Abstract
nsp1 protein of severe acute respiratory syndrome coronavirus (SARS-CoV), a group 2b CoV, suppresses host gene expression by promoting host mRNA degradation and translation inhibition. The present study analyzed the activities of nsp1 proteins from the group 2 bat CoV strains Rm1, 133, and HKU9-1, belonging to groups 2b, 2c, and 2d, respectively. The host mRNA degradation and translational suppression activities of nsp1 of SARS-CoV and Rm1 nsp1 were similar and stronger than the activities of the nsp1 proteins of 133 and HKU9-1. Rm1 nsp1 expression in trans strongly inhibited the induction of type I interferon (IFN-I) and IFN-stimulated genes in cells infected with an IFN-inducing SARS-CoV mutant, while 133 and HKU9-1 nsp1 proteins had relatively moderate IFN-inhibitory activities. The results of our studies suggested a conserved function among nsp1 proteins of SARS-CoV and group 2 bat CoVs.
Severe acute respiratory syndrome coronavirus (SARS-CoV) is the etiological agent of a newly emerged disease, SARS, which originated in southern China in 2002 and spread to various areas of the world in a 2003 epidemic (reviewed in reference 2). Bats are the natural reservoir of a variety of group 1 and 2 CoVs, including viruses closely related to SARS-CoV, SARS-like CoVs (SLCoVs) (11, 13, 21, 23). Considering the potential of bat CoVs as emerging pathogens for humans and animals, it is of the utmost importance to characterize these viruses.
SARS-CoV nsp1 is a 180-amino-acid protein that is translated from the most 5′ coding region of the SARS-CoV genome (19). SARS-CoV nsp1 protein induces host mRNA degradation and translational suppression both in nsp1-expressing cells and in SARS-CoV-infected cells (9, 14). Studies of the nsp1 proteins of mouse hepatitis virus (MHV) and SARS-CoV, which belong to CoV groups 2a and 2b, respectively, suggest that nsp1 plays important roles in the suppression of host innate immune functions and contributes to viral pathogenesis (9, 14, 22, 26). These observations and the fact that SARS-CoV nsp1 exhibits sequence similarity to the nsp1 proteins of other group 2 CoVs but not to those of group 1 CoVs (18) led us to hypothesize that nsp1 proteins of the newly identified group 2 bat CoVs have similar functions to suppress host gene expression and block host antiviral immune responses.
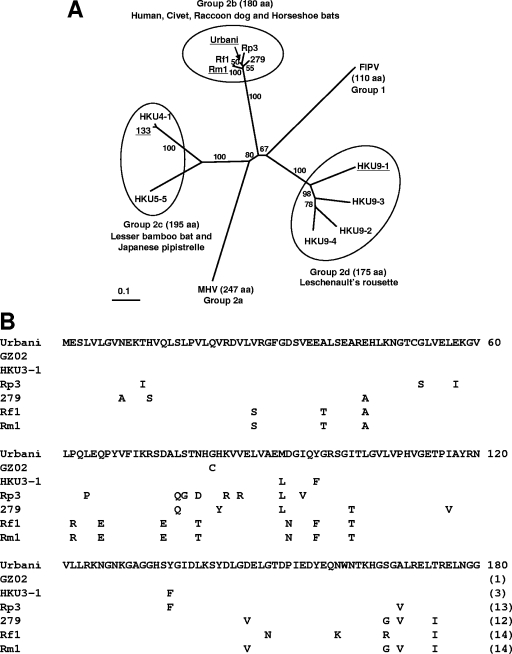
The present study explored the activities of nsp1 proteins of group 2 bat CoVs for the suppression of host gene expression. At the initiation of our studies, group 2 bat CoVs had been further divided tentatively into three subgroups, including 2b and the putative subgroups 2c and 2d (24). We have chosen to characterize the nsp1 proteins of three group 2 bat CoV strains, Rm1 (15), 133 (20), and HKU9-1 (24), belonging to the subgroups 2b, 2c, and 2d, respectively. The levels of amino acid sequence identity of Rm1, 133, and HKU9-1 nsp1 proteins to SARS-CoV nsp1 protein are 92.2, 19.7, and 30.9%, respectively. The low levels of amino acid sequence homology between nsp1 proteins of group 2c and 2d CoVs and SARS-CoV nsp1 are comparable to the levels of amino acid sequence homology between nsp1 proteins of the group 2a CoVs, including MHV and bovine CoV, and SARS-CoV nsp1; the degrees of amino acid sequence identity of MHV nsp1 and bovine CoV nsp1 to SARS-CoV nsp1 are 20.6 and 17.3%, respectively (18). Figure 1A shows the phylogenetic relationships of the group 2 CoV nsp1 proteins, including the group 2 bat CoV nsp1 proteins analyzed in this study.
FIG. 1.
Unrooted phylogenetic tree of CoV nsp1 proteins and amino acid sequence variations among group 2b CoV nsp1 proteins. (A) The unrooted phylogenetic tree of CoV nsp1 proteins was constructed by the neighbor-joining method and the Treeview program and based on 100 bootstrapped data sets. The names of virus strains with nsp1 proteins that were analyzed in this study are underlined. The virus strains used in the phylogenetic analysis are as follows: Urbani, human isolate of SARS-CoV; Rm1 and 279, bat isolates from R. macrotis (horseshoe bat); Rf1, bat isolate from R. ferrumequinum; Rp3, bat isolate from R. pearsoni; 133 and HKU4-1, bat isolates from Tylonycteris pachypus (lesser bamboo bat); HKU5-5, bat isolate from Pipistrellus abramus (Japanese pipistrelle); HKU9-1, HKU9-2, HKU9-3, and HKU9-4, bat isolates from Rousettus leschenaulti (Leschenault's rousette); MHV strain A59; and feline infectious peritonitis virus (FIPV) strain WSU-79/1146. The amino acid (aa) lengths of nsp1 proteins of group 2b, 2c, and 2d CoVs are indicated in parentheses. The scale bar indicates the estimated number of substitutions per 10 amino acids. (B) The full-length SARS-CoV Urbani nsp1 amino acid sequence is represented. Only the amino acid residues that differ between SARS-CoV nsp1 and other group 2b CoV nsp1 proteins are shown. In addition to the group 2b CoV amino acid sequences used for the phylogenetic tree, those of GZ02 (human isolate of SARS-CoV) and HKU3-1 (bat isolate from R. sinicus) were compared. The total number of amino acid differences in each nsp1 protein compared with SARS-CoV nsp1 is indicated in parentheses. The number at the end of each line of the SARS-CoV Urbani nsp1 sequence indicates the amino acid position.
To examine the effects of group 2 bat CoV nsp1 proteins on reporter gene expression, cDNAs were synthesized by Bio Basic Inc. according to the nsp1 amino acid sequences of Rm1 (180 amino acids) (15), 133 (195 amino acids) (20), and HKU9-1 (175 amino acids) (24), with the appropriate codon modifications for optimized translations in human cells. The plasmids pCAGGS-Rm1, pCAGGS-133, and pCAGGS-HKU9-1 were constructed by inserting the Rm1 nsp1 open reading frame (ORF), the 133 nsp1 ORF, and the HKU9-1 nsp1 ORF, respectively, into pCAGGS-MCS, each with a sequence encoding a C-terminal myc tag. As controls, the parental plasmid pCAGGS; pCAGGS-Nsp1-WT, encoding the SARS-CoV nsp1 protein (9); and pCAGGS-Nsp1-mt, encoding a mutant form of SARS-CoV nsp1 (SCoVnsp1-mt) containing alanines in place of the positively charged amino acids K164 and H165 in nsp1 of SARS-CoV (14), were used; expressed SARS-CoV nsp1 protein, but not SCoVnsp1-mt protein, suppresses host gene expression (14).
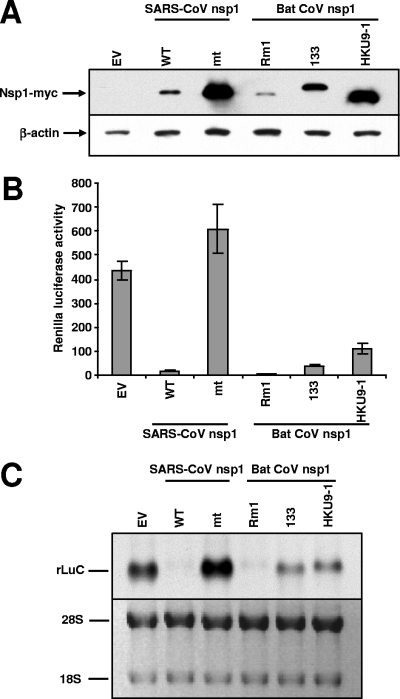
293 cells grown in 24-well plates were cotransfected in triplicate with 0.1 μg of pRL-SV40, in which the Renilla luciferase (RLuc) gene was cloned downstream of the simian virus 40 promoter (9), and 0.5 μg of the pCAGGS-based nsp1 expression plasmids described above by using TransIT-293 reagent (Mirus). At 20 h posttransfection, cell extracts were prepared. Consistent with the findings of our previous studies (9, 14), Western blot analysis showed efficient accumulation of SCoVnsp1-mt protein and low accumulation levels of SARS-CoV nsp1 protein (Fig. 2A). HKU9-1 nsp1 protein accumulated efficiently, yet to a slightly lower level than the SCoVnsp1-mt protein. The level of accumulation of 133 nsp1 protein was lower than that of the HKU9-1 nsp1 protein but higher than that of the Rm1 nsp1 protein. These data suggested that like SARS-CoV nsp1 protein, Rm1 and 133 nsp1 proteins suppressed their own gene expression.
FIG. 2.
Effects of expression of bat CoV nsp1 on reporter gene expression. 293 cells were independently cotransfected with pRL-SV40 and one of the following plasmids: pCAGGS (empty vector [EV]), pCAGGS-Nsp1-WT (WT), pCAGGS-Nsp1-mt (mt), pCAGGS-Rm1 (Rm1), pCAGGS-133 (133), or pCAGGS-HKU9-1 (HKU9-1). At 20 h posttransfection, cell extracts were prepared. (A) Western blot analysis using anti-myc monoclonal antibody (Upstate) and anti-β-actin antibody (Santa Cruz Biotechnology) demonstrated the accumulation of expressed nsp1 proteins and β-actin, respectively. The data are representative of results from three independent experiments. (B) RLuc activities were measured and represented as the average of results from three independent experiments. Activities are expressed in relative light units. (C) The abundance of expressed RLuc RNA transcripts from pRL-SV40 was examined by Northern blot analysis using an RLuc probe. The data are representative of results from three independent experiments. The abundance of 28S and 18S rRNAs in each sample is also shown.
A reporter assay using the cell samples lysed in RLuc lysis buffer (Promega) showed that the expression of SARS-CoV nsp1 protein, but not that of SCoVnsp1-mt protein, suppressed RLuc activity (Fig. 2B); these data were consistent with the results in our previous report (9, 14). Rm1 nsp1 protein inhibited RLuc activity as efficiently as SARS-CoV nsp1 protein, while 133 and HKU9-1 nsp1 proteins had relatively moderate suppressive activities (Fig. 2B). The abundance of RLuc mRNA was very low in cells expressing SARS-CoV nsp1 protein but not in those expressing SCoVnsp1-mt protein (Fig. 2C). Rm1 protein suppressed the accumulation of RLuc mRNA as efficiently as SARS-CoV nsp1 protein, while the reduction in the abundance of RLuc mRNA in cells expressing 133 or HKU9-1 nsp1 protein was less drastic. These data clearly showed that the three group 2 bat CoV nsp1 proteins suppressed reporter gene expression and reporter gene mRNA accumulation with different efficiencies.
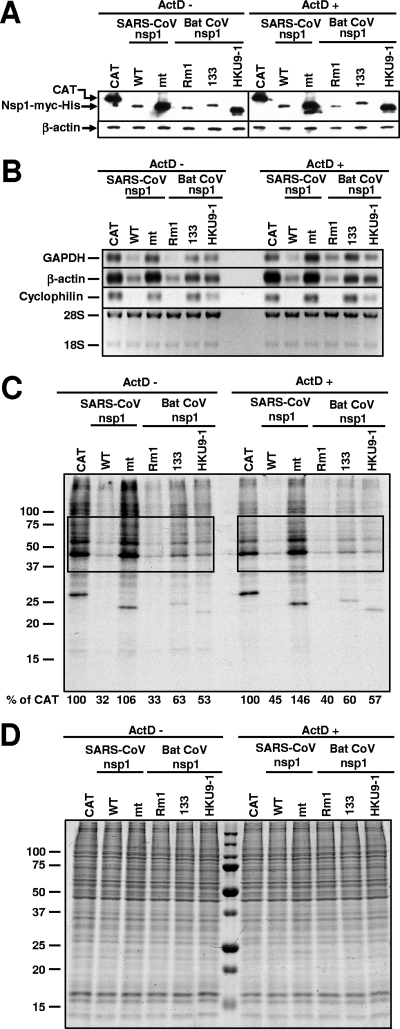
To know whether the group 2 bat CoV nsp1 proteins promoted host mRNA degradation, subconfluent 293 cells were transfected independently with one of six capped and polyadenylated RNA transcripts, each encoding Rm1 nsp1, 133 nsp1, HKU9-1 nsp1, SARS-CoV nsp1, SCoVnsp1-mt, or chloramphenicol acetyltransferase (CAT) protein, by using TransIT mRNA (Mirus); all of these proteins had a C-terminal myc tag. At 1 h posttransfection, the cells were incubated in the absence or presence of 4 μg/ml of actinomycin D (ActD; Sigma), which blocks new RNA synthesis and facilitates the analysis of the fate of preexisting cellular mRNAs; this concentration of ActD severely blocks host RNA synthesis (9). If the expression of bat CoV nsp1 proteins resulted in a reduction in the abundance of host endogenous mRNAs in the presence of ActD, then the data would suggest that the expression of the bat nsp1 proteins promotes host mRNA degradation. Similar to the plasmid transfection experiments (Fig. 2A), these RNA transfection assays revealed that the expression levels of SARS-CoV nsp1, Rm1 nsp1, and 133 nsp1 proteins were significantly lower than those of CAT, SCoVnsp1-mt, and HKU9-1 nsp1 proteins (Fig. 3A). ActD treatment had little effect on the abundance of endogenous glyceraldehyde-3-phosphate dehydrogenase (GAPDH), β-actin, and cyclophilin mRNAs in the cells expressing CAT protein (Fig. 3B). These results were not surprising because these mRNAs have long half-lives, due to which ActD treatment for less than 10 h often has a minimal effect on their abundance (1, 3, 12, 17, 25). The levels of these endogenous mRNAs in SARS-CoV nsp1-expressing cells and Rm1 nsp1-expressing cells were significantly lower than those in other samples, both in the presence and in the absence of ActD (Fig. 3B). The expression of 133 and HKU9-1 nsp1 proteins induced a moderate reduction in β-actin mRNA levels, and the abundance of GAPDH and cyclophilin mRNAs in the cells expressing HKU9-1 nsp1 protein was lower than that in cells expressing CAT or SCoVnsp1-mt protein; ActD treatment did not alter these trends. In the absence of ActD, 133 nsp1 expression had little effect on the abundance of cyclophilin mRNA yet it moderately suppressed GAPDH mRNA accumulation. We have repeated these Northern blot analyses at least three times and obtained similar results. These data strongly suggested that the expressed Rm1 nsp1 promoted the degradation of endogenous mRNAs as efficiently as expressed SARS-CoV nsp1, whereas the expressed 133 and HKU9-1 nsp1 proteins had less prominent activities to promote the degradation of these host mRNAs.
FIG. 3.
Effect of bat CoV nsp1 expression on host mRNA stability and protein synthesis. 293 cells were independently transfected with in vitro-synthesized RNA transcripts encoding CAT (CAT), SARS-CoV nsp1 (WT), SCoVnsp1-mt (mt), Rm1 nsp1 (Rm1), 133 nsp1 (133), or HKU9-1 nsp1 (HKU9-1) protein. One hour after RNA transfection, cells were incubated in the absence of ActD (ActD −) or the presence of ActD (ActD +). (A) At 8 h posttransfection, total proteins were extracted. Western blot analysis was performed using anti-myc or anti-β-actin antibodies. (B) Total RNAs were extracted at 8 h posttransfection and used for Northern blot analysis with riboprobes specific for GAPDH, β-actin, and cyclophilin. The abundance of 28S and 18S rRNAs is shown at the bottom. (C and D) Cells were radiolabeled with 20 μCi/ml of [35S]methionine from 6.5 to 7 h after the ActD addition. Equivalent amounts of intracellular proteins were analyzed by sodium dodecyl sulfate-12.5% polyacrylamide gel electrophoresis. (C) The gel was exposed to X-ray film. Phosphorimager analysis was performed to determine the level of host protein synthesis, and the numbers below the lanes represent the percentages of radioactivity relative to that for cells transfected with CAT RNA (% of CAT). The boxes indicate the regions of the gel used for phosphorimager analysis. Representative data from two independent experiments are shown. (D) The gel was stained with colloid Coomassie blue.
Next, the effect of bat CoV nsp1 expression on host protein synthesis was examined. 293 cells were independently transfected with one of the six RNA transcripts as described in the legend to Fig. 3A. The cells were either treated with ActD at 1 h posttransfection or left untreated and radiolabeled with [35S]methionine from 6.5 to 7 h after the ActD addition. After radiolabeling, the cell extracts were prepared and analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. The amounts of radioactivity in the selected regions of the gel were determined by using a Storm 860 PhosphorImager (Molecular Dynamics). As expected, SARS-CoV nsp1 protein expression, but not SCoVnsp1-mt expression, strongly inhibited host protein synthesis (Fig. 3C). The expression of Rm1, 133, and HKU9-1 nsp1 proteins also suppressed host protein synthesis (Fig. 3C). The amounts of radiolabeled proteins in SARS-CoV nsp1-expressing cells and in Rm1 nsp1-expressing cells were lower than those in cells expressing 133 nsp1 protein or HKU9-1 nsp1 protein (Fig. 3C). Colloid Coomassie blue staining of the gel confirmed the similar loading amounts of proteins in the gel (Fig. 3D). These data revealed that the Rm1, 133, and HKU9-1 nsp1 proteins efficiently inhibited host protein synthesis and also highlighted a difference in the levels of host translational suppression activity among the group 2 bat CoV nsp1 proteins.
The data presented above led us to hypothesize that the group 2 bat CoV nsp1 proteins also inhibit the expression of genes that are involved in host innate immune functions in infected cells. We tested this hypothesis by evaluating the effects of group 2 bat CoV nsp1 protein expression on the induction of beta interferon (IFN-β) and two IFN-stimulated genes (ISGs), ISG15 and ISG56, in the cells infected with the SARS-CoV nsp1 mutant (SARS-CoV-mt), which induces high levels of IFN-β and ISG proteins in infected 293/ACE2 cells (14); the SARS-CoV-mt nsp1 gene and the SCoVnsp1-mt gene have the same mutation (14). We speculated that host cells probably use the same pattern recognition receptors (16) to recognize unidentified pathogen-associated molecular patterns of replicating SARS-CoV and group 2 bat CoVs and trigger the signaling pathways that lead to type I IFN and ISG induction. If nsp1 proteins of the group 2 bat CoVs suppress the induction of IFN-β and ISGs in SARS-CoV-mt-infected cells, then it is reasonable to extrapolate these results and suggest that the proteins possess the activity to suppress the expression of the IFN-β gene and ISGs in bat CoV-infected cells.
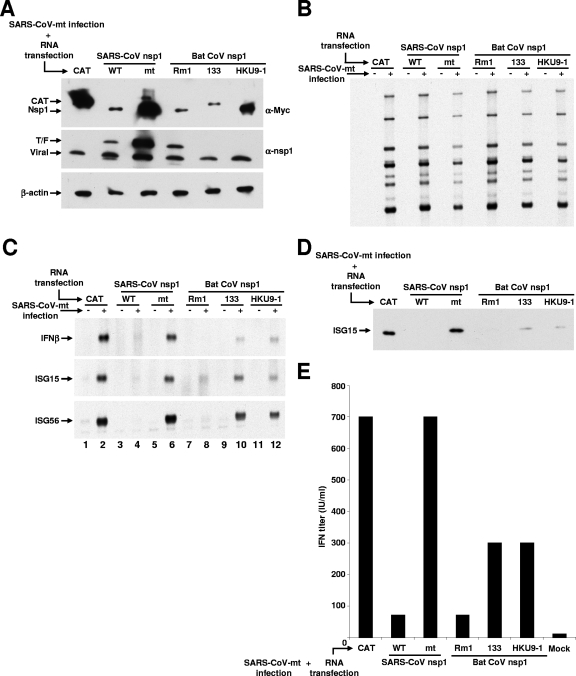
293/ACE2 cells (9) were either mock infected or infected with SARS-CoV-mt (14). One hour after infection, cells were transfected with capped and polyadenylated RNA transcripts encoding Rm1 nsp1, 133 nsp1, HKU9-1 nsp1, SARS-CoV nsp1, SCoVnsp1-mt, or CAT. The total intracellular RNAs and proteins were extracted at 18 h postinfection (h p.i.), while supernatants were collected at 43 h p.i. to assay the production of biologically active IFN-I. The amounts of the expressed SARS-CoV, Rm1, and 133 nsp1 proteins were significantly lower than those of expressed CAT protein or SCoVnsp1-mt and HKU9-1 nsp1 proteins (Fig. 4A, top panel). These results, which were similar to the data shown in Fig. 3A, suggested that the virally encoded SCoVnsp1-mt protein did not alter the nsp1 expression profile in cells transfected with RNA transcripts and that the differences in the abundance of the expressed nsp1 proteins probably reflected the activity of each nsp1 to suppress its own expression. The abundance of virally encoded SCoVnsp1-mt protein and that of β-actin were similar in all the samples (Fig. 4A, middle and bottom panels). Anti-nsp1 antibody detected the myc-tagged SARS-CoV nsp1 protein and SCoVnsp1-mt protein as expected (Fig. 4A, middle panel), while it also recognized Rm1 nsp1 protein (Fig. 4A, middle panel). Efficient viral mRNA accumulation occurred in all the samples, yet for an unknown reason, the abundance of viral mRNAs was slightly lower in the cells expressing SCoVnsp1-mt, 133 nsp1, or HKU9-1 nsp1 than in those expressing CAT, SARS-CoV nsp1, or Rm1 nsp1 (Fig. 4B). IFN-β, ISG15, and ISG56 mRNAs were not detected in the mock-infected cells transfected with RNA transcripts (Fig. 4C, odd-numbered lanes), whereas these mRNAs accumulated in the SARS-CoV-mt-infected cells transfected with the CAT RNA transcript (Fig. 4C, lane 2), demonstrating that SARS-CoV-mt replication, but not transfection with RNA transcripts, induced the accumulation of IFN-β, ISG15, and ISG56 mRNAs. SARS-CoV nsp1 expression efficiently suppressed the accumulation of IFN-β, ISG15, and ISG56 mRNAs (Fig. 4C, lane 4). These data suggested that the efficiencies of RNA transfection in 293/ACE2 cells were high and also reinforced our previous finding that SARS-CoV nsp1 plays a critical role in the inhibition of the expression of innate immune response genes (14). The expression of SCoVnsp1-mt protein did not prevent the accumulation of IFN-β, ISG15, and ISG56 mRNAs (Fig. 4C, lane 6). Expressed Rm1 nsp1 protein suppressed the accumulation of IFN-β, ISG15, and ISG56 mRNAs as efficiently as SARS-CoV nsp1 protein (Fig. 4C, lane 8), while expressed 133 and HKU9-1 nsp1 proteins showed moderate activities to suppress the accumulation of these mRNAs. ISG15 protein accumulated efficiently in SARS-CoV-mt-infected cells expressing CAT protein or SCoVnsp1-mt protein but not in those expressing SARS-CoV nsp1 or Rm1 nsp1 (Fig. 4D). Low levels of ISG15 protein accumulation in SARS-CoV-mt-infected cells expressing 133 or HKU9-1 nsp1 protein were found. Overall, the abundance profile of the ISG15 protein roughly correlated with those of ISG15 mRNAs. A biological assay of secreted IFN-I showed significant production of IFN-I from SARS-CoV-mt-infected cells expressing CAT protein or SCoVnsp1-mt protein, whereas the titers of IFN-I in infected cells expressing Rm1 nsp1 or SARS-CoV nsp1 protein were very low (Fig. 4E). The levels of IFN-I produced from infected cells expressing 133 and HKU9-1 nsp1 proteins were lower than those produced from CAT or SCoVnsp1-mt protein-expressing cells and higher than those produced from SARS-CoV nsp1 or Rm1 nsp1 protein-expressing cells (Fig. 4E). The profile of IFN-I production roughly correlated with the levels of IFN-β mRNA accumulation, as shown in Fig. 4C. These data clearly demonstrated that the expressed group 2 bat CoV nsp1 proteins and SARS-CoV nsp1 protein were able to function in trans to inhibit the upregulation of IFN-I and ISGs in SARS-CoV-mt-infected cells.
FIG. 4.
Effect of bat CoV nsp1 expression on IFN-I and ISG induction in SARS-CoV-mt-infected cells. 293/ACE2 cells were either mock infected or infected with SARS-CoV-mt at a multiplicity of infection of 3. One hour after infection, cells were independently transfected with RNA transcripts encoding CAT (CAT), SARS-CoV nsp1 (WT), SCoVnsp1-mt (mt), Rm1 nsp1 (Rm1), 133 nsp1 (133), or HKU9-1 nsp1 (HKU9-1) protein. At 18 h p.i., total intracellular proteins and RNAs were extracted. (A) Western blot analysis was performed using anti-myc (α-myc), anti-nsp1, and anti-β-actin antibodies to demonstrate the accumulations of nsp1 proteins expressed from the RNA transcripts in transfected cells, the virally encoded nsp1-mt protein, and the host β-actin protein, respectively. (B) The viral mRNAs were detected by Northern blot analysis using a probe that binds to the 3′ end of the SARS-CoV genome (nucleotides 29,084 to 29,608) (14). +, infection; −, mock infection. (C) The accumulation of endogenous IFN-β, ISG15, and ISG56 mRNAs was evaluated by Northern blot analysis using riboprobes specific for the IFN-β gene, ISG15, and ISG56, respectively. (D) Western blot analysis was performed using anti-ISG15 protein polyclonal antibody (Cell Signaling Technology) to detect ISG15 protein. (E) At 43 h p.i., culture supernatants were collected from mock-infected cells (mock) and the SARS-CoV-mt-infected cells that were transfected with the indicated RNA transcripts. After complete inactivation of viruses by 60Co irradiation (2 × 106 rad) (8), the samples were subjected to acid treatment (14). After neutralization of the samples, the human IFN-I activity was measured by a standard plaque reduction assay using Sindbis virus on Vero cells (5). The units of IFN are expressed as the reciprocal of the dilution that inhibited the formation of 50% of the viral plaques. The data are the averages of results from two independent experiments. IU, international units.
The amino acid sequence of SARS-CoV nsp1 is highly conserved, and only one nonconservative amino acid variation (C82G) among nsp1 sequences of SARS-CoVs isolated from humans and civets is known (7), while nsp1 proteins of bat SLCoVs, which were isolated from four species of horseshoe bats (genus Rhinolophus) and phylogenetically categorized into group 2b with SARS-CoVs, differed from the SARS-CoV nsp1 protein by 3 to 14 amino acids in 31 positions (Fig. 1B). The sequence of Rm1 nsp1 has 14 amino acid changes, with a degree of sequence identity of 92.2%, compared with SARS-CoV nsp1. We noted that the amino acid sequences at the N-terminal regions of Rm1 nsp1 and the nsp1 protein of the group 2b bat CoV Rf1 are identical and that the amino acid sequences at the C-terminal regions of Rm1 nsp1 and the nsp1 protein of another group 2b bat CoV, 279, are identical (Fig. 1B). Also, the primary sequence of Rm1, downstream of the nsp1 gene, is similar to that of 279 (21). These data suggest that homologous RNA recombination within the nsp1 gene occurred between Rf1-like and 279-like SLCoVs to generate Rm1 or its progenitor virus. Because Rm1, Rf1, and 279 replicate in the horseshoe bat species Rhinolophus macrotis, R. ferrumequinum, and R. macrotis, respectively, in Hubei province, China (13, 20), the putative homologous RNA recombination event that generated Rm1 may have occurred following the interspecies transmission of Rf1-like and 279-like SLCoVs between R. macrotis and R. ferrumequinum.
While a previous report (26) and the findings of the present study have suggested that group 2 CoV nsp1 proteins, despite their low levels of primary sequence homology, share a common biological function to inhibit host gene expression and modulate host innate immune responses, our studies also highlighted the differences in their biological activities. While Rm1 and SARS-CoV nsp1 proteins exhibited strong host mRNA degradation activities, the 133 and HKU9-1 nsp1 proteins did not promote extensive host mRNA degradation (Fig. 3B), suggesting that the mechanism of action of group 2b bat and SARS-CoV nsp1 proteins may be different from that of other group 2 bat CoV nsp1 proteins. We also noted that while the abundance of the expressed HKU9-1 nsp1 protein was clearly higher than that of the 133 nsp1 protein (Fig. 2A and 3A), the expression of HKU9-1 nsp1 protein and that of 133 nsp1 protein resulted in similar levels of host protein synthesis inhibition (Fig. 3C). Poor accumulation of SARS-CoV, Rm1, and 133 nsp1 proteins in cells expressing these proteins (Fig. 2A and 3A) suggested that these nsp1 proteins suppressed their own gene expression, while the expression of HKU9-1 nsp1 had little effect on its own gene expression, suggesting that the mechanisms of host gene expression inhibition mediated by HKU9-1 nsp1 also differed from those mediated by the 133 nsp1 protein.
Assuming that 133 and HKU9-1 also efficiently suppress host innate immune responses like SARS-CoV does (9, 14), the finding that the nsp1 proteins of 133 and HKU9-1 had only moderate activities to suppress the expression of host innate immune response genes suggests that another viral gene(s) carried by these bat CoVs may play an important role in suppressing innate immune functions. In addition to nsp1 (9, 14, 22), past studies showed that nsp3 (4), ORF 3b (10), ORF 6 (6, 10), and N (10) proteins of SARS-CoV have activities to suppress the host innate immune functions. It is likely that one or more of the corresponding proteins and/or another protein(s) encoded in the 133 and HKU9-1 genomes complements the moderate suppressive activities of nsp1 proteins of these viruses to achieve efficient suppression of the host innate immune functions. It is also possible that the biological activities of nsp1 proteins of group 2 CoVs are influenced by the host cell types and species; nsp1 proteins of 133 and HKU9-1 may efficiently suppress host gene expression in certain types of bat-derived cells.
Acknowledgments
This work was supported by Public Health Service grant AI72493 from NIH. Y.T. was supported by a grant from the Ministry of Education, Culture, Sports, Science and Technology of Japan. W.K. was supported by the James W. McLaughlin Fellowship Fund.
Footnotes
Published ahead of print on 4 March 2009.
REFERENCES
- 1.Atasoy, U., S. L. Curry, I. López de Silances, A.-B. Shyu, V. Casolaro, M. Gorospe, and C. Stellato. 2003. Regulation of eotaxin gene expression by TNF-α and IL-4 through mRNA stabilization: involvement of the RNA-binding protein HuR. J. Immunol. 1714369-4378. [DOI] [PubMed] [Google Scholar]
- 2.Cheng, V. C. C., S. K. P. Lau, P. C. Y. Woo, and K. Y. Yuen. 2007. Severe acute respiratory syndrome coronavirus as an agent of emerging and reemerging infection. Clin. Microbiol. Rev. 20660-694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Condeelis, J., and R. H. Singer. 2005. How and why does β-actin mRNA target? Biol. Cell 9797-110. [DOI] [PubMed] [Google Scholar]
- 4.Devaraj, S. G., N. Wang, Z. Chen, Z. Chen, M. Tseng, N. Barretto, R. Lin, C. J. Peters, C.-T. K. Tseng, S. C. Bake, and K. Li. 2007. Regulation of IRF-3-dependent innate immunity by the papain-like protease domain of the severe acute respiratory syndrome coronavirus. J. Biol. Chem. 28232208-32221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ferreira, P. C. P., M. L. P. Peixoto, M. A. V. Silva, and R. R. Golgher. 1979. Assay of human interferon in Vero cells by several methods. J. Clin. Microbiol. 9471-475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Frieman, M., B. Yount, M. Heise, S. A. Kopecky-Bromberg, P. Palese, and R. S. Baric. 2007. Severe acute respiratory syndrome coronavirus ORF6 antagonizes STAT1 function by sequestering nuclear import factors on the rough endoplasmic reticulum/Golgi membrane. J. Virol. 819812-9824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Graham, R. L., J. S. Sparks, L. D. Eckerle, A. C. Sims, and M. R. Denison. 2008. SARS coronavirus replicase proteins in pathogenesis. Virus Res. 13388-100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ito, N., E. C. Mossel, K. Narayanan, V. L. Popov, C. Huang, T. Inoue, C. J. Peters, and S. Makino. 2005. Severe acute respiratory syndrome coronavirus 3a protein is a viral structural protein. J. Virol. 793182-3186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kamitani, W., K. Narayanan, C. Huang, K. Lokugamage, T. Ikegami, N. Ito, H. Kubo, and S. Makino. 2006. Severe acute respiratory syndrome coronavirus nsp1 protein suppress host gene expression by promoting host mRNA degradation. Proc. Natl. Acad. Sci. USA 10312885-12890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kopecky-Bromberg, S. A., L. Martínez-Sobrino, M. Frieman, R. A. Baric, and P. Palese. 2007. Severe acute respiratory syndrome coronavirus open reading frame (ORF) 3b, ORF6, and nucleocapsid proteins function as interferon antagonists. J. Virol. 81548-557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lau, S. K. P., P. C. Y. Woo, K. S. M. Li, Y. Huang, H.-W. Tsoi, B. H. L. Wong, S. S. Y. Wong, S.-Y. Leung, K.-H. Chan, and K.-Y. Yuen. 2005. Severe acute respiratory syndrome coronavirus-like virus in Chinese horseshoe bats. Proc. Natl. Acad. Sci. USA 10214040-14045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Leclerc, G. J., G. M. Leclerc, and J. C. Barredo. 2002. Real-time RT-PCR analysis of mRNA decay: half-life of beta-actin mRNA in human leukemia CCRF-CEM and Nalm-6 cell lines. Cancer Cell Int. 21-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li, W., Z. Shi, M. Yu, W. Ren, C. Smith, J. H. Epstein, H. Wang, G. Crameri, Z. Hu, H. Zhang, J. Zhang, J. McEachern, H. Field, P. Daszak, B. T. Eaton, S. Zhang, and L.-F. Wang. 2005. Bats are natural reservoirs of SARS-like coronaviruses. Science 310676-679. [DOI] [PubMed] [Google Scholar]
- 14.Narayanan, K., C. Huang, K. Lokugamage, W. Kamitani, T. Ikegami, C.-T. K. Tseng, and S. Makino. 2008. Severe acute respiratory syndrome coronavirus nsp1 suppresses host gene expression, including that of type I interferon, in infected cells. J. Virol. 824471-4479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ren, W., W. Li, M. Yu, P. Hao, Y. Zhang, P. Zhou, S. Zhang, G. Zhao, Y. Zhong, S. Wang, L.-F. Wang, and Z. Shi. 2006. Full-length genome sequences of two SARS-like coronaviruses in horseshoe bats and genetic variation analysis. J. Gen. Virol. 873355-3359. [DOI] [PubMed] [Google Scholar]
- 16.Saito, T., and M. Gale, Jr. 2007. Principles of intracellular viral recognition. Curr. Opin. Immunol. 1917-23. [DOI] [PubMed] [Google Scholar]
- 17.Shih, N.-Y., and G. Floyd-Smith. 1996. Protein kinase C-δ mRNA is down-regulated transcriptionally and post-transcriptionally by 12-O-tetradecanoylphorbol-13-acetate. J. Biol. Chem. 27116040-16046. [DOI] [PubMed] [Google Scholar]
- 18.Snijder, E. J., P. J. Bredenbeek, J. C. Dobbe, V. Thiel, J. Ziebuhr, L. L. M. Poon, Y. Guan, M. Rozanov, W. J. M. Spaan, and A. E. Gorbalenya. 2003. Unique and conserved features of genome and proteome of SARS-coronavirus, an early split-off from the coronavirus group 2 lineage. J. Mol. Biol. 331991-1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stadler, K., V. Masignani, M. Eickmann, S. Becker, S. Abrignani, H.-D. Klenk, and R. Rappuoli. 2003. SARS—beginning to understand a new virus. Nat. Rev. Microbiol. 1209-218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tang, X. C., J. X. Zhang, S. Y. Zhang, P. Wang, X. H. Fan, L. F. Li, G. Li, B. Q. Dong, W. Liu, C. L. Cheung, K. M. Xu, W. J. Song, D. Vijaykrishna, L. L. M. Poon, J. S. M. Peiris, G. J. D. Smith, H. Chen, and Y. Guan. 2006. Prevalence and genetic diversity of coronaviruses in bats from China. J. Virol. 807481-7490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vijaykrishna, D., G. J. D. Smith, J. X. Zhang, J. S. M. Peiris, H. Chen, and Y. Guan. 2007. Evolutionary insights into the ecology of coronaviruses. J. Virol. 814012-4020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wathelet, M. G., M. Orr, M. B. Frieman, and R. S. Baric. 2007. Severe acute respiratory syndrome coronavirus evades antiviral signaling: role of nsp1 and rational design of an attenuated strain. J. Virol. 8111620-11633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Woo, P. C. Y., S. K. P. Lau, K. S. M. Li, R. W. S. Poon, B. H. L. Wong, H.-W. Tsoi, B. C. K. Yip, Y. Huang, K.-H. Chan, and K.-Y. Yuen. 2006. Molecular diversity of coronaviruses in bats. Virology 351180-187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Woo, P. C. Y., M. Wang, S. K. P. Lau, H. Xu, R. W. S. Poon, R. Guo, B. H. L. Wong, K. Gao, H.-W. Tsoi, Y. Huang, K. S. M. Li, C. S. F. Lam, K.-H. Chan, B.-J. Zheng, and K.-Y. Yuen. 2007. Comparative analysis of twelve genomes of three novel group 2c and group 2d coronaviruses reveals unique group and subgroup features. J. Virol. 811574-1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zelenaia, O. A., and M. B. Robinson. 2000. Degradation of glial glutamate transporter mRNA is selectively blocked by inhibition of cellular transcription. J. Neurochem. 752252-2258. [DOI] [PubMed] [Google Scholar]
- 26.Züst, R., L. Cervantes-Barrangán, T. Kuri, G. Blakqori, F. Weber, B. Ludewig, and V. Thiel. 2007. Coronavirus non-structural protein 1 is a major pathogenicity factor: implications for the rational design of coronavirus vaccines. PloS Pathog. 31062-1072. [DOI] [PMC free article] [PubMed] [Google Scholar]