0.05 ± 0.03 | −0.04 (−0.12 to 0.05) | 0.37 |
| Urinary albumin/24 h (mg) | 33.0 ± 7.4 | 20.2 ± 4.6 | −12.8 ± 7.0 | 55.0 ± 37.2 | 69.5 ± 47.2 | 14.6 ± 12.0 | −27.4 (−55.1 to 0.3) | 0.05 |
| Log albumin/24 h | 1.17 ± 0.07 | 1.04 ± 0.06 | −0.13 ± 0.058 | 1.09 ± 0.07 | 1.05 ± 0.08 | −0.04 ± 0.05 | −0.09 (−0.22 to 0.04) | 0.18 |
| C-reactive protein (mg/L) | 4.6 ± 0.8 | 2.6 ± 0.5 | −1.9 ± 0.69 | 6.9 ± 1.4 | 4.5 ± 0.9 | −2.4 ± 0.89 | −5.0 (−1.5 to 2.5) | 0.65 |
| Blood pressure, systolic (mm Hg; n = 48 vegan)10 | 123.8 ± 2.5 | 123.8 ± 2.4 | −0.0 ± 2.0 | 122.9 ± 2.1 | 126.6 ± 2.4 | 3.7 ± 1.9 | −3.7 (−9.2 to 1.8) | 0.19 |
| Blood pressure, diastolic (mm Hg; n = 48 vegan)10 | 77.9 ± 1.6 | 74.0 ± 1.4 | −3.9 ± 1.39 | 80.0 ± 1.5 | 77.3 ± 1.5 | −2.7 ± 1.18 | −1.2 (−4.6 to 2.2) | 0.48 |
Hb A1c, glycated hemoglobin. To convert HDL, LDL, and total cholesterol to SI units (mmol/L), multiply by 0.0259; to convert triglycerides to SI units (mmol/L), multiply by 0.0113.
Final values are last available values, based on intention-to-treat analyses.
Values in this column represent differences between the mean change scores of the vegan group and those of the conventional diet group; 95% CIs in parentheses.
Refers to t tests for between-group (vegan compared with conventional) comparisons of changes from baseline to final values.
Mean ± SE (all such values).
Significantly different from baseline (within-group t test comparison): 6P < 0.0001, 7P < 0.001, 8P < 0.05, 9P < 0.01.
Blood pressure was not determined for one participant in the vegan group because of equipment failure.
Glycemic control
For Hb A1c, the repeated-measures analysis of variance showed significant effects for time (P < 0.0001) and for diet group-by-time interaction (P = 0.03). The effects for time were both linear and curvilinear. The group-by-time interaction was followed with t test comparisons between groups for changes from baseline to 74 wk. In an intention-to-treat analysis, including all participants without regard to medication adjustments, Hb A1c changes from baseline to 74 wk or to the last available value were −0.34 for the vegan group and −0.14 for the conventional diet group (P = 0.43) (Table 3).
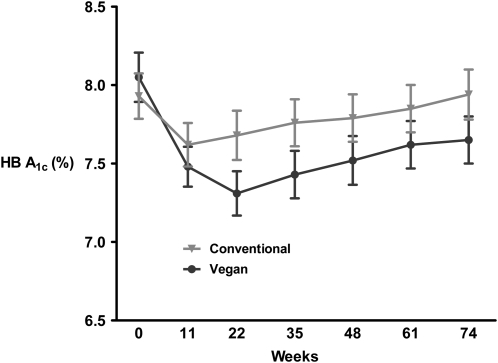
In an effort to remove the confounding effect of medication changes, 2 additional analyses were conducted. Hb A1c changes from baseline to 74 wk or to the last available value before any medication adjustment were −0.40 and 0.01 in vegan and conventional diet groups, respectively (P = 0.03) (Table 4; Figure 1). Among 74-wk completers whose medications remained unchanged throughout (14 vegan, 21 conventional), Hb A1c changes were −0.82 (vegan; within-group P = 0.04) and −0.21 (conventional; within-group P = 0.35) (P = 0.14).
TABLE 4.
Glycemia, plasma lipids, and blood pressure before medication changes1
| Vegan group (n = 49, except as noted) |
Conventional diet group (n = 50, except as noted) |
|||||||
| Baseline | Final2 | Change | Baseline | Final | Change | Effect size3 | P4 | |
| Hb A1c (%) | 8.05 ± 0.165 | 7.65 ± 0.15 | −0.40 ± 0.146 | 7.93 ± 0.14 | 7.94 ± 0.16 | +0.01 ± 0.13 | −0.41 (−0.78 to −0.04) | 0.03 |
| Fasting plasma glucose (mg/dL) | 163.5 ± 7.6 | 149.4 ± 8.1 | −14.1 ± 5.87 | 160.4 ± 5.8 | 153.9 ± 8.1 | −6.5 ± 6.7 | −7.6 (−25.3 to 10.1) | 0.40 |
| Total cholesterol (mg/dL) | 187.0 ± 5.3 | 166.6 ± 4.0 | −20.4 ± 3.38 | 198.9 ± 6.2 | 192.1 ± 6.4 | −6.8 ± 4.3 | −13.7 (−24.4 to −2.9) | 0.01 |
| Non-HDL cholesterol (mg/dL) | 134.7 ± 5.6 | 115.2 ± 4.2 | −19.5 ± 2.98 | 149.0 ± 6.2 | 142.9 ± 6.3 | −6.1 ± 4.6 | −13.4 (−24.2 to −2.6) | 0.02 |
| LDL cholesterol (mg/dL; n = 49 conventional) | 104.4 ± 4.7 | 90.8 ± 3.7 | −13.5 ± 2.68 | 117.7 ± 5.9 | 114.3 ± 5.9 | −3.4 ± 3.9 | −10.1 (−19.4 to −0.8) | 0.03 |
| HDL cholesterol (mg/dL) | 52.3 ± 2.8 | 51.4 ± 2.8 | −0.9 ± 1.1 | 49.8 ± 2.1 | 49.2 ± 2.1 | −0.7 ± 1.7 | −0.3 (−4.3 to 3.8) | 0.90 |
| Total cholesterol-to-HDL ratio | 4.0 ± 0.2 | 3.6 ± 0.2 | −0.4 ± 0.19 | 4.3 ± 0.2 | 4.2 ± 0.2 | −0.1 ± 0.2 | −0.3 (−0.7 to 0.1) | 0.14 |
| VLDL cholesterol (mg/dL; n = 47 vegan, n = 47 conventional) | 26.2 ± 2.1 | 22.2 ± 1.7 | −3.9 ± 1.36 | 26.8 ± 2.0 | 24.6 ± 1.8 | −2.2 ± 1.8 | −1.8 (−6.2 to 2.7) | 0.43 |
| Triglycerides (mg/dL) | 148.1 ± 16.1 | 120.0 ± 10.2 | −28.1 ± 10.36 | 158.1 ± 18.8 | 158.1 ± 29.1 | 0.0 ± 28.7 | −28.1 (−89.0 to 32.9) | 0.36 |
| Log triglycerides | 2.08 ± 0.04 | 2.01 ± 0.03 | −0.07 ± 0.026 | 2.12 ± 0.04 | 2.08 ± 0.04 | −0.03 ± 0.03 | −0.03 (−0.11 to 0.05) | 0.41 |
| Blood pressure, systolic (mm Hg; n = 48 vegan)10 | 123.8 ± 2.5 | 123.3 ± 2.5 | −0.6 ± 1.5 | 122.9 ± 2.1 | 121.8 ± 2.2 | −1.1 ± 1.3 | 0.5 (−3.4 to 4.5) | 0.79 |
| Blood pressure, diastolic (mm Hg; n = 48 vegan)10 | 77.9 ± 1.6 | 75.1 ± 1.5 | −2.8 ± 1.17 | 80.0 ± 1.5 | 77.7 ± 1.4 | −2.3 ± 1.17 | −0.5 (−3.6 to 2.5) | 0.73 |
Hb A1c, glycated hemoglobin. To convert HDL, LDL, and total cholesterol to SI units (mmol/L), multiply by 0.0259; to convert triglycerides to SI units (mmol/L), multiply by 0.0113.
Final glycemia, lipid, and blood pressure values are last available values before any change in hypoglycemic, lipid-lowering, or blood pressure–lowering medications, respectively.
Values in this column represent differences between the mean change scores of the vegan group and those of the conventional diet group; 95% CIs in parentheses.
P values refer to t tests for between-group (vegan compared with conventional) comparisons of changes from baseline to final values.
Mean ± SE (all such values).
Significantly different from baseline (within-group t test comparison): 6P < 0.01, 7P < 0.05, 8P < 0.0001, 9P < 0.001.
Blood pressure was not determined for one participant in the vegan group because of equipment failure.
FIGURE 1.
Glycated hemoglobin (HB A1c) values for all participants (n = 49 vegan diet; n = 50 conventional diet). The mean (±SD) data shown are last values before any change to diabetes medications carried forward. t Test for between-group comparison of changes from baseline to final values, P = 0.03.
A regression model was constructed that included baseline Hb A1c, weight change, and diet group as predictors of Hb A1c change from baseline to 74 wk or last available value. In this model, baseline Hb A1c (P < 0.0001) and weight change (P = 0.007) were associated with Hb A1c change, but the effect of diet group was no longer significant (P = 0.12).
Controlling for diet group and baseline Hb A1c values, weight change was significantly associated with Hb A1c change; each kilogram of weight loss was associated with a 0.09 decrease in Hb A1c. For the subgroup with no net change in diabetes medications (n = 41), the Pearson's correlation of weight change with Hb A1c change was r = 0.50, P = 0.001.
Plasma lipids
Reductions in total, LDL-, and non–HDL-cholesterol concentrations from baseline to 74 wk (or to last available value) were observed for both groups, as were reductions in triglyceride and VLDL-cholesterol concentrations in the vegan group, with no between-group differences in intention-to-treat analyses (Table 3). In analyses adjusted for medication changes, reductions in LDL and non-HDL cholesterol were significantly greater in the vegan group (Table 4). No treatment-related serious adverse events were observed.
DISCUSSION
In this 74-wk study, both diets were associated with significant weight loss. In analyses including all participants without regard to medication changes, both diets were associated with reductions in Hb A1c; the mean change was slightly but not significantly greater in the vegan group. In analyses controlling for medication changes, significantly greater reductions were seen in Hb A1c values and in total and LDL-cholesterol concentrations in the vegan group.
Much of the effect of the intervention diets on glycemia appears to be mediated by weight reduction. However, the 2 diets appear to have altered energy intake by different mechanisms. Although overweight individuals in the conventional diet group were prescribed an explicit energy deficit, a low-fat vegan diet typically elicits significant weight loss in the absence of prescribed energy intake limits (2). This is likely because reduced dietary fat and increased dietary fiber reduce dietary energy density (15, 16).
Individuals following self-selected plant-based diets typically have lower body weights, compared with omnivores (17). In a US Department of Agriculture survey of 10,014 adults, vegetarians and individuals on high-carbohydrate, low-fat diets had the lowest body mass indexes of the groups studied (18). In clinical trials, the use of plant-based diets is associated with weight reduction that is partially preserved over the long term. In a study that used a low-fat vegan diet without added exercise in overweight postmenopausal women, median weight reduction was 4.9 kg at 1 y and 3.1 kg at 2 y, both of which were greater than weight changes associated with a comparison diet based on the National Cholesterol Education Program guidelines (19). Among individuals with cardiac disease, a lifestyle program that included a low-fat vegetarian diet and mild exercise was associated with net weight loss of 10.9 kg at 1 y and 5.8 kg at 5 y (20).
A vegan diet may also be associated with reductions in intramyocellular lipid, which is strongly associated with insulin sensitivity (21). A case-control study found that the median soleus muscle intramyocellular lipid concentration was 31% lower in a group of 21 vegans compared with 25 omnivores matched for age and body weight (P = 0.01) (22).
The long-term effect of both diet interventions on glycemia was reduced in comparison with the short-term findings from this study (6). Among participants in the vegan group with no changes to diabetes medications, Hb A1c had fallen 1.23 by 22 wk (n = 24) and 0.82 by 74 wk (n = 14). Among medication-stable participants in the conventional diet group, the Hb A1c reduction was 0.38 at 22 wk (n = 33), and 0.21 (n = 21) at 74 wk. Both groups retained most of their weight loss, suggesting that their diets had not returned to their baseline patterns.
The principal diet change in the conventional diet group was a reduction in energy intake, which appears to be due to reduced portion sizes rather than to changes in macronutrient balance, which was close to the recommended percentages for protein, carbohydrate, and monounsaturated fat at baseline and changed only slightly during the study. This change in energy intake was nonetheless sufficient to lead to sustained weight loss. Several studies that used conventional diets (23–26) have reported more dramatic Hb A1c reductions (0.5–1.9%) than were observed in the conventional diet group in our study. However, each of those studies lasted ≤6 mo, and none accounted for medication alterations or dropouts in reports of Hb A1c changes. The current study was approximately 1 y longer than prior studies, and during this time changes in medications are likely to occur. We have therefore taken steps to report results both with and without accounting for medication changes. Our participant population had had diabetes for >8 years, on average, before study entry.
Vegetarian and vegan diets are often more effective than other diets for lipid control (27), an important consideration, given that coronary heart disease is the main cause of mortality in diabetes. The reduction in triglycerides associated with the vegan diet contrasts with earlier studies suggesting that diets high in refined carbohydrate may transiently increase triglyceride concentrations for some individuals (2, 28). It appears that high-fiber, low–glycemic index foods, such as were used in the present study, have a salutary effect on triglyceride concentrations (29), and the observed weight loss may also have contributed to triglyceride reductions.
The acceptability of low-fat vegetarian and vegan diets has been shown to be similar to that of other therapeutic diets in studies of individuals with cardiovascular disease (8), young women with dysmenorrhea (9), overweight but otherwise healthy postmenopausal women (10), and the current study population (30). Although low-fat vegan diets are generally lower in fat, saturated fat, and cholesterol and higher in fiber and complex carbohydrate, compared with omnivorous diets (31, 32), planning is important for micronutrient adequacy, particularly for sources of vitamin B-12, vitamin D, and calcium (7).
The study's strengths include its randomized design, extended duration, inclusion of individuals with long-standing diabetes, analysis of dependent measures without regard to variations in dietary adherence, statistical methods aimed at reducing the effect of medication changes, and applicability outside the research setting. The study also has weaknesses. The medication changes required for patient safety or for appropriate medical management present a challenge in outpatient diabetes studies of more than a few months' duration. In the present study, most participants in both groups altered medications, often because of clinical success (low blood glucose values), presenting a confounding variable that required special analyses. Carrying forward the last value before any medication change, as in the current study, may either under- or overestimate effects (33, 34). Limiting the analysis to those with no medication changes reduces sample size and statistical power. It is reassuring that all statistical models (repeated-measures analysis, use of final data from all participants without regard to medication use or dietary adherence, and use of the last value before medication change) led to similar conclusions, showing a mean long-term Hb A1c–lowering effect ranging from 0.3 to 0.4 units for the vegan group and from 0 to 0.1 for the conventional diet group. Moreover, the persistent mean weight loss confirms the participants' reports that dietary changes were at least partially maintained.
In conclusion, in individuals with type 2 diabetes participating in a research study, both a low-fat vegan diet and a diet based on 2003 ADA guidelines facilitated long-term weight reduction. In analyses controlling for medication changes, the vegan diet appeared to be more effective for control of glycemia and plasma lipid concentrations. Whether the observed differences provide clinical benefit for the macro- or microvascular complications of diabetes remains to be established. (Other articles in this supplement to the Journal include references 35–61.)
Acknowledgments
We thank Paul Poppen, of the George Washington University, for conducting statistical analyses.
The authors' responsibilities were as follows—NDB: participated in the study design and data analysis and drafted the manuscript; JC and DJAJ: participated in the study design and data analysis and reviewed the manuscript for critical content; GT-M: participated in recruiting, nutrition instruction, and manuscript preparation; LG: participated in nutrition instruction and reviewed the manuscript for critical content; AG: participated in nutrition instruction, conducted nutrient analyses, and reviewed the manuscript; and HF: participated in data analysis and reviewed the manuscript for critical content.
NDB is president of the Physicians Committee for Responsible Medicine and the Cancer Project, organizations that promote the use of low-fat, plant-based diets, and writes books and gives lectures about therapeutic diets, including vegan diets. He is the author of Dr. Neal Barnard's Program for Reversing Diabetes and receives royalties from its sales. None of the other authors had any personal or financial conflict of interest.
REFERENCES
- 1.Jenkins DJA, Kendall CWC, Marchie A, et al. Type 2 diabetes and the vegetarian diet. Am J Clin Nutr 2003;78(suppl):610S–6S [DOI] [PubMed] [Google Scholar]
- 2.Barnard ND, Scialli AR, Turner-McGrievy G, Lanou AJ, Glass J. The effects of a low-fat, plant-based dietary intervention on body weight, metabolism, and insulin sensitivity. Am J Med 2005;118:991–7 [DOI] [PubMed] [Google Scholar]
- 3.Pischke CR, Weidner G, Elliott-Eller M, et al. Comparison of coronary risk factors and quality of life in coronary artery disease patients with versus without diabetes mellitus. Am J Cardiol 2006;97:1267–73 [DOI] [PubMed] [Google Scholar]
- 4.Anderson JW, Ward K. High-carbohydrate, high-fiber diets for insulin-treated men with diabetes mellitus. Am J Clin Nutr 1979;32:2312–21 [DOI] [PubMed] [Google Scholar]
- 5.Barnard RJ, Jung T, Inkeles SB. Diet and exercise in the treatment of NIDDM: the need for early emphasis. Diabetes Care 1994;17:1469–72 [DOI] [PubMed] [Google Scholar]
- 6.Barnard ND, Cohen J, Jenkins DJ, et al. A low-fat, vegan diet improves glycemic control and cardiovascular risk factors in a randomized clinical trial in individuals with type 2 diabetes. Diabetes Care 2006;29:1777–83 [DOI] [PubMed] [Google Scholar]
- 7.American Dietetic Association Position of the American Dietetic Association and Dietitians of Canada: Vegetarian diets. J Am Diet Assoc 2003;103:748–65 [DOI] [PubMed] [Google Scholar]
- 8.Barnard ND, Scherwitz LW, Ornish D. Adherence and acceptability of a low-fat, vegetarian diet among patients with cardiac disease. J Cardiopulm Rehabil 1992;12:423–31 [Google Scholar]
- 9.Barnard ND, Scialli AR, Bertron P, Hurlock D, Edmonds K. Acceptability of a therapeutic low-fat, vegan diet in premenopausal women. J Nutr Educ 2000;32:314–9 [Google Scholar]
- 10.Barnard ND, Scialli AR, Turner-McGrievy GM, Lanou AJ. Acceptability of a very-low-fat, vegan diet compares favorably to a more moderate low-fat diet in a randomized, controlled trial. J Cardiopulm Rehabil 2004;24:229–35 [DOI] [PubMed] [Google Scholar]
- 11.Wilson DH, Bogacz JP, Forsythe CM, et al. Fully automated assay of glycohemoglobin with the Abbott IMx analyzer: novel approaches for separation and detection. Clin Chem 1993;39:2090–7 [PubMed] [Google Scholar]
- 12.Franz MJ, Bantle JP, Beebe CA, et al. ; American Diabetes Association Evidence-based nutrition principles and recommendations for the treatment and prevention of diabetes and related complications. Diabetes Care 2003;26(suppl):S51–61 [DOI] [PubMed] [Google Scholar]
- 13.Schakel SF, Sievert YA, Buzzard IM. Sources of data for developing and maintaining a nutrient database. J Am Diet Assoc 1988;88:1268–71 [PubMed] [Google Scholar]
- 14.Bouchard C, Tremblay A, LeBlanc C, Lortie G, Savard R, Theriault G. A method to assess energy expenditure in children and adults. Am J Clin Nutr 1983;37:461–7 [DOI] [PubMed] [Google Scholar]
- 15.Kendall A, Levitsky DA, Strupp BJ, Lissner L. Weight loss on a low-fat diet: consequence of the imprecision of the control of food intake in humans. Am J Clin Nutr 1991;53:1124–9 [DOI] [PubMed] [Google Scholar]
- 16.Howarth NC, Saltzman E, Roberts SB. Dietary fiber and weight regulation. Nutr Rev 2001;59:129–39 [DOI] [PubMed] [Google Scholar]
- 17.Berkow SE, Barnard N. Vegetarian diets and weight status. Nutr Rev 2006;64:175–88 [DOI] [PubMed] [Google Scholar]
- 18.Kennedy ET, Bowman SA, Spence JT, Freedman M, King J. Popular diets: correlation to health, nutrition, and obesity. J Am Diet Assoc 2001;101:411–20 [DOI] [PubMed] [Google Scholar]
- 19.Turner-McGrievy GM, Barnard ND, Scialli AR. A two-year randomized weight loss trial comparing a vegan diet to a more moderate low-fat diet. Obesity (Silver Spring) 2007;15:2276–81 [DOI] [PubMed] [Google Scholar]
- 20.Ornish D, Scherwitz LW, Billings JH, et al. Intensive lifestyle changes for reversal of coronary heart disease. JAMA 1998;280:2001–7 [DOI] [PubMed] [Google Scholar]
- 21.Petersen KF, Dufour S, Befroy D, Garcia R, Shulman GI. Impaired mitochondrial activity in the insulin-resistant offspring of patients with type 2 diabetes. N Engl J Med 2004;350:664–71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Goff LM, Bell JD, So PW, Dornhorst A, Frost GS. Veganism and its relationship with insulin resistance and intramyocellular lipid. Eur J Clin Nutr 2005;59:291–8 [DOI] [PubMed] [Google Scholar]
- 23.Franz MJ, Monk A, Barry B, et al. Effectiveness of medical nutrition therapy provided by dietitians in the management of non-insulin-dependent diabetes mellitus: a randomized controlled trial. J Am Diet Assoc 1995;95:1009–17 [DOI] [PubMed] [Google Scholar]
- 24.Miller CK, Edwards L, Kissling G, Sanville L. Nutrition education improves metabolic outcomes among older adults with diabetes mellitus: results from a randomized control trial. Prev Med 2002;34:252–9 [DOI] [PubMed] [Google Scholar]
- 25.Ziemer DC, Berkowitz KJ, Panayioto RM, et al. A simple meal plan emphasizing healthy food choices is as effective as an exchange-based meal plan for urban African Americans with type 2 diabetes. Diabetes Care 2003;26:1719–24 [DOI] [PubMed] [Google Scholar]
- 26.Goldhaber-Fiebert JD, Goldhaber-Fiebert SN, Tristán ML, Nathan DM. Randomized controlled community-based nutrition and exercise intervention improves glycemia and cardiovascular risk factors in type 2 diabetic patients in rural Costa Rica. Diabetes Care 2003;26:24–9 [DOI] [PubMed] [Google Scholar]
- 27.Jenkins DJ, Kendall CW, Marchie A, et al. Effects of a dietary portfolio on cholesterol-lowering foods vs lovastatin on serum lipids and C-reactive protein. JAMA 2003;290:502–10 [DOI] [PubMed] [Google Scholar]
- 28.Ornish D, Brown SE, Scherwitz LW, Billings JH, Armstrong WT, Ports TA. Can lifestyle changes reverse coronary heart disease? Lancet 1990;336:129–33 [DOI] [PubMed] [Google Scholar]
- 29.Jenkins DJ, Wolever TM, Kalmusky J, et al. Low-glycemic index diet in hyperlipidemia: use of traditional starchy foods. Am J Clin Nutr 1987;46:66–71 [DOI] [PubMed] [Google Scholar]
- 30.Barnard ND, Gloede L, Cohen J, et al. A low-fat vegan diet elicits greater macronutrient changes, but is comparable in adherence and acceptability, compared with a more conventional diabetes diet among individuals with type 2 diabetes. J Am Diet Assoc 2009;109:263–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Turner-McGrievy GM, Barnard ND, Scialli AR, Lanou AJ. Effects of a low-fat, vegan diet and a Step II diet on macro- and micronutrient intakes in overweight, postmenopausal women. Nutrition 2004;20:738–46 [DOI] [PubMed] [Google Scholar]
- 32.Turner-McGrievy GM, Barnard ND, Cohen J, Jenkins DJA, Gloede L, Green AA. Changes in nutrient intake and dietary quality among participants with type 2 diabetes following a low-fat vegan diet or a conventional diabetes diet for 22 weeks. J Am Diet Assoc 2008;108:1636–45 [DOI] [PubMed] [Google Scholar]
- 33.Ware JH. Interpreting incomplete data in studies of diet and weight loss. N Engl J Med 2003;348:2136–7 [DOI] [PubMed] [Google Scholar]
- 34.Beunckens C, Molenberghs G, Kenward MG. Direct likelihood analysis versus simple forms of imputation for missing data in randomized clinical trials. Clin Trials 2005;2:379–86 [DOI] [PubMed] [Google Scholar]
- 35.Rajaram S, Sabaté J. Preface. Am J Clin Nutr 2009;89(suppl):1541S–2S [DOI] [PubMed] [Google Scholar]
- 36.Jacobs DR, Jr, Gross MD, Tapsell LC. Food synergy: an operational concept for understanding nutrition. Am J Clin Nutr 2009;89(suppl):1543S–8S [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jacobs DR, Jr, Haddad EH, Lanou AJ, Messina MJ. Food, plant food, and vegetarian diets in the US dietary guidelines: conclusions of an expert panel. Am J Clin Nutr 2009;89(suppl):1549S–2S [DOI] [PubMed] [Google Scholar]
- 38.Lampe JW. Interindividual differences in response to plant-based diets: implications for cancer risk. Am J Clin Nutr 2009;89(suppl):1553S–7S [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Simon JA, Chen Y-H, Bent S. The relation of α-linolenic acid to the risk of prostate cancer: a systematic review and meta-analysis. Am J Clin Nutr 2009;89(suppl):1558S–64S [DOI] [PubMed] [Google Scholar]
- 40.Pierce JP, Natarajan L, Caan BJ, et al. Dietary change and reduced breast cancer events among women without hot flashes after treatment of early-stage breast cancer: subgroup analysis of the Women's Healthy Eating and Living Study. Am J Clin Nutr 2009;89(suppl):1565S–71S [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Newby PK. Plant foods and plant-based diets: protective against childhood obesity? Am J Clin Nutr 2009;89(suppl):1572S–87S [DOI] [PubMed] [Google Scholar]
- 42.Mangat I. Do vegetarians have to eat fish for optimal cardiovascular protection? Am J Clin Nutr 2009;89(suppl):1597S–601S [DOI] [PubMed] [Google Scholar]
- 43.Willis LM, Shukitt-Hale B, Joseph JA. Modulation of cognition and behavior in aged animals: role for antioxidant- and essential fatty acid–rich plant foods. Am J Clin Nutr 2009;89(suppl):1602S–6S [DOI] [PubMed] [Google Scholar]
- 44.Fraser GE. Vegetarian diets: what do we know of their effects on common chronic diseases? Am J Clin Nutr 2009;89(suppl):1607S–12S [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Key TJ, Appleby PN, Spencer EA, Travis RC, Roddam AW, Allen NE. Cancer incidence in vegetarians: results from the European Prospective Investigation into Cancer and Nutrition (EPIC-Oxford). Am J Clin Nutr 2009;89(suppl):1620S–6S [DOI] [PubMed] [Google Scholar]
- 46.Key TJ, Appleby PN, Spencer EA, Travis RC, Roddam AW, Allen NE. Mortality in British vegetarians: results from the European Prospective Investigation into Cancer and Nutrition (EPIC-Oxford). Am J Clin Nutr 2009;89(suppl):1613S–9S [DOI] [PubMed] [Google Scholar]
- 47.Craig WJ. Health effects of vegan diets. Am J Clin Nutr 2009;89(suppl):1627S–33S [DOI] [PubMed] [Google Scholar]
- 48.Weaver CM. Should dairy be recommended as part of a healthy vegetarian diet? Point. Am J Clin Nutr 2009;89(suppl):1634S–7S [DOI] [PubMed] [Google Scholar]
- 49.Lanou AJ. Should dairy be recommended as part of a healthy vegetarian diet? Counterpoint. Am J Clin Nutr 2009;89(suppl):1638S–42S [DOI] [PubMed] [Google Scholar]
- 50.Sabaté J, Ang Y. Nuts and health outcomes: new epidemiologic evidence. Am J Clin Nutr 2009;89(suppl):1643S–8S [DOI] [PubMed] [Google Scholar]
- 51.Ros E. Nuts and novel biomarkers of cardiovascular disease. Am J Clin Nutr 2009;89(suppl):1649S–56S [DOI] [PubMed] [Google Scholar]
- 52.Rajaram S, Haddad E, Mejia A, Sabaté J. Walnuts and fatty fish influence different serum lipid fractions in normal to mildly hyperlipidemic individuals: a randomized controlled study. Am J Clin Nutr 2009;89(suppl):1657S–63S [DOI] [PubMed] [Google Scholar]
- 53.Lampe JL. Is equol the key to the efficacy of soy foods? Am J Clin Nutr 2009;89(suppl):1664S–7S [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Badger TM, Gilchrist JM, Pivik RT, et al. The health implications of soy infant formula. Am J Clin Nutr 2009;89(suppl):1668S–72S [DOI] [PubMed] [Google Scholar]
- 55.Messina M, Wu AH. Perspectives on the soy–breast cancer relation. Am J Clin Nutr 2009;89(suppl):1673S–9S [DOI] [PubMed] [Google Scholar]
- 56.Lönnerdal B. Soybean ferritin: implications for iron status of vegetarians. Am J Clin Nutr 2009;89(suppl):1680S–5S [DOI] [PubMed] [Google Scholar]
- 57.Chan J, Jaceldo-Siegl K, Fraser GE. Serum 25-hydroxyvitamin D status of vegetarians partial vegetarians, and nonvegetarians: the Adventist Health Study-2. Am J Clin Nutr 2009;89(suppl):1686S–92S [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Elmadfa I, Singer I. Vitamin B-12 and homocysteine status among vegetarians: a global perspective. Am J Clin Nutr 2009;89(suppl):1693S–8S [DOI] [PubMed] [Google Scholar]
- 59.Marlow HJ, Hayes WK, Soret S, Carter RL, Schwab ER, Sabaté J. Diet and the environment: does what you eat matter? Am J Clin Nutr 2009;89(suppl):1699S–703S [DOI] [PubMed] [Google Scholar]
- 60.Carlsson-Kanyama A, González AD. Potential contributions of food consumption patterns to climate change. Am J Clin Nutr 2009;89(suppl):1704S–9S [DOI] [PubMed] [Google Scholar]
- 61.Eshel G, Martin PA. Geophysics and nutritional science: toward a novel, unified paradigm. Am J Clin Nutr 2009;89(suppl):1710S–6S [DOI] [PubMed] [Google Scholar]