Abstract
OBJECTIVE
Obesity is associated with increased metabolic and cardiovascular risk. The ectopic fat hypothesis suggests that subcutaneous fat may be protective, but this theory has yet to be fully explored.
RESEARCH DESIGN AND METHODS
Participants from the Framingham Heart Study (n = 3,001, 48.5% women) were stratified by visceral adipose tissue (VAT) into sex-specific tertiles. Within these tertiles, age-adjusted abdominal subcutaneous adipose tissue (SAT) tertiles were examined in relation to cardiometabolic risk factors.
RESULTS
In the lowest VAT tertile, risk factor prevalence was low, although systolic blood pressure in women and rates of high triglycerides, impaired fasting glucose, hypertension, and the metabolic syndrome in men increased with increasing SAT tertile (all P < 0.04). In contrast, in the top VAT tertile, lower triglycerides were observed in men with increasing SAT (64.4% high triglycerides in SAT tertile 1 vs. 52.7% in SAT tertile 3, P = 0.03). Similar observations were made for women, although results were not statistically significant (50.6% high triglycerides in SAT tertile 1 vs. 41.0% in tertile 3, P = 0.10). Results in the highest VAT tertile were notable for a lack of increase in the prevalence of low HDL in men and women and in rates of impaired fasting glucose in men with increasing subcutaneous fat, despite sizable differences in BMI across SAT tertiles (27.1 to 36.3 kg/m2[women]; 28.1 to 35.7 kg/m2[men]).
CONCLUSIONS
Although adiposity increases the absolute risk of metabolic and cardiovascular disease, abdominal subcutaneous fat is not associated with a linear increase in the prevalence of all risk factors among the obese, most notably, high triglycerides.
Obesity is associated with multiple cardiometabolic risk factors, including insulin resistance (1), diabetes (2), hypertension (3), and dyslipidemia (4). Variations in fat distribution may mediate such risks, with visceral adipose tissue (VAT) associated with more adverse risk factor profiles than abdominal subcutaneous adipose tissue (SAT) (5,6). The ectopic fat hypothesis proposes that obesity represents a failure of adipocyte growth and differentiation, resulting in “acquired lipodystrophy” and fat deposition in liver, skeletal muscle, and pancreatic β-cells (7). Such ectopic fat stores are hypothesized to contribute to the pathogenesis of impaired insulin secretion and insulin resistance and to mediate obesity-related cardiovascular disease (8).
In addition to the detrimental effects of VAT, human and animal studies have suggested a possible protective role for subcutaneous fat. In humans, increased subcutaneous leg fat is associated with decreased risk of disturbed glucose metabolism and dyslipidemia, independent of abdominal fat (9). Thiazolidinedione treatment, which increases total fat mass, mostly in subcutaneous fat stores, improves insulin sensitivity (10). Removal of VAT by omentectomy results in decreased glucose and insulin levels in humans, (11), whereas removal of SAT by liposuction does not always result in improvements in glucose metabolism or lipid levels (12,13). Transplantation of subcutaneous fat into visceral compartments in mice produces decreases in body weight and total fat mass and improved glucose metabolism, suggesting that subcutaneous fat may be intrinsically different from visceral fat in ways that are beneficial (14).
Therefore, the purpose of the present study was to test the hypothesis that abdominal subcutaneous fat is a protective fat depot in terms of cardiometabolic risk factor prevalence. We theorized that among those with similar levels of VAT, increasing SAT might be associated with decreases in cardiometabolic risk factor prevalence despite increasing BMI and total abdominal fat.
RESEARCH DESIGN AND METHODS
The Framingham Heart Study is a prospective cohort study that began in 1948. In 1971, the offspring and spouses of the original cohort were enrolled in the Offspring Study, and in 2002 the children of the original cohort's offspring were enrolled in the Third Generation Study. Between June 2002 and April 2005, 3,529 Offspring and Third Generation participants underwent chest and abdominal computed tomographic (CT) scanning in conjunction with the Multi-Detector Computed Tomography (MDCT) substudy. The present study sample consisted of the 3,001 participants (1,455 women and 1,546 men) who had interpretable CT scans (both chest and abdominal), who were free of cardiovascular disease, and who had complete covariate information.
The institutional review boards of the Boston University Medical Center and Massachusetts General Hospital approved the study protocol. All subjects provided informed written consent.
Abdominal adipose tissue imaging and volumetric measurements
Participants underwent eight-slice MDCT abdominal scanning in a supine position (LightSpeed Ultra, General Electric, Milwaukee, WI). Twenty-five contiguous 5-mm thick slices (120 kVp, 400 mA, gantry rotation time 500 ms, and table feed 3:1) were obtained, covering 125 mm above the level of S1.
VAT and SAT volumes were quantified from CT scans using a dedicated offline workstation (Aquarius 3D Workstation; TeraRecon, San Mateo, CA) using a semiautomatic segmentation technique. A reader manually traced the abdominal muscular wall separating the two layers. An image display window width of −195 to −45 Hounsfield units and a window center of −120 Hounsfield units were used to identify pixels containing fat. VAT was defined as adipose tissue inside the abdominal muscular wall and SAT as adipose tissue outside the abdominal muscular wall. Interclass correlations for inter-reader comparisons were 0.997 for SAT and 0.992 for VAT on a random sample of 100 scans; high intra-reader correlations were similarly obtained.
Risk factor and covariate assessment
Covariates were measured at the seventh Framingham Offspring examination (1998–2001) and the first Third Generation examination (2002–2005). BMI was calculated as weight in kilograms divided by the square of height in meters. Waist circumference was measured at the umbilicus. Serum triglycerides, total and HDL cholesterol, and fasting plasma glucose were measured in fasting morning samples obtained from attendees. Diabetes was defined as fasting plasma glucose ≥126 mg/dl or treatment with a hypoglycemic agent or insulin; impaired fasting glucose was defined as a fasting plasma glucose level of 100–125 mg/dl in the absence of diabetes treatment. Hypertension was defined as systolic blood pressure ≥140 mmHg and diastolic blood pressure as ≥90 mmHg or antihypertensive treatment. High triglycerides were defined as ≥150 mg/dl or lipid treatment. Low HDL was defined as <40 mg/dl for men or <50 mg/dl for women. Current smoking was defined as smoking ≥1 cigarette per day in the past year. Alcohol use was evaluated through a physician-administered questionnaire and categorized as more or less than 14 drinks per week (men) or 7 drinks per week (women). Women were classified as postmenopausal if periods had stopped for ≥1 year. Metabolic syndrome was defined by modified Adult Treatment Panel III criteria (impaired fasting glucose defined as fasting plasma glucose 100–125 mg/dl in the absence of treatment of diabetes, the high triglyceride component was defined as triglycerides≥150 mg/dl or treatment with a lipid-lowering agent, and blood pressure criteria were modified to include treatment for hypertension).
Statistical analysis
Study participants were stratified by VAT into sex-specific tertiles. Within each tertile, subjects were categorized further into sex-specific abdominal SAT tertiles. Age-adjusted means and prevalences of risk factors including BMI, waist circumference, fasting glucose, diabetes, HDL cholesterol, triglycerides, hypertension, and the metabolic syndrome were compared across the SAT tertiles. Because overall and visceral adiposity increases with age, all analyses were age-adjusted. As a secondary analysis, participants were separately stratified into SAT tertiles within each clinical BMI category (normal weight, overweight, and obese), and the analyses were repeated. All analyses were performed using SAS (version 9.1); two-tailed values of P < 0.05 were considered significant.
RESULTS
Study sample characteristics
Overall, 1,455 women (mean age 51.4 years) and 1,546 men (mean age 48.6 years) comprised the study sample. The average BMI was 26.7 kg/m2 for women and 28.3 kg/m2 for men, with 24.1% of women and 27.3% of men classified as obese. Approximately one-quarter of women had high triglycerides, low HDL, hypertension, and the metabolic syndrome, whereas 41.1% of men had high triglycerides, 31.1% had low HDL, 28.5% had hypertension, and 35.2% had the metabolic syndrome. Additional study sample characteristics are listed in Table 1.
Table 1.
Study sample characteristics
| Women | Men | |
|---|---|---|
| n | 1,455 | 1,546 |
| Age (years) | 51.4 ± 9.5 | 48.6 ± 10.1 |
| BMI (kg/m2) | 26.7 ± 5.4 | 28.3 ± 4.4 |
| Waist circumference (cm) | 92.0 ± 14.4 | 100.3 ± 11.5 |
| Systolic blood pressure (mmHg) | 119.5 ± 17.4 | 123.1 ± 14.3 |
| Diastolic blood pressure (mmHg) | 73.5 ± 9.1 | 78.5 ± 8.9 |
| Fasting plasma glucose (mg/dl) | 94.9 ± 16.6 | 101.0 ± 20.0 |
| Triglycerides (mg/dl) | 109.8 ± 65.2 | 139.6 ± 102.9 |
| Impaired fasting glucose* | 251 (18.0) | 558 (38.5) |
| Diabetes | 62 (4.3) | 95 (6.2) |
| High triglycerides† | 354 (24.3) | 635 (41.1) |
| Low HDL‡ | 352 (24.2) | 481 (31.1) |
| Hypertension | 352 (24.2) | 440 (28.5) |
| Metabolic syndrome | 361 (24.8) | 543 (35.2) |
| Current smoking | 178 (12.2) | 203 (13.1) |
| Alcohol (≥14 drinks/week in men/≥7 drinks/week in women) | 222 (15.3) | 247 (16.0) |
| Postmenopausal | 694 (47.7) | — |
| Hormone replacement therapy | 333 (22.9) | — |
| SAT (cm3) | 3,076 ± 1,449 | 2,599 ± 1,185 |
| VAT (cm3) | 1,309 ± 809 | 2,158 ± 967 |
Data are means ± SD orn(%).
*Defined as fasting plasma glucose 100–125 mg/dl in the absence of diabetes treatment.
†Defined as triglycerides ≥150 mg/dl or lipid treatment.
‡Defined as HDL <40 mg/dl (men) or <50 mg/dl (women).
Age-adjusted tertile analysis
All measures of adiposity (BMI, waist circumference, SAT, and VAT) increased significantly for women and men across SAT tertiles within each VAT tertile (Table 2). Among those in the lowest VAT tertile, risk factor prevalence was uniformly low and increased with increasing SAT tertile (Table 3). Among women, the age-adjusted linear trend was significant for systolic blood pressure (109 mmHg in SAT tertile 1 vs. 114 mmHg in SAT tertile 3, P < 0.001). For men, linear trends were significant for several risk factors. Of particular note are the significant increases in prevalence rates of impaired fasting glucose (19.1% in SAT tertile 1 vs. 29.1% in SAT tertile 3, P = 0.03) and high triglycerides (12.1% in SAT tertile 1 vs. 20.4% in SAT tertile 3, P = 0.04).
Table 2.
-Age-adjusted measures of adiposity by sex-specific SAT tertiles within sex-specific VAT tertiles
| Women |
Men |
|||||||
|---|---|---|---|---|---|---|---|---|
| SAT tertile 1 | SAT tertile 2 | SAT tertile 3 | Age-adjusted SAT linear P value | SAT tertile 1 | SAT tertile 2 | SAT tertile 3 | Age-adjusted SAT linear P value | |
| VAT tertile 1 | ||||||||
| n | 161 | 163 | 161 | — | 171 | 172 | 172 | — |
| BMI (kg/m2) | 20.6 ± 0.2 | 22.3 ± 0.2 | 24.6 ± 0.2 | <0.0001 | 22.9 ± 0.2 | 24.8 ± 0.2 | 27.3 ± 0.2 | <0.0001 |
| WC (cm) | 74.2 ± 0.5 | 79.0 ± 0.5 | 84.6 ± 0.5 | <0.0001 | 84.6 ± 0.5 | 90.9 ± 0.5 | 97.5 ± 0.5 | <0.0001 |
| SAT (cm3) | 1,067 ± 32 | 1,780 ± 32 | 2,701 ± 32 | <0.0001 | 1,048 ± 33 | 1,774 ± 33 | 2,770 ± 33 | <0.0001 |
| SAT range (cm3) | 170.8–1,463.2 | 1,463.4–2,084.9 | 2,085.0–5,130.2 | — | 121.5–1,497.3 | 1,501.8–2,069.2 | 2,071.5–5,636.0 | — |
| VAT (cm3)* | 421 ± 9 | 561 ± 9 | 631 ± 9 | <0.0001 | 895 ± 22 | 1,216 ± 22 | 1,352 ± 22 | <0.0001 |
| VAT tertile 2 | ||||||||
| n | 161 | 162 | 162 | — | 172 | 172 | 172 | — |
| BMI (kg/m2) | 23.7 ± 0.2 | 25.5 ± 0.2 | 29.4 ± 0.2 | <0.0001 | 26.2 ± 0.2 | 27.9 ± 0.2 | 30.5 ± 0.2 | <0.0001 |
| WC (cm) | 84.3 ± 0.5 | 90.9 ± 0.5 | 99.3 ± 0.5 | <0.0001 | 94.0 ± 0.5 | 99.3 ± 0.5 | 107.4 ± 0.5 | <0.0001 |
| SAT (cm3) | 2,139 ± 40 | 2,921 ± 40 | 4,300 ± 40 | <0.0001 | 1,756 ± 44 | 2,453 ± 44 | 3,697 ± 44 | <0.0001 |
| SAT range (cm3) | 727.2–2,555.6 | 2,555.8–3,337.2 | 3,348.7–7,460.9 | — | 1,080.2–2,134.2 | 2,138.6–2,817.2 | 2,826.6–7,259.0 | — |
| VAT (cm3)† | 1,061 ± 16 | 1,135 ± 16 | 1,230 ± 16 | <0.0001 | 2,024 ± 17 | 2,068 ± 17 | 2,114 ± 18 | <0.001 |
| VAT tertile 3 | ||||||||
| n | 161 | 162 | 162 | — | 171 | 172 | 172 | — |
| BMI (kg/m2) | 27.1 ± 0.3 | 30.9 ± 0.3 | 36.3 ± 0.3 | <0.0001 | 28.1 ± 0.2 | 30.8 ± 0.2 | 35.7 ± 0.2 | <0.0001 |
| WC (cm) | 95.0 ± 0.8 | 104.6 ± 0.8 | 116.3 ± 0.8 | <0.0001 | 100.8 ± 0.5 | 107.7 ± 0.5 | 119.9 ± 0.5 | <0.0001 |
| SAT (cm3) | 2,910 ± 49 | 4,123 ± 48 | 5,730 ± 48 | <0.0001 | 2,106 ± 46 | 3,076 ± 45 | 4,700 ± 45 | <0.0001 |
| SAT range (cm3) | 1,677.8–3,480.8 | 3,481.5–4,692.0 | 4,713.1–8,876.7 | — | 1,090.5–2,614.1 | 2,614.5–3,608.2 | 3,612.1–8,368.0 | — |
| VAT (cm3)‡ | 1,977 ± 47 | 2,222 ± 46 | 2,535 ± 46 | <0.0001 | 3,027 ± 46 | 3,173 ± 45 | 3,548 ± 46 | <0.0001 |
Data are means ± SEM.
*VAT range: 202.3–800.9 cm3(women), 172.7–1679.2 cm3(men).
†VAT range: 803.2–1,545.1 cm3(women), 1,679.3–2,497.4 cm3(men).
‡VAT range: 1,546.7–5,548.0 cm3(women), 2,497.6–6,212.7 cm3(men). WC, waist circumference.
Table 3.
Age-adjusted risk factors by sex-specific SAT tertile within sex-specific VAT tertiles
| Women |
Men |
|||||||
|---|---|---|---|---|---|---|---|---|
| SAT tertile 1 | SAT tertile 2 | SAT tertile 3 | Age-adjusted SAT linear P value | SAT tertile 1 | SAT tertile 2 | SAT tertile 3 | Age-adjusted SAT linear P value | |
| VAT tertile 1 | ||||||||
| Age | 46.9 ± 0.6 | 47.8 ± 0.6 | 47.2 ± 0.6 | 0.74 | 45.2 ± 0.6 | 45.0 ± 0.6 | 44.3 ± 0.6 | 0.34 |
| SBP | 109 ± 1 | 111 ± 1 | 114 ± 1 | <0.001 | 117 ± 1 | 118 ± 1 | 118 ± 1 | 0.13 |
| Fasting glucose | 87.8 ± 0.8 | 89.3 ± 0.8 | 89.2 ± 0.8 | 0.22 | 94.2 ± 1.3 | 97.7 ± 1.3 | 96.7 ± 1.3 | 0.16 |
| Log triglycerides | 4.2 ± 0.03 | 4.2 ± 0.03 | 4.3 ± 0.03 | 0.13 | 4.4 ± 0.04 | 4.5 ± 0.04 | 4.5 ± 0.04 | <0.001 |
| Impaired fasting glucose* | 3.1 | 4.8 | 3.7 | 0.76 | 19.1 | 21.0 | 29.1 | 0.03 |
| Diabetes | 0.0 | 2.1 | 1.1 | 0.95 | 1.4 | 1.4 | 3.1 | 0.25 |
| High triglycerides† | 2.8 | 3.6 | 7.4 | 0.06 | 12.1 | 19.7 | 20.4 | 0.04 |
| Low HDL‡ | 9.7 | 11.6 | 15.9 | 0.10 | 14.0 | 13.4 | 16.7 | 0.49 |
| Hypertension | 6.3 | 7.7 | 7.9 | 0.55 | 7.2 | 12.8 | 15.4 | 0.02 |
| Metabolic syndrome | 0.0 | 0.4 | 2.5 | 0.93 | 3.5 | 5.2 | 8.8 | 0.04 |
| VAT tertile 2 | ||||||||
| Age | 53.6 ± 0.7 | 50.8 ± 0.7 | 50.0 ± 0.7 | <0.001 | 49.5 ± 0.7 | 49.3 ± 0.7 | 45.8 ± 0.7 | <0.001 |
| SBP | 117 ± 1 | 119 ± 1 | 122 ± 1 | 0.01 | 123 ± 1 | 124 ± 1 | 123 ± 1 | 0.69 |
| Fasting glucose | 94.2 ± 1.0 | 93.3 ± 1.0 | 94.0 ± 1.0 | 0.91 | 97.5 ± 1.1 | 101.5 ± 1.1 | 101.2 ± 1.1 | 0.02 |
| Log triglycerides | 4.5 ± 0.04 | 4.6 ± 0.04 | 4.5 ± 0.04 | 0.96 | 4.9 ± 0.04 | 4.9 ± 0.04 | 4.9 ± 0.04 | 0.29 |
| Impaired fasting glucose* | 15.4 | 13.9 | 18.2 | 0.50 | 31.4 | 37.9 | 47.5 | 0.003 |
| Diabetes | 3.3 | 2.3 | 1.2 | 0.21 | 2.2 | 3.3 | 3.9 | 0.32 |
| High triglycerides† | 20.0 | 22.8 | 21.5 | 0.74 | 55.4 | 44.9 | 47.3 | 0.14 |
| Low HDL‡ | 22.3 | 17.8 | 24.5 | 0.65 | 37.7 | 34.7 | 36.1 | 0.77 |
| Hypertension | 14.9 | 19.3 | 19.7 | 0.27 | 21.2 | 26.4 | 31.3 | 0.04 |
| Metabolic syndrome | 8.3 | 14.1 | 27.2 | <0.0001 | 22.5 | 33.1 | 50.7 | <0.0001 |
| VAT tertile 3 | ||||||||
| Age | 58.5 ± 0.7 | 55.2 ± 0.7 | 52.8 ± 0.7 | <0.0001 | 56.0 ± 0.8 | 51.9 ± 0.8 | 50.7 ± 0.8 | <0.0001 |
| SBP | 127 ± 1 | 127 ± 1 | 130 ± 1 | 0.10 | 127 ± 1 | 128 ± 1 | 131 ± 1 | 0.02 |
| Fasting glucose | 98.1 ± 1.7 | 104.3 ± 1.6 | 104.3 ± 1.7 | 0.01 | 107.9 ± 1.9 | 105.4 ± 1.9 | 107.3 ± 1.9 | 0.82 |
| Log triglycerides | 4.8 ± 0.04 | 4.9 ± 0.04 | 4.9 ± 0.04 | 0.75 | 5.0 ± 0.04 | 4.9 ± 0.04 | 4.9 ± 0.04 | 0.05 |
| Impaired fasting glucose* | 30.0 | 30.3 | 41.7 | 0.03 | 49.6 | 50.4 | 48.6 | 0.85 |
| Diabetes | 4.0 | 8.4 | 7.6 | 0.13 | 10.8 | 9.2 | 14.9 | 0.26 |
| High triglycerides† | 50.6 | 45.2 | 41.0 | 0.10 | 64.4 | 52.5 | 52.7 | 0.03 |
| Low HDL‡ | 36.3 | 37.0 | 40.5 | 0.46 | 48.4 | 39.8 | 38.8 | 0.08 |
| Hypertension | 40.2 | 40.9 | 49.1 | 0.14 | 43.3 | 38.3 | 52.0 | 0.12 |
| Metabolic syndrome | 51.2 | 51.8 | 65.1 | 0.02 | 55.6 | 61.8 | 73.0 | 0.001 |
Data are means ± SEM or %.
*Defined as fasting plasma glucose 100–125 mg/dl in the absence of diabetes treatment.
†Defined as triglycerides ≥150 mg/dl or lipid treatment.
‡Defined as HDL <40 mg/dl (men) or 50 mg/dl (women). SBP, systolic blood pressure.
In the middle (second) VAT tertile, risk factor prevalence was generally higher than that in VAT tertile 1 and again displayed a pattern of increase across SAT tertiles. Among women, linear trends were significant for systolic blood pressure (117 mmHg in SAT tertile 1 vs. 122 mmHg in SAT tertile 3, P = 0.01) and the metabolic syndrome (8.3% in SAT tertile 1 vs. 27.2% in SAT tertile 3, P < 0.0001). Among men, linear trends were also significant and increasing for the metabolic syndrome (22.5% in SAT tertile 1 vs. 50.7% in SAT tertile 3, P < 0.0001), as well as for fasting glucose, impaired fasting glucose, and hypertension.
In the top VAT tertile, absolute risk factor prevalence was high and increased significantly with increasing SAT for several risk factors. For example, linear trends for rates of metabolic syndrome were significant for both women (51.2% in SAT tertile 1 vs. 65.1% in SAT tertile 3, P = 0.02) and men (55.6% in SAT tertile 1 vs. 73.0% in SAT tertile 3, P = 0.001). However, the pattern of increase in risk factor prevalence across SAT tertiles present among the bottom two VAT tertiles was notably absent among both men and women in the highest VAT tertile for low HDL and among men for fasting glucose and impaired fasting glucose despite large increases in BMI across SAT tertiles (27.1 vs. 36.3 kg/m2 for women and 28.1 vs. 35.7 kg/m2 for men). In addition, prevalence rates of high triglycerides in men decreased significantly with increasing SAT (64.4% in SAT tertile 1 vs. 52.7% in SAT tertile 3, P = 0.03). A similar trend was seen in women, although it was not statistically significant (50.6% in SAT tertile 1 vs. 41.0% in SAT tertile 3, P = 0.10).
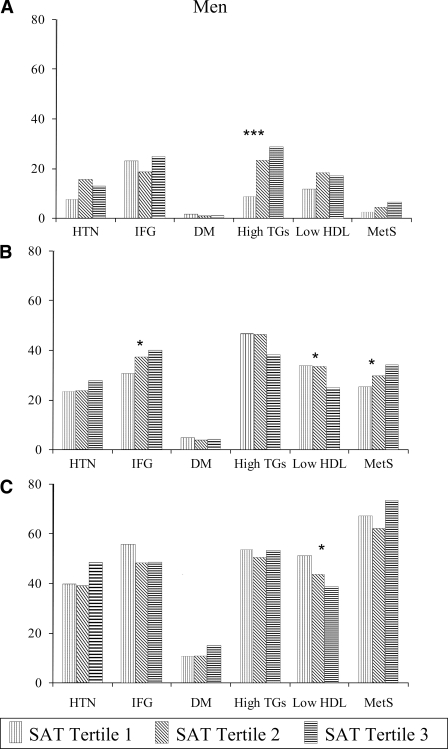
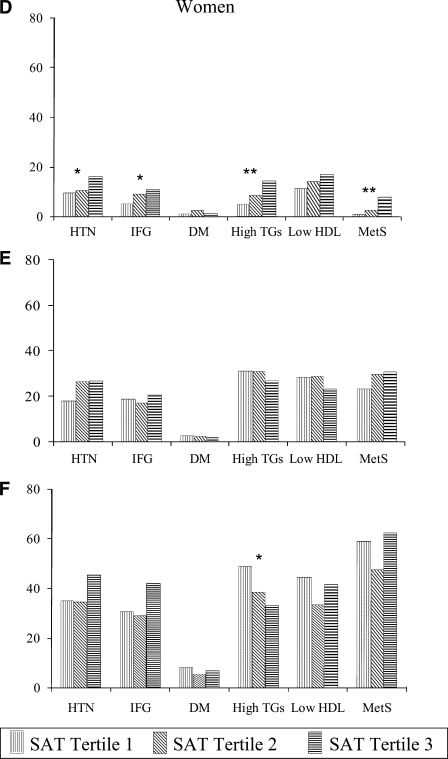
When the study sample was stratified into BMI categories (normal weight, overweight, and obese), similar patterns were observed (Fig. 1). The prevalence of all risk factors markedly increased through the BMI categories. Among those of normal weight, rates of high triglycerides increased significantly with SAT in both women (5.2% in SAT tertile 1 vs. 14.4% in SAT tertile 3, P = 0.002) and men (8.4% in SAT tertile 1 vs. 28.6% in SAT tertile 3, P < 0.001). Among the overweight and obese, however, the prevalence of high triglycerides did not increase with SAT and decreased significantly among obese women (48.8% in SAT tertile 1 vs. 33.0% in SAT tertile 3, P = 0.02).
Figure 1.
Age-adjusted levels of risk factors by tertile of SAT in normal-weight (A and D), overweight (B and E), and obese (C and F) subjects. *P < 0.05, **P < 0.01, and ***P < 0.001 for linear trend across tertiles. DM, diabetes; HTN, hypertension; IFG, impaired fasting glucose; MetS, metabolic syndrome; TGs, triglycerides.
CONCLUSIONS
Principal findings
Cardiometabolic risk increases with increasing visceral adiposity and BMI. Among those in the bottom two VAT tertiles, increasing subcutaneous fat is generally associated with increases in risk factor prevalence. Among those in the highest VAT tertile, however, increasing SAT is not associated with uniform increases in risk factors prevalence, despite significant increases in BMI, waist circumference SAT, and VAT. Most striking, linear trends for triglycerides actually decrease with increasing SAT in the top VAT tertile, suggesting that subcutaneous fat may be associated with beneficial effects on triglyceride levels in those with the most visceral fat.
In the context of the current literature
Although total adiposity is strongly associated with metabolic and cardiovascular risk, it is becoming increasingly clear that different fat compartments contribute differentially to these risks. Abdominal visceral fat is a stronger correlate of cardiovascular disease risk than BMI, waist circumference, or abdominal subcutaneous fat (5,6). Central obesity, in which fat mass is predominantly intra-abdominal, is more strongly associated with insulin resistance, dyslipidemia, and atherosclerosis than is peripheral obesity, in which fat is predominantly gluteofemoral (1,15). Weight loss through diet and exercise, which results in reductions in visceral fat (16), is associated with improvements in insulin sensitivity (17), blood pressure (18), serum lipids (19), and inflammatory markers (20). Similarly, loss of VAT by omentectomy leads to decreases in glucose and insulin levels (11), whereas loss of SAT by liposuction does not always produce the same beneficial effects (12,13), suggesting that VAT is more likely to be responsible for the metabolic abnormalities associated with obesity.
A few studies have suggested a possible beneficial role for SAT, observing that increased hip and thigh fat mass is associated with lower glucose and lipid levels, independent of abdominal fat (9). Patients with lipodystrophic loss of SAT have an increased risk for insulin resistance, diabetes, and dyslipidemia (21). In mice, transplantation of subcutaneous fat into visceral compartments leads to decreases in body weight and total fat mass and improved glucose metabolism, whereas transplantation of visceral fat into either subcutaneous or visceral compartments results in no such improvements. This suggests that subcutaneous fat may differ from visceral fat in ways that are metabolically beneficial (14).
Our results confirm that cardiometabolic risk increases significantly with increases in VAT. The prevalence of many risk factors, including hypertension and the metabolic syndrome, also increases with increasing SAT for all VAT and BMI tertiles. Among those in the lower two-thirds of the VAT distribution, more SAT is also associated with increased risk of most other risk factors examined, suggesting that SAT is not protective in these individuals. Among those with the most VAT, however, increased SAT is associated with lower triglycerides, suggesting that SAT may be associated with beneficial effects on triglyceride levels in the obese.
Mechanisms
The ectopic fat hypothesis suggests that a hallmark of obesity is fat deposition in liver, skeletal muscle, and pancreatic β-cells resulting from insufficient adipocyte growth and differentiation in the setting of nutritional excess (22). Such ectopic fat stores are theorized to affect tissue and organ function by physical compression, the secretion of various locally acting substances, and cell dysfunction or cell death of nonadipose cells, a phenomenon known as lipotoxicity (7). In line with this theory, SAT represents a proper expansion of nonpathogenic adipocytes and therefore may be considered a protective fat depot (22). Improvements in insulin sensitivity with thiazolidinedione treatment, which increases subcutaneous fat stores, are suggestive of a protective effect of SAT (10). Our results are consistent with a potential protective role for SAT in the case of triglycerides among the obese.
Another possible explanation for obesity-related cardiometabolic disease is the portal vein hypothesis, which proposes that increased visceral fat leads to higher free fatty acid concentrations in the portal vein, increased systemic fatty acid flux, and increased hepatic lipase activity, which removes lipids from LDL and HDL, and may lead to dyslipidemia (23). Although our results do not contradict the detrimental effects of VAT proposed by the portal vein hypothesis (the prevalence of all risk factors increased with VAT tertile in our sample), they do support the notion that such a theory is, at best, incomplete because it does not explain either the detrimental or the beneficial effects of SAT, which does not drain through the portal vein, on cardiometabolic risk.
VAT and SAT differ not only in anatomic location but also in cytokine secretion profile. SAT releases 2–3 times more leptin than VAT (24), whereas VAT secretes more adiponectin, interleukin-6, interleukin-8, plasminogen activator inhibitor 1, and angiotensin than does SAT (23). Although the relationships between VAT and SAT secretion profiles and cardiometabolic pathogenesis are, at present, unclear, it may be that paracrine and perhaps endocrine factors contribute to the differential effects of VAT and SAT.
It is important to note that our results suggest a possible protective effect of SAT only among those in the highest tertile of VAT. Indeed, in the lower VAT tertiles, increasing SAT is associated only with increases in risk factor prevalence. One explanation for this difference is that there is a relatively larger increase in VAT in the lower tertiles and that SAT is simply a marker for increased VAT in these groups. SAT by itself may not be deleterious or beneficial in the absence of the positive energy balance of obesity. Accordingly, SAT may only be protective in obese individuals because these individuals are already in positive energy balance and SAT provides a nonpathological energy storage depot. It is also important to note that the lack of increase in the prevalence of risk factors such as low LDL and impaired fasting glucose with increasing SAT among the obese may be due to a threshold effect, whereby risk factor prevalence is already so high that increasing SAT is not associated with further increases in prevalence.
Strengths and limitations
Compared with previous work on subcutaneous fat and cardiometabolic risk, the strengths of the present study include a large sample size with a wide range of BMI and age, thereby reducing the likelihood of ascertainment bias in the results. In addition, the study benefits from the quantification of fat volumes from CT scans rather than a single-slice measurement of fat area. A major limitation of the study is its lack of data on subcutaneous fat stores aside from the abdomen; therefore, we cannot generalize our findings to gluteofemoral fat. Further, we are unable to separate deep versus superficial SAT in the abdomen. It may be that superficial SAT is less pathogenic than deep SAT (25). In addition, analysis by VAT and SAT tertiles increases the number of statistical tests performed, increasing the likelihood of false-positive results at the P < 0.05 significance level. Other limitations include the cross-sectional study design, which precludes inferences of causality, and the lack of ethnic diversity, which may limit generalizability to nonwhite groups.
In summary, whereas abdominal adiposity is associated with a higher absolute risk of metabolic and cardiovascular disease, subcutaneous abdominal fat is not associated with a linear increase in the prevalence of all risk factors among the obese. Indeed, in the case of high triglycerides, SAT may actually be a protective fat depot in obese individuals.
Acknowledgments
This work was supported by the National Heart, Lung, and Blood Institute (NHLBI) Framingham Heart Study (Grant N01-HC-25195). S.A.P. is supported by the PASTEUR Program and the Harvard Medical School Office of Enrichment Programs. R.S.V. is supported in part by Grant 2K24HL04334 (NHLBI, National Institutes of Health).
No potential conflicts of interest relevant to this article were reported.
Footnotes
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
References
- 1.Despres JP: The insulin resistance-dyslipidemic syndrome of visceral obesity: effect on patients' risk. Obes Res 1998; 6 ( Suppl. 1): 8S– 17S [DOI] [PubMed] [Google Scholar]
- 2.Mokdad AH, Ford ES, Bowman BA, Dietz WH, Vinicor F, Bales VS, Marks JS: Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. JAMA 2003; 289: 76– 79 [DOI] [PubMed] [Google Scholar]
- 3.Harris MM, Stevens J, Thomas N, Schreiner P, Folsom AR: Associations of fat distribution and obesity with hypertension in a bi-ethnic population: the ARIC study. Atherosclerosis Risk in Communities Study. Obes Res 2000; 8: 516– 524 [DOI] [PubMed] [Google Scholar]
- 4.Lemieux I, Pascot A, Couillard C, Lamarche B, Tchernof A, Almeras N, Bergeron J, Gaudet D, Tremblay G, Prud'homme D, Nadeau A, Despres JP: Hypertriglyceridemic waist: A marker of the atherogenic metabolic triad (hyperinsulinemia; hyperapolipoprotein B; small, dense LDL) in men? Circulation 2000; 102: 179– 184 [DOI] [PubMed] [Google Scholar]
- 5.Despres JP, Lemieux S, Lamarche B, Prud'homme D, Moorjani S, Brun LD, Gagne C, Lupien PJ: The insulin resistance-dyslipidemic syndrome: contribution of visceral obesity and therapeutic implications. Int J Obes Relat Metab Disord 1995; 19 ( Suppl. 1): S76– S86 [PubMed] [Google Scholar]
- 6.Fox CS, Massaro JM, Hoffmann U, Pou KM, Maurovich-Horvat P, Liu CY, Vasan RS, Murabito JM, Meigs JB, Cupples LA, D'Agostino RB, Sr, O'Donnell CJ: Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation 2007; 116: 39– 48 [DOI] [PubMed] [Google Scholar]
- 7.Unger RH: The physiology of cellular liporegulation. Annu Rev Physiol 2003; 65: 333– 347 [DOI] [PubMed] [Google Scholar]
- 8.Montani JP, Carroll JF, Dwyer TM, Antic V, Yang Z, Dulloo AG: Ectopic fat storage in heart, blood vessels and kidneys in the pathogenesis of cardiovascular diseases. Int J Obes Relat Metab Disord 2004; 28 ( Suppl. 4): S58– S65 [DOI] [PubMed] [Google Scholar]
- 9.Snijder MB, Visser M, Dekker JM, Goodpaster BH, Harris TB, Kritchevsky SB, De RN, Kanaya AM, Newman AB, Tylavsky FA, Seidell JC: Low subcutaneous thigh fat is a risk factor for unfavourable glucose and lipid levels, independently of high abdominal fat. The Health ABC Study. Diabetologia 2005; 48: 301– 308 [DOI] [PubMed] [Google Scholar]
- 10.Miyazaki Y, Mahankali A, Matsuda M, Mahankali S, Hardies J, Cusi K, Mandarino LJ, DeFronzo RA: Effect of pioglitazone on abdominal fat distribution and insulin sensitivity in type 2 diabetic patients. J Clin Endocrinol Metab 2002; 87: 2784– 2791 [DOI] [PubMed] [Google Scholar]
- 11.Thorne A, Lonnqvist F, Apelman J, Hellers G, Arner P: A pilot study of long-term effects of a novel obesity treatment: omentectomy in connection with adjustable gastric banding. Int J Obes Relat Metab Disord 2002; 26: 193– 199 [DOI] [PubMed] [Google Scholar]
- 12.Klein S, Fontana L, Young VL, Coggan AR, Kilo C, Patterson BW, Mohammed BS: Absence of an effect of liposuction on insulin action and risk factors for coronary heart disease. N Engl J Med 2004; 350: 2549– 2557 [DOI] [PubMed] [Google Scholar]
- 13.Giugliano G, Nicoletti G, Grella E, Giugliano F, Esposito K, Scuderi N, D'Andrea F: Effect of liposuction on insulin resistance and vascular inflammatory markers in obese women. Br J Plast Surg 2004; 57: 190– 194 [DOI] [PubMed] [Google Scholar]
- 14.Tran TT, Yamamoto Y, Gesta S, Kahn CR: Beneficial effects of subcutaneous fat transplantation on metabolism. Cell Metab 2008; 7: 410– 420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Scherzer R, Shen W, Bacchetti P, Kotler D, Lewis CE, Shlipak MG, Heymsfield SB, Grunfeld C: Simple anthropometric measures correlate with metabolic risk indicators as strongly as magnetic resonance imaging-measured adipose tissue depots in both HIV-infected and control subjects. Am J Clin Nutr 2008; 87: 1809– 1817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Langendonk JG, Kok P, Frolich M, Pijl H, Meinders AE: Decrease in visceral fat following diet-induced weight loss in upper body compared to lower body obese premenopausal women. Eur J Intern Med 2006; 17: 465– 469 [DOI] [PubMed] [Google Scholar]
- 17.Uusitupa M, Lindi V, Louheranta A, Salopuro T, Lindstrom J, Tuomilehto J: Long-term improvement in insulin sensitivity by changing lifestyles of people with impaired glucose tolerance: 4-year results from the Finnish Diabetes Prevention Study. Diabetes 2003; 52: 2532– 2538 [DOI] [PubMed] [Google Scholar]
- 18.Stevens VJ, Obarzanek E, Cook NR, Lee IM, Appel LJ, Smith WD, Milas NC, Mattfeldt-Beman M, Belden L, Bragg C, Millstone M, Raczynski J, Brewer A, Singh B, Cohen J: Long-term weight loss and changes in blood pressure: results of the Trials of Hypertension Prevention, phase II. Ann Intern Med 2001; 134: 1– 11 [DOI] [PubMed] [Google Scholar]
- 19.Dattilo AM, Kris-Etherton PM: Effects of weight reduction on blood lipids and lipoproteins: a meta-analysis. Am J Clin Nutr 1992; 56: 320– 328 [DOI] [PubMed] [Google Scholar]
- 20.Ziccardi P, Nappo F, Giugliano G, Esposito K, Marfella R, Cioffi M, D'Andrea F, Molinari AM, Giugliano D: Reduction of inflammatory cytokine concentrations and improvement of endothelial functions in obese women after weight loss over one year. Circulation 2002; 105: 804– 809 [DOI] [PubMed] [Google Scholar]
- 21.Garg A: Acquired and inherited lipodystrophies. N Engl J Med 2004; 350: 1220– 1234 [DOI] [PubMed] [Google Scholar]
- 22.Heilbronn L, Smith SR, Ravussin E: Failure of fat cell proliferation, mitochondrial function and fat oxidation results in ectopic fat storage, insulin resistance and type II diabetes mellitus. Int J Obes Relat Metab Disord 2004; 28 ( Suppl. 4): S12– S21 [DOI] [PubMed] [Google Scholar]
- 23.Yang X, Smith U: Adipose tissue distribution and risk of metabolic disease: does thiazolidinedione-induced adipose tissue redistribution provide a clue to the answer? Diabetologia 2007; 50: 1127– 1139 [DOI] [PubMed] [Google Scholar]
- 24.Tritos NA, Mantzoros CS: Leptin: its role in obesity and beyond. Diabetologia 1997; 40: 1371– 1379 [DOI] [PubMed] [Google Scholar]
- 25.Kelley DE, Thaete FL, Troost F, Huwe T, Goodpaster BH: Subdivisions of subcutaneous abdominal adipose tissue and insulin resistance. Am J Physiol Endocrinol Metab 2000; 278: E941– E948 [DOI] [PubMed] [Google Scholar]