Abstract
Context: Perivascular cell membrane-bound tissue factor (TF) initiates hemostasis via thrombin generation. The identity and potential regulation of TF-expressing cells at the human maternal-fetal interface that confers hemostatic protection during normal and preterm delivery is unclear.
Objectives: The objective of the study were to identify TF-expressing cells at the maternal-fetal interface in term and preterm decidual sections by immunohistochemistry and evaluate progestin, thrombin, TNF-α, and IL-1β effects on TF expression by cultured human term decidual cells (DCs).
Interventions and Main Outcome Measures: Serial placental sections were immunostained for TF. Leukocyte-free term DC monolayers were incubated with 10−8 m estradiol (E2) or E2 plus 10−7 m medroxyprogestrone acetate (MPA) ± thrombin or TNF-α or IL-1β. ELISA and Western blotting assessed TF in cell lysates. Quantitative real-time RT-PCR measured TF mRNA levels.
Results: Immunolocalized TF in DC membranes in preterm and term placental sections displayed higher Histologic Scores than villous mesenchymal cells (P < 0.05). TF was undetected in interstitial or extravillous trophoblasts. Compared with DCs incubated with E2, MPA and 2.5 U/ml thrombin each doubled TF levels (P < 0.05) and E2 + MPA + thrombin further doubled TF levels (P < 0.05), whereas TNF-α and IL-1β were ineffective. Western blotting confirmed the ELISA results. Quantitative RT-PCR revealed corresponding changes in TF mRNA levels.
Conclusions: In human term placental sections, DC-expressed TF exceeds that of other cell types at the maternal-fetal interface and is localized at the cell membranes in which it can bind to factor VII and meet the hemostatic demands of labor and delivery via thrombin formation. Unlike the general concept that TF is constitutive in cells that highly express it, MPA and thrombin significantly enhanced TF expression in term DC monolayers.
Elevated decidual cell-expressed tissue factor at the maternal-fetal interface is positioned to promote hemostasis and protect against hemorrhage during labor and delivery.
Progesterone transforms estradiol (E2)-primed human endometrial stromal cells to decidual cells (DCs) around blood vessels and glands during the midluteal phase of the menstrual cycle. Under continued progesterone stimulation, decidualization spreads throughout the late luteal phase and pregnant endometrium (decidua) (1,2). The use of in situ hybridization and immunohistochemistry (IHC) has localized tissue factor (TF) mRNA and protein, respectively, to decidualized stromal cells of luteal phase and early gestational endometrium (3,4,5). TF is a glycoprotein that consists of a membrane-spanning hydrophobic domain, a cytoplasmic tail, and a hydrophilic extracellular domain. Vascular injury initiates clotting when plasma-derived factor VII binds to the extracellular domain of perivascular cell membrane-bound TF. The resulting TF/VIIa complex promotes hemostasis via a series of changes initiated by the extracellular cleavage of prothrombin to thrombin (6).
Human uterine DC-expressed TF is spatially and temporally positioned to prevent hemorrhage, initially as blastocyst-derived extravillous trophoblasts (EVTs) breach decidual capillaries to provide the embryo with oxygen and nutrients before placentation (7) and subsequently as EVTs enter and remodel spiral arteries and arterioles into low-resistance, high-capacitance vessels that increase blood flow to the intervillous space required by the developing fetoplacental unit (8). In mice, TF expression by the uterine epithelium, decidua, and trophoblasts are reported to prevent fatal hemorrhage in the immediate postpartum period after detachment of the placenta from the uterine wall (9,10). In late human pregnancy, reports are conflicting as to the extent of TF expression by trophoblasts (9,10,11,12), whereas TF expression has not been assessed in the decidua. The crucial role of TF in protecting against potentially fatal hemorrhage during labor and delivery prompted use of IHC in the current study to determine the specific cellular location and extent of TF expression at the maternal-fetal interface of preterm and term placental sections.
Placental abruption (i.e. decidual hemorrhage) markedly enhances thrombin generation from DC-expressed TF. Severe cases lead to hypofibrinogenemia and disseminated intravascular coagulation. The involvement of abruptions with enhanced thrombin generation from DC-expressed TF is indicated by the high sensitivity and specificity with which elevated circulating thrombin-antithrombin complex levels predict the occurrence of preterm delivery due to preterm labor and/or premature rupture of fetal membranes (PPROM) (13,14,15). Thrombin also mediates several biological effects by binding to cell membrane-associated protease-activated receptors (16). Previously we found that thrombin acts via protease-activated receptor-1 to enhance matrix metalloproteinase (MMP) expression in term human DCs (17,18). Degradation of the fetal membrane and choriodecidual extracellular matrix (ECM) by DC-derived MMPs is consistent with the strong association between abruptions and PPROM (19).
In tissues that are highly vulnerable to hemorrhage such as the brain and placental villi, high constitutively expressed TF is postulated to prevent bleeding from damaged vessels (6,11). However, regulation of TF expression has been directly assessed in relatively few cell types. The critical role played by progesterone in maintaining pregnancy and preventing prematurity (20) taken together with the potentially fatal consequences of abruption prompted evaluation of the effects of medroxyprogesterone acetate (MPA) and thrombin on TF expression in cultured term DCs. Because deciduitis and chorioamnionitis are major antecedents of PPROM and preterm delivery (21,22,23,24), the effects of the classic proinflammatory cytokines, IL-1β and TNF-α, were also assessed on TF expression in cultured term DCs.
Patients and Methods
Patients and tissue
After receiving written informed consent, biopsies of decidua basalis used for IHC obtained from placentas of both idiopathic preterm (n = 10) and term (n = 5) deliveries were formalin fixed and paraffin embedded under approval of the Yale University School of Medicine Human Investigation Committee. Control placental specimens were obtained after either cesarean or vaginal delivery, and none displayed any clinical or histological evidence of chorioamnionitis or chronic villitis. Mean gestational ages ± sem were 38.9 ± 0.3 wk for term controls and 33.1 ± 1.5 wk for preterm controls.
IHC
Formalin-fixed, paraffin-embedded sections were deparaffinized in xylene and rehydrated through a graded series of ethanol. Antigen retrieval was performed in citrate buffer (10 mm; pH 6.0) under slow boil for 8 min. Endogenous peroxidase activity was quenched by incubating the slides in 3% H2O2 for 15 min. The slides were then incubated in 10% normal donkey serum (Jackson ImmunoResearch, West Grove, PA) in PBS with avidin blocking kit (Vector Laboratories, Burlingame, CA) for 1 h at room temperature in a humidified chamber. After blotting off excess serum, slides were incubated with primary antibodies (goat antihuman anticoagulation factor III/tissue factor antibody, 1:500 dilution; R&D Systems, Minneapolis, MN; and goat antihuman antivimentin antibody, 1:100 dilution; Abcam, Cambridge, MA) prepared in PBS with 0.1% Tween 20 and biotin blocking kit, overnight at 6 C. Afterward the slides were washed and then incubated with secondary antibody (biotinylated donkey antigoat antibody; Jackson ImmunoResearch) for 30 min at room temperature. After subsequent washing, the antigen-antibody complex was detected using an avidin-biotin-peroxidase kit (Vector Laboratories), with diaminobenzidine (3,3-diaminobenzidine tetrahydrochloride dehydrate) (Vector Laboratories) used as the chromogen. The sections were then counterstained with hematoxylin (Vector Laboratories) and mounted.
TF immunostaining intensity was semiquantitatively evaluated according to the following scale: 0 (no staining), 1+ (weak but detectable staining), 2+ (moderate or distinct staining), 3+ (intense staining). For each tissue, an Histologic Score (HSCORE) value was determined by calculating the sum of the percentage of cells that stain at each intensity scale and then multiplying that value by the weighted intensity scale, using the formula HSCORE = Σ π (i + 1), where i is the intensity scale and π is the percentage of cells staining at that scale. For each slide, five different areas and at least 100 cells per area were evaluated under a light microscope at a ×400 magnification. HSCOREs were determined at different times by two blinded investigators; the average score was then used. Vimentin-positive staining in close serial sections was used to localize DCs.
Isolation and culture of DCs
After receiving written informed consent, placentas and attached fetal membranes were obtained at term from 10 nonlaboring patients with uncomplicated pregnancies undergoing repeat cesarean deliveries at Yale-New Haven Hospital under Human Investigation Committee approval. A small portion of each specimen was formalin-fixed and paraffin-embedded and then examined histologically to exclude underlying acute or chronic inflammation.
After the amnion was removed from the placental membranes, the decidua was scraped from the maternal surface of the chorion, minced, and digested in Ham’s F-10 + 10% charcoal-stripped calf serum (Flow Laboratories, Rockville, MD) containing 2.5 mg/ml collagenase (200 U/mg) (Worthington Biochemical Corp., Freehold, NJ) in a shaking water bath at 37 C for 30 min (1 g of tissue per 10 ml of solution). After adding 6.25 U of deoxyribonuclease (Sigma-Aldrich, St. Louis, MO) per milliliter of digestate, the incubation was continued for another 45 min. The final digestate was passed through a 23-gauge needle five times to dissociate remaining cell clusters. Isolated cells were centrifuged at 1500 rpm for 5 min at 4 C and then washed in Ham’s F-10. This procedure was repeated three times, and the final cell pellet was resuspended (1 g of tissue per milliliter) in 20% Percoll (Sigma-Aldrich), layered on a (60:50:40) discontinuous Percoll gradient, and centrifuged at 22,000 rpm for 20 min at 4 C. The top cell layer was collected, washed, resuspended in Ham’s F-10 without serum, and centrifuged at 1500 rpm for 5 min at 4 C. After repeating this procedure, the resulting cell pellet was resuspended in 40% Percoll (1 g of tissue per milliliter), layered on a discontinuous (55:50:40) Percoll gradient, and centrifuged at 22,000 rpm for 20 min at 4 C. The top cell layer was washed twice in serum-free Ham’s F-10 and then centrifuged at 1500 rpm for 5 min at 4 C. The cell pellet was resuspended in Ham’s F-10 + 10% stripped calf serum, and DCs were counted in a hemocytometer. Trypan blue exclusion identified greater than 95% of isolated DCs as viable.
Isolated DCs (5 × 105 cells/ml) were suspended in basal medium, a phenol red-free 1:1 (vol/vol) mix of DMEM (Sigma) and Ham’s F-12 (Flow Laboratories) with 100 U/ml penicillin, 100 μg/ml streptomycin, and 0.25 μg/ml Fungizone supplemented with 10% stripped calf serum. DCs were seeded onto polystyrene tissue culture dishes coated with 2% type B gelatin (Sigma). The cultures were grown to confluence in a standard 95% air/5% CO2 incubator at 37 C. Cells were harvested using trypsin/EDTA and analyzed by flow cytometry with anti-CD45 and anti-CD14 monoclonal antibodies (BD PharMingen, San Diego, CA) to monitor the presence of leukocytes after each passage. After three to four passages, cell cultures were found to be leukocyte free (<1%). The latter were used for experimental cell incubations. The cultured cells were vimentin positive and cytokeratin negative by IHC. After five passages, the cells were seeded onto polystyrene tissue culture-treated flasks without gelatin coating.
Experimental cell incubations
Confluent DCs were primed for 7 d in basal medium supplemented with stripped calf serum containing either 10−8 m E2 or E2 + 10−7 m medroxyprogesterone acetate (Sigma) used in place of native progesterone, which is rapidly metabolized in vitro (25), with one change of medium. The cultures were washed twice with Hanks’ balanced salt solution to remove residual serum components and switched to a serum-free defined medium (DM) consisting of basal medium plus insulin/transferrin/selenium + (BD Biosciences, Bedford, MA), 5 μmol/liter FeSO4, 0.5 μmol/liter ZnSO4, 1 nmol/liter CuSO4, 50 μg/ml ascorbic acid (Sigma), and 50 ng/ml epidermal growth factor (BD Biosciences) with corresponding steroid(s) with or without thrombin (American Diagnostica, Stanford, CT) or TNF-α or IL-1β (R&D Systems). After 24 h, conditioned DM supernatants and cell lysates were frozen. RNA was extracted from parallel incubations after 6 h.
ELISA
A specific ELISA measured immunoreactive human TF levels in DC lysates (American Diagnostica, Stamford, CT). The results were normalized to total cell protein as measured by a modified Lowry assay (Bio-Rad Laboratories, Inc., Hercules, CA). The sensitivity of the ELISA is 10 pg/ml with intraassay and interassay coefficients of variation of 4.5% and 7.5%, respectively.
Western blotting
Western blot analysis was performed on 40 μg per sample of DC lysates, which were diluted in reducing Laemmli sample buffer and then boiled for 5 min. The samples were subjected to SDS-PAGE on a 4–15% Tris-HCl linear gradient gel (Bio-Rad) with subsequent electroblotting transfer onto a 0.2-μm nitrocellulose membrane (Bio-Rad). After transfer, the membrane was blocked overnight in Tris-buffered saline (Fisher, Fairlawn, NJ) with 4% nonfat dry milk and then incubated for 2 h with 2.0 μg/ml of a goat antihuman TF polyclonal antibody (American Diagnostica). The membrane was washed in PBS and 0.2% Tween 20 before and after incubation with horseradish peroxidase-conjugated rabbit antigoat IgG (ICN Biomedicals, Aurora, OH). Chemiluminescence was detected with enhanced chemiluminescence reagents (PerkinElmer Life Sciences, Boston, MA) and autoradiography film (Amersham Pharmacia, Buckinghamshire, UK) according to the manufacturer’s instructions. The membrane blot was then incubated in stripping buffer [100 mm 2-mercaptoethanol, 2% sodium dodecyl sulfate, 62.5 mm Tris-HCl (pH 6.7)] at 50 C for 30 min and then washed and blocked, as described above, before reprobing with 1:5000 of a mouse antihuman β-actin monoclonal antibody (Sigma-Aldrich) overnight at 4 C. The membrane was washed as described above before and after incubation with horseradish peroxidase-conjugated goat antimouse IgG (ICN Biomedicals). Subsequent chemiluminescence was detected as described above.
Real-time quantitative RT-PCR
To verify that the TF and β-actin probes yielded the correct bands, extracted RNA from experimental cell incubations were subjected to semiquantitative RT-PCR using a kit from Invitrogen (Carlsbad, CA), performing 35 cycles with the Mastercycler (Eppendorf, Westbury, NY). For quantitative real-time RT-PCR, reverse transcription was initially performed with avian myeloma virus reverse transcriptase (Invitrogen). A quantitative standard curve was created between 40 pg and 2.5 ng cDNA with a Light Cycler (Roche, Indianapolis, IN) by monitoring increasing fluorescence of PCR products during amplification. On establishing the standard curve, quantification of the unknown samples was determined with the Roche Light Cycler and adjusted to the quantitative expression of β-actin from the corresponding unknowns. Melting curve analysis determined the specificity of the amplified products and the absence of primer-dimer formation. All products obtained yielded correct melting temperatures. Products were then run on a 1.2% agarose gel along with a 100-bp DNA ladder and then stained with ethidium bromide for visualization. The following primers were synthesized and gel-purified at the Yale DNA Synthesis Laboratory, Critical Technologies: β-actin sense, 5′-CGTACCACTGGCATCGTGAT-3′, antisense, 5′-GTGTTGGCGTACAGGTCTTTG-3′, 452-bp product; TF sense, 5′-GAAGCAGACGTACTTGGCACGG-3′, antisense, 5′-CCGAGGTTTGTCTCCAGGTA-3′, 121-bp product.
Statistical analysis
The ELISA and quantitative RT-PCR results were not normally distributed, and therefore, pair-wise multiple comparisons were analyzed with nonparametric ANOVA on ranks (Kruskal-Wallis test), followed by post hoc Student-Newman-Keuls test. Control and treatment groups were compared with a P < 0.05 representing statistical significance. Immunostaining HSCOREs were normally distributed, as determined by the Kolmogorov-Smirnov test, and the results were analyzed by one-way ANOVA with post hoc Holm-Sidak testing. Statistical significance was defined as P < 0.05. Statistical calculations were performed using Sigma Stat version 3.0 (Jandel Scientific Corp., San Rafael, CA).
Results
Immunostaining of TF at the decidual/placental interface
Decidual/placental specimens from idiopathic preterm and term deliveries were immunostained to localize tissue factor and vimentin expression in close serial sections (Fig. 1). Vimentin immunostaining was performed to identify decidual cells (Fig. 1B). In all specimen groups, TF expression in the decidua basalis was localized almost exclusively to the perimembranous area of DCs and was essentially absent from interstitial trophoblasts. This contrast in differential TF localization is particularly evident when interstitial trophoblasts are immediately adjacent to and intercalate with DCs (Fig. 1, A and C, idiopathic preterm and term tissues, respectively). In placental villi, TF immunostaining was present in the villous mesoderm, in which it was localized primarily in perivascular adventitia (Fig. 1D, idiopathic preterm specimen). Syncytiotrophoblasts and cytotrophoblasts showed virtually no staining. Negative controls for TF staining using an isotype-matched nonspecific antibody revealed no signals (Fig. 1E). HSCORE analysis (Fig. 1F) indicated that, in both specimen groups, TF immunostaining intensity was strongest in the perimembranous area of DCs, in which it was significantly higher than in interstitial trophoblasts and the villous mesoderm (P < 0.05). Immunostaining in the villous mesoderm exhibited moderate intensity and was significantly higher than in interstitial trophoblasts (P < 0.05). There were no significant HSCORE differences between specimen groups in DCs (HSCORE mean ± sem: 200 ± 12 vs. 238 ± 12 in preterm vs. term specimens, respectively), interstitial trophoblasts (15 ± 5 vs. 26 ± 5 in preterm vs. term specimens, respectively), or the villous mesoderm (106 ± 17 vs. 136 ± 19 in preterm vs. term specimens, respectively).
Figure 1.
Immunohistochemical analysis of tissue factor expression at the decidual/placental interface. Serial sections of decidual basalis specimens were immunostained for TF and vimentin, as shown in an idiopathic preterm specimen (A, TF; B, vimentin). DCs (arrows), identified by positive vimentin staining, exhibited strong perimembranous TF staining. TF staining was virtually absent in interstitial trophoblasts (arrowheads). Similar results were seen in term specimens (C). Placental villi exhibited moderate TF staining in the mesoderm (M), in which the staining was primarily localized to perivascular adventitia (small arrows), as shown in a preterm specimen (D). Syncytiotrophoblasts (S) and cytotrophoblasts (C) exhibited no TF staining. Similar results were seen in term specimens (not shown). Negative control immunostaining using a nonspecific isotype-matched antibody revealed no positive signals (E). TF staining intensity (F); bars indicate HSCORE mean ± sem) among cell types was highest in DCs (*, vs. interstitial trophoblasts and the villous mesoderm of the same specimen group, P < 0.05), with moderate staining in the villous mesoderm (†, vs. interstitial trophoblasts of the same specimen group, P < 0.05). There were no significant differences in HSCOREs between specimen groups.
TF protein expression in cultured DCs
Figure 2 displays the separate and interactive effects of MPA and thrombin on immunoreactive levels of TF in cell lysates after incubation with leukocyte-free, term decidual cells. The choice of E2 as the control incubation for E2 plus MPA was based on elevated circulating E2 and progesterone levels during gestation. Compared with basal TF levels of 248.5 ± 69.3 pg/ml · μg cell protein, thrombin and MPA each significantly enhanced TF levels by more than 2-fold to 606.5 ± 150.7 pg/ml · μg cell protein for thrombin (P < 0.05) and 634 ± 171.5 pg/ml · μg cell protein for MPA (P < 0.05). The response to thrombin added with E2 + MPA was at least additive, compared with E2 alone, to 1976.1 ± 904 pg/ml · μg cell protein.
Figure 2.
Effects of MPA and thrombin on TF expression in DCs. Term DCs were primed in E2 or E2 + MPA and then switched to DM with corresponding steroids ± 2.5 U/ml thrombin (Th) for 24 h. ELISAs were performed for TF in cell lysates with the results normalized to total cell protein: n = 10 (mean ± sem). *, vs. E2; **, vs. E2 + MPA and E2 + Th; P < 0.05.
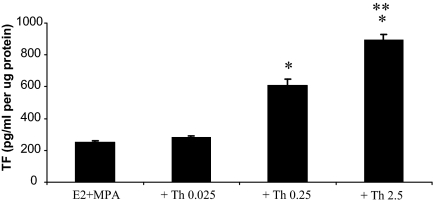
Figure 3 indicates that in cultured DCs that were primed with and then maintained in E2 plus MPA, thrombin elicited a concentration-dependent increase in TF expression with statistical significance attained at 0.25 U/ml (P < 0.05). Figure 4 reveals the specificity of thrombin-enhanced TF expression by demonstrating that in contrast with the significant elevation elicited by incubation with 2.5 U/ml of thrombin, neither IL-1β nor TNF-α significantly enhanced TF expression.
Figure 3.
Concentration-response effects of thrombin on TF expression in DCs. Term DCs were primed in E2 + MPA and then switched to DM with steroids ± thrombin (Th) at varying concentrations in units per milliliter for 24 h. ELISAs were performed for TF in cell lysates and measured by ELISA. Results are normalized to total cell protein (n = 3, mean ± sem). *, vs. E2 + MPA; **, vs. + Th 0.25; P < 0.05.
Figure 4.
Effects of thrombin, IL-1β, and TNF-α on TF expression in DCs. Term DCs were primed in E2 or E2 + MPA and then switched to DM with corresponding steroids ± 2.5 U/ml thrombin (Th) or 1 ng/ml of TNF-α or IL-1β for 24 h. ELISAs were performed for TF in cell lysates with the results normalized to total cell protein: n = 8 (mean ± sem). *, vs. E2 + MPA; P < 0.05.
Figure 5 displays the results of Western blotting for the presence in term DC lysates of TF (upper panel) and the β-actin loading control (lower panel). A single band was present that conformed to the mobility of authentic TF (47 kDa) and β-actin (42 kDa). Consistent with the ELISA results, thrombin and MPA each increased the magnitude of the TF band with greater induction elicited by E2 + MPA.
Figure 5.
Western blot of thrombin effects on TF output by DCs. Term DCs were primed in E2 or E2 + MPA and then switched to DM with corresponding steroids ± 2.5 U/ml thrombin (Th) for 24 h. Cell lysates were subjected to Western blotting after SDS-PAGE. See Patients and Methods for details.
TF mRNA expression in cultured DCs
Figure 6 displays TF mRNA levels in the term DC monolayers as determined by quantitative real-time RT-PCR. Compared with basal levels in term DCs treated with E2 alone, thrombin and MPA each significantly enhanced steady-state TF mRNA levels by 2.7 ± 0.2- and 3.4 ± 1.0-fold, respectively (mean ± sem; P < 0.05). The combination of E2 + MPA with thrombin increased TF mRNA levels by 5.4 ± 1.3-fold compared with E2 alone, (mean ± sem; n = 9; P < 0.05).
Figure 6.
Effects of MPA and thrombin on TF mRNA expression in DCs. Term DCs were primed in E2 or E2 + MPA and then switched to DM with corresponding steroids ± 2.5 U/ml thrombin (Th) for 6 h. TF mRNA was measured by quantitative real-time RT-PCR and normalized to β-actin mRNA; n = 9 (mean ± sem). *, vs. E2; P < 0.05.
Discussion
Our previous reports indicated that TF expression by perivascular DCs provides hemostatic protection against potentially pregnancy-terminating hemorrhage in the periimplantational period during EVT invasion of decidual capillaries and later during placentation when EVTs penetrate and remodel spiral arteries and arterioles (7,8). The current study indicates that such protection afforded by DCs can be extended to include the hemostatic demands of labor and delivery. Specifically, it observed more prominent TF immunostaining at the cell membranes of DCs than in neighboring cell types of the maternal-fetal interface in term placental sections. Moreover, the general concept that TF is constitutive in high expressing cells is contradicted by our observations that MPA and thrombin significantly enhanced TF expression in cultured, leukocyte-free term DCs.
In humans and mice, TF expression conforms to a similar tissue-specific pattern with low levels in liver, spleen, skeletal muscle, and thymus and high levels in skin, heart, kidney, lung, brain, uterus, and placenta. In low TF-expressing tissues, hemostasis depends primarily on the intrinsic coagulation pathway, which may explain why patients with hemophilia experience spontaneous hemorrhage into skeletal muscles (10). In tissues that express high levels of TF, the TF/VIIa complex of the extrinsic coagulation pathway provides enhanced hemostatic protection against potentially fatal hemorrhage via thrombin generation and resulting platelet activation and clot formation (6,10). The absence of any TF-deficient condition in humans emphasizes its importance to species survival and precludes direct evaluation of the effects of TF deficiency. Transgenic TF knockout mice develop fragile vessels and die of hemorrhage in uteri. However, incorporation of a human minigene-expressing TF at only 1% of its wild-type level rescued the knockouts to produce live born pups, although they do display decidual hemorrhage during gestation and have a high frequency of fatal postpartum hemorrhage. Adult low-TF mice experience hemostatic defects in tissues that normally express high levels of TF (6,10).
In the current study, IHC analysis of the maternal-fetal interface of preterm and term placental sections found that villous mesenchymal cells displayed significant TF immunostaining but with HSCORE values that were markedly lower than those for DCs (P < 0.05). In contrast to elevated TF expression in DCs, interstitial trophoblasts adjacent to, and intercalating with, DCs as well as villous and extravillous trophoblasts displayed little or no TF staining. Previously the only notable difference in TF expression between human and mice was observed in the kidney with TF localized in the glomerulus in humans and primarily in the tubules in mice (26). However, our findings indicate that unlike elevated expression of TF in mouse trophoblasts (9), but consistent with some reports in humans (11,12), third-trimester human trophoblasts express little or no TF. Moreover, it reveals for the first time that elevated TF expression by DCs exceeds that of other cell types at the maternal-fetal interface and is localized (primarily) at the cell membranes in which it is positioned to meet the hemostatic demands of labor and delivery by binding to factor VII and activating the extrinsic coagulation pathway via thrombin generation. The clinical implication of our finding is that in the high TF-expressing DCs impaired TF expression would result in potentially catastrophic hemorrhage across all of gestation.
In this report we also observed high TF expression in perivascular adventitial stromal cells at the maternal fetal interface of preterm and term placental sections. Such constitutively expressed TF forms a hemostatic envelope that prevents bleeding from damaged vessels (11,27). Although the expression of TF is generally characterized as constitutive in high TF expressing tissues (10), the current study found that MPA significantly enhanced TF mRNA and protein expression in leukocyte-free term DC monolayers. Our previous studies demonstrated that progestin induces TF expression during in vitro decidualization of human endometrial stromal cells by a mechanism involving the specificity protein-1 transcription factor (28) and that TF expression is elevated in DCs of endometrial sections from the progesterone-dominated luteal phase of the menstrual cycle and early pregnancy (3,4).
The integral role played by progesterone-induced myometrial quiescence in maintaining pregnancy is well established in humans as well as many other mammalian species (20,29). However, our previous and current results suggests that, in humans, progesterone also plays a crucial role in maintaining hemostasis throughout pregnancy and in the immediate postpartum period by chronically up-regulating TF expression. Strong evidence for this assertion includes hemorrhage associated with pregnancy complications linked to absent or impaired decidualization (i.e. ectopic pregnancy and placenta accreta). The lack of progesterone withdrawal in humans is crucial to the avoidance of potentially catastrophic hemorrhage during parturition and the immediate postpartum period. Indeed, considered from an evolutionary perspective, we posit that the highly invasive human hemochorial placenta renders humans particularly vulnerable to uteroplacental and decidual hemorrhage. Thus, the principal reason that progesterone withdrawal does not occur in human is likely to maintain decidual TF expression.
This study also found that thrombin significantly enhanced TF mRNA and protein expression in term DC monolayers with at least additive effects produced by coincubation with thrombin and MPA (P < 0.05). Unlike the response to thrombin, mediators of infection-associated PPROM and pre-term delivery, IL-1β and TNF-α (21,22,23,24) did not enhance TF expression when added at a concentration shown to markedly augment the expression of several end points in cultured term DCs (30,31,32). Previously we found that incubation of leukocyte-free term DCs with thrombin significantly augmented mRNA and protein levels of interstitial collagenase (MMP-1) (18) and stromelysin-1 (MMP-3) (17) as well as the primary neutrophil chemoattractant and activator IL-8 (33). We also observed a dense neutrophil infiltrate, in the decidua of placental sections from abruption-induced PPROM in the absence of infection (33). Although the association between PPROM and intraamniotic infections is well established, strong evidence also links PPROM with placental abruption in the absence of infection (19). Specifically the amniotic fluid of patients experiencing PPROM exhibits inflammation in nearly half of cases, but only half of these display evidence of infection (23).
In human pregnancies, the interactive effects of a fibrillar collagen-rich ECM in the amnion and choriodecidua provide greater than additive tensile strength and structural integrity that withstands disruptive forces stemming from the fetus and myometrium (34). However, these ECMs can be degraded by proteases derived from decidual neutrophils, decidual cells, or fetal membranes (35). Previous studies suggest that under sterile conditions, synergy between neutrophil-derived elastase, collagenase, and gelatinase (MMP-9) (36,37) with enhanced expression of MMP-1 and MMP-3 by thrombin in DCs (17,18) and relaxin in the fetal membranes (38,39) would promote PPROM by degrading the amnion and choriodecidual ECM. This intense proteolytic and proinflammatory milieu risks vascular disruption, which can then elicit catastrophic hemorrhage and/or exacerbation of the initiating abruption. Counteracting these pathological outcomes, our observation that thrombin augments TF expression in term DCs suggests the existence of a mechanism by which decidual hemorrhage triggers PTD to reduce maternal morbidity and mortality.
Several of the effects of MPA on cultured term DCs can be extrapolated to the results of a multicenter clinical trial demonstrating that administration of 17-hydroxyprogesterone caproate early in the second trimester reduces the incidence of pre-term delivery (40). Progestin-enhanced TF expression in term DCs demonstrated in the current study is expected to protect against potentially catastrophic hemorrhage during labor and delivery and postpartum (6,27). Moreover, inhibition of thrombin-enhanced MMP-1 and MMP-3 expression by progestin (17,18) should counteract the effects of abruption-associated PPROM and PTD. By contrast, the failure of MPA to neutralize thrombin-induced IL-8 expression in term DCs (33) suggests a limitation in the clinical efficacy of progesterone therapy in the setting of abruption.
Footnotes
This work was supported by Grants 2 R01HD33937-05 and 1 R01 HL070004-04 (to C.J.L.) from the National Institutes of Health.
Disclosure Summary: C.J.L., W.M., U.K., L.F.B., S.-T.H., E.F.F., G.K., and F.S. have nothing to declare.
First Published Online March 10, 2009
Abbreviations: DC, Decidual cell; DM, defined medium; E2, estradiol; ECM, extracellular matrix; EVT, extravillous trophoblast; HSCORE, Histologic Score; IHC, immunohistochemistry; MMP, matrix metalloproteinase; MPA, medroxyprogestrone acetate; PPROM, premature rupture of fetal membranes; TF, tissue factor.
References
- Bell SC 1990 Decidualization and relevance to menstruation. In: D'Arcangues C, Fraser IS, Newton JR, Odlind V, eds. Contraception and mechanisms of endometrial bleeding. Cambridge, UK: Cambridge University Press; 188 [Google Scholar]
- Tabanelli S, Tang B, Gurpide E 1992 In vitro decidualization of human endometrial stromal cells. J Steroid Biochem Mol Biol 42:337–344 [DOI] [PubMed] [Google Scholar]
- Lockwood CJ, Krikun G, Papp C, Toth-Pal E, Markiewicz L, Wang EY, Kerenyi T, Zhou X, Hausknecht V, Papp Z 1994 The role of progestationally regulated stromal cell tissue factor and type-1 plasminogen activator inhibitor (PAI-1) in endometrial hemostasis and menstruation. Ann NY Acad Sci 734:57–79 [DOI] [PubMed] [Google Scholar]
- Lockwood CJ, Nemerson Y, Guller S, Krikun G, Alvarez M, Hausknecht V, Gurpide E, Schatz F 1993 Progestational regulation of human endometrial stromal cell tissue factor expression during decidualization. J Clin Endocrinol Metab 76:231–236 [DOI] [PubMed] [Google Scholar]
- Runic R, Schatz F, Krey L, Demopoulos R, Thung S, Wan L, Lockwood CJ 1997 Alterations in endometrial stromal cell tissue factor protein and messenger ribonucleic acid expression in patients experiencing abnormal uterine bleeding while using Norplant-2 contraception. J Clin Endocrinol Metab 82:1983–1988 [DOI] [PubMed] [Google Scholar]
- Mackman N 2004 Role of tissue factor in hemostasis, thrombosis, and vascular development. Arterioscler Thromb Vasc Biol 24:1015–1022 [DOI] [PubMed] [Google Scholar]
- Moore KL 1988 Formation of the bilaminar embryo. In: Wonsiewicz M, ed. The developing human; clinically oriented embryology. 4th ed. Philadelphia: WB Saunders; 38–49 [Google Scholar]
- Pijnenborg R, Vercruysse L, Hanssens M 2006 The uterine spiral arteries in human pregnancy: facts and controversies. Placenta 27:939–958 [DOI] [PubMed] [Google Scholar]
- Erlich J, Parry GC, Fearns C, Muller M, Carmeliet P, Luther T, Mackman N 1999 Tissue factor is required for uterine hemostasis and maintenance of the placental labyrinth during gestation. Proc Natl Acad Sci USA 96:8138–8143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tilley R, Mackman N 2006 Tissue factor in hemostasis and thrombosis. Semin Thromb Hemost 32:5–10 [DOI] [PubMed] [Google Scholar]
- Drake TA, Morrissey JH, Edgington TS 1989 Selective cellular expression of tissue factor in human tissues. Implications for disorders of hemostasis and thrombosis. Am J Pathol 134:1087–1097 [PMC free article] [PubMed] [Google Scholar]
- Fleck RA, Rao LV, Rapaport SI, Varki N 1990 Localization of human tissue factor antigen by immunostaining with monospecific, polyclonal anti-human tissue factor antibody. Thromb Res 59:421–437 [DOI] [PubMed] [Google Scholar]
- Chaiworapongsa T, Espinoza J, Yoshimatsu J, Kim YM, Bujold E, Edwin S, Yoon BH, Romero R 2002 Activation of coagulation system in preterm labor and preterm premature rupture of membranes. J Matern Fetal Neonatal Med 11:368–373 [DOI] [PubMed] [Google Scholar]
- Elovitz MA, Baron J, Phillippe M 2001 The role of thrombin in preterm parturition. Am J Obstet Gynecol 185:1059–1063 [DOI] [PubMed] [Google Scholar]
- Rosen T, Kuczynski E, O'Neill LM, Funai EF, Lockwood CJ 2001 Plasma levels of thrombin-antithrombin complexes predict preterm premature rupture of the fetal membranes. J Matern Fetal Med 10:297–300 [DOI] [PubMed] [Google Scholar]
- Coughlin SR 2005 Protease-activated receptors in hemostasis, thrombosis and vascular biology. J Thromb Haemost 3:1800–1814 [DOI] [PubMed] [Google Scholar]
- Mackenzie AP, Schatz F, Krikun G, Funai EF, Kadner S, Lockwood CJ 2004 Mechanisms of abruption-induced premature rupture of the fetal membranes: Thrombin enhanced decidual matrix metalloproteinase-3 (stromelysin-1) expression. Am J Obstet Gynecol 191:1996–2001 [DOI] [PubMed] [Google Scholar]
- Rosen T, Schatz F, Kuczynski E, Lam H, Koo AB, Lockwood CJ 2002 Thrombin-enhanced matrix metalloproteinase-1 expression: a mechanism linking placental abruption with premature rupture of the membranes. J Matern Fetal Neonatal Med 11:11–17 [DOI] [PubMed] [Google Scholar]
- Harger JH, Hsing AW, Tuomala RE, Gibbs RS, Mead PB, Eschenbach DA, Knox GE, Polk BF 1990 Risk factors for preterm premature rupture of fetal membranes: a multicenter case-control study. Am J Obstet Gynecol 163:130–137 [DOI] [PubMed] [Google Scholar]
- Zakar T, Hertelendy F 2007 Progesterone withdrawal: key to parturition. Am J Obstet Gynecol 196:289–296 [DOI] [PubMed] [Google Scholar]
- Newton ER 2005 Preterm labor, preterm premature rupture of membranes, and chorioamnionitis. Clin Perinatol 32:571–600 [DOI] [PubMed] [Google Scholar]
- Saji F, Samejima Y, Kamiura S, Sawai K, Shimoya K, Kimura T 2000 Cytokine production in chorioamnionitis. J Reprod Immunol 47:185–196 [DOI] [PubMed] [Google Scholar]
- Shim SS, Romero R, Hong JS, Park CW, Jun JK, Kim BI, Yoon BH 2004 Clinical significance of intra-amniotic inflammation in patients with preterm premature rupture of membranes. Am J Obstet Gynecol 191:1339–1345 [DOI] [PubMed] [Google Scholar]
- Zaga-Clavellina V, Garcia-Lopez G, Flores-Herrera H, Espejel-Nunez A, Flores-Pliego A, Soriano-Becerril D, Maida-Claros R, Merchant-Larios H, Vadillo-Ortega F 2007 In vitro secretion profiles of interleukin (IL)-1β, IL-6, IL-8, IL-10, and TNF α after selective infection with Escherichia coli in human fetal membranes. Reprod Biol Endocrinol 5:46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arici A, Marshburn PB, MacDonald PC, Dombrowski RA 1999 Progesterone metabolism in human endometrial stromal and gland cells in culture. Steroids 64:530–534 [DOI] [PubMed] [Google Scholar]
- Luther T, Flossel C, Mackman N, Bierhaus A, Kasper M, Albrecht S, Sage EH, Iruela-Arispe L, Grossmann H, Strohlein A, Zhang Y, Nawroth PP, Carmeliet P, Loskutoff DJ, Muller M 1996 Tissue factor expression during human and mouse development. Am J Pathol 149:101–113 [PMC free article] [PubMed] [Google Scholar]
- Mackman N, Tilley RE, Key NS 2007 Role of the extrinsic pathway of blood coagulation in hemostasis and thrombosis. Arterioscler Thromb Vasc Biol 27:1687–1693 [DOI] [PubMed] [Google Scholar]
- Krikun G, Schatz F, Mackman N, Guller S, Demopoulos R, Lockwood CJ 2000 Regulation of tissue factor gene expression in human endometrium by transcription factors Sp1 and Sp3. Mol Endocrinol 14:393–400 [DOI] [PubMed] [Google Scholar]
- Mesiano S, Welsh TN 2007 Steroid hormone control of myometrial contractility and parturition. Semin Cell Dev Biol 18:321–331 [DOI] [PubMed] [Google Scholar]
- Lockwood CJ, Arcuri F, Toti P, Felice CD, Krikun G, Guller S, Buchwalder LF, Schatz F 2006 Tumor necrosis factor-α and interleukin-1β regulate interleukin-8 expression in third trimester decidual cells: implications for the genesis of chorioamnionitis. Am J Pathol 169:1294–1302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lockwood CJ, Matta P, Krikun G, Koopman LA, Masch R, Toti P, Arcuri F, Huang ST, Funai EF, Schatz F 2006 Regulation of monocyte chemoattractant protein-1 expression by tumor necrosis factor-α and interleukin-1β in first trimester human decidual cells: implications for preeclampsia. Am J Pathol 168:445–452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oner C, Schatz F, Kizilay G, Murk W, Buchwalder LF, Kayisli UA, Arici A, Lockwood CJ 2008 Progestin-inflammatory cytokine interactions affect matrix metalloproteinase-1 and -3 expression in term decidual cells: implications for treatment of chorioamnionitis-induced preterm delivery. J Clin Endocrinol Metab 93:252–259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lockwood CJ, Toti P, Arcuri F, Paidas M, Buchwalder L, Krikun G, Schatz F 2005 Mechanisms of abruption-induced premature rupture of the fetal membranes: thrombin-enhanced interleukin-8 expression in term decidua. Am J Pathol 167:1443–1449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore RM, Mansour JM, Redline RW, Mercer BM, Moore JJ 2006 The physiology of fetal membrane rupture: insight gained from the determination of physical properties. Placenta 27:1037–1051 [DOI] [PubMed] [Google Scholar]
- Parry S, Strauss 3rd JF 1998 Premature rupture of the fetal membranes. N Engl J Med 338:663–670 [DOI] [PubMed] [Google Scholar]
- Opdenakker G, Van den Steen PE, Dubois B, Nelissen I, Van Coillie E, Masure S, Proost P, Van Damme J 2001 Gelatinase B functions as regulator and effector in leukocyte biology. J Leukoc Biol 69:851–859 [PubMed] [Google Scholar]
- Helmig BR, Romero R, Espinoza J, Chaiworapongsa T, Bujold E, Gomez R, Ohlsson K, Uldbjerg N 2002 Neutrophil elastase and secretory leukocyte protease inhibitor in prelabor rupture of membranes, parturition and intra-amniotic infection. J Matern Fetal Neonatal Med 12:237–246 [DOI] [PubMed] [Google Scholar]
- Bryant-Greenwood GD, Kern A, Yamamoto SY, Sadowsky DW, Novy MJ 2007 Relaxin and the human fetal membranes. Reprod Sci 14:42–45 [DOI] [PubMed] [Google Scholar]
- Millar LK, Boesche MH, Yamamoto SY, Killeen J, DeBuque L, Chen R, Bryant-Greenwood GD 1998 A relaxin-mediated pathway to preterm premature rupture of the fetal membranes that is independent of infection. Am J Obstet Gynecol 179:126–134 [DOI] [PubMed] [Google Scholar]
- Meis PJ, Klebanoff M, Thom E, Dombrowski MP, Sibai B, Moawad AH, Spong CY, Hauth JC, Miodovnik M, Varner MW, Leveno KJ, Caritis SN, Iams JD, Wapner RJ, Conway D, O'Sullivan MJ, Carpenter M, Mercer B, Ramin SM, Thorp JM, Peaceman AM, Gabbe S 2003 Prevention of recurrent preterm delivery by 17α-hydroxyprogesterone caproate. N Engl J Med 348:2379–2385 [DOI] [PubMed] [Google Scholar]