Abstract
Background
The association of aggressive periodontitis with Kindler syndrome was based on a single case in 1996 and later confirmed with a larger population. Since then, significant research has greatly increased our understanding of the molecular pathology of this disorder. We review recent advances in the molecular mechanisms of the syndrome and present a maintenance case report of a patient who has been followed in our clinic.
Methods
A female patient who was diagnosed with Kindler syndrome and aggressive periodontitis at the age of 16 years has been followed and treated in our clinic for 12 years. Her main treatment has been maintenance therapy following her initial treatment and restorative work previously documented. Gingival biopsies obtained during the recent extraction of hopeless maxillary molars were used for histologic assessment of gingival tissue attachment apparatus and to isolate gingival fibroblasts. Reverse transcription-polymerase chain reaction (RT-PCR) was performed using these cells to confirm the lack of expression of kindlin-1.
Results
RT-PCR showed the total loss of kindlin-1 mRNA in cultured gingival fibroblasts, supporting the clinical diagnosis of Kindler syndrome. Tissue biopsies revealed atypical pocket epithelium. Maintenance therapy has been moderately successful. Teeth that were recently lost had a poor prognosis at the initial assessment. The patient’s gingiva and oral mucosa continue to be fragile with episodes of sloughing and inflammation.
Conclusions
Periodontitis in Kindler syndrome responds to maintenance therapy, but the gingiva and oral mucosa continue to display an abnormal appearance with white patches. Histologic findings suggest that the junctional epithelium in Kindler syndrome may be abnormal and could explain why these patients have periodontal disease. Attachment loss progressed around teeth with an initial guarded or poor prognosis. Teeth that started with a good or fair prognosis continue to have a fair prognosis. Limited dental implant treatment is being considered.
Keywords: Case report, kindlin-1, periodontitis, treatment outcome
The association of aggressive periodontitis with Kindler syndrome (KS) was proposed based on a single case in 1996 and later confirmed with a larger population of patients.1,2 KS is an autosomal recessive disorder‡ that was first described in 1954 by Dr. Theresa Kindler.3 The first patient described by Kindler had gingiva that readily bled, skin on the dorsa of her hands and feet with a thin, wrinkled appearance, and webbing between the second and third toes of both feet. By the age of 10 years, the blistering and sun sensitivity had resolved, but the skin remained thin and fragile. Gingival fragility and early and rapidly progressive periodontitis (aggressive periodontitis) were common features in this original case and other cases of KS.2 Other features vary between cases and include: photosensitivity; acral hyperkeratosis; nail dystrophy; webbing and contractures of the fingers and toes; alopecia; actinic changes; mucosal involvement, including urethral, vaginal, anal, esophageal, and oral commissural stenosis (narrowing or constricting); eversion of the eyelids; pigmentation of the lips; and malformation of the nails. Common dermatologic findings include congenital skin blistering that resolves slowly with age; mild photosensitivity that improves with age; and early, generalized, progressive poikiloderma with extensive atrophy.4
MOLECULAR PATHOLOGY
Recently, the gene responsible for KS was localized on the short arm of chromosome 20 and named kindlin-1.5,6 Two transcripts for kindlin-1 have been described in public databases predicting proteins of 230 and 677 amino acids. Kindlin-1 is closely related to other members of the three-member kindlin family of cytoplasmic focal adhesion proteins (kindlin-1, -2, and -3).7 Expression of the large variant of kindlin-1 is high in keratinocytes but relatively low in fibroblasts.6–8 The small splice variant of kindlin-1 has been detected in kidney, colon, and small intestine, but it remains unknown whether truncated protein is expressed or has a function.7 Kindlin-2 (also known as Mig-2) is expressed in all tissue examined, whereas kindlin-3 expression is limited to hematopoietic cells.7 Proliferation of keratinocytes from patients with KS is reduced, and kindlin-1–deficient keratinocytes show increased apoptosis.9 Kindlin-1 is significantly overexpressed in lung and colon carcinomas.10 Kindlin-1 localizes mainly at the basal aspect of the basal keratinocytes in vivo.6
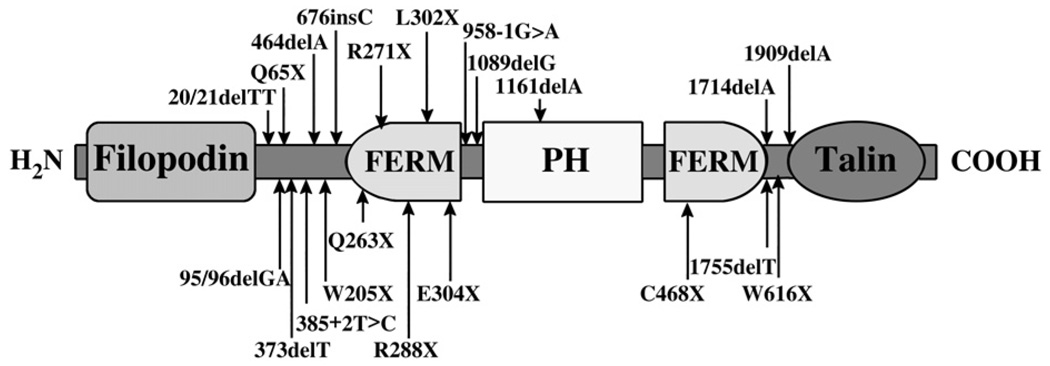
Kindlin-1 contains several domains (Fig. 1) that are homologous to a group of proteins that link the cytoskeleton to the cell membrane. The four-point one, ezrin, radixin, moesin (FERM) domain found in kindlin-1 is present in several other proteins that link to the plasma membrane, such as talin, ezrin-radixin-moesin family proteins, and several tyrosine kinases and phosphatases.11 The FERM domain in talin is involved in the binding of the integrin tail and integrin activation.12,13 The pleckstrin homology (PH) domain, which can bind directly to phosphoinositides, is another plasma membrane–targeting domain that is found in kindlin-1.14 It is likely that kindlin-1 participates in the formation of focal adhesions through the binding and recruiting of other molecules, as has been shown for kindlin-2.15,16 Kindlin-1 could serve as a direct link between extracellular matrix–sensing integrins and actin-binding cytoplasmic proteins because it can bind cytoplasmic tails of β1 and β3 integrins, and it colocalizes with actin microfilaments and paxillin in focal adhesions.7,8 Colocalization seems to be stronger in kindlin-1–overexpressing transfected fibroblasts (that normally express low levels of this protein) compared to keratinocytes that exhibit an abundant intracellular perinuclear pool of kindlin-1,7 suggesting that kindlin-1 could also possess functions outside focal adhesions. Kindlin-1–deficient keratinocytes show reduced cell adhesion and proliferation and increased apoptosis,9 which might explain the atrophic features of the skin and mucosa in patients who have KS.
Figure 1.
The kindlin-1 protein with FERM and PH domains and regions of homology with filipodin and talin. Locations of reported positions of all loss-of-function mutations within the KIND1 gene are shown, including nonsense, frameshift, or splice mutations (indicated by arrows) but excluding large deletions. COOH = carboxylic acid.
A number of single-nucleotide mutations have been reported in patients with KS in several publications5,6,17–20 since the identification of the kindlin gene (Fig. 1). The vast majority of these mutations tend to concentrate around the area coding the FERM domain. Recently, a novel large genomic deletion of ∼3.9 kilobases (kb) was discovered in four patients.17 This mutation deletes exons 10 and 11 from the KIND1 mRNA and leads to truncated kindlin-1 protein. Excluding the large deletions, an up-to-date summary of the reported mutations for the KIND1 gene is depicted in Figure 1.
HISTOLOGIC FEATURES
Histologically, skin lesions of patients affected by KS are characterized by epidermal atrophy, focal vacuolization of the basal layer of the epithelium, and pigmentary incontinence in the upper dermis, in addition to a mild lymphocytic infiltrate, consistent with poikiloderma. Electron microscopy shows extensive reduplication and disruption of the lamina densa along the dermoepidermal junction beneath the basal cells and cleft formation that occurs in the lamina lucida.5,21–24 These histologic features, although not specific to KS, are useful in distinguishing the disorder from dystrophic epidermolysis bullosa. Before identification of mutations in the KIND1 gene, the ultrastructural reduplication of the lamina densa was one of the most useful clues in diagnosing KS. Desmosomes, hemidesmosomes, tonofilaments, anchoring filaments, and fibrils appear normal. In an immunofluorescence study by Wiebe and Larjava,21 the distribution of the basement membrane zone components appeared normal, with the exception of breaks. In addition, type VII collagen in KS is found in abnormal locations deep in the connective tissue stroma.21,25 The molecular mechanism through which the lack of kindlin-1 leads to duplication of basement membrane and abnormal distribution of type VII collagen is not known.
CASE REPORT
This is a follow-up of a case first presented by Wiebe et al.1 Initially, the patient presented as a 16-year-old female on referral from a local children’s hospital dermatology department with a chief complaint of painful and bleeding gums. She had been diagnosed as having “Weary-Kindler” syndrome, which at the time was poorly understood and was assigned based on clinical features and medical history. KS has since been differentiated from Weary’s hereditary acrokeratotic poikiloderma26 and a form of epidermolysis bullosa.
During the initial examination, the patient presented with seven missing mandibular teeth, no caries, generalized tooth mobility, and limited localized plaque and calculus but with significant spontaneous bleeding and bleeding on probing. The gingiva was thin and fragile, and the epithelium sloughed with minor abrasion. The gingival margins were rolled and cratered and lifted away from the tooth surface with a gentle blast of air. Pocketing of 6 to 11 mm was noted as well as localized areas of recession of 1 to 3 mm. Radiographically, the roots appeared relatively short, and the bone loss extended into the apical third around the maxillary central incisors and to the middle third around many posterior teeth. Bacterial tests were negative for Aggregatibacter actinomycetemcomitans (previously Actinobacillus actinomycetemcomitans) and showed only low levels of Porphyromonas gingivalis, Prevotella intermedia, Eikenella corrodens, Fusobacterium nucleatum, Campylobacter rectus, Tannerella forsythia (previously T. forsythensis), and Treponema denticola.1
Conservative periodontal therapy was provided along with extraction of one maxillary central incisor. A gingivectomy was done on the palatal aspect of the maxillary anterior teeth. A lower partial denture, designed to keep the major connectors away from the gingival margins, was constructed, and a splinted bridge was placed in the maxilla from canine to canine. Root canal therapy was performed later on the remaining maxillary incisors that became non-vital. Although the prognosis remained guarded to poor for many of the retained teeth, the treatment was kept conservative given the concerns of having a denture resting on the fragile oral mucosa and the unknown response to surgery and osseointegrated dental implants.
Scaling and root planing and regular maintenance cleanings produced a significant improvement in the condition of the gingiva and her oral comfort. The number of sites with bleeding on probing decreased from 198 prior to treatment to 10, and the appearance of the gingiva improved, although it was not normal.1
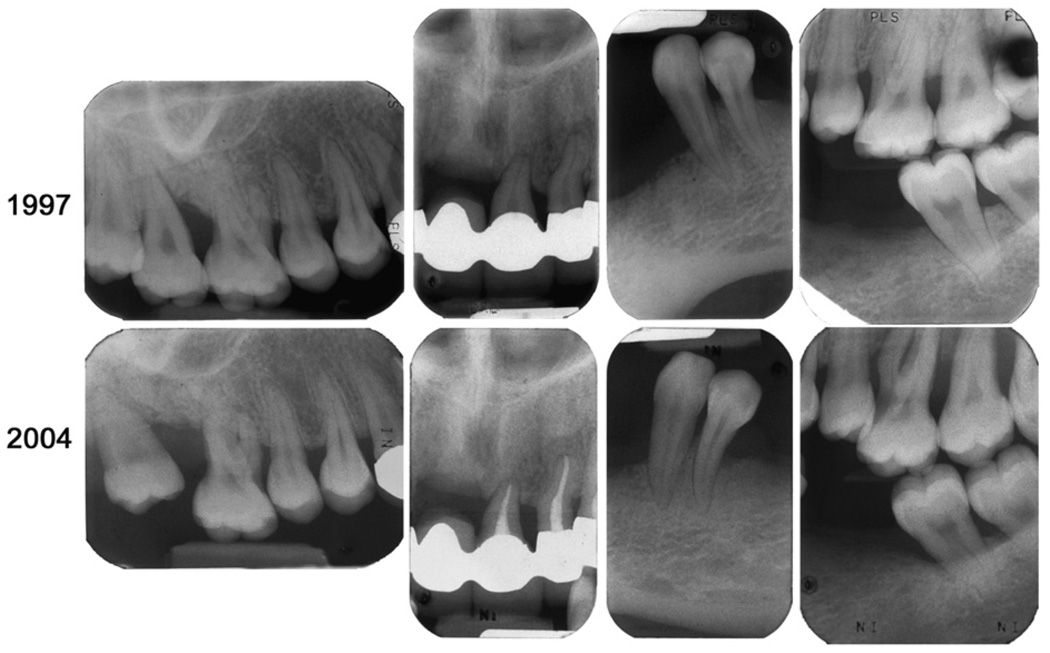
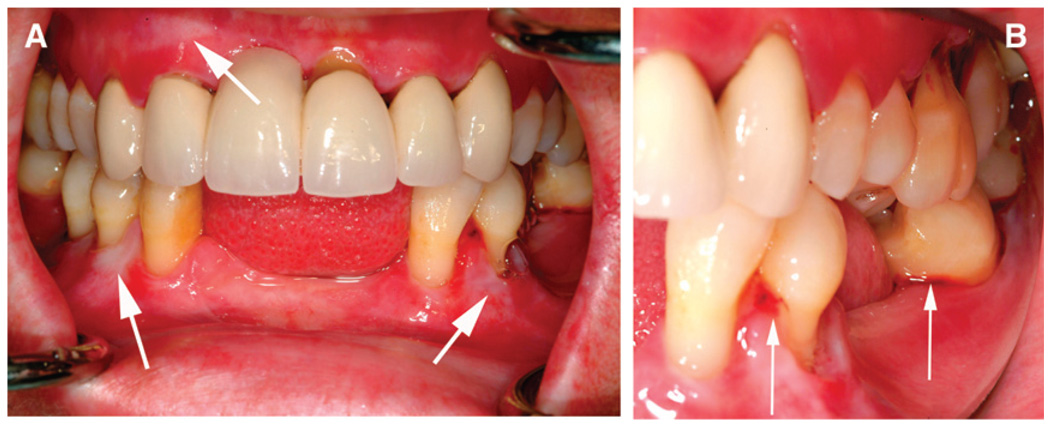
From 1996 to 2006, the only treatment provided was maintenance cleanings at ~4-month intervals. During the 12-year follow-up period, the patient started smoking cigarettes although she was advised against it. This continued for 3 years. Following her original periodontal and restorative treatment,1 she had only eight sites with probing depths >4 mm. At the most recent examination, several sites showed radiographic evidence of the progression of bone loss, and, clinically, pocket depths >4 mm were noted at 24 sites (Fig. 2). There was generalized mobility from “+” to “3” and Class 2 or 3 furcation involvement on all molars. Generalized recession of 1 to 6 mm was present. The effectiveness of plaque removal has been variable. The gingiva and mucosa continue to appear white and fragile (Fig. 3), with occasional spontaneous bleeding episodes, and her maxillary molars were sensitive to temperature. For those reasons, the patient requested to have her remaining maxillary molars removed in 2006. These teeth were extracted with a small amount of gingival tissue attached (approved by the Clinical Research Ethics Board, University of British Columbia) for isolation of gingival fibroblasts (for confirmation of her clinical diagnosis) and routine histopathologic assessment.
Figure 2.
Selective radiographic comparison of the bone levels 1 year after her initial treatment was completed (1997) and at the most recent complete survey (2004). Note the progression of bone loss around all of the maxillary molars, whereas some teeth with minimal bone support (remaining maxillary incisors) show no major alterations.
Figure 3.
Clinical intraoral photographs reveal the thin lichenoid appearance of the gingiva (arrows) (A) and localized sites of spontaneous bleeding (arrows) (B).
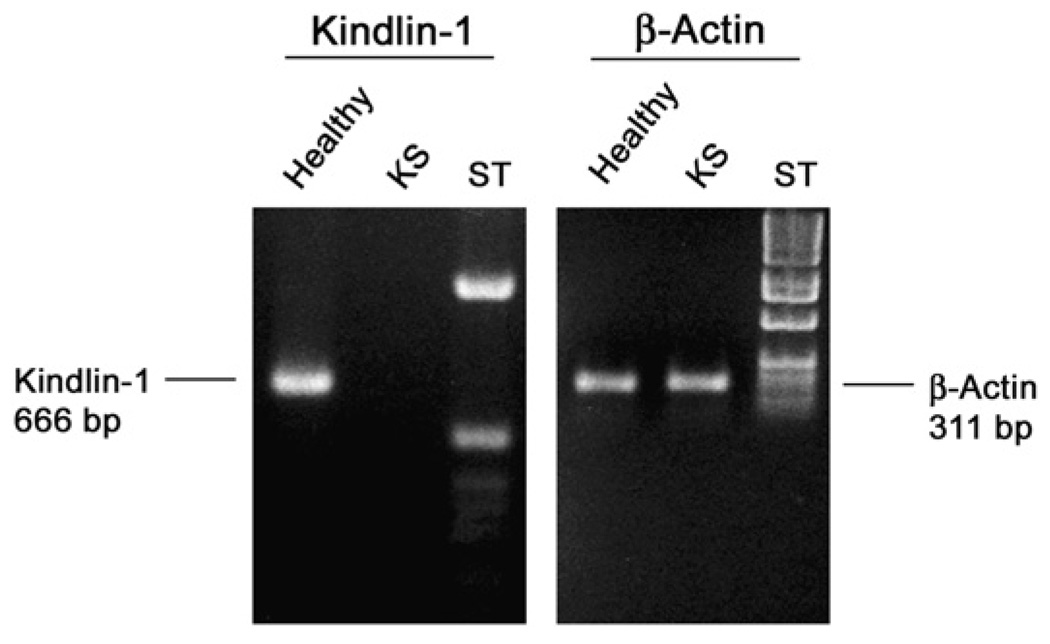
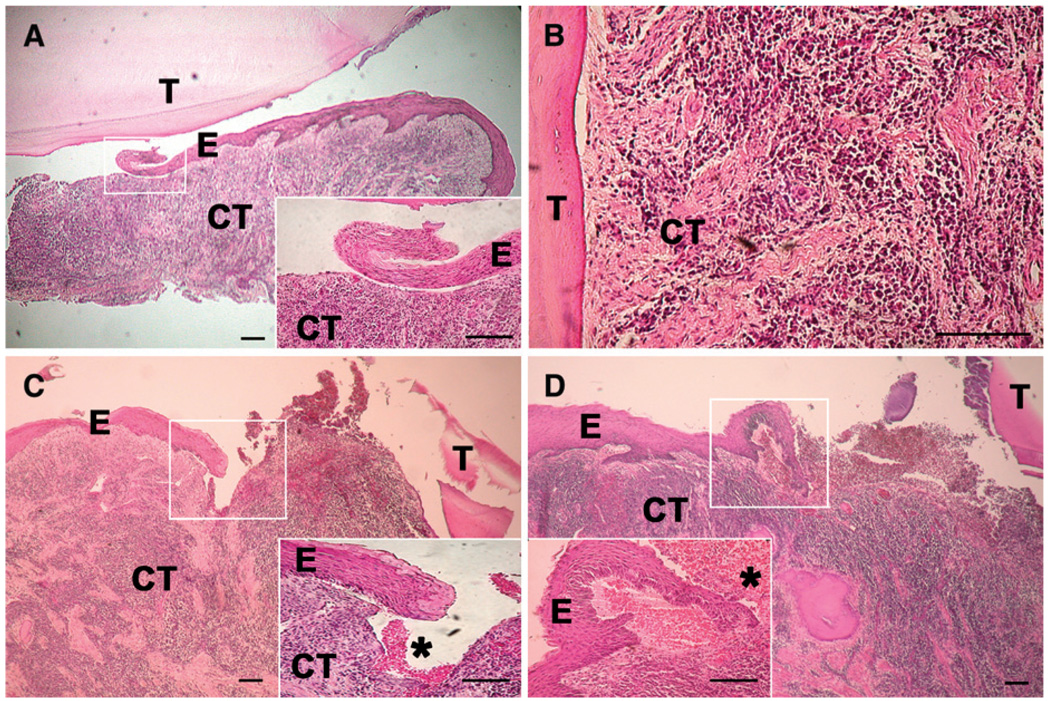
The exact mutation in this patient remained unknown. The initial screening by Siegel et al.6 failed to identify the mutation using a genomic polymerase chain reaction (PCR) strategy. It is possible that the mutation was cryptic or there was a defect in the promoter. Therefore, we investigated whether her cells were expressing kindlin-1. Gingival fibroblasts from the patient and from a healthy control were cultured using routine techniques.27 RNA was extracted and used for kindlin-1 reverse transcription (RT)-PCR (Fig. 4). Gingival fibroblasts were cultured in Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum. Total RNA was extracted from the cells.§ Three micrograms total RNA was reverse transcribed using oligodeoxythymidylic acid primer with a system.‖ For each PCR, 2 µl reverse-transcribed reaction mixture was combined with a total volume of 50 µl containing 20 mM Tris-HCl (pH 8.8), 10 mM KCl, 10 mM (NH4)2SO4, 2 mM MgSO4, 0.1% Triton-X100, 10mM deoxynucleotide triphosphates, 10 µM of each oligonucleotide primer, and 1 µl 1 unit/µl DNA polymerase. An initial denaturation step at 94°C for 1 minute and 30 seconds was followed by 25 cycles for β-actin and 50 cycles for kindlin-1 consisting of denaturation at 94°C, annealing at 62°C, and extension at 72°C, each for 40 seconds. A final extension step was carried out at 72°C for 5 minutes. The designed primers for kindlin-1 were 5′-TGGTTCAGTGACAGCCCTTTGA-3′ and 5′-CACAACTTCGCAGCCTCTAAG-3′, corresponding to positions 613 to 635 and 1,258 to 1,279 of kindlin-1 cDNA, respectively. The primers for β-actin were 5′-GAGACCTTCAACACCCCAGCC-3 and 5′-GGCCATCTCTTGCTCGAAGTC-3, which were described by Redlich et al.28 RT-PCR products were separated by 1% agarose gel electrophoresis and stained with ethidium bromide for visualization under an ultraviolet light. RT-PCR showed a total loss of kindlin-1 mRNA in cultured gingival fibroblasts from the affected patient (Fig. 4). Tissue biopsies revealed the presence of heavy inflammation, but no typical pocket epithelium or junctional epithelium was found in any of the specimens analyzed (Fig. 5).
Figure 4.
Lack of expression of kindlin-1 RNA in cells cultured from a patient with KS. ST = 1-kb DNA ladder; bp = base pairs.
Figure 5.
Histologic assessment of periodontal tissue of maxillary molars of the patient with KS. The teeth were removed gently with forceps, with a small amount of gingival tissue attached, and processed for routine histology. Atypical junctional/pocket epithelium was present in all specimens that showed proper morphology (three separate blocks shown in A, C, and D). In two specimens (A and C), the epithelium showed a blunt end with no attachment to the tooth surface. In one specimen (D), the end of the epithelium resembled epithelium migrating into a wound. Red blood cells (*) were visible in many samples, indicating trauma or spontaneous bleeding (as complained about by the patient). Heavy chronic inflammation was present in all specimens examined (A through D). The inflammatory infiltrates were often approaching the root surface deep in the connective tissue without the epithelial enclosure that is seen in classic periodontitis specimens (B, which represents a deeper area of the same specimen shown in A). Considering the extent of inflammation, the rete ridges were poorly developed or non-existent with signs of epithelial separation from the connective tissue (A, C, and D). T = tooth; E = epithelium; CT = connective tissue. Bars = 200 µm.
Limited implant therapy is under consideration. Unfortunately, the patient has limited opening of her mouth and an inadequate amount of bone coronal to the sinus to retain implants unless a sinus elevation procedure is performed. In addition to the technical difficulties of placing implants in the posterior maxilla, there is a lack of experience in determining the soft tissue response around implants in patients with KS.
DISCUSSION
Initially, this patient responded well to standard scaling and root planing. Many teeth that had a guarded or poor prognosis at the time of the initial examination were maintained for >10 years but continued to lose periodontal attachment. It is likely that the clinical picture would be more favorable if the patient had remained on a tighter maintenance schedule, had better home care, and had not taken up smoking. Nevertheless, it is likely that these teeth would have continued to lose attachment over time. Teeth that had a fair prognosis at the initial examination continue to be fair. The maxillary cuspid-to-cuspid bridge seems to have served her well, as has the cast lower partial denture that was constructed to have the major connector clear of the gingival margins.
In a cross-sectional study2 involving 20 subjects with KS, periodontitis had an early onset (teenage years), and its rate of progression was rapid, resembling aggressive periodontitis, compared to individuals without KS of the same geographic and ethnic origin. Generally, these patients did not receive dental care, and home care was minimal. The clinical manifestation of periodontal disease in patients with KS may show considerable variation, as is seen for other clinical symptoms, even with the same type of mutations.6 Because many patients with this syndrome have unreported periodontal status, the variety of disease manifestation remains to be explored. This case report provides some hope for these patients that their periodontal disease can be arrested, at least in part, with maintenance therapy. However, the risk for losing more attachment is high. We found no evidence of epithelial seal (i.e., junctional epithelium) to the teeth or the presence of proper pocket epithelium in any of the extracted teeth. If these findings are true, KS represents the first pathologic periodontal condition in which bacterial biofilm has direct access to periodontal connective tissue, similar to open chronic wounds. Because of its ability to participate in actin filament organization and cell adhesion, kindlin-1 may serve as a key intracellular protein mediating the adhesion of epithelial cells directly to the tooth.29
CONCLUSIONS
Traditional non-surgical periodontal treatment has been beneficial for this patient with KS, although teeth with a poor prognosis at the initial examination continued to lose support. Histopathologic findings suggest that the kindlin-1 protein may play a major role in maintaining adhesion of the junctional epithelium to the tooth.
ACKNOWLEDGMENTS
The authors thank Mrs. Ingrid Ellis, research grant facilitator, University of British Columbia, for editorial assistance in the final preparation of the manuscript. This work was supported by a grant (DE016099) from the National Institutes of Health, Bethesda, Maryland. The authors report no conflicts of interest related to this case report.
Footnotes
Online Mendelian Inheritance in Man (OMIM) #173650, Johns Hopkins University, Baltimore, MD.
RNeasy Mini Kit, Qiagen, Mississauga, ON.
SuperScript First-Strand Synthesis System, Invitrogen, Burlington, ON.
REFERENCES
- 1.Wiebe CB, Silver JG, Larjava HS. Early-onset periodontitis associated with Weary-Kindler syndrome. J Periodontol. 1996;67:1004–1010. doi: 10.1902/jop.1996.67.10.1004. [DOI] [PubMed] [Google Scholar]
- 2.Wiebe CB, Penagos H, Luong N, et al. Clinical and microbiologic study of periodontitis associated with Kindler syndrome. J Periodontol. 2003;74:25–31. doi: 10.1902/jop.2003.74.1.25. [DOI] [PubMed] [Google Scholar]
- 3.Kindler T. Congenital poikiloderma with traumatic bulla formation and progressive cutaneous atrophy. Br J Dermatol. 1954;66:104–111. doi: 10.1111/j.1365-2133.1954.tb12598.x. [DOI] [PubMed] [Google Scholar]
- 4.Patrizi A, Pauluzzi P, Neri I, Trevisan G, De Giorgi LB, Pasquinelli G. Kindler syndrome: Report of a case with ultrastructural study and review of the literature. Pediatr Dermatol. 1996;13:397–402. doi: 10.1111/j.1525-1470.1996.tb00709.x. [DOI] [PubMed] [Google Scholar]
- 5.Jobard F, Bouadjar B, Caux F, et al. Identification of mutations in a new gene encoding a FERM family protein with a pleckstrin homology domain in Kindler syndrome. Hum Mol Genet. 2003;12:925–935. doi: 10.1093/hmg/ddg097. [DOI] [PubMed] [Google Scholar]
- 6.Siegel DH, Ashton GH, Penagos HG, et al. Loss of kindlin-1, a human homolog of the Caenorhabditis elegans actin-extracellular matrix linker protein UNC-112, causes Kindler syndrome. Am J Hum Genet. 2003;73:174–187. doi: 10.1086/376609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ussar S, Wang HV, Linder S, Fassler R, Moser M. The kindlins: Subcellular localization and expression during murine expression. Exp Cell Res. 2006;312:3142–3151. doi: 10.1016/j.yexcr.2006.06.030. [DOI] [PubMed] [Google Scholar]
- 8.Kloeker S, Major MB, Calderwood DA, Ginsberg MH, Jones DA, Beckerle MC. The Kindler syndrome protein is regulated by transforming growth factor-β and involved in integrin-mediated adhesion. J Biol Chem. 2004;279:6824–6833. doi: 10.1074/jbc.M307978200. [DOI] [PubMed] [Google Scholar]
- 9.Herz C, Aumailley M, Schulte C, Schlotzer-Schrehardt U, Bruckner-Tuderman L, Has C. Kindlin-1 is a phosphoprotein involved in regulation of polarity, proliferation, and motility of epidermal keratinocytes. J Biol Chem. 2006;281:36082–36090. doi: 10.1074/jbc.M606259200. [DOI] [PubMed] [Google Scholar]
- 10.Weinstein EJ, Bourner M, Head R, Zakeri H, Bauer C, Mazzarella R. URP1: A member of a novel family of PH and FERM domain-containing membrane-associated proteins is significantly over-expressed in lung and colon carcinomas. Biochim Biophys Acta. 2003;1637:207–216. doi: 10.1016/s0925-4439(03)00035-8. [DOI] [PubMed] [Google Scholar]
- 11.Louvet-Vallee S. ERM proteins: From cellular architecture to cell signaling. Biol Cell. 2000;92:305–316. doi: 10.1016/s0248-4900(00)01078-9. [DOI] [PubMed] [Google Scholar]
- 12.Calderwood DA, Zent R, Grant R, Rees DJ, Hynes RO, Ginsberg MH. The Talin head domain binds to integrin beta subunit cytoplasmic tails and regulates integrin activation. J Biol Chem. 1999;274:28071–28074. doi: 10.1074/jbc.274.40.28071. [DOI] [PubMed] [Google Scholar]
- 13.Calderwood DA, Yan B, de Pereda JM, et al. The phosphotyrosine binding-like domain of talin activates integrins. J Biol Chem. 2002;277:21749–21758. doi: 10.1074/jbc.M111996200. [DOI] [PubMed] [Google Scholar]
- 14.Maffucci T, Falasca M. Specificity in pleckstrin homology (PH) domain membrane targeting: A role for a phosphoinositide-protein co-operative mechanism. FEBS Lett. 2001;506:173–179. doi: 10.1016/s0014-5793(01)02909-x. [DOI] [PubMed] [Google Scholar]
- 15.Tu Y, Wu S, Shi X, Chen K, Wu C. Migfilin and Mig-2 link focal adhesions to filamin and the actin cytoskeleton and function in cell shape modulation. Cell. 2003;113:37–47. doi: 10.1016/s0092-8674(03)00163-6. [DOI] [PubMed] [Google Scholar]
- 16.Wu C. Migfilin and its binding partners: From cell biology to human diseases. J Cell Sci. 2005;118:659–664. doi: 10.1242/jcs.01639. [DOI] [PubMed] [Google Scholar]
- 17.Has C, Wessagowit V, Pascucci M, et al. Molecular basis of Kindler syndrome in Italy: Novel and recurrent Alu/Alu recombination, splice site, nonsense, and frameshift mutations in the KIND1 gene. J Invest Dermatol. 2006;126:1776–1783. doi: 10.1038/sj.jid.5700339. [DOI] [PubMed] [Google Scholar]
- 18.Ashton GHS, McLean WHI, South AP, et al. Recurrent mutations in kindlin-1, a novel keratinocyte focal contact protein, in the autosomal recessive skin fragility and photosensitivity disorder, Kindler syndrome. J Invest Dermatol. 2004;122:78–83. doi: 10.1046/j.0022-202X.2003.22136.x. [DOI] [PubMed] [Google Scholar]
- 19.Sethuraman G, Fassihi H, Ashton GHS, et al. An Indian child with Kindler syndrome resulting from a new homozygous nonsense mutation (C468X) in the KIND1 gene. Clin Exp Dermatol. 2005;30:286–288. doi: 10.1111/j.1365-2230.2004.01712.x. [DOI] [PubMed] [Google Scholar]
- 20.Sadler E, Klausegger A, Muss W, et al. Novel KIND1 gene mutation in Kindler syndrome with severe gastrointestinal tract involvement. Arch Dermatol. 2006;142:1619–1624. doi: 10.1001/archderm.142.12.1619. [DOI] [PubMed] [Google Scholar]
- 21.Wiebe CB, Larjava HS. Abnormal deposition of type VII collagen in Kindler syndrome. Arch Dermatol Res. 1999;291:6–13. doi: 10.1007/s004030050377. [DOI] [PubMed] [Google Scholar]
- 22.Yasukawa K, Sato-Matsumura KC, McMillan J, Tsuchiya K, Shimizu H. Exclusion of COL7A1 mutation in Kindler syndrome. J Am Acad Dermatol. 2002;46:447–450. doi: 10.1067/mjd.2002.117523. [DOI] [PubMed] [Google Scholar]
- 23.Forman AB, Prendiville JS, Esterly NB, et al. Kindler syndrome: Report of two cases and review of the literature. Pediatr Dermatol. 1989;6:91–101. doi: 10.1111/j.1525-1470.1989.tb01004.x. [DOI] [PubMed] [Google Scholar]
- 24.Ashton GHS. Kindler syndrome. Clin Exp Dermatol. 2004;29:116–121. doi: 10.1111/j.1365-2230.2004.01465.x. [DOI] [PubMed] [Google Scholar]
- 25.Shimizu H, Sato M, Ban M, et al. Immunohistochemical, ultrastructural, and molecular features of Kindler syndrome distinguish it from dystrophic epidermolysis bullosa. Arch Dermatol. 1997;133:1111–1117. [PubMed] [Google Scholar]
- 26.Weary PE, Manley WF, Graham GF. Heriditary acrokeratotic poikiloderma. Arch Dermatol. 1971;103:409–422. [PubMed] [Google Scholar]
- 27.Larjava H, Uitto VJ, Eerola E, Haapasalo M. Inhibition of gingival fibroblast growth by Bacteroides gingivalis. Infect Immun. 1987;55:201–205. doi: 10.1128/iai.55.1.201-205.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Redlich M, Reichenberg E, Harari D, Zaks B, Shoshan S, Palmon A. The effect of mechanical force on mRNA levels of collagenase, collagen type I, and tissue inhibitors of metalloproteinases in gingivae of dogs. J Dent Res. 2001;80:2080–2084. doi: 10.1177/00220345010800121101. [DOI] [PubMed] [Google Scholar]
- 29.Salonen JI, Kautsky MB, Dale BA. Changes in cell phenotype during regeneration of junctional epithelium of human gingiva in vitro. J Periodontal Res. 1989;24:370–377. doi: 10.1111/j.1600-0765.1989.tb00885.x. [DOI] [PubMed] [Google Scholar]