Abstract
Matrix metalloproteinase-9 (MMP-9) secreted from macrophages plays an important role in tissue destruction and inflammation through degradation of matrix proteins and proteolytic activation of cytokines/chemokines. Whereas the MEK-ERK and PI3K-Akt pathways up-regulate MMP-9 expression, regulation of MMP-9 by JNK remains controversial. Presently, we aimed to determine the role of JNK in MMP-9 regulation in Raw 264.7 cells. Inhibition of JNK by the JNK inhibitor SP600125 induced MMP-9 in the absence of serum and suppressed the expression of TNF-α, IL-6 and cyclooxygenase-2 in LPS-treated Raw 264.7 cells. In a knockdown experiment with small interfering RNA, suppression of JNK1 induced MMP-9 expression. Interestingly, mouse serum suppressed SP600125-mediated MMP-9 induction, similar to IFN-γ. However, the inhibitory activity of mouse serum was not affected by pyridone 6, which inhibits Janus kinase downstream to IFN-γ. In addition to mouse serum, conditioned media of Raw 264.7 cells contained the inhibitory factor(s) larger than 10 kDa, which suppressed SP600125- or LPS-induced MMP-9 expression. Taken together, these data suggest that JNK1 suppresses MMP-9 expression in the absence of serum. In addition, the inhibitory factor(s) present in serum or secreted from macrophages may negatively control MMP-9 expression.
Keywords: anthra(1,9-cd)pyrazol-6(2H)-one; autocrine communication; JNK mitogen-activated protein kinases; macrophages; matrix metalloproteinase 9; serum
Introduction
Macrophages may be crucial in inflammatory diseases including chronic obstructive pulmonary disease (Shapiro et al., 1999), atherosclerosis (Deguchi et al., 2006), arthritis (Blom et al., 2007), and tubulointerstitial kidney disease (Eardley et al., 2005) via the release of pro-inflammatory mediators. Macrophages additionally contribute to tissue destruction by the release of several proteases including matrix metalloproteinase-9 (MMP-9; also called gelatinase B) (Van de Steen et al., 2000; Vaday et al., 2001). MMP-9 is a protease that degrades extracellular matrix proteins including gelatin, collagen, elastin, and laminin (Opdenakker et al., 2001; Chakrabarti and Patel, 2005). In addition, MMP-9 modulates the activities of other proteases, growth factors, cytokines, and chemokines through proteolytic cleavage (Opdenakker et al., 2001; Atkinson et al., 2003; Lin et al., 2008). These activities indicate the important roles of MMP-9 in tissue destruction and also in tissue remodeling and inflammation.
MMP-9 regulation involves transcriptional regulation, post-translational cleavage, and antagonism by physiological inhibitors (Chakraborti et al., 2003; Yan and Boyd, 2007). In transcriptional regulation, MMP-9 expression is controlled by transcriptional factors including AP-1 and NF-κB, which bind to the corresponding binding sites in the MMP-9 promoter region (Benbow et al., 1997). In various kinds of cells, different stimuli induce MMP-9 expression through activation of the MEK-ERK or phosphoinositide 3-kinase (PI3K)-Akt signaling pathways, which subsequently activate AP-1 and NF-κB (Gum et al., 1997; Genersch et al., 2000; Chung et al., 2004; Moon et al., 2004; Woo et al., 2004a; Cheng et al., 2006; Han et al., 2006). Also, p38 MAPK up-regulates MMP-9 expression in Raw 264.7 cells stimulated with LPS (Woo et al., 2004) and CpG oligodeoxynucleotide (Lim et al., 2007).
In comparison with ERK and p38 MAPK, however, the regulatory role of JNK in MMP-9 expression remains contentious. Inhibition of JNK activity by the JNK inhibitor SP600125 decreases MMP-9 expression in ovarian carcinoma cells stimulated with PMA (Shin et al., 2002), astrocytes stimulated with IL-1β (Wu et al., 2004), and cardiac fibroblasts stimulated with IL-1β (Xie et al., 2004). Similarly, knockdown of JNK2 by siRNA also inhibits MMP-9 expression induced by TNF-α in A549 cells (Lin et al., 2008). In contrast, SP600125 does not suppress MMP-9 expression in Raw 264.7 cells stimulated with LPS (Woo et al., 2004) and does not inhibit MMP-9 expression induced by PMA in rat astrocytes, even though PMA activates JNK (Arai et al., 2003).
In this study, we demonstrate that JNK1, but not JNK2, suppresses LPS-induced MMP-9 expression in Raw 264.7 cells in the absence of serum. Additionally, we reveal that the presence of the inhibitory factor(s) in serum that represses MMP-9 expression induced by JNK inhibition. Finally, we report the presence of a similar autocrine inhibitory activity in the conditioned media of Raw 264.7 cells.
Results
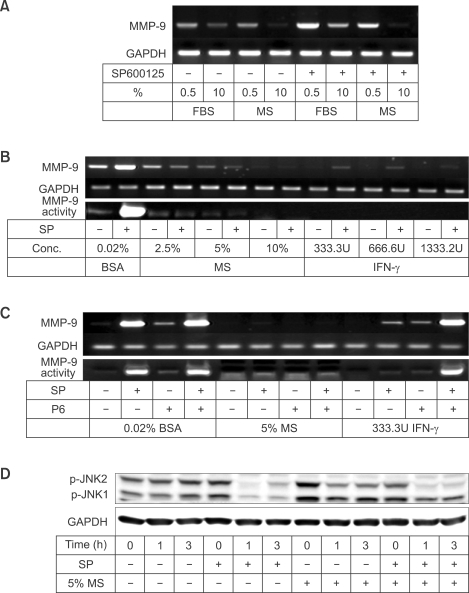
SP600125 augments MMP-9 induction in Raw 264.7 cells stimulated by LPS or TNF-α
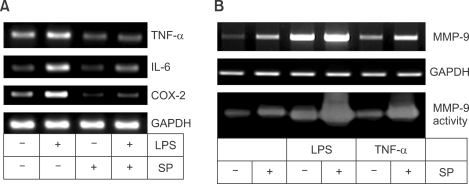
We first checked the effect of the JNK inhibitor SP600125 on induction of MMP-9 in comparison with other genes already known to be up-regulated through JNK by LPS stimulation (Matsuguchi et al., 2001; Waetzig et al., 2005). Before stimulation, Raw 264.7 cells were deprived of serum overnight with DMEM containing 0.02% BSA. Then, 10 µM SP600125 was added to the cells 1 h prior to stimulation with 100 ng/ml LPS. Total RNA or culture medium was collected after 8 h for MMP-9 RT-PCR or after 24 h for MMP-9 gelatin zymography. As shown in Figure 1A, LPS induced the expression of TNF-α, IL-6, and COX-2 mRNA in Raw 264.7 cells. These inductions were abrogated by 10 µM SP600125. These observations indicate that induction of TNF-α, IL-6, or COX-2 mRNA by LPS is up-regulated by JNK in Raw 264.7 cells.
Figure 1.
SP600125 augments MMP-9 induction in Raw 264.7 cells stimulated by LPS or TNF-α. (A) Raw 264.7 cells were pretreated with 10 µM SP600125 1 h prior to stimulation with 100 ng/ml LPS. Total RNAs were collected 8 h later and RT-PCR was performed with specific primers for TNF-α, IL-6, or COX-2. (B) Raw 264.7 cells were pretreated with 10 µM SP600125 1 h prior to stimulation with 100 ng/ml LPS or 100 ng/ml TNF-α. Total RNAs or culture media were collected after 8 h for MMP-9 RT-PCR or after 24 h for MMP-9 gelatin zymography.
Similarly, LPS and TNF-α increased both MMP-9 mRNA expression and MMP-9 secretion (a pro-form of MMP-9, Figure 1B). However, in contrast to TNF-α, IL-6, and COX-2 expression, both MMP-9 expression and MMP-9 secretion was augmented by 10 µM SP600125. Interestingly, SP600125 elevated basal levels of MMP-9 mRNA expression and MMP-9 secretion without stimulation of LPS or TNF-α.
SP600125 induces basal expression of MMP-9 in a time- and concentration-dependent manner
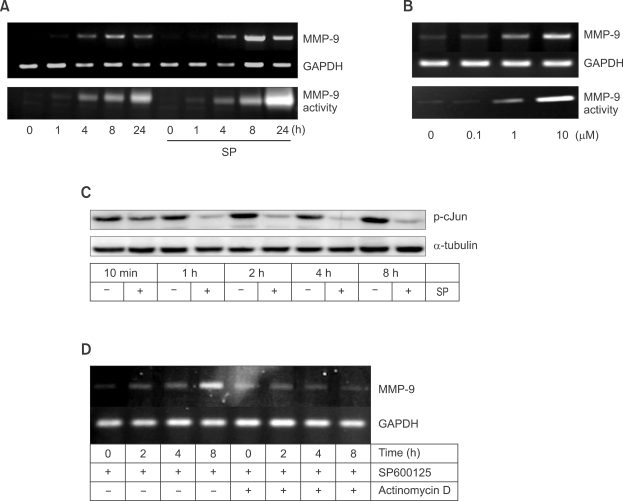
As SP600125 increased the basal expression of MMP-9 even without LPS or TNF-α (Figure 1B), we next characterized the effect of SP600125 on basal MMP-9 expression. In the absence of SP600125, both MMP-9 mRNA expression and MMP-9 secretion gradually increased according to the time of incubation in Raw 264.7 cells cultured in 0.02% BSA-containing medium (Figure 2A). However, SP600125 elevated MMP-9 mRNA expression to a much greater extent at 8 h and 24 h. Accordingly, a marked increase in MMP-9 activity was observed in SP600125-treated Raw 264.7 cells at 24 h. The inductive effect of SP600125 on MMP-9 expression was concentration-dependent (Figure 2B). Both MMP-9 mRNA expression and MMP-9 secretion were increased by SP600125 in a concentration-dependent manner.
Figure 2.
SP600125 induces basal expression of MMP-9. (A) Time-dependent induction of MMP-9 by SP600125. Raw 264.7 cells were treated with 10 µM SP600125. At the indicated times, total RNAs and culture media were collected for MMP-9 RT-PCR and MMP-9 zymography, respectively. (B) Concentration-dependent MMP-9 induction. Raw 264.7 cells were treated with various concentrations of SP600125. Total RNAs or culture media were collected after 8 h for MMP-9 RT-PCR or after 24 h for MMP-9 gelatin zymography. (C) After Raw 264.7 cells were treated with 10 µM SP600125, the cells were collected for determination of c-jun phosphorylation by Western blot. (D) For determination of regulation level of MMP-9 expression, Raw 264.7 cells were pre-treated with actinomycin D (5 µg/ml) 1 h prior to 10 µM SP600125. At the indicated times, the cells were collected and MMP-9 expression was determined.
To confirm whether the inducing effect of SP600125 on MMP-9 expression was mediated through JNK activity, phosphorylation of the JNK substrate c-Jun was determined after treatment with SP600125. As shown in Figure 2C, phosphorylation of c-Jun decreased from 10 min to 8 h after SP600125 treatment.
Next, we determined the level of regulation of MMP-9 expression by SP600125. Raw 264.7 cells were pre-treated with actinomycin D 1 h prior to 10 µM SP600125. As shown in Figure 2D, actinomycin D successfully inhibited MMP-9 induction by SP600125 at 8 h. This data indicates that SP600125 induced MMP-9 mRNA at transcriptional level.
Effects of SP600125 on ERK, p38 MAPK, Akt, and NF-κB
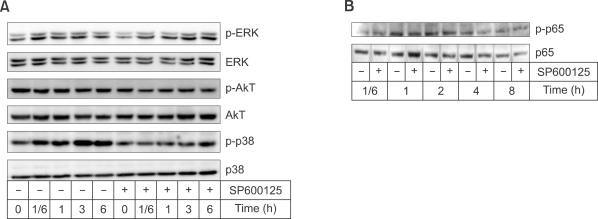
As ERK, p38 MAPK, or Akt mediate MMP-9 induction (Chung et al., 2004), we tried to determine the effects of SP600125 on the activation of ERK, p38 MAPK, or Akt. As shown in Figure 3A, SP600125 did not elevate the phosphorylation of ERK, p38 MAPK, or Akt. Furthermore, SP600125 did not affect the phosphorylation of p65, which is a component of NF-κB involved in the induction of MMP-9 (Figure 3B). These results suggest that SP600125 may not induce MMP-9 expression through the activation of ERK, p38 MAPK, Akt, or p65 of NF-κB.
Figure 3.
Effects of SP600125 on MAPK, AKT, and NF-κB. After Raw 264.7 cells were treated with 10 µM SP600125, the cells were collected at the indicated times for Western blot. (A) Phosphorylation of ERK, Akt, or p38 MAPK. (B) Phosphorylation of p65.
Suppression of MMP-9 expression by JNK1
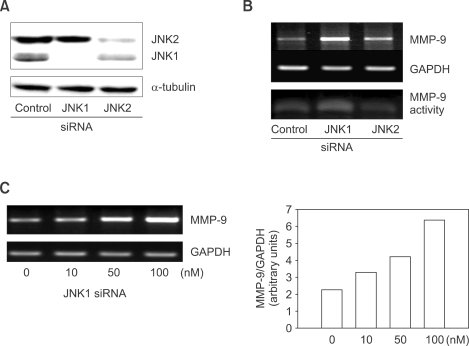
Next, we performed a knock-down experiment to exclude a nonspecific effect of SP600125 and to identify JNK isoform involved in the negative regulation of MMP-9 expression, in light of different potential biological roles of JNK1 and JNK2 (Bogoyevitch, 2006). JNK1 or JNK2 siRNA successfully suppressed levels of JNK1 or JNK2 protein, respectively (Figure 4A). Knock-down of JNK1 by JNK1 siRNA increased both MMP-9 mRNA expression and MMP-9 secretion (Figure 4B). In addition, JNK1 siRNA increased MMP-9 expression in a concentration-dependent manner (Figure 4C). In contrast, JNK2 siRNA induced MMP-9 expression to a much lesser degree. However, it may not be conclusive that JNK2 siRNA caused MMP-9 induction, because JNK2 siRNA slightly inhibited JNK1 expression. These data suggest that JNK1 can specifically suppress basal MMP-9 expression in Raw 264.7 cells.
Figure 4.
Suppression of MMP-9 expression by JNK1. Raw 264.7 cells (2 × 105) were transfected with negative control, JNK1, or JNK2 siRNA. (A) Levels of JNK1and JNK2 proteins in Western blot, and (B) MMP-9 mRNA expression and MMP-9 secretion after transfection of 100 nM each siRNA. (C) Induction of MMP-9 expression in a concentration-dependent manner. Raw 264.7 cells were transfected with increasing concentrations of JNK siRNA. After 72 h, total RNAs were collected for MMP-9 RT-PCR.
Serum inhibits SP600125-mediated MMP-9 induction
The above experiments were performed in the absence of serum to exclude influence of serum. Next, we tried to confirm SP600125-mediated MMP-9 induction in the presence of different concentrations of mouse serum and FBS. Surprisingly, SP600125-mediated induction of MMP-9 was attenuated by 10% mouse serum or FBS (Figure 5A). FBS displayed weaker inhibitory effect than did mouse serum. Next, Raw 264.7 cells were treated with different concentrations of mouse serum (2.5-10%). Mouse serum inhibited both basal and SP60015-induced MMP-9 expression in a concentration-dependent manner (Figure 5B). Both basal and SP600125-induced MMP-9 expression was almost completely abolished by 10% mouse serum. As IFN-γ is known to inhibit MMP-9 expression (Xie et al., 1994), we investigated whether IFN-γ was responsible for MMP-9 suppression in mouse serum. INF-γ reduced both basal and SP600125-induced MMP-9 expression similar to the mouse serum in a concentration-dependent manner (Figure 5B). However, while P6, a pan-JAK inhibitor, completely restored SP600125-mediated MMP-9 induction, it did not affect the inhibitory activity of mouse serum on MMP-9 expression (Figure 5C). As JNK1 siRNA induced MMP-9 expression and mouse serum suppressed MMP-9 induction by SP600125, we determined effect of mouse serum on activated status of JNK. In Figure 5D, SP600125 inhibited phosphorylation of JNK1 and JNK2, and mouse serum restored phosphorylation of JNK1 but not JNK2. These data imply that inhibitory factor(s) other than IFN-γ suppress MMP-9 expression and mouse serum suppressed MMP-9 expression possibly through maintenance of JNK1 activity.
Figure 5.
Serum inhibits SP600125-mediated MMP-9 induction. (A) Raw 264.7 cells were treated with 0.5 or 10% FBS or mouse serum 1 h prior to 10 µM SP600125. After 8 h, total RNAs were collected for MMP-9 RT-PCR. (B) Raw 264.7 cells were pretreated with increasing concentrations of mouse serum or mouse IFN-γ. 1 h later, 10 µM SP600125 was added. Total RNAs or culture media were collected after 8 h for MMP-9 RT-PCR or after 24 h for MMP-9 gelatin zymography. (C) Raw 264.7 cells were pretreated with 250 nM pyridone 1 h prior to mouse serum or IFN-γ. 1 h later, 10 µM SP600125 was added. Total RNAs or culture media were collected after 8 h for MMP-9 RT-PCR or after 24 h for MMP-9 gelatin zymography. (D) Raw 264.7 cells were treated with 5% mouse serum 30 min prior to 10 µM SP600125. At the indicated times, the cells were collected for determination of JNK phosphorylation by Western blot.
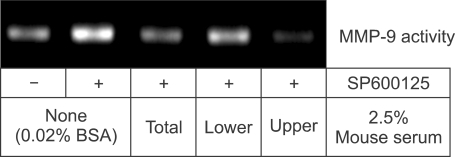
The inhibitory factor(s) in the mouse serum exceed 10 kDa
To characterize the nature of the inhibitory factor(s) in the mouse serum, the serum was fractionated and concentrated 2-fold by an ultrafiltration unit having a membrane with a 10 kDa molecular weight cutoff. Whereas the lower fraction passing through the membrane did not suppress SP600125-induced MMP-9 secretion, the upper fraction containing molecules >10 kDa inhibited the increase in MMP-9 secretion to a greater extent than unfiltered mouse serum (Figure 6).
Figure 6.
The inhibitory factor(s) in the mouse serum is larger than 10 kDa. Mouse serum was fractionated and concentrated 2-fold with an ultrafiltration unit having a 10 kDa molecular weight cutoff. Raw 264.7 cells were pretreated with 0.02% BSA, 2.5% mouse serum, 2.5% filtered fraction (lower), or 2.5% of unfiltered fraction (upper) 4 h prior to the addition of 10 µM SP600125. Culture media was collected 24 h later for MMP-9 zymography.
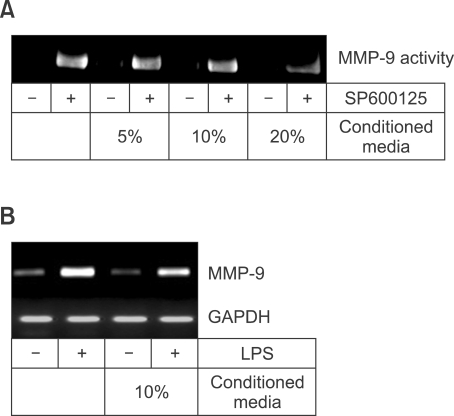
Presence of inhibitory factor(s) in conditioned media of Raw 264.7 cells
We hypothesized that inhibitory factor(s) may be secreted from Raw 264.7 cells, because basal and SP600125-induced expression of MMP-9 mRNA were lower at 24 h than at 8 h in Figure 2A. The inhibitory activity on MMP-9 expression was determined in the conditioned media of Raw 24.7 cells. To obtain the media, Raw 264.7 cells were cultured in the absence of serum and the conditioned media were added to fresh culture media at final concentrations from 5-20% (Figure 7A). SP600125-mediated increase in MMP-9 secretion was inhibited by 20% conditioned media. In addition, the concentrated conditioned media also inhibited MMP-9 induction by LPS (Figure 7B). These data are consistent with the suggestion that the presence of inhibitory factor(s) secreted from Raw 264.7 cells can suppress MMP-9 expression induced by JNK inhibition or LPS stimulation.
Figure 7.
Presence of inhibitory factor(s) in conditioned media of Raw 264.7 cells. (A) Suppression of MMP-9 secretion by the conditioned media. The conditioned media was obtained after 1 day culture of Raw 264.7 cells in the absence of serum and BSA. Conditioned media were added to the fresh culture media at final concentrations from 5-20%. 4 h later, 10 µM SP600125 was added to the cells. Culture media was collected 24 h later for MMP-9 zymography. (B) The conditioned media obtained in (A) was concentrated 15-fold and was added to the cells at a final concentration of 10%. 4 h later, the cells were stimulated with 100 ng/ml LPS. Total RNAs were collected after 8 h for MMP-9 RT-PCR.
Discussion
In this study, we have demonstrated that knockdown of JNK1, but not JNK2, induces MMP-9 mRNA expression and MMP-9 secretion in Raw 264.7 cells. Even with structural and biochemical similarities, JNK1 and JNK2 do not simply redundantly perform the same cellular and biological functions. For example, whereas JNK1 phosphorylates and activates transcriptional activity of c-Jun, JNK2 is preferentially bound to c-Jun in unstimulated cells and contributes to the degradation of c-Jun (Sabapathy et al., 2004). Ablation of JNK1 decreases TATA-binding protein expression, whereas ablation of JNK2 enhances it (Zhong et al., 2007). In addition, JNK1, but not JNK2, plays a predominant role in the induction of pro-inflammatory cytokine from bone marrow macrophages in response to LPS and TNF-α (Sánchez-Tilló et al., 2007). However, differential regulation of MMP-9 by JNK1 or JNK2 has remained unclear due to an absence of comparative experimental data, even though JNK1 may induce MMP-9 (Wang et al., 2008; Crowe et al., 2001) without comparison with JNK2. In contrast, our data clearly indicate the distinct role of JNK1 in the regulation of MMP-9 expression in comparison with JNK2.
The presently documented suppressive role of JNK1 in MMP-9 expression is contradictory to previous findings. IL-1β-activated JNK increases MMP-9 expression in rat cardiac fibroblasts (Xie et al., 2004) and rat brain astrocytes (Wu et al., 2004). In ovarian carcinoma cells, inhibition of JNK reduces the secretion of MMP-9 induced by PMA (Shin et al., 2002). Activation of JNK is related to increased MMP-9 expression induced by TNF-α in A549 cells (Lin et al., 2008). In contrast to these reports, Heidinger et al. (2006) observed similar results to ours. In THP-1 monocytic cells, MMP-9 expression was augmented by SP600125 together with upregulation of ERK phosphorylation. They proposed that MMP-9 is up-regulated by TNF-α released in an autocrine fashion. However, we did not observe any increase in ERK phosphorylation by SP600125 treatment in Raw 264.7 cells. Interestingly, Heidinger et al. (2006) cultured THP-1 cells in absence of serum as we did.
Serum may be a cause of data discrepancy in MMP-9 studies. Similar to JNK, p38 MAPK produced a different response in MMP-9 expression that was dependent on the presence of serum in the culture media. In the presence of 10% serum, inhibition of p38 MAPK reduces MMP-9 induction in LPS-activated Raw 264.7 cells (Woo et al., 2004), whereas inhibition of p38 MAPK augments MMP-9 expression in LPS-treated rat astrocytes (Shin et al., 2007), or THP-1 monocytic cells (Heidinger et al., 2006) under serum deprivation.
Even though our observations implicate the existence of the inhibitory factor(s) in serum that suppress MMP-9 expression, our experiments were not designed to pinpoint possible inhibitory factor(s). IFN-γ suppresses MMP-9 expression through the JAK/STAT pathway (Ma et al., 2005). However, IFN-γ could presently be excluded as a candidate, because pyridone 6 did not block the inhibitory effect of mouse serum on MMP-9 expression. TGF-β, IL-4, or IL-10 may also be a candidate as the inhibitory factor in mouse serum. IL-4 suppresses MMP-9 expression in human monocytes stimulated with ConA (Corcoran et al., 1992). TGF-β also reduces MMP-9 expression induced by TNF-α in MonoMac-6 monocytic cells (Vaday et al., 2001). In addition, IL-10 inhibits MMP-9 induction by ConA in human monocytes (Mertz et al., 1994). However, the contribution of these factors to the suppression of MMP-9 expression is questionable, since we obtained serum from healthy mice. Similar to serum, the conditioned media also showed the inhibition of MMP-9 induction. However, it is not clear whether both inhibitory factors are identical. At present, we are purifying the inhibitory factor(s) in the conditioned media of Raw 264.7 cells.
In this study, we observed little change in phosphorylation of p65 of NF-κB, which is a well-known transcriptional factor to induce MMP-9 expression. However, we cannot exclude possibility that JNK1 might inhibit NF-κB activity, because we did not determine the transcriptional activity of NF-κB on the promoter of MMP-9 gene. In drosophila SL2 cells, JNK negatively regulated expression of NF-κB target genes (Kim et al., 2005). JNK caused AP-1 complex to bind to promoters activated by NF-κB, resulting in reduced NF-κB binding. Interestingly, AP-1 binding led to recruitment of histone deacetylase dHDAC1, and induction of the NF-κB target gene was augmented by inhibition of HDAC activity. From these findings, they proposed that the inhibitory action of JNK/AP-1 is a switch to terminate activation of a group of NF-κB target genes. Therefore, in this context, it needs to be clarified whether the negative regulatory mechanism of JNK/AP-1 is working on MMP-9 expression, and which components of AP-1 complex are responsible for this inhibitory action in the future study.
Methods
Reagents
DMEM, BSA, and FBS were purchased from Gibco (Gaithersburg, MD). Control, JNK1, or JNK2 siRNA was purchased from Dharmacon (Chicago, IL). SP600125 was purchased from Alexis (San Diego, CA) and the JAK inhibitor pyridone 6 (P6) was from Calbiochem (San Diego, CA). Antibodies to p-JNK, p-ERK, p-Akt, Akt, p-c-Jun, p-p38 MAPK, and p-p65 were from Cell Signaling Technology (Boston, MA). Antibodies to ERK, α-tubulin, and p38 MAPK were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Antibody to p65 was from Upstate Biotechnology (Lake Placid, NY). LPS (O26:B6), IFN-γ, gelatin, and other chemicals were acquired from Sigma-Aldrich (St. Louis, MO).
Cell culture
Raw 264.7 cells from Korean Cell Line Bank (Seoul, Korea) were grown in DMEM containing 10% FBS, 100 U/ml penicillin, and 100 µg/ml streptomycin at 37℃ in a 5% CO2 humidified incubator. Before experiments, the cells were incubated in DMEM containing 0.02% BSA overnight, and the media was replaced again 4 h prior to each experiment. All the data are representative of triplicate experiments.
RT-PCR of MMP-9 mRNA
RT-PCR was used to determine mRNA expression of MMP-9 and GAPDH. Total RNAs were obtained using a commercial kit (Intron, Sungnam, Korea) according to the manufacturer's instructions. To obtain cDNA, 0.2 µg total RNAs were added to a 20 µl reaction mixture containing 15 U/µl SSII ribonuclease H-reverse transcriptase (Superscript II; Invitrogen), 10 mM Tris-HCl (pH 8.3), 50 mM KCl, 5 mM MgCl2, 0.5 mM dNTPs, 2.5 µM random hexamers, and 2.5 U/µl RNase inhibitor. PCR primers for mouse MMP-9 and GAPDH were as follows: MMP-9, 5'-CAAACCCTGCGTATTTCC-3' (sense) and 5'-AGAGTACTGCTTGCCCAGGA-3' (antisense); GAPDH, 5'-CTCATGACCACAGTCCATGC-3' (sense) and 5'-TTCATCGGGATGACCTT-3' (antisense). PCR was performed with 2 µl of cDNA and 0.2 mM primers using a touchdown PCR; 2 cycles of PCR at 94℃ for 1 min, 64℃ for 1 min and 72℃ for 1 min, followed by 25 cycles (MMP-9) or 20 cycles (GAPDH) of PCR at 94 ℃ for 1 min, 59℃ for 1 min and 72℃ for 1 min. PCR products (MMP-9, 223 bps; GAPDH, 155 bps) were resolved in 1.5% agarose gels containing ethidium bromide. Images were taken by a Gel-Doc apparatus (Kodak, Rochester, NY), and analyzed with Image Gauge software (Fuji, Japan).
Assay of MMP-9 activity by gelatin zymography
The gelatinolytic activity of MMP-9 secreted into the culture medium was determined by gelatin zymography (Chung et al., 2004). Briefly, conditioned media freed of cell debris by centrifugation were mixed with Laemmli buffer lacking reducing agents. After electrophoresis in an 8% SDS-PAGE gel containing 1 mg/ml gelatin, the gel was incubated in a developing buffer (20 mM Tris/HCl, pH 7.8, 1% Triton X-100, 10 mM CaCl2, 5 µM ZnCl2) for 24 h at 37℃. Thereafter, the gel was stained with 1% Coomassie Brilliant Blue R-250. Images of gelatinolytic activities were taken and analysis done as described above.
Preparation of conditioned media of Raw 264.7 cells
Raw 264.7 cells were cultured in DMEM containing 10% FBS until 80% confluence. The cells were washed with PBS and incubated in serum- and BSA-free DMEM. Twenty-four hours later, the conditioned medium was collected, and cell debris was removed by centrifugation at 400 g. The conditioned medium was concentrated and fractionated by ultrafiltration using a 10 kDa molecular weight cutoff (Millipore, Billerica, MA).
Western blot analysis
RAW 264.7 cells were washed with cold PBS and lysed in a cold lysis buffer (20 mM Tris, pH 7.5, 150 mM NaCl, 1 mM EDTA, 1 mM EGTA, 1% Triton X-100, 0.1% SDS, 1% NP-40, 2.5 mM sodium pyrophosphate, 1 mM β-glycerophosphate, 1 mM Na3VO4, 2 mM PNPP, and 1 × protease inhibitor cocktail). After centrifugation at 16,000 g, 30 µg of protein recovered from the supernatant was separated using 12% SDS-PAGE and the separated proteins were transferred to a PVDF membrane. After blocking with 5% skim milk in T-TBS (0.1% Tween 20-Tris buffered saline) for 1 h at room temperature, the membranes were incubated with primary antibodies diluted 1:1,000 at 4℃ overnight, and subsequently with HRP-conjugated secondary antibodies diluted 1:5,000 for 1 h at room temperature. The proteins were visualized using enhanced chemiluminescence reagents. Images were taken by LAS-3000 (Fuji, Japan), and analyzed with Image Gauge software (Fuji).
Transfection of siRNA
Transient transfection of siRNA was performed using HiperFect transfection reagent (Qiagen, Valencia, CA) according to the manufacturer's instructions. Raw 264.7 cells (2 × 105) were seeded into wells of a 24-well plate. siRNA (10-100 nM) was mixed with the transfection reagent, and added to the cells. After 72 h, JNK1 and JNK2 protein levels were determined by Western blotting, and total RNAs or culture media were collected for MMP-9 RT-PCR or MMP-9 zymography, respectively.
Acknowledgments
This work was supported by the Korea Research Foundation Grant funded by the Korean Government (KRF-2007-521-E00027).
Abbreviation
- MMP-9
matrix metalloproteinase-9
References
- 1.Arai K, Lee SR, Lo EH. Essential role for ERK mitogen-activated protein kinase in matrix metalloproteinase-9 regulation in rat cortical astrocytes. Glia. 2003;43:254–264. doi: 10.1002/glia.10255. [DOI] [PubMed] [Google Scholar]
- 2.Atkinson JJ, Senior RM. Matrix metalloproteinase-9 in lung remodeling. Am J Respir Cell Mol Biol. 2003;28:12–24. doi: 10.1165/rcmb.2002-0166TR. [DOI] [PubMed] [Google Scholar]
- 3.Benbow U, Brinckerhoff CE. The AP-1 site and MMP gene regulation: what is all the fuss about? Matrix Biol. 1997;15:519–526. doi: 10.1016/s0945-053x(97)90026-3. [DOI] [PubMed] [Google Scholar]
- 4.Blom AB, van Lent PL, Libregts S, Holthuysen AE, van der Kraan PM, van Rooijen N, van den Berg WB. Crucial role of macrophages in matrix metalloproteinase-mediated cartilage destruction during experimental osteoarthritis: involvement of matrix metalloproteinase 3. Arthritis Rheum. 2007;56:147–157. doi: 10.1002/art.22337. [DOI] [PubMed] [Google Scholar]
- 5.Bogoyevitch MA. The isoform-specific functions of the c-Jun N-terminal Kinases (JNKs): differences revealed by gene targeting. Bioessays. 2006;28:923–934. doi: 10.1002/bies.20458. [DOI] [PubMed] [Google Scholar]
- 6.Chakrabarti S, Patel KD. Matrix metalloproteinase-2 (MMP-2) and MMP-9 in pulmonary pathology. Exp Lung Res. 2005;31:599–621. doi: 10.1080/019021490944232. [DOI] [PubMed] [Google Scholar]
- 7.Chakraborti S, Mandal M, Das S, Mandal A, Chakraborti T. Regulation of matrix metalloproteinases: an overview. Mol Cell Biochem. 2003;253:269–285. doi: 10.1023/a:1026028303196. [DOI] [PubMed] [Google Scholar]
- 8.Cheng JC, Chou CH, Kuo ML, Hsieh CY. Radiation-enhanced hepatocellular carcinoma cell invasion with MMP-9 expression through PI3K/Akt/NF-kappaB signal transduction pathway. Oncogene. 2006;25:7009–7018. doi: 10.1038/sj.onc.1209706. [DOI] [PubMed] [Google Scholar]
- 9.Chung TW, Lee YC, Kim CH. Hepatitis B viral HBx induces matrix metalloproteinase-9 gene expression through activation of ERK and PI-3K/AKT pathways: involvement of invasive potential. FASEB J. 2004;18:1123–1125. doi: 10.1096/fj.03-1429fje. [DOI] [PubMed] [Google Scholar]
- 10.Corcoran ML, Stetler-Stevenson WG, Brown PD, Wahl LM. Interleukin 4 inhibition of prostaglandin E2 synthesis blocks interstitial collagenase and 92-kDa type IV collagenase/gelatinase production by human monocytes. J Biol Chem. 1992;267:515–519. [PubMed] [Google Scholar]
- 11.Crowe DL, Tsang KJ, Shemirani B. Jun N-terminal kinase 1 mediates transcriptional induction of matrix metalloproteinase 9 expression. Neoplasia. 2001;3:27–32. doi: 10.1038/sj.neo.7900135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Deguchi JO, Aikawa M, Tung CH, Aikawa E, Kim DE, Ntziachristos V, Weissleder R, Libby P. Inflammation in atherosclerosis: visualizing matrix metalloproteinase action in macrophages in vivo. Circulation. 2006;114:55–62. doi: 10.1161/CIRCULATIONAHA.106.619056. [DOI] [PubMed] [Google Scholar]
- 13.Eardley KS, Cockwell P. Macrophages and progressive tubulointerstitial disease. Kidney Int. 2005;68:437–455. doi: 10.1111/j.1523-1755.2005.00422.x. [DOI] [PubMed] [Google Scholar]
- 14.Genersch E, Hayess K, Neuenfeld Y, Haller H. Sustained ERK phosphorylation is necessary but not sufficient for MMP-9 regulation in endothelial cells: involvement of Ras-dependent and -independent pathways. J Cell Sci. 2000;113:4319–4330. doi: 10.1242/jcs.113.23.4319. [DOI] [PubMed] [Google Scholar]
- 15.Gum R, Wang H, Lengyel E, Juarez J, Boyd D. Regulation of 92 kDa type IV collagenase expression by the jun aminoterminal kinase- and the extracellular signal-regulated kinase-dependent signaling cascades. Oncogene. 1997;14:1481–1493. doi: 10.1038/sj.onc.1200973. [DOI] [PubMed] [Google Scholar]
- 16.Han S, Ritzenthaler JD, Sitaraman SV, Roman J. Fibronectin increases matrix metalloproteinase 9 expression through activation of c-Fos via extracellular-regulated kinase and phosphatidylinositol 3-kinase pathways in human lung carcinoma cells. J Biol Chem. 2006;281:29614–29624. doi: 10.1074/jbc.M604013200. [DOI] [PubMed] [Google Scholar]
- 17.Heidinger M, Kolb H, Krell HW, Jochum M, Ries C. Modulation of autocrine TNF-alpha-stimulated matrix metalloproteinase 9 (MMP-9) expression by mitogen-activated protein kinases in THP-1 monocytic cells. Biol Chem. 2006;387:69–78. doi: 10.1515/BC.2006.010. [DOI] [PubMed] [Google Scholar]
- 18.Kim T, Yoon J, Cho H, Lee WB, Kim J, Song YH, Kim SN, Yoon JH, Kim-Ha J, Kim YJ. Downregulation of lipopolysaccharide response in Drosophila by negative crosstalk between the AP1 and NF-kappaB signaling modules. Nat Immunol. 2005;6:211–218. doi: 10.1038/ni1159. [DOI] [PubMed] [Google Scholar]
- 19.Lim EJ, Lee SH, Lee JG, Kim JR, Yun SS, Baek SH, Lee C. Toll-like receptor 9 dependent activation of MAPK and NF-kB is required for the CpG ODN-induced matrix metalloproteinase-9 expression. Exp Mol Med. 2007;39:239–245. doi: 10.1038/emm.2007.27. [DOI] [PubMed] [Google Scholar]
- 20.Lin CC, Tseng HW, Hsieh HL, Lee CW, Wu CY, Cheng CY, Yang CM. Tumor necrosis factor-alpha induces MMP-9 expression via p42/p44 MAPK, JNK, and nuclear factor-kappaB in A549 cells. Toxicol Appl Pharmacol. 2008;229:386–398. doi: 10.1016/j.taap.2008.01.032. [DOI] [PubMed] [Google Scholar]
- 21.Ma Z, Chang MJ, Shah RC, Benveniste EN. Interferon-γ-activated STAT-1α suppresses MMP-9 gene transcription by sequestration of the coactivators CBP/p300. J Leukoc Biol. 2005;78:515–523. doi: 10.1189/jlb.0205112. [DOI] [PubMed] [Google Scholar]
- 22.Matsuguchi T, Musikacharoen T, Johnson TR, Kraft AS, Yoshikai Y. A novel mitogen-activated protein kinase phosphataseis an important negative regulator of lipopolysaccharide-mediated c-Jun N-terminal kinase activation in mouse macrophage cell lines. Mol Cell Biol. 2001;21:6999–7009. doi: 10.1128/MCB.21.20.6999-7009.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mertz PM, DeWitt DL, Stetler-Stevenson WG, Wahl LM. Interleukin 10 suppression of monocyte prostaglandin H synthase-2. Mechanism of inhibition of prostagland-independent matrix metalloproteinase production. J Biol Chem. 1994;269:21322–21329. [PubMed] [Google Scholar]
- 24.Moon SK, Cha BY, Kim CH. ERK1/2 mediates TNF-alpha-induced matrix metalloproteinase-9 expression in human vascular smooth muscle cells via the regulation of NF-kappaB and AP-1: Involvement of the ras dependent pathway. J Cell Physiol. 2004;198:417–427. doi: 10.1002/jcp.10435. [DOI] [PubMed] [Google Scholar]
- 25.Opdenakker G, Van den Steen PE, Dubois B, Nelissen I, Van Coillie E, Masure S, Proost P, Van Damme J. Gelatinase B functions as regulator and effector in leukocyte biology. J Leukoc Biol. 2001;69:851–859. [PubMed] [Google Scholar]
- 26.Sabapathy K, Hochedlinger K, Nam SY, Bauer A, Karin M, Wagner EF. Distinct roles for JNK1 and JNK2 in regulating JNK activity and c-Jun-dependent cell proliferation. Mol Cell. 2004;15:713–725. doi: 10.1016/j.molcel.2004.08.028. [DOI] [PubMed] [Google Scholar]
- 27.Sánchez-Tilló E, Comalada M, Xaus J, Farrera C, Valledor AF, Caelles C, Lloberas J, Celada A. JNK1 Is required for the induction of Mkp1 expression in macrophages during proliferation and lipopolysaccharide-dependent activation. J Biol Chem. 2007;282:12566–12573. doi: 10.1074/jbc.M609662200. [DOI] [PubMed] [Google Scholar]
- 28.Shapiro SD. The maxcrophage in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1999;160:S29–S32. doi: 10.1164/ajrccm.160.supplement_1.9. [DOI] [PubMed] [Google Scholar]
- 29.Shin CY, Lee WJ, Choi JW, Choi MS, Park GH, Yoo BK, Han SY, Ryu JR, Choi EY, Ko KH. Role of p38 MAPK on the down-regulation of matrix metalloproteinase-9 expression in rat astrocytes. Arch Pharm Res. 2007;30:624–633. doi: 10.1007/BF02977658. [DOI] [PubMed] [Google Scholar]
- 30.Shin M, Yan C, Boyd D. An inhibitor of c-jun aminoterminal kinase (SP600125) represses c-Jun activation, DNA-binding and PMA-inducible 92-kDa type IV collagenase expression. Biochim Biophys Acta. 2002;1589:311–316. doi: 10.1016/s0167-4889(02)00195-7. [DOI] [PubMed] [Google Scholar]
- 31.Vaday GG, Schor H, Rahat MA, Lahat N, Lider O. Transforming growth factor-beta suppresses tumor necrosis factor alpha-induced matrix metalloproteinase-9 expression in monocytes. J Leukoc Biol. 2001;69:613–621. [PubMed] [Google Scholar]
- 32.Van den Steen PE, Proost P, Wuyts A, Van Damme J, Opdenakker G. Neutrophil gelatinase B potentiates interleukin-8 tenfold by aminoterminal processing, whereas it degrades CTAP-III, PF-4, and GRO-alpha and leaves RANTES and MCP-2 intact. Blood. 2000;96:2673–2681. [PubMed] [Google Scholar]
- 33.Waetzig V, Czeloth K, Hidding U, Mielke K, Kanzow M, Brecht S, Goetz M, Lucius R, Herdegen T, Hanisch UK. c-Jun N-terminal kinases (JNKs) mediate pro-inflammatory actions of microglia. Glia. 2005;50:235–246. doi: 10.1002/glia.20173. [DOI] [PubMed] [Google Scholar]
- 34.Woo CH, Lim JH, Kim JH. Lipopolysaccharide induces matrix metalloproteinase-9 expression via a mitochondrial reactive oxygen species-p38 kinase-activator protein-1 pathway in Raw 264.7 cells. J Immunol. 2004a;173:6973–6980. doi: 10.4049/jimmunol.173.11.6973. [DOI] [PubMed] [Google Scholar]
- 35.Wu CY, Hsieh HL, Jou MJ, Yang CM. Involvement of p42/p44 MAPK, p38 MAPK, JNK and nuclear factor-kappa B in interleukin-1beta-induced matrix metalloproteinase-9 expression in rat brain astrocytes. J Neurochem. 2004;90:1477–1488. doi: 10.1111/j.1471-4159.2004.02682.x. [DOI] [PubMed] [Google Scholar]
- 36.Xie B, Dong Z, Fidler IJ. Regulatory mechanisms for the expression of type IV collagenases/gelatinases in murine macrophages. J Immunol. 1994;152:3637–3644. [PubMed] [Google Scholar]
- 37.Xie Z, Singh M, Singh K. Differential regulation of matrix metalloproteinase-2 and -9 expression and activity in adult rat cardiac fibroblasts in response to interleukin-1β. J Biol Chem. 2004;279:39513–39519. doi: 10.1074/jbc.M405844200. [DOI] [PubMed] [Google Scholar]
- 38.Yan C, Boyd DD. Regulation of matrix metalloproteinase gene expression. J Cell Physiol. 2007;211:19–26. doi: 10.1002/jcp.20948. [DOI] [PubMed] [Google Scholar]
- 39.Zhong S, Fromm J, Johnson DL. TBP is differentially regulated by c-Jun N-terminal kinase 1 (JNK1) and JNK2 through Elk-1, controlling c-Jun expression and cell proliferation. Mol Cell Biol. 2007;27:54–64. doi: 10.1128/MCB.01365-06. [DOI] [PMC free article] [PubMed] [Google Scholar]