Abstract
During development, dorsal root ganglion (DRG) neurons extend their axons toward the dorsolateral part of the spinal cord and enter the spinal cord through the dorsal root entry zone (DREZ). After entering the spinal cord, these axons project into the dorsal mantle layer after a “waiting period” of a few days. We revealed that the diffusible axonal guidance molecule netrin-1 is a chemorepellent for developing DRG axons. When DRG axons orient themselves toward the DREZ, netrin-1 proteins derived from the ventral spinal cord prevent DRG axons from projecting aberrantly toward the ventral spinal cord and help them to project correctly toward the DREZ. In addition to the ventrally derived netrin-1, the dorsal spinal cord cells adjacent to the DREZ transiently express netrin-1 proteins during the waiting period. This dorsally derived netrin-1 contributes to the correct guidance of DRG axons to prevent them from invading the dorsal spinal cord. In general, there is a complete lack of sensory axonal regeneration after a spinal cord injury, because the dorsal column lesion exerts inhibitory activities toward regenerating axons. Netrin-1 is a novel candidate for a major inhibitor of sensory axonal regeneration in the spinal cord; because its expression level stays unchanged in the lesion site following injury, and adult DRG neurons respond to netrin-1-induced axon repulsion. Although further studies are required to show the involvement of netrin-1 in preventing the regeneration of sensory axons in CNS injury, the manipulation of netrin-1-induced repulsion in the CNS lesion site may be a potent approach for the treatment of human spinal injuries.
Key words: netrin-1, dorsal root ganglion, axon guidance, chemorepellent, Unc5, spinal cord, axon regeneration
Developing axons navigate to their targets by responding to attractive and repulsive guidance cues working in a contact-dependent or diffusible fashion in their environment (reviewed in ref. 1). During early development of the primary sensory system, centrally projecting sensory axons from dorsal root ganglion (DRG) neurons extend toward the dorsolateral region of the spinal cord (Fig. 1A and C), where they enter the spinal cord exclusively through the dorsal root entry zone (DREZ), and never orient themselves toward the notochord or the ventral spinal cord (Fig. 1A; reviewed in ref. 2). We previously showed that the notochord but not the ventral spinal cord secretes semaphorin 3A (Sema3A), which is known to be a chemorepellent for DRG axons at early developmental stages (Fig. 1A).3 This is the reason why DRG axons never project toward the notochord. Along the same line, it is highly possible that the ventral spinal cord may secrete some chemorepulsive cue other than Sema3A for DRG axons.
Figure 1.
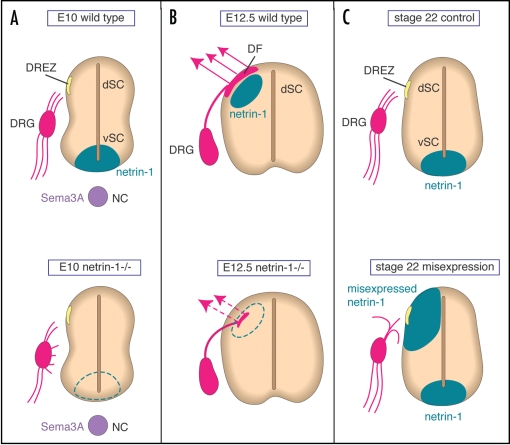
Netrin-1 plays a critical role in sensory axonal guidance as an axon chemorepellent. (A) A schematic diagram of a thoracic transverse section of an E10 mouse embryo, summarizing the possible mechanism of netrin-1 action in early DRG axonal guidance. When DRG axons project toward the DREZ in the dorsal spinal cord (dSC), ventrally derived netrin-1 chemorepels DRG axons to prevent them from orienting aberrantly toward the ventral spinal cord (vSC) (upper). NC; notochord. In netrin-1-deficient embryos, some DRG axons misorient themselves toward the ventral spinal cord, because of the absence of netrin-1 proteins in the ventral spinal cord (lower). (B) At E12.5 when DRG axons grow to the marginal zone of the spinal cord longitudinally (arrows) to form the dorsal funiculus (DF), netrin-1 proteins are transiently expressed in a subpopulation of dorsal spinal cord cells adjacent to the dorsal funiculus (upper). In netrin-1-deficient embryos, the dorsal funiculus is disorganized because DRG axons are no longer waiting for invading the dorsal mantle layer (lower). (C) Gain-of-function experiments by electroporation confirm the repulsive activity of netrin-1 toward DRG axons. When netrin-1 is misexpressed in the dorsal spinal cord, the number of DRG axons that enter the DREZ is significantly reduced compared with the control, because some DRG axons fail to project toward the DREZ and turn in the wrong direction.
After entering the spinal cord, DRG axons grow to the marginal zone of the spinal cord longitudinally to form the dorsal funiculus without projecting to the dorsal mantle layer for a few days (this delay of the axonal projection to the mantle layer is referred to as the ‘waiting period;’ Fig. 1B). A few days later, proprioceptive afferents of DRGs begin to send collaterals into the dorsal layers, and cutaneous afferents project ventrally through the dorsal layers.4 This evidence raises the possibility that some repulsive cues transiently prevent the collaterals of DRGs from penetrating the dorsal spinal cord during this waiting period.
Netrins are a family of secreted proteins that play a key role in axonal guidance, cell migration, morphogenesis and angiogenesis.5 Netrin-1 is a bifunctional axonal guidance cue, attracting some axons including commissural axons via the Deleted in Colorectal Cancer (DCC) receptor and repelling others via Unc5 receptors (reviewed in ref. 6). However, it has not been clear whether netrin-1 plays a role in sensory axonal guidance during development.
Several observations strongly suggest a role for netrin-1 in DRG axonal guidance as a repulsive guidance cue during development.7,8 First, in the mouse embryo at embryonic day (E) 10–11.5 when many DRG axons orient themselves to reach the DREZ, netrin-1 is strongly expressed in the floor plate of the ventral spinal cord but not in the dorsal spinal cord (Fig. 1A). Second, at E12.5 when DRG neurons extend their axons longitudinally along the dorsolateral margin of the spinal cord, netrin-1 is expressed in the dorsolateral region adjacent to the DREZ (Fig. 1B), but its expression is down-regulated in the dorsal spinal cord at E13.5 when many collaterals have entered the mantle layer. Third, repulsive netrin-1 receptor Unc5c is expressed in the DRG neurons during development.
These observations motivated us to explore whether netrin-1/Unc5c signaling contributes to DRG axonal guidance. We used cell and tissue cultures combined with tissues from netrin-1-deficient mice. We clearly showed that netrin-1 exerts a chemorepulsive activity toward developing DRG axons and that the ventral spinal cord-derived repulsive activity depends on netrin-1 in vitro.8 Additional evidence for a chemorepulsive role of netrin-1 came from the observation of DRG axonal trajectories in netrin-1-deficient mice.7,8 In netrin-1-deficient embryos at E10, we showed that some DRG axons became misoriented toward the ventral spinal cord, probably because of the absence of netrin-1 proteins in the ventral spinal cord (Fig. 1A). In addition, at E12.5 when DRG axons grow to the marginal zone of the spinal cord longitudinally to form the dorsal funiculus, the dorsal funiculus is disorganized in netrin-1-deficient embryos, because in the absence of netrin-1 DRG axons are not waiting for invading the dorsal mantle layer adjacent to the dorsal funiculus (Fig. 1B). Gain-of-function experiments further confirmed the repulsive activity of netrin-1 toward DRG axons (Fig. 1C). These lines of evidence lead us to the conclusion that dorsally derived netrin-1 plays an important role in providing the ‘waiting period’ for extension of collaterals from sensory afferents and that ventrally derived netrin-1 prevents sensory axons from misorienting themselves toward the ventral spinal cord.
At later developmental stages (E13.5), DRG axons still possess a weak responsiveness to the chemorepulsive activity of netrin-1 in vitro.8 In addition, both postnatal and adult DRG neurons respond to netrin-1-induced axon inhibition.9 Consistent with these results, DRG neurons at not only later developmental stages (E13.5) but also postnatal stages express the repulsion-mediating netrin-1 receptor Unc5c.8,9
Generally, lesioning of the dorsal column projection of sensory axons results in a complete lack of regeneration. The possible explanation for the complete lack of regeneration is that the environment, the lesion site itself and/or oligodendrocytes adjacent to the lesion, may be non-permissive for regenerating axons.10 Sema3A and chondroitin sulfate proteoglycans (CSPGs) are candidates as major inhibitors of sensory axonal regeneration in the spinal cord, because they are expressed in the lesion site and can inhibit DRG axonal growth in vitro.3,11–14 Recently, Kaneko et al. showed that a selective inhibitor of Sema3A also enhances axonal regeneration and functional recovery in a subpopulation of sensory neurons after lesioning of the dorsal column.12 More recently, McMahon's group clearly demonstrated that enzymatic degradation of CSPGs on the dorsal column lesion of the spinal cord promotes sensory axonal regeneration and functional recovery.13,14 Although these treatments greatly improved functional recovery, complete sensory axonal growth and functional recovery have not been yet achieved after the spinal cord injury. To promote further recovery of sensory axonal regeneration in the CNS, we should focus on other candidate inhibitors of CNS injury sites.
Following spinal cord injury, the expression of the attraction- mediating netrin-1 receptor DCC decreases, while the expression level of the repulsive receptor Unc5c returns to normal.15 Levels of netrin-1 expression also stay unchanged in neurons and oligodendrocytes adjacent to the lesion site. Together with the in vitro evidence described above, these data strongly suggest a possible role for netrin-1 as a novel inhibitor of CNS myelin for regenerating DRG axons in the dorsal column-lesioned spinal cord. Further studies will be required to show directly the functional recovery of sensory axons in the spinal cord by perturbation of netrin-1 in and around the lesion site after spinal cord injury.
Acknowledgements
This work was supported by grants from the program Grants-in-Aid for Scientific Research of the MEXT, Japan, and by grants from the Brain Science Foundation, Japan.
Footnotes
Previously published online as a Cell Adhesion & Migration E-publication: http//www.landesbioscience.com/journals/celladhesion/article/7837
References
- 1.Tessier-Lavigne M, Goodman CS. The molecular biology of axon guidance. Science. 1996;274:1123–1133. doi: 10.1126/science.274.5290.1123. [DOI] [PubMed] [Google Scholar]
- 2.Masuda T, Shiga T. Chemorepulsion and cell adhesion molecules in patterning initial trajectories of sensory axons. Neurosci Res. 2005;51:337–347. doi: 10.1016/j.neures.2005.01.007. [DOI] [PubMed] [Google Scholar]
- 3.Masuda T, Tsuji H, Taniguchi M, Yagi T, Tessier-Lavigne M, Fujisawa H, et al. Differential non-target-derived repulsive signals play a critical role in shaping initial axonal growth of dorsal root ganglion neurons. Dev Biol. 2003;254:289–302. doi: 10.1016/s0012-1606(02)00087-8. [DOI] [PubMed] [Google Scholar]
- 4.Ozaki S, Snider WD. Initial trajectories of sensory axons toward laminar targets in the developing mouse spinal cord. J Comp Neurol. 1997;380:215–229. [PubMed] [Google Scholar]
- 5.Cirulli V, Yebra M. Netrins: beyond the brain. Nat Rev Mol Cell Biol. 2007;8:296–306. doi: 10.1038/nrm2142. [DOI] [PubMed] [Google Scholar]
- 6.Moore SW, Tessier-Lavigne M, Kennedy TE. Netrins and their receptors. Adv Exp Med Biol. 2007;621:17–31. doi: 10.1007/978-0-387-76715-4_2. [DOI] [PubMed] [Google Scholar]
- 7.Watanabe K, Tamamaki N, Furuta T, Ackerman SL, Ikenaka K, Ono K. Dorsally derived netrin 1 provides an inhibitory cue and elaborates the ‘waiting period’ for primary sensory axons in the developing spinal cord. Development. 2006;133:1379–1387. doi: 10.1242/dev.02312. [DOI] [PubMed] [Google Scholar]
- 8.Masuda T, Watanabe K, Sakuma C, Ikenaka K, Ono K, Yaginuma H. Netrin-1 acts as a repulsive guidance cue for sensory axonal projections toward the spinal cord. J Neurosci. 2008;28:10380–10385. doi: 10.1523/JNEUROSCI.1926-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Park JI, Seo IA, Lee HK, Park HT, Shin SW, Park YM, et al. Netrin-1 inhibits regenerative axon growth of adult dorsal root ganglion neurons in vitro. J Korean Med Sci. 2007;22:641–645. doi: 10.3346/jkms.2007.22.4.641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bradbury EJ, McMahon SB, Ramer MS. Keeping in touch: sensory neurone regeneration in the CNS. Trends Pharmacol Sci. 2000;21:389–394. doi: 10.1016/s0165-6147(00)01536-4. [DOI] [PubMed] [Google Scholar]
- 11.Masuda T, Fukamauchi F, Takeda Y, Fujisawa H, Watanabe K, Okado N, et al. Developmental regulation of notochord-derived repulsion for dorsal root ganglion axons. Mol Cell Neurosci. 2004;25:217–227. doi: 10.1016/j.mcn.2003.10.005. [DOI] [PubMed] [Google Scholar]
- 12.Kaneko S, Iwanami A, Nakamura M, Kishino A, Kikuchi K, Shibata S, et al. A selective Sema3A inhibitor enhances regenerative responses and functional recovery of the injured spinal cord. Nat Med. 2006;12:1380–1389. doi: 10.1038/nm1505. [DOI] [PubMed] [Google Scholar]
- 13.Bradbury EJ, Moon LDF, Popat RJ, King VR, Bennett GS, Patel PN, et al. Chondroitinase ABC promotes functional recovery after spinal cord injury. Nature. 2002;416:636–640. doi: 10.1038/416636a. [DOI] [PubMed] [Google Scholar]
- 14.Cafferty WBJ, Bradbury EJ, Lidierth M, Jones M, Duffy PJ, Pezet S, et al. Chondroitinase ABC-mediated plasticity of spinal sensory function. J Neurosci. 2008;28:11998–12009. doi: 10.1523/JNEUROSCI.3877-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Manitt C, Wang D, Kennedy TE, Howland DR. Positioned to inhibit: netrin-1 and netrin receptor expression after spinal cord injury. J Neurosci Res. 2006;84:1808–1820. doi: 10.1002/jnr.21070. [DOI] [PubMed] [Google Scholar]