Abstract
BACKGROUND:
Endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) is the preferred modality for the cytological diagnosis of various cancers. Onsite cytopathology interpretation is not available in most centres.
OBJECTIVE:
To assess whether the the adequacy of tissue sampling assessed by an onsite cytotechnologist improves the diagnostic accuracy of EUS-FNA.
METHODS:
The present study is a retrospective review of all patients undergoing solid mass EUS-FNA between September 2005 and August 2007. Patients in group I (September 2005 to August 2006) had cytology slides prepared by an endoscopy nurse. Patients in group II (September 2006 to August 2007) had cytology slides prepared, stained and assessed for adequacy of tissue sampling by a cytotechnologist in the endoscopy suite. The final cytopathological diagnosis (definitely positive, definitely negative or inconclusive) was compared between the two groups.
RESULTS:
A total of 49 EUS-FNA procedures were performed in 47 patients in group I and 60 EUS-FNA procedures in 55 patients in group II. Pancreatic masses were the most common target site in both groups. The total number of needle passes was 105 in group I (mean 2.14 passes per patient; range one to five needle passes) and 158 in group II (mean 2.63 passes per patient; range one to four needle passes). The difference in the number of needle passes was not statistically significant between groups. The final diagnosis was definite in 53% in group I compared with 77% in group II (P=0.01). The percentage of inconclusive diagnoses was 47% in group I and 23% in group II (P=0.001).
CONCLUSION:
Onsite cytotechnologist interpretation of adequacy of tissue sampling significantly improves the diagnostic yield of EUS-FNA. This appears to be independent of the total number of needle passes undertaken for tissue sampling.
Keywords: Biopsy, Endosonography, Outcome
Abstract
HISTORIQUE :
L’aspiration à l’aiguille guidée par endoscopie (BAA-GE) est la modalité favorisée pour le diagnostic cytologique de divers cancers. Dans la plupart des centres, on ne peut interpréter les cytopathologies sur place.
OBJECTIF :
Les auteurs ont évalué si la validité de l’échantillonnage de tissus par un cytotechnologiste sur place améliore le taux de diagnostic de BAA-GE.
MÉTHODOLOGIE :
La présente étude rétrospective porte sur tous les patients ayant subi la BAA-GE d’une masse solide entre septembre 2005 et août 2007. Pour les patients du groupe I (septembre 2005 à août 2006), les coupes de cytologie étaient préparées par une infirmière en endoscopie. Pour les patients du groupe II (septembre 2006 à août 2007), les coupes de cytologie étaient préparées, colorées et évaluées en vue d’établir la validité de l’échantillonnage de tissus par un cytotechnologiste de l’unité d’endoscopie. On a comparé le diagnostic cytopathologique définitif (catégoriquement positif, catégoriquement négatif ou non concluant) entre les deux groupes.
RÉSULTATS :
Au total, 49 BAA-GE ont été effectuées chez 47 patients du groupe I et 60, chez 55 patients du groupe II. Dans les deux groupes, les masses pancréatiques représentaient le foyer le plus ciblé. On a dénombré un total de 105 passages d’aiguille dans le groupe I (moyenne de 2,14 par patient; plage de un à cinq) et de 158 dans le groupe II (moyenne de 2,63 par patient; plage de un à quatre). La différence du nombre de passages d’aiguille n’était pas statistiquement significative entre les deux groupes. Le diagnostic final était catégorique dans 53 % des cas du groupe I par rapport à 77 % des cas du groupe II (P=0,01). Le pourcentage de diagnostics non concluants s’élevait à 47 % dans le groupe I et de 23 % dans le groupe II (P=0,001).
CONCLUSION :
L’interprétation de la validité de l’échantillonnage de tissus par un cytotechnologiste sur place améliore considérablement le taux de diagnostic des BAA-GE. Ce résultat semble indépendant du nombre total de passages d’aiguille effectués pour l’échantillonnage des tissus.
Endoscopic ultrasound (EUS) is a well-established modality in diagnosing and staging various gastrointestinal, pancreaticobiliary and pulmonary cancers (1–4). However, with an accuracy of 82%, EUS alone is limited in its ability to discriminate between malignant and benign lesions (5–7). In the early 1990s, EUS-guided fine-needle aspiration (EUS-FNA) emerged as a minimally invasive technique to obtain cytological specimens from lesions within, or immediately adjacent to, the upper or lower gastrointestinal tract. In experienced hands, EUS-FNA has a proven accuracy with a diagnostic yield for the detection of various malignancies of approximately 90% (8–13).
A successful cytological diagnosis following EUS-FNA requires an experienced endosonographer, a specimen of adequate cellularity, a high-quality cytology slide preparation and an astute cytopathologist. Up to 32% of FNA interpretation may be nondiagnostic due to scant cellularity and/or poor slide preparation causing ‘crush artefact’ when performed by inexperienced personnel (14). Onsite interpretation of FNA specimens is beneficial for rapid clinical diagnosis and decision-making (14–17). Because of a shortage of trained cytopathologists, most centres do not have provisions for onsite diagnosis of EUS-FNA specimens (5,14,18,19). Under these circumstances, it is recommended to perform between five and seven needle passes for pancreatic mass aspiration and between three and five needle passes for liver mass and lymph node aspiration to enhance diagnostic accuracy (20,21). The cytology slides are usually prepared by an endoscopy nurse and later sent to the pathology department for subsequent review by a cytopathologist.
A skilled cytotechnologist, if available in the endoscopy suite during EUS-FNA, can enhance the quality of direct smears to limit the amount of crush and air drying artefact and perform onsite staining to assess for adequacy of tissue sampling, thereby increasing the accuracy for obtaining a definitive diagnosis and reducing the need for additional needle passes.
The aim of the present study was to determine whether onsite cytotechnologist interpretation improves the diagnostic yield of solid mass EUS-FNA.
PATIENTS AND METHODS
Between September 2005 and August 2007, all patients undergoing EUS-FNA for solid mass lesions at the University of Alberta Hospital, Edmonton, Alberta, were reviewed and divided into two groups.
EUS-FNA examination
During the study period, all procedures were performed by a single experienced endosonographer using the Pentax EG3630U or EG3630UT curvilinear array echoendoscope (Pentax Precision Instruments, USA). EUS-FNA was performed using a 22-gauge Wilson-Cook Echotip or Echotip-ultra needle (Wilson-Cook, USA). All patients provided informed consent before undergoing the procedure. Procedures were performed in the endoscopy suite under conscious sedation with midazolam and meperidine. A second procedure was completed in selected patients if the initial EUS-FNA was considered inconclusive for diagnosis.
Group I: Without onsite cytotechnologist
Group I included patients who had undergone EUS-FNA between September 1, 2005, and August 31, 2006. The number of needle passes for FNA was at the discretion of the endosonographer and was determined by the degree of difficulty or when it appeared that adequate material had been obtained. The aspirated specimen was smeared onto cytology slides by the assisting endoscopy nurse. One-half of the prepared slides were fixed with an alcohol-based fixative and the other half were air-dried. All slides were then sent to the pathology department for final cytopathological interpretation.
Group II: onsite cytotechnologist
Group II included patients who had undergone EUS-FNA between September 1, 2006, and August 31, 2007. At the start of the procedure, the cytotechnologist was notified so that he/she could be in the endoscopy suite by the time the first needle pass was completed. The cytotechnologist prepared all slides and stained them in the endoscopy suite with the Diff-Quik method (American Scientific Products, USA). The stained specimen was then assessed microscopically for adequacy of tissue sampling. If the target lesion was considered to have been satisfactorily sampled, additional needle passes were undertaken only at the discretion of the endosonographer. These additional specimens, when obtained, were stored in a liquid medium for subsequent cell block preparation. All specimens obtained and/or stained in the endoscopy suite were then taken by the cytotechnologist to the pathology department for final cytopathological interpretation.
Outcome measures
Based on the pathologist’s report, the final cytopathological diagnosis was categorized into two major categories: definite (definitely positive or definitely negative); and inconclusive. The primary outcome was the difference in the number of definite and inconclusive diagnostic categories between group I and group II. A secondary outcome was the difference in the number of needle passes undertaken in the two groups.
Ethics
The study was approved by the institutional Health Research Ethics Board of the University of Alberta including review of patient charts.
Statistical analysis
The statistical analysis was performed using the Internet-based Simple Interactive Statistical Analysis software. The t-test was used to analyze the difference between groups I and II (95% CIs) and confirmed the P values with the χ2 and Pearson statistic. Statistical significance was set at P=0.05
RESULTS
The patient demographics and procedure characteristics for group I and group II are shown in Table 1. In both groups, sex distribution was almost equal (51% women). The patients in group I were slightly older than group II with a mean age of 65 years (range 23 to 84 years) versus the mean age of 61 years (range 26 to 86 years) for group II, respectively. The distribution of EUS-FNA target sites was comparable between the two groups (Table 1). Solid masses in the pancreas were the most common FNA target site in both groups (45% in group I versus 37% in group II). This was followed by mediastinal lymph nodes and abdominal lymph nodes or masses (25% and 18% in group I versus 25% and 23% in group II, respectively). A total of 49 EUS-FNA procedures were performed in 47 patients in group I whereas 60 EUS-FNA procedures were performed in 55 patients in group II. The total number of needle passes in group I was 105 (mean 2.14 per patient; range one to five needle passes) versus 158 in group II (mean 2.63 per patient; range one to four needle passes). This difference was not statistically significant.
TABLE 1.
Patient demographics and procedure characteristics
| Group I | Group II | P | |
|---|---|---|---|
| Patients, n | 47 | 55 | |
| EUS-FNA procedures, n | 49 | 60 | |
| Age, years, mean (range) | 65 (23–84) | 61 (26–86) | |
| Men:women | 23:24 | 27:28 | |
| Fine-needle aspiration target site, n (%) | |||
| Pancreatic mass | 22 (45) | 22 (37) | |
| Mediastinal mass | 12 (25) | 15 (27) | |
| Abdominal mass | 9 (18) | 14 (22) | |
| Submucosal mass | 4 (8) | 4 (7) | |
| Miscellaneous | 2 (4) | 5 (8) | |
| Needle passes, n | |||
| Total | 105 | 158 | NS |
| Per patient, mean (range) | 2.14 (1–5) | 2.63 (1–4) | NS |
| Diagnosis, n (%) | |||
| Definite | 26 (53) | 46 (77) | 0.01 |
| Inconclusive | 23 (47) | 14 (23) | 0.001 |
EUS-FNA Endoscopic ultrasound-guided fine-needle aspiration; NS Not significant
The final cytopathological diagnosis was reported as definite (definitely positive or definitely negative) in 26 of 49 (53%) EUS-FNA procedures in group I compared with 46 of 60 (77%) EUS-FNA procedures in group II (P=0.01; 95% CI 0.04 to 0.30). Table 2 displays a breakdown of the specific EUS-FNA diagnoses. In the present cohort, three of 26 (12%) patients in group I and nine of 46 (20%) patients in group II were diagnosed as definitely negative, repectively (Table 2). All patients with a definitely negative diagnosis in each group were confirmed benign on surgical histology.
TABLE 2.
Diagnoses and rates of definitely positive and definitely negative endoscopic ultrasound-guided fine-needle aspiration cytology
| Diagnosis | Group I: Definitely positive (88%) | Group II: Definitely positive (80%) |
|---|---|---|
| Pancreatic cancer | 11 | 8 |
| Pancreatic neuroendocrine tumour | 2 | 4 |
| Esophageal cancer | 3 | 3 |
| Non-small-cell lung cancer | 3 | 12 |
| Small-cell lung cancer | 1 | 1 |
| Metastatic adenocarcinoma | 1 | 5 |
| Lymphoma | 1 | 1 |
| Gastrointestinal stromal tumour | 1 | 1 |
| Miscellaneous | – | 2 |
| Subtotal | 23 | 37 |
| Definitely negative* (12%) | Definitely negative* (20%) | |
| Benign lymph nodes | 2 | 1 |
| Pancreatic pseudocyst | 1 | 1 |
| Benign epithelial hyperplasia | – | 3 |
| Lymphadenitis | – | 1 |
| Reactive lymphoid hyperplasia | – | 1 |
| Fat necrosis | – | 1 |
| Chronic pancreatitis | – | 1 |
| Subtotal | 3 | 9 |
| Total | 26 | 46 |
All confirmed on surgical histology
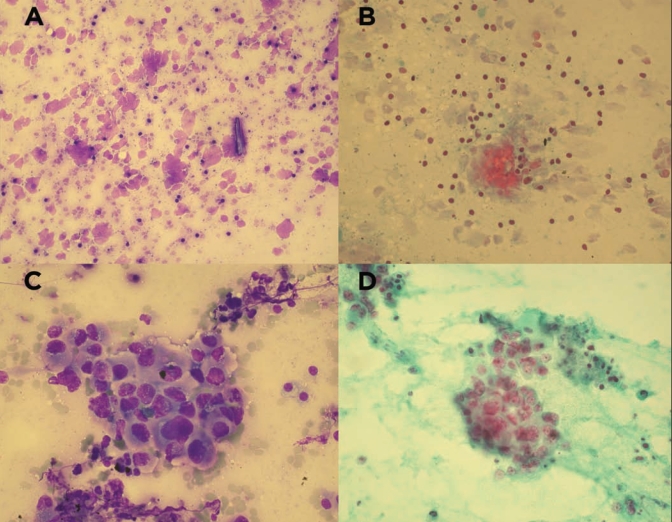
Inconclusive diagnoses were reported in 23 of 49 (47%) EUS-FNA procedures in group I and 14 of 60 (23%) EUS-FNA procedures in group II (P=0.001; 95% CI −0.48 to 0.13). Of the 23 EUS-FNA procedures with an inconclusive diagnosis in group I, there were four cases (17%) that were reported to have crush artefact (Figure 1) whereas none of the 14 cases with an inconclusive diagnosis in group II were reported to have crush artefact.
Figure 1).
A Diff-Quik (American Scientific Products, USA) staining shows significant air-drying artefact with nuclear fragmentation, original magnification ×200; B Papanicolaou staining shows neoplastic nuclear crushing due to air-drying artefact while smaller lymphocytes are not affected (original magnification ×200); C, D Diff-Quik and Papanicolaou staining, respectively, of moderately differentiated adenocarcinoma showing preserved nucleocytoplasmic architecture and no crush or air-drying artefact (original magnification ×400)
DISCUSSION
EUS-FNA is an accurate, safe and cost-effective technique to obtain tissue samples for the diagnosis and staging of various luminal and nonluminal gastrointestinal cancers, as well as staging of lung cancer. Published data suggest that the presence of a cytopathologist in the endoscopy suite during EUS-FNA is useful and cost-effective. In a series of 67 patients with metastatic lung cancer (22), EUS-FNA with onsite cytopathology interpretation reported a diagnostic accuracy of 92%, thereby avoiding the need for surgery in 68% of patients. Klapman et al (23) also demonstrated that the presence of an onsite cytopathologist improved the diagnostic yield of EUS-FNA. In this study, EUS-FNA results from two university hospitals were reviewed. At centre 1, 108 patients underwent EUS-FNA in the presence of an onsite cytopathologist. At centre 2, where an onsite cytopathologist was not available, 87 patients underwent EUS-FNA. All procedures at both hospitals were performed by the same endosonographer. At centre 1, a definite diagnosis of positive or negative for malignancy was reported in 78% compared with 52% for centre 2 (P=0.001), and the proportion of patients with an unsatisfactory specimen was 9% compared with 20% in centre 2 (P=0.035). Erickson et al (21) also found that the diagnostic yield of EUS-FNA dropped by 10% to 15% when a cytopathologist was not available onsite, in addition to prolonging procedure time and potentially increasing the procedural risk from multiple needles passes.
These data are not limited to EUS-FNA alone. A review of 5688 patients by Nasuti et al (14) proved the diagnostic accuracy and cost-effectiveness of onsite cytopathology evaluation. In this study, all FNA procedures performed by ultrasound, computed tomography, bronchoscopy, fluoroscopy and EUS over a five-year period were included. The proportion of non-diagnostic or inconclusive specimens, defined as inadequate cellularity, was 0.98%. This rate was compared with the published rate of nondiagnostic FNA (20%) when onsite cytopathology evaluation was not used. The authors also report a cost benefit of US$404,000 per year by reducing the number of repeat FNA procedures for nondiagnostic specimens. Cost savings by preventing repeat procedures have also been shown by other investigators (24,25).
Chang et al (9) reported that in the presence of an onsite cytopathologist during EUS-FNA, adequate specimens were obtained in 100% of patients compared with only 29% in the absence of an onsite cytopathologist. This discrepancy ultimately caused a change in policy at their centre requiring that all EUS-FNA procedures be performed in the presence of an onsite cytopathologist. However, resource and staff limitations do not support the widespread implementation of such a policy.
Cytotechnologists, on the other hand, are less resourceintensive but trained to optimally process cytological specimens and assess for adequacy of target tissue sampling. This minimizes the incidence of crush and air-drying artefacts seen with slides prepared by less-experienced individuals, and ensures adequacy of sample cellularity. In our study, we have demonstrated that onsite processing and interpretation of cytological specimens by a cytotechnologist had a significant impact on the diagnostic yield of EUS-FNA. To our knowledge, the impact of an onsite cytotechnologist has not been reported previously.
There are, however, some limitations in the design of our study. The present study was a single-centre retrospective review of a relatively small number of patients. Although the same endosonographer performed all procedures, not all samples were handled and prepared by the same endoscopy nurse or cytotechnologist and they were not all interpreted by the same cytopathologist. This may have led to differences in preparation and interpretation, but these limitations cannot be avoided in tertiary hospitals that have multiple personnel providing a specific service. A factor that has not been well described in the literature is the ability of individual cytotechnologists to differentiate the glandular cells obtained from the target lesion from those contaminating the needle during its passage across the glandular epithelium of the gastrointestinal wall. Perhaps this could explain some of the false-positive interpretations of adequate specimen cellularity in group II and could possibly be an explanation for the relatively low diagnostic yield of 77% in this group. This, however, is purely speculative and must be studied in further detail, especially if provisions for onsite cytotechnology interpretation are to increase in the future.
Another drawback of our study is the number of needle passes undertaken for EUS-FNA. Although there was no significant difference in the mean number of needle passes between the two groups, group I (without an onsite cytotechnologist) only had a mean of 2.14 needle passes per patient. Depending on the targeted site, it is recommended to perform five to seven needle passes on pancreatic lesions and two to five needle passes on lymph nodes when a cytopathologist is not available in the endoscopy suite (20,21). Therefore, we recognize this as a potential shortcoming of our study and realize that the diagnostic accuracy could have been increased in group I by increasing the number of needle passes undertaken. However, even if we speculate that the diagnostic yield of EUS-FNA can be improved significantly by increasing the number of needle passes, we have shown that with the presence of an onsite cytotechnologist, we can achieve high diagnostic yields by reducing the average number of needle passes although EUS-FNA, generally speaking, is a fairly safe intervention. Limiting the number of needle passes, in our experience, are in situations of technical difficulty such as those encountered with tumours of the uncinate process of the pancreas or when tissue vascularity precludes multiple needle passes.
CONCLUSION
Our study shows that onsite cytotechnologist interpretation during EUS-FNA has a significant clinical impact by increasing the diagnostic yield of EUS-FNA. Although a cost analysis was not performed as part of the present study, one can speculate this approach to be cost-saving by preventing repeat procedures. We therefore recommend allocating a cytotechnologist for onsite interpretation for adequacy of tissue sampling in centres where the services of a cytopathologist are not available.
Acknowledgments
The authors acknowledge Stephen Wreakes, Medical Photography, Capital Health, for his assistance with the photographic images and Raj Sandha for her assistance with the statistical analysis.
Footnotes
NOTE: The present study was presented as a poster at the Annual Meeting of the Canadian Association of Gastroenterology/Canadian Digestive Diseases Week, February 29-March 4, 2008, Montreal, Quebec.
REFERENCES
- 1.Rice TW, Boyce GA, Sivak MV, Adelstein DJ, Kirby TJ. Esophageal carcinoma: esophageal ultrasound assessment of preoperative chemotherapy. Ann Thorac Surg. 1992;53:972–7. doi: 10.1016/0003-4975(92)90369-f. [DOI] [PubMed] [Google Scholar]
- 2.Boyce GA, Sivak MV, Jr, Lavery, et al. Endoscopic ultrasound in the pre-operative staging of rectal carcinoma. Gastrointest Endosc. 1992;38:468–71. doi: 10.1016/s0016-5107(92)70478-7. [DOI] [PubMed] [Google Scholar]
- 3.Heintz A, Mildenberger P, Georg M, Braunstein S, Junginger T. Endoscopic ultrasonography in the diagnosis of regional lymph nodes in esophageal and gastric cancer – results of studies in vitro. Endoscopy. 1993;25:231–5. doi: 10.1055/s-2007-1010298. [DOI] [PubMed] [Google Scholar]
- 4.Eloubeidi MA. Endoscopic ultrasound-guided fine-needle aspiration in the staging and diagnosis of patients with lung cancer. Semin Thorac Cardiovasc Surg. 2007;19:206–11. doi: 10.1053/j.semtcvs.2007.07.008. [DOI] [PubMed] [Google Scholar]
- 5.Pellise Urquiza M, Fernandez-Esparrach G, Sole M, et al. Endoscopic ultrasound-guided fine needle aspiration: predictive factors of accurate diagnosis and cost-minimization analysis of on-site pathologist. Gastroenterol Hepatol. 2007;30:319–24. doi: 10.1157/13107565. [DOI] [PubMed] [Google Scholar]
- 6.Eloubeidi MA, Jhala D, Chhieng DC, et al. Yield of endoscopic ultrasound-guided fine-needle aspiration biopsy in patients with suspected pancreatic carcinoma. Cancer. 2003;99:285–92. doi: 10.1002/cncr.11643. [DOI] [PubMed] [Google Scholar]
- 7.Brand B, Pfaff T, Binmoeller KF, Sriram, et al. Endoscopic ultrasound for differential diagnosis of focal pancreatic lesions, confirmed by surgery. Scand J Gastroenterol. 2000;35:1221–8. doi: 10.1080/003655200750056736. [DOI] [PubMed] [Google Scholar]
- 8.Aithal GP, Anagnostopoulos GK, Tam W, et al. EUS-guided tissue sampling: Comparison of “dual sampling” (Trucut biopsy plus FNA) with “sequential sampling” (Trucut biopsy and then FNA as required) Endoscopy. 2007;39:725–30. doi: 10.1055/s-2007-966400. [DOI] [PubMed] [Google Scholar]
- 9.Chang KJ, Katz KD, Durbin TE, et al. Endoscopic ultrasound-guided fine-needle aspiration. Gastrointest Endosc. 1994;40:694–9. [PubMed] [Google Scholar]
- 10.Storch I, Jorda M, Thurer R, et al. Advantage of EUS Trucut biopsy combined with fine-needle aspiration without immediate on-site cytopathologic examination. Gastrointest Endosc. 2006;64:505–11. doi: 10.1016/j.gie.2006.02.056. [DOI] [PubMed] [Google Scholar]
- 11.Chhieng DC, Jhala D, Jhala N, et al. Endoscopic ultrasound-guided fine-needle aspiration biopsy: A study of 103 cases. Cancer. 2002;96:232–9. doi: 10.1002/cncr.10714. [DOI] [PubMed] [Google Scholar]
- 12.Gress FG, Hawes RH, Savides TJ, Ikenberry SO, Lehman GA. Endoscopic ultrasound-guided fine-needle aspiration biopsy using linear array and radial scanning endosonography. Gastrointest Endosc. 1997;45:243–50. doi: 10.1016/s0016-5107(97)70266-9. [DOI] [PubMed] [Google Scholar]
- 13.Bentz JS, Kochman ML, Faigel DO, Ginsberg GG, Smith DB, Gupta PK. Endoscopic ultrasound-guided real-time fine-needle aspiration: Clinicopathologic features of 60 patients. Diagn Cytopathol. 1998;18:98–109. doi: 10.1002/(sici)1097-0339(199802)18:2<98::aid-dc4>3.0.co;2-p. [DOI] [PubMed] [Google Scholar]
- 14.Nasuti JF, Gupta PK, Baloch ZW. Diagnostic value and cost-effectiveness of on-site evaluation of fine-needle aspiration specimens: Review of 5,688 cases. Diagn Cytopathol. 2002;27:1–4. doi: 10.1002/dc.10065. [DOI] [PubMed] [Google Scholar]
- 15.Ducatman BS, Hogan CL, Wang HH. A triage system for processing fine needle aspiration cytology specimens. Acta Cytol. 1989;33:797–9. [PubMed] [Google Scholar]
- 16.Mazza E, Maddau C, Ricciardi A, Falchini M, Matucci M, Ciarpallini T. On-site evaluation of percutaneous CT-guided fine needle aspiration of pulmonary lesions. A study of 321 cases. Radiol Med (Torino) 2005;110:141–8. [PubMed] [Google Scholar]
- 17.Campisi P, Accinelli G, De Angelis C, Pacchioni D, Bussolati G. [On-site evaluation and triage for endoscopic ultrasound-guided fine needle aspiration cytology. The Turin experience] Minerva Med. 2007;98:395–400. [PubMed] [Google Scholar]
- 18.Layfield LJ, Bentz JS, Gopez EV. Immediate on-site interpretation of fine-needle aspiration smears: A cost and compensation analysis. Cancer. 2001;93:319–22. doi: 10.1002/cncr.9046. [DOI] [PubMed] [Google Scholar]
- 19.Diacon AH, Schuurmans MM, Theron J, et al. Utility of rapid on-site evaluation of transbronchial needle aspirates. Respiration. 2005;72:182–8. doi: 10.1159/000084050. [DOI] [PubMed] [Google Scholar]
- 20.LeBlanc JK, Ciaccia D, Al-Assi MT, et al. Optimal number of EUS-guided fine needle passes needed to obtain a correct diagnosis. Gastrointest Endosc. 2004;59:475–81. doi: 10.1016/s0016-5107(03)02863-3. [DOI] [PubMed] [Google Scholar]
- 21.Erickson RA, Sayage-Rabie L, Beissner RS. Factors predicting the number of EUS-guided fine-needle passes for diagnosis of pancreatic malignancies. Gastrointest Endosc. 2000;51:184–90. doi: 10.1016/s0016-5107(00)70416-0. [DOI] [PubMed] [Google Scholar]
- 22.Tournoy KG, Praet MM, Van Maele G, Van Meerbeeck JP. Esophageal endoscopic ultrasound with fine-needle aspiration with an on-site cytopathologist: High accuracy for the diagnosis of mediastinal lymphadenopathy. Chest. 2005;128:3004–9. doi: 10.1378/chest.128.4.3004. [DOI] [PubMed] [Google Scholar]
- 23.Klapman JB, Logrono R, Dye CE, Waxman I. Clinical impact of on-site cytopathology interpretation on endoscopic ultrasound-guided fine needle aspiration. Am J Gastroenterol. 2003;98:1289–94. doi: 10.1111/j.1572-0241.2003.07472.x. [DOI] [PubMed] [Google Scholar]
- 24.Sauer T, Freng A, Djupesland P. Immediate interpretation of FNA smears from the head and neck region. Diagn Cytopathol. 1992;8:116–8. doi: 10.1002/dc.2840080205. [DOI] [PubMed] [Google Scholar]
- 25.Silverman JF, Finley JL, O’Brien KF, et al. Diagnostic accuracy and role of immediate interpretation of fine needle aspiration biopsy specimens from various sites. Acta Cytol. 1989;33:791–6. [PubMed] [Google Scholar]