Abstract
Alzheimer’s disease (AD) is incredibly common. Increasing longevity ensures its prevalence will rise even further. Ongoing efforts to understand AD pathogenesis reveal numerous tantalizing leads. Formulating a comprehensive AD pathogenesis theory capable of incorporating these disparate leads, though, has proven difficult. This review discusses current attempts to formulate a comprehensive AD pathogenesis theory. In doing so, it focuses on clinical and molecular relationships between AD and aging. A better understanding of these relationships could inform and impact future development of AD-directed treatment strategies.
Keywords: Alzheimer’s disease, aging, amyloid, cascade, mitochondria
Introduction
Alzheimer’s disease (AD) is the most common disease of aging. Countless clinicians and scientists have dedicated careers to its investigation. For some time the field has been too large to comprehensively summarize. While this review broadly discusses both molecular and clinical AD-relevant themes, it considers these themes within the narrower context of aging. By focusing in this way, it hopes to provide a better understanding of aging-AD relationships. Considering AD as a disease of aging provides perspective on recent therapeutic development efforts, which are discussed later on. First, though, it is necessary to define AD and describe why it is so intimately related to aging. While doing so, it is necessary to consider which is more appropriate – to consider AD a condition of aging, or to consider it as a distinct disease entity. Although this argument is largely a semantic one, it does have important implications for future AD research.
What is Alzheimer’s disease?
This review will first consider the term “Alzheimer’s disease”. This is a worthwhile endeavor because AD perspectives abound. Affected patients and their family members may not distinguish AD from the generic dementia clinical syndrome. Physicians not specializing in AD often don’t go much further, using it as a default diagnosis for persons dementing despite negative diagnostic tests. To AD sub-specialists able to identify and categorize different patterns of cognitive decline, AD is less a diagnosis of exclusion and more a recognizable entity. AD sub-specialists can predict with reasonable accuracy the histopathologic features existing within the brains of those with an AD-consistent clinical syndrome (Gearing et al 1995). However, the pathogenic relevance of these histopathologic features is itself unclear.
The first defined histopathologic features of AD were extracellular amyloid plaques and intracellular neurofibrillary tangles. More recently recognized histopathologic features include synaptic degeneration, hippocampal neuronal loss, and aneuploidy. AD histopathologic criteria, though, currently take into account only plaques and tangles. Several AD histopathologic criteria are commonly used (Khachaturian 1985; Mirra et al 1991; Consensus Criteria 1997). Approximately 90% of the time, when pathologists evaluate brains from persons diagnosed with AD by trained clinicians, enough plaques and tangles are found to meet histopathologic diagnostic standards (Gearing et al 1995). Interestingly, for some time pathologists have recognized non-demented elderly individuals will also qualify for a histopathologic diagnosis of AD (reviewed in Swerdlow 2006). This has caused fundamental problems for the AD field. If plaque-tangle quantity represents the AD “gold standard”, then clinical dementia becomes less relevant to the diagnosis.
De-emphasizing dementia as a required component raises several conceptual questions. First, how should one consider deceased individuals with plaque-laden brains but no dementia? The term “preclinical AD” is often used in these situations (Price et al 2001). Preclinical AD assumes histopathologic changes can precede clinical changes, and that if preclinical AD subjects lived longer dementia would manifest. Until recently this assumption has not been testable. The advent of in vivo plaque and plaque-tangle imaging may facilitate more critical assessment of this hypothesis. To date, such imaging has corroborated the pathology literature, in that non-demented elderly subjects often have plaque burdens equivalent to those with clinically diagnosed AD (Shoghi-Jadid et al 2002; Klunk et al 2004; Fagan et al 2006; Rentz et al 2006).
The second conceptual question relates to how many people either have AD or the potential to get AD. Certainly, how to address this question depends heavily on the definition used. If histopathology represents the gold standard, then up to 95% of those making it over 100 years of age have AD (Jicha et al 2005). One large survey of brains from persons over 85 found at least some degree of AD histopathology in all brains examined (Polvikoski et al 2005). A possible interpretation of this is that all individuals, should they live long enough, have the potential to develop AD. While this point is also not testable, it is minimally relevant. Rather, it is more important to consider whether in the most at-risk demographic groups it becomes more common to have AD than not to have AD. This does appear to be the case. Since approximately 75%–90% of centenarians have a dementia syndrome and 85%–95% meet at least minimal histopathologic criteria for AD (Mizutani and Shamada 1992; Thomassen et al 1998; Blansjaar et al 2000; Perls 2004; Jicha et al 2005), it seems reasonable to conclude centenarians are expected to have AD, and those who do not have it are the exception. Therefore, it is worth considering that at some point in the aging continuum AD ceases to become a disease because it becomes the norm.
The third conceptual question relates to the fact individuals commonly experience age-related revision of their cognitive skills. The neuropsychology literature suggests that upon reaching adulthood, insidious cognitive changes ensue (Snowdon et al 1996; Smyth et al 2004). These changes, which occur over decades, generally represent decline from previously higher levels of function as opposed to lateral shifts in cognitive strategies. While associated with at most subtle functional consequences not felt to be clinically relevant on an individual basis, it is worthwhile considering whether these changes do in fact represent part of an AD continuum. The relevance of this question is emphasized by the recently evolved concept of mild cognitive impairment (MCI) (Petersen et al 1999). MCI was originally intended to define a transitional state between normal cognition and dementia. Retrospective perspectives now suggest the majority of those with MCI are actually manifesting AD in its earliest recognizable clinical form (Morris 2006). The operational definition of MCI is similar to that of an older term, age-associated cognitive decline (AACD) (Levy 1994). AACD was formulated to classify those with “benign” cognitive changes. The main difference between AACD and MCI is therefore one of perspective. AACD was intended as an optimistic diagnosis, whereas MCI is considerably more pessimistic. Whether the physiology underlying age-associated cognitive changes is fundamentally different from that underlying AD-associated cognitive change remains unknown.
It is now common to lump under the AD umbrella all dementia syndromes manifesting plaque and tangle accumulation. This in turn has driven classification of various AD subtypes. Dementia of the Alzheimer’s Type (DAT) corresponds to the classic presenile form identified by Alois Alzheimer. Senile dementia of the Alzheimer’s Type (SDAT) includes individuals that until the 1970’s may have been assigned a diagnosis of senility, hardening of the arteries, or just getting older. Late-onset AD (LOAD) generally applies to those developing signs and symptoms after the age of 65. Early-onset AD variably refers to those developing signs or symptoms before the age of 55, 60, or 65. The exact upper age limit is therefore arbitrarily defined. Familial AD (FAD) loosely refers to those AD subjects with a positive AD family history, but without additional clarification this designation is confusing. The reason is that if the simple presence of a single affected relative is enough to qualify a patient for FAD, then FAD is incredibly common. If more stringent criteria are used, such as the presence of a recognizable Mendelian inheritance pattern, then FAD comprises an exceedingly small percentage (less than 1%) of the total number of AD cases. Most Mendelian FAD cases are autosomal dominant and have a presenile onset. Nevertheless, even among early-onset cases clear-cut autosomal dominant inheritance patterns are rare. Despite their rarity, the autosomal dominant FAD cases demonstrate genetic heterogeneity, as mutations in at least three different genes cause the phenotype (Table 1). Therefore, when considering AD pathogenesis, it is perhaps expedient to consider the fact that what we now consider AD actually appears to consist of multiple pathogenically different Alzheimer’s diseases.
Table 1.
Genes implicated in familial autosomal dominant AD
| Gene | Chromosome | Proportion of AD |
|---|---|---|
| Amyloid precursor protein | 21 | ~25 known families |
| Presenilin 1 | 14 | ~315 known families |
| Presenilin 2 | 1 | ~18 known families |
Disease of aging versus aging
Alois Alzheimer’s first “Alzheimer’s disease” patient, Auguste D., was reported in detail in 1907 (Alzheimer 1907). Auguste D. presented early in her sixth decade, and so could officially be considered a case of presenile dementia. On autopsy, her brain manifested the plaques and tangles that have since been associated with the AD condition. As no prior descriptions of plaque and tangle presenile dementia existed, the door was open for naming a new disease, which was shortly thereafter accomplished by Emil Kraepelin, the chairman of Alzheimer’s academic department (Kraepelin 1910). Simultaneously, Oscar Fischer (1907) at a rival institution recognized brains of elderly demented individuals also contained plaques. In subsequent years the medical community assimilated these observations so that the presenile tangle-and-plaque dementia was considered a disease, AD. The other situation (senile tangle-and-plaque dementia) was considered a normal part of aging, and for many decades hence syndromically referred to as senile dementia (Amaducci et al 1986; Boller and Forbes 1998). In the decades following this, AD remained a rather uncommon entity.
While life expectancy increased in westernized countries during the course of the twentieth century, senile dementia became increasingly prevalent. Interest in treating the demented elderly therefore grew, and investigators increasingly noted the similarities between AD and senile dementia. As Katzman pointed out in 1976, “neither the clinician, the neuropathologist, nor the electron microscopist can distinguish between the two disorders, except by the age of the patient” (Katzman 1976). It was further argued “Alzheimer disease and senile dementia are a single process and should therefore be considered a single disease” (Katzman 1976). This view justified making dementia syndrome research a national health priority and invigorated research into the phenomenon. Expanding the definition of AD to include those with senile dementia swelled the ranks of those diagnosed to such an extent it inextricably linked AD to aging. The incidence of AD rises with increasing age, so that among centenarians the histopathologic and clinical changes needed to justify a diagnosis of AD probably coexist in greater than 75% (Mizutani and Shamada 1992; Thomassen et al 1998; Blansjaar et al 2000; Perls 2004; Jicha et al 2005). One oft-quoted study concluded almost half those over age 85 have AD or at least an AD syndrome (Evans et al 1989).
Relevant to this debate are recent data suggesting AD may develop in individuals over the course of decades (Snowdon et al 1996; Smyth et al 2004). If this view is correct, then AD is a disease most people are in the process of developing throughout adulthood. From an epidemiologic standpoint, therefore, the debate about whether tangle-and-plaque dementia is a distinct disease or part of aging remains unresolved. An in-depth discussion of the aging-AD controversy has been reviewed elsewhere (Swerdlow 2006).
Does the histopathology drive the disease, or does the disease drive the histopathology?
With AD, the cause-consequence debate goes back to Alzheimer’s time. Alzheimer personally believed the histopathologic features he observed were a marker of an upstream process and not the root cause of the disease (Davis and Chisholm 1999). There are biochemical studies that support this view. For example, inhibition of the electron transport chain (ETC) enzyme cytochrome oxidase alters amyloid precursor protein (APP) processing to an amyloidogenic derivative (Gabuzda et al 1994). Oxidative stress activates beta secretase (BACE) activity, a requisite event in the processing of APP to Aβ (Tamagno et al 2002). Data such as these suggest amyloidosis in AD is a secondary event.
On the other hand, it is clear AD sometimes represents a primary amyloidosis. In 1991, APP gene mutation was shown to cause an early-onset, autosomal dominant AD variant (Goate et al 1991). Mutation of two other genes, presenilin 1 and presenilin 2, were subsequently found to also cause early-onset, autosomal dominant AD (Levy-Lahad et al 1995; Sherrington et al 1995). Functional studies revealed these mutations alter APP processing. In each case, Aβ42 to Aβ40 ratios increase (Scheuner et al 1996).
Whether most AD represents a primary or secondary amyloidosis goes to the heart of the AD pathogenesis debate. Two pathogenic AD hypotheses are discussed below, the amyloid cascade hypothesis (which assumes AD is always a primary amyloidosis), and the mitochondrial cascade hypothesis (which assumes most AD is a secondary amyloidosis).
The amyloid cascade hypothesis
The cortical plaques of AD brains largely consist of Aβ protein. Aβ is produced via processing of its parent protein, APP. The gene encoding APP resides on chromosome 21. Specific APP physiologic roles are not entirely clear, but in a general sense it is felt to contribute to proper neuronal function and perhaps cerebral development (Zheng et al 1995).
Because plaques contain Aβ and Down’s syndrome patients with trisomy 21 manifest both plaque pathology and presenile cognitive decline below their baseline, the role of the APP gene in AD was previously considered. In 1991, an APP gene mutation was uncovered in a family with autosomal dominant, early onset AD (Goate et al 1991). Soon after this, the amyloid cascade hypothesis was formulated. In its original form, the amyloid cascade hypothesis proposed altered APP processing drove Aβ production, Aβ gave rise to plaques, plaques induced neurodegeneration, and this neuronal loss resulted in the clinical dementia syndrome typical of AD (Hardy and Allsop 1991; Hardy and Higgins 1992).
While subsequent research failed to show APP mutation was a common cause of AD, the findings of other genetic and molecular research also lent support to the amyloid cascade hypothesis. Specifically, mutations in two other genes, presenilin 1 on chromosome 14 and presenilin 2 on chromosome 1, were also shown to cause variants of early onset, autosomal dominant AD (Levy-Lehad et al 1995; Sherrington et al 1995). While the functional role of presenilin proteins was unknown at the time of these discoveries, it was nevertheless found that as was the case with APP mutations, presenilin mutations enhanced production of the 42 amino acid APP C-terminal degradation product (Aβ42) at the expense of the 40 amino acid C-terminal APP C-terminal degradation product (Aβ40) (Scheuner et al 1996). Aβ42 is toxic to cells in culture, prone to aggregation, and found in plaques. More recent data suggest the presenilin gene products comprise part of the γ-secretase complex that is so intimately involved in APP processing (Wolfe et al 1999; Kimberly et al 2000).
The amyloid cascade hypothesis (Figure 1) has substantially evolved since its initial formulation. While Aβ sequestered in plaques was at first proposed to represent the critical toxic species, more recent versions of the hypothesis assume Aβ that is not sequestered in plaques actually drives the disease (Lesne and Kotilinek 2005; Walsh et al 2005). Even so, the amyloid cascade hypothesis seems most applicable in cases of early onset, autosomal dominant AD. Certainly, these cases are the ones most likely to represent a primary amyloidosis. Such cases comprise far less than 1% of AD, though, and it is not clear whether it is reasonable to etiologically extrapolate to the late-onset form (which afflicts the vast majority of those affected). Whether individuals with late-onset AD also carry genetic variations that promote a primary Aβ amyloidosis remains to be shown. If this turns out not to be the case, the possibility that amyloidosis in late-onset AD is secondary to a more upstream event will require consideration.
Figure 1.
The amyloid cascade hypothesis. A black box is shown in the middle of the figure, since mechanisms through which Aβ42 drives downstream pathology are not well defined.
Pursuit of the amyloid cascade hypothesis has yielded in depth insight into how APP processing actually occurs. Enzymes are capable of cutting APP in several places near its C-terminal end. One group of enzymes called α-secretases cut APP 83 amino acids from its carboxyl terminus. Enzymes with α-secretase activity include the ADAM (“A Disintegrin and Metalloproteinase”) protease family. The β-secretase enzymes consist of at least two different complexes named BACE1 and BACE2. The BACE enzymes cut APP 99 amino acids from the carboxyl terminus. The third type of APP cut is rendered by the γ-secretase enzyme complex. The catalytic function of γ-secretase is mediated by either presenilin 1 or presenilin 2. The γ-secretase cuts APP twice. One of these cuts occurs 50 amino acids from the APP carboxyl terminus. This generates a 50 amino acid peptide consisting of the extreme APP C-terminal end, which is titled the amyloid intracellular domain (AICD). The other γ-secretase cut is somewhat variable, but tends to occur 57, 59, or 61 amino acids from the APP C-terminus.
The APP secretases work in combination. Sequential processing by the α and γ-secretases results in a large N-terminal peptide called soluble APPα (sAPPα) and a smaller 3 kD peptide called P3. Proteolysis by enzymes with α-secretase activity precludes sequential β-γ secretase activity. When both the β and γ secretases process APP, the β-secretase cut produces a large N-terminal peptide called soluble APPβ (sAPPβ), as well as a smaller C-terminal fragment called CTFβ. The γ-secretase cuts CTFβ, and the more upstream (from the APP carboxyl terminus) 4 kD peptide defined by the γ and β secretases is the Aβ peptide. With sequential β-γ secretase activity the exact γ secretase cut site varies, yielding an Aβ peptide which is typically 38–43 amino acids.
Aβ degradation is also enzymatically mediated. Enzymes that degrade Aβ include neprilysin and insulin degrading enzyme (IDE). Interestingly, in the common non-autosomal dominant forms of AD Aβ overproduction is accompanied by IDE downregulation (Cook et al 2003). This suggests amyloidosis in AD is not a toxic accident, but rather part of a coordinated cell response to an upstream event. Defining this event could potentially drive further evolution of the amyloid cascade hypothesis. As it currently stands, the amyloid cascade hypothesis assumes all AD is a primary amyloidosis. This assumption is extrapolated from a handful of AD cases which almost certainly are primary amyloidoses. If an upstream event is shown to initiate AD amyloidosis, though, it would suggest amyloidosis in the vast majority of AD cases is a secondary event. Taking this argument one step further, some have even argued amyloidosis in AD not only represents a secondary event, but also a compensatory one (Castellani et al 2006).
Other clues to AD pathogenesis
Plaque-oriented research dominates the AD field. Much investigation, though, has focused on neurofibrillary tangles. These intracellular aggregations contain abnormally configured, excessively phosphorylated tau protein. In differentiated cells tau is normally unphosphorylated. It associates with microtubule cytoskeleton elements. This differs from undifferentiated cells, in which microtubules and tau do not form a permanent cytoskeleton and tau is phosphorylated. Mutations in the tau gene are associated with familial frontotemporal dementia, especially in cases with concomitant parkinsonism and tangle histopathology (Neary et al 2005). Thus, while primary tauopathy can drive neurodegeneration, it is not recognized to cause an AD phenotype.
Interestingly, AD brains exhibit neuronal cell cycle re-entry (Vincent et al 1996; McShea et al 1999; Arendt et al 2000; Yang et al 2001; Bowser and Smith 2002; Zhu et al 2004). Cell cycle re-entry refers to a phenomenon in which differentiated, non-dividing cells manifest molecular changes typically associated with cell division. These manifestations include an increase in cyclin-dependent kinase (CDK) activities and DNA content. Cell cycle re-entry produces aneuploid neuronal nuclei with replicated chromosomes. AD neurons can actually reach G2, the penultimate stage of the cell cycle that immediately precedes mitosis (M). Nevertheless, these neurons are unable to complete mitosis, which leaves them in a state called G2-M arrest.
CDK proteins also phoshorylate tau. It is tempting to speculate tau phosphorylation and cell cycle re-entry pathology are related. A mechanistically critical relationship between cell-cycle re-entry, oxidative stress, and neuronal demise was recently postulated (Zhu et al 2004).
As discussed above, AD and aging are epidemiologically intertwined. The strength of this relationship suggests these processes share mechanistic commonalities. Clinically, AD is not an all-or-nothing entity. It is a continuum. Mild, moderate, and severe stages are arbitrarily defined. Preceding AD proper is a recognizable transition phase, MCI. Neuropsychological testing now suggests MCI is itself preceded by a period of cognitive change (reviewed in Swerdlow 2006). Some call this period pre-MCI (Swerdlow 2006). Pre-MCI may last decades; some data suggest it starts early in adulthood (Snowdon et al 1996; Smyth et al 2004). Pre-MCI may provide a conceptual framework for “cognitive reserve” phenomena in AD (Stern 2006). Those with higher education levels, a cognitive reserve surrogate, are said to have reduced AD risk. It is heuristically reasoned high cognitive reserve individuals begin their decades-long cognitive decline further from the dementia finish line than low cognitive reserve individuals. Given equal pre-MCI cognitive decline trajectories, low cognitive reserve individuals will cross dementia thresholds before high cognitive reserve individuals. Pre-MCI decline trajectories, though, probably vary between individuals. A combination of genetic and environmental factors may influence decline trajectories. By this reasoning, those with the most cognitive reserve and slowest decline trajectories are more likely to develop clinical AD at older ages than those with less cognitive reserve and faster decline trajectories (Figure 2).
Figure 2.
Alzheimer’s disease dementia develops over decades. Those with more cognitive reserve and slower decline trajectories dement at the oldest ages. Those with less cognitive reserve and more rapid decline trajectories dement at younger ages.
Apolipoprotein E (APOE) alleles influence AD related-markers in ways potentially relevant to pre-MCI cognitive decline trajectories (Small et al 1995; Reiman et al 1996; Caselli et al 1999). The APOE gene on chromosome 19 encodes a protein, apolipoprotein E, which was previously recognized to play a role in cholesterol transport. Three APOE alleles are distributed throughout the population. Among these, the APOE4 allele associates with a younger age of AD onset than the APOE3 or APOE2 alleles (Corder et al 1993; Locke et al 1995; Blacker and Tanzi 1998). For perhaps this reason APOE4 is also associated with a greater lifetime AD risk. The underlying biochemical basis for this association is unknown, but hypotheses abound (Saunders 2000). The cysteine to arginine substitutions that define APOE4 may minimize any inherent ability of the protein to mitigate oxidative stress (Miyata and Smith 1996). Apolipoprotein E protein variations may affect cholesterol transport, thereby indirectly affecting amyloidosis (Poirier 2000). Direct effects on Aβ cerebrovascular transport might explain the association (DeMattos et al 2002; Zlokovic et al 2005). Apoliprotein E fragments also accumulate in mitochondria and affect mitochondrial function (Chang et al 2005). Apolipoprotein E4 produces more of these toxic fragments than apolipoprotein E3.
Mitochondrial dysfunction clearly exists in AD subjects (Parker et al 1990; reviewed in Swerdlow and Kish 2002). Structural and functional changes are evident. Mitochondrial pathology in AD is not limited to the brain. Mitochondria are implicated in aging, amyloidosis, oxidative stress, tau phosphorylation, and cell cycling. The protean manifestations of mitochondrial dysfunction and systemic nature of mitochondrial dysfunction in AD suggest to some a central pathogenic role for these organelles (Parker et al 1990; Swerdlow and Khan 2004).
The mitochondrial cascade hypothesis
The mitochondrial cascade hypothesis attempts a unified explanation for the clinical, biochemical, and histologic features of AD (Swerdlow and Khan 2004). The mitochondrial cascade hypothesis takes several conceptual liberties. It assumes similar physiologic mechanisms underlie AD and brain aging. It postulates because AD mitochondrial dysfunction is systemic, it cannot simply represent a consequence of neurodegeneration. The mitochondrial cascade hypothesis argues non-Mendelian genetic factors contribute to non-autosomal dominant AD. Finally, it posits AD brain mitochondrial dysfunction drives amyloidosis, tau phosphorylation, and cell cycle re-entry.
Certainly, mitochondria are indirectly featured in past theories of aging and directly in current aging theory. The rate of living hypothesis arose in the 1920’s from observations that animals with low metabolic rates tend to outlive those with high metabolic rates (Pearl 1928). Harman refined this when he proposed the free radical theory of aging in 1956 (Harman 1956). The free radical theory of aging specifically postulated over time cells accumulate structural damage from oxidative byproducts. By the 1970’s mitochondria were recognized sites of free radical production, and for many the free radical theory of aging morphed into the mitochondrial theory of aging (Harman 1972; Miquel et al 1980). The late 1980’s envisaged a role for somatic mitochondrial DNA (mtDNA) damage in aging (Linnane et al 1989; Wallace 1992). Corroborating this possibility are recent studies showing mtDNA mutation acquisition accelerates aging in laboratory animals (Trifunovic et al 2004; Kujoth et al 2005).
Mitochondrial dysfunction is observed in multiple AD tissues (reviewed in Swerdlow and Kish 2002). At least brain, platelet, and fibroblast mitochondria are involved. Defects of three mitochondrial enzymes are reported. This includes reduced activities of pyruvate dehydrogenase complex, alpha ketoglutarate dehydrogenase complex, and cytochrome oxidase (Gibson et al 1998). Spectral analysis of cytochrome oxidase indicates AD brains contain normal amounts of cytochrome oxidase, but the enzyme itself is structurally altered (Parker and Parks 1995). Various mechanisms, such as oxidative stress and proteasome dysfunction, have been postulated to facilitate mitochondrial dysfunction in neurodegenerative diseases such as AD (Ding et al 2006). Also, cytoplasmic hybrid (cybrid) studies indicate mtDNA at least in part accounts for reduced cytochrome oxidase activity in AD, and through this perhaps oxidative stress (reviewed in Swerdlow 2007).
Cybrid studies involve transfer of exogenous mtDNA to cultured cells depleted of endogenous mtDNA (reviewed in Khan et al 2006). “ρ” DNA is an alternative term for mtDNA. These mtDNA-depleted cells are therefore called ρ0 cells. They do not produce mtDNA-encoded proteins and lack cytochrome oxidase activity. Mitochondrial DNA transferred to ρ0 cells replicates and accomplishes mtDNA replacement. This enables expression of mtDNA-encoded ETC subunits and restoration of cytochrome oxidase activity. When cybrid lines containing mtDNA from AD subject platelets are compared to cybrid lines containing mtDNA from age-matched controls without AD, cytochrome oxidase activity is lower in the cybrid lines containing AD subject mtDNA (Swerdlow et al 1997; reviewed in Swerdlow 1997). Because nuclear genetic and cell culture conditions are equivalent between all cybrid cell lines, differences between mtDNA from the donor subjects likely account for the observed differences in cytochrome oxidase activity.
The exact nature of the implied AD cybrid-control cybrid mtDNA difference is unclear. This uncertainty is partly due to mtDNA-related complexities. Mitochondrial genetics and nuclear genetics differ in several key ways. One difference is that cells contain multiple copies of mtDNA. The mtDNA molecules within a cell can be identical, a state referred to as homoplasmy. However, the nucleotide sequences of mtDNA molecules within a cell can also vary. This is called heteroplasmy. If mtDNA sequence variation is present within a cell, it is necessary to consider whether it represents a homoplasmic or heteroplasmic variation. If the variation is heteroplasmic, it is necessary to further consider whether it represents a majority (high abundance heteroplasmy) or minority (low abundance heteroplasmy) of that cell’s mtDNA copies.
Distinct homoplasmic or high abundance heteroplasmic mutations of mtDNA cytochrome oxidase (CO) likely account for at most a very small percentage of AD (Hamblet et al 2006), and therefore should not account for AD-control cybrid differences. Mitochondrial DNA polymorphisms could potentially account for AD-control cybrid differences, as these are a common source of mtDNA inter-individual variability. Although AD-mtDNA polymorphism associations are reported (Chagnon et al 1999; reviewed in Swerdlow and Kish 2002; van der Walt et al 2004), the effect of specific polymorphisms or linked polymorphisms (haplogroups) on cybrid cytochrome oxidase activity is unstudied. Low abundance heteroplasmy might also cause AD-control cybrid cytochrome oxidase differences. A recent study did in fact suggest unique low abundance mtDNA heteroplasmies occur in AD (Coskun et al 2004). Other data indicate low abundance mtDNA heteroplasmies manifest with increasing age, and these heteroplasmies are associated with reduced cytochrome oxidase activity (Lin et al 2002). However, the presence and role of low abundance mtDNA heteroplasmies in AD cybrids has not been critically evaluated.
Whether somatic or inherited mtDNA features account for AD-control cybrid cytochrome oxidase differences also requires consideration. Data pertinent to this question permit speculation. First, complex I activity is normal in AD cybrids (Ghosh et al 1999). Complex I contains seven mtDNA-encoded subunits. Absence of complex I dysfunction suggests somatic mutation does not account for reduced cytochrome oxidase activity in AD cybrids, since acquired mtDNA somatic mutation should also reduce complex I activity. Second, reduction of cytochrome oxidase activity in multiple non-degenerating tissues is more consistent with mtDNA inheritance than somatic mutation. Third, plotting one AD cybrid study’s cytochrome oxidase data by mtDNA donor age actually reveals the youngest AD subjects show the biggest activity reductions (Figure 3). These data suggest mtDNA signatures might affect intrinsic cognitive decline trajectories. One interpretation of this is the lower the cytochrome oxidase activity, the sooner the subject reaches the dementia threshold. Epidemiologic data showing maternal AD status correlates better with offspring AD status than paternal AD status is consistent with this possibility (Edland et al 1996).
Figure 3.
Cytochrome oxidase activity may influence cognitive decline rates. Cytochrome oxidase activity was assayed in cybrid lines expressing mtDNA from 15 different AD subjects. The youngest AD mtDNA donors tended towards lower activies. Nine cybrid lines expressing mtDNA from control subjects indicate at most subtle age-related activity reductions.
Any comprehensive theory of AD pathogenesis must explain the different pathologies observed in Alzheimer’s disease. AD cybrids with reduced cytochrome oxidase activity overproduce Aβ42 and Aβ40 (Khan et al 2000). Under in vitro conditions sodium azide, a cytochrome oxidase inhibitor, alters APP processing towards amyloidogenic pathways (Gabuzda et al 1994). Administering sodium azide to mice also causes tau phosphorylation (Szabados et al 2004). Fibroblasts from FAD subjects phosphorylate tau following exposure to CCCP, a mitochondrial uncoupler (Blass et al 1990). Mitochondrial ETC dysfunction increases free radical production (reviewed in Swerdlow 2002). Enhanced reliance on anaerobic metabolism is associated with cell cycling (reviewed in Swerdlow and Khan 2004).
Interestingly, Aβ inhibits ETC activity in general and cytochrome oxidase activity specifically (Periera et al 1998; Casley et al 2002; Crouch et al 2005; Devi et al 2006). Therefore, a reciprocal relationship exists between mitochondrial function and amyloidosis. Several independent observations reinforce this concept. Neuronal-like NT2 human teratocarcinoma cells exposed to Aβ show high rates of demise. NT2 ρ0 cells, though, are impervious to Aβ (Cardoso et al 2001). The main difference between native NT2 cells and NT2 ρ0 cells is the absence of a functional ETC in the ρ0 cells. This suggests under cell culture conditions the mitochondrial ETC mediates Aβ toxicity. Also, APP, Aβ, and the entire γ-secretase complex are found either within mitochondria or mitochondrial membranes (Anandtheerthavarada et al 2003; Lustbader et al 2004; Hansson et al 2004; Crouch et al 2005; Manczak et al 2006). The role APP, Aβ, and γ-secretase play in mitochondrial function is currently unknown. Recent work showing oligomeric β-sheet proteins permeabilize membranes (Glabe and Kayed 2006) raises the possibility these proteins allow cells to disable mitochondria when certain conditions are met. If mitochondrial dysfunction is one of these conditions, one physiologic role of APP or Aβ may be to “shut down” abnormal mitochondria.
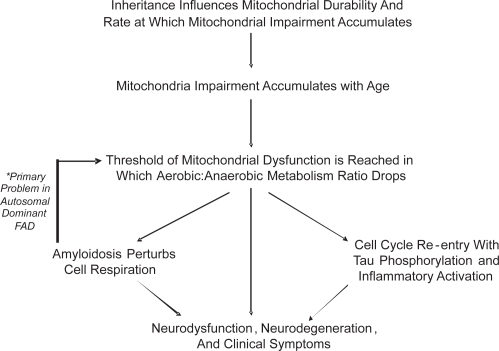
The mitochondrial cascade hypothesis takes aging phenomena into account, and applies to individuals long before they develop AD. The hypothesis postulates among individuals, inherited ETC differences influence mitochondrial durability. More durable mitochondria maintain adequate function longer than less durable mitochondria, and this in part determines aging success. Eventually, though, the balance between aerobic and anaerobic metabolism is not sustainable. At this point various cell responses are triggered. Cell cycle re-entry occurs. Tau phosphorylation ensues as neurons commit to de-differentiation. Amyloidosis facilitates this commitment by further altering aerobic metabolism. Ultimately, the neuronal de-differentiation response fails. Neurodegeneration results.
Just as the mitochondrial cascade hypothesis identifies with aging theory, links to the amyloid cascade hypothesis are postulated. In early onset, autosomal dominant AD altered APP or Aβ physiology initiate pathogenesis. If altered APP or Aβ physiology inadvertently induces mitochondrial dysfunction, it would trigger the same series of events as aging. Mitochondrial failure may therefore represent a final common pathway between autosomal dominant AD and non-autosomal dominant AD. Such would explain the clinical and histologic similarities observed between the different Alzheimer’s diseases (Figure 4).
Figure 4.
The mitochondrial cascade hypothesis.
Treatment development and clinical trials: Critical assessments of pathogenic hypotheses?
Current drug development strategies fall broadly into one of two categories. The first category includes treatments specifically designed to reduce the burden of brain Aβ. The second category includes all other strategies.
In order to reduce brain amyloid levels, approaches to both reducing Aβ production and enhancing its removal are undergoing evaluation. As discussed above, Aβ is produced through the β and γ-secretase mediated processing of APP. β-secretase inhibitors have gone to phase II human trials. Agents that specifically inhibit γ-secretase could prove problematic from a side effect perspective, as γ-secretase is also critical for processing Notch3, a protein of developmental importance and perhaps brain maintenance. Notch3 mutations cause another dementia syndrome, cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL).
Certain NSAIDS (ibuprofen and flurbiprofen) influence the γ-secretase but do not inhibit it outright. In general, these “selective amyloid lowering agents” (SALAs) alter where γ-secretase cuts the APP protein. Under in vitro conditions, ibuprofen and flurbiprofen reduce production of the Aβ42 APP derivative, with a secondary increase in the production of shorter Aβ fragments (Ericksen et al 2003). An enantiomer of flurbiprofen, R-flurbiprofen, recently completed a phase II human trial and is slated for a phase III efficacy trial.
Immunotherapy approaches have been studied as a way to enhance Aβ removal. The most extensive investigation involved AN1792, an Aβ-based vaccine (Aβ linked to an adjuvant). This vaccine first showed efficacy in transgenic mice that produce human Aβ. Such transgenic mice are engineered to express human APP transgenes containing mutations known to cause autosomal dominant, early-onset AD, and are widely used for preclinical drug screening in AD. In vaccinated mice, Aβ loads were reduced, there was preservation of cognitive abilities, and the vaccine was well tolerated (Schenk et al 1999). When phase I human studies did not reveal obvious toxicity, a phase II trial was initiated. This trial was prematurely halted after a substantial percentage of those mounting a robust immune response to the vaccine experienced encephalitis, a potentially life-threatening brain inflammation. However, there were over 40 individuals in this study who were vaccinated, developed a robust immune response, and did not develop encephalitis. Most of these subjects, as well as the trial’s placebo group, received ongoing clinical follow-up. One-year post-vaccination neuropsychological data on these subjects have been published (Gilman et al 2005). On no single endpoint was a statistical benefit shown. Subjects in both treatment and placebo groups continued to decline. For most of the individual endpoints, trends towards lesser decline in the vaccination group were identified, and on a z score analysis these trends were significant, suggesting it was possible the vaccine group was progressing slightly more slowly than the placebo group. Other data pertinent to the AN1792 study derive from autopsies of vaccinated subjects that subsequently died for various reasons. Brain histopathology from these deceased subjects demonstrated clear-cut reductions in brain parenchyma Aβ (Nicoll et al 2003; Ferrer et al 2004; Masliah et al 2005). To summarize the AN1792 experience from an efficacy perspective, available clinical data are inconclusive, but indicate over a one-year period activating the immune system to remove Aβ does not have a robust impact on cognition.
The next generation of amyloid clearance therapies are currently under development. These include modifications of the active immunization approach that will hopefully not trigger encephalitis. Passive immunization approaches via antibody infusions are also under investigation. The use of unique Aβ antibodies is being explored. The utility of treating AD subjects with intravenous immunoglobulin preparations, which naturally contain antibodies to Aβ, is being evaluated in an open label study (Relkin et al 2005).
The second category of AD treatment development includes efforts not specifically intended to reduce brain Aβ levels. For example, neuroscientists are unraveling intracellular pathways involved in cell information processing, and drugs that can modulate these pathways are under consideration. Drugs that retard neurofibrillary tangle formation in mice expressing mutant human tau transgenes, such as valproic acid, are being tested in humans. Although standard antioxidants to date have shown no-to-minimal evidence of therapeutic efficacy, new antioxidants that target specific free radical production sites are in preclinical development (Reddy 2006). The thiazolidinedione drugs rosiglitazone and pioglitazone, which reduce insulin resistance and which may also have anti-inflammatory effects, are undergoing testing in humans with AD (Risner et al 2006; Geldmacher et al 2006). One small open label trial has involved intracranial implantation of fibroblasts engineered to produce neurotrophic factors (Tuszynski et al 2005).
Obviously, successful development of new AD treatments depends on elucidating AD’s true underlying pathophysiology. If AD is a primary amyloidosis, as is postulated by the amyloid cascade hypothesis, then reducing Aβ would seem a rationale way to proceed with drug development. If AD is not a primary amyloidosis, then the impact anti-amyloid therapies have on the disease will be limited at best. Moreover, if AD is not a primary amyloidosis, the usefulness of Aβ-overproducing transgenic animals for preclinical drug testing is called into question. Finally, at genetic and epidemiologic levels it is now possible to define several different Alzheimer’s diseases. It is reasonable to consider whether treatments useful in one type of AD may not benefit patients with another type. In any case, Aβ-oriented treatment development will likely provide a practical assessment of the amyloid cascade hypothesis. If treatments that efficiently reduce Aβ production or remove Aβ fail to arrest disease progression, it would argue amyloidosis is not the primary pathology in most of those with AD.
Conclusion
Unlike many recent AD reviews, this one assumes critical questions remain about AD pathogenesis. As AD now stands, there are at least several different Alzheimer’s diseases. The rare autosomal dominant forms are primary amyloidoses. Amyloidosis in the common age-related forms may prove secondary. Until the causes of Aβ accumulation in age-related AD are revealed, it seems wise not to assume Aβ aggregation drives the disease, or that Aβ removal will cure it.
AD is now associated with increasing numbers of clinical, biochemical, and histologic markers. The fidelity of these associations, though, does not necessarily address pathogenesis. After all, it would be wrong to conclude serum troponin causes myocardial infarcts simply because levels are consistently elevated in myocardial infarct states. On the other hand, the more AD phenomena we recognize, the more likely it is the pieces of the AD puzzle will one day come together.
Footnotes
Grant information
Supported by the National Institute of Aging (AG022407).
References
- Alzheimer A. Uber eine eigenartige Erkrankung der Hirnrinde. Allg Z Psychiat Psych-Gerichtl Med. 1907;64:146–8. [Google Scholar]
- Amaducci LA, Rocca WA, Schoenberg BS. Origin of the distinction between Alzheimer’s disease and senile dementia: How history can clarify nosology. Neurology. 1986;36:1497–9. doi: 10.1212/wnl.36.11.1497. [DOI] [PubMed] [Google Scholar]
- Anandtheerthavarada HK, Biswas G, Robin MA, Avadhani MG. Mitochondrial targeting and a novel transmembrane arrest of Alzheimer’s precursor protein impairs mitochondrial function in neuronal cells. J Cell Biol. 2003;16:41–54. doi: 10.1083/jcb.200207030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arendt T. Alzheimer’s disease as a loss of differentiation control in a subset of neurons that retain immature features in the adult brain. Neurobiol Aging. 2000;21:783–96. doi: 10.1016/s0197-4580(00)00216-5. [DOI] [PubMed] [Google Scholar]
- Blacker D, Tanzi RE. The genetics of Alzheimer disease: Current status and future prospects. Arch Neurol. 1998;55:294–6. doi: 10.1001/archneur.55.3.294. [DOI] [PubMed] [Google Scholar]
- Blansjaar BA, Thomassen R, Van Schaick HW. Prevalence of dementia in centenarians. Int J Geriatr Psychiatry. 2000;15:219–25. doi: 10.1002/(sici)1099-1166(200003)15:3<219::aid-gps97>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- Blass JP, Baker AC, Ko L, Black RS. Induction of Alzheimer antigens by an uncoupler of oxidative phosphorylation. Arch Neurol. 1990;47:864–9. doi: 10.1001/archneur.1990.00530080046009. [DOI] [PubMed] [Google Scholar]
- Boller F, Forbes MM. History of dementia and dementia in history: An overview. J Neurol Sci. 1998;158:125–33. doi: 10.1016/s0022-510x(98)00128-2. [DOI] [PubMed] [Google Scholar]
- Bowser R, Smith MA. Cell cycle proteins in Alzheimer’s disease: Plenty of wheels but no cycle. J Alzheimers Dis. 2002;4:249–54. doi: 10.3233/jad-2002-4316. [DOI] [PubMed] [Google Scholar]
- Cardoso SM, Santos S, Swerdlow RH, Oliveira CR. Functional mitochondria are required for amyloid beta-mediated neurotoxicity. FASEB J. 2001;15:1439–41. doi: 10.1096/fj.00-0561fje. [DOI] [PubMed] [Google Scholar]
- Caselli RJ, Graff-Radford NR, Reiman EM, et al. Preclinical memory decline in cognitively normal apolipoprotein E-ɛ4 homozygotes. Neurology. 1999;53:201–7. doi: 10.1212/wnl.53.1.201. [DOI] [PubMed] [Google Scholar]
- Casley CS, Canevari L, Land JM, Clark JB, Sharpe MA. Beta-amyloid inhibits integrated mitochondrial respiration and key enzyme activities. J Neurochem. 2002;80:91–100. doi: 10.1046/j.0022-3042.2001.00681.x. [DOI] [PubMed] [Google Scholar]
- Castellani RJ, Lee HG, Zhu X, Nunomura A, Perry G, Smith MA. Neuropathology of Alzheimer disease: Pathognomonic but not pathogenic. Acta Neuropathol. 2006;111:503–9. doi: 10.1007/s00401-006-0071-y. [DOI] [PubMed] [Google Scholar]
- Chagnon P, Gee M, Filion M, et al. Phylogenetic analysis of the mitochondrial genome indicates significant differences between patients with Alzheimer disease and controls in a French-Canadian founder population. Am J Med Genet. 1999;85:20–30. doi: 10.1002/(sici)1096-8628(19990702)85:1<20::aid-ajmg6>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- Chang S, ran Ma T, Miranda RD, et al. Lipid- and receptor-binding regions of apolipoprotein E4 fragments act in concert to cause mitochondrial dysfunction and neurotoxicity. Proc Natl Acad Sci USA. 2005;102:18694–9. doi: 10.1073/pnas.0508254102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Consensus recommendations for the postmortem diagnosis of Alzheimer’s disease The national institute on aging, and reagan institute working group on diagnostic criteria for the neuropathological assessment of Alzheimer’s disease. Neurobiol Aging. 1997;18(4 Suppl):S1–2. [PubMed] [Google Scholar]
- Cook DG, Leverenz JB, McMillan PJ, et al. Reduced hippocampal insulin-degrading enzyme in late-onset Alzheimer’s disease is associated with the apolipoprotein E-epsilon4 allele. Am J Pathol. 2003;162:313–9. doi: 10.1016/s0002-9440(10)63822-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corder EH, Saunders AM, Strittmatter WJ, et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science. 1993;261:921–3. doi: 10.1126/science.8346443. [DOI] [PubMed] [Google Scholar]
- Coskun PE, Beal MF, Wallace DC. Alzheimer’s brains harbor somatic mtDNA control-region mutations that suppress mitochondrial transcription and replication. Proc Natl Acad Sci USA. 2004;101:10726–31. doi: 10.1073/pnas.0403649101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crouch PJ, Blake R, Duce JA, et al. Copper-dependent inhibition of human cytochrome c oxidase by a dimeric conformer of amyloid-beta1–42. J Neurosci. 2005;25:672–9. doi: 10.1523/JNEUROSCI.4276-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis JN, 2nd, Chisholm JC. Alois Alzheimer and the amyloid debate. Nature. 1999;26:810. doi: 10.1038/23571. [DOI] [PubMed] [Google Scholar]
- DeMattos RB, Bales KR, Parsadanian M, et al. Plaque-associated disruption of CSF and plasma amyloid-beta (Abeta) equilibrium in a mouse model of Alzheimer’s disease. J Neurochem. 2002;81:229–36. doi: 10.1046/j.1471-4159.2002.00889.x. [DOI] [PubMed] [Google Scholar]
- Devi L, Prabhu BM, Galati DF, Avadhani NG, Anandatheerthavarada HK. Accumulation of amyloid precursor protein in the mitochondrial import channels of human Alzheimer’s disease brain is associated with mitochondrial dysfunction. J Neurosci. 2006;26:9057–68. doi: 10.1523/JNEUROSCI.1469-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding Q, Dimayuga E, Keller JN. Proteasome regulation of oxidative stress in aging and age-related diseases of the CNS. Antioxid Redox Signal. 2006;8:163–72. doi: 10.1089/ars.2006.8.163. [DOI] [PubMed] [Google Scholar]
- Edland SD, Silverman JM, Peskind ER, et al. Increased risk of dementia in mothers of Alzheimer’s disease cases: Evidence for maternal inheritance. Neurology. 1996;47:254–6. doi: 10.1212/wnl.47.1.254. [DOI] [PubMed] [Google Scholar]
- Eriksen JL, Sagi SA, Smith TE, et al. NSAIDs and enantiomers of flurbiprofen target gamma-secretase and lower Abeta 42 in vivo. J Clin Invest. 2003;112:440–9. doi: 10.1172/JCI18162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans DA, Funkenstein HH, Albert MS, et al. Prevalence of Alzheimer’s disease in a community population of older persons. Higher than previously reported. JAMA. 1989;262:2551–6. [PubMed] [Google Scholar]
- Fagan AM, Mintun MA, Mach RH, et al. Inverse relation between in vivo amyloid imaging load and cerebrospinal fluid Abeta(42) in humans. Ann Neurol. 2006;59:512–9. doi: 10.1002/ana.20730. [DOI] [PubMed] [Google Scholar]
- Ferrer I, Boada Rovira M, Sanchez Guerra ML, Rey MJ, Costa-Jussa F. Neuropathology and pathogenesis of encephalitis following amyloid-beta immunization in Alzheimer’s disease. Brain Pathol. 2004;14:11–20. doi: 10.1111/j.1750-3639.2004.tb00493.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer O. Miliare Nekrosen mit drusigen Wucherungen der Neurofibrillen, eine regelmabige Veranderung der Hirnrinde bei seniler Demenz. Monatsschr Psychiatr Neurol. 1907;22:361–72. [Google Scholar]
- Gabuzda D, Busciglio J, Chen LB, Matsudaira P, Yankner BA. Inhibition of energy metabolism alters the processing of amyloid precursor protein and induces a potentially amyloidogenic derivative. J Biol Chem. 1994;269:13623–8. [PubMed] [Google Scholar]
- Gearing M, Mirra SS, Hedreen JC, et al. The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part X. Neuropathology confirmation of the clinical diagnosis of Alzheimer’s disease. Neurology. 1995;45:461–6. doi: 10.1212/wnl.45.3.461. [DOI] [PubMed] [Google Scholar]
- Geldmacher DS, Fritsch T, McClendon MJ, Lerner AJ, Landreth GE.2006A double-blind, placebo controlled, 18 momth pilot study of the PPAR-gamma agonist pioglitazone in Alzheimer’s disease Program and abstracts of the 10th International Conference on Alzheimer’s Disease and Related Disorders, Abstract P2–408. [Google Scholar]
- Ghosh SS, Swerdlow RH, Miller SW, et al. Use of cytoplasmic hybrid cell lines for elucidating the role of mitochondrial dysfunction in Alzheimer’s disease and Parkinson’s disease. Ann N Y Acad Sci. 1999;893:176–91. doi: 10.1111/j.1749-6632.1999.tb07825.x. [DOI] [PubMed] [Google Scholar]
- Gibson GE, Sheu KF, Blass JP. Abnormalities of mitochondrial enzymes in Alzheimer’s disease. J Neural Transm. 1998;105:855–70. doi: 10.1007/s007020050099. [DOI] [PubMed] [Google Scholar]
- Gilman S, Koller M, Black RS, et al. Clinical effects of Abeta immunization (AN1792) in patients with AD in an interrupted trial. Neurology. 2005;64:1553–62. doi: 10.1212/01.WNL.0000159740.16984.3C. [DOI] [PubMed] [Google Scholar]
- Glabe CG, Kayed R. Common structure and toxic function of amyloid oligomers implies a common mechanism of pathogenesis. Neurology. 2006;66(2 Suppl 1):S74–8. doi: 10.1212/01.wnl.0000192103.24796.42. [DOI] [PubMed] [Google Scholar]
- Goate A, Chartier-Harlin MC, Mullan M, et al. Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature. 1991;349:704–6. doi: 10.1038/349704a0. [DOI] [PubMed] [Google Scholar]
- Hamblet NS, Ragland B, Ali M, et al. Mutations in mitochondrial-encoded cytochrome c-oxidase subunits I, II, and III genes detected in Alzheimer’s disease using single-strand conformation polymorphism. Electrophoresis. 27:398–408. doi: 10.1002/elps.200500420. [DOI] [PubMed] [Google Scholar]
- Hansson CA, Frykman S, Farmery MR, et al. Nicastrin, presenilin, APH-1, and PEN-2 form active gamma-secretase complexes in mitochondria. J Biol Chem. 2004;279:51654–60. doi: 10.1074/jbc.M404500200. [DOI] [PubMed] [Google Scholar]
- Hardy J, Allsop D. Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends Pharmacol Sci. 1991;12:383–8. doi: 10.1016/0165-6147(91)90609-v. [DOI] [PubMed] [Google Scholar]
- Hardy JA, Higgins GA. Alzheimer’s disease: the amyloid cascade hypothesis. Science. 1992;256:184–5. doi: 10.1126/science.1566067. [DOI] [PubMed] [Google Scholar]
- Harman D. Aging: a theory based on free radical and radiation chemistry. J Gerontol. 1956;11:298–300. doi: 10.1093/geronj/11.3.298. [DOI] [PubMed] [Google Scholar]
- Harman D. A biological clock: The mitochondria? J Am Ger Soc. 1972;20:145–7. doi: 10.1111/j.1532-5415.1972.tb00787.x. [DOI] [PubMed] [Google Scholar]
- Jicha GA, Parisi JE, Dickson DW, et al. Alzheimer and Lewy body pathology in centenarian case series. Neurology. 2005;6(Suppl 1):A275. [Google Scholar]
- Katzman R. The prevalence and malignancy of Alzheimer’s disease: a major killer. Arch Neurol. 1976;33:217–8. doi: 10.1001/archneur.1976.00500040001001. [DOI] [PubMed] [Google Scholar]
- Khachaturian ZS. Diagnosis of Alzheimer’s disease. Arch Neurol. 1985;42:1097–106. doi: 10.1001/archneur.1985.04060100083029. [DOI] [PubMed] [Google Scholar]
- Khan SM, Cassarino DS, Abramova NN, et al. Alzheimer’s disease cybrids replicate beta-amyloid abnormalities through cell death pathways. Ann Neurol. 2000;48:148–55. [PubMed] [Google Scholar]
- Khan SM, Smigrodzki RM, Swerdlow R.2006Cell and animal models of mtDNA biology: Progress and prospects Am J Physiol Cell Physiol August9[Epub ahead of print]. PMID 16899549. [DOI] [PubMed] [Google Scholar]
- Kimberly WT, Xia W, Rahmatic J, Wolfe MS, Selkoe DJ. The transmembrane aspartates in presenilin 1 and 2 are obligatory for gamma-wcretase activity and amyloid beta-protein generation. J Biol Chem. 2000;275:3173–8. doi: 10.1074/jbc.275.5.3173. [DOI] [PubMed] [Google Scholar]
- Klunk WE, Engler H, Nordberg A, et al. Imaging brain amyloid in Alzheimer’s disease with Pittsburgh Compound-B. Ann Neurol. 2004;55:306–19. doi: 10.1002/ana.20009. [DOI] [PubMed] [Google Scholar]
- Kraepelin E.Psychiatrie. 1910. Ein Lehrbuch fur Studierende und Artze. II. Band, Klinische Psychiatrie. Verlag Johann Ambrosius Barth, Leipzig.
- Kujoth GC, Hiona A, Pugh TD, et al. 309. Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging Science 309481–4. [DOI] [PubMed] [Google Scholar]
- Lesne S, Kotilinek L. Amyloid plaques and amyloid-beta oligomers: An ongoing debate. J Neurosci. 2005;25:9319–20. doi: 10.1523/JNEUROSCI.3246-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy R. Aging-associated cognitive decline. Working Party of the International Psychogeriatric Association in collaboration with the World Health Organization. Int Psychogeriatr. 1994;6:63–8. [PubMed] [Google Scholar]
- Levy-Lahad E, Wasco W, Poorkaj P, et al. Candidate gene for the chromosome 1 familial Alzheimer’s disease locus. Science. 1995;269:973–7. doi: 10.1126/science.7638622. [DOI] [PubMed] [Google Scholar]
- Lin MT, Simon DK, Ahn CH, Kim LM, Beal MF. High aggregate burden of somatic mtDNA point mutations in aging and Alzheimer’s disease brain. Hum Mol Genet. 2002;11:133–45. doi: 10.1093/hmg/11.2.133. [DOI] [PubMed] [Google Scholar]
- Linnane AW, Marzuki S, Ozawa T, Tanaka M. Mitochondrial DNA mutations as an important contributor to ageing and degenerative diseases. Lancet. 1989;1:642–5. doi: 10.1016/s0140-6736(89)92145-4. [DOI] [PubMed] [Google Scholar]
- Locke PA, Conneally PM, Tanzi RE, Gusella JF, Haines JL. Apolipoprotein E, survival in Alzheimer’s disease patients, and the competing risks of death and Alzheimer’s disease. Neurology. 1995;45:1323–8. doi: 10.1212/wnl.45.7.1323. [DOI] [PubMed] [Google Scholar]
- Lustbader JW, Cirilli M, Lin C, et al. ABAD directly links Abeta to mitochondrial toxicity in Alzheimer’s disease. Science. 2004;304:448–52. doi: 10.1126/science.1091230. [DOI] [PubMed] [Google Scholar]
- Manczak M, Anekonda TS, Henson E, et al. Mitochondria are a direct site of A beta accumulation in Alzheimer‘s disease neurons: implications for free radical generation and oxidative damage in disease progression. Hum Mol Genet. 2006;15:1437–49. doi: 10.1093/hmg/ddl066. [DOI] [PubMed] [Google Scholar]
- Masliah E, Hansen L, Adame A, et al. Abeta vaccination effects on plaque pathology in the absence of encephalitis in Alzheimer disease. Neurology. 2005;64:129–31. doi: 10.1212/01.WNL.0000148590.39911.DF. [DOI] [PubMed] [Google Scholar]
- McShea A, Wahl AF, Smith MA. Re-entry into the cell cycle: A mechanism for neurodegeneration in Alzheimer disease. Med Hypotheses. 1999;52:525–7. doi: 10.1054/mehy.1997.0680. [DOI] [PubMed] [Google Scholar]
- Miquel J, Economos AC, Fleming J, Johnson JE., Jr Mitochondrial role in cell aging. Exp Gerontol. 1980;15:575–91. doi: 10.1016/0531-5565(80)90010-8. [DOI] [PubMed] [Google Scholar]
- Mirra SS, Heyman A, McKeel D, et al. The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). II. Standardization of the neuropathological assessment of Alzheimer’s disease. Neurology. 1991;41:479–86. doi: 10.1212/wnl.41.4.479. [DOI] [PubMed] [Google Scholar]
- Miyata M, Smith JD. Apolipoprotein E allele-specific antioxidant activity and effects on cytotoxicity by oxidative insults and beta-amyloid peptides. Nat Genet. 1996;14:55–61. doi: 10.1038/ng0996-55. [DOI] [PubMed] [Google Scholar]
- Mizutani T, Shimada H. Neuropathological background of twenty-seven centenarian brains. J Neurol Sci. 1992;108:168–77. doi: 10.1016/0022-510x(92)90047-o. [DOI] [PubMed] [Google Scholar]
- Morris JC. Mild cognitive impairment is early-stage Alzheimer disease: Time to revise diagnostic criteria. Arch Neurol. 2006;63:15–6. doi: 10.1001/archneur.63.1.15. [DOI] [PubMed] [Google Scholar]
- Neary D, Snowden J, Mann D. Frontotemporal dementia. Lancet Neurol. 2005;4:771–80. doi: 10.1016/S1474-4422(05)70223-4. [DOI] [PubMed] [Google Scholar]
- Nicoll JA, Wilkinson D, Holmes C, et al. Neuropathology of human Alzheimer disease after immunization with amyloid-beta peptide: A case report. Nat Med. 2003;9:448–52. doi: 10.1038/nm840. [DOI] [PubMed] [Google Scholar]
- Parker WD, Jr, Filley CM, Parks JK. Cytochrome oxidase deficiency in Alzheimer’s disease. Neurology. 1990;40:1302–3. doi: 10.1212/wnl.40.8.1302. [DOI] [PubMed] [Google Scholar]
- Parker WD, Jr, Parks JK. Cytochrome c oxidase in Alzheimer’s disease brain: purification and characterization. Neurology. 1995;45:482–6. doi: 10.1212/wnl.45.3.482. [DOI] [PubMed] [Google Scholar]
- Pearl R. The rate of living, being an account of some experimental studies on the biology of life duration. New York: Alfred A. Knopf; 1928. [Google Scholar]
- Pereira C, Santos MS, Oliveira C. Mitochondrial function impairment induced by amyloid beta-peptide on PC12 cells. Neuroreport. 1998;9:1749–55. doi: 10.1097/00001756-199806010-00015. [DOI] [PubMed] [Google Scholar]
- Perls T. Dementia-free centenarians. Exp Gerontol. 2004;39:1587–93. doi: 10.1016/j.exger.2004.08.015. [DOI] [PubMed] [Google Scholar]
- Petersen RC, Smith GE, Waring SC, et al. Mild cognitive impairment: Clinical characterization and outcome. Arch Neurol. 1999;56:303–8. doi: 10.1001/archneur.56.3.303. [DOI] [PubMed] [Google Scholar]
- Poirier J. Apoliproptein E and Alzheimer’s disease. A role in amyloid catabolism. Ann NY Acad Sci. 2000;924:81–90. doi: 10.1111/j.1749-6632.2000.tb05564.x. [DOI] [PubMed] [Google Scholar]
- Polvikoski T, Sulkava R, Rastas S, et al. Incidence of dementia in very elderly individuals: A clinical, neuropathological and molecular genetic study. Neuroepidemiology. 2005;26:76–82. doi: 10.1159/000090252. [DOI] [PubMed] [Google Scholar]
- Price JL, Ko AI, Wade MJ, et al. Neuron number in the entorhinal cortex and CA1 in preclinical Alzheimer disease. Arch Neurol. 58:1395–402. doi: 10.1001/archneur.58.9.1395. [DOI] [PubMed] [Google Scholar]
- Reddy PH. Mitochondrial oxidative damage in aging and Alzheimer’s disease: Implications for mitochondrially targeted antoxidant therapeutics. J Biomed Biotechnol. 2006;2006:31372. doi: 10.1155/JBB/2006/31372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiman EM, Caselli RJ, Yun LS, et al. Preclinical evidence of Alzheimer’s disease in persons homozygous for the epsilon 4 allele for apoliprotein E. NEJM. 1996;334:752–8. doi: 10.1056/NEJM199603213341202. [DOI] [PubMed] [Google Scholar]
- Relkin N, Szabo P, Adamiak B, et al. Intravenous immunoglobulin (IVIg) treatment causes dose-dependent alterations in β-amyloid (Aβ) levels and anti-Aβ antibody titers in plasma and cerebrospinal fluid (CSF) of Alzheimer’s disease (AD) patients. Neurology. 2005;64:A144. [Google Scholar]
- Rentz DM, Becker JA, Moran EK, et al. Amyloid imaging with Pittsburgh Compound-B (PIB) in AD, MCI, and highly intelligent older adults. Neurology. 2006;66(suppl 2):A66. [Google Scholar]
- Risner ME, Saunders AM, Altman JF, et al. Efficacy of rosiglitazone in a genetically defined population with mild-to-moderate Alzheimer’s disease. Pharmacogenomics J. 2006;6:246–45. doi: 10.1038/sj.tpj.6500369. [DOI] [PubMed] [Google Scholar]
- Saunders AM. Apolipoprotein E and Alzheimer disease: An update on genetic and functional analyses. J Neuropath Exp Neurol. 2000;59:751–8. doi: 10.1093/jnen/59.9.751. [DOI] [PubMed] [Google Scholar]
- Schenk D, Barbour R, Dunn W, et al. Immunization with amyloid-beta attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature. 1999;400:173–7. doi: 10.1038/22124. [DOI] [PubMed] [Google Scholar]
- Scheuner D, Eckman C, Jensen M, et al. Secreted amyloid beta-protein similar to that in the senile plaques of Alzheimer’s disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer’s disease. Nat Med. 1996;2:864–70. doi: 10.1038/nm0896-864. [DOI] [PubMed] [Google Scholar]
- Sherrington R, Rogaeva EI, Liang Y, et al. Cloning of a gene bearing missense mutations in early-onset familial Alzheimer’s disease. Nature. 1995;375:754–60. doi: 10.1038/375754a0. [DOI] [PubMed] [Google Scholar]
- Shoghi-Jadid K, Small GW, Agdeppa ED, et al. Localization of neurofibrillary tangles and beta amyloid plaques in the brains of living patients with Alzheimer disease. Am J Geriatr Psychiatry. 2002;10:24–35. [PubMed] [Google Scholar]
- Small GW, Mazziotta JC, Collins MT, et al. Apolipoprotein E type 4 allele and cerebral glucose metabolism in relatives at risk for familial Alzheimer disease. JAMA. 1995;273:942–7. [PubMed] [Google Scholar]
- Smyth KA, Fritsch T, Cook TB, et al. Worker functions and traits associated with occupations and the development of AD. Neurology. 2004;63:498–503. doi: 10.1212/01.wnl.0000133007.87028.09. [DOI] [PubMed] [Google Scholar]
- Snowdon DA, Kemper SJ, Mortimer JA, et al. Linguistic ability in early life and cognitive function and Alzheimer’s disease in late life. Findings from the Nun Study. JAMA. 1996;275:528–32. [PubMed] [Google Scholar]
- Stern Y. Cognitive reserve and Alzheimer disease. Alzheimer Dis Assoc Disord. 2006;20(3 suppl 2):S69–74. doi: 10.1097/00002093-200607001-00010. [DOI] [PubMed] [Google Scholar]
- Swerdlow RH, Parks JK, Cassarino DS, et al. Cybrids in Alzheimer’s disease: A cellular model of the disease? Neurology. 1997;49:918–25. doi: 10.1212/wnl.49.4.918. [DOI] [PubMed] [Google Scholar]
- Swerdlow RH. Mitochondrial DNA – related mitochondrial dysfunction in neurodegenerative diseases. Arch Pathol Lab Med. 2002;126:271–80. doi: 10.5858/2002-126-0271-MDRMDI. [DOI] [PubMed] [Google Scholar]
- Swerdlow RH, Kish SJ. Mitochondria in Alzheimer’s disease. Int Rev Neurobiol. 2002;53:341–85. doi: 10.1016/s0074-7742(02)53013-0. [DOI] [PubMed] [Google Scholar]
- Swerdlow RH, Khan SM. A “mitochondrial cascade hypothesis” for sporadic Alzheimer’s disease. Med Hypotheses. 2004;63:8–20. doi: 10.1016/j.mehy.2003.12.045. [DOI] [PubMed] [Google Scholar]
- Swerdlow RH.2006Is aging part of Alzheimer’s disease, or is Alzheimer’s disease part of aging? Neurobiol Aging July28[Epub ahead of print] PMID: 16876913. [DOI] [PubMed]
- Swerdlow RH.2007Mitochondria in cybrids containing mtDNA from persons with mitochondriopathies J Neurosci Res January22[Epub ahead of print] PMID: 17243174. [DOI] [PubMed]
- Szabados T, Dul C, Majtenyi K, et al. A chronic Alzheimer’s model evoked by mitochondrial poison sodium azide for pharmacological investigations. Behav Brain Res. 2004;154:31–40. doi: 10.1016/j.bbr.2004.01.016. [DOI] [PubMed] [Google Scholar]
- Tamagno E, Bardini P, Obbili A, et al. Oxidative stress increases expression and activity of BACE in NT2 neurons. Neurobiol Dis. 2002;10:279–88. doi: 10.1006/nbdi.2002.0515. [DOI] [PubMed] [Google Scholar]
- Thomassen R, van Schaick HW, Blansjaar BA. Prevalence of dementia over age 100. Neurology. 1998;50:283–6. doi: 10.1212/wnl.50.1.283. [DOI] [PubMed] [Google Scholar]
- Trifunovic A, Wredenberg A, Falkenberg M, et al. Premature ageing in mice expressing defective mitochondrial DNA polymerase. Nature. 2004;429:417–423. doi: 10.1038/nature02517. [DOI] [PubMed] [Google Scholar]
- Tuszynski MH, Thal L, Pay M, et al. A phase 1 clinical trial of nerve growth factor gene therapy for Alzheimer disease. Nat Med. 2005;11:551–5. doi: 10.1038/nm1239. [DOI] [PubMed] [Google Scholar]
- van der Walt JM, Dementieva YA, Martin ER, et al. Analysis of European mitochondrial haplogroups with Alzheimer disease risk. Neurosci Lett. 2004;365:28–32. doi: 10.1016/j.neulet.2004.04.051. [DOI] [PubMed] [Google Scholar]
- Vincent I, Rosado M, Davies P. Mitotic mechanisms in Alzheimer’s disease? J Cell Biol. 1996;132:413–25. doi: 10.1083/jcb.132.3.413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace DC. Mitochondrial genetics: a paradigm for aging and degenerative diseases? Science. 1992;256:628–32. doi: 10.1126/science.1533953. [DOI] [PubMed] [Google Scholar]
- Walsh DM, Klyubin I, Shankar GM, et al. The role of cell-derived oligomers of Abeta in Alzheimer’s disease and avenues for therapeutic intervention. Biochem Soc Trans. 2005;33:1087–90. doi: 10.1042/BST20051087. [DOI] [PubMed] [Google Scholar]
- Wolfe WT, Xia W, Ostaszewski BL, et al. Two transmembrane aspartates in presenilin-1 required for presenilin endoproteolysis and gamma-secretase activity. Nature. 1999;398:513–7. doi: 10.1038/19077. [DOI] [PubMed] [Google Scholar]
- Yang Y, Geldmacher DS, Herrup K. DNA replication precedes neuronal cell death in Alzheimer’s disease. J Neurosci. 2001;21:2662–8. doi: 10.1523/JNEUROSCI.21-08-02661.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng H, Jiang M, Trumbauer ME, et al. Beta-amyloid precursor protein-deficient mice show reactive gliosis and decreased locomoter activity. Cell. 1995;81:525–31. doi: 10.1016/0092-8674(95)90073-x. [DOI] [PubMed] [Google Scholar]
- Zhu X, Raina AK, Perry G, Smith MA. Alzheimer’s disease: The two-hit hypothesis. Lancet Neurol. 2004;3:219–26. doi: 10.1016/S1474-4422(04)00707-0. [DOI] [PubMed] [Google Scholar]
- Zlokovic BV. Neurovascular mechanisms of Alzheimer’s neurodegeneration. Trends Neurosci. 2005;28:202–8. doi: 10.1016/j.tins.2005.02.001. [DOI] [PubMed] [Google Scholar]