Abstract
Aim
To investigate the association between sex hormone levels and the risk of falling in older men and women.
Methods
199 men and 246 women age 65 or older living at home followed for 3 years after baseline assessment of sex hormones. Analyses controlled for age, bmi, baseline 25-hydroxyvitamin D, sex hormone binding globulin, physical activity, smoking, alcohol use, number of comorbidities, maximal follow-up, and vitamin D plus calcium treatment.
Results
Compared to the lowest quartile, men and women in the highest sex-specific quartile of total testosterone had a 78% and 66% decreased risk of falling (men: OR = 0.22; 95% CI [0.07,0.72]/ women: OR = 0.34; 95% CI [0.14,0.83]); if those individuals also took calcium plus vitamin D the anti-fall effect was enhanced to 84% among men and 85% among women (men: OR = 0.16; 95% CI [0.03,0.90] / women: OR = 0.15; 95% CI [0.04,0.57]). Women in the top quartile of dihydroepiandrosterone sulfate (DHEA-S) had a significant 61% lower risk of falling compared to those in the lowest quartile (OR = 0.39; 95% CI [0.16,0.93]). Other sex hormones and SHBG did not predict falling in men or women.
Conclusion
Higher physiologic total testosterone in both genders and higher DHEA-S levels in women predicted a more than 60% lower risk of falling. The anti-fall benefit of higher physiologic testosterone levels is enhanced to over 80% by additional calcium and vitamin D supplementation.
Keywords: falls, testosterone, sex hormones, older individuals
INTRODUCTION
An inverse association between sex hormone levels and hip fracture risk has been documented1, explained by benefits on bone remodeling2 and bone density3,4. Alternatively, lower sex hormone levels may be associated with fall risk as an alternative pathway explaining elevated hip fracture risk with age. Given falling being a primary risk factor of hip fracture risk among older individuals, factors that relate to falling need careful evaluation, especially if deficiencies are amendable to treatment.
Testosterone levels decline with age in both men and women5 and testosterone replacement may increase lean body mass6 and muscle strength7 in men with low testosterone levels. Among healthy older men with normal to mildly decreased testosterone levels, testosterone supplementation increased lean body mass6-9, although strength and functional performance were unchanged with testosterone replacement in some studies performed in men age 65 and older7,10. Regarding fall risk and testosterone levels, observational studies have been inconsistent with an inverse association documented in one11 of two prospective studies12 among older men. Whether higher testosterone levels reduce fall risk among women, where levels of the hormone are far lower, is unclear4,12.
Bioavailable and free estradiol levels decline with age in men and women13. While some studies found that estrogen therapy may enhance muscle strength in older women14,15, others did not find a benefit16,17. Limited data from one observational study suggested no association between estrogen levels and muscle strength or fall risk in older men or women12.
DHEA-S exerts its action indirectly after its conversion to androgens and estrogens in peripheral tissues. Its association with falling among older individuals is unclear with two observational studies suggesting a positive correlation of the hormone with muscle strength in older men and women18,19. Whether sex hormone binding globuline (SHBG) is associated with falling has not been explored. However, SHBG may play an important adverse role in older individuals as it increases with age and possibly contributes to a decreased bioavailability of sex hormones4.
Given the scarcity of studies on the role of sex hormone levels and the risk of falling in older persons, we assess the association of baseline sex hormone levels and subsequent risk of falling over a 3 –year follow-up. Taking advantage of the blinded intervention with vitamin D plus calcium within this data set, we were able to explore a possible additive benefit of higher hormone levels and treatment with vitamin D plus calcium. Vitamin D similar to testosterone and estrogen, is a steroid hormone, and previous randomized controlled trials suggested a significant improvement of strength20 and reduction of falls21-23 with vitamin D treatment. In the same data set, vitamin D plus calcium significantly reduced the odds of falling in women (odds ratio [OR], 0.54; 95% confidence interval [CI], 0.30−0.97), but not in men (OR, 0.93; 95% CI, 0.50−1.72)23. If there was an additive benefit, future intervention studies targeting falls may optimize both hormone levels and vitamin D plus calcium intake to decrease fall risk most efficiently. Finally, we studied the association of sex hormone levels with total and leg lean body mass.
METHODS
Subjects
The Boston Stop-It trial is a 3-year double-blind randomized controlled trial on the effect of vitamin D3 (700 IU per day) plus calcium (500 mg per day) on bone mineral density and fractures24. Of 848 persons who were prescreened with questionnaires, 545 were invited for screening. The final study sample was 445 subjects (199 men and 245 women). Apart from DXA measurements and fractures, falls were assessed throughout the trial, as well as sex hormone levels at baseline. The latter data were used in the present analyses. All participants provided written informed consent and the study protocol was approved by the Investigation Review Board at Tufts University.
Exclusion criteria
The trial enrolled healthy ambulatory older men and women age 65 or older living in the community24. The trial excluded individuals with Parkinsons disease or hemiplegia, cancer or hyperparathyroidism. The criteria of exclusion also included a kidney stone in the past 5 years, renal disease (serum creatinine > 1.2 mg/dl), liver disease, bilateral hip surgery, dietary calcium intake exceeding 1500 mg/day, therapy with testosterone, estrogen, tamoxifen, bisphosphonate, fluoride or calcitonin in the past two years.
Follow-up
Of 445 subjects who were randomized, 389 attended the 3-year follow-up visit and 318 were still on study medication at the 3-year follow-up visit. For this analysis, we used all 445 individuals randomized.
Fall definition and ascertainment
Falls were ascertained by postcards. Participants were asked to send a postcard after every fall, which was then followed by a phone call from a staff member to assess the circumstances of the fall. In addition, falls were ascertained at every 6-month follow-up visit.
We included all reported fall events. Falls were defined as ”unintentionally coming to rest on the ground, floor, or other lower level“25. Falls due severe trauma involving external force or vehicles were not counted as a fall. Falling at least once was the primary outcome of the analyses.
Measurements
Baseline BMI is weight in kilograms divided by height in meters squared measured at the study center. Physical activity included leisure, household, and occupational activity as estimated by the Physical Activity Scale for the Elderly (PASE) questionnaire26. Tobacco use and use of alcoholic beverages was assessed by a questionnaire at baseline.
Comorbid conditions assessed at baseline with a questionnaire were summarized with a comorbidity score, which represents the sum of the following comorbid conditions: diabetes, hyperthyroidism, hypertension, cancer, low back surgery, previous hip fracture, and stomach surgery.
Total and leg lean body mass was measured by dual-energy x-ray absorptiometry using a DPX-L scanner (Lunar Radiation, Madison Wisconsin).
Laboratory investigations
On the baseline visit, venous blood was collected between 7:00 and 9:30 a.m. after the subjects had fasted for at least 8 hours. Plasma 25-OHD levels were measured by competitive protein binding assay, as described by Preece et al., with intra- and interassay CVs of 5.6 to 7.7%27. All following hormones were measured in the laboratory of the late Dr. Christopher Longcope at the University of Massachusetts in Worcester. Estrone and estradiol were measured in serum by radioimmunoassay following solvent extraction and celite chromatography. The intra- and inter-assay CVs for estrone were 5.0 and 10.0% and for estradiol were 7.0 and 13.2%, respectively. The estradiol assay had a detection level of 5 to 7 pg/ml. Androstenedione was measured in serum using radioimmunoassay kits from Diagnostic System Laboratories (Webster, TX). This antibody is highly specific with negligible cross-reaction with other steroids. The intraand inter-assay CVs were 7.3 and 9.8%, respectively. DHEA-S was measured in serum using radioimmunoassay kits from ICN Biomedical (CostaMesa, CA) with relatively high cross-reactions, 30 − 60%, with dehydroepiandrosterone and androstenedione. Since DHEA-S circulates at levels at least 1,000 times those of the other two steroids, this cross-reaction does not interfere with the assay. The intra- and inter-assay CVs were 4.3 and 8.6%, respectively. Total testosterone was measured in serum using radioimmunoassay kits from Diagnostic Products Corp (Los Angeles, CA). There are no significant cross-reactions with other natural steroids. The intra- and inter-assay CVs were 5.9 and 8.7%, respectively. Free testosterone was measured by the method of Hammond et al.28. The measurement was done with centrifugal ultrafiltration and the inter- and intra-assay CVs are 8.9 and 5.2%, respectively.
Statistical analysis
We used logistic regression to evaluate the effect of quartiles of sex hormone levels with the lowest quartile as the reference on a person's risk of falling at least once during the 3-year follow-up. In men and women, all analyses were controlled for age in years, baseline BMI in kg/m2 (< 25, 25−29, > 30), baseline plasma 25-OHD levels, baseline PASE status for physical activity assessment, baseline smoking status (never, current, former smoker), baseline use of alcoholic beverages (yes/no), baseline number of comorbid conditions, treatment with vitamin D plus calcium or placebo, and length of follow-up in days. The analyses for total testosterone, estrone and estradiol were also controlled for SHBG in men and women.
In addition, we compared the baseline mean total lean body mass and the baseline mean leg lean body mass among quartiles of baseline sex hormone levels using a multiple linear regression model while adjusting for age in years, baseline BMI in kg/m2 (< 25, 25−29, > 30), baseline plasma 25-OHD levels, baseline PASE, baseline smoking status (never, current, former smoker), baseline use of alcoholic beverages (yes/no) and baseline number of comorbid conditions. Least square means were used to express the adjusted mean percent difference in lean body mass by quartiles of sex hormone levels.
All analyses were conducted with SAS (Version 8.2; SAS Institute Inc., Cary, NC, USA). All p-values were 2-sided.
RESULTS
Baseline characteristics by sex are displayed in Table 1. While mean age was the same for men and women, women were less physically active, had lower 25-hydroxyvitamin D levels and were less likely to be ever smokers. Hormone levels, differed by sex significantly with one exception, estrone. Overall, 49% (97) of men and 57% (134) of women fell during the 3 year follow-up.
Table 1. Characteristics of the study population.
PASE is the Physical Activity Score for the Elderly.
| Characteristic Mean ± Standard Deviation | Men No. = 199 | Women No. = 247 | p-value |
|---|---|---|---|
| Age (yr) | 71 ± 5 | 71 ± 5 | 0.29 |
| BMI (kg/m2) | 27.0 ± 3.4 | 26.7 ± 4.8 | 0.44 |
| 25-OHD (ng/l) | 33.0 ± 14.2 | 26.6 ± 12.7 | < 0.0001 |
| Physical Activity (PASE) | 124 ± 57 | 106 ± 50 | 0.003 |
| Ever Smokers (percent) | 67% | 52% | 0.004 |
| Alcoholic Bev. / day (percent) | 74% | 65% | 0.05 |
| Testosterone (ng/ml) | 4.98 + 1.54 | 0.37 + 0.27 | < 0.0001 |
| Free testosterone (pg/ml) | 1.42 + 0.36 | 1.04 + 0.49 | < 0.0001 |
| SHBG (ng/l) | 56.0 + 22.3 | 75.1 + 36.4 | < 0.0001 |
| DHEA-S (μg/ml) | 0.98 + 0.69 | 0.60 + 0.45 | < 0.0001 |
| Estrone (pg/ml) | 29.9 + 14.0 | 30.1 + 18.9 | 0.87 |
| Estradiol (pg/ml) | 28.3 + 11.2 | 25.5 + 16.9 | 0.04 |
| Androstenedione (ng/ml) | 1.28 + 0.54 | 1.00 + 0.55 | < 0.0001 |
Risk of falling by hormone levels
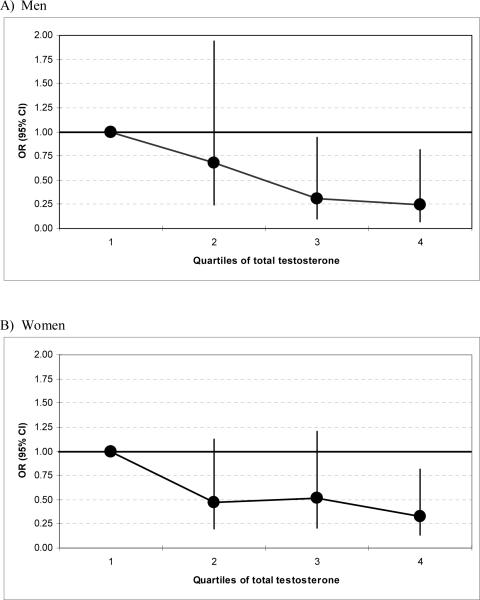
Among men, total testosterone levels independent of SHBG were significantly associated with the odds of falling. Men in the highest quartile of total testosterone with serum levels of 5.68 ng/ml or above had a 78% decreased odds of falling compared to men in the lowest quartile with total testosterone levels of 3.77 ng/ml or less (OR = 0.22; 95% CI [0.07,0.72]). There was a significant trend between a lower odds of falling and higher total testosterone levels (p = 0.005). See Figure 1 for exact boundaries of quartiles and illustration of the observed trend.
Figure 1. Odds of falling by quartile of total testosterone in men and in women.
Independent of age, body mass index, physical activity, SHBG levels, 25(OH)D levels, vitamin D plus calcium treatment, number of comorbid conditions, smoking and alcohol consumption, there was a significant trend in men (test for trend: p = 0.005) and women (test for trend: p = 0.03) suggesting a decrease in the odds of falling with higher testosterone levels. Comparing the two extreme quartiles, men in the highest quartile had a 78% and women had a 66% lower odds of falling. Results were similar with or without adjustment for SHBG.
Among women, similar to men, there was a significant trend between a lower odds of falling and higher testosterone levels (p = 0.03). Women in the top quartile of testosterone with serum levels of 0.49 ng/ml and above had a 66% decreased risk of falling compared to those in the lowest quartile with serum levels of 0.20 ng/ml or less (OR = 0.34; 95% CI [0.14,0.83]). See Figure 1 for exact boundaries of quartiles and illustration of the observed trend.
Also, women in the top quartile of DHEA-S had a 61% lower risk of falling compared to those in the lowest quartile (OR = 0.39; 95% CI [0.16,0.93] (see Table 2). There appeared to be a threshold effect with a benefit in all women reaching serum levels of above 0.30 μg/ml, the upper end of the lowest quartile. Among men, there was a similar directionality with a decreased risk of falling with higher DHEA-S levels. However, this was not significant.
Table 2. Odds of falling across quartiles of estradiol, DHEA-S, and SHBG.
All analyses controlled for age, baseline BMI, baseline plasma 25-OHD levels, baseline PASE status for physical activity assessment, baseline smoking status, baseline use of alcoholic beverages, baseline number of comorbid conditions, treatment with vitamin D plus calcium or placebo, and length of follow-up in days. Similar to estrone, there was no significant association between the odds of falling and estradiol and androstenedione levels.
| Range of top and lowest quartile by sex | Effect Men OR [95% CI] p-value for trend | Effect Women OR [95% CI] p-value for trend | |||
|---|---|---|---|---|---|
| Estradiol (pg/ml) | |||||
| Men bottom: | ≤ 17 | - | Reference | ||
| Men second: | 18.0 − 24.0 | 0.42 | [0.16,1.16] | ||
| Men third: | 25.0 − 31.0 | 0.63 | [0.24,1.65] | ||
| Men top: | > 31.0 | 0.88 | [0.35−2.23] | ||
| *p = 0.98 | |||||
| Women bottom: | ≤ 14 | - | Reference | ||
| Women second: | 15.0 − 21.0 | 1.79 | [0.75−4.22] | ||
| Women third: | 22.0 − 30.0 | 1.36 | [0.59−3.13] | ||
| Women top: | > 30.0 | 0.92 | [0.40−2.13] | ||
| |
|
|
|
|
*p = 0.76 |
| DHEA-S (μg/ml) | |||||
| Men bottom: | ≤ 0.53 | - | Reference | ||
| Men second: | 0.54 − 0.78 | 0.90 | [0.33,2.44] | ||
| Men third: | 0.79 − 1.20 | 0.49 | [0.18,1.31] | ||
| Men top: | > 1.20 | 0.56 | [0.21−1.46] | ||
| *p = 0.14 | |||||
| Women bottom: | ≤ 0.30 | - | Reference | ||
| Women second: | 0.31 − 0.45 | 0.27 | [0.11,0.63]** | ||
| Women third: | 0.46 − 0.75 | 0.43 | [0.18,1.02] | ||
| Women top: | > 0.75 | 0.39 | [0.16−0.93]** | ||
| |
|
|
|
|
*p = 0.08 |
| SHBG (ng/ml) | |||||
| Men bottom: | ≤ 41.05 | - | Reference | ||
| Men second: | 41.06 − 52.93 | 2.86 | [1.08,7.54]** | ||
| Men third: | 52.94 − 65.46 | 0.98 | [0.38,2.54] | ||
| Men top: | > 65.46 | 1.54 | [0.57−4.14] | ||
| *p = 0.89 | |||||
| Women bottom: | ≤ 51.53 | - | Reference | ||
| Women second: | 51.54 − 65.08 | 0.66 | [0.27,1.60] | ||
| Women third: | 65.09 − 90.25 | 1.21 | [0.49,2.95] | ||
| Women top: | > 90.25 | 0.57 | [0.22−1.41] | ||
| *p = 0.47 | |||||
p-value for trend test.
Significantly different from reference.
Estrone, estradiol, free testosterone, androstenedione, and SHBG were not significantly associated with the odds of falling.
As this was a double-blind RCT with vitamin D plus calcium compared to placebo23, we were able to explore a possible additive benefit of higher testosterone levels and vitamin D plus calcium supplementation on fall prevention in Table 3. Among men and women, there was an additional benefit of being in the sex-specific top quartile of serum testosterone plus being randomized to vitamin D and calcium. The OR of falling among men in the top quartile of serum testosterone declined from 0.23 in the placebo group to 0.16 in the vitamin D plus calcium group. Similarly, the OR of falling among women in the top quartile of serum testosterone declined from 0.41 in the placebo group to 0.15 in the vitamin D plus calcium group.
Table 3. Effect of higher testosterone levels on falling by vitamin D + calcium supplementation.
All analyses controlled for age, baseline BMI, baseline plasma 25-OHD levels, baseline PASE status for physical activity assessment, baseline smoking status, baseline use of alcoholic beverages, baseline number of comorbid conditions, and length of follow-up in days. Our data suggest an additive benefit of high testosterone levels and vitamin D (700 IU per day) plus calcium (500 mg per day) supplementation in older men and women.
| Sex-Specific total testosterone quartiles |
Results adjusted for treatment OR [95% CI] |
Results in Placebo group OR [95% CI] |
Results in vitamin D + calcium group OR [95% CI] |
|---|---|---|---|
| Men | |||
| Comparing top to bottom quartile |
0.22 [0.07,0.72] |
0.23 [0.04,1.40] |
0.16 [0.03,0.90] |
| Women | |||
| Comparing top to bottom quartile | 0.34 [0.14,0.83] | 0.41 [0.09,1.86] | 0.15 [0.04,0.57] |
Hormone levels and lean body mass
There was a significant inverse association between baseline total and leg lean body mass, and quartiles of SHBG in both men and women (see adjusted means in Figure 2 A and B). All sex hormones were not appreciably associated with lean body mass.
Figure 2. Adjusted mean baseline leg and total body lean mass by quartiles of baseline SHBG.
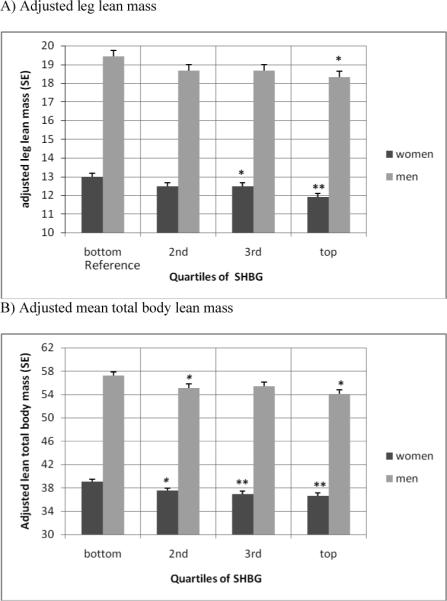
Mean adjusted lean mass by quartiles of SHBG is adjusted for age, physical activity, 25(OH)D levels, number of comorbid conditions, smoking and alcohol consumption. P-values for comparison of quartiles with reference quartile (bottom quartile for SHBG): * < 0.05, ** < 0.001.
Among men, the test for trend was significant for total body (p = 0.005) and leg (0.03) lean mass. Among women, the test for trend was significant among women for total body (p = 0.005) and leg (p = 0.0002) lean mass.
DISCUSSION
We found that fall risk declines with higher physiological testosterone levels among older ambulatory men and women independent of age, SHBG levels, body mass index, physical activity, smoking, alcohol consumption, number of comorbid conditions, vitamin D plus calcium treatment, and length of follow-up. Men and women in the top quartile of sex specific testosterone levels had a 78% respectively 66% lower risk of falling compared to individuals in the lowest quartile. This benefit was augmented if individuals in the top quartile had additional vitamin D plus calcium supplementation. In this subgroup fall risk was reduced by 84% in men and 85% in women.
The additional benefit of vitamin D plus calcium, may be explained by evidence from several randomized controlled trials showing that vitamin D supplementation reduces the risk of falls in older individuals by enhancing muscle strength and balance20,21,23,29,30. From a clinical perspective the possible additive benefit of higher physiologic testosterone levels and vitamin D plus calcium supplementation is of interest as the additional benefit appears to be significant, applies to both sexes, and both components can be altered by treatment. In fact, our earlier findings that did not take testosterone levels into considerations, suggested a significant reduction in falls among women (OR = 0.54; 95% confidence interval [CI], 0.30−0.97), but not in men (OR, 0.93; 95% CI, 0.50−1.72with vitamin D plus calcium supplementation compared to placebo23. Thus, especially among men, the additional correction of testosterone levels to the upper end of the physiologic range may be important.
There was no association between estrogen levels and falls in either sex. Our findings are consistent with two prospective cohort studies, which did not find an association between estrogen levels and incident falls among older men11,12 or women12. Furthermore, one double-blind randomized controlled trial with falls as the outcome did not find a protective effect of hormone replacement therapy in ambulatory older women31. Thus, in contrast to the previously documented benefit of estrogen on bone density in men32 and women33, higher estrogen levels may not protect from falls in either sex.
We found that higher DHEA-S levels may reduce the odds of falling by 61% in women and there appeared to be a similar direction in men. Our study adds to the literature suggesting that higher physiological levels, above 0.31 μg/l, may be beneficial for fall prevention in women. Additional data are needed in both men and women.
There was no association of the directly measured free testosterone on falls in both genders. Physiologically, it is expected that free testosterone reflects the bioavailable part of total testosterone that enters the cells and is most sensitive to outcomes, such as falls. However, it has been suggested that measurement of free testosterone may present with difficulties or may not best represent the bioavailable fraction of testosterone34. Alternatively, we controlled for SHBG when assessing the benefits of testosterone, which may approximates the truely bioavailable fraction of testosterone. Androstenedione levels and SHBG levels were not associated with falling in men or women.
Only SHBG showed a significant inverse association with total and leg lean body mass in men and women. Specifically, based on our adjusted results, men in the top quartile of serum SHBG concentrations had a 6% lower leg lean mass and top quartile women had a 9% lower leg lean mass. Higher SHGB levels have previously been identified as a risk factor for hip fractures in the Study of Osteoporotic Fracture (SOF)35. Our results support these findings indirectly, as low leg lean body mass is a correlate of quadriceps weakness36 and poor structural parameters of bone37, which are risk factors for hip fractures36. However, leg lean mass is a surrogate of muscle force with limitations and direct strength measures are preferable if the target endpoint is falling. Mechanistically, a higher protein intake may decrease SHBG levels resulting in a higher lean body mass. In the Massachusetts Male Aging Study, protein intake showed a negative association with SHBG levels38.
Despite its protective role in regard to falls, there was no association between baseline total testosterone levels and baseline total or leg lean body mass. One explanation may be that the effect was too small to be picked up cross-sectionally or that the physiologic range did not provide large enough contrasts. Previous studies on testosterone replacement among hypogonadal men found a significant increase in lean body mass6 and muscle strength7. Also, three cohort studies documented a significant association between lower testosterone levels and risk of sarcopenia39, reduced physical performance or reduced muscle strength11,12.
In summary, higher physiological testosterone levels may be advantageous for fall prevention in older men and women. Fall prevention may be further improved by providing vitamin D plus calcium supplementation to individuals with higher physiologic testosterone levels. Future intervention studies are needed to test the effect of testosterone supplementation on falls in combination with vitamin D and calcium supplementation. These studies may aim to bring older men and women up to the upper reference range for sex-specific physiologic testosterone levels, as this may be sufficient for fall prevention according to our results. The combined benefit of higher testosterone and vitamin D plus calcium appears to be additive.
Based on our cross-sectional findings, body composition is not associated with sex hormones but SHBG levels. Lean mass was highest among men and women with low SHBG levels. Thus, factors that impact on SHBG status may need further exploration.
Funding/Support
This study was supported by a grant from the Charles H. Farnsworth Trust, Boston, Mass (US Trust Company, trustee), and by grant AG10353 from the National Institutes of Health, Bethesda, Md, and a Swiss National Foundation Professorship Grant.
Footnotes
Disclosures: None
Role of the Sponsors: No sponsors participated in the design and conduct of the study; in the collection, analysis, and interpretation of the data; or in the preparation, review, or approval of the manuscript.
Literature
- 1.Amin S, Zhang Y, Felson DT, Sawin CT, Hannan MT, Wilson PW, Kiel DP. Estradiol, testosterone, and the risk for hip fractures in elderly men from the Framingham Study. Am J Med. 2006;119(5):426–33. doi: 10.1016/j.amjmed.2005.10.048. [DOI] [PubMed] [Google Scholar]
- 2.Fink HA, Ewing SK, Ensrud KE, Barrett-Connor E, Taylor BC, Cauley JA, Orwoll ES. Association of testosterone and estradiol deficiency with osteoporosis and rapid bone loss in older men. J Clin Endocrinol Metab. 2006;91(10):3908–15. doi: 10.1210/jc.2006-0173. Epub 2006 Jul 18. [DOI] [PubMed] [Google Scholar]
- 3.Mellstrom D, Johnell O, Ljunggren O, Eriksson AL, Lorentzon M, Mallmin H, Holmberg A, Redlund-Johnell I, Orwoll E, Ohlsson C. Free testosterone is an independent predictor of BMD and prevalent fractures in elderly men: MrOS Sweden. J Bone Miner Res. 2006;21(4):529–35. doi: 10.1359/jbmr.060110. Epub 2006 Apr 5. [DOI] [PubMed] [Google Scholar]
- 4.Riggs BL, Khosla S, Melton LJ., 3rd Sex steroids and the construction and conservation of the adult skeleton. Endocr Rev. 2002;23(3):279–302. doi: 10.1210/edrv.23.3.0465. [DOI] [PubMed] [Google Scholar]
- 5.Pfeilschifter J, Scheidt-Nave C, Leidig-Bruckner G, Woitge HW, Blum WF, Wuster C, Haack D, Ziegler R. Relationship between circulating insulin-like growth factor components and sex hormones in a population-based sample of 50-to 80-year-old men and women. J Clin Endocrinol Metab. 1996;81(7):2534–40. doi: 10.1210/jcem.81.7.8675573. [DOI] [PubMed] [Google Scholar]
- 6.Bhasin S, Harman SM, Blackman MR, Wittert GA, Chapman IM, Haren MT, Mackintosh S, Coates P, Morley JE, Crawford BA, Liu PY, Kean MT, Bleasel JF, Handelsman DJ. Testosterone supplementation for aging-associated sarcopenia. J Gerontol A Biol Sci Med Sci. 2003;58(11):1002–8. doi: 10.1093/gerona/58.11.m1002. [DOI] [PubMed] [Google Scholar]
- 7.Giannoulis MG, Sonksen PH, Umpleby M, Breen L, Pentecost C, Whyte M, McMillan CV, Bradley C, Martin FC. The Effects of Growth Hormone and/or Testosterone in Healthy Elderly Men: A Randomized Controlled Trial. J Clin Endocrinol Metab. 2005;6:6. doi: 10.1210/jc.2005-0957. [DOI] [PubMed] [Google Scholar]
- 8.Blackman MR, Sorkin JD, Munzer T, Bellantoni MF, Busby-Whitehead J, Stevens TE, Jayme J, O'Connor KG, Christmas C, Tobin JD, Stewart KJ, Cottrell E, St Clair C, Pabst KM, Harman SM. Growth hormone and sex steroid administration in healthy aged women and men: a randomized controlled trial. Jama. 2002;288(18):2282–92. doi: 10.1001/jama.288.18.2282. [DOI] [PubMed] [Google Scholar]
- 9.Wittert GA, Chapman IM, Haren MT, Mackintosh S, Coates P, Morley JE. Oral testosterone supplementation increases muscle and decreases fat mass in healthy elderly males with low-normal gonadal status. J Gerontol A Biol Sci Med Sci. 2003;58(7):618–25. doi: 10.1093/gerona/58.7.m618. [DOI] [PubMed] [Google Scholar]
- 10.Ottenbacher KJ, Ottenbacher ME, Ottenbacher AJ, Acha AA, Ostir GV. Androgen treatment and muscle strength in elderly men: A meta-analysis. J Am Geriatr Soc. 2006;54(11):1666–73. doi: 10.1111/j.1532-5415.2006.00938.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Orwoll E, Lambert LC, Marshall LM, Blank J, Barrett-Connor E, Cauley J, Ensrud K, Cummings SR. Endogenous testosterone levels, physical performance, and fall risk in older men. Arch Intern Med. 2006;166(19):2124–31. doi: 10.1001/archinte.166.19.2124. [DOI] [PubMed] [Google Scholar]
- 12.Schaap LA, Pluijm SM, Smit JH, van Schoor NM, Visser M, Gooren LJ, Lips P. The association of sex hormone levels with poor mobility, low muscle strength and incidence of falls among older men and women. Clin Endocrinol (Oxf) 2005;63(2):152–60. doi: 10.1111/j.1365-2265.2005.02315.x. [DOI] [PubMed] [Google Scholar]
- 13.Orwoll E, Lambert LC, Marshall LM, Phipps K, Blank J, Barrett-Connor E, Cauley J, Ensrud K, Cummings S, Mellstrom D, Johnell O, Ljunggren O, Eriksson AL, Lorentzon M, Mallmin H, Holmberg A, Redlund-Johnell I, Ohlsson C. Testosterone and estradiol among older men Free testosterone is an independent predictor of BMD and prevalent fractures in elderly men: MrOS Sweden. J Clin Endocrinol Metab. 2006;91(4):1336–44. doi: 10.1210/jc.2005-1830. Epub 2005 Dec 20. [DOI] [PubMed] [Google Scholar]
- 14.Taaffe DR, Newman AB, Haggerty CL, Colbert LH, de Rekeneire N, Visser M, Goodpaster BH, Nevitt MC, Tylavsky FA, Harris TB. Estrogen replacement, muscle composition, and physical function: The Health ABC Study. Med Sci Sports Exerc. 2005;37(10):1741–7. doi: 10.1249/01.mss.0000181678.28092.31. [DOI] [PubMed] [Google Scholar]
- 15.Taaffe DR, Sipila S, Cheng S, Puolakka J, Toivanen J, Suominen H. The effect of hormone replacement therapy and/or exercise on skeletal muscle attenuation in postmenopausal women: a yearlong intervention. Clin Physiol Funct Imaging. 2005;25(5):297–304. doi: 10.1111/j.1475-097X.2005.00628.x. [DOI] [PubMed] [Google Scholar]
- 16.Kenny AM, Dawson L, Kleppinger A, Iannuzzi-Sucich M, Judge JO. Prevalence of sarcopenia and predictors of skeletal muscle mass in nonobese women who are long-term users of estrogen-replacement therapy. J Gerontol A Biol Sci Med Sci. 2003;58(5):M436–40. doi: 10.1093/gerona/58.5.m436. [DOI] [PubMed] [Google Scholar]
- 17.Bemben DA, Langdon DB. Relationship between estrogen use and musculoskeletal function in postmenopausal women. Maturitas. 2002;42(2):119–27. doi: 10.1016/s0378-5122(02)00033-6. [DOI] [PubMed] [Google Scholar]
- 18.Valenti G, Denti L, Maggio M, Ceda G, Volpato S, Bandinelli S, Ceresini G, Cappola A, Guralnik JM, Ferrucci L. Effect of DHEAS on skeletal muscle over the life span: the InCHIANTI study. J Gerontol A Biol Sci Med Sci. 2004;59(5):466–72. doi: 10.1093/gerona/59.5.m466. [DOI] [PubMed] [Google Scholar]
- 19.Kostka T, Arsac LM, Patricot MC, Berthouze SE, Lacour JR, Bonnefoy M. Leg extensor power and dehydroepiandrosterone sulfate, insulin-like growth factor-I and testosterone in healthy active elderly people. Eur J Appl Physiol. 2000;82(1−2):83–90. doi: 10.1007/s004210050655. [DOI] [PubMed] [Google Scholar]
- 20.Bischoff HA, Stahelin HB, Dick W, Akos R, Knecht M, Salis C, Nebiker M, Theiler R, Pfeifer M, Begerow B, Lew RA, Conzelmann M. Effects of vitamin D and calcium supplementation on falls: a randomized controlled trial. J Bone Miner Res. 2003;18(2):343–51. doi: 10.1359/jbmr.2003.18.2.343. [DOI] [PubMed] [Google Scholar]
- 21.Bischoff-Ferrari HA, Dawson-Hughes B, Willett CW, Staehelin HB, Bazemore MG, Zee RY, Wong JB. Effect of vitamin D on falls: a meta-analysis. JAMA. 2004;291(16):1999–2006. doi: 10.1001/jama.291.16.1999. [DOI] [PubMed] [Google Scholar]
- 22.Flicker L, MacInnis RJ, Stein MS, Scherer SC, Mead KE, Nowson CA, Thomas J, Lowndes C, Hopper JL, Wark JD. Should all older people in residential care receive vitamin D to prevent falls? Results of a randomized trial. JBMR. 2004;19(Suppl 1):S99. doi: 10.1111/j.1532-5415.2005.00468.x. abstract F459. [DOI] [PubMed] [Google Scholar]
- 23.Bischoff-Ferrari HA, Orav EJ, Dawson-Hughes B. Effect of cholecalciferol plus calcium on falling in ambulatory older men and women: a 3-year randomized controlled trial. Arch Intern Med. 2006;166(4):424–30. doi: 10.1001/archinte.166.4.424. [DOI] [PubMed] [Google Scholar]
- 24.Dawson-Hughes B, Harris SS, Krall EA, Dallal GE. Effect of calcium and vitamin D supplementation on bone density in men and women 65 years of age or older. N Engl J Med. 1997;337(10):670–6. doi: 10.1056/NEJM199709043371003. [DOI] [PubMed] [Google Scholar]
- 25.Buchner DM, Hornbrook MC, Kutner NG, Tinetti ME, Ory MG, Mulrow CD, Schechtman KB, Gerety MB, Fiatarone MA, Wolf SL, et al. Development of the common data base for the FICSIT trials. J Am Geriatr Soc. 1993;41(3):297–308. doi: 10.1111/j.1532-5415.1993.tb06708.x. [DOI] [PubMed] [Google Scholar]
- 26.Washburn RA, Smith KW, Jette AM, Janney CA. The Physical Activity Scale for the Elderly (PASE): development and evaluation. J Clin Epidemiol. 1993;46(2):153–62. doi: 10.1016/0895-4356(93)90053-4. [DOI] [PubMed] [Google Scholar]
- 27.Preece MA, O'Riordan JL, Lawson DE, Kodicek E. A competitive protein-binding assay for 25-hydroxycholecalciferol and 25-hydroxyergocalciferol in serum. Clin Chim Acta. 1974;54(2):235–42. doi: 10.1016/0009-8981(74)90241-1. [DOI] [PubMed] [Google Scholar]
- 28.Hammond GL, Nisker JA, Jones LA, Siiteri PK. Estimation of the percentage of free steroid in undiluted serum by centrifugal ultrafiltration-dialysis. J Biol Chem. 1980;255(11):5023–6. [PubMed] [Google Scholar]
- 29.Pfeifer M, Begerow B, Minne HW, Abrams C, Nachtigall D, Hansen C. Effects of a short-term vitamin D and calcium supplementation on body sway and secondary hyperparathyroidism in elderly women. J Bone Miner Res. 2000;15(6):1113–8. doi: 10.1359/jbmr.2000.15.6.1113. [DOI] [PubMed] [Google Scholar]
- 30.Flicker L, MacInnis RJ, Stein MS, Scherer SC, Mead KE, Nowson CA, Thomas J, Lowndes C, Hopper JL, Wark JD. Should older people in residential care receive vitamin D to prevent falls? Results of a randomized trial. J Am Geriatr Soc. 2005;53(11):1881–8. doi: 10.1111/j.1532-5415.2005.00468.x. [DOI] [PubMed] [Google Scholar]
- 31.Gallagher JC, Fowler SE, Detter JR, Sherman SS. Combination treatment with estrogen and calcitriol in the prevention of age-related bone loss. J Clin Endocrinol Metab. 2001;86(8):3618–28. doi: 10.1210/jcem.86.8.7703. [DOI] [PubMed] [Google Scholar]
- 32.Gennari L, Merlotti D, Martini G, Gonnelli S, Franci B, Campagna S, Lucani B, Dal Canto N, Valenti R, Gennari C, Nuti R. Longitudinal association between sex hormone levels, bone loss, and bone turnover in elderly men. J Clin Endocrinol Metab. 2003;88(11):5327–33. doi: 10.1210/jc.2003-030736. [DOI] [PubMed] [Google Scholar]
- 33.Cauley JA, Robbins J, Chen Z, Cummings SR, Jackson RD, LaCroix AZ, LeBoff M, Lewis CE, McGowan J, Neuner J, Pettinger M, Stefanick ML, Wactawski-Wende J, Watts NB. Effects of estrogen plus progestin on risk of fracture and bone mineral density: the Women's Health Initiative randomized trial. Jama. 2003;290(13):1729–38. doi: 10.1001/jama.290.13.1729. [DOI] [PubMed] [Google Scholar]
- 34.Miller KK, Rosner W, Lee H, Hier J, Sesmilo G, Schoenfeld D, Neubauer G, Klibanski A. Measurement of free testosterone in normal women and women with androgen deficiency: comparison of methods. J Clin Endocrinol Metab. 2004;89(2):525–33. doi: 10.1210/jc.2003-030680. [DOI] [PubMed] [Google Scholar]
- 35.Cummings SR, Browner WS, Bauer D, Stone K, Ensrud K, Jamal S, Ettinger B. Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N Engl J Med. 1998;339(11):733–8. doi: 10.1056/NEJM199809103391104. [DOI] [PubMed] [Google Scholar]
- 36.Cummings SR, Nevitt MC, Browner WS, Stone K, Fox KM, Ensrud KE, Cauley J, Black D, Vogt TM. Risk factors for hip fracture in white women. Study of Osteoporotic Fractures Research Group. N Engl J Med. 1995;332(12):767–73. doi: 10.1056/NEJM199503233321202. [DOI] [PubMed] [Google Scholar]
- 37.Szulc P, Beck TJ, Marchand F, Delmas PD. Low skeletal muscle mass is associated with poor structural parameters of bone and impaired balance in elderly men--the MINOS study. J Bone Miner Res. 2005;20(5):721–9. doi: 10.1359/JBMR.041230. [DOI] [PubMed] [Google Scholar]
- 38.Longcope C, Feldman HA, McKinlay JB, Araujo AB. Diet and sex hormone-binding globulin. J Clin Endocrinol Metab. 2000;85(1):293–6. doi: 10.1210/jcem.85.1.6291. [DOI] [PubMed] [Google Scholar]
- 39.Szulc P, Duboeuf F, Marchand F, Delmas PD. Hormonal and lifestyle determinants of appendicular skeletal muscle mass in men: the MINOS study. Am J Clin Nutr. 2004;80(2):496–503. doi: 10.1093/ajcn/80.2.496. [DOI] [PubMed] [Google Scholar]