Abstract
Allergic airway hyperresponsiveness (AHR) in OVA-sensitized and challenged mice, mediated by allergen-specific Th2 cells and Th2-like iNKT cells, develops under the influence of enhancing and inhibitory γδ T cells. The AHR-enhancing cells belong to the Vγ1+ γδ T cell subset, cells that are capable of increasing IL-5 and IL-13 levels in the airways in a manner like Th2 cells. They also synergize with iNKT cells in mediating AHR. However, unlike Th2 cells, the AHR-enhancers arise in untreated mice, and we show here that they exhibit their functional bias already as thymocytes, at an HSAhi maturational stage. In further contrast to Th2 cells and also unlike iNKT cells, they could not be stimulated to produce IL-4 and IL-13, consistent with their synergistic dependence on iNKT cells in mediating AHR. Mice deficient in IFN-γ, TNFRp75 or IL-4 did not produce these AHR-enhancing γδ T cells, but in the absence of IFN-γ, their spontaneous development was restored by adoptive transfer of IFN-γ competent dendritic cells from untreated donors. Intra-peritoneal injection of OVA/alum restored development of the AHR-enhancers in all of the mutant strains, indicating that the enhancers still can be induced when they fail to develop spontaneously, and that they themselves need not express TNFRp75, IFN-γ or IL-4 in order to exert their function. We conclude that both the development and the cytokine potential of the AHR-enhancing γδ T cells differs critically from that of Th2 cells and NKT cells, despite similar influences of these cell populations on AHR.
Keywords: T cells, Allergy, Lung, Spleen and Lymph Nodes, Transgenic/Knockout Mice
Introduction
Exposure to pathogens induces protective responses of antigen (Ag)-specific CD4+ T lymphocytes, which are associated with distinct cytokine patterns. Those involving interferon-γ (IFN-γ), interleukin-2 (IL-2) and tumor necrosis factor β (TNFβ) have been classified as T-helper 1 (Th1), those involving IL-4, -5, -9, and -13 as T-helper 2 (Th2) and those involving IL-17 as T-helper 17 (Th17) cells (1). All of these responses depend on Ag-priming, and on the induced differentiation of CD4+ αβ T cells into functionally polarized Th-types.
Polarized Th2 cells in particular promote humoral responses against extracellular parasites such as certain helminths (2), but they are also responsible for the development of hypersensitivity reactions against non-infectious allergens (3). Exactly what leads to the development of Th2-responses in vivo is not clear. According to one theory, weak Ag-stimulation favors Th2-reactivity. A Th2-bias also exists after birth when the immune system is still immature (4). However, it is frequently observed that Th2 responses are induced by specific infectious pathogens (5), and by allergens (6). Th2-cytokines such as IL-4 further aid in the development of Th2-immunity (7, 8). Recent studies have implicated the innate immune system in this process with its ability to recognize non-self motifs of extra-cellular or intra-cellular organisms and cell-damage in the initiation of induced Th2-reactivity (9–12). For example, it has been proposed that the innate system is able to recognize protease activity, which plays an essential role in the life cycle of helminths, and that allergens, many of which are proteases also, are recognized by essentially the same mechanism (13, 14). The stimulatory properties of aluminum adjuvants, which are commonly used to induce Th2-type immunity to inert protein antigens and allergens, were shown to be based on the activation of an intracellular innate response system, known as the Nalp3 inflammasome, which then induces the release of specific cytokines capable of polarizing the developing Th2 cells (11).
In mice infected with the Th1-inducing bacterium Listeria monocytogenes or the Th2-inducing extracellular parasite Nippostrongylus brasiliensis, γδ T cells also produced IFN-γ and IL-4, respectively, suggesting initially that γδ T cells in general might differentiate in response to Th-polarizing conditions in a manner similar to uncommitted αβ T cells (15). However, several studies showed that subsets of γδ T cells, defined by their expression of TCR-Vγ, exhibit differential and sometimes opposed Th-like reactivities (16, 17). In particular, cells expressing Vγ1, but not other γδ T cells, selectively responded to the Th2-inducing parasite Schistosoma mansonii (18), inhibited host resistance to the Th1-inducing L. monocytogenes (17) and increased Th2-cytokines and allergic airway hyperresponsiveness (AHR) in mice sensitized and challenged with ovalbumin (OVA) (19). To determine if γδ T cells must be induced to develop their Th-like functions, we established a model of adoptive cell-transfer, in which transferred γδ T cells can be examined for their ability to modulate OVA-induced AHR (20, 21). In this model, we previously observed that development of AHR-suppressive Vγ4+ γδ T cells had to be induced through repeated airway challenge of the donor mice, although matched Ag was not required (22). In marked contrast, we now demonstrate that development of the AHR-enhancing Vγ1+ γδ T cells does not require any treatment and is already established in the thymus, different from Th2 cells but similar to iNKT cells. Moreover, unlike both the induced AHR-mediating Th2 cells and the spontaneously developing AHR-mediating iNKT cells, the spontaneously developing AHR-enhancing γδ T cells do not exert their effect by producing the Th2 cytokines IL-4 and IL-13.
Materials and Methods
Animals
C57BL/6 mice and several mutant strains with the same genetic background (B6.TCR-β−/−, B6.TCR-δ−/−, B6.TCR-β−/−/δ−/−, B6.IL-4−/−, B6.CD8α−/−) were purchased from The Jackson Laboratory (Bar Harbor, Maine). B6.TCR-β−/−/IFN-γ−/− mice were generated by crossing the single mutants and breeding double mutants identified in the F2 generation, and B6.TNFRp75−/− mice were a gift from Dr. D. Lynch (Immunex, Seattle, WA). The mice were 8–12 wk old at the time of the experiments. All mice were maintained on an OVA-free diet, and were cared for at National Jewish Medical and Research Center (Denver, Colorado), following guidelines for immune deficient animals. All experiments were conducted under a protocol approved by the Institutional Animal Care and Use Committee.
Sensitization and airway challenge
Groups of mice were sensitized by i.p. injection of 20 μg OVA (ovalbumin grade V; Sigma-Aldrich, St. Louis, MO) emulsified in 2.25 mg aluminum hydroxide (AlumImject; Pierce Chemical, Rockford, IL) in a total volume of 100 μl on days 0 and 14 (2ip). These mice were either subsequently challenged or they served as donors of Vγ1+ γδ T cells. Mice were challenged via the airways with OVA (10 mg/ml in saline) for 20 min each on days 28, 29 and 30, by ultrasonic nebulization (particle size 1–5 mm; De Vilbiss, Somerset, PA) (3N). Lung resistance (RL) and dynamic compliance (Cdyn) were assessed 48h after the last allergen challenge, directly following provocation with aerosolized methacholine (MCh), and the mice were subsequently sacrificed to obtain tissues and cells for additional analyses.
Broncho-alveolar lavage (BAL)
Immediately following measurements of AHR, lungs were lavaged, and BAL fluid was recovered. Total leukocyte numbers were measured (Coulter Counter; Coulter Electronics, FL). Differential cell counts were performed by light microscopy of cyto-centrifuged preparations (Cytospin2, Cytospin; Thermo Shandon), stained with Leukostat (Fisher Scientific). For each sample, at least 200 cells were counted and differentiated by standard hematological procedures.
Cell purification and adoptive transfer of T cells and DC
C57BL/6 or B6.TCR-β−/− mice were used as cell donors. Their spleens and thymi were homogenized, treated with Gey’s solution for red blood cell-removal, and passed through nylon wool columns for T cell-enrichment. Non-adherent (NAD) cells were collected for further purification. NAD cells were stained with biotinylated anti Vγ mAbs, then washed and incubated with streptavidin-conjugated magnetic beads (Streptavidin Microbeads, Miltenyi Biotec, Bergisch Gladbach, Germany), and passed through magnetic columns as previously described in detail (22). In particular, repeated positive selection starting with NAD spleen cells from B6.TCR-β−/− mice produced a cell population containing > 90% viable Vγ1+ cells. Negative selection starting with NAD spleen cells from B6.TCR-β−/− mice involved removal of Vγ4+ cells and produced a cell population containing > 65% viable Vγ1+ cells as determined by two-color staining with anti-TCR-δ and anti-Vγ1 mAbs. In order to obtain subsets of Vγ1+ cells, NAD cells were stained with biotinylated subset-specific mAbs (anti-Vδ6.3 mAb 17-C or anti-HSA (CD24) mAb M1/69 plus streptavidin-PE and FITC-conjugated anti-Vγ1 mAb 2.11, then sorted on a MoFlo cell sorter (Dako Cytomation, Inc.)) and collected at a purity of > 95%. The purified cells were washed and re-suspended in BSS, and injected via the tail vein into OVA-sensitized mice (B6.TCR-δ−/−), less than 1 h prior to the first airway challenge.
To purify CD8+ DC, a CD8+ DC for reconstitution experiments, a CD8+ DC purification kit (Miltenyi Biotec, Bergisch Gladbach, Germany) was used as per the manufacturer’s recommendations. Briefly, red cell-depleted splenocytes derived from untreated T cell-deficient mice (B6.TCR-β−/−/δ −/−) were incubated with biotinylated antibody cocktail (anti-CD90, anti-CD45R and anti-CD49b) in PBS/5% FBS (10min on ice), then washed and incubated with anti-biotin microbeads (15min on ice), then washed and passed over a LD magnetic column. These enriched DC were then incubated with anti-CD8α (Ly-2) microbeads (30mins on ice), washed and passed twice over a MS magnetic column retaining the CD8+ DC. This protocol produced a preparation containing 60–70% viable CD8α+ DC, based on the additional criteria of dual MHC class II and CD11c expression as previously described (23). 1×105 wt CD8+ DCs were transferred i.v. to B6.TCR-β−/−/IFN-γ−/− recipients, which were used as donors of Vγ1+ cells 4 days later.
Note: Throughout this paper, we use the nomenclature for murine Vγ genes introduced by Tonegawa and colleagues (24). We use the term “enhancing cells” to refer to purified Vγ1+ γδ T cells capable of enhancing AHR upon adoptive cell transfer into OVA-sensitized and challenged recipients, and the term “suppressive cells” to refer to purified Vγ4+ γδ T cells derived from OVA-sensitized and challenged mice, which are capable of suppressing AHR.
Determination of airway responsiveness
Airway responsiveness was assessed as a change in lung function after provocation with aerosolized MCh using a method previously described in detail (25). MCh was administered for 10s (60 breaths/min, 0.5 ml tidal volume) in increasing concentrations. Maximum values of lung resistance (RL) and minimum values of dynamic compliance (Cdyn) were recorded and expressed as percent change from the baseline obtained after saline aerosol treatment.
Relative frequencies of activated and cytokine producing cells
Thymus and spleen (B6.TCR-β−/−) were homogenized and treated with Gey’s solution to remove red blood cells. For analysis of cytokine-competence, cells were cultured at 1–2×106/ml for 4 hours at 37° in the presence of PMA/ionomycin and Brefeldin A to activate them while blocking their cytokine release. After this, cells were stained with anti-Vγ1 (mAb 2.11-FITC) and anti-Vδ6.3(biotinylated mAb 17C and streptavidin-APC) mAbs, then fixed in 1% paraformaldehyde. After permeabilizing cells in 0.5% saponin buffer, PE-labeled anti-mIL4 (BVD4-1D11), anti-mIL13 (eBio13A), anti-mIL17 (TC11-18H10), anti-mIFNγ (XMG1.2) and anti-TNFα (MP6-XT22) mAbs (from Pharmingen, eBiosciences and BD Biosciences) were then added to detect intracellular cytokines. For analysis of the state of activation, freshly isolated cells were stained immediately with antibodies against these makers (anti CD25 (PC-61), anti CD44 (IM7), anti CD45RB (16A), anti CD62L (Mel-14), anti CD69 (H1.2F3), all from BD Pharmingen), in addition to the same TCR-specific antibodies as in the analysis of cytokine-expression. Fluorescent staining profiles were analyzed on a FACscan (Dako Cytomation, Inc).
Histology
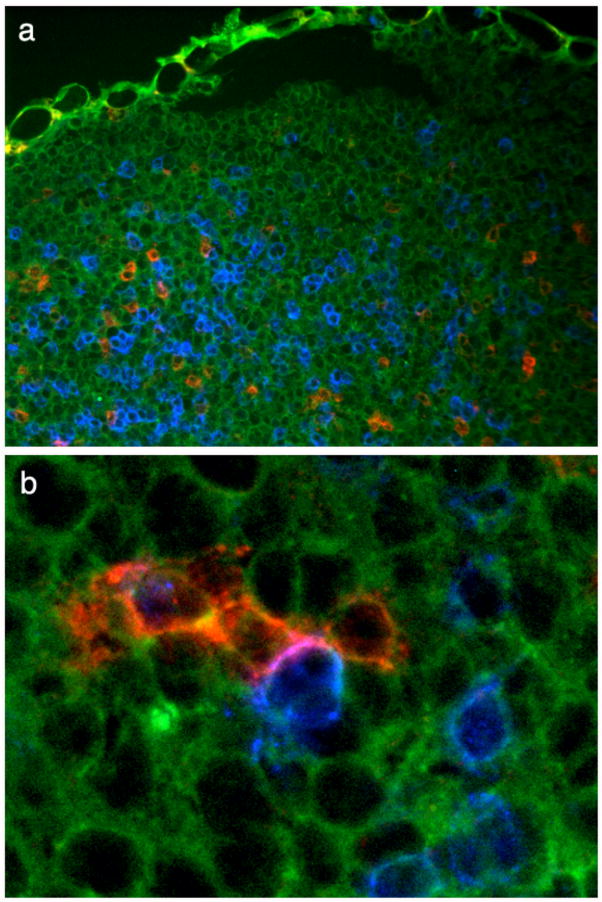
Frozen sections of thymus tissue from adult B6.TCR-β−/− mice were dehydrated and stained with antibodies as previously described in detail (23, 26). Abs used for histology were anti-TCR-δ (mAb GL3), and Vγ1 (mAb 2.11), anti CD8α (mAb 53–6.7) plus secondary reagents as described (26). Stained sections were mounted and slides viewed using a Leica DM-RXMA upright fluorescent confocal microscope using 25× (Fig. 7A) and 40× oil immersion objectives (Fig. 7B). Digital images were captured using a Cooke SensiCam (Romulus, MI), and processed on an Apple MacIntosh Computer using the SlideBook Imaging program (3I Inc.).
Figure 7.
Vγ1+ thymocytes encounter CD8α+ cells in the thymus. Frozen sections of adult B6.TCR-β−/− thymus were stained with antibodies specific for TCR-δ (red) and CD8α (blue) (A) or Vγ1 (red) and CD8α (blue) (B). Autofluorescence (green) shows thymus tissue. The digital image in panel B is 7.4× enlarged relative to panel A. The arrow in panel B points to the area of contact between the γδ T cell (red) and the CD8+ cell (blue).
Statistical analysis
Data are presented as means +/− standard error of the mean (SEM). The paired t test was used for two-group comparisons, and Two Way-ANOVA for analysis of differences in three or more groups, followed by pair-wise post Bonferroni test. Statistically significant levels were set at a p value of < 0.05.
Results
AHR-enhancing γδ T cells develop in normal untreated mice
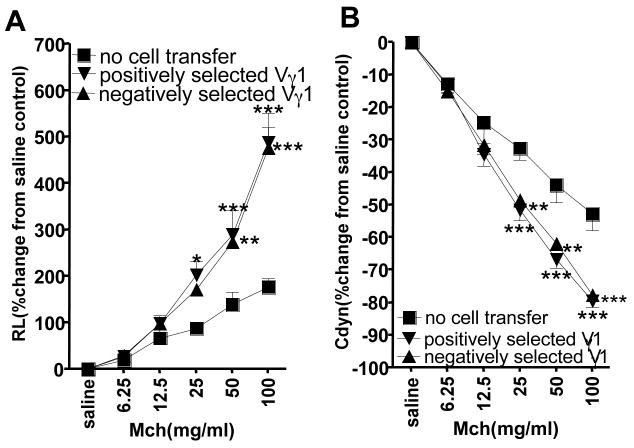
We have previously shown that γδ T cells expressing Vγ1 enhance AHR and also airway cytokines (IL-5 and IL-13) in C57BL/6 mice sensitized and challenged with OVA, using both TCR-depletion (in vivo treatment with anti TCR mAbs, which inactivates the targeted T cells) and complementary adoptive cell transfer (19, 21). In the cell transfer model, mice deficient for all γδ T cells (B6.TCR-δ−/−), which fail to develop AHR following OVA-sensitization and challenge when they are young, develop AHR at levels similar to wild-type mice when they receive small numbers of Vγ1+ γδ T cells prior to the first challenge. In addition, substantial increases in IL-5 and IL-13, and a decrease in IL-10 were evident in BAL fluid. The Th2-like AHR-enhancers can be detected in both OVA-sensitized wild-type C57BL/6 mice and genetically matched mice deficient in all αβ T cells (B6.TCR-β−/−), indicating that they arise independently from αβ T cells (21). The AHR-enhancers are also present in untreated donors, suggesting that they develop without need for any external stimulation (21). However, because the transferred cells in these experiments were purified in vitro using anti TCR-mAbs and magnetic beads, the possibility remained that they became activated and acquired their function through this process of positive selection. To address this issue, we prepared enriched Vγ1+ cells from the spleen of non-sensitized B6.TCR-β−/− mice by negative selection, and examined their AHR-enhancing potential in comparison with positively selected cells (Fig. 1A, B). Upon transfer into OVA-sensitized B6.TCR-δ−/− mice, the negatively selected Vγ1+ cells enhanced AHR to the same level as the positively selected cells, indicating that the process of positive selection does not contribute to their enhancing function. In contrast to their potent AHR-inducing effect, neither type of transferred cells substantially altered the inflammatory infiltrate in the airways (data not shown), consistent with our earlier findings (21).
Figure 1.
Vγ1+ γδ T cells from naïve donors mediate AHR regardless of whether they were purified by positive or negative selection. AHR was monitored by measuring lung resistance (A) and dynamic compliance (B). OVA-sensitized B6.TCR-δ−/− mice received 1 ×104 splenic Vγ1+ γδ T cells from untreated B6.TCR-β−/− donors, prior to airway challenge. The Vγ1+ cells were enriched by positive or negative selection, as described in the Methods. Recipients that were sensitized and challenged but did not receive cells (no cells transferred) are also shown. Results for each group are presented as means +/− SEM (n=7–8). Significant differences between “no cell transfer” and “selected Vγ1” cells transferred groups are indicated: *P<0.05, **P<0.01, ***P<0.001.
AHR-enhancing γδ T cells exhibit their functional potential already as thymocytes
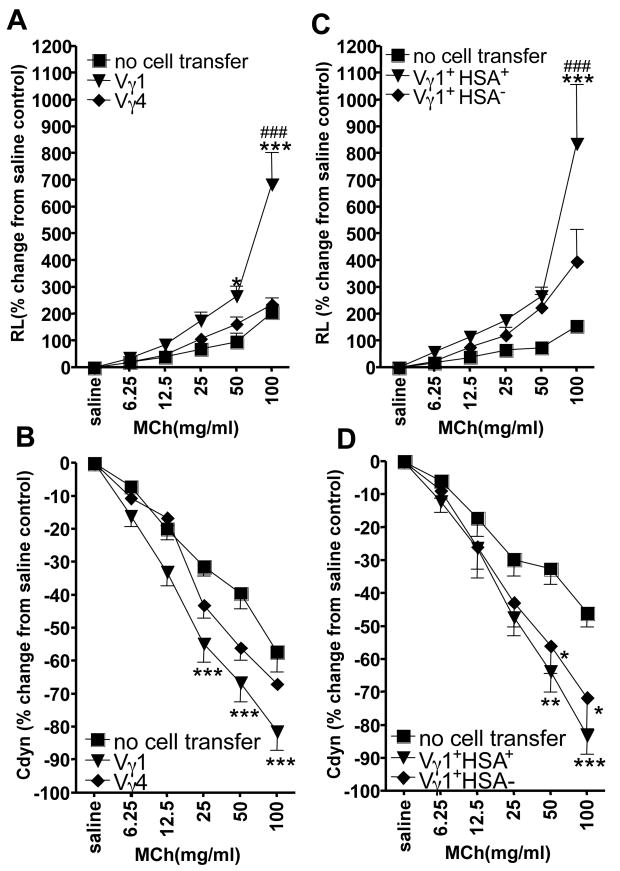
Because donors of the splenic AHR-enhancers did not require sensitization or any other prior treatment, it seemed possible that Vγ1+ γδ T cells acquire their AHR-enhancing potential already during intra-thymic maturation. To investigate this possibility, Vγ1+ and Vγ4+ thymocytes were purified from the thymi of untreated 8–10 wk old B6.TCR-β−/− mice, and transferred into OVA-sensitized B6.TCR-δ−/− recipients, prior to airway challenge and the measurement of airway function. Whereas Vγ4+ thymocytes had no effect, Vγ1+ thymocytes enhanced AHR though not as strongly as Vγ1+ splenocytes (Fig. 2A, B). As was observed with the splenic γδ T cells, the transferred thymocytes did not change the composition of the cellular infiltrate in the airways (data not shown). Because some peripheral γδ T cells return to the thymus (27), the possibility remained that the thymic AHR-enhancers belong to this returning mature population. Therefore, we fractionated Vγ1+ thymocytes based on their expression of heat stable antigen (HSA, CD24), a marker of thymocyte maturation (28, 29). In general, HSAhi thymocytes are less mature than HSAlo thymocytes, and peripheral T cells remain HSAlo. When we compared the HSAhi and HSAlo Vγ1+ thymocytes, we found that both cell types enhanced AHR (Fig. 2C, D). Again, the transferred thymocytes did not substantially alter the composition of the cellular infiltrate in the airways (data not shown).
Figure 2.
Vγ1+ thymocytes mediate AHR. AHR was monitored by measuring lung resistance (A, C) and dynamic compliance (B, D). OVA-sensitized B6.TCR-δ−/− mice received 1×104 splenic Vγ1+ thymocytes from untreated B6. TCR-β−/− donors, prior to airway challenge. The thymocytes were selected for Vγ1 or Vγ4 expression, and Vγ1+ thymocytes were further divided into HSAhi and HSAlo cells, as described in the Methods. Recipients that were sensitized and challenged but did not receive cells (no cell transfer), are also shown. Results for each group are presented as means +/− SEM (n=4–7). Significant differences between “no cell transfer” and “Vγ1+ cells” transferred groups are indicated: *P<0.05, **P<0.01, ***P<0.001. Significant differences between Vγ1+ and Vγ4+ cells transferred groups, or Vγ1/HSA+ and Vγ1/HSA− cells transferred groups are also indicated: ###P<0.001.
Vγ1+ AHR-enhancers fail to produce IL-4/13
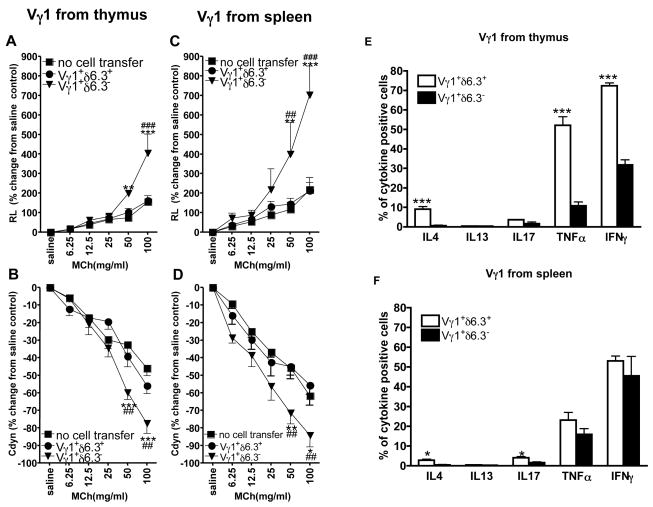
We also examined Vγ1+ thymocytes co-expressing Vδ6.3, for two reasons. First, these cells have been previously reported to return to the thymus from peripheral locations (27), raising the possibility that thymic AHR-enhancers might consist of this more mature population. Second, these Vγ1+/Vδ6.3+ cells have been likened to iNKT cells, which do not require Th2-polarization to produce IL-4 and IL-13 and are capable of mediating AHR (30). However, when we compared Vγ1+Vδ6.3+ and Vγ1+Vδ6.3− thymocytes, only the latter enhanced AHR (Fig. 3A, B), and when we extended the comparison to splenocytes, the AHR-enhancers again were contained within the Vγ1+Vδ6.3− fraction (Fig. 3C, D), consistent with our earlier finding that AHR-enhancing γδ T cells in the spleen express Vγ1/Vδ5 (21). Clearly, the AHR-enhancing γδ T cells in the thymus are not derived from the re-circulating NKT-like subset of γδ T cells (27). Next, we compared the same cell populations (Vγ1+Vδ6.3+ and Vγ1+Vδ6.3− cells) in thymus and spleen for their ability to produce cytokines when stimulated with PMA/ionomycin (Fig. 3E, G). Not surprisingly, large fractions of the NKT-like Vδ6.3+ cells in the thymus produced cytokines, including IL-4, TNF-α and IFN-γ. Far fewer cells among the Vδ6.3− thymocytes, which contain the AHR-enhancers, produced TNF-α and IFN-γ, and IL-4-producing cells were absent. Only the Vδ6.3+ cells contained IL-13 producers. IL-4/13-producing cells among Vγ1+ splenocytes were even less frequent. Only small fractions of Vγ1+ cells in thymus and spleen produced IL-17, and there was little if any difference between Vδ6.3+ and Vδ6.3− cells. The two fractions of Vγ1+ cells in the thymus also differed in their state of activation (Fig. 3F), with Vδ6.3− cells expressing the adhesion-molecule CD62L at significantly higher frequency, and more of the Vδ6.3+ cells expressing CD44 and CD45RB, two markers of activation/memory, but these differences mostly disappeared in the spleen (Fig. 3H). Taken together, these results illustrate the heterogeneity of the Vγ1+ γδ T cells, especially in the thymus, and they provide support for the notion that Vγ1+ γδ T cells capable of mediating AHR become functional in the thymus. In addition, they suggest that the AHR-enhancing γδ T cells exert their effect on the basis of a mechanism that is fundamentally different from that employed by AHR-mediating Th2 cells and iNKT cells, both of which produce IL-4 and IL-13.
Figure 3.
Vγ1+/Vδ6.3− γδ T cells mediate AHR but fail to express Th2 cytokines. AHR was monitored by measuring lung resistance (A, C) and dynamic compliance (B, D). Cytokine-producing cells were enumerated by intra-cellular staining and cytofluorometry (E, F), and activated cells were enumerated by cell-surface staining and cytofluorometry (G, H). To determine cellular effects on AHR, OVA-sensitized B6.TCR-δ−/− mice received 1×104 Vγ1+ thymocytes or splenic Vγ1+ cells from untreated B6.TCR-β−/− donors, prior to airway challenge. The Vγ1+ cells were further selected into Vδ6.3+ and Vδ6.3− fractions, as described in the Methods. Recipients that were sensitized and challenged but did not receive cells (no cell transfer) are also shown. Results for each group are presented as means +/− SEM (n= 4–6). Significant differences between “no cell transfer” and “Vγ1+ cells” transferred groups are indicated: *P<0.05, **P<0.01, ***P<0.001. Significant differences between “Vγ1/Vδ6.3+” and “Vγ1/Vδ6.3−“ cells transferred groups are also indicated: ##P<0.01, ###P<0.001. To determine frequencies of cells with potential for cytokine production, Vγ1 cells from B6.TCR-β−/− thymus and spleen were activated in vitro with PMA/ionophore in the presence of Brefeldin A, stained for TCR-expression and intra-cellular cytokines. Percent fractions of cytokine producing cells among Vγ1+/Vδ6.3+ and Vγ1+/Vδ6.3− subsets are shown (E, F). To determine frequencies of cells expression activation/memory markers, freshly isolated cells were stained directly for TCR expression and cell-surface markers. Percent fractions of marker-positive cells among Vγ1+/Vδ6.3+ and Vγ1+/Vδ6.3− subsets are shown (G, H). Results for each group are presented as mean±SEM (n=8–11). Significant differences between Vγ1+/Vδ6.3− and spleen Vγ1+/V6.3+ groups are indicated: *P<0.05, ** P<0.01, ***P<0.001.
Requirements for the development/function of AHR-enhancing γδ T cells
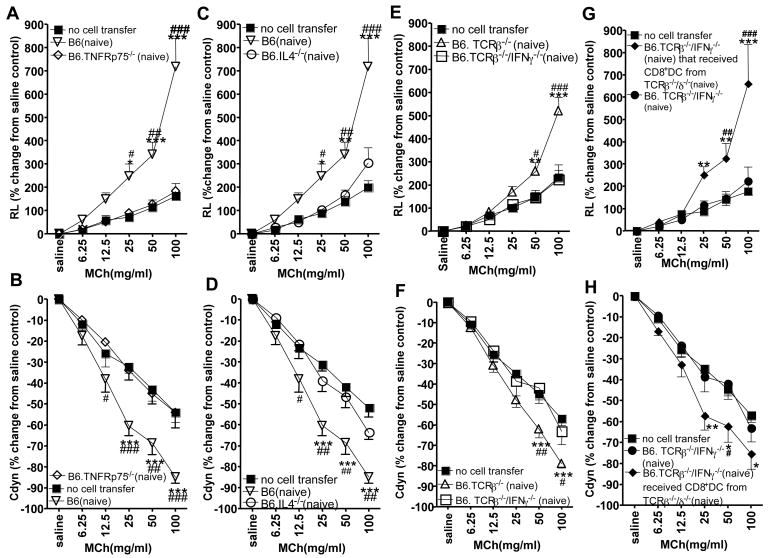
We previously found that γδ T cells derived from untreated mice express the high affinity TNF-receptor (TNF-Rp75) at higher levels, and are more sensitive to TNF-α compared to αβ T cells (31). For this reason, we considered the possibility that signaling through TNF-Rp75 might be required in their functional development. Indeed, Vγ1+ cells from the spleen of untreated C57BL/6 mice genetically deficient in TNF-Rp75 completely failed to enhance AHR in the cell-transfer model (Fig. 4A, B). This finding suggested that signaling through TNF-Rp75 is somehow required for the AHR-enhancing function of the Vγ1+ cells. Subsequently, we examined strains of mice deficient for additional cytokine genes, and found that Vγ1+ cells from untreated C57BL/6 mice genetically deficient in IL-4, and from untreated B6.TCR-β−/− mice deficient in IFN-γ, also have lost their AHR-enhancing potential (Fig. 4C-F). However, at least for Vγ1+ cells in the IFN-γ deficient mice (B6.TCR-β−/−/IFN-γ−/−), this functional potential was restored following transfer of CD8+ DC, purified from the spleen of untreated mice (Fig. 4G, H) by a previously described technique (23), confirming that the AHR-enhancers themselves need not express IFN-γ. For these experiments, the transferred DC were obtained from donors capable of producing IFN-γ (B6.TCR-β −/−/δ−/−), and might themselves have provided this cytokine. Taken together, these data revealed a role for several cytokines in connection with the AHR-enhancers, and suggested that IFN-γ and CD8+ DC might be involved in their development.
Figure 4.
Vγ1+ γδ T cells from naïve donors deficient in TNFRp75, IL-4 and IFN-γ fail to mediate AHR, and transferred CD8+ DC restore this ability in Vγ1+ cells from IFN-γ deficient mice. AHR was monitored by measuring lung resistance (A, C, E, G) and dynamic compliance (B, D, F, H). OVA-sensitized B6.TCR-δ−/− mice received 1×104 splenic Vγ1+ γδ T cells from untreated donors, or from donors that received 1×105 CD8+ splenic DC (G, H, see Methods), prior to airway challenge. In addition to the cells from the cytokine/cytokine-receptor-deficient mice, cells from matched normal controls (C57BL6 or B6.TCR-β−/− mice) were also transferred. Responses of recipients that were sensitized and challenged but did not receive cells (no cell transfer), are also shown. Results for each group are presented as means +/− SEM (A, B n=6; C, D n=8; E, F n=6). Significant differences between “no cells transfer” and “Vγ1+” cells transferred groups are indicated: *P<0.05, **P<0.01, ***P<0.001. Significant differences between wild-type or mutant-derived Vγ1 cells transferred, or between Vγ1+ cells derived from IFN-γ deficient or from IFN-γ-deficient DC-reconstituted mice, are also indicated: #P<0.05, ##P<0.01, ###P<0.001.
OVA/alum injection restores the AHR-enhancing Vγ1+ cells in mice that fail to produce them spontaneously
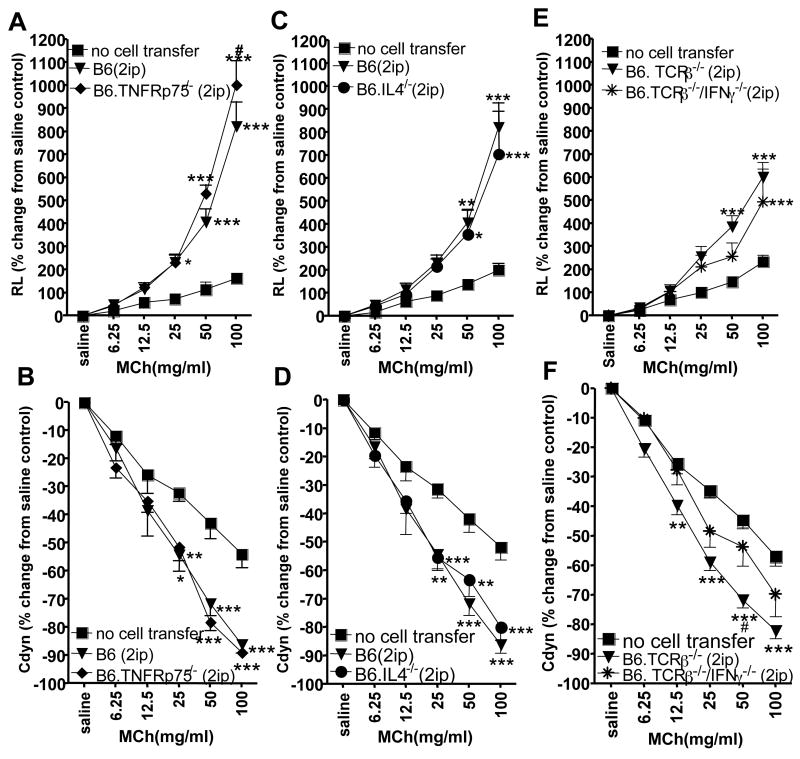
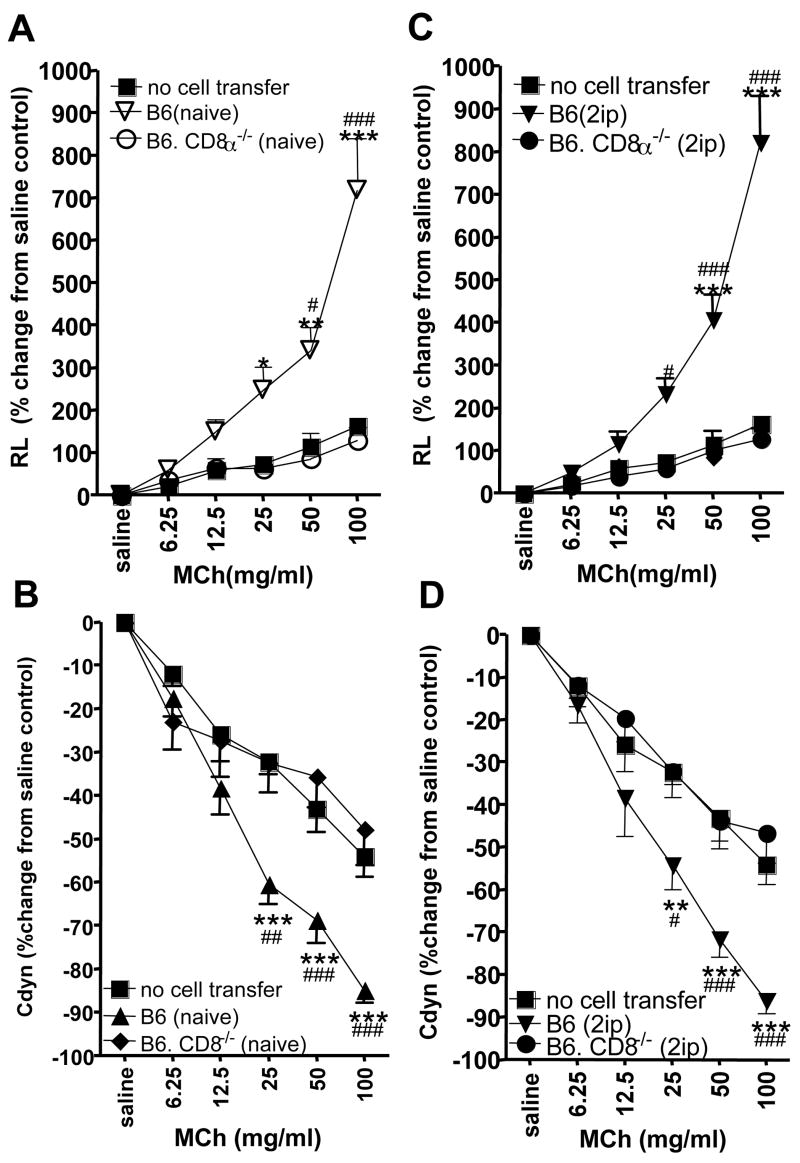
It has been shown that aluminum hydroxide (alum) enables Gr1+, IL-4-secreting innate cells to prime B lymphocytes (32). Moreover, alum together with a second stimulus such as LPS stimulates Nalp3 inflammasome activation and Th2 polarization (11), and a single i.p. injection of OVA/alum is sufficient to induce an antigen-specific IgE response (33). Having identified several genetically altered mouse strains in which AHR-enhancers fail to develop “automatically” (mice lacking the expression of IL-4, TNF-Rp75 or IFN-γ), we re-examined cells of these mice after two i.p. injections of OVA/alum. In all cases, the sensitized mice were now able to produce Vγ1+ γδ T cells capable of enhancing AHR (Fig. 5A–F), suggesting that there is also an inducible pathway for the development of the AHR-enhancing γδ T cells. Moreover, neither TNF-Rp75 nor IFN-γ or even IL-4 is absolutely required in the functional repertoire of these cells, which develop in the absence of these molecules. Finally, as a way to assess the limit of OVA/alum-mediated AHR-E-induction, we examined mice lacking CD8α expression, which also fail to produce AHR-enhancers, but can be restored in this regard by adoptive transfer of CD8α+ DC (23). Here, in marked contrast to the cytokine or cytokine-receptor-deficient mice, sensitization with OVA/alum alone did not restore the AHR-enhancers (Fig. 6A–D), suggesting that CD8+ DC are indispensable for the development of the AHR-enhancing γδ T cells. We have shown previously that CD8+ DC and γδ T cells encounter each other in the spleen, and that the DC are required for the development of the γδ T cells (23). Because the AHR-enhancing γδ T cells acquire their functional potential already in the thymus (this study), we examined the thymus histologically (Fig. 7). Here also, as in the spleen, CD8+ non-T cells could be seen frequently in close proximity or direct contact with γδ T cells.
Figure 5.
Vγ1+ γδ T cells from 2ip-sensitized donors deficient in TNFRp75, IL-4 and IFN-γ mediate AHR. AHR was monitored by measuring lung resistance (A, C, E) and dynamic compliance (B, D, F). OVA-sensitized B6.TCR-δ−/− mice received 1×104 splenic Vγ1+ γδ T cells from 2ip-sensitized donors, prior to airway challenge. In addition to the cells from the cytokine/cytokine-receptor-deficient mice, cells from 2ip-sensitized normal controls (C57BL6 or B6.TCR-β−/− mice) were also transferred. Responses of recipients that were sensitized and challenged but did not receive cells (no cell transfer), are also shown. Results for each group are presented as means +/− SEM (A, B n=6; C, D n=8; E, F n=5). Significant differences between “no cell transfer” and “Vγ1+” cells transferred groups are indicated: *P<0.05, **P<0.01, ***P<0.001. Significant differences between wild-type and mutant-derived “Vγ1+ cells transferred (F), are also indicated: #P<0.01.
Figure 6.
Vγ1+ γδ T cells from CD8α-deficient donors fail to mediate AHR even when the donors are 2ip-sensitized. AHR was monitored by measuring lung resistance (A, C) and dynamic compliance (B, D). OVA-sensitized B6.TCR-δ−/− mice received 1 ×104 splenic Vγ1+ γδ T cells from untreated (A, B) or 2ip-sensitized donors, prior to airway challenge. Recipients that were sensitized and challenged but did not receive cells (no cell transfer), are also shown. Results for each group are presented as means +/− SEM (A, B n=3; C, D n=5). Significant differences between “no cell transfer” and “Vγ1+” cells transferred groups are indicated: *P<0.05, **P<0.01, ***P<0.001. Significant differences between wild-type and mutant-derived Vγ1 cells are also indicated: #P<0.05, ##P<0.001, ###P<0.0001.
Discussion
The development and functions of γδ T cells remain poorly understood. Previous studies from one group suggested that γδ T cells in general acquire Th-like functional profiles when they develop under Th-polarizing conditions, so that they might become Th1-like during one type of infection and Th2-like during another (15). In contrast, another group provided evidence to indicate that γδ T cells develop with a strong bias to produce IFN-γ (34). Our earlier studies and those of others suggested that functional profiles in γδ T cells actually segregate with TCR-defined subsets, which may be Th-like or NKT-like (16, 19, 27). Investigating mice sensitized and challenged with OVA, which develop allergic airway inflammation and AHR dependent on several types of T cell including Th2 cells, CD8+ T cells, and iNKT cells, we found that Vγ1+ γδ T cells enhanced and Vγ4+ cells suppressed AHR (19). The Vγ4+ AHR-suppressors produced IFN-γ and depended on IFN-γ for their function (35), in this regard reminiscent of Th1 cells. The Vγ1+ cells exhibited less of a tendency to produce IFN-γ, and caused IL-5 and IL-13 to increase in the airways in a manner like Th2 cells and iNKT cells (19), suggesting that they might be categorized as Th2-like lymphocytes. However, the findings of the current study modify and refine this picture: they reveal that the AHR-enhancing γδ T cells develop quite differently from Th2 cells, and that their functional profile in terms of cytokine production differs from those of Th2 cells or Th2-like iNKT cells. The first set of observations in this study suggested that the spontaneously developing Vγ1+ AHR-enhancing γδ T cells might resemble AHR-enhancing iNKT cells, which also were found to develop without induction (30). In contrast to conventional Th2 cells, both cell-types appear to reach at least some degree of functional competence already in the thymus, in the case of the γδ T cells demonstrated by their enhancing effect on AHR following adoptive cell-transfer (this study), and in the case of the iNKT cells, by their ability to produce Th2-type cytokines in large quantity (36). Although the AHR-enhancing effect of the thymocytes was smaller than that of splenic γδ T cells, it is not insignificant when one considers that only 104 cells were transferred. These γδ T cells might mature even earlier than the iNKT cells, since they exhibited their function already as HSAhi thymocytes, whereas the iNKT cells acquire their functions at a HSAlo maturational stage (37), assuming that the innate cell types follow a maturational pathway in the thymus which resembles that of classical T cells. Because of the developmental similarity with iNKT cells, it seemed possible that the AHR-enhancing γδ T cells actually might belong to a previously identified Vγ1+ subpopulation with NKT-like properties (38). These cells frequently co-express Vδ6.3, and like iNKT cells, some express IL-4 already as thymocytes. Phenotypically, they resemble activated/memory-type cells. Our data confirmed these observations, but showed that the AHR-enhancing Vγ1+ thymocytes do not belong to this NKT-like subset. This is consistent with our earlier finding that the AHR-enhancing γδ T cells are NK1.1−, unlike the NKT-like population of γδ T cells (21). It remains to be addressed whether the thymic precursors of the AHR-enhancers must be selected in order to acquire their functional potential. Thymic selection clearly plays a role in the development of iNKT cells (39, 40), which also transition from HSAhi to HSAlo maturational stages. Thymic iNKT cells are thought to undergo a Th2-to-Th1 conversion such that the least mature cells only express IL-4, whereas intermediate cells express IL-4/IFN-γ and the most mature cells express IFN-γ (36). A maturational conversion also has been described for T10/T22-reactive γδ T cells, which were found to express IL-17 in the absence of an expressed ligand in the thymus, and IFN-γ in its presence, suggesting that the ligand-experience in the thymus changes their cytokine profile (41). A TCR-ligand for the AHR-enhancers is not known. However, consistent with a role for the TCR, we found that they exclusively express Vγ1Vδ5 (21), and only very few Vγ1+ thymocytes in this study could be induced to produce IL-17 while many produced IFN-γ.
The AHR-enhancing Vγ1+ γδ T cells may develop similarly to NKT cells, but they are not otherwise NKT-like. This difference is further emphasized by the induced cytokine profile of these cells. NKT cells upon induction produce large amounts of cytokine and typically exhibit a mixed profile, including both Th1 and Th2 cytokines (42). Similarly, we found relatively increased frequencies of IFN-γ, TNF-α and IL-4-producers among the NKT-like Vγ1+Vδ6.3+ thymocytes. However, Vγ1+Vδ6.3− thymocytes, which contain the AHR-enhancers, did not contain IL-4-producers, and fewer among them produced IFN-γ and TNF-α. These differences are not merely a reflection of different maturational states because the Vγ1+ cells in the spleen also failed to produce Th2 cytokines. Both Vγ1+Vδ6.3+ and Vγ1+Vδ6.3− spleen cells hardly contained any IL-13 or IL-17-producers. Therefore, although the effect of the γδ T cells on airway responsiveness resembles that of Th2-cells and iNKT cells, the mechanism by which they influence AHR probably is very different. This makes sense in view of our earlier findings that the AHR-enhancing γδ T cells depend on αβ T cells in order to exert their enhancing effect (19), and that Vγ1+ γδ T cells and iNKT cells synergize to promote AHR (21). When adoptively transferred into mice deficient of all other T cells, the two types of innate T cells depended on each other and either type alone could not induce AHR. It thus appears that the γδ T cells can exert their AHR-enhancing function only in synergy with a cellular source of essential Th2-type cytokines, especially IL-13, which is not provided by the γδ T cells. This remains to be shown, however, and the nature of the γδ T cell-contribution is still unclear.
The AHR-enhancing γδ T cells, which promote Th2-reactivity, might arise constitutively as part of a developmental program, or become induced by background levels of immune stimulation. To detect molecular and cellular requirements in their development, we examined the already known requirements for the Th2-response. Some time ago, TNF-α was identified as a critical component in the induction of protective Th2-responses during helminth infections (43), and it also appears to play a role in the induction of Th2-dependent allergic airway inflammation (10). Our data provide indirect evidence that signals from this cytokine are required for the development of the AHR-enhancers in untreated mice. Similarly, IL-4 can play an critical role at the onset of Th2-responses (9), and our data indicate that IL-4 is required for the development of the AHR-enhancers in untreated mice. Why IFN-γ might be required also is not obvious given that conventional Th2 responses are inhibited by IFN-γ (44), but it might prevent differentiation of the γδ T cells along a competing alternative pathway (45), or the requirement might relate to an auxiliary cell rather than the γδ T cells themselves. In fact, we already reported evidence that CD8α+ DC are needed for the development of the AHR-enhancers in untreated mice (23). Since the AHR-enhancing γδ T cells do not depend on antigen-priming, the role of the DC remains enigmatic (46). However, as DC also have been implicated in autophagy (47), they might provide autologous ligands for the γδ T cells. Histological evidence for CD8α+ DC encounters with Vγ1+ cells in the thymus is provided in Fig. 7. Furthermore, we show in the current study that reconstituting IFN-γ-deficient mice with wild-type CD8+DC is sufficient to restore the development of the AHR-enhancers (Fig. 4).
That i.p. injection of OVA/alum can restore development of the AHR-enhancers in the IFN-γ- or IL-4-deficient mice further emphasizes that these cytokines have no essential role in the AHR-enhancing function of these cells. Perhaps, sensitization with OVA/alum overcomes the various deficiencies because it stimulates both TLR-dependent and Nalp3 inflammasome-dependent pathways of innate-activation which help in the induction of Th2 cells (10, 11), and which also might provide inducing signals for γδ T cell-differentiation. In sum, our data suggest that the AHR-enhancing γδ T cells play an important part in the Th2-response, and that this part is synergistic and complementary rather than additive. In this way, a small number of γδ T cells might be able to jump-start and regulate the far larger populations of Th2 and Th2-like cells. How they do this and what might control their engagement remains to be determined.
Acknowledgments
We wish to acknowledge the valuable support of Dr. Katsuyuki Takeda.
Footnotes
This work was supported by NIH grants AI40611 and HL65410 to W.K.B., AI44920 and AI063400 to R.L.O. and HL36577 to E.W.G.
References
- 1.Steinman L. A brief history of TH17, the first major revision in the TH1/TH2 hypothesis of T cell-mediated tissue damage. Nature Medicine. 2007;13:139–145. doi: 10.1038/nm1551. [DOI] [PubMed] [Google Scholar]
- 2.Else KJ, Finkelman FD. Intestinal nematode parasites, cytokines and effector mechanisms. Int J Parasitol. 1998;28:145–1158. doi: 10.1016/s0020-7519(98)00087-3. [DOI] [PubMed] [Google Scholar]
- 3.Ramalingam TR, Reiman RM, Wynn TA. Exploiting worm and allergy models to understand Th2 cytokine biology. Current Opinion in Allergy and Clinical Immunology. 2005;5:392–398. doi: 10.1097/01.all.0000182542.30100.6f. [DOI] [PubMed] [Google Scholar]
- 4.Li L, Lee HH, Bell JJ, Gregg RK, Ellis JS, Gessner A, Zaghouani H. IL-4 utilizes an alternative receptor to drive apoptosis of Th1 cells and skews neonatal immunity toward Th2. Immunity. 2004;20:429–440. doi: 10.1016/s1074-7613(04)00072-x. [DOI] [PubMed] [Google Scholar]
- 5.Kaiko GE, Phipps S, Hickey DK, Lam CE, Hansbro PM, Foster PS, Beagley KW. Chlamydia muridarum infection subverts dendritic cell function to promote Th2 immunity and airways hyperreactivity. J Immunol. 2008;180:2225–2232. doi: 10.4049/jimmunol.180.4.2225. [DOI] [PubMed] [Google Scholar]
- 6.Wills-Karp M. Immunologic basis of antigen-induced airway hyperresponsiveness. Annu Rev Immunol. 1999;17:255–281. doi: 10.1146/annurev.immunol.17.1.255. [DOI] [PubMed] [Google Scholar]
- 7.Chen L, Grabowski KA, Xin J-P, Coleman J, Huang Z, Espiritu B, Alkan S, Xie HB, Zhu Y, White FA, Clancy J, Jr, Huang H. IL-4 induces differentiation and expansion of Th2 cytokine-producing eosinophils. J Immunol. 2004;172:2059–2066. doi: 10.4049/jimmunol.172.4.2059. [DOI] [PubMed] [Google Scholar]
- 8.Kim EY, Battaile JT, Patel AC, You Y, Agapov E, Grayson MH, Benoit LA, Byers DE, Alevy Y, Tucker J, Swanson S, Tidwell R, Tyner JW, Morton JD, Castro M, Polineni D, Patterson GA, Schwendener RA, Allard JD, Peltz G, Holtzman MJ. Persistent activation of an innate immune response translates respiratory viral infection into chronic lung disease. Nature Medicine. 2008;14:633–640. doi: 10.1038/nm1770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Min B, Paul WE. Basophils and type 2 immunity. Current Opinion in Hematology. 2008;15:59–63. doi: 10.1097/MOH.0b013e3282f13ce8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Eisenbarth SC, Piggott DA, Huleatt JW, Visintin I, Herrick CA, Bottomly K. Lipopolysaccharide-enhanced, Toll-like receptor 4-dependent T helper cell type 2 responses to inhaled antigen. J Exp Med. 2002;196:1645–1651. doi: 10.1084/jem.20021340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Eisenbarth SC, Colegio OR, O’Connor W, Jr, Sutterwala FS, Flavell RA. Crucial role for the Nalp3 inflammasome in the immunostimulatory properties of aluminium adjuvants. Nature. 2008;453:1122–1127. doi: 10.1038/nature06939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gordon S. Alternative activation of macrophages. Nature Reviews Immunology. 2002;3:23–35. doi: 10.1038/nri978. [DOI] [PubMed] [Google Scholar]
- 13.Zhu Z, Zheng T, Homer RJ, Kim YK, Chen NY, Cohn L, Hamid Q, Elias JA. Acidic mammalian chitinase in asthmatic Th2 inflammation and IL-13 pathway activation. Science. 2004;304:1678–1682. doi: 10.1126/science.1095336. [DOI] [PubMed] [Google Scholar]
- 14.Sokol CL, Barton GM, Farr AG, Medzitov R. A mechanism for the initiation of allergen-induced T helper type 2 responses. Nature Immunology. 2008 doi: 10.1038/ni1558. online Epap. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ferrick DA, Schrenzel MD, Mulvania T, Hsieh B, Ferlin WG, Lepper H. Differential production of interferon-γ and interleukin-4 in response to Th1- and Th2-stimulating pathogens by γδ T cells in vivo. Nature. 1995;373:255–257. doi: 10.1038/373255a0. [DOI] [PubMed] [Google Scholar]
- 16.Huber SA, Graveline D, Newell MK, Born WK, O’Brien RL. Vγ1+ T cells suppress and Vγ4+ T cells promote susceptibility to coxsackievirus B3-induced myocarditis in mice. J Immunology. 2000;165:4174–4181. doi: 10.4049/jimmunol.165.8.4174. [DOI] [PubMed] [Google Scholar]
- 17.O’Brien RL, Xiang Y, Huber S, Ikuta K, Born WK. Depletion of a γδ T cell subset can increase host resistance to a bacterial infection. J Immunol. 2000;165:6472–6479. doi: 10.4049/jimmunol.165.11.6472. [DOI] [PubMed] [Google Scholar]
- 18.Sandor M, Sperling AI, Cook GA, Weinstock JV, Lynch RG, Bluestone JA. Two waves of γδ T cells expressing different Vδ genes are recruited into schistosome-induced liver granuloma. J Immunol. 1995;155:275–284. [PubMed] [Google Scholar]
- 19.Hahn Y-S, Taube C, Jin N, Sharp L, Wands JM, Kemal Aydintug M, Lahn M, Huber SA, O’Brien RL, Gelfand EW, Born WK. Different potentials of γδ T cell subsets in regulating airway responsiveness: Vγ1+ cells, but not Vγ4+ cells, promote airway hyperreactivity, TH2 cytokines, and airway inflammation. J Immunol. 2004;172:2894–2902. doi: 10.4049/jimmunol.172.5.2894. [DOI] [PubMed] [Google Scholar]
- 20.Hahn YS, Taube C, Jin N, Takeda K, Park JW, Wands JM, Aydintug MK, Roark CL, Lahn M, O’Brien RL, Gelfand EW, Born WK. Vγ4+ T cells regulate airway hyperreactivity to methacholine in ovalbumin-sensitized and challenged mice. J Immunol. 2003;171:3170–3178. doi: 10.4049/jimmunol.171.6.3170. [DOI] [PubMed] [Google Scholar]
- 21.Jin N, Miyahara N, Roark CL, French JD, Aydintug MK, Gapin MJLL, O’Brien RL, Gelfand EW, Born WK. Airway hyperresponsiveness through synergy of gd T cells and NKT cells. J Immunol. 2007;179:2961–2968. doi: 10.4049/jimmunol.179.5.2961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jin N, Taube C, Sharp L, Hahn YS, Yin X, Wands JM, Roark CL, O’Brien RL, Gelfand EW, Born WK. Mismatched antigen prepares gd T cells for suppression of airway hyperresponsiveness. J Immunol. 2005;174:2671–2679. doi: 10.4049/jimmunol.174.5.2671. [DOI] [PubMed] [Google Scholar]
- 23.Cook L, Miyahara N, Jin N, Wands JM, Taube C, Roark CL, Potter TA, Gelfand EW, O’Brien RL, Born WK. Evidence that CD8+ dendritic cells enable the development of gammadelta T cells that modulate airway hperresponsiveness. J Immunol. 2008;181:309–319. doi: 10.4049/jimmunol.181.1.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Heilig JS, Tonegawa S. Diversity of murine gamma genes and expression in fetal and adult T lymphocytes. Nature. 1986;322:836–840. doi: 10.1038/322836a0. [DOI] [PubMed] [Google Scholar]
- 25.Lahn M, Kanehiro A, Takeda K, Joetham A, Schwarze J, Koehler G, O’Brien R, Gelfand EW, Born W. Negative regulation of airway responsiveness that is dependent on γδ T cells and independent of αβ T cells. Nature Medicine. 1999;5:1150–1156. doi: 10.1038/13476. [DOI] [PubMed] [Google Scholar]
- 26.Wands JM, Roark CL, Aydintug MK, Jin N, Hahn YS, Cook L, Yin X, Dalporto J, Lahn M, Hyde DM, Gelfand EW, Mason DY, O’Brien RL, Born WK. Distribution and leukocyte contacts of gd T cells in the lung. J Leukocyte Biology. 2005;78:1086–1096. doi: 10.1189/jlb.0505244. [DOI] [PubMed] [Google Scholar]
- 27.Azuara V, Levraud JP, Lembezat MP, Pereira P. A novel subset of adult gamma delta thymocytes that secretes a distinct pattern of cytokines and expresses a very restricted T cell receptor repertoire. Eur J Immunol. 1997;27:544–553. doi: 10.1002/eji.1830270228. [DOI] [PubMed] [Google Scholar]
- 28.Egerton M, Shortman K, Scollay R. The kinetics of immature murine thymocyte development in vivo. Int Immunol. 1990;2:501–507. doi: 10.1093/intimm/2.6.501. [DOI] [PubMed] [Google Scholar]
- 29.Pereira P, Zijlstra M, McMaster J, Loring JM, Jaenisch R, Tonegawa S. Blockade of transgenic γδ T cell development in β2-microglobulin deficient mice. EMBO J. 1992;11:25–31. doi: 10.1002/j.1460-2075.1992.tb05023.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Akbari O, Stock P, Meyer E, Kronenberg M, Sidobre S, Nakayama T, Taniguchi M, Grusby MJ, DeKruyff RH, Umetsu DT. Essential role of NKT cells producing IL-4 and IL-13 in the development of allergen-induced airway hyperreactivity. Nature Medicine. 2003;9:582–588. doi: 10.1038/nm851. [DOI] [PubMed] [Google Scholar]
- 31.Lahn M, Kalataradi H, Mittelstadt P, Pflum E, Vollmer M, Cady C, Mukasa A, Vella A, Ikle D, Harbeck R, O’Brien R, Born W. Early preferential stimulation of γδ T cells by TNF-α. J Immunol. 1998;160:5221–5230. [PubMed] [Google Scholar]
- 32.Jordan MB, Mills DM, Kappler J, Marrack P, Cambier JC. Promotion of B cell immune responses via an alum-induced myeloid cell population. Science. 2004;304:1808–1810. doi: 10.1126/science.1089926. [DOI] [PubMed] [Google Scholar]
- 33.McMenamin C, Pimm C, McKersey M, Holt PG. Regulation of IgE responses to inhaled antigen in mice by antigen-specific γδ T cells. Science. 1994;265:1869–1871. doi: 10.1126/science.7916481. [DOI] [PubMed] [Google Scholar]
- 34.Yin Z, Chen C, Szabo SJ, Glimcher LH, Ray A, Craft J. T-bet expression and failure of GATA-3 cross-regulation lead to default production of IFN-γ by γδ T cells. J Immunol. 2002;168:1566–1571. doi: 10.4049/jimmunol.168.4.1566. [DOI] [PubMed] [Google Scholar]
- 35.Lahn M, Kanehiro A, Takeda K, Terry J, Hahn YS, Aydintug MK, Konowal A, Ikuta K, O’Brien RL, Gelfand EW, Born WK. MHC class I-dependent Vγ4+ pulmonary T cells regulate αβ T cell-independent airway responsiveness. Proc Natl Acad Sci (USA) 2002;99:8850–8855. doi: 10.1073/pnas.132519299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Benlagha K, Kyin T, Beavis A, Teyton L, Bendelac A. A thymic precursor to the NT T cell lineage. Science. 2002;296:553–555. doi: 10.1126/science.1069017. [DOI] [PubMed] [Google Scholar]
- 37.Benlagha K, Wei DG, Veiga J, Teyton L, Bendelac A. Characterization of the early stages of thymic NKT cell development. J Exp Med. 2005;202:485–492. doi: 10.1084/jem.20050456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gerber DJ, Azuara V, Levraud J-P, Huang SY, Lembezat M-P, Pereira P. IL-4-producing γδ T cells that express a very restricted TCR repertoire are preferentially localized in liver and spleen. J Immunol. 1999;163:3076–3082. [PubMed] [Google Scholar]
- 39.Bendelac A, Savage PB, Teyton L. The biology of NKT cells. Annu Rev Immunol. 2007;25:297–336. doi: 10.1146/annurev.immunol.25.022106.141711. [DOI] [PubMed] [Google Scholar]
- 40.Matsuda JL, Mallevaey T, Scott-Browne JP, Gapin L. CD1d-restricted iNKT cells, the ‘Swiss-Army knife’ of the immune system. Current Opinion in Immunology. 2008;20:358–368. doi: 10.1016/j.coi.2008.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jensen KDC, Su X, Shin S, Li L, Youssef S, Yamasaki S, Steinman L, Saito T, Locksley RM, Davis MM, Baumgarth N, Chien YH. Thymic selection determines gammadelta T cell effector fate: antigen-naive cells make interleukin-17 and antigen-experienced cells make interferon gamma. Immunity. 2008;29:90–100. doi: 10.1016/j.immuni.2008.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Matsuda JL, Gapin L. Developmental program of mouse Vα14i NKT cells. Current Opinion in Immunology. 2005;17:122–130. doi: 10.1016/j.coi.2005.01.002. [DOI] [PubMed] [Google Scholar]
- 43.Artis D, Humphreys NE, Bancroft AJ, Rothwell NJ, Potten CS, Grencis RK. Tumor necrosis factor alpha is a critical component of interleukin 13-mediated protective T helper cell type 2 responses during helminth infections. J Exp Med. 1999;190:953–962. doi: 10.1084/jem.190.7.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wynn TA. IL-13 effector functions. Annu Rev Immunol. 2003;21:425–456. doi: 10.1146/annurev.immunol.21.120601.141142. [DOI] [PubMed] [Google Scholar]
- 45.Roark CL, Simonian PL, Fontenot AP, Born WK, O’Brien RL. Gamma/delta T cells: an important source of IL-17. Current Opinon in Immunology. 2008;20:353–357. doi: 10.1016/j.coi.2008.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lin ML, Zhan Y, Villadangos JA, Lew AM. The cell biology of cross-presentation and the role of dendritic cell subsets. Immunol Cell Biol. 2008;86:353–362. doi: 10.1038/icb.2008.3. [DOI] [PubMed] [Google Scholar]
- 47.Vyas JM, Van der Veen AG, Ploegh HL. The known unknowns of antigen processing and presentation. Nature Reviews Immunology. 2008;8:607–618. doi: 10.1038/nri2368. [DOI] [PMC free article] [PubMed] [Google Scholar]