Abstract
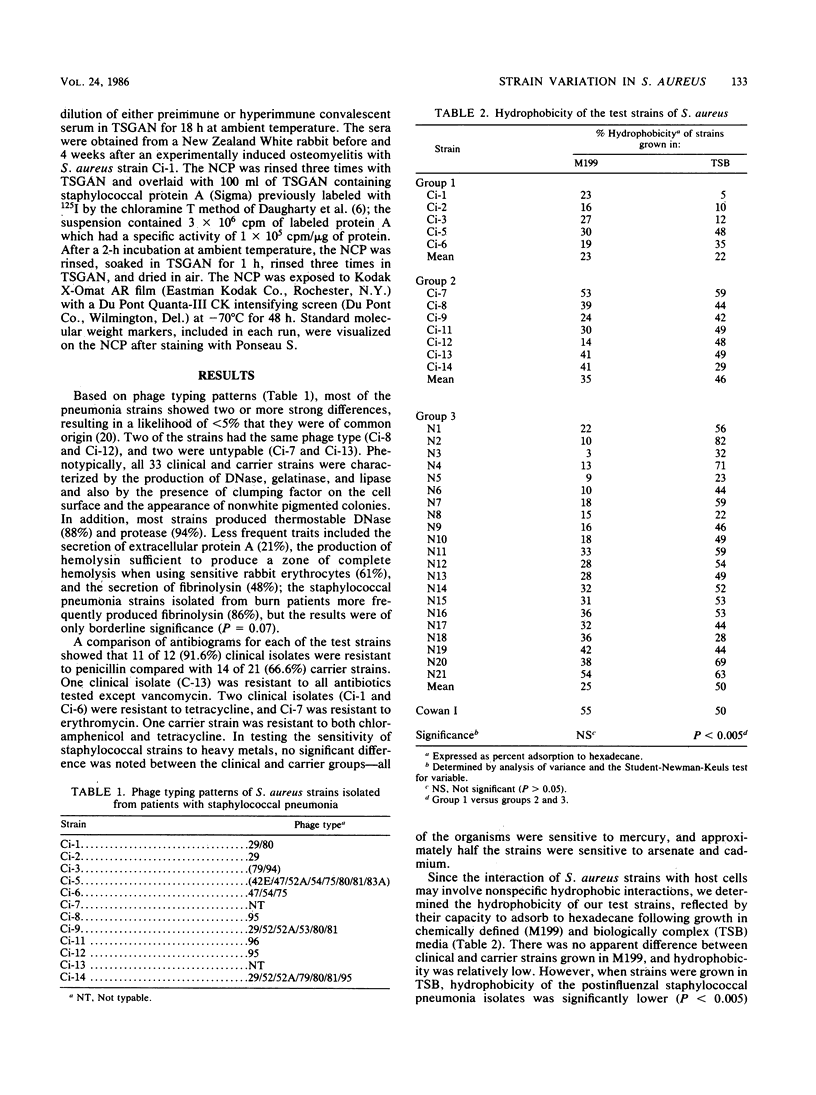
A total of 5 Staphylococcus aureus strains from patients with postinfluenzal staphylococcal pneumonia, 7 from burn patients with staphylococcal pneumonia, and 21 from the nasopharynx of carriers were phenotypically characterized. All or most strains produced coagulase, clumping factor, DNase, thermostable DNase, protease, gelatinase, lipase, and pigment; the strains were low to moderate producers of extracellular protein A, fibrinolysin, and alpha-hemolysin. All strains were sensitive to mercury, half were sensitive to arsenate and cadmium, and 67 to 92% were resistant to penicillin. Differences between strains were not statistically significant. Cell surface hydrophobicity was determined by measuring percent adsorption to hexadecane. Hydrophobicity of postinfluenzal staphylococcal pneumonia strains was significantly lower than that of pneumonia strains from burn patients and carriers (P less than 0.005). Immunoblot experiments with sera immune to one clinical test strain allowed the separation of all strains into three groups based on probe-positive reactions with primarily four staphylococcal polypeptides (154,200, 130,000, 77,100, and 64,400 molecular weight). The difference in distribution of clinical and carrier strains was highly significant (P = 0.007).
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barbour A. G. Vaginal isolates of Staphylococcus aureus associated with toxic shock syndrome. Infect Immun. 1981 Aug;33(2):442–449. doi: 10.1128/iai.33.2.442-449.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown M. R., Foster J. H. A simple diagnostic milk medium for Pseudomonas aeruginosa. J Clin Pathol. 1970 Mar;23(2):172–177. doi: 10.1136/jcp.23.2.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatterjee A. N., Mirelman D., Singer H. J., Park J. T. Properties of a novel pleiotropic bacteriophage-resistant mutant of Staphylococcus aureus H. J Bacteriol. 1969 Nov;100(2):846–853. doi: 10.1128/jb.100.2.846-853.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen M. L., Falkow S. Protein antigens from Staphylococcus aureus strains associated with toxic-shock syndrome. Science. 1981 Feb 20;211(4484):842–844. doi: 10.1126/science.7466361. [DOI] [PubMed] [Google Scholar]
- Daugharty H., Warfield D. T., Davis M. L. Solid-phase radioimmunoassay of total and influenza-specific immunoglobulin G. Appl Microbiol. 1972 Feb;23(2):360–367. doi: 10.1128/am.23.2.360-367.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davison V. E., Sanford B. A. Adherence of staphylococcus aureus to influenza A virus-infected Madin-Darby canine kidney cell cultures. Infect Immun. 1981 Apr;32(1):118–126. doi: 10.1128/iai.32.1.118-126.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forsgren A., Nordström K., Philipson L., Sjöquist J. Protein A mutants of Staphylococcus aureus. J Bacteriol. 1971 Jul;107(1):245–250. doi: 10.1128/jb.107.1.245-250.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goding J. W. Use of staphylococcal protein A as an immunological reagent. J Immunol Methods. 1978;20:241–253. doi: 10.1016/0022-1759(78)90259-4. [DOI] [PubMed] [Google Scholar]
- Köndell P. A., Nord C. E., Nordenram G. Characterization of Staphylococcus aureus isolates from oral surgical outpatients compared to isolates from hospitalized and non-hospitalized individuals. Int J Oral Surg. 1984 Oct;13(5):416–422. doi: 10.1016/s0300-9785(84)80068-x. [DOI] [PubMed] [Google Scholar]
- Lachica R. V., Genigeorgis C., Hoeprich P. D. Metachromatic agar-diffusion methods for detecting staphylococcal nuclease activity. Appl Microbiol. 1971 Apr;21(4):585–587. doi: 10.1128/am.21.4.585-587.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Langone J. J. Protein A of Staphylococcus aureus and related immunoglobulin receptors produced by streptococci and pneumonococci. Adv Immunol. 1982;32:157–252. [PubMed] [Google Scholar]
- Ljungh A., Hjertén S., Wadström T. High surface hydrophobicity of autoaggregating Staphylococcus aureus strains isolated from human infections studied with the salt aggregation test. Infect Immun. 1985 Feb;47(2):522–526. doi: 10.1128/iai.47.2.522-526.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melly M. A., Duke L. J., Liau D. F., Hash J. H. Biological properties of the encapsulated Staphylococcus aureus M. Infect Immun. 1974 Aug;10(2):389–397. doi: 10.1128/iai.10.2.389-397.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlievert P. M., Osterholm M. T., Kelly J. A., Nishimura R. D. Toxin and enzyme characterization of Staphylococcus aureus isolates from patients with and without toxic shock syndrome. Ann Intern Med. 1982 Jun;96(6 Pt 2):937–940. doi: 10.7326/0003-4819-96-6-937. [DOI] [PubMed] [Google Scholar]
- Schwarzmann S. W., Adler J. L., Sullivan R. J., Jr, Marine W. M. Bacterial pneumonia during the Hong Kong influenza epidemic of 1968-1969. Arch Intern Med. 1971 Jun;127(6):1037–1041. [PubMed] [Google Scholar]
- Sokol P. A., Ohman D. E., Iglewski B. H. A more sensitive plate assay for detection of protease production by Pseudomanas aeruginosa. J Clin Microbiol. 1979 Apr;9(4):538–540. doi: 10.1128/jcm.9.4.538-540.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Todd J. K., Franco-Buff A., Lawellin D. W., Vasil M. L. Phenotypic distinctiveness of Staphylococcus aureus strains associated with toxic shock syndrome. Infect Immun. 1984 Aug;45(2):339–344. doi: 10.1128/iai.45.2.339-344.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]



