Abstract
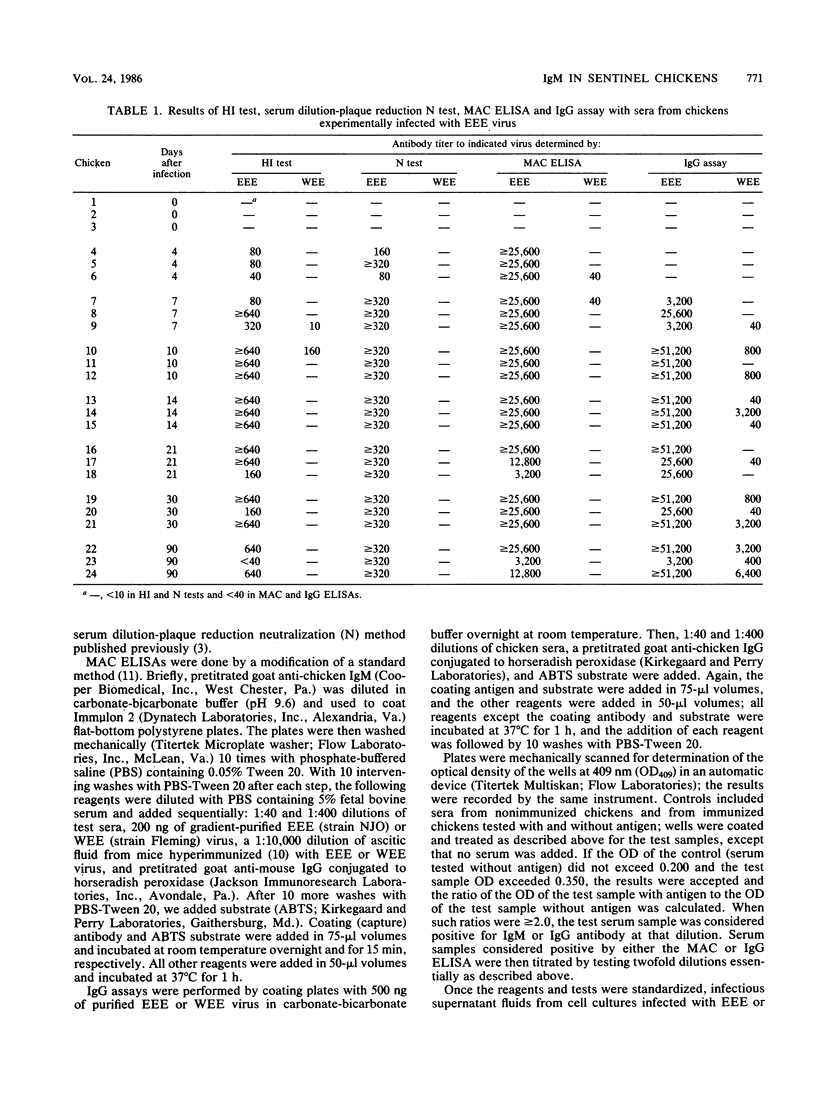
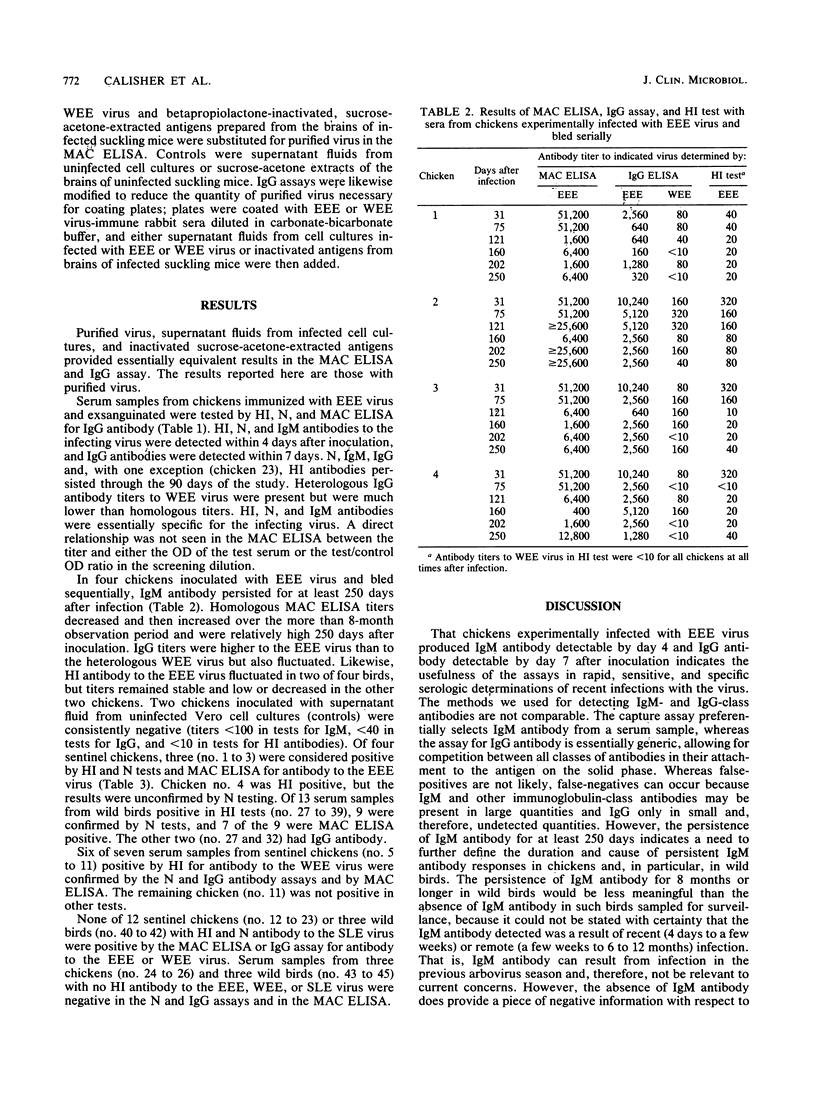
Young chickens were inoculated with 5,000 PFU of eastern equine encephalitis (EEE) virus and bled at intervals thereafter for determinations of hemagglutination-inhibiting (HI), neutralizing (N), immunoglobulin M (IgM), and IgG antibodies. HI, N, and IgM antibodies were first detected 4 days after infection, and IgG was detected 7 days after infection. All four antibodies persisted through day 90 after infection. HI, N, and IgM antibody titers remained elevated and were not cross-reactive with the related alphavirus western equine encephalitis (WEE) virus. IgG antibody titers also remained high, but heterologous reactivity to WEE virus increased with time after infection. Serum samples from sentinel chickens and wild birds infected in nature with EEE, WEE, or St. Louis encephalitis virus and submitted to this laboratory from state and local health departments were tested for IgM antibody by using anti-chicken IgM for capture and for IgG antibodies to the EEE and WEE viruses. There was essentially complete correlation between HI, N, and either IgM (indicating recent infections) or IgG (indicating more remote infections) antibody. We conclude that the IgM antibody capture enzyme immunoassay can be used as a specific and sensitive assay to replace the routinely used HI test for detecting antibody in sentinel chickens and in young, wild birds used for arbovirus surveillance. The test is rapid and relatively inexpensive and can be performed in essentially all adequately supplied laboratories.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BYRNE R. J., ROBBINS M. L. Mortality patterns and antibody response in chicks inoculated with Eastern equine encephalomyelitis virus. J Immunol. 1961 Jan;86:13–16. [PubMed] [Google Scholar]
- CLARKE D. H., CASALS J. Techniques for hemagglutination and hemagglutination-inhibition with arthropod-borne viruses. Am J Trop Med Hyg. 1958 Sep;7(5):561–573. doi: 10.4269/ajtmh.1958.7.561. [DOI] [PubMed] [Google Scholar]
- Calisher C. H., Monath T. P., Karabatsos N., Trent D. W. Arbovirus subtyping: applications to epidemiologic studies, availability of reagents, and testing services. Am J Epidemiol. 1981 Nov;114(5):619–631. doi: 10.1093/oxfordjournals.aje.a113234. [DOI] [PubMed] [Google Scholar]
- Chamberlain R. W. Arbovirology--then and now. Am J Trop Med Hyg. 1982 May;31(3 Pt 1):430–437. doi: 10.4269/ajtmh.1982.31.430. [DOI] [PubMed] [Google Scholar]
- Griffiths B. B., McClain O. Immunological response of chickens to eastern equine encephalomyelitis virus. Res Vet Sci. 1985 Jan;38(1):65–68. [PubMed] [Google Scholar]
- Holden P., Muth D., Shriner R. B. Arbovirus hemagglutinin-inhibition in avian sera: inactivation with protamine sulfate. Am J Epidemiol. 1966 Jul;84(1):67–73. doi: 10.1093/oxfordjournals.aje.a120628. [DOI] [PubMed] [Google Scholar]
- OGATA M., BYRNE R. J. Relationships between eastern and western equine encephalomyelitis viruses as demonstrated by the hemagglutination-inhibition antibody response of experimentally infected chickens. Am J Vet Res. 1961 Mar;22:266–270. [PubMed] [Google Scholar]
- Slaght S. S., Yang T. J., van der Heide L. Adaptation of enzyme-linked immunosorbent assay to the avian system. J Clin Microbiol. 1979 Nov;10(5):698–702. doi: 10.1128/jcm.10.5.698-702.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tikasingh E. S., Spence L., Downs W. G. The use of adjuvant and sarcoma 180 cells in the production of mouse hyperimmune ascitic fluids to arboviruses. Am J Trop Med Hyg. 1966 Mar;15(2):219–226. doi: 10.4269/ajtmh.1966.15.219. [DOI] [PubMed] [Google Scholar]


