Abstract
Our knowledge of the uptake and transport of dietary fat and fat-soluble vitamins has advanced considerably. Researchers have identified several new mechanisms by which lipids are taken up by enterocytes and packaged as chylomicrons for export into the lymphatic system or clarified the actions of mechanisms previously known to participate in these processes. Fatty acids are taken up by enterocytes involving protein-mediated as well as protein-independent processes. Net cholesterol uptake depends on the competing activities of NPC1L1, ABCG5, and ABCG8 present in the apical membrane. We have considerably more detailed information about the uptake of products of lipid hydrolysis, the active transport systems by which they reach the endoplasmic reticulum, the mechanisms by which they are resynthesized into neutral lipids and utilized within the endoplasmic reticulum to form lipoproteins, and the mechanisms by which lipoproteins are secreted from the basolateral side of the enterocyte. apoB and MTP are known to be central to the efficient assembly and secretion of lipoproteins. In recent studies, investigators found that cholesterol, phospholipids, and vitamin E can also be secreted from enterocytes as components of high-density apoB-free/apoAI-containing lipoproteins. Several of these advances will probably be investigated further for their potential as targets for the development of drugs that can suppress cholesterol absorption, thereby reducing the risk of hypercholesterolemia and cardiovascular disease.
Keywords: intestine, dietary fat, triacylglycerol, cholesterol, phospholipids, fat-soluble vitamins, microsomal triglyceride transfer protein
dietary fats consist of a wide array of polar and nonpolar lipids (32, 33). Triacylglycerol (TAG) is the dominant fat in the diet, contributing 90–95% of the total energy derived from dietary fat. Dietary fats also include phospholipids (PLs), sterols (e.g., cholesterol), and many other lipids (e.g., fat-soluble vitamins). The predominant PL in the intestinal lumen is phosphatidylcholine (PC), which is derived mostly from bile (10–20 g/day in humans) but also from the diet (∼1–2 g/day). The predominant dietary sterols are cholesterol (mostly of animal origin) and β-sitosterol (the major plant sterol). Although β-sitosterol accounts for 25% of dietary sterols, it is not absorbed by humans under physiological conditions.
Researchers have been investigating the various steps involved in lipid digestion, absorption, and metabolism to identify factors that could serve as targets for the development of drugs capable of reducing the risk of lipid-associated disorders, including dyslipidemias and cardiovascular disease. In so doing, they have explored key aspects of lipid digestion hydrolysis, emulsification, and micelle formation. They have also explored key issues of absorption, e.g., the uptake of the products of lipid hydrolysis by enterocytes (the epithelial cells lining the walls of the intestinal lumen) and their transport to intracellular compartments, where fatty acids and sterols are transformed into neutral lipids. Efficient absorption ensures that dietary fat is available to be used as a source of energy that supports various cellular functions or to be stored and serve as reservoirs for lipoprotein trafficking, bile acid synthesis, steroidogenesis, membrane formation and maintenance, and epidermal integrity in various mammalian cells. Excess fat is stored in cytosolic lipid droplets until it is needed to support intracellular processes.
In this review, we will concentrate on the biochemical processes involved in the digestion and absorption of the most common dietary lipids: TAGs, PLs, cholesterol, and fat-soluble vitamins.
DIGESTION AND ABSORPTION OF LIPIDS: A BRIEF OVERVIEW
The digestion of lipids begins in the oral cavity through exposure to lingual lipases, which are secreted by glands in the tongue to begin the process of digesting triglycerides. Digestion continues in the stomach through the effects of both lingual and gastric enzymes. The stomach is also the major site for the emulsification of dietary fat and fat-soluble vitamins, with peristalsis a major contributing factor. Crude emulsions of lipids enter the duodenum as fine lipid droplets and then mix with bile and pancreatic juice to undergo marked changes in chemical and physical form. Emulsification continues in the duodenum along with hydrolysis and micellization in preparation for absorption across the intestinal wall. (For details about the emulsification, hydrolysis, and micellization of fats, see Refs. 131 and 144.)
Bile and pancreatic juice provide pancreatic lipase, bile salts, and colipase, which function cooperatively to ensure the efficiency of lipid digestion and absorption. The importance of bile to the efficiency of these processes is indicated by the decreased rate of lipid absorption in humans with bile fistulas. The greatly reduced concentration of bile acid in the duodenum of such individuals suggests that bile salts, although possibly not absolutely necessary for digestion, are essential for the complete absorption of dietary fats (150). Elevated concentrations of bile salts have been shown to inhibit pancreatic lipase activity in the duodenum (190). Such inhibition is offset, however, by colipase, which has been shown in vitro to restore pancreatic lipase activity under such circumstances (112, 128). The importance of colipase in the digestion of fat was indicated in a clinical report of steatorrhea in two brothers with a congenital absence of colipase, which indicated that the steatorrhea decreased with the administration of colipase (76). It has also been demonstrated in colipase-deficient mice (41), which, when placed on a high-fat diet, developed steatorrhea that was so severe that undigested lipids could be seen in their feces. When these mice were placed on a low-fat diet, their ability to digest fat returned to normal. These findings suggest that colipase plays a critical, but not essential, role in the digestion of dietary lipids by pancreatic lipases.
Triacylglycerides
Digestion and absorption of TAGs.
TAG is digested primarily by pancreatic lipase in the upper segment of the jejunum. This process generates a liquid-crystalline interface at the surface of the emulsion particles (13, 161). The activity of pancreatic lipase on the sn-1 and sn-3 positions of the TAG molecule results in the release of 2-monoacylglycerol (2-MAG) and free fatty acids (FFAs) (122–124); 2-MAG is the predominant form in which MAG is absorbed from the small intestine. The formation of 2-MAG (and 1-MAG) through isomerization in an aqueous medium occurs more slowly than the uptake of 2-MAG from the small intestine (20). Further hydrolysis of 1- or 2-MAG by pancreatic lipase results in the formation of glycerol and FFAs (78); cholesterol esterase can also hydrolyze the acyl group at the sn-2 position to form glycerol and FFAs (111).
Free fatty acids are taken up from the intestinal lumen into the enterocytes and used for the biosynthesis of neutral fats. A protein-independent diffusion model and protein-dependent mechanisms have been proposed for the uptake and transport of fatty acids (FAs) across the apical membrane of the enterocyte (for details, see Ref. 119). A number of candidate proteins have been proposed to take part in protein-dependent uptake mechanisms. FAT/CD36 plays a key role in the uptake of FAs (1, 19). FAT/CD36 is highly expressed in the intestine (37), and its expression is upregulated by the presence of dietary fat (148), genetic obesity, and diabetes mellitus (64). Studies in CD36-null mice suggest that it is intimately involved in the uptake and transport of FAs targeted for transport to the lymphatic system through their use in the assembly of chylomicrons (45). This relationship is suggested by the finding that the uptake of FAs is not impaired in CD36-null animals. However, lipids tend to accumulate in the proximal small intestine of these animals primarily because of decreased transport of FAs to the lymphatic system (45). FA transport proteins (FATPs) are well represented in the small intestine by their FATP4 isoform, which is thought to help facilitate the uptake of FAs by the enterocytes (162).
TAG synthesis.
Once inside the enterocyte, the products of TAG hydrolysis must traverse the cytoplasm to reach the endoplasmic reticulum (ER), where they are used to synthesize complex lipids. Specific binding proteins carry FAs and MAG to the intracellular site, where they will be used for TAG biosynthesis. The two major fatty acid-binding proteins (FABPs) found in enterocytes are liver FABP and intestinal FABP (2, 16, 69). Most TAG biosynthesis in the enterocyte occurs along the MAG pathway, in which MAG and fatty acyl-CoA are covalently joined to form diacylglycerol (DAG) in a reaction catalyzed by monoacylglycerol acyltransferases (MGATs) (40, 197). Further acylation of DAG by diacylglycerol acyltransferase (DGAT) leads to the synthesis of TAG. Two DGATs have been identified and characterized: DGAT1 and DGAT2. [For additional information about DGATs, refer to the recent review (197).] DGAT2 is expressed mainly in the liver and intestine. Its importance in the absorption of fat has been demonstrated by a reduction in fat absorption in DGAT2-knockout (KO) animals. DGAT1, which is expressed in several tissues (including liver, intestine, and skin), differs from DGAT2 in that defective fat absorption is not seen in DGAT1-KO mice. In recent studies, investigators have found that MGAT2 (29, 31) and MGAT3 (30) possess DGAT activity (26, 170). Some TAG synthesis also occurs through the dephosphorylation of phosphatidic acid and acylation of the resultant DAG. Thus, several enzymes take part in the biosynthesis of TAG in intestinal cells.
Use of TAG in lipoprotein assembly.
The existence of multiple pools of DAG (159, 160) and TAG (51, 55, 60, 104, 175, 183, 195) has been known for some time. Using differentiated human colon carcinoma (Caco-2) cells, Luchoomun and Hussain (115) showed that nascent TAG is used preferentially for chylomicron assembly. TAG is also synthesized de novo from fatty acyl chains and glycerol phosphate. When generated through this pathway, however, TAG is only partly used to assemble nascent apolipoprotein (apo)B-containing lipoproteins. Most of the TAG synthesized through this pathway enters the cytosolic pool to be used to generate a distinct pool of DAG. (For a review of this topic, see Ref. 202.) DAG esterification also occurs in the ER lumen (142), where the resultant TAG binds to the microsomal triglyceride transport protein (MTP), which participates in the assembly and secretion of neutral lipids in chylomicrons. This process is described further in a subsequent section in this review. (For additional information, also see Refs. 18 and 119.)
Phospholipids
The predominant PL in the lumen of the small intestine is PC, which is found in mixed micelles that also contain cholesterol and bile salts. The digestion of PLs is carried out primarily by pancreatic phospholipase A2 (pPLA2) and other lipases secreted by the pancreas in response to food intake. These lipases interact with PLs at the sn-2 position to yield FFAs and lysophosphatidylcholine (21, 182). These products of lipolysis are removed from the water-oil interface when they are incorporated into the mixed micelles that form spontaneously when they interact with bile salts. Having both hydrophilic and hydrophobic components, bile salts are able to facilitate micelle formation; MAG and PL enhance their ability to form mixed micelles. pPLA2-KO mice are indistinguishable from wild-type controls when fed regular chow, except for their resistance to diet-induced obesity (84). Increased excretion of TAG, on a high-fat diet, in the feces of pPLA2-KO mice indicates that pPLA2 deficiency has a greater effect on the digestion of TAG than that of PL hydrolysis (84). It does not affect PL hydrolysis and absorption, possibly because its activity is compensated for by other PLA2 enzymes (158).
Cholesterol
Cholesterol in the body represents both endogenous sources (produced in the liver and peripheral tissues) and dietary sources absorbed from the intestine (186). The human diet provides ∼400 mg of cholesterol daily, and the liver secretes ∼1 g daily (66, 194). Approximately 50% of the cholesterol in the intestine is absorbed; the remainder is excreted in feces (10, 39).
Digestion.
Only nonesterified cholesterol can be incorporated into bile acid micelles and absorbed by enterocytes. Most dietary cholesterol exists in the form of the free sterol, with only 10–15% existing as the cholesteryl ester. The latter must be hydrolyzed by cholesterol esterase to release free cholesterol for absorption. Cholesterol is only minimally soluble in an aqueous environment (82, 177) and thus must be partitioned into bile salt micelles prior to absorption. It usually enters these micelles along with TAGs and PLs, ionized and nonionized FAs, MAGs, and lysophospholipids to form mixed micelles (74, 196). These micelles are transported to the brush border of the enterocyte, where cholesterol is absorbed. Its absorption depends on the presence of bile acids in the intestinal lumen (191) and correlates directly with the total bile acid pool (149).
Absorption: cholesterol uptake.
Cholesterol must pass through a diffusion barrier at the intestinal lumen-enterocyte membrane interface before it can interact with transporter proteins responsible for its uptake and subsequent transport across the cellular brush border. Bile salt micelles facilitate the transfer of cholesterol across the unstirred water layer. The mechanism by which the cholesterol in micelles is taken up by the cell and crosses the brush-border membrane is still under investigation. Cholesterol absorption had long been considered an energy-independent, simple, passive diffusion process. However, it is taken up by the enterocyte with relatively high efficiency compared with structurally similar phytosterols (130). New evidence strongly suggests that a transporter-facilitated mechanism is involved. Interindividual differences and interstrain variations in the efficiency of intestinal cholesterol absorption (195, 196) support this suggestion, as does the discovery that multiple genes (4, 14, 106, 113) participate in the regulation of cholesterol absorption and several molecules appear to inhibit it (59).
Absorption: cholesterol transport.
Several proteins have been investigated for their potential roles as intestinal cholesterol transporters, but evidence for their direct role in the uptake of cholesterol remains elusive. Studies in genetically modified animal models have helped investigators gain important insights into the mechanisms of transport and identity of transporter proteins. Through these studies, investigators have identified Niemann-Pick C1 like 1 (NPC1L1) as a cholesterol uptake transporter (4) and the ATP-binding cassette (ABC) proteins ABCG5 and ABCG8 as cholesterol efflux transporters (14, 106, 113). These three molecules appear to be key players in the control of the cholesterol absorption from the intestinal lumen.
ABCG5 and ABCG8, which function as a heterodimer (62), are critical for the control of sterol absorption. Mutations in the genes encoding human ABCG5 and ABCG8 transporters cause β-sitosterolemia (14, 106, 113), which is characterized by the accumulation of plant sterols in blood and other tissues as a result of their enhanced absorption from the intestines and decreased removal in bile. These proteins are localized at the canalicular membrane of hepatocytes and at the brush border of enterocytes. Abcg5/g8 deficiency in mice results in reduced biliary cholesterol secretion (199) and enhanced phytosterol absorption (102, 146, 199) but has only minimal effects on the efficiency of cholesterol absorption (146, 199). The pharmacological induction or overexpression of Abcg5 and Abcg8 in mice (199–201) results in a reduction in fractional cholesterol absorption (i.e., the percentage of cholesterol absorbed from the intestine, which is determined using a dual-isotope feeding technique) and indicates that ABCG5 and ABCG8 play a role in the control of cholesterol absorption under certain conditions.
The identification of NPC1L1 as a putative cholesterol transporter in the enterocytes (4) was facilitated by the discovery of the cholesterol absorption inhibitor ezetimibe (4, 59), which reduces diet-induced hypercholesterolemia (49, 56, 103, 187, 203). NPC1L1 is a glycosylated protein localized at the brush-border membrane of the enterocyte (95). The deletion of Npc1l1 in mice results in a reduction in fractional cholesterol absorption (4). Ezetimibe has been shown to bind to NPC1L1-expressing cells and to the intestinal brush border (59). Deletion of Npc1l1 also results in the elimination of the binding capacity of the brush border (59), which indicates that NPC1L1 is a target of ezetimibe.
A sterol regulatory element in the promoter and a sterol-sensing domain of NPC1L1 appear to regulate cholesterol absorption in response to cholesterol intake. Expression of Npc1l1 is enhanced in the cholesterol-depleted porcine intestine and suppressed in mice placed on a cholesterol-rich diet (83). Most of the NPC1L1 in the body is found in intracellular membranes. However, cholesterol deprivation induces its translocation to the plasma membrane, where it can pick up cholesterol and transport it to the ER for esterification and packaging into nascent lipoproteins (50, 198).
Reducing the expression of NPC1L1 at the level of transcription may reduce cholesterol absorption. Activation of the nuclear receptor peroxisome proliferator-activated receptor (PPAR)δ/β by the synthetic agonist GW610742 has been shown to reduce cholesterol absorption by decreasing Npc1l1 expression without altering the expression of Abcg5 and Abcg8 (185). A decrease in Npc1l1 expression has also been observed following treatment of human colon-derived Caco-2 cells with ligands for PPARδ/β but not for PPARγ or PPARα (185).
Absorption: other regulatory factors.
The nuclear liver X receptors (LXRs) LXRα (expressed mainly in the liver, kidney, intestine, spleen, and adrenals) and LXRβ (expressed ubiquitously) regulate pathways involved in the metabolism of cholesterol and in lipid biosynthesis. LXR target genes have been shown to be involved in cholesterol and lipid homeostasis. (For a list of target genes and their regulation, see Ref. 174.) After activation by natural ligands (e.g., oxysterols), the LXR forms a heterodimer with the retinoid X receptor (96, 97) and binds to specific LXR response elements in the promoter regions of their target genes to activate gene transcription. LXR target genes include those that express proteins involved in the efflux of cholesterol from the cell (155, 157, 174, 189) as well as bile acid synthesis (174) and lipogenesis (174). Thus, global LXR activation by synthetic agonists has a plethora of effects, including elevated high-density lipoprotein (HDL) levels (25, 28, 98, 126, 147, 163, 178), hypertriglyceridemia (156, 163), hepatic steatosis (65), increased excretion of cholesterol in bile (147, 201), reduced efficiency of cholesterol absorption from the intestine (103, 157, 159, 191, 206), and increased loss of neutral sterols in feces (201).
Other transporters, such as scavenger receptor class B type I (SR-BI), which is localized both at the apical and basolateral membranes of enterocytes (27), have also been suspected of having a role in the control of cholesterol absorption. The possibility that SR-BI is a cholesterol transporter is suggested by the observation that intestine-specific overexpression of SR-BI in mice leads to an increase in cholesterol and TAG absorption in short-term absorption experiments (17). The importance of the FA translocase CD36 (which is also expressed in epithelial cells lining the small intestine) in FA absorption is well established. CD36 has also been implicated as the cholesterol transporter in brush-border membranes. Additionally, overexpression of CD36 in COS-7 cells has been shown to enhance cholesterol uptake from micellar substrates (184). In another study, CD36-null mice showed a significant reduction in cholesterol transport from the intestinal lumen to the lymphatic system (133). Decreased cholesterol uptake has been shown in brush-border membrane vesicles prepared from the proximal (but not distal) intestine of SR-BI-KO mice (184) and in brush-border membrane vesicles and Caco-2 cells preincubated with antibodies to SR-BI (71). However, targeted disruption of SR-BI in mice has little effect on in vivo intestinal cholesterol absorption (3, 120, 192), which suggests that SR-BI might not be essential for absorption of cholesterol from the intestine.
Another potential step in the regulation of cholesterol absorption involves two ER membrane-localized enzymes, acyl-CoA:cholesterol acyltransferase 1 (ACAT1) and ACAT2, that catalyze the esterification of intracellular cholesterol (107). ACAT1 is expressed in many tissues (36, 61), but its level of expression in the mouse small intestine is very low (125). By contrast, ACAT2 expression is restricted to the small intestine and liver (6, 35, 137). ACAT2 is highly specific for cholesterol and does not esterify plant sterols. It is the predominant enzyme responsible for the synthesis of cholesterol esters and their subsequent secretion with lipoproteins. ACAT2 deficiency results in a considerable decrease in the rate of cholesterol absorption (26). The rate of cholesterol esterification in the presence of these enzymes is notably enhanced by substrate availability, but, as we demonstrated recently, it is inhibited by product accumulation. This inhibition is relieved by MTP (94), which transfers cholesterol esters from the ER membranes to nascent apoB-lipoproteins. The importance of MTP in cholesterol absorption has been well documented (85, 86, 89).
Approximately 70–80% of cholesterol entering the lymphatic system is esterified, which suggests that esterification is important for bulk entry of cholesterol into nascent chylomicrons. Reesterification of the absorbed cholesterol within the enterocyte enhances the diffusion gradient to favor the entry of intraluminal cholesterol into the cell and is therefore an important regulator of cholesterol absorption from the intestine. Thus, inhibition of ACAT by pharmacological intervention (38, 73) or deletion of Acat2 (26) significantly reduces the rate of cholesterol absorption.
It has also been suggested that ATP-binding cassette transporter A1 (ABCA1) plays a role in the control of cholesterol absorption. ABCA1 was initially thought to be localized to the apical membrane (157); more recent studies have provided evidence suggesting that it is present in the basolateral membrane of chicken enterocytes (132) and human Caco-2 cells (138). Our studies in Caco-2 cells and in the apoA1-KO mouse model showed for the first time that basolateral efflux (secretion) of cholesterol occurs in high-density apoB-free/apoA-I-containing lipoproteins (Fig. 1) (91, 93). The importance of ABCA1 in the biogenesis of HDL in the intestine was demonstrated by deleting intestinal Abca1, which resulted in a 30% decrease in plasma HDL cholesterol levels (24). Enterocytes deficient in ABCA1 absorb smaller amounts of cholesterol (24). Thus, HDL contributes considerably to cholesterol absorption from the intestine. The origin of HDL particles in mesenteric lymph fluid has been a subject of considerable controversy (11, 53, 117, 139, 152, 169, 188). The measurement of cholesterol in lymph led to the hypothesis that HDL is not important in cholesterol transport. However, Brunham et al. (24) recently demonstrated that the HDL in lymph is dependent on the activity of hepatic ABCA1 and enters the lymph from plasma. Thus, quantification of HDL in lymph may not provide an accurate assessment of the extent of cholesterol absorption via this pathway.
Fig. 1.
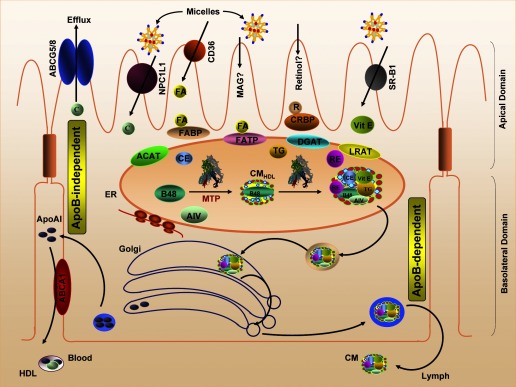
Intestinal lipid absorption. Products of lipid hydrolysis are solubilized in micelles and presented to the apical membranes of enterocytes. This membrane harbors several transport proteins that participate in the uptake of various types of lipids. Niemann-Pick C1 like 1 (NPC1L1) is a protein involved in cholesterol uptake. CD36 and fatty acid (FA) transport protein (FATP) have been shown to participate in FA transport, whereas scavenger receptor class B type I (SR-BI) is involved in vitamin E (Vit E) uptake. In the cytosol, FA-binding protein (FABP) and cellular retinol-binding protein (CRBP) transport FAs and retinol (R), respectively. Acyl-CoA:cholesterol acyltransferase (ACAT), diacylglycerol acyltransferase (DGAT), and lecithin:retinol acyltransferase (LRAT) are found in the endoplasmic reticulum (ER) membrane, where they facilitate the esterification of cholesterol, monoacylglycerols (MAG), and retinol, respectively. These esterified products are incorporated into apolipoprotein (apo)B48-containing chylomicrons (CM) in a microsomal triglyceride (TG) transport protein (MTP)-dependent manner. The newly synthesized prechylomicrons are transported in specialized vesicles to the Golgi apparatus for further processing and for secretion. In addition, enterocytes express ATP-binding cassette (ABC) transporter A1 on the basolateral membrane to facilitate the efflux of cholesterol. C, free cholesterol; RE, retinyl ester; CE, cholesteryl ester; AIV, apolipoprotein A-IV.
Vitamin E
Vitamin E is one of the most abundant lipid-soluble antioxidants found in the plasma and somatic cells of higher mammals. Vitamin E exists in two forms, as tocopherols and tocotrienols, that vary dramatically in terms of biological activity due to variations in bioavailability and in intrinsic antioxidant properties. The hydrophobic nature of vitamin E creates a major challenge to living organisms in maintaining adequate uptake, transport, and delivery of this vitamin to various tissues.
Vitamin E absorption.
Micelle formation is required for the absorption of vitamin E (57, 171). Pancreatic enzymes may also aid in its absorption (121); this is suggested by the finding of vitamin E deficiency in patients with cystic fibrosis, who do not secrete pancreatic enzymes (12, 48, 70, 172, 193).
The mechanism of absorption of vitamin E from the intestine is similar to the mechanism involved in the transport of other lipid molecules in vivo and involves molecular, biochemical, and cellular mechanisms closely related to overall lipid and lipoprotein homeostasis (22, 67, 75, 100, 101, 179–181). The uptake of vitamin E from the intestine has traditionally been assumed to be a simple process of passive diffusion. However, in a study in Caco-2 cells, it was shown to be a rapid, saturable, temperature-dependent process (7). Recent studies suggest that SR-BI plays a role in vitamin E absorption (154). Vitamin E absorption from the intestine is thought to occur predominantly through the secretion of chylomicrons into the lymphatic system. Consistent with this hypothesis is the observation that vitamin E absorption is dependent on the availability of oleic acid for triglyceride synthesis and chylomicron assembly and is inhibited by MTP inhibitors (7). The importance of alternative pathways for vitamin E absorption has also been suggested by the observation that symptoms of vitamin E deficiency are ameliorated after treatment with high doses of vitamin E in patients deficient in chylomicrons. Indeed, intestinal vitamin E absorption may also occur via direct secretion from epithelial cells into the portal venous circulation by HDL efflux. This HDL-dependent mechanism was recently characterized in Caco-2 cells (7). Whether intestinal ABCA1 or other ABC transporters are also critical for the absorption of vitamin E from the intestine and for steady-state plasma α-tocopherol levels remains to be seen.
Vitamin A.
The de novo synthesis of compounds with vitamin A activity is limited to plants and microorganisms. Higher animals must obtain vitamin A from the diet. The main dietary forms of preformed vitamin A are carotenoids in fruits and vegetables and long-chain FA esters of retinol in foods of animal origin (145). Carotenoids are either cleaved to generate retinol or absorbed intact, whereas retinyl esters must be completely hydrolyzed within the intestinal lumen to release free retinol before retinol can be taken up by enterocytes. Hydrolysis of the retinyl esters requires lipase activity. The products of TAG hydrolysis provide a better milieu for the solubilization of vitamin E in mixed micelles.
Free retinol is taken up by intestinal mucosal cells (44). In studies conducted in Caco-2 cells (151) and intestinal segments, investigators have found that saturable carrier-mediated processes at physiological concentrations (81) and nonsaturable diffusion-dependent processes at pharmacological concentrations (80) of free retinol are involved in retinol uptake. After cellular uptake, the intercellular retinol-binding proteins (CRBPs) CRBP-I and CRBP-II immediately sequester free retinol. CRBP-I is expressed in many tissues, whereas CRBP-II is expressed primarily in the absorptive cells of the small intestine. CRBP-II is one of the most abundant soluble proteins in the jejunal mucosa, which indicates that it may be uniquely suited for the absorption of retinol from the intestine (109, 136, 140, 141). In studies conducted in retinoid-deficient rats (153) and in rats placed on diets containing long-chain FAs (176), investigators found that CRBP-II mRNA expression increased in the small intestine. In studies carried out in Caco-2 cells, investigators found that overexpression of CRBP-II or treatment with retinoic acid (which is associated with increased CRBP-II mRNA expression) resulted in increased absorption and intracellular esterification of retinol. In CRBP-II-KO mice, investigators found that CRBP-II plays an important (albeit not essential) role in the absorption of vitamin A from the intestine (47). Some of the free retinol in the enterocytes remains associated with CRBP, but much of the retinol is usually esterified by lecithin:retinol acyltransferases (LRATs) and stored within the cell (116). The recent characterization of the LRAT-KO mouse [in which no detectable tissue retinyl esters were found (9)] has largely resolved the question of whether enzymes such as ARAT are physiologically involved in retinol esterification in the intestine.
Metabolic studies have revealed that most of the retinyl esters in plasma are present in small chylomicrons; significant amounts are found in large chylomicrons, and smaller amounts are found in very-low-density lipoproteins (VLDL) (108). Studies have been carried out in differentiated Caco-2 cells under postprandial conditions to determine the mechanism of retinol secretion by the intestine. In these studies, investigators found that a significant amount of retinyl ester was secreted mainly with chylomicrons independent of the rate of retinol uptake and intracellular levels of free or esterified retinol (134). Pluronic L81, which inhibits the secretion of chylomicrons, decreased the secretion of retinyl ester and did not result in their increased secretion with smaller lipoproteins. This suggests that intestinal retinyl ester secretion is a highly specific and regulated process that is dependent on the assembly and secretion of chylomicrons. A significant amount of retinol is also secreted into the portal circulation, probably as free retinol; this is expected to be physiologically significant in pathological conditions that affect the secretion of chylomicrons (79). However, very little is known about the regulation of the retinol secretion by this pathway. Under fasting conditions, Caco-2 cells secrete mainly free retinol unassociated with lipoproteins (134). The secretion of free retinol may require facilitated transport. This notion is supported by the marked inhibition of the free retinol efflux into the basolateral medium by glyburide, a known inhibitor of the ABCA1 transporter (46). Mechanisms regulating retinol output through these pathways are not well understood.
ASSEMBLY, SECRETION, AND REGULATION OF INTESTINAL LIPOPROTEINS
The major lipoproteins secreted by the intestine are VLDL and chylomicrons. Of these, the chylomicrons are synthesized exclusively in the intestine to transport dietary fat and fat-soluble vitamins into the blood (Fig. 1). Chylomicrons are primarily very large, spherical TAG-rich particles (5, 58) that also contain PLs, cholesterol, vitamin E, vitamin A, and protein. The lipoprotein core contains TAG, cholesteryl esters, and fat-soluble vitamins, whereas the surface contains a monolayer of PLs (mainly phosphatidylcholine), free cholesterol, and protein (127, 204, 205). A key structural component of the chylomicron is the huge, hydrophobic, nonexchangeable protein apoB48. Other proteins associated with nascent chylomicrons (which are synthesized in enterocytes) are apoA-I, apoA-IV, and apoCs. apoB48 synthesis is generally believed to be constitutive (88). However, the amount of lipids transported during the postprandial state is severalfold greater than that transported during the fasting state. Increased transport of fat with similar amounts of apoB48 occurs because of an increase in the size of the particles during the postprandial state (42, 43, 54, 63, 72, 173). Fat feeding also increases the expression of apoA-IV, which serves as a surface component for apoB48 particles in the enterocyte (77, 135).
It has been proposed that chylomicron assembly involves the synthesis of “primordial lipoproteins” followed by their core expansion, which results in the formation of “nascent lipoproteins” (88). This basic proposal was later expanded to suggest that chylomicron assembly may involve three independent steps: assembly of primordial lipoproteins, formation of lipid droplets, and core expansion (85). Subsequently, different biochemical signposts for various biosynthetic milestones were articulated (89). The association of a preformed PL with nascent apoB was proposed to determine the assembly of primordial lipoproteins. An increase in the amount of nascent TAG would serve as an indicator of the synthesis of larger lipoproteins, and retinyl esters were proposed as markers of the completion of chylomicron assembly.
Chylomicron assembly occurs mainly in the ER. These particles are then transported to the cis-Golgi in prechylomicron transport vesicles (PCTVs) (18, 105, 119). Budding of the PCTVs has been shown to require liver FABP (135). Prechylomicrons are very large and carry a unique cargo compared with vesicles that carry nascent proteins (205). Siddiqi et al. (165) showed that chylomicrons are exported from the ER in large vesicles (250 nm) by a coat protein complex II (COPII)-independent mechanism, although COPII proteins are found on PCTVs. These investigators demonstrated further that COPII-interacting proteins (Sar1, Sec23/24, and Sec13/31) are needed to form lipid vesicles that can fuse with the Golgi complex. The unique presence of vesicle-associated membrane protein 7 in the intestinal ER and on PCTVs has been suggested to play a role in the export of chylomicrons from the ER to the cis-Golgi (166). Recently, it was shown that an isoform of protein kinase C (PKC), PKCζ, is required for PCTV budding (167). Thus, chylomicrons are transported on unique particles, different from those used for protein transport, by intestinal cells. Similarly, unique particles have been shown to transport hepatic lipoproteins in the liver (164).
Various mechanisms have been shown to regulate the secretion of lipoproteins from the intestine. Intestinal lipoprotein production is affected by the transcription of apoB, which has generally been believed to be constitutive. Previously, it was known that apoB levels change primarily through co- and posttranslational mechanisms; however, Singh et al. (168) showed that apoB secretion is affected by a modest change in its transcription. In recent studies, investigators have found that apoA-IV levels also affect intestinal lipoprotein assembly and secretion. Increased secretion of nascent TAG and PLs with chylomicrons has been shown in intestinal porcine epithelial cells (a newborn swine enterocyte cell line) overexpressing apoA-IV (114). Similarly, apical supplementation of Caco-2 cells with lipid micelles has been shown to increase apoA-IV mRNA and protein levels, which, in turn, facilitates lipid secretion (34).
Lipoprotein assembly and secretion is also regulated by MTP, which transfers several lipids and assists in the formation of primordial apoB lipoproteins (87, 90). MTP is modulated by various factors and is typically regulated at the transcriptional level (68). The initial incorporation of lipids into apoB by MTP prevents it from proteosome-mediated degradation. Genetic deficiency or pharmacological inhibition of MTP allows continued synthesis of apoB, but the protein is misfolded and is thus destroyed by ER-associated degradation (52, 110). Changes in MTP activity affect plasma apoB lipoprotein levels significantly. In an in vitro cell-free system, investigators found that MTP plays a pivotal role in facilitating lipid recruitment by acting as a chaperone to assist in apoB folding (99). They also found evidence suggesting that PL recruitment is intrinsic in the NH2-terminal domain of apoB during the translational process and may facilitate protein folding. Recently, we showed that a deficiency in inositol-requiring enzyme 1β (IRE1β) in mice placed on a high-fat, high-cholesterol diet enhances intestinal MTP expression, which leads to increased lipid absorption and chylomicron secretion (92). IRE1β was shown to cause endolytic cleavage of MTP mRNA between exons 2 and 7. The cleaved products are subsequently degraded by 3′-5′ Ski2 nuclease and 5′-3′-exoribonuclease (XRN)1 and XRN2 exonucleases. Thus, IRE1β decreases MTP mRNA levels by enhancing its posttranscriptional degradation. Three potential mechanisms of MTP mRNA degradation have been proposed: 1) because both IRE1β and MTP mRNA translation are translated within the ER membrane, it is possible that, under the conditions that increase MTP mRNA synthesis (e.g., high cholesterol and lipid levels), IRE1β and MTP become juxtaposed and thus facilitate the specific degradation of MTP mRNA; 2) IRE1β may activate or recruit another nuclease that initiates MTP mRNA degradation; or 3) under the conditions of increased chylomicron assembly, the protein translational machinery is temporarily stalled, leading to the recognition of the MTP mRNA by endoribonucleases and its subsequent degradation. IRE1β is a mammalian ER stress sensor protein with prominent expression within intestinal epithelial cells. Within intestine, IRE1β protein is detectable in the stomach, small intestine, and colon. Furthermore, it is expressed mainly in the intestinal epithelial cells (15). By modulating MTP mRNA levels and, consequently, the extent of apoB lipidation, IRE1β may 1) protect the ER from the consequences of rapid fluctuations in membrane PL, neutral lipids, or vitamin E levels or 2) mediate downregulation of chylomicron secretion in response to satiety signals or respond to endocrine factors released by the liver and adipose tissue in the presence of excess accumulation of lipid throughout the body. Thus, modulation of IRE1β activity can provide a way to modulate intestine-specific regulation of lipoprotein assembly and lipid absorption. In this mechanism, upregulation of IRE1β may be useful for avoiding a diet-induced hyperlipidemia.
Intestinal lipid absorption has been shown to exhibit diurnal variations in rodents and humans (8, 23, 118, 129, 143). Although plasma lipid concentrations are maintained within a narrow physiological range, several factors (e.g., plasma clearance, cholesterol biosynthesis, and hormonal changes) can lead to changes in plasma lipids. It was recently demonstrated that diurnal variations in plasma lipid levels in rodents are due to changes in MTP levels (143). Lipid absorption was higher at 2400 than at 1200 because of the rodents' nocturnal feeding behavior. Using in situ loops and isolated enterocytes, it was demonstrated that circadian variations were due to changes in intestinal activities and not because of variations in gastric functions. Further studies have revealed that intestinal expression of MTP also has diurnal variations. Consistent with this finding, the transcription rate for microsomal triglyceride transfer protein (mttp) was high at 2400 compared with 1200. Thus, it appears that MTP expression and lipid absorption are maximized to absorb more lipids at mealtime. The diurnal variations in MTP, in turn, may be responsible for the change in total plasma lipids and apoB lipoproteins (143).
CONCLUSIONS
The absorption of lipids from the intestinal lumen into the enterocytes and their subsequent secretion into circulation is a complex process. Membrane transporters regulate lipid uptake on the apical surface of the brush border of the enterocyte. An essential role for NPC1L1 in cholesterol absorption and for the coordinated activities of ABCG5 and ABCG8 in limiting excess cholesterol uptake is well established. Several proteins involved in FA uptake have also been identified. Transport of lipids from the plasma membrane to the ER involves the activity of intracellular trafficking proteins. Lipids that have been absorbed into the ER are resynthesized and packaged into chylomicrons; this process is dependent on apoB and MTP activity. Specialized vesicles carry these chylomicrons to the basolateral membrane of the cell for secretion. Posttranscriptional degradation of MTP by IRE1β provides a way to modulate intestine-specific regulation of lipoprotein assembly and absorption and may be useful for avoiding diet-induced hyperlipidemias. Recent findings concerning the basolateral efflux of cholesterol as high-density apoA-I-containing lipoproteins will help us identify additional targets for the development of drugs that may suppress cholesterol absorption to reduce hypercholesterolemia and the risk for cardiovascular disease.
GRANTS
This work was supported in part by National Institute of Diabetes and Digestive and Kidney Diseases Grant DK-46700 and a Grant-in-Aid from the American Heart Association to M. M. Hussain.
REFERENCES
- 1.Abumrad NA CD36 may determine our desire for dietary fats. J Clin Invest 115: 2965–2967, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Agellon LB, Toth MJ, Thomson AB. Intracellular lipid binding proteins of the small intestine. Mol Cell Biochem 239: 79–82, 2002. [PubMed] [Google Scholar]
- 3.Altmann SW, Davis HR Jr, Yao X, Laverty M, Compton DS, Zhu LJ, Crona JH, Caplen MA, Hoos LM, Tetzloff G, Priestley T, Burnett DA, Strader CD, Graziano MP. The identification of intestinal scavenger receptor class B, type I (SR-BI) by expression cloning and its role in cholesterol absorption. Biochim Biophys Acta 1580: 77–93, 2002. [DOI] [PubMed] [Google Scholar]
- 4.Altmann SW, Davis HR Jr, Zhu LJ, Yao X, Hoos LM, Tetzloff G, Iyer SP, Maguire M, Golovko A, Zeng M, Wang L, Murgolo N, Graziano MP. Niemann-Pick C1 Like 1 protein is critical for intestinal cholesterol absorption. Science 303: 1201–1204, 2004. [DOI] [PubMed] [Google Scholar]
- 5.Anderson LJ, Boyles JK, Hussain MM. A rapid method for staining large chylomicrons. J Lipid Res 30: 1819–1824, 1989. [PubMed] [Google Scholar]
- 6.Anderson RA, Joyce C, Davis M, Reagan JW, Clark M, Shelness GS, Rudel LL. Identification of a form of acyl-CoA:cholesterol acyltransferase specific to liver and intestine in nonhuman primates. J Biol Chem 273: 26747–26754, 1998. [DOI] [PubMed] [Google Scholar]
- 7.Anwar K, Kayden HJ, Hussain MM. Transport of vitamin E by differentiated Caco-2 cells. J Lipid Res 47: 1261–1273, 2006. [DOI] [PubMed] [Google Scholar]
- 8.Balasubramaniam S, Szanto A, Roach PD. Circadian rhythm in hepatic low-density-lipoprotein (LDL)-receptor expression and plasma LDL levels. Biochem J 298: 39–43, 1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Batten ML, Imanishi Y, Maeda T, Tu DC, Moise AR, Bronson D, Possin D, Van Gelder RN, Baehr W, Palczewski K. Lecithin-retinol acyltransferase is essential for accumulation of all-trans-retinyl esters in the eye and in the liver. J Biol Chem 279: 10422–10432, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bays H Ezetimibe. Expert Opin Investig Drugs 11: 1587–1604, 2002. [DOI] [PubMed] [Google Scholar]
- 11.Bearnot HR, Glickman RM, Weinberg L, Green PH, Tall AR. Effect of biliary diversion on rat mesenteric lymph apolipoprotein-I and high density lipoprotein. J Clin Invest 69: 210–217, 1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bennett MJ, Medwadowski BF. Vitamin A, vitamin E, and lipids in serum of children with cystic fibrosis or congenital heart defects compared with normal children. Am J Clin Nutr 20: 415–421, 1967. [DOI] [PubMed] [Google Scholar]
- 13.Benzonana G, Desnuelle P. Kinetic study of the action of pancreatic lipase on emulsified triglycerides. Enzymology assay in heterogeneous medium. Biochim Biophys Acta 105: 121–136, 1965. [PubMed] [Google Scholar]
- 14.Berge KE, Tian H, Graf GA, Yu L, Grishin NV, Schultz J, Kwiterovich P, Shan B, Barnes R, Hobbs HH. Accumulation of dietary cholesterol in sitosterolemia caused by mutations in adjacent ABC transporters. Science 290: 1771–1775, 2000. [DOI] [PubMed] [Google Scholar]
- 15.Bertolotti A, Wang X, Novoa I, Jungreis R, Schlessinger K, Cho JH, West AB, Ron D. Increased sensitivity to dextran sodium sulfate colitis in IRE1beta-deficient mice. J Clin Invest 107: 585–593, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Besnard P, Niot I, Poirier H, Clement L, Bernard A. New insights into the fatty acid-binding protein (FABP) family in the small intestine. Mol Cell Biochem 239: 139–147, 2002. [PubMed] [Google Scholar]
- 17.Bietrix F, Yan D, Nauze M, Rolland C, Bertrand-Michel J, Comera C, Schaak S, Barbaras R, Groen AK, Perret B, Terce F, Collet X. Accelerated lipid absorption in mice overexpressing intestinal SR-BI. J Biol Chem 281: 7214–7219, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Black DD Development and Physiological Regulation of Intestinal Lipid Absorption. I. Development of intestinal lipid absorption: cellular events in chylomicron assembly and secretion. Am J Physiol Gastrointest Liver Physiol 293: G519–G524, 2007. [DOI] [PubMed] [Google Scholar]
- 19.Bonen A, Han XX, Habets DD, Febbraio M, Glatz JF, Luiken JJ. A null mutation in skeletal muscle FAT/CD36 reveals its essential role in insulin- and AICAR-stimulated fatty acid metabolism. Am J Physiol Endocrinol Metab 292: E1740–E1749, 2007. [DOI] [PubMed] [Google Scholar]
- 20.Borgstroem B Influence of bile salt, pH, and time on the action of pancreatic lipase; physiological implications. J Lipid Res 5: 522–531, 1964. [PubMed] [Google Scholar]
- 21.Borgstrom B, Dahlqvist A, Lundh G, Sjovall J. Studies of intestinal digestion and absorption in the human. J Clin Invest 36: 1521–1536, 1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Brigelius-Flohe R, Kelly FJ, Salonen JT, Neuzil J, Zingg JM, Azzi A. The European perspective on vitamin E: current knowledge and future research. Am J Clin Nutr 76: 703–716, 2002. [DOI] [PubMed] [Google Scholar]
- 23.Bruckdorfer KR, Kang SS, Khan IH, Bourne AR, Yudkin J. Diurnal changes in the concentrations of plasma lipids, sugars, insulin and corticosterone in rats fed diets containing various carbohydrates. Horm Metab Res 6: 99–106, 1974. [DOI] [PubMed] [Google Scholar]
- 24.Brunham LR, Kruit JK, Iqbal J, Fievet C, Timmins JM, Pape TD, Coburn BA, Bissada N, Staels B, Groen AK, Hussain MM, Parks JS, Kuipers F, Hayden MR. Intestinal ABCA1 directly contributes to HDL biogenesis in vivo. J Clin Invest 116: 1052–1062, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Brunham LR, Kruit JK, Pape TD, Parks JS, Kuipers F, Hayden MR. Tissue-specific induction of intestinal ABCA1 expression with a liver X receptor agonist raises plasma HDL cholesterol levels. Circ Res 99: 672–674, 2006. [DOI] [PubMed] [Google Scholar]
- 26.Buhman KK, Accad M, Novak S, Choi RS, Wong JS, Hamilton RL, Turley S, Farese RV Jr. Resistance to diet-induced hypercholesterolemia and gallstone formation in ACAT2-deficient mice. Nat Med 6: 1341–1347, 2000. [DOI] [PubMed] [Google Scholar]
- 27.Cai L, Eckhardt ER, Shi W, Zhao Z, Nasser M, de Villiers WJ, van der Westhuyzen DR. Scavenger receptor class B type I reduces cholesterol absorption in cultured enterocyte CaCo-2 cells. J Lipid Res 45: 253–262, 2004. [DOI] [PubMed] [Google Scholar]
- 28.Cao G, Beyer TP, Yang XP, Schmidt RJ, Zhang Y, Bensch WR, Kauffman RF, Gao H, Ryan TP, Liang Y, Eacho PI, Jiang XC. Phospholipid transfer protein is regulated by liver X receptors in vivo. J Biol Chem 277: 39561–39565, 2002. [DOI] [PubMed] [Google Scholar]
- 29.Cao J, Burn P, Shi Y. Properties of the mouse intestinal acyl-CoA:monoacylglycerol acyltransferase, MGAT2. J Biol Chem 278: 25657–25663, 2003. [DOI] [PubMed] [Google Scholar]
- 30.Cao J, Cheng L, Shi Y. Catalytic properties of MGAT3, a putative triacylgycerol synthase. J Lipid Res 48: 583–591, 2007. [DOI] [PubMed] [Google Scholar]
- 31.Cao J, Lockwood J, Burn P, Shi Y. Cloning and functional characterization of a mouse intestinal acyl-CoA:monoacylglycerol acyltransferase, MGAT2. J Biol Chem 278: 13860–13866, 2003. [DOI] [PubMed] [Google Scholar]
- 32.Carey MC, Small DM. The characteristics of mixed micellar solutions with particular reference to bile. Am J Med 49: 590–608, 1970. [DOI] [PubMed] [Google Scholar]
- 33.Carey MC, Small DM, Bliss CM. Lipid digestion and absorption. Annu Rev Physiol 45: 651–677, 1983. [DOI] [PubMed] [Google Scholar]
- 34.Carriere V, Vidal R, Lazou K, Lacasa M, Delers F, Ribeiro A, Rousset M, Chambaz J, Lacorte JM. HNF-4-dependent induction of apolipoprotein A-IV gene transcription by an apical supply of lipid micelles in intestinal cells. J Biol Chem 280: 5406–5413, 2005. [DOI] [PubMed] [Google Scholar]
- 35.Cases S, Novak S, Zheng YW, Myers HM, Lear SR, Sande E, Welch CB, Lusis AJ, Spencer TA, Krause BR, Erickson SK, Farese RV Jr. ACAT-2, a second mammalian acyl-CoA:cholesterol acyltransferase. Its cloning, expression, and characterization. J Biol Chem 273: 26755–26764, 1998. [DOI] [PubMed] [Google Scholar]
- 36.Chang CC, Huh HY, Cadigan KM, Chang TY. Molecular cloning and functional expression of human acyl-coenzyme A:cholesterol acyltransferase cDNA in mutant Chinese hamster ovary cells. J Biol Chem 268: 20747–20755, 1993. [PubMed] [Google Scholar]
- 37.Chen M, Yang Y, Braunstein E, Georgeson KE, Harmon CM. Gut expression and regulation of FAT/CD36: possible role in fatty acid transport in rat enterocytes. Am J Physiol Endocrinol Metab 281: E916–E923, 2001. [DOI] [PubMed] [Google Scholar]
- 38.Clark SB, Tercyak AM. Reduced cholesterol transmucosal transport in rats with inhibited mucosal acyl CoA:cholesterol acyltransferase and normal pancreatic function. J Lipid Res 25: 148–159, 1984. [PubMed] [Google Scholar]
- 39.Clearfield MB A novel therapeutic approach to dyslipidemia. J Am Osteopath Assoc 103: S16–S20, 2003. [PubMed] [Google Scholar]
- 40.Coleman RA, Haynes EB. Monoacylglycerol acyltransferase. Evidence that the activities from rat intestine and suckling liver are tissue-specific isoenzymes. J Biol Chem 261: 224–228, 1986. [PubMed] [Google Scholar]
- 41.D'Agostino D, Cordle RA, Kullman J, Erlanson-Albertsson C, Muglia LJ, Lowe ME. Decreased postnatal survival and altered body weight regulation in procolipase-deficient mice. J Biol Chem 277: 7170–7177, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Davidson NO, Kollmer ME, Glickman RM. Apolipoprotein B synthesis in rat small intestine: regulation by dietary triglyceride and biliary lipid. J Lipid Res 27: 30–39, 1986. [PubMed] [Google Scholar]
- 43.Davidson NO, Magun AM, Brasitus TA, Glickman RM. Intestinal apolipoprotein A-I and B-48 metabolism: effects of sustained alterations in dietary triglyceride and mucosal cholesterol flux. J Lipid Res 28: 388–402, 1987. [PubMed] [Google Scholar]
- 44.Dew SE, Ong DE. Specificity of the retinol transporter of the rat small intestine brush border. Biochemistry 33: 12340–12345, 1994. [DOI] [PubMed] [Google Scholar]
- 45.Drover VA, Ajmal M, Nassir F, Davidson NO, Nauli AM, Sahoo D, Tso P, Abumrad NA. CD36 deficiency impairs intestinal lipid secretion and clearance of chylomicrons from the blood. J Clin Invest 115: 1290–1297, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.During A, Harrison EH. Mechanisms of provitamin A (carotenoid) and vitamin A (retinol) transport into and out of intestinal Caco-2 cells. J Lipid Res 48: 2283–2294, 2007. [DOI] [PubMed] [Google Scholar]
- 47.E X, Zhang L, Lu J, Tso P, Blaner WS, Levin MS, Li E. Increased neonatal mortality in mice lacking cellular retinol-binding protein II. J Biol Chem 277: 36617–36623, 2002. [DOI] [PubMed] [Google Scholar]
- 48.Elias E, Muller DP, Scott J. Association of spinocerebellar disorders with cystic fibrosis or chronic childhood cholestasis and very low serum vitamin E. Lancet 2: 1319–1321, 1981. [DOI] [PubMed] [Google Scholar]
- 49.Ezzet F, Wexler D, Statkevich P, Kosoglou T, Patrick J, Lipka L, Mellars L, Veltri E, Batra V. The plasma concentration and LDL-C relationship in patients receiving ezetimibe. J Clin Pharmacol 41: 943–949, 2001. [DOI] [PubMed] [Google Scholar]
- 50.Field FJ, Watt K, Mathur SN. Ezetimibe interferes with cholesterol trafficking from the plasma membrane to the endoplasmic reticulum in CaCo-2 cells. J Lipid Res 48: 1735–1745, 2007. [DOI] [PubMed] [Google Scholar]
- 51.Fielding BA, Callow J, Owen RM, Samra JS, Matthews DR, Frayn KN. Postprandial lipemia: the origin of an early peak studied by specific dietary fatty acid intake during sequential meals. Am J Clin Nutr 63: 36–41, 1996. [DOI] [PubMed] [Google Scholar]
- 52.Fisher EA, Ginsberg HN. Complexity in the secretory pathway: the assembly and secretion of apolipoprotein B-containing lipoproteins. J Biol Chem 277: 17377–17380, 2002. [DOI] [PubMed] [Google Scholar]
- 53.Forester GP, Tall AR, Bisgaier CL, Glickman RM. Rat intestine secretes spherical high density lipoproteins. J Biol Chem 258: 5938–5943, 1983. [PubMed] [Google Scholar]
- 54.Fraser R, Cliff WJ, Courtice FC. The effect of dietary fat load on the size and composition of chylomicrons in thoracic duct lymph. Q J Exp Physiol Cogn Med Sci 53: 390–398, 1968. [DOI] [PubMed] [Google Scholar]
- 55.Fukuda N, Azain MJ, Ontko JA. Altered hepatic metabolism of free fatty acids underlying hypersecretion of very low density lipoproteins in the genetically obese Zucker rats. J Biol Chem 257: 14066–14072, 1982. [PubMed] [Google Scholar]
- 56.Gagne C, Bays HE, Weiss SR, Mata P, Quinto K, Melino M, Cho M, Musliner TA, Gumbiner B. Efficacy and safety of ezetimibe added to ongoing statin therapy for treatment of patients with primary hypercholesterolemia. Am J Cardiol 90: 1084–1091, 2002. [DOI] [PubMed] [Google Scholar]
- 57.Gallo-Torres HE Obligatory role of bile for the intestinal absorption of vitamin E. Lipids 5: 379–384, 1970. [DOI] [PubMed] [Google Scholar]
- 58.Gantz D, Bennett CS, Derksen A, Small DM. Size and shape determination of fixed chylomicrons and emulsions with fluid or solid surfaces by three-dimensional analysis of shadows. J Lipid Res 31: 163–171, 1990. [PubMed] [Google Scholar]
- 59.Garcia-Calvo M, Lisnock J, Bull HG, Hawes BE, Burnett DA, Braun MP, Crona JH, Davis HR Jr, Dean DC, Detmers PA, Graziano MP, Hughes M, Macintyre DE, Ogawa A, O'neill KA, Iyer SP, Shevell DE, Smith MM, Tang YS, Makarewicz AM, Ujjainwalla F, Altmann SW, Chapman KT, Thornberry NA. The target of ezetimibe is Niemann-Pick C1-Like 1 (NPC1L1). Proc Natl Acad Sci USA 102: 8132–8137, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Glaumann H, Bergstrand A, Ericsson JL. Studies on the synthesis and intracellular transport of lipoprotein particles in rat liver. J Cell Biol 64: 356–377, 1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Goodman DS Cholesterol ester metabolism. Physiol Rev 45: 747–839, 1965. [DOI] [PubMed] [Google Scholar]
- 62.Graf GA, Li WP, Gerard RD, Gelissen I, White A, Cohen JC, Hobbs HH. Coexpression of ATP-binding cassette proteins ABCG5 and ABCG8 permits their transport to the apical surface. J Clin Invest 110: 659–669, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Green PH, Glickman RM. Intestinal lipoprotein metabolism. J Lipid Res 22: 1153–1173, 1981. [PubMed] [Google Scholar]
- 64.Greenwalt DE, Scheck SH, Rhinehart-Jones T. Heart CD36 expression is increased in murine models of diabetes and in mice fed a high fat diet. J Clin Invest 96: 1382–1388, 1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Grefhorst A, Elzinga BM, Voshol PJ, Plosch T, Kok T, Bloks VW, van der Sluijs FH, Havekes LM, Romijn JA, Verkade HJ, Kuipers F. Stimulation of lipogenesis by pharmacological activation of the liver X receptor leads to production of large, triglyceride-rich very low density lipoprotein particles. J Biol Chem 277: 34182–34190, 2002. [DOI] [PubMed] [Google Scholar]
- 66.Grundy SM, Metzger AL. A physiological method for estimation of hepatic secretion of biliary lipids in man. Gastroenterology 62: 1200–1217, 1972. [PubMed] [Google Scholar]
- 67.Hacquebard M, Carpentier YA. Vitamin E: absorption, plasma transport and cell uptake. Curr Opin Clin Nutr Metab Care 8: 133–138, 2005. [DOI] [PubMed] [Google Scholar]
- 68.Hagan DL, Kienzle B, Jamil H, Hariharan N. Transcriptional regulation of human and hamster microsomal triglyceride transfer protein genes. Cell type-specific expression and response to metabolic regulators. J Biol Chem 269: 28737–28744, 1994. [PubMed] [Google Scholar]
- 69.Hanhoff T, Lucke C, Spener F. Insights into binding of fatty acids by fatty acid binding proteins. Mol Cell Biochem 239: 45–54, 2002. [PubMed] [Google Scholar]
- 70.Harries JT, Muller DP. Absorption of different doses of fat soluble and water miscible preparations of vitamin E in children with cystic fibrosis. Arch Dis Child 46: 341–344, 1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Hauser H, Dyer JH, Nandy A, Vega MA, Werder M, Bieliauskaite E, Weber FE, Compassi S, Gemperli A, Boffelli D, Wehrli E, Schulthess G, Phillips MC. Identification of a receptor mediating absorption of dietary cholesterol in the intestine. Biochemistry 37: 17843–17850, 1998. [DOI] [PubMed] [Google Scholar]
- 72.Hayashi H, Fujimoto K, Cardelli JA, Nutting DF, Bergstedt S, Tso P. Fat feeding increases size, but not number, of chylomicrons produced by small intestine. Am J Physiol Gastrointest Liver Physiol 259: G709–G719, 1990. [DOI] [PubMed] [Google Scholar]
- 73.Heider JG, Pickens CE, Kelly LA. Role of acyl CoA:cholesterol acyltransferase in cholesterol absorption and its inhibition by 57–118 in the rabbit. J Lipid Res 24: 1127–1134, 1983. [PubMed] [Google Scholar]
- 74.Hernell O, Staggers JE, Carey MC. Physical-chemical behavior of dietary and biliary lipids during intestinal digestion and absorption. 2. Phase analysis and aggregation states of luminal lipids during duodenal fat digestion in healthy adult human beings. Biochemistry 29: 2041–2056, 1990. [DOI] [PubMed] [Google Scholar]
- 75.Herrera E, Barbas C. Vitamin E: action, metabolism and perspectives. J Physiol Biochem 57: 43–56, 2001. [PubMed] [Google Scholar]
- 76.Hildebrand H, Borgström B, Békássy A, Erlanson-Albertsson C, Helin I. Isolated co-lipase deficiency in two brothers. Gut 23: 243–246, 1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Hockey KJ, Anderson RA, Cook VR, Hantgan RR, Weinberg RB. Effect of the apolipoprotein A-IV Q360H polymorphism on postprandial plasma triglyceride clearance. J Lipid Res 42: 211–217, 2001. [PubMed] [Google Scholar]
- 78.Hofmann AF, Borgstrom B. Hydrolysis of long-chain monoglycerides in micellar solution by pancreatic lipase. Biochim Biophys Acta 70: 317–331, 1963. [DOI] [PubMed] [Google Scholar]
- 79.Hollander D Retinol lymphatic and portal transport: influence of pH, bile, and fatty acids. Am J Physiol Gastrointest Liver Physiol 239: G210–G214, 1980. [DOI] [PubMed] [Google Scholar]
- 80.Hollander D Intestinal absorption of vitamins A, E, D, and K. J Lab Clin Med 97: 449–462, 1981. [PubMed] [Google Scholar]
- 81.Hollander D, Muralidhara KS. Vitamin A1 intestinal absorption in vivo: influence of luminal factors on transport. Am J Physiol Endocrinol Metab Gastrointest Physiol 232: E471–E477, 1977. [DOI] [PubMed] [Google Scholar]
- 82.Holt PR, Fairchild BM, Weiss J. A liquid crystalline phase in human intestinal contents during fat digestion. Lipids 21: 444–446, 1986. [DOI] [PubMed] [Google Scholar]
- 83.Huff MW, Pollex RL, Hegele RA. NPC1L1: evolution from pharmacological target to physiological sterol transporter. Arterioscler Thromb Vasc Biol 26: 2433–2438, 2006. [DOI] [PubMed] [Google Scholar]
- 84.Huggins KW, Boileau AC, Hui DY. Protection against diet-induced obesity and obesity-related insulin resistance in Group 1B PLA2-deficient mice. Am J Physiol Endocrinol Metab 283: E994–E1001, 2002. [DOI] [PubMed] [Google Scholar]
- 85.Hussain MM A proposed model for the assembly of chylomicrons. Atherosclerosis 148: 1–15, 2000. [DOI] [PubMed] [Google Scholar]
- 86.Hussain MM, Fatma S, Pan X, Iqbal J. Intestinal lipoprotein assembly. Curr Opin Lipidol 16: 281–285, 2005. [DOI] [PubMed] [Google Scholar]
- 87.Hussain MM, Iqbal J, Anwar K, Rava P, Dai K. Microsomal triglyceride transfer protein: a multifunctional protein. Front Biosci 8: s500–s506, 2003. [DOI] [PubMed] [Google Scholar]
- 88.Hussain MM, Kancha RK, Zhou Z, Luchoomun J, Zu H, Bakillah A. Chylomicron assembly and catabolism: role of apolipoproteins and receptors. Biochim Biophys Acta 1300: 151–170, 1996. [DOI] [PubMed] [Google Scholar]
- 89.Hussain MM, Kedees MH, Singh K, Athar H, Jamali NZ. Signposts in the assembly of chylomicrons. Front Biosci 6: D320–D331, 2001. [DOI] [PubMed] [Google Scholar]
- 90.Hussain MM, Shi J, Dreizen P. Microsomal triglyceride transfer protein and its role in apoB-lipoprotein assembly. J Lipid Res 44: 22–32, 2003. [DOI] [PubMed] [Google Scholar]
- 91.Iqbal J, Anwar K, Hussain MM. Multiple, independently regulated pathways of cholesterol transport across the intestinal epithelial cells. J Biol Chem 278: 31610–31620, 2003. [DOI] [PubMed] [Google Scholar]
- 92.Iqbal J, Dai K, Seimon T, Jungreis R, Oyadomari M, Kuriakose G, Ron D, Tabas I, Hussain MM. IRE1beta inhibits chylomicron production by selectively degrading MTP mRNA. Cell Metab 7: 445–455, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Iqbal J, Hussain MM. Evidence for multiple complementary pathways for efficient cholesterol absorption in mice. J Lipid Res 46: 1491–1501, 2005. [DOI] [PubMed] [Google Scholar]
- 94.Iqbal J, Rudel LL, Hussain MM. Microsomal triglyceride transfer protein enhances cellular cholesteryl esterification by relieving product inhibition. J Biol Chem 283: 19967–19980, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Iyer SP, Yao X, Crona JH, Hoos LM, Tetzloff G, Davis HR Jr, Graziano MP, Altmann SW. Characterization of the putative native and recombinant rat sterol transporter Niemann-Pick C1 Like 1 (NPC1L1) protein. Biochim Biophys Acta 1722: 282–292, 2005. [DOI] [PubMed] [Google Scholar]
- 96.Janowski BA, Grogan MJ, Jones SA, Wisely GB, Kliewer SA, Corey EJ, Mangelsdorf DJ. Structural requirements of ligands for the oxysterol liver X receptors LXRalpha and LXRbeta. Proc Natl Acad Sci USA 96: 266–271, 1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Janowski BA, Willy PJ, Devi TR, Falck JR, Mangelsdorf DJ. An oxysterol signalling pathway mediated by the nuclear receptor LXR alpha. Nature 383: 728–731, 1996. [DOI] [PubMed] [Google Scholar]
- 98.Jiang XC, Beyer TP, Li Z, Liu J, Quan W, Schmidt RJ, Zhang Y, Bensch WR, Eacho PI, Cao G. Enlargement of high density lipoprotein in mice via liver X receptor activation requires apolipoprotein E and is abolished by cholesteryl ester transfer protein expression. J Biol Chem 278: 49072–49078, 2003. [DOI] [PubMed] [Google Scholar]
- 99.Jiang ZG, Liu Y, Hussain MM, Atkinson D, McKnight CJ. Reconstituting initial events during the assembly of apolipoprotein B-containing lipoproteins in a cell-free system. J Mol Biol 383: 1181–1194, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Kaempf-Rotzoll DE, Traber MG, Arai H. Vitamin E and transfer proteins. Curr Opin Lipidol 14: 249–254, 2003. [DOI] [PubMed] [Google Scholar]
- 101.Kayden HJ, Traber MG. Absorption, lipoprotein transport, and regulation of plasma concentrations of vitamin E in humans. J Lipid Res 34: 343–358, 1993. [PubMed] [Google Scholar]
- 102.Klett EL, Lu K, Kosters A, Vink E, Lee MH, Altenburg M, Shefer S, Batta AK, Yu H, Chen J, Klein R, Looije N, Oude-Elferink R, Groen AK, Maeda N, Salen G, Patel SB. A mouse model of sitosterolemia: absence of Abcg8/sterolin-2 results in failure to secrete biliary cholesterol. BMC Med 2: 5, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Knopp RH, Gitter H, Truitt T, Bays H, Manion CV, Lipka LJ, LeBeaut AP, Suresh R, Yang B, Veltri EP. Effects of ezetimibe, a new cholesterol absorption inhibitor, on plasma lipids in patients with primary hypercholesterolemia. Eur Heart J 24: 729–741, 2003. [DOI] [PubMed] [Google Scholar]
- 104.Kondrup J, Damgaard SE, Fleron P. Metabolism of palmitate in perfused rat liver. Computer models of subcellular triacylglycerol metabolism. Biochem J 184: 73–81, 1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Kumar NS, Mansbach CM. Prechylomicron transport vesicle: isolation and partial characterization. Am J Physiol Gastrointest Liver Physiol 276: G378–G386, 1999. [DOI] [PubMed] [Google Scholar]
- 106.Lee MH, Lu K, Hazard S, Yu H, Shulenin S, Hidaka H, Kojima H, Allikmets R, Sakuma N, Pegoraro R, Srivastava AK, Salen G, Dean M, Patel SB. Identification of a gene, ABCG5, important in the regulation of dietary cholesterol absorption. Nat Genet 27: 79–83, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lee RG, Willingham MC, Davis MA, Skinner KA, Rudel LL. Differential expression of ACAT1 and ACAT2 among cells within liver, intestine, kidney, and adrenal of nonhuman primates. J Lipid Res 41: 1991–2001, 2000. [PubMed] [Google Scholar]
- 108.Lemieux S, Fontani R, Uffelman KD, Lewis GF, Steiner G. Apolipoprotein B-48 and retinyl palmitate are not equivalent markers of postprandial intestinal lipoproteins. J Lipid Res 39: 1964–1971, 1998. [PubMed] [Google Scholar]
- 109.Li E, Norris AW. Structure/function of cytoplasmic vitamin A-binding proteins. Annu Rev Nutr 16: 205–234, 1996. [DOI] [PubMed] [Google Scholar]
- 110.Liao W, Chan L. Apolipoprotein B, a paradigm for proteins regulated by intracellular degradation, does not undergo intracellular degradation in CaCo2 cells. J Biol Chem 275: 3950–3956, 2000. [DOI] [PubMed] [Google Scholar]
- 111.Lombardo D, Guy O. Studies on the substrate specificity of a carboxyl ester hydrolase from human pancreatic juice. II. Action on cholesterol esters and lipid-soluble vitamin esters. Biochim Biophys Acta 611: 147–155, 1980. [DOI] [PubMed] [Google Scholar]
- 112.Lowe ME The triglyceride lipases of the pancreas. J Lipid Res 43: 2007–2016, 2002. [DOI] [PubMed] [Google Scholar]
- 113.Lu K, Lee MH, Hazard S, Brooks-Wilson A, Hidaka H, Kojima H, Ose L, Stalenhoef AF, Mietinnen T, Bjorkhem I, Bruckert E, Pandya A, Brewer HB Jr, Salen G, Dean M, Srivastava A, Patel SB. Two genes that map to the STSL locus cause sitosterolemia: genomic structure and spectrum of mutations involving sterolin-1 and sterolin-2, encoded by ABCG5 and ABCG8, respectively. Am J Hum Genet 69: 278–290, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Lu S, Yao Y, Meng S, Cheng X, Black DD. Overexpression of apolipoprotein A-IV enhances lipid transport in newborn swine intestinal epithelial cells. J Biol Chem 277: 31929–31937, 2002. [DOI] [PubMed] [Google Scholar]
- 115.Luchoomun J, Hussain MM. Assembly and secretion of chylomicrons by differentiated Caco-2 cells. Nascent triglycerides and preformed phospholipids are preferentially used for lipoprotein assembly. J Biol Chem 274: 19565–19572, 1999. [DOI] [PubMed] [Google Scholar]
- 116.MacDonald PN, Ong DE. Evidence for a lecithin-retinol acyltransferase activity in the rat small intestine. J Biol Chem 263: 12478–12482, 1988. [PubMed] [Google Scholar]
- 117.Magun AM, Brasitus TA, Glickman RM. Isolation of high density lipoproteins from rat intestinal epithelial cells. J Clin Invest 75: 209–218, 1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Maillot F, Garrigue MA, Pinault M, Objois M, Theret V, Lamisse F, Hoinard C, Antoine JM, Lairon D, Couet C. Changes in plasma triacylglycerol concentrations after sequential lunch and dinner in healthy subjects. Diabetes Metab 31: 69–77, 2005. [DOI] [PubMed] [Google Scholar]
- 119.Mansbach CM 2nd, Gorelick F. Development and Physiological Regulation of Intestinal Lipid Absorption. II. Dietary lipid absorption, complex lipid synthesis, and the intracellular packaging and secretion of chylomicrons. Am J Physiol Gastrointest Liver Physiol 293: G645–G650, 2007. [DOI] [PubMed] [Google Scholar]
- 120.Mardones P, Quiñones V, Amigo L, Moreno M, Miquel JF, Schwarz M, Miettinen HE, Trigatti B, Krieger M, VanPatten S, Cohen DE, Rigotti A. Hepatic cholesterol and bile acid metabolism and intestinal cholesterol absorption in scavenger receptor class B type I-deficient mice. J Lipid Res 42: 170–180, 2001. [PubMed] [Google Scholar]
- 121.Mathias PM, Harries JT, Peters TJ, Muller DP. Studies on the in vivo absorption of micellar solutions of tocopherol and tocopheryl acetate in the rat: demonstration and partial characterization of a mucosal esterase localized to the endoplasmic reticulum of the enterocyte. J Lipid Res 22: 829–837, 1981. [PubMed] [Google Scholar]
- 122.Mattson FH, Beck LW. The specificity of pancreatic lipase for the primary hydroxyl groups of glycerides. J Biol Chem 219: 735–740, 1956. [PubMed] [Google Scholar]
- 123.Mattson FH, Volpenhein RA. The digestion and absorption of triglycerides. J Biol Chem 239: 2772–2777, 1964. [PubMed] [Google Scholar]
- 124.Mattson FH, Volpenhein RA. Hydrolysis of primary and secondary esters of glycerol by pancreatic juice. J Lipid Res 9: 79–84, 1968. [PubMed] [Google Scholar]
- 125.Meiner V, Tam C, Gunn MD, Dong LM, Weisgraber KH, Novak S, Myers HM, Erickson SK, Farese RV Jr. Tissue expression studies on the mouse acyl-CoA: cholesterol acyltransferase gene (Acact): findings supporting the existence of multiple cholesterol esterification enzymes in mice. J Lipid Res 38: 1928–1933, 1997. [PubMed] [Google Scholar]
- 126.Miao B, Zondlo S, Gibbs S, Cromley D, Hosagrahara VP, Kirchgessner TG, Billheimer J, Mukherjee R. Raising HDL cholesterol without inducing hepatic steatosis and hypertriglyceridemia by a selective LXR modulator. J Lipid Res 45: 1410–1417, 2004. [DOI] [PubMed] [Google Scholar]
- 127.Miller KW, Small DM. Surface-to-core and interparticle equilibrium distributions of triglyceride-rich lipoprotein lipids. J Biol Chem 258: 13772–13784, 1983. [PubMed] [Google Scholar]
- 128.Momsen WE, Brockman HL. Inhibition of pancreatic lipase B activity by taurodeoxycholate and its reversal by colipase. J Biol Chem 251: 384–388, 1976. [PubMed] [Google Scholar]
- 129.Mondola P, Gambardella P, Santangelo F, Santillo M, Greco AM. Circadian rhythms of lipid and apolipoprotein pattern in adult fasted rats. Physiol Behav 58: 175–180, 1995. [DOI] [PubMed] [Google Scholar]
- 130.Moreau RA, Whitaker BD, Hicks KB. Phytosterols, phytostanols, and their conjugates in foods: structural diversity, quantitative analysis, and health-promoting uses. Prog Lipid Res 41: 457–500, 2002. [DOI] [PubMed] [Google Scholar]
- 131.Mu H, Hoy CE. The digestion of dietary triacylglycerols. Prog Lipid Res 43: 105–133, 2004. [DOI] [PubMed] [Google Scholar]
- 132.Mulligan JD, Flowers MT, Tebon A, Bitgood JJ, Wellington C, Hayden MR, Attie AD. ABCA1 is essential for efficient basolateral cholesterol efflux during the absorption of dietary cholesterol in chickens. J Biol Chem 278: 13356–13366, 2003. [DOI] [PubMed] [Google Scholar]
- 133.Nauli AM, Nassir F, Zheng S, Yang Q, Lo CM, Vonlehmden SB, Lee D, Jandacek RJ, Abumrad NA, Tso P. CD36 is important for chylomicron formation and secretion and may mediate cholesterol uptake in the proximal intestine. Gastroenterology 131: 1197–1207, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Nayak N, Harrison EH, Hussain MM. Retinyl ester secretion by intestinal cells: a specific and regulated process dependent on assembly and secretion of chylomicrons. J Lipid Res 42: 272–280, 2001. [PubMed] [Google Scholar]
- 135.Neeli I, Siddiqi SA, Siddiqi S, Mahan J, Lagakos WS, Binas B, Gheyi T, Storch J, Mansbach CM. Liver fatty acid-binding protein initiates budding of pre-chylomicron transport vesicles from intestinal endoplasmic reticulum. J Biol Chem 282: 17974–17984, 2007. [DOI] [PubMed] [Google Scholar]
- 136.Newcomer ME, Jamison RS, Ong DE. Structure and function of retinoid-binding proteins. Subcell Biochem 30: 53–80, 1998. [DOI] [PubMed] [Google Scholar]
- 137.Oelkers P, Behari A, Cromley D, Billheimer JT, Sturley SL. Characterization of two human genes encoding acyl coenzyme A:cholesterol acyltransferase-related enzymes. J Biol Chem 273: 26765–26771, 1998. [DOI] [PubMed] [Google Scholar]
- 138.Ohama T, Hirano K, Zhang Z, Aoki R, Tsujii K, Nakagawa-Toyama Y, Tsukamoto K, Ikegami C, Matsuyama A, Ishigami M, Sakai N, Hiraoka H, Ueda K, Yamashita S, Matsuzawa Y. Dominant expression of ATP-binding cassette transporter-1 on basolateral surface of Caco-2 cells stimulated by LXR/RXR ligands. Biochem Biophys Res Commun 296: 625–630, 2002. [DOI] [PubMed] [Google Scholar]
- 139.Oliveira HC, Nilausen K, Meinertz H, Quintao EC. Cholesteryl esters in lymph chylomicrons: contribution from high density lipoprotein transferred from plasma into intestinal lymph. J Lipid Res 34: 1729–1736, 1993. [PubMed] [Google Scholar]
- 140.Ong DE Vitamin A-binding proteins. Nutr Rev 43: 225–232, 1985. [DOI] [PubMed] [Google Scholar]
- 141.Ong DE Cellular transport and metabolism of vitamin A: roles of the cellular retinoid-binding proteins. Nutr Rev 52: S24–S31, 1994. [DOI] [PubMed] [Google Scholar]
- 142.Owen MR, Corstorphine CC, Zammit VA. Overt and latent activities of diacylglycerol acytransferase in rat liver microsomes: possible roles in very-low-density lipoprotein triacylglycerol secretion. Biochem J 323: 17–21, 1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Pan X, Hussain MM. Diurnal regulation of microsomal triglyceride transfer protein and plasma lipid levels. J Biol Chem 282: 24707–24719, 2007. [DOI] [PubMed] [Google Scholar]
- 144.Phan CT, Tso P. Intestinal lipid absorption and transport. Front Biosci 6: D299–D319, 2001. [DOI] [PubMed] [Google Scholar]
- 145.Plack PA Occurrence, absorption and distribution of vitamin A. Proc Nutr Soc 24: 146–153, 1965. [DOI] [PubMed] [Google Scholar]
- 146.Plösch T, Bloks VW, Terasawa Y, Berdy S, Siegler K, Van Der Sluijs F, Kema IP, Groen AK, Shan B, Kuipers F, Schwarz M. Sitosterolemia in ABC-transporter G5-deficient mice is aggravated on activation of the liver-X receptor. Gastroenterology 126: 290–300, 2004. [DOI] [PubMed] [Google Scholar]
- 147.Plösch T, Kok T, Bloks VW, Smit MJ, Havinga R, Chimini G, Groen AK, Kuipers F. Increased hepatobiliary and fecal cholesterol excretion upon activation of the liver X receptor is independent of ABCA1. J Biol Chem 277: 33870–33877, 2002. [DOI] [PubMed] [Google Scholar]
- 148.Poirier H, Degrace P, Niot I, Bernard A, Besnard P. Localization and regulation of the putative membrane fatty-acid transporter (FAT) in the small intestine. Comparison with fatty acid-binding proteins (FABP). Eur J Biochem 238: 368–373, 1996. [DOI] [PubMed] [Google Scholar]
- 149.Ponz de Leon M, Loria P, Iori R, Carulli N. Cholesterol absorption in cirrhosis: the role of total and individual bile acid pool size. Gastroenterology 80: 1428–1437, 1981. [PubMed] [Google Scholar]
- 150.Porter HP, Saunders DR, Tytgat G, Brunser O, Rubin CE. Fat absorption in bile fistula man. A morphological and biochemical study. Gastroenterology 60: 1008–1019, 1971. [PubMed] [Google Scholar]
- 151.Quick TC, Ong DE. Vitamin A metabolism in the human intestinal Caco-2 cell line. Biochemistry 29: 11116–11123, 1990. [DOI] [PubMed] [Google Scholar]
- 152.Quintao EC, Drewiacki A, Stechhaln K, de Faria EC, Sipahi AM. Origin of cholesterol transported in intestinal lymph: studies in patients with filarial chyluria. J Lipid Res 20: 941–945, 1979. [PubMed] [Google Scholar]
- 153.Rajan N, Kidd GL, Talmage DA, Blaner WS, Suhara A, Goodman DS. Cellular retinoic acid-binding protein messenger RNA: levels in rat tissues and localization in rat testis. J Lipid Res 32: 1195–1204, 1991. [PubMed] [Google Scholar]
- 154.Reboul E, Klein A, Bietrix F, Gleize B, Malezet-Desmoulins C, Schneider M, Margotat A, Lagrost L, Collet X, Borel P. Scavenger receptor class B type I (SR-BI) is involved in vitamin E transport across the enterocyte. J Biol Chem 281: 4739–4745, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Repa JJ, Berge KE, Pomajzl C, Richardson JA, Hobbs H, Mangelsdorf DJ. Regulation of ATP-binding cassette sterol transporters ABCG5 and ABCG8 by the liver X receptors alpha and beta. J Biol Chem 277: 18793–18800, 2002. [DOI] [PubMed] [Google Scholar]
- 156.Repa JJ, Liang G, Ou J, Bashmakov Y, Lobaccaro JM, Shimomura I, Shan B, Brown MS, Goldstein JL, Mangelsdorf DJ. Regulation of mouse sterol regulatory element-binding protein-1c gene (SREBP-1c) by oxysterol receptors, LXRalpha and LXRbeta. Genes Dev 14: 2819–2830, 2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Repa JJ, Turley SD, Lobaccaro JA, Medina J, Li L, Lustig K, Shan B, Heyman RA, Dietschy JM, Mangelsdorf DJ. Regulation of absorption and ABC1-mediated efflux of cholesterol by RXR heterodimers. Science 289: 1524–1529, 2000. [DOI] [PubMed] [Google Scholar]
- 158.Richmond BL, Boileau AC, Zheng S, Huggins KW, Granholm NA, Tso P, Hui DY. Compensatory phospholipid digestion is required for cholesterol absorption in pancreatic phospholipase A(2)-deficient mice. Gastroenterology 120: 1193–1202, 2001. [DOI] [PubMed] [Google Scholar]
- 159.Rustow B, Kunze D. Diacylglycerol synthesized in vitro from sn-glycerol 3-phosphate and the endogenous diacylglycerol are different substrate pools for the biosynthesis of phosphatidylcholine in rat lung microsomes. Biochim Biophys Acta 835: 273–278, 1985. [DOI] [PubMed] [Google Scholar]
- 160.Rustow B, Kunze D. Further evidence for the existence of different diacylglycerol pools of the phosphatidylcholine synthesis in microsomes. Biochim Biophys Acta 921: 552–558, 1987. [DOI] [PubMed] [Google Scholar]
- 161.Sarda L, Desnuelle P. Actions of pancreatic lipase on esters in emulsions. Biochim Biophys Acta 30: 513–521, 1958. [DOI] [PubMed] [Google Scholar]
- 162.Schaffer JE, Lodish HF. Expression cloning and characterization of a novel adipocyte long chain fatty acid transport protein. Cell 79: 427–436, 1994. [DOI] [PubMed] [Google Scholar]
- 163.Schultz JR, Tu H, Luk A, Repa JJ, Medina JC, Li L, Schwendner S, Wang S, Thoolen M, Mangelsdorf DJ, Lustig KD, Shan B. Role of LXRs in control of lipogenesis. Genes Dev 14: 2831–2838, 2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Siddiqi SA VLDL exits from the endoplasmic reticulum in a specialized vesicle, the VLDL transport vesicle, in rat primary hepatocytes. Biochem J 413: 333–342, 2008. [DOI] [PubMed] [Google Scholar]
- 165.Siddiqi SA, Gorelick FS, Mahan JT, Mansbach CM. COPII proteins are required for Golgi fusion but not for endoplasmic reticulum budding of the pre-chylomicron transport vesicle. J Cell Sci 116: 415–427, 2003. [DOI] [PubMed] [Google Scholar]
- 166.Siddiqi SA, Mahan J, Siddiqi S, Gorelick FS, Mansbach CM. Vesicle-associated membrane protein 7 is expressed in intestinal ER. J Cell Sci 119: 943–950, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Siddiqi SA, Mansbach CM. PKC zeta-mediated phosphorylation controls budding of the pre-chylomicron transport vesicle. J Cell Sci 121: 2327–2338, 2008. [DOI] [PubMed] [Google Scholar]
- 168.Singh K, Batuman OA, Akman HO, Kedees MH, Vakil V, Hussain MM. Differential, tissue-specific, transcriptional regulation of apolipoprotein B secretion by transforming growth factor beta. J Biol Chem 277: 39515–39524, 2002. [DOI] [PubMed] [Google Scholar]
- 169.Sipahi AM, Oliveira HC, Vasconcelos KS, Castilho LN, Bettarello A, Quintao EC. Contribution of plasma protein and lipoproteins to intestinal lymph: comparison of long-chain with medium-chain triglyceride duodenal infusion. Lymphology 22: 13–19, 1989. [PubMed] [Google Scholar]
- 170.Smith SJ, Cases S, Jensen DR, Chen HC, Sande E, Tow B, Sanan DA, Raber J, Eckel RH, Farese RV Jr. Obesity resistance and multiple mechanisms of triglyceride synthesis in mice lacking Dgat. Nat Genet 25: 87–90, 2000. [DOI] [PubMed] [Google Scholar]
- 171.Sokol RJ, Heubi JE, Iannaccone S, Bove KE, Balistreri WF. Mechanism causing vitamin E deficiency during chronic childhood cholestasis. Gastroenterology 85: 1172–1182, 1983. [PubMed] [Google Scholar]
- 172.Sokol RJ, Reardon MC, Accurso FJ, Stall C, Narkewicz M, Abman SH, Hammond KB. Fat-soluble-vitamin status during the first year of life in infants with cystic fibrosis identified by screening of newborns. Am J Clin Nutr 50: 1064–1071, 1989. [DOI] [PubMed] [Google Scholar]
- 173.Sorci-Thomas M, Wilson MD, Johnson FL, Williams DL, Rudel LL. Studies on the expression of genes encoding apolipoproteins B100 and B48 and the low density lipoprotein receptor in nonhuman primates. Comparison of dietary fat and cholesterol. J Biol Chem 264: 9039–9045, 1989. [PubMed] [Google Scholar]
- 174.Steffensen KR, Gustafsson JA. Putative metabolic effects of the liver X receptor (LXR). Diabetes 53, Suppl 1: S36–S42, 2004. [DOI] [PubMed] [Google Scholar]
- 175.Storch J, Zhou YX, Lagakos WS. Metabolism of apical versus basolateral sn-2-monoacylglycerol and fatty acids in rodent small intestine. J Lipid Res 49: 1762–1769, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Suruga K, Suzuki R, Goda T, Takase S. Unsaturated fatty acids regulate gene expression of cellular retinol-binding protein, type II in rat jejunum. J Nutr 125: 2039–2044, 1995. [DOI] [PubMed] [Google Scholar]
- 177.Swell L, Trout EC Jr, Hopper JR, Field H Jr, Treadwell CR. Specific function of bile salts in cholesterol absorption. Proc Soc Exp Biol Med 98: 174–176, 1958. [DOI] [PubMed] [Google Scholar]
- 178.Tang W, Ma Y, Jia L, Ioannou YA, Davies JP, Yu L. Niemann-Pick C1-like 1 is required for an LXR agonist to raise plasma HDL cholesterol in mice. Arterioscler Thromb Vasc Biol 28: 448–454, 2008. [DOI] [PubMed] [Google Scholar]
- 179.Traber MG, Arai H. Molecular mechanisms of vitamin E transport. Annu Rev Nutr 19: 343–355, 1999. [DOI] [PubMed] [Google Scholar]
- 180.Traber MG, Burton GW, Hamilton RL. Vitamin E trafficking. Ann NY Acad Sci 1031: 1–12, 2004. [DOI] [PubMed] [Google Scholar]
- 181.Traber MG, Sies H. Vitamin E in humans: demand and delivery. Annu Rev Nutr 16: 321–347, 1996. [DOI] [PubMed] [Google Scholar]
- 182.van den Bosch H, Postema NM, de Haas GH, van Deenen LL. On the positional specificity of phospholipase A from pancreas. Biochim Biophys Acta 98: 657–659, 1965. [DOI] [PubMed] [Google Scholar]
- 183.van Greevenbroek MM, de Bruin TW. Chylomicron synthesis by intestinal cells in vitro and in vivo. Atherosclerosis 141, Suppl 1: S9–S16, 1998. [DOI] [PubMed] [Google Scholar]
- 184.van Bennekum A, Werder M, Thuahnai ST, Han CH, Duong P, Williams DL, Wettstein P, Schulthess G, Phillips MC, Hauser H. Class B scavenger receptor-mediated intestinal absorption of dietary beta-carotene and cholesterol. Biochemistry 44: 4517–4525, 2005. [DOI] [PubMed] [Google Scholar]
- 185.van der Veen JN, Kruit JK, Havinga R, Baller JF, Chimini G, Lestavel S, Staels B, Groot PH, Groen AK, Kuipers F. Reduced cholesterol absorption upon PPARdelta activation coincides with decreased intestinal expression of NPC1L1. J Lipid Res 46: 526–534, 2005. [DOI] [PubMed] [Google Scholar]
- 186.van Heek M, Farley C, Compton DS, Hoos L, Alton KB, Sybertz EJ, Davis HR Jr. Comparison of the activity and disposition of the novel cholesterol absorption inhibitor, SCH58235, and its glucuronide, SCH60663. Br J Pharmacol 129: 1748–1754, 2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.van Heek M, Farley C, Compton DS, Hoos LM, Smith-Torhan A, Davis HR. Ezetimibe potently inhibits cholesterol absorption but does not affect acute hepatic or intestinal cholesterol synthesis in rats. Br J Pharmacol 138: 1459–1464, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Vasconcelos KS, Sipahi AM, Oliveira HC, Castilho LN, De Luccia N, Quintão EC. Origin of intestinal lymph cholesterol in rats: contribution from luminal absorption, mucosal synthesis and filtration from plasma. Lymphology 22: 4–12, 1989. [PubMed] [Google Scholar]
- 189.Venkateswaran A, Repa JJ, Lobaccaro JM, Bronson A, Mangelsdorf DJ, Edwards PA. Human white/murine ABC8 mRNA levels are highly induced in lipid-loaded macrophages. A transcriptional role for specific oxysterols. J Biol Chem 275: 14700–14707, 2000. [DOI] [PubMed] [Google Scholar]
- 190.Verger R Pancreatic lipase. In: Lipases, edited by Borgstrom B and Brockman HL. Amsterdam: Elsevier, 1984, p. 84–150.
- 191.Voshol PJ, Schwarz M, Rigotti A, Krieger M, Groen AK, Kuipers F. Down-regulation of intestinal scavenger receptor class B, type I (SR-BI) expression in rodents under conditions of deficient bile delivery to the intestine. Biochem J 356: 317–325, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Wang DQ, Carey MC. Susceptibility to murine cholesterol gallstone formation is not affected by partial disruption of the HDL receptor SR-BI. Biochim Biophys Acta 1583: 141–150, 2002. [DOI] [PubMed] [Google Scholar]
- 193.Willison HJ, Muller DP, Matthews S, Jones S, Kriss A, Stead RJ, Hodson ME, Harding AE. A study of the relationship between neurological function and serum vitamin E concentrations in patients with cystic fibrosis. J Neurol Neurosurg Psychiatry 48: 1097–1102, 1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Wilson MD, Rudel LL. Review of cholesterol absorption with emphasis on dietary and biliary cholesterol. J Lipid Res 35: 943–955, 1994. [PubMed] [Google Scholar]
- 195.Yang LY, Kuksis A, Myher JJ. Biosynthesis of chylomicron triacylglycerols by rats fed glyceryl or alkyl esters of menhaden oil fatty acids. J Lipid Res 36: 1046–1057, 1995. [PubMed] [Google Scholar]
- 196.Yao L, Heubi JE, Buckley DD, Fierra H, Setchell KD, Granholm NA, Tso P, Hui DY, Woollett LA. Separation of micelles and vesicles within lumenal aspirates from healthy humans: solubilization of cholesterol after a meal. J Lipid Res 43: 654–660, 2002. [PubMed] [Google Scholar]
- 197.Yen CL, Stone SJ, Koliwad S, Harris C, Farese RV Jr. Thematic review series: glycerolipids. DGAT enzymes and triacylglycerol biosynthesis. J Lipid Res 49: 2283–2301, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Yu L, Bharadwaj S, Brown JM, Ma Y, Du W, Davis MA, Michaely P, Liu P, Willingham MC, Rudel LL. Cholesterol-regulated translocation of NPC1L1 to the cell surface facilitates free cholesterol uptake. J Biol Chem 281: 6616–6624, 2006. [DOI] [PubMed] [Google Scholar]
- 199.Yu L, Hammer RE, Li-Hawkins J, Von BK, Lutjohann D, Cohen JC, Hobbs HH. Disruption of Abcg5 and Abcg8 in mice reveals their crucial role in biliary cholesterol secretion. Proc Natl Acad Sci USA 99: 16237–16242, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Yu L, Li-Hawkins J, Hammer RE, Berge KE, Horton JD, Cohen JC, Hobbs HH. Overexpression of ABCG5 and ABCG8 promotes biliary cholesterol secretion and reduces fractional absorption of dietary cholesterol. J Clin Invest 110: 671–680, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Yu L, York J, von Bergmann K, Lutjohann D, Cohen JC, Hobbs HH. Stimulation of cholesterol excretion by the liver X receptor agonist requires ATP-binding cassette transporters G5 and G8. J Biol Chem 278: 15565–15570, 2003. [DOI] [PubMed] [Google Scholar]
- 202.Zammit VA Role of insulin in hepatic fatty acid partitioning: emerging concepts. Biochem J 314: 1–14, 1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Ziajka PE, Reis M, Kreul S, King H. Initial low-density lipoprotein response to statin therapy predicts subsequent low-density lipoprotein response to the addition of ezetimibe. Am J Cardiol 93: 779–780, 2004. [DOI] [PubMed] [Google Scholar]
- 204.Zilversmit DB The composition and structure of lymph chylomicrons in dog, rat, and man. J Clin Invest 44: 1610–1622, 1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Zilversmit DB Formation and transport of chylomicrons. Fed Proc 26: 1599–1605, 1967. [PubMed] [Google Scholar]


