Abstract
Background/Aims
The androgen dehydroepiandrosterone (DHEA) attenuates allergic inflammatory airway reactions by down-regulating the Th2 response in mice. The purpose of this study was to investigate whether DHEA suppresses Th2 cytokine production in cultured peripheral blood mononuclear cells (PBMCs) from asthmatic patients.
Methods
Sixty-one consecutive suspected asthmatic or non-asthmatic men underwent tests for asthma. PBMCs from each subject were cultured with and without DHEA (0.01~10 µM) for 48 h. The concentrations of interferon (IFN)-γ, interleukin (IL)-5, and IL-10 in the culture supernatant were measured via an enzyme-linked immunosorbent assay.
Results
In PBMCs from subjects exhibiting methacholine airway hyperresponsiveness (AHR), DHEA significantly suppressed IL-10, IL-5, and IFN-γ production in a dose-dependent manner (all p<0.001) and tended to increase the IFN-γ/IL-5 ratio (p=0.087). DHEA (10 µM) suppressed cytokine production to a greater degree in subjects with AHR compared with those without AHR (IL-5: 24.0±7.8% vs. 40.9±3.6%, p<0.01; IFN-γ: 29.7±7.0% vs. 54.5±5.1%, p<0.01). Cytokine suppression was significantly related to AHR, serum total IgE levels, and skin reactivity to house dust mites.
Conclusions
DHEA suppressed both Th1 and Th2 responses, with a Th1 bias, and the degree of suppression was associated with the severity of AHR or atopy. Therefore, DHEA may be a useful therapy for asthma.
Keywords: Asthma; Atopy; Dehydroepiandrosterone; Mononuclear cells, Th2
INTRODUCTION
Asthma is associated with an enhanced Th2 immune response1). Th1 and Th2 cells are regulated reciprocally, and it has been shown that Th1 enhancers such as bacille Calmette-Guérin vaccine2) and CpG oligodeoxynucleotides3) suppress asthmatic reactions in animal models. The adrenal androgen dehydroepiandrosterone (DHEA) is also reported to increase the production of Th1 cytokines, including interleukin (IL)-24, 5) and interferon (IFN)-γ5), and to prevent or attenuate allergic inflammatory airway responses in an animal model of asthma6, 7).
In a study on human peripheral blood mononuclear cells (PBMCs), the number of cells secreting IFN-γ was related with the serum level of DHEA sulfate ester (DHEAS)8). In addition, patients with asthma9, 10) or atopic dermatitis11), another Th2-associated disease, showed significantly lower levels of serum DHEA compared with healthy controls. Moreover, Tabata et al.11) showed that DHEA significantly reduced IL-4 production in PBMCs from patients with atopic dermatitis.
The purpose of this study was to examine whether DHEA suppresses Th2 cytokine production in cultured PBMCs from asthmatic patients.
MATERIALS AND METHODS
Subjects
Sixty-one consecutive suspected asthmatic or non-asthmatic men (age range, 18~34 years) were recruited from Chonnam National University Hospital. Most of the patients with suspected asthma were young men who required a medical certificate for asthma before they could be exempted from obligatory military service. Most non-asthmatic subjects were medical students and doctors. Female subjects were also recruited, but only 18 subjects responded (age range, 21~38 years). Participants were classified according to gender and the presence of airway hyperresponsiveness (AHR), based on their responses to a methacholine bronchoprovocation test. All controller medications for asthma, including oral steroids, were withheld for more than 1 week before enrollment. The Institutional Review Board of Chonnam National University Hospital approved this study. All subjects were informed of the experimental procedures and provided written informed consent.
Study design
On the day of the study, a medical history was obtained and a physical examination was performed. Allergy skin-prick tests, baseline spirometry, and a methacholine bronchoprovocation test to detect AHR were conducted, in addition to several laboratory analyses, including complete blood counts, total IgE levels, and quantification of serum DHEAS and cortisol. A blood sample was drawn for PBMC culture. The fresh blood was immediately processed to isolate PBMCs, which were cultured with and without DHEA. The culture supernatant was assayed for Th1 and Th2 cytokines via an enzyme-linked immunosorbent assay (ELISA).
Asthma tests
Allergy skin-prick tests were carried out using the following 10 common aeroallergen extracts (Allergopharma, Reinbek, Germany): Dermatophagoides farinae, D. pteronyssinus, cockroach, cat, dog, Aspergillus fumigatus, hazel, birch, timothy, and ragweed. Histamine (1 mg/mL) and saline (0.9%) solutions were used as positive and negative controls, respectively. Reactivity was graded on a scale of 0~4+ for each allergen, based on the ratio of the size of the allergen-induced wheal to the size of the histamine-induced wheal. The sum of all 10 grades was used as an overall "atopy score," and the sum of the scores for D. farinae and D. pteronyssinus was designated the "house dust mite score." Total serum IgE levels were measured by nephelometry (Behring Diagnostics GmbH, Germany).
The bronchial challenge tests followed a standardized tidal breathing method as described previously12). Aerosols were generated using a DeVilbiss 646 nebulizer (DeVilbiss Co., Somerset, PA, USA; output, 0.13 mL/min), and airway responses to methacholine (Sigma, St. Louis, MO, USA) aerosols were measured using a spirometer (Spiro Analyzer ST-250, Fukuda Sangyo, Tokyo, Japan). The provocation concentration of methacholine (PC20) that resulted in a 20% decrease in forced expiratory volume in 1 s (FEV1) was calculated by linear interpolation of the log dose-response curve. AHR was defined as methacholine PC20 < 16 mg/mL. An index of bronchial reactivity (BRindex) was calculated as log10 [10 + the maximal% fall in FEV1/log10 (dose in mg/dl of the stimulus required to produce maximal% fall)]13).
Hormone and cytokine assay
Serum concentrations of DHEAS and cortisol were measured via radioimmunoassay using commercially available kits (Diagnostic Products Corp., Los Angeles, CA, USA). Radioactivity was quantified using a gamma counter (Cobra 5010 Quantum; Packard Instrument Co., Meriden, CT, USA).
PBMCs were isolated by centrifugation over a Ficoll-Hypaque density gradient (Lymphoprep; Nycomed Pharma, Oslo, Norway). The PBMCs were washed five times and re- suspended at a concentration of 1×106 cells/mL in RPMI medium 1640 (Gibco BRL, Paisley, UK) supplemented with 10% fetal bovine serum (FBS; Gibco, Grand Island, NY, USA) and 1% penicillin/streptomycin/amphotericin B (BioWhittaker, Walkersville, MD, USA). The PBMCs were stimulated with a T-cell-specific mitogen, concanavalin A (Con A; 2.5 µg/mL; Sigma), after pre-incubation with DHEA (10 µmole; Sigma) for 1 h at 37℃. Some PBMCs were also pre-incubated with 0.01~1 µmole DHEA. After 48 h of culture, cell culture supernatants were collected and stored at 70℃ until analysis. The concentrations of IFN-γ, IL-5, and IL-10 were measured using commercially available ELISA kits (Biosource, Camarillo, CA, USA). Assay sensitivities for IFN-γ, IL-5, and IL-10 were 4, 4, and 1 pg/mL, respectively.
Statistical analysis
Data are presented as the mean±standard error of the mean (SEM). PC20 values and IgE, hormone, and cytokine levels were log-transformed before analysis. The intra-group difference in cytokine levels was determined using the Wilcoxon signed rank and Friedman tests. The Mann-Whitney U test was used to detect significant differences between groups. Associations between variables were examined using Spearman's rank correlation coefficient. A value of p<0.05 was considered statistically significant.
RESULTS
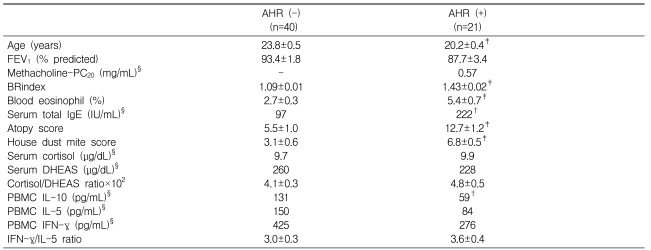
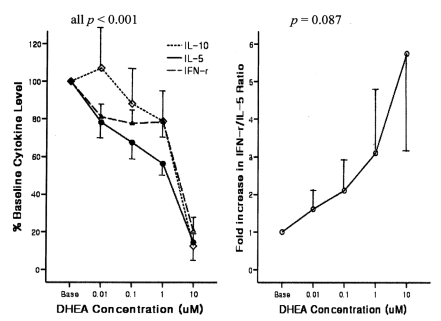
Male subjects with AHR were younger and showed significantly higher BRindices, eosinophil proportions, total IgE levels, atopy scores, and house dust mite scores, but lower IL-10 levels compared with men without AHR (Table 1). In subjects with AHR, the addition of DHEA to Con A-stimulated PBMCs significantly suppressed the production of IL-10, IL-5, and IFN-γ in a dose-dependent manner (all p<0.001) and tended to increase the IFN-γ/IL-5 ratio (p=0.087; Figure 1). In subjects without AHR, DHEA also significantly suppressed the production of IL-10, IL-5, and IFN-γ in a dose-dependent manner (all p<0.001) but did not affect the IFN-γ/IL-5 ratio.
Table 1.
Comparisons of subject characteristics
AHR, airway hyperresponsiveness; FEV1, forced expiratory volume in 1 s; PC20, the provocation concentration resulting in a 20% decrease in FEV1; BRindex, bronchial responsiveness index; Atopy score, sum of all grades for skin test reactivity in response to 10 common aeroallergens; House dust mite score, sum of grades for Dermatophagoides farinae and D. pteronyssinus reactivity; DHEAS, dehydroepiandrosterone sulfate; IFN, interferon; IL, interleukin; PBMC, peripheral blood mononuclear cells; §Geometric mean values; †p<0.05 and ‡p<0.01 compared with negative AHR.
Figure 1.
Dose-response relationship for the suppressive effect of dehydroepiandrosterone (DHEA) on cytokine production in cultured peripheral blood mononuclear cells (expressed as % of baseline value) and the resulting fold increase in the interferon (IFN)-γ/interleukin (IL)-5 ratio in subjects with airway hyperresponsiveness.
The observed changes in cytokine levels and the IFN-γ/IL-5 ratio from baseline after treatment with the highest dose (10 µM) of DHEA were significant in all comparisons, except for changes in the IFN-γ/IL-5 ratio in men with AHR, which showed a trend toward increasing (p=0.088) (Table 2). The maximal dose of DHEA markedly suppressed IL-5 production in subjects with AHR (24.0±7.8%) compared with those without AHR (40.9±3.6%, p<0.01). The suppression of IFN-γ production was also significantly greater in subjects with AHR (29.7±7.0%) than in those without AHR 54.5±5.1%, p<0.01). Although the resulting increase in the IFN-γ/IL-5 ratio tended to be higher in subjects with AHR compared with those without AHR, the difference between these groups did not reach statistical significance (p=0.71).
Table 2.
The suppressive effect of 10 µM dehydroepiandrosterone on concanavalin A-stimulated cytokine production in cultured peripheral blood mononuclear cells (expressed as % of baseline value) and the resulting fold increase in the interferon (IFN)-γ/interleukin (IL)-5 ratio
AHR, airway hyperresponsiveness; *p<0.01 compared with baseline; ‡p<0.01 compared with negative AHR.
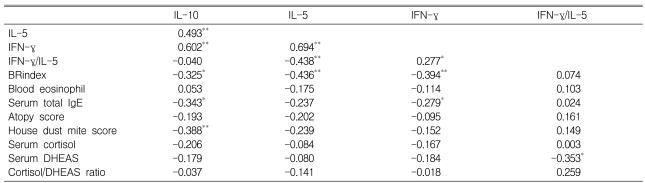
In male subjects, significant positive relationships were detected among IL-10, IL-5, and IFN-γ in terms of the degree of DHEA (10 µM)-induced suppression (expressed as % of baseline value; Table 3). The suppressive effect of DHEA on IL-10 was significantly related to AHR, serum total IgE level, and reactivity to house dust mites. In addition, both IL-5 and IFN-γ suppression were significantly related to AHR, IFN-γ suppression was significantly related to serum total IgE concentrations, and the IFN-γ/IL-5 ratio was significantly related to serum DHEAS.
Table 3.
Relationships among the suppressive effect of 10 µM dehydroepiandrosterone on cytokine production (expressed as % of baseline value), the resulting fold increase in the interferon (IFN)-γ/interleukin (IL)-5 ratio, and subject characteristics.
BRindex, bronchial responsiveness index; Atopy score, sum of all grades for skin test reactivity in response to 10 common aeroallergens; House dust mite score, sum of grades for Dermatophagoides farinae and D. pteronyssinus reactivity; DHEAS, dehydroepiandrosterone sulfate. *p<0.05 and **p<0.01.
Compared with women without AHR (n=15), men without AHR (n=40) showed a significantly higher level of DHEAS (geometric mean: 153 vs. 260 µg/dL, ; p<0.01) but not cortisol (8.2 vs. 9.7 µg/dL), which resulted in a significantly lower cortisol/DHEAS ratio (6.1±0.9 vs. 4.1±0.3%, p<0.05). Compared with female subjects, male subjects showed significantly higher eosinophil proportions, total IgE levels, and atopy scores, but lower IL-10 levels (199 vs. 131 pg/mL, p<0.01). Similar to male subjects, female subjects with AHR exhibited trends toward cytokine suppression (IL-10: 13.3±1.9%, IL-5: 44.2±7.0%, IFN-γ: 68.4±7.4%; all p<0.01) and a Th1 shift (ΔIFN-γ/IL-5 ratio: 2.5±0.7; p<0.01). However, we were unable to evaluate the effect of DHEA on cytokine production in female subjects with AHR owing to the small sample size (n=3), despite the similar trends in the suppression and Th1 shift.
DISCUSSION
In this study, DHEA suppressed the production of IL-5 in cultured PBMCs, resulting in a non-significant increase in the IFN-γ/IL-5 ratio in subjects with AHR. It has been reported that DHEA suppresses allergic inflammatory airway responses6, 7) and serum concentrations of IgE and IL-614) in animal models of asthma, and that DHEA reduces IL-4 production in cultured PBMCs from patients with atopic dermatitis11). The mechanism by which DHEA suppresses allergic responses is thought to involve a shift in the Th1/Th2 balance toward Th1. Our results support this hypothesis.
The Th1 cytokine IFN-γ inhibits Th2 cells15) and plays an important role in regulating the allergic airway reaction16, 17). However, in contrast to some reports that DHEA increases IFN-γ production5, 18), we found that DHEA suppressed IFN-γ production in a dose-dependent manner. Similar results have been reported by Yu et al.6, 7) and Araneo et al.19). Therefore, our results suggest that a mechanism mediated by some other factor (i.e., not IFN-γ) mediates the DHEA-associated suppression of the Th2 cytokine IL-5. Unfortunately, we did not quantify the number of PBMCs in our study; however, it has been reported that DHEA suppresses the proliferation of Con A-stimulated splenocytes in mice20). In addition, DHEA antagonizes glucocorticoid-induced deviation of the immune response toward a Th2 cytokine profile, probably through the peroxisome proliferator-activated receptor α21). Alternatively, DHEA may down-regulate both IFN-γ and IL-5 production while maintaining a Th1 bias, which would suggest that the balance between Th1 and Th2 is more important than the actual level of Th1 cytokines in determining the influence of DHEA on asthma.
IL-10 drives the generation of T regulatory 1 cells22), which produce IL-10, and in turn, IL-10 inhibits both Th1 and Th2 cytokine production23). Asthma is associated with diminished IL-10 expression in the allergic airway24), which is consistent with our result showing that the baseline IL-10 levels were lower in male subjects with AHR than in those without AHR. However, the IL-10 concentration in PBMCs was significantly reduced by treatment with DHEA, and we observed a significant positive correlation between the changes in IL-10 and IL-5 levels. Yu et al.6) also showed that DHEA decreased the production of both IL-5 and IL-10. Although it has been demonstrated that DHEA induces IL-10 production in spleen cells25), the decreased production of IL-10 may be the result of suppressed cell proliferation20) and other mechanisms.
Relatively more severe atopy or asthma was associated with a greater degree of DHEA-induced cytokine suppression in this study. Because inflammatory/immune cells are activated during active allergic disease processes and because DHEA up-regulates DHEA receptor activity in activated human T lymphocytes 26), DHEA would likely be more effective in suppressing inflammatory/immune responses during active allergic diseases.
As DHEA is an adrenal androgen, it is expected that the serum DHEAS level and cortisol/DHEAS ratio would differ significantly between male and female subjects without AHR. It has been reported that IFN-γ production in PBMCs is positively correlated with the plasma DHEA level and inversely correlated with the cortisol/DHEA ratio27). Therefore, the effects of DHEA treatment are likely influenced by gender. In the present study, exogenous DHEA increased the IFN-γ/IL-5 ratio in an inverse relationship with the serum DHEAS level, even though cytokine suppression and increased IFN-γ/IL-5 ratios tended to occur in both genders. These results are consistent with our previous study suggesting that in men, the higher background levels of DHEA and other androgens associated with the Th1 bias may result in an adaption to DHEA, and thus greater Th1-inducing power may be necessary to overcome this adaptation in men28).
The number of female subjects with AHR in this study was too small to evaluate the effects of DHEA among AHR subjects and between AHR and non-AHR subjects. Thus, further investigation involving a much larger sample size is required. Furthermore, because serum concentrations of DHEAS and cortisol are influenced by many factors, precise measurement is required in this regard.
Collectively, our study demonstrated that DHEA decreased both IL-5 and IFN-γ production in cultured PBMCs and skewed the Th1/Th2 balance toward Th1. In addition, the suppressive effect of DHEA on cytokine production was related to the severity of AHR or atopy. Therefore, DHEA supplementation may have therapeutic benefits for asthmatics.
Footnotes
This study was supported by a research grant from the Chonnam National University Research Institute of Medical Sciences.
References
- 1.Ricci M, Rossi O, Bertoni M, Matucci A. The importance of Th2-like cells in the pathogenesis of airway allergic inflammation. Clin Exp Allergy. 1993;23:360–369. doi: 10.1111/j.1365-2222.1993.tb00340.x. [DOI] [PubMed] [Google Scholar]
- 2.Herz U, Gerhold K, Gruber C, Braun A, Wahn U, Renz H, Paul K. BCG infection suppresses allergic sensitization and development of increased airway reactivity in an animal model. J Allergy Clin Immunol. 1998;102:867–874. doi: 10.1016/s0091-6749(98)70030-2. [DOI] [PubMed] [Google Scholar]
- 3.Shirota H, Sano K, Kikuchi T, Tamura G, Shirato K. Regulation of T-helper Type 2 cell and airway eosinophilia by transmucosal coadministration of antigen and oligodeoxynucleotides containing CpG motifs. Am J Respir Cell Mol Biol. 2000;22:176–182. doi: 10.1165/ajrcmb.22.2.3772. [DOI] [PubMed] [Google Scholar]
- 4.Daynes RA, Dudley DJ, Araneo BA. Regulation of murine lymphokine production in vivo: II. dehydroepiandrosterone is a natural enhancer of interleukin 2 synthesis by helper T cells. Eur J Immunol. 1990;20:793–802. doi: 10.1002/eji.1830200413. [DOI] [PubMed] [Google Scholar]
- 5.Araghi-Niknam M, Zhang Z, Jiang S, Call O, Eskelson CD, Watson RR. Cytokine dysregulation and increased oxidation is prevented by dehydroepiandrosterone in mice infected with murine leukemia retrovirus. Proc Soc Exp Biol Med. 1997;216:386–391. doi: 10.3181/00379727-216-44186. [DOI] [PubMed] [Google Scholar]
- 6.Yu CK, Yang BC, Lei HY, Chen YC, Liu YH, Chen CC, Liu CW. Attenuation of house dust mite Dermatophagoides farinae-induced airway allergic responses in mice by dehydroepiandrosterone is correlated with down-regulation of TH2 response. Clin Exp Allergy. 1999;29:414–422. doi: 10.1046/j.1365-2222.1999.00484.x. [DOI] [PubMed] [Google Scholar]
- 7.Yu CK, Liu YH, Chen CL. Dehydroepiandrosterone attenuates allergic airway inflammation in Dermatophagoides farinae-sensitized mice. J Microbiol Immunol Infect. 2002;35:199–202. [PubMed] [Google Scholar]
- 8.Verthelyi D, Klinman DM. Sex hormone levels correlate with the activity of cytokine-secreting cells in vivo. Immunology. 2000;100:384–390. doi: 10.1046/j.1365-2567.2000.00047.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Feher KG, Koo E, Feher T. Adrenocortical function in bronchial asthma. Acta Med Hung. 1983;40:125–131. [PubMed] [Google Scholar]
- 10.Dunn PJ, Mahood CB, Speed JF, Jury DR. Dehydroepiandrosterone sulphate concentrations in asthmatic patients: pilot study. N Z Med J. 1984;97:805–808. [PubMed] [Google Scholar]
- 11.Tabata N, Tagami H, Terui T. Dehydroepiandrosterone may be one of the regulators of cytokine production in atopic dermatitis. Arch Dermatol Res. 1997;289:410–414. doi: 10.1007/s004030050213. [DOI] [PubMed] [Google Scholar]
- 12.Choi IS, Lee S, Kim DH, Chung SW, Lee YC, Cho JY, Lee WJ. Airways are more reactive to histamine than to methacholine in patients with mild airway hyperresponsiveness, regardless of atopy. Korean J Intern Med. 2007;22:164–170. doi: 10.3904/kjim.2007.22.3.164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Burrows B, Sears MR, Flannery EM, Herbison GP, Holdaway MD. Relationships of bronchial responsiveness assessed by methacholine to serum IgE, lung function, symptoms, and diagnoses in 11-year-old New Zealand children. J Allergy Clin Immunol. 1992;90:376–385. doi: 10.1016/s0091-6749(05)80018-1. [DOI] [PubMed] [Google Scholar]
- 14.Sudo N, Yu XN, Kubo C. Dehydroepiandrosterone attenuates the spontaneous elevation of serum IgE level in NC/Nga mice. Immunol Lett. 2001;79:177–179. doi: 10.1016/s0165-2478(01)00285-1. [DOI] [PubMed] [Google Scholar]
- 15.Fernandez-Botran R, Sanders VM, Mosmann TR, Vitetta ES. Lymphokine-mediated regulation of the proliferative response of clones of T helper 1 and T helper 2 cells. J Exp Med. 1988;168:543–558. doi: 10.1084/jem.168.2.543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yoshida M, Leigh R, Matsumoto K, Wattie J, Ellis R, O'Byrne, Inman MD. Effect of interferon-gamma on allergic airway responses in interferon-gamma-deficient mice. Am J Respir Crit Care Med. 2002;166:451–456. doi: 10.1164/rccm.200202-095OC. [DOI] [PubMed] [Google Scholar]
- 17.Huang TJ, MacAry PA, Eynott P, Moussavi A, Daniel KC, Askenase PW, Kemeny DM, Chung KF. Allergen-specific Th1 cells counteract efferent Th2 cell-dependent bronchial hyperresponsiveness and eosinophilic inflammation partly via IFN-gamma. J Immunol. 2001;166:207–217. doi: 10.4049/jimmunol.166.1.207. [DOI] [PubMed] [Google Scholar]
- 18.Padgett DA, Loria RM. Endocrine regulation of murine macrophage function: effects of dehydroepiandrosterone, androstenediol, and androstenetriol. J Neuroimmunol. 1998;84:61–68. doi: 10.1016/s0165-5728(97)00244-0. [DOI] [PubMed] [Google Scholar]
- 19.Araneo BA, Dowell T, Diegel M, Daynes RA. Dihydrotestosterone exerts a depressive influence on the production of interleukin-4 (IL-4), IL-5, and gamma-interferon, but not IL-2 by activated murine T cells. Blood. 1991;78:688–699. [PubMed] [Google Scholar]
- 20.Loria RM, Padgett DA. Control of the immune response by DHEA and its metabolites. Rinsho Byori. 1998;46:505–517. [PubMed] [Google Scholar]
- 21.Hernandez-Pando R, De La Luz Streber M, Orozco H, Arriaga K, Pavon L, Al-Nakhli SA, Rook GA. The effects of androstenediol and dehydroepiandrosterone on the course and cytokine profile of tuberculosis in BALB/c mice. Immunology. 1998;95:234–241. doi: 10.1046/j.1365-2567.1998.00601.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Groux H, O'Garra A, Bigler M, Rouleau M, Antonenko S, de Vries JE, Roncarolo MG. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature. 1997;389:737–742. doi: 10.1038/39614. [DOI] [PubMed] [Google Scholar]
- 23.Umetsu DT, Akbari O, DeKruyff RH. Regulatory T cells control the development of allergic disease and asthma. J Allergy Clin Immunol. 2003;112:480–487. [PubMed] [Google Scholar]
- 24.Borish L, Rosenwasser LJ. Cytokines in allergic inflammation. In: Adkinson NF Jr, Yunginger JW, Busse WW, Bochner BS, Holgate ST, Simons FE, editors. Middleton's allergy principles & practice. 6th ed. Philadelphia: Mosby; 2003. pp. 135–157. [Google Scholar]
- 25.Cheng GF, Tseng J. Regulation of murine interleukin-10 production by dehydroepiandrosterone. J Interferon Cytokine Res. 2000;20:471–478. doi: 10.1089/10799900050023889. [DOI] [PubMed] [Google Scholar]
- 26.Okabe T, Haji M, Takayanagi R, Adachi M, Imasaki K, Kurimoto F, Watanabe T, Nawata H. Up-regulation of high-affinity dehydroepiandrosterone binding activity by dehydroepiandrosterone in activated human T lymphocytes. J Clin Endocrinol Metab. 1995;80:2993–2996. doi: 10.1210/jcem.80.10.7559886. [DOI] [PubMed] [Google Scholar]
- 27.Bozza VV, D'Attilio L, Mahuad CV, Giri AA, del Rey A, Besedovsky H, Bottasso O, Bay ML. Altered cortisol/DHEA ratio in tuberculosis patients and its relationship with abnormalities in the mycobacterial-driven cytokine production by peripheral blood mononuclear cells. Scand J Immunol. 2007;66:97–103. doi: 10.1111/j.1365-3083.2007.01952.x. [DOI] [PubMed] [Google Scholar]
- 28.Cui Y, Choi IS, Koh YA, Lin XH, Cho YB, Won YH. Effects of combined BCG and DHEA treatment in preventing the development of asthma. Immunol Invest. 2008;37:191–202. doi: 10.1080/08820130801967833. [DOI] [PubMed] [Google Scholar]