Abstract
Age, sex steroids, and abdominal-visceral fat (AVF) jointly affect pulsatile growth hormone (GH) secretion. Pulsatile GH secretion in turn is controlled by GH-releasing hormone (GHRH), GH-releasing peptide (GHRP), and somatostatin. Marked stimulation of pulsatile GH secretion is achieved via GHRH-GHRP synergy. Nonetheless, how key modulators of GH secretion, such as age, sex steroids, and body mass index, modify GHRH-GHRP synergy is not known. The present strategy was to 1) infuse GHRH and GHRP-2 simultaneously to evoke synergy and 2) downregulate the gonadal axis with leuprolide and then restore placebo (Pl) or testosterone (T) to clamp the sex steroid milieu. Forty-seven men [18–74 yr of age, T = 7–1,950 ng/dl, estradiol (E2) = 5–79 pg/ml, insulin-like growth factor (IGF)-I = 115–817 μg/l, AVF = 11–349 cm2] were studied. GHRH-GHRP synergy correlated negatively with age and AVF (both P < 0.001) and positively with IGF-I (P < 0.001) and IGF-binding protein (IGFBP)-3 (P = 0.031). Unstimulated basal (nonpulsatile) GH secretion correlated positively with T (P = 0.015) and E2 (P = 0.004) concentrations. Fasting pulsatile GH secretion varied negatively with age (P = 0.017) and positively with IGF-I (P = 0.002) and IGFBP-3 (P = 0.001). By stepwise forward-selection multivariate analyses, AVF, IGF-I, and IGFBP-3 together explained 60% of the variability in GHRH-GHRP synergy (P < 0.001), E2 accounted for 17% of the variability in basal GH secretion (P = 0.007), and IGF-I explained 20% of the variability in fasting pulsatile GH secretion (P = 0.002). In conclusion, a paradigm examining GHRH-GHRP synergy under a sex steroid clamp reveals highly selective control of basal, pulsatile, and synergistic peptide-driven GH secretion by AVF, E2, and IGF-I in healthy men.
Keywords: androgen, fat, human, male, insulin-like growth factor I, pulsatility, secretion, age
pulsatile secretion accounts for >85% of total daily growth hormone (GH) production (20). Burst-like GH release conveys significant physiological information to target tissues, including insulin-like growth factor I (IGF-I) production (22, 40, 41). Conversely, attenuation of GH pulse-dependent STAT5b signaling lowers IGF-I concentrations and causes growth failure (11, 20, 40). From a mechanistic perspective, a triad of pivotal peptides controls the size of GH secretory bursts: two amplifying signals, GH-releasing hormone (GHRH) and GH-releasing peptide (GHRP), and an inhibitory signal, somatostatin (SS) (4, 6, 16, 29, 34). Factors such as sex steroids, age, body composition, IGF-I, and GH feedback, thyroxine, glucocorticoids, free fatty acids, cytokines, neurotransmitters, exercise, and glycemia can modulate pulsatile GH secretion putatively by altering the release and/or actions of GHRH, ghrelin/GHRP, and SS (20, 31, 40). How such factors influence GHRH-GHRP synergy is not known.
The majority of investigations in humans and animals have evaluated the individual actions of GHRH, GHRP, or SS on GH secretion (3, 21). Some analyses have combined l-arginine with GHRH or GHRP, since l-arginine can antagonize GH feedback-induced hypothalamic SS outflow (2, 10, 19, 23, 33). A few studies have used simultaneous GHRH and GHRP infusions to evaluate GH reserve in potentially hypopituitary patients (4, 24, 36, 38). Transgenic, biomathematical, pharmacological, electrophysiological, and clinical models establish that GHRH and ghrelin/GHRP stimulate GH secretion synergistically (13, 16, 28, 34, 40). However, very little is known about the physiological factors that modulate GHRH-GHRP synergy in healthy individuals (36, 38). The present study evaluates modulation of two-peptide synergy by age, sex steroids, body composition, IGF-I, and IGF-binding protein (IGFBP)-3 concentrations during experimental sex steroid depletion and repletion in healthy men.
METHODS
Subjects.
Volunteers provided written informed consent approved by the Mayo Institutional Review Board and reviewed by the US Food and Drug Administration under investigator-initiated new drug numbers for GHRP-2 and GHRH. Exclusion criteria were exposure to psychotropic or neuroactive drugs within five biological half-lives; body mass index (BMI) <18 and >32.5 kg/m2; anemia (hemoglobin <12.8 g/dl); recent or active drug or alcohol abuse, psychosis, depression, mania, or anxiety state; acute or chronic organ system disease; use of testosterone (T), other anabolic steroids, or glucocorticoids; endocrinopathy, other than primary thyroid failure treated with l-thyroxine replacement; night-shift work or recent transmeridian travel (exceeding 3 time zones within 7 days of study); acute weight change (loss or gain of >2 kg in 6 wk); allergy to administered peptides; and unwillingness to provide written informed consent. Each subject had an unremarkable medical history and physical examination and normal screening laboratory tests of hepatic, renal, endocrine, metabolic, and hematological function. The men reported normal sexual development and function.
Visceral fat mass.
Intra-abdominal-visceral fat (AVF) mass was estimated by single-slice abdominal CT scan at L3–L4, exactly as reported previously (35).
Protocol.
The study design was parallel cohort, double blind, and prospectively randomized. Twenty-four healthy young men and 23 healthy older men each received two consecutive intramuscular injections of depot leuprolide acetate (3.75 mg, 3 wk apart) to deplete systemic T and estradiol (E2) concentrations. Beginning on the day of the second leuprolide injection, volunteers were given saline (n = 13 young, n = 12 older) or a pharmacological dose of 200 mg of T enanthate (n = 11 young, n = 11 older) intramuscularly weekly in three doses (designated days 0, 7, and 14) in prospective, randomized double-blind order. The dual-secretagogue infusion was scheduled during days 12–16. Each participant was studied in the Clinical Translational Research Unit (CRU) after a standardized meal and an overnight fast.
Subjects were admitted to the CRU before 1700 and stayed overnight. To limit nutritional confounds, a constant meal (vegetarian or nonvegetarian; 8 kcal/kg, distributed as 50% carbohydrate, 20% protein, and 30% fat) was given in the CRU at 1800 on the night before the study. Volunteers then fasted and abstained from alcohol and caffeine overnight until the end of the infusion on the next day.
In the CRU, catheters were placed in contralateral forearm veins at 0700 to allow simultaneous infusion of secretagogues and blood sampling every 10 min for a total of 6 h beginning at 0800. Sampling encompassed a 3-h baseline and a 3-h stimulation interval. The latter comprised combined continuous intravenous infusions of 1 μg·kg−1·h−1 GHRH (GEREF, Serono, Norwalk, MA) and 1 μg·kg−1·h−1 GHRP-2 (Takeda Pharmaceuticals, Deerfield, IL), which are maximally stimulatory in adults (3, 38).
Blood was also withdrawn at 0800 for later assay of serum E2, T, luteinizing hormone (LH), follicle-stimulating hormone (FSH), IGF-I, IGFBP-1, IGFBP-3, albumin, and sex hormone-binding globulin (SHBG) concentrations. Lunch was provided after sampling before discharge from the CRU.
Hormone assays.
Serum GH concentrations were determined in duplicate by automated ultrasensitive two-site immunoenzymatic chemiluminescence assay performed on the DxI automated system (Beckman Instruments, Chaska, MN). Interassay coefficients of variation (CVs) were 6.1, 4.3, 5.0, and 4.8% at 0.46, 3.0, 7.2, and 13.6 μg/l, respectively. Intra-assay CVs were 4.7, 3.5, and 3.2% at 0.37, 2.5, and 14.8 μg/l, respectively. The lowest detectable GH concentration at 95% confidence is 0.008 μg/l determined by processing a 6-point calibration curve, 5 quality controls, and 10 replicates of zero calibrator in multiple assays.
E2 and T were measured by tandem liquid chromatography-ion spray mass spectrometry. For E2, intra-assay CVs were 18, 3.8, and 7.2% at 3.6, 40, and 297 pg/ml (multiply by 3.68 for pmol/l). Interassay CVs were 8.1, 4.7, and 4.9% at 16, 31, and 119 pg/ml, respectively. For T, the analytic range is 7–2,000 ng/dl (multiply by 0.0347 for nmol/l) for a 0.1-ml volume. Intra-assay CVs were 3.3, 2.8, 2.2, and 2.0% at T concentrations of 16, 64, 184, and 927 ng/dl, respectively; corresponding interassay CVs were 5.1, 3.8, 3.7, and 2.8%.
IGFBP-1, IGFBP-3, and total IGF-I concentrations were quantified by immunoradiometric assays (Diagnostic Systems Laboratories, Webster, TX). Intra-assay CVs for IGF-I were 3.4, 3, and 1.5% at 9.4, 55, and 264 μg/l, respectively, and interassay CVs were 9 and 6.2% at 64 and 157 μg/l.
Statistical analysis.
One-way ANOVA was used to compare baseline (unstimulated) hormone concentrations in the four groups. Two-way ANOVA (2 × 2 factorial design) was used to examine the individual and interactive effects of normal vs. low T or E2 (2 independent factors) and age (young vs. older) on the summed mass of GH secreted in pulses over the 3 h after secretagogue infusion. Post hoc contrasts were made via Tukey's honestly significantly difference test (15). Linear regression analysis was applied to examine the relationship between GH secretion and age, AVF, and IGF-I, IGFBP-1, and IGFBP-3 concentrations.
Values are means ± SE. Experiment-wise P < 0.05 was construed as statistically significant.
Statistical power.
Data from 18 studies in hypogonadal or normal men (n = 149 subjects total) indicate that parenteral T supplementation increases mean GH concentrations by a weighted-mean effect size [standard deviations (SDs)] of 1.8 (7, 8, 12, 17, 18, 25, 30, 32, 37, 39). In women, the effect size of age on GHRH/GHRP-2 stimulation was 1.2 SDs (38). Power analysis assumed that T or E2 depletion and age in men mediate effects of similar magnitudes. If comparisons are made by two-way ANOVA at P < 0.05, then analysis of data from 45 subjects would achieve >90% power to detect the smaller effect size.
Deconvolution analysis.
GH concentration-time series were analyzed using a recently developed automated deconvolution method, which was verified mathematically by direct statistical proof and validated empirically by hypothalamopituitary sampling and simulated pulsatile time series (9). First, the Matlab-based algorithm detrends the data and normalizes concentrations to the unit interval [0,1] (27). Second, the program creates multiple sets of potential pulse times via an incremental smoothing process (a nonlinear adaptation of the heat-diffusion equation). Third, a maximum-likelihood estimation method calculates all secretion and elimination parameters for each of the multiple candidate pulse-time sets. Deconvolution parameters comprise basal secretion (β0), two half-lives (α1, α2), secretory-burst mass (η0, η1), random effects on burst mass (σA), procedural/measurement error (σε), and a three-parameter flexible gamma secretory-burst waveform (β1, β2, β3). The fast half-life of GH was represented as 3.5 min, constituting 37% of the decay amplitude, and the slow half-life as 20.8 min (14). The Akaike information criterion (1) is used to select the optimal pulse-time set from the multiple candidate sets. Interpulse intervals are then described by a two-parameter Weibull process (a more general form of a Poisson process), which defines the frequency (number of bursts per sampling period, λ of a Weibull distribution) and the regularity of interpulse intervals (unitless γ of Weibull). Other parameters are basal and pulsatile secretion rates (concentration units/session), mass secreted per burst (concentration units), and waveform mode (time delay to maximal secretion after burst onset).
RESULTS
Mean ages of the young and older cohorts were 24 ± 0.72 yr (n = 24) and 60 ± 1.6 yr (n = 23), respectively. The age range overall was 18–74 yr. BMI and AVF were higher in older subjects (Table 1). The overall range of BMI was 20–34 kg/m2, and that of AVF was 11–349 cm2. Comparison of baseline (pre-leuprolide) endocrine data in the young and older cohorts disclosed similar concentrations of total T, LH, thyroid-stimulating hormone, and IGFBP-1. Older men had lower IGF-I, IGFBP-3, albumin, prolactin, bioavailable T, and free T and higher SHBG and FSH concentrations than young men.
Table 1.
Baseline hormone data
|
Preintervention Measurements |
P | ||||
|---|---|---|---|---|---|
|
Young |
Older
|
||||
| Pl (n =13) | T (n =11) | Pl (n =12) | T (n =11) | ||
| Age, yr | 23±1.1* | 24±0.99* | 57±1.7† | 64±2.4† | < 0.001 |
| BMI, kg/m2 | 25±0.82 | 24±0.86 | 27±1.0 | 26±0.84 | 0.087 |
| IGFBP-1, μg/l | 14±2.7 | 16±3.2 | 22±3.8 | 15±2.2 | 0.242 |
| IGFBP-3, μg/l | 4,732±222*† | 4,876±283* | 3,994±156†‡ | 3,710±175‡ | < 0.001 |
| IGF-I, μg/l | 434±35* | 420±27* | 242±24† | 264±37† | < 0.001 |
| SHBG, nmol/l | 23±3.3* | 27±3.5*† | 36±4† | 40±4.4† | 0.002 |
| Albumin, g/dl | 4.7±0.053* | 4.7±0.089*† | 4.3±0.11†‡ | 4.2±0.065‡ | < 0.001 |
| LH, IU/l | 3.9±0.45 | 3.2±0.39 | 3.3±0.51 | 3.5±0.75 | 0.657 |
| FSH, IU/l | 3.5±0.37* | 3.3±0.63* | 5.0±0.80*† | 7.0±1.0† | 0.003 |
| Prolactin, μg/l | 9.0±0.92*† | 12±2.1* | 7.0±0.56† | 7.9±0.69*† | 0.040 |
| E2, pg/ml | 31±2.2 | 25±1.5 | 25±2.8 | 25±3.3 | 0.149 |
| T, ng/dl | |||||
| Total | 595±45 | 548±44 | 533±70 | 503±51 | 0.258 |
| Bioavailable | 263±29* | 194±17* | 130±14† | 107±11† | < 0.001 |
| Free | 20±2.2* | 15±1.2*† | 11±1.1†‡ | 9.0±0.86‡ | < 0.001 |
| TSH, mIU/l | 2.5±0.40 | 1.6±0.18 | 2.6±0.42 | 2.8±0.52 | 0.063 |
Values are means ± SE. BMI, body mass index; IGFBP, insulin-like growth factor (IGF)-binding protein; SHBG, sex hormone-binding globulin; LH, luteinizing hormone; FSH, follicle-stimulating hormone; E2, estradiol (multiply by 3.68 to convert to pmol/l); T, testosterone (multiply by 0.0347 to convert to nmol/l); TSH, thyroid-stimulating hormone. P values were determined by ANOVA. Means in any given row with unshared (unique) symbols ( *, †, ‡) differ significantly by Tukey's post hoc test: * differs from †, but neither * nor † differs from *‡.
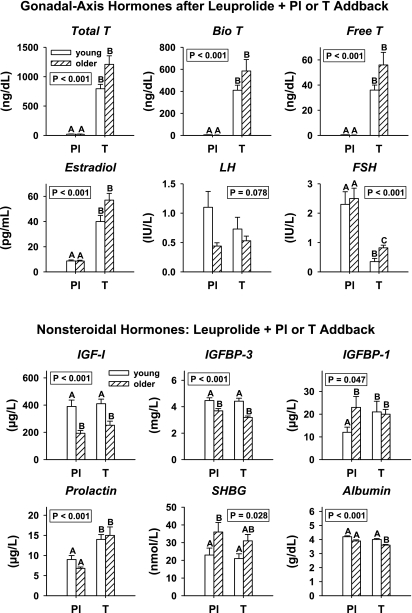
Figure 1A presents endocrine measurements obtained after leuprolide administration with restoration of placebo (Pl) vs. T in young and older individuals. Age per se did not influence T, E2, or LH concentrations when separately assessed in the hypogonadal and eugonadal contexts. The overall range of T was 7–1,950 ng/dl (all subjects), and of that of E2 was 5–79 pg/ml. FSH was suppressed less after T in older than young men. IGF-I and IGFBP-3 concentrations were lower in older than younger individuals, whether Pl or T was restored (Fig. 1B). For the combined groups, IGF-I ranged from 115 to 817 μg/dl and IGFBP-3 from 2,705 to 5,822 μg/l. Higher prolactin concentrations were induced by T than by Pl supplementation in both age groups. Age elevated IGFBP-1 and SHBG concentrations only in the low-T/E2 milieu. Albumin was lower in older men receiving leuprolide with T than in any other group.
Fig. 1.
Concentrations of sex steroids and gonadotropins (top) and extragonadal hormones (bottom) in 47 men stratified by age (young and older) and randomly assigned to 3 wk of placebo (Pl) or testosterone (T) repletion after administration of the gonadotropin-releasing hormone agonist (leuprolide). LH, luteinizing hormone; FSH, follicle-stimulating hormone; IGF-I, insulin-like growth factor I; IGFBP, IGF-binding protein; SHBG, sex hormone-binding globulin. Values are means ± SE. P values were determined by ANOVA. Different (unshared) superscripts (A, B, C) denote significantly different means by Tukey's post hoc test.
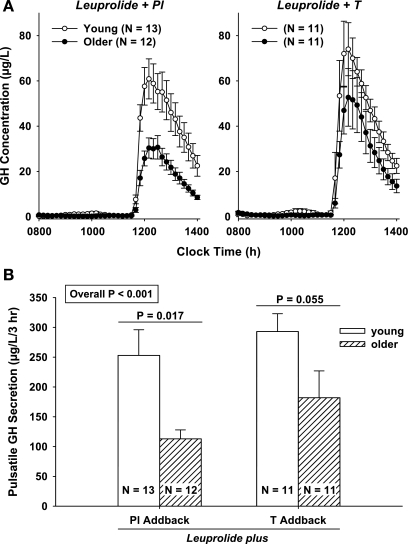
Simultaneous infusion of GHRH and GHRP-2 evoked marked time-dependent release of GH (Fig. 2A). Peak GH concentrations differed significantly among cohorts (P = 0.005 by ANOVA), with maximal values in young men given leuprolide with T (P = 0.006 vs. older men given leuprolide + Pl and P = 0.008 vs. older men given leuprolide + T; Table 2). Peak GH concentrations did not differ among the young men given leuprolide with Pl, young men given leuprolide with T, and older men given leuprolide with T. Combined-peptide stimulation of pulsatile GH secretion as assessed by deconvolution analysis yielded the same descending order of responses (P = 0.001 by ANOVA; Fig. 2B). Unstimulated pulsatile GH secretion was 5.9-fold higher in the young than the older men given leuprolide with T (P = 0.045 ANOVA). Basal (nonpulsatile) GH secretion and secretory-burst shape (mode) did not differ among the four study groups (Table 2).
Fig. 2.
A: time course of growth hormone (GH) concentrations monitored every 10 min for 3 h during saline infusion followed by 3 h of simultaneous infusion of GH-releasing hormone (GHRH) and GH-releasing peptide (GHRP)-2 (both at 1 μg·kg−1·h−1). B: pulsatile GH secretion estimated in the 4 groups of volunteers receiving combined GHRH-GHRP-2 infusion. Values are means ± SE. P values reflect ANOVA and Tukey's post hoc comparisons.
Table 2.
ANOVA of pulsatile GH secretion under GHRH-GHRP-2 drive
| End Point |
Young |
Older
|
P | ||
|---|---|---|---|---|---|
| Pl (n =13) | T (n =11) | Pl (n =12) | T (n =11) | ||
| Peak GH concn, μg/l | 64±9.0*‡ | 80±14* | 34±5.1† | 55±13†‡ | 0.005 |
| GH secretion, μg·l−1·3 h−1 | |||||
| Pulsatile | 253±43*‡ | 293±30* | 113±15† | 182±45†‡ | 0.001 |
| Basal | 3.8±1.5 | 3.1±1.3 | 2.2±0.55 | 3.9±0.85 | 0.48 |
| Secretory-burst mode, min | 15±1.4 | 15±0.67 | 13±1.5 | 16±2.4 | 0.79 |
Values are means ± SE. GH, growth hormone; GHRH, GH-releasing hormone; GHRP, GH-releasing peptide. P values were determined by ANOVA. Different (unshared) symbols ( *, †, ‡) within a row denote significant post hoc differences by Tukey's test.
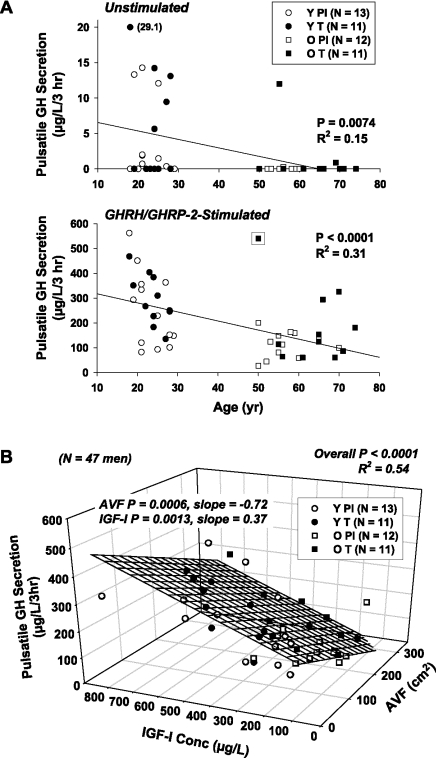
Regression analysis was used to evaluate modulation of GHRH-GHRP-2 synergy (dependent variable) by AVF, age, and sex steroid concentrations (independent variables). The effect of continuous combined GHRH-GHRP-2 stimulation on pulsatile GH secretion was explained univariately by age (negatively, R2 = 0.24, P < 0.001), AVF (negatively, R2 = 0.40, P < 0.01), IGF-I (positively, R2 = 0.39, P < 0.001), and IGFBP-3 (positively but weakly, R2 = 0.10, P = 0.031; Fig. 3A). Bivariate regression analysis revealed that AVF and IGF-I jointly predict pulsatile GH secretion during GHRH-GHRP-2 drive (overall R2 = 0.54, P < 0.0001; Fig. 3B). Individual partial R2 contributions were highly significant (P = 0.0006 for AVF and P = 0.0013 for IGF-I). On the other hand, the positive individual effects of IGFBP-3 and IGF-I were accounted for fully by IGF-I in bivariate regression (R2 = 0.41, P < 0.001). Analogously, when age and IGF-I were allowable independent variables, IGF-I emerged as the dominant predictor of GHRH-GHRP-2 synergy (R2 = 0.47, P < 0.001). When age or IGFBP-3 was paired with T as possible joint determinants of GHRH-GHRP-stimulated GH secretion, age and IGFBP-3 alone were the respective primary correlates (both P < 0.001, R2 = 0.44 and R2 = 0.26).
Fig. 3.
A: linear regression of unstimulated (saline-infused; top) or GHRH-GHRP-2-stimulated (bottom) pulsatile GH secretion on age [young (Y) and older (O)]. Data point enclosed in a box was a statistical outlier at P < 0.001 based on Studentized residuals. B: joint linear regression of GHRH- and GHRP-2-stimulated pulsatile GH secretion (vertical axis) on IGF-I concentrations (horizontal axis) and abdominal-visceral fat (AVF; oblique axis) in 47 men. Bivariate R2 and P values, along with partial P values and slopes for the individual covariates IGF-I and AVF, are shown.
On the basis of the outcomes of univariate and bivariate regression, stepwise forward-selection multivariate analyses was applied. This approach indicated that AVF (negatively), IGF-I (positively), and IGFBP-3 (negatively) together explained 59.8% of the variability in GHRH-GHRP synergy (P < 0.0001).
Basal GH secretion correlated positively with total T (R2 = 0.14, P = 0.015) and total E2 (R2 = 0.20 P = 0.004) by univariate analysis. AVF, age, IGF-I, and IGFBP-3 were not significant determinants. Bivariate regression disclosed that age (P = 0.007) and IGF-I (P = 0.034) were joint predictors of basal GH release (R2 = 0.18, P = 0.021). Stepwise forward-selection multivariate regression identified E2 as the principal predictor of basal GH output (multivariate R2 = 0.17, P = 0.007).
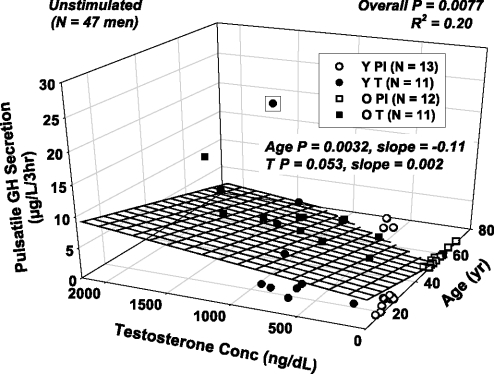
According to univariate regression, pulsatile GH secretion during saline infusion was related negatively to age (R2 = 0.15 P = 0.017) and positively to IGF-I (R2 = 0.20, P = 0.002) and IGFBP-3 (R2 = 0.21, P = 0.001) concentrations. Bivariate regression analysis revealed that age negatively (P = 0.003) and T positively (P = 0.053) together predicted 20% of the variability in fasting pulsatile GH secretion (overall P = 0.0077; Fig. 4). Age and E2 together acted similarly (R2 = 0.19, P = 0.014; age P = 0.006, E2 P = 0.060), but the E2 trend was not significant. Multivariate analyses showed that IGF-I was the primary statistical determinant of fasting pulsatile GH secretion (R2 = 0.20, P = 0.002).
Fig. 4.
Conjoint negative effect of age (oblique axis) and positive effect of T concentrations (horizontal axis) on unstimulated (saline-infused) pulsatile GH secretion (vertical axis) in 47 healthy men. See Fig. 3 legend for format.
DISCUSSION
Salient aspects of this work are 1) introduction of an experimental paradigm of T/E2 depletion and repletion in young and older individuals to dissect the separate and interrelated effects of age and sex steroids on GH secretion, 2) combined continuous intravenous infusion of maximally stimulatory doses of GHRH and GHRP-2 over 3 h to appraise secretagogue synergy, 3) inclusion of men with a wide range of body compositions, so as to estimate the concomitant influence of AVF, and 4) use of a validated deconvolution method to separately quantify pulsatile and basal GH secretion. Under these conditions, stepwise forward-selection multivariate analysis revealed that 1) AVF (negatively), IGF-I (positively), and IGFBP-3 (positively) jointly determine pulsatile GH secretion stimulated by GHRH-GHRP-2, accounting for 60% of total response variability; 2) E2 concentrations constitute a positive correlate of basal (nonpulsatile) GH secretion independently of age, AVF, and IGF-I, IGFBP-3, and T concentrations, explaining 17% of interindividual variations in this measure; and 3) IGF-I concentrations positively predict fasting pulsatile GH secretion during saline infusion, representing 20% of intersubject variance. These outcomes define distinct physiological modulators of basal, fasting pulsatile, and synergistically GHRH-GHRP-driven pulsatile GH secretion.
Two-way ANOVA disclosed that age and T/E2 availability influence the magnitude of GHRH-GHRP-2 synergy (overall P < 0.001 with no statistical interaction). In the low-T/E2 milieu, the magnitude of dual-peptide synergy in older men was 45% of that in young men (P = 0.017). The age-related deficit was less striking in the replaced-T/E2 milieu (older/young response percentage 62%, P = 0.055). The lack of an age × T/E2 interaction at good (>90%) statistical power could indicate that young and older men respond similarly to a change in sex steroid milieu. This postulate would be consistent with dose-response studies showing age-comparable effects of T on skeletal muscle (5). GHRH-GHRP synergy is putatively important physiologically when GHRH and ghrelin are jointly available in high concentrations (40) and when SS outflow is low (20).
Multivariate analysis of primary factors determining GHRH-GHRP-2 synergy unveiled composite contributions by AVF (negative), IGF-I (positive), and IGFBP-3 (negative), with overall R2 = 0.60 and each partial P ≤ 0.01. The relative strengths of different determinants can also be expressed by standardized regression coefficients [slope normalized to SD of the independent variable (42)]. Absolute values of standardized coefficients were (10−3 slope units) 10.3, 3.87, and 0.074, for AVF, IGF-I, and IGFBP-3, respectively. Whereas the contribution of IGF-I was positive, that of IGFBP-3 was negative. The latter finding could suggest that IGFBP-3 mutes pulsatile GH responses to GHRH and GHRP. In this regard, transgenetic silencing of IGFBP-3 expression can enhance linear growth, suggesting an inhibitory effect of the binding protein. Inhibition might be mediated by direct cellular effects of IGFBP-3 and/or by sequestration of otherwise unbound (free) IGF-I (40).
Univariate regression analysis identified E2 (R2 = 0.31) and T (R2 = 0.14) as significant individual positive determinants of basal GH secretion. Bivariate regression showed that age and IGF-I concentrations were positive correlates. When age, AVF, T, E2, IGF-I, and IGFBP-3 were allowed to enter into stepwise forward-selection multivariate regression, E2 emerged as the dominant positive correlate of basal GH secretion (R2 = 0.17). With the assumption of a GH distribution volume of 57 ml/kg in men (20), each 10 pg/ml increase in E2 concentration would predict an increment of 26 μg/day of basal GH secretion. The positive correlation between E2 and basal GH secretion might reflect direct estrogenic stimulation of somatotrope secretion, as recognized in pituitary cell cultures and heterotopic transplants (26).
By univariate assessment, age (negatively, R2 = 0.13), IGF-I (positively, R2 = 0.20), and IGFBP-3 (positively, R2 = 0.21) predicted unstimulated pulsatile GH secretion. In bivariate analyses, the principal paired correlates were age (negative) and T (positive), with overall R2 = 0.20 and P = 0.008. Each 100 ng/dl decrement in T or 5-yr increment in age was associated with respective decreases in unstimulated pulsatile GH secretion of 6.4 and 35 μg/day. According to multivariate regression analysis, IGF-I was the dominant determinant (R2 = 0.20, P = 0.002), such that each 100 μg/l increment in IGF-I translated into 45 μg more GH secreted daily in pulses. The correspondence between IGF-I concentrations and pulsatile GH secretion may reflect the fact that GH pulses activate STAT5b signaling and that STAT5b mediates stimulation of IGF-I synthesis (11).
Unresolved questions include whether longer-term T/E2 depletion would exert comparable or greater effects. In addition, how AVF, age, and sex steroids together influence feedback by GH and IGF-I remains to be defined. Paradigms comparable to that presented here should have utility in further dissecting multifactorial mechanisms that regulate GH secretion.
In summary, a paradigm of fixed maximal combined GHRH-GHRP stimulation and experimentally controlled T/E2 milieus in healthy men reveals E2-predominant regulation of basal (nonpulsatile) GH secretion; a strong positive association between IGF-I and fasting pulsatile GH secretion; and conjoint AVF (negative), IGFBP-3 (negative), and IGF-I (positive) determination of GHRH-GHRP synergy. These outcomes indicate the need for more extended physiological models of GH-axis regulation that incorporate individual and interactive effects of age, T, E2, AVF, IGF-I, and IGFBP-3.
GRANTS
This work was supported in part by Center for Translational Science Activities Grant 1 UL 1 RR-024150 to the Mayo Clinic and Foundation from the National Center for Research Resources and National Institute on Aging Grant R01 AG-19695.
Acknowledgments
We thank Donna Scott for manuscript preparation, Ashley Bryant for data analysis and graphics, the Mayo Immunochemical Laboratory for assay assistance, and the Mayo research nursing staff for implementation of the protocol.
REFERENCES
- 1.Akaike H A new look at the statistical model identification. IEEE Trans Autom Control 19: 716–723, 1974. [Google Scholar]
- 2.Alba-Roth J, Muller OA, Schopohl J, Von Werder K. Arginine stimulates growth hormone secretion by suppressing endogenous somatostatin secretion. J Clin Endocrinol Metab 67: 1186–1189, 1988. [DOI] [PubMed] [Google Scholar]
- 3.Anderson SM, Shah N, Evans WS, Patrie JT, Bowers CY, Veldhuis JD. Short-term estradiol supplementation augments growth hormone (GH) secretory responsiveness to dose-varying GH-releasing peptide infusions in healthy postmenopausal women. J Clin Endocrinol Metab 86: 551–560, 2001. [DOI] [PubMed] [Google Scholar]
- 4.Arvat E, Maccario M, Di Vito L, Broglio F, Benso A, Gottero C, Papotti M, Muccioli G, Dieguez C, Casanueva FF, Deghenghi R, Camanni F, Ghigo E. Endocrine activities of ghrelin, a natural growth hormone secretagogue (GHS), in humans: comparison and interactions with hexarelin, a nonnatural peptidyl GHS, and GH-releasing hormone. J Clin Endocrinol Metab 86: 1169–1174, 2001. [DOI] [PubMed] [Google Scholar]
- 5.Bhasin S, Woodhouse L, Casaburi R, Singh AB, Mac RP, Lee M, Yarasheski KE, Sinha-Hikim I, Dzekov C, Dzekov J, Magliano L, Storer TW. Older men are as responsive as young men to the anabolic effects of graded doses of testosterone on the skeletal muscle. J Clin Endocrinol Metab 90: 678–688, 2005. [DOI] [PubMed] [Google Scholar]
- 6.Bluet-Pajot MT, Epelbaum J, Gourdji D, Hammond C, Kordon C. Hypothalamic and hypophyseal regulation of growth hormone secretion. Cell Mol Neurobiol 18: 101–123, 1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bondanelli M, Ambrosio MR, Margutti A, Franceschetti P, Zatelli MC, degli Uberti EC. Activation of the somatotropic axis by testosterone in adult men: evidence for a role of hypothalamic growth hormone-releasing hormone. Neuroendocrinology 77: 380–387, 2003. [DOI] [PubMed] [Google Scholar]
- 8.Chalew SA, Udoff LC, Hanukoglu A, Bistritzer T, Armour KM, Kowarski AA. The effect of testosterone therapy on spontaneous growth hormone secretion in boys with constitutional delay. Am J Dis Child 142: 1345–1348, 1988. [DOI] [PubMed] [Google Scholar]
- 9.Chattopadhyay S, Veldhuis JD, Keenan DM. Probabilistic recovery of pulsatile, secretory and kinetic structure: an alternating discrete and continuous schema. Q Appl Math 66: 401–421, 2008. [Google Scholar]
- 10.Darzy KH, Aimaretti G, Wieringa G, Gattamaneni HR, Ghigo E, Shalet SM. The usefulness of the combined growth hormone (GH)-releasing hormone and arginine stimulation test in the diagnosis of radiation-induced GH deficiency is dependent on the postirradiation time interval. J Clin Endocrinol Metab 88: 95–102, 2003. [DOI] [PubMed] [Google Scholar]
- 11.Davey HW, Park SH, Grattan DR, McLachlan MJ, Waxman DJ. STAT5b-deficient mice are growth hormone pulse-resistant. Role of STAT5b in sex-specific liver p450 expression. J Biol Chem 274: 35331–35336, 1999. [DOI] [PubMed] [Google Scholar]
- 12.Eakman GD, Dallas JS, Ponder SW, Keenan BS. The effects of testosterone and dihydrotestosterone on hypothalamic regulation of growth hormone secretion. J Clin Endocrinol Metab 81: 1217–1223, 1996. [DOI] [PubMed] [Google Scholar]
- 13.Farhy LS, Veldhuis JD. Deterministic construct of amplifying actions of ghrelin on pulsatile GH secretion. Am J Physiol Regul Integr Comp Physiol 288: R1649–R1663, 2005. [DOI] [PubMed] [Google Scholar]
- 14.Faria ACS, Veldhuis JD, Thorner MO, Vance ML. Half-time of endogenous growth hormone (GH) disappearance in normal man after stimulation of GH secretion by GH-releasing hormone and suppression with somatostatin. J Clin Endocrinol Metab 68: 535–541, 1989. [DOI] [PubMed] [Google Scholar]
- 15.Fisher LD, van Belle G. Descriptive statistics. In: Biostatistics: A Methodology for the Health Sciences. New York: Wiley, 1996, p. 58–74.
- 16.Frohman LA New insights into the regulation of somatotrope function using genetic and transgenic models. Metabolism 45: 1–3, 1996. [DOI] [PubMed] [Google Scholar]
- 17.Fryburg DA, Weltman A, Jahn LA, Weltman JY, Samolijik E, Veldhuis JD. Short-term modulation of the androgen milieu alters pulsatile but not exercise or GHRH-stimulated GH secretion in healthy men. J Clin Endocrinol Metab 82: 3710–3719, 1997. [DOI] [PubMed] [Google Scholar]
- 18.Gentili A, Mulligan T, Godschalk M, Clore J, Patrie J, Iranmanesh A, Veldhuis JD. Unequal impact of short-term testosterone repletion on the somatotropic axis of young and older men. J Clin Endocrinol Metab 87: 825–834, 2002. [DOI] [PubMed] [Google Scholar]
- 19.Ghigo E, Aimaretti G, Arvat E, Camanni F. Growth hormone-releasing hormone combined with arginine or growth hormone secretagogues for the diagnosis of growth hormone deficiency in adults. Endocrine 15: 29–38, 2001. [DOI] [PubMed] [Google Scholar]
- 20.Giustina A, Veldhuis JD. Pathophysiology of the neuroregulation of growth hormone secretion in experimental animals and the human. Endocr Rev 19: 717–797, 1998. [DOI] [PubMed] [Google Scholar]
- 21.Guillaume V, Magnan E, Cataldi M, Dutour A, Sauze N, Renard M, Razafindraibe H, Conte-Devolx B, Deghenghi R, Lenaerts V. Growth hormone (GH)-releasing hormone secretion is stimulated by a new GH-releasing hexapeptide in sheep. Endocrinology 135: 1073–1076, 1994. [DOI] [PubMed] [Google Scholar]
- 22.Gustafsson JA, Eden S, Eneroth P, Hokfelt T, Isaksson O, Jansson JO, Mode A, Norstedt G. Regulation of sexually dimorphic hepatic steroid metabolism by the somatostatin-growth hormone axis. J Steroid Biochem 19: 691–698, 1983. [DOI] [PubMed] [Google Scholar]
- 23.Ham JN, Ginsberg JP, Hendell CD, Moshang T Jr. Growth hormone releasing hormone plus arginine stimulation testing in young adults treated in childhood with cranio-spinal radiation therapy. Clin Endocrinol (Oxf) 62: 628–632, 2005. [DOI] [PubMed] [Google Scholar]
- 24.Hataya Y, Akamizu T, Takaya K, Kanamoto N, Ariyasu H, Saijo M, Moriyama K, Shimatsu A, Kojima M, Kangawa K, Nakao K. A low dose of ghrelin stimulates growth hormone (GH) release synergistically with GH-releasing hormone in humans. J Clin Endocrinol Metab 86: 4552, 2001. [DOI] [PubMed] [Google Scholar]
- 25.Hobbs CJ, Plymate SR, Rosen CJ, Adler RA. Testosterone administration increases insulin-like growth factor-I levels in normal men. J Clin Endocrinol Metab 77: 776–779, 1993. [DOI] [PubMed] [Google Scholar]
- 26.Jansson JO, Carlsson L, Seeman H. Estradiol—but not testosterone—stimulates the secretion of growth hormone in rats with the pituitary gland autotransplanted to the kidney capsule. Acta Endocrinol (Copenh) 103: 212–218, 1983. [Google Scholar]
- 27.Keenan DM, Chattopadhyay S, Veldhuis JD. Composite model of time-varying appearance and disappearance of neurohormone pulse signals in blood. J Theor Biol 236: 242–255, 2005. [DOI] [PubMed] [Google Scholar]
- 28.Kojima M, Kangawa K. Ghrelin: structure and function. Physiol Rev 85: 495–522, 2005. [DOI] [PubMed] [Google Scholar]
- 29.Low MJ, Otero-Corchon V, Parlow AF, Ramirez JL, Kumar U, Patel YC, Rubinstein M. Somatostatin is required for masculinization of growth hormone-regulated hepatic gene expression but not of somatic growth. J Clin Invest 107: 1571–1580, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mauras N, Blizzard RM, Link K, Johnson ML, Rogol AD, Veldhuis JD. Augmentation of growth hormone secretion during puberty: evidence for a pulse amplitude-modulated phenomenon. J Clin Endocrinol Metab 64: 596–601, 1987. [DOI] [PubMed] [Google Scholar]
- 31.Muller EE, Locatelli V, Cocchi D. Neuroendocrine control of growth hormone secretion. Physiol Rev 79: 511–607, 1999. [DOI] [PubMed] [Google Scholar]
- 32.Muniyappa R, Sorkin JD, Veldhuis JD, Harman SM, Munzer T, Bhasin S, Blackman MR. Long-term testosterone supplementation augments overnight growth hormone secretion in healthy older men. Am J Physiol Endocrinol Metab 293: E769–E775, 2007. [DOI] [PubMed] [Google Scholar]
- 33.Popovic V, Pekic S, Doknic M, Micic D, Damjanovic S, Zarkovic M, Aimaretti G, Corneli G, Ghigo E, Deiguez C, Casanueva FF. The effectiveness of arginine + GHRH test compared with GHRH + GHRP-6 test in diagnosing growth hormone deficiency in adults. Clin Endocrinol (Oxf) 59: 251–257, 2003. [DOI] [PubMed] [Google Scholar]
- 34.Shuto Y, Shibasaki T, Otagiri A, Kuriyama H, Ohata H, Tamura H, Kamegai J, Sugihara H, Oikawa S, Wakabayashi I. Hypothalamic growth hormone secretagogue receptor regulates growth hormone secretion, feeding, and adiposity. J Clin Invest 109: 1429–1436, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vahl N, Jorgensen JO, Skjaerback C, Veldhuis JD, Orskov H, Christiansen J. Abdominal adiposity rather than age and sex predicts the mass and patterned regularity of growth hormone secretion in mid-life healthy adults. Am J Physiol Endocrinol Metab 272: E1108–E1116, 1997. [DOI] [PubMed] [Google Scholar]
- 36.Van den Berghe G, de Zegher F, Veldhuis JD, Wouters P, Verbruggen W, Awouters M, Schetz M, Verwaest C, Lauwers P, Bouillon R, Bowers CY. The somatotropic axis in critical illness: effects of continuous GHRH and GHRP-2 infusion. J Clin Endocrinol Metab 82: 590–599, 1997. [DOI] [PubMed] [Google Scholar]
- 37.Veldhuis JD, Anderson SM, Iranmanesh A, Bowers CY. Testosterone blunts feedback inhibition of GH secretion by experimentally elevated IGF-I concentrations. J Clin Endocrinol Metab 90: 1613–1617, 2005. [DOI] [PubMed] [Google Scholar]
- 38.Veldhuis JD, Evans WS, Bowers CY. Impact of estradiol supplementation on dual peptidyl drive of growth-hormone secretion in postmenopausal women. J Clin Endocrinol Metab 87: 859–866, 2002. [DOI] [PubMed] [Google Scholar]
- 39.Veldhuis JD, Evans WS, Iranmanesh A, Weltman AL, Bowers CY. Short-term testosterone supplementation relieves growth hormone autonegative feedback in men. J Clin Endocrinol Metab 89: 1285–1290, 2004. [DOI] [PubMed] [Google Scholar]
- 40.Veldhuis JD, Roemmich JN, Richmond EJ, Bowers CY. Somatotropic and gonadotropic axes linkages in infancy, childhood, and the puberty-adult transition. Endocr Rev 27: 101–140, 2006. [DOI] [PubMed] [Google Scholar]
- 41.Waxman DJ, Pampori NA, Ram PA, Agrawal AK, Shapiro BH. Interpulse interval in circulating growth hormone patterns regulates sexually dimorphic expression of hepatic cytochrome P450. Proc Natl Acad Sci USA 88: 6868–6872, 1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Weltman A, Weltman JY, Hartman ML, Abbott RD, Rogol AD, Evans WS, Veldhuis JD. Relationship between age, percentage body fat, fitness, and 24-hour growth hormone release in healthy young adults: effects of gender. J Clin Endocrinol Metab 78: 543–548, 1994. [DOI] [PubMed] [Google Scholar]