Abstract
The in vitro susceptibilities of 59 avian Aspergillus fumigatus strains to amphotericin B, itraconazole, and voriconazole were determined using the standard microdilution broth method (CLSI M38-A2). Four isolates showed acquired resistance to itraconazole and voriconazole, harboring implications for the treatment of aspergillosis in both birds and humans.
Aspergillosis is the most common fungal disease of the avian respiratory tract and is caused primarily by Aspergillus fumigatus. Prognosis of avian aspergillosis is often poor due to delayed diagnostics and treatment failure. Acquired resistance to antimycotics in humans has been reported (6, 12). However, Dannaoui et al. (5) and Verweij et al. (22) found that the prevalences of high MICs of amphotericin B (>2 μg/ml), itraconazole (>16 μg/ml), and voriconazole (>1 μg/ml) among clinical isolates of A. fumigatus in France and The Netherlands were low (0%, ∼2%, and 0%, respectively). A 2003 survey of isolates from human patients hospitalized in North America, Europe, and Latin America showed that A. fumigatus was generally less susceptible to amphotericin B and voriconazole than did a 2000 survey (18). Aspergillus isolates collected from medical centers worldwide between 2000 and 2006 with MICs of voriconazole and itraconazole of >2 μg/ml were found to be rare (<1% and 0% of the isolates, respectively) (19). Recently, 1,912 clinical A. fumigatus isolates from a university medical center in The Netherlands (1994 to 2007) were investigated and indicated that itraconazole resistance had emerged since 2000, with annual prevalences of 1.7 to 6% (21). The widespread uses of azole prophylaxis in patients (5, 6) and in agriculture (11) are thought to be the major driver of azole resistance in human medicine. Also, in avian medicine, azoles are used for antifungal therapy (including prophylaxis). To date, little is known about acquired resistance to antimycotics in isolates from birds suffering from aspergillosis. The novel antifungal agent voriconazole has become the preferred drug for treatment of respiratory and disseminated aspergillosis in humans and has also been used for treatment of fungal infections in birds (3, 9, 10).
In this study, 59 A. fumigatus isolates from domestic and wild birds were included. The isolates were obtained from Anseriformes (n = 5), Charadriiformes (n = 3), Ciconiiformes (n = 1), Columbiformes (n = 6), Falconiformes (n = 10), Galliformes (n = 4), Passeriformes (n = 5), Piciformes (n = 1), Psittaciformes (n = 14), Sphenisciformes (n = 2), Strigiformes (n = 2), and Struthioniformes (n = 1) birds in Belgium and The Netherlands. The isolates were identified based on the macro- and micromorphology of the fungus. Determination of partial DNA sequences of the β-tubulin and rodlet A genes (1) and the ability to grow at 48°C was used to confirm species identity. To exclude repeated testing of identical strains, microsatellite length polymorphism was performed (4). The MICs of amphotericin B, itraconazole, and voriconazole were determined using the broth microdilution method as described in the Clinical and Laboratory Standards Institute (CLSI) document M38-A2, with Candida krusei IHEM 9560 (ATCC 6258) as a quality control.
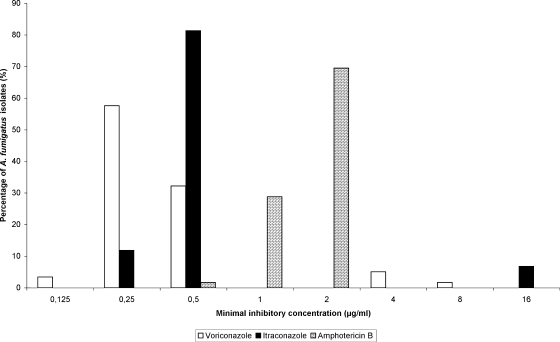
The results of the in vitro susceptibility testing of amphotericin B, itraconazole, and voriconazole for the 59 avian A. fumigatus isolates are summarized in Table 1 and Fig. 1. For amphotericin B, a monomodal MIC distribution was seen, indicating the absence of acquired resistance. Figure 1, however, shows a bimodal MIC distribution for itraconazole and voriconazole, indicating acquired resistance in the four isolates in the higher range of MICs. These isolates were genetically different and were obtained from Pyrrhura sp., Psittacus erithacus, Pionus sp., and Geranoaetus melanoleucus birds. Two of these domestic birds had been treated with itraconazole (treatment schedule unknown).
TABLE 1.
MIC ranges, MIC50s, and MIC90s of amphotericin B, itraconazole, and voriconazole for 59 avian A. fumigatus isolatesa
| Antifungal agent | MIC range (μg/ml) | MIC50 (μg/ml) | MIC90 (μg/ml) |
|---|---|---|---|
| Amphotericin B | 0.5-2 | 2 | 2 |
| Itraconazole | 0.25->16 | 0.5 | 0.5 |
| Voriconazole | 0.13-8 | 0.25 | 0.5 |
MICs were determined using the standard microdilution broth method (CLSI M38-A2).
FIG. 1.
Distribution of MICs of amphotericin B, itraconazole, and voriconazole for 59 avian A. fumigatus isolates. MICs were determined using the standard microdilution broth method (CLSI M38-A2).
The main goal of in vitro susceptibility testing of antifungal agents is to predict the clinical outcome of therapy. However, interpretive breakpoints for any mold-drug combination have not been established yet, although high itraconazole and amphotericin B MICs have been associated with treatment failure in humans and/or mice (6, 8, 12, 15). Data regarding association between MICs and clinical outcome are not available for voriconazole. In this study, the MIC of itraconazole was at least 32 times higher and that of voriconazole at least 8 to 16 times higher in the four isolates with acquired resistance than in the other isolates. The likelihood that birds or humans infected with these isolates will respond well to treatment with these agents should be considered to be low.
Various mechanisms of azole resistance have been described (2, 7, 13, 17). To date, the most prevalent mechanism of azole resistance appears to be the modification of the cyp51A gene, encoding the target enzyme 14α-sterol demethylase (16). These mutations, reducing the binding of azoles to the enzymatic site, have been associated with different antifungal susceptibility profiles, depending on the azole structure and concomitant binding affinities (14). Hence, mutations resulting in resistance to itraconazole appear to differ from those providing resistance to voriconazole (20, 23). In this study, the four isolates having itraconazole MICs of >16 μg/ml were also less susceptible to voriconazole (MICs of 4 to 8 μg/ml), which might indicate cross-resistance. When elevated MICs for itraconazole and voriconazole arise, resistance mechanisms resulting in elevations of all azole MICs seem to be most likely. Possible mechanisms are amino acid substitutions of methionine at position 220 in the cyp51A gene product and substitution of leucine at position 98 for histidine in the cyp51A gene product, together with two copies of a 34-bp sequence in tandem in the cyp51A gene promoter (16, 17, 20, 21).
The source of the resistant isolates found in these birds is unclear. In human medicine, acquisition of itraconazole resistance after prolonged therapy has been documented (6). In this study, two of the four resistant strains were isolated from birds that received itraconazole. The possible effect of antimycotic treatment in birds is unknown, and because isolates were not available before treatment, this could not be further investigated. Determination of resistant isolates in birds can also be considered an indication of the presence of acquired resistance in the surrounding environment. Acquisition of resistance in the environment might be induced by the extensive use of azoles for plant protection (11), which consequently gain access to susceptible birds and humans. Hence, further investigation of this phenomenon is necessary, as it harbors implications for the treatment of aspergillosis in both birds and humans.
To our knowledge, this is the first report of birds infected with A. fumigatus strains that harbor acquired resistance to both itraconazole and voriconazole.
Acknowledgments
This work was supported by the Institute for the Promotion of Innovation through Science and Technology in Flanders (IWT Vlaanderen), Brussels, Belgium.
We have no conflicts of interest regarding the present work.
The skillful technical assistance of S. De Bruycker is greatly appreciated.
Footnotes
Published ahead of print on 2 March 2009.
REFERENCES
- 1.Alcazar-Fuoli, L., E. Mellado, A. Alastruey-Izquierdo, M. Cuenca-Estrella, and J. L. Rodriguez-Tudela. 2008. Aspergillus section Fumigati: antifungal susceptibility patters and sequence-based identification. Antimicrob. Agents Chemother. 52:1244-1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alcazar-Fuoli, L., E. Mellado, G. Garcia-Effron, J. F. Lopez, J. O. Grimalt, J. M. Cuenca-Estrella, and J. L. Rodriguez-Tudela. 2008. Ergosterol biosynthesis pathway in Aspergillus fumigatus. Steroids 73:339-347. [DOI] [PubMed] [Google Scholar]
- 3.Beernaert, L. A., K. Baert, P. Marin, K. Chiers, P. De Backer, F. Pasmans, and A. Martel. 2008. Designing voriconazole treatment for racing pigeons: balancing between hepatic enzyme auto induction and toxicity. Med. Mycol. 5:1-10 [Epub ahead of print.]. doi: 10.1080/1 369378080 2262115. [DOI] [PubMed] [Google Scholar]
- 4.Beernaert, L. A., F. Pasmans, F. Haesebrouck, and A. Martel. 2008. Modelling Aspergillus fumigatus infections in racing pigeons (Columba livia domestica). Avian Pathol. 37:545-549. [DOI] [PubMed] [Google Scholar]
- 5.Dannaoui, E., F. Persat, M. F. Monier, E. Borel, M. A. Piens, and S. Picot. 1999. In-vitro susceptibility of Aspergillus spp. isolates to amphotericin B and itraconazole. J. Antimicrob. Chemother. 44:553-555. [DOI] [PubMed] [Google Scholar]
- 6.Dannaoui, E., E. Borel, M. F. Monier, M. A. Piens, S. Picot, and F. Persat. 2001. Acquired itraconazole resistance in Aspergillus fumigatus. J. Antimicrob. Chemother. 47:333-340. [DOI] [PubMed] [Google Scholar]
- 7.da Silva Ferreira, M. E., J. L. Capellaro, E. dos Reis Marques, I. Malavazi, D. Perlin, S. Park, J. B. Anderson, A. L. Colombo, B. A. Arthington-Skaggs, M. H. S. Goldman, and G. H. Goldman. 2004. In vitro evolution of itraconazole resistance in Aspergillus fumigatus involves multiple mechanisms of resistance. Antimicrob. Agents Chemother. 48:4405-4413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Denning, D. W., S. A. Radford, K. L. Oakley, L. Hall, E. M. Johnson, and D. W. Warnock. 1997. Correlation between in-vitro susceptibility testing to itraconazole and in-vivo outcome of Aspergillus fumigatus infection. J. Antimicrob. Chemother. 40:401-414. [DOI] [PubMed] [Google Scholar]
- 9.Di Somma, A., T. Bailey, C. Silvanose, and C. Garcia-Martinez. 2007. The use of voriconazole for the treatment of aspergillosis in falcons (Falco species). J. Avian Med. Surg. 21:307-316. [DOI] [PubMed] [Google Scholar]
- 10.Gothard, P., and T. R. Rogers. 2004. Voriconazole for serious fungal infections. Int. J. Clin. Pract. 58:74-80. [DOI] [PubMed] [Google Scholar]
- 11.Hoff, H. 2001. Critical annotations to the use of azole antifungals for plant protection. Antimicrob. Agents Chemother. 45:2987-2990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lass-Flörl, C., G. Kofler, G. Kropshofer, J. Hermans, A. Kreczy, M. P. Dierich, and D. Niederwieser. 1998. In-vitro testing of susceptibility to amphotericin B is a reliable predictor of clinical outcome in invasive aspergillosis. J. Antimicrob. Chemother. 42:497-502. [DOI] [PubMed] [Google Scholar]
- 13.Manavathu, E. K., J. A. Vazquez, and P. H. Chandrasekar. 1999. Reduced susceptibility in laboratory-selected mutants of Aspergillus fumigatus to itraconazole due to decreased intracellular accumulation of the antifungal agent. Int. J. Antimicrob. Agents 12:213-219. [DOI] [PubMed] [Google Scholar]
- 14.Manavathu, E. K., O. C. Abraham, and P. H. Chandrasekar. 2001. Isolation and in vitro susceptibility to amphotericin B, itraconazole and posaconazole of voriconazole-resistant laboratory isolates of Aspergillus fumigatus. Clin. Microbiol. Infect. 7:130-137. [DOI] [PubMed] [Google Scholar]
- 15.Manavathu, E. K., J. L. Cutright, and P. H. Chandrasekar. 2005. In vivo resistance of a laboratory-selected Aspergillus fumigatus isolate to amphotericin B. Antimicrob. Agents Chemother. 49:428-430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mellado, E., G. Garcia-Effron, L. Alcazar-Fuoli, M. Cuenca-Estrella, and J. L. Rodriguez-Tudela. 2004. Substitutions at methionine 220 in the 14α-sterol demethylase (Cyp51A) of Aspergillus fumigatus are responsible for resistance in vitro to azole antifungal drugs. Antimicrob. Agents Chemother. 48:2747-2750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mellado, E., G. Garcia-Effron, L. Alcazar-Fuoli, W. J. G. Melchers, P. E. Verweij, M. Cuenca-Estrella, and J. L. Rodriguez-Tudela. 2007. A new Aspergillus fumigatus resistance mechanism conferring in vitro cross-resistance to azole antifungals involves a combination of cyp51A alterations. Antimicrob. Agents Chemother. 51:1897-1904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Messer, S. A., R. N. Jones, and T. R. Fritsche. 2006. International surveillance of Candida spp. and Aspergillus spp.: report from the SENTRY antimicrobial surveillance program (2003). J. Clin. Microbiol. 44:1782-1787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pfaller, M. A., S. A. Messer, L. Boyken, C. Rice, S. Tendolkar, R. J. Hollis, and D. J. Diekema. 2008. In vitro survey of triazole cross-resistance among more than 700 clinical isolates of Aspergillus species. J. Clin. Microbiol. 46:2568-2572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rodriguez-Tudela, J. L., L. Alcazar-Fuoli, E. Mellado, A. Alastruey-Izquierdo, A. Monzon, and M. Cuenca-Estrella. 2008. Epidemiological cutoffs and cross-resistance to azole drugs in Aspergillus fumigatus. Antimicrob. Agents Chemother. 52:2468-2472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Snelders, E., H. A. L. van der Lee, J. Kuijpers, A. J. M. M. Rijs, J. Varga, R. A. Samson, E. Mellado, A. R. T. Donders, W. J. G. Melchers, and P. E. Verweij. 2008. Emergence of azole resistance in Aspergillus fumigatus and spread of a single resistance mechanism. PLoS Med. 5:e219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Verweij, P. E., D. T. A. Te Dorsthorst, A. J. M. M. Rijs, H. G. De Vries-Hospers, J. F. G. M. Meis, and the Dutch Interuniversity Working Party for Invasive Mycoses. 2002. Nationwide survey of in vitro activities of itraconazole and voriconazole against clinical Aspergillus fumigatus isolates cultured between 1945 and 1998. J. Clin. Microbiol. 40:2648-2650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xiao, L., V. Madison, A. S. Chau, D. Loebenberg, R. E. Palermo, and P. M. McNicholas. 2004. Three-dimensional models of wild-type and mutated forms of cytochrome P450 14α-sterol demethylases from Aspergillus fumigatus and Candida albicans provide insights into posaconazole binding. Antimicrob. Agents Chemother. 48:568-574. [DOI] [PMC free article] [PubMed] [Google Scholar]