Abstract
Inositol 1,3,4-trisphosphate 5/6-kinase (ITPK1) is a key regulatory enzyme at the branch point for the synthesis of inositol hexakisphosphate (IP6), an intracellular signaling molecule implicated in the regulation of ion channels, endocytosis, exocytosis, transcription, DNA repair, and RNA export from the nucleus. IP6 also has been shown to be an integral structural component of several proteins. We have generated a mouse strain harboring a β-galactosidase (βgal) gene trap cassette in the second intron of the Itpk1 gene. Animals homozygous for this gene trap are viable, fertile, and produce less ITPK1 protein than wild-type and heterozygous animals. Thus, the gene trap represents a hypomorphic rather than a null allele. Using a combination of immunohistochemistry, in situ hybridization, and βgal staining of mice heterozygous for the hypomorphic allele, we found high expression of Itpk1 in the developing central and peripheral nervous systems and in the paraxial mesoderm. Examination of embryos resulting from homozygous matings uncovered neural tube defects (NTDs) in some animals and axial skeletal defects or growth retardation in others. On a C57BL/6 × 129(P2)Ola background, 12% of mid-gestation embryos had spina bifida and/or exencephaly, whereas wild-type animals of the same genetic background had no NTDs. We conclude that ITPK1 is required for proper development of the neural tube and axial mesoderm.
Keywords: exencephaly, hypomorphic allele, inositol signaling, spina bifida
Inositol polyphosphates are water-soluble signaling molecules derived by phosphorylation of combinations of the 6 hydroxyl groups on the inositol ring. These compounds regulate various cellular processes. The most abundant inositol polyphosphate, inositol hexakisphosphate (IP6), plays an essential role in mRNA export (1–4), DNA repair (5, 6), transcriptional regulation (7), protein kinase regulation (8–10), endocytosis (11), adenosine deaminase function (12), and nonvisual arrestin oligomerization (13).
Inositol 1,3,4-trisphosphate 5/6 kinase (ITPK1) is a pivotal enzyme in the IP6 production pathway. In mammalian cells, ITPK1 uses inositol 1,3,4-trisphosphate [I(1,3,4)P3] as a substrate to generate inositol 1,3,4,5-tetrakisphosphate (IP4) and inositol 1,3,4,6-P4 (14), which is further phosphorylated to inositol 1,3,4,5,6-pentakisphosphate (IP5) by a 5-kinase (15, 16) and then to IP6 by a 2-kinase (17, 18). ITPK1 also can phosphorylate inositol 3,4,5,6-P4 [I(3,4,5,6)P4] at the 1-position (19, 20), as well as function as a phosphatase and as an isomerase (21–23). Overexpression of ITPK1 leads to increased levels of IP4 isomers, IP5, and IP6, and depletion of ITPK1 by RNAi results in decreased levels of IP4 isomers, IP5, and IP6 (18). Underscoring the importance of the highly phosphorylated inositols in cells, null mutations in either of the other 2 genes in mice encoding kinases leading to the synthesis of IP6 results in embryonic lethality (24, 25).
Although biochemical studies suggest that ITPK1 plays a key role in intracellular inositol phosphate metabolism, little is known about the developmental expression of this kinase. Northern blot analysis has shown that human ITPK1 is widely expressed, with the highest mRNA levels in the brain (14). To characterize the developmental expression profile and in vivo function of ITPK1, we generated mice harboring a β-galactosidase (βgal) gene trap cassette in the Itpk1 gene. Examination of embryos homozygous for the gene trap allele showed the presence of neural tube defects (NTDs) in some of these embryos. NTDs are common birth defects in humans (26). Several studies have shown that women with low serum folate levels are at an increased risk of giving birth to a child with an NTD (27–29). The pivotal role of maternal folate level in human NTDs was conclusively demonstrated in the 1991 Medical Research Council Vitamin Study investigating NTD prevention, which reported a 70% protective effect from periconceptual folic acid supplementation (30). That leaves 30% of cases of NTD considered folate-resistant.
In some animal models for folate-resistant NTDs, inositol supplementation or depletion has been shown to affect the prevalence of NTDs (31–35). In this report, we show that mice with reduced levels of ITPK1 develop NTDs. We propose that disruption of inositol signaling might be responsible for the NTDs seen in these animals.
Results
Characterization of a Transgenic Mouse Harboring a βgal Reporter in the Itpk1 Gene.
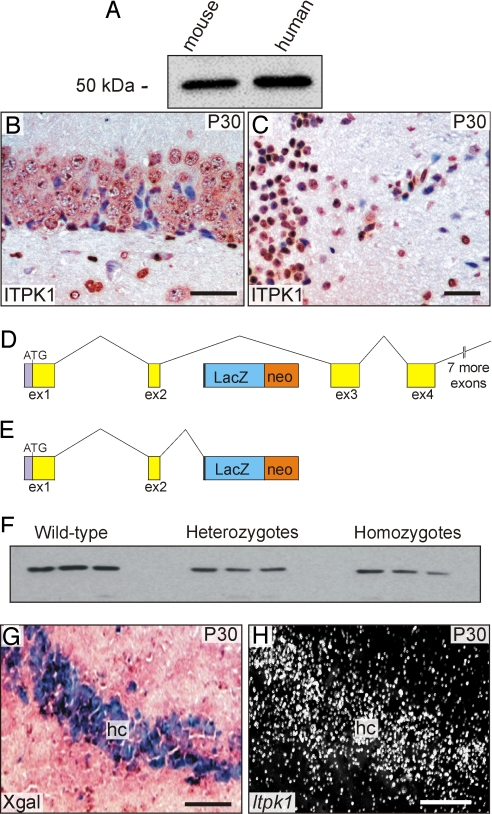
To gain insight into the in vivo function of ITPK1, we generated mice harboring a βgal gene trap cassette in the second intron of the Itpk1 gene. To assess the expression of ITPK1 in these mice, we tested whether an antibody produced against human ITPK1 would cross-react with the mouse protein. Western blot analysis revealed a 50-kDa protein in soluble extracts from mouse and human brain (Fig. 1A). Using this antibody, tissue sections from adult mouse brain were stained for ITPK1 immunoreactivity. Hippocampal neurons (Fig. 1B), brainstem neurons (Fig. 1C), periventricular neurons [supporting information (SI) Fig. S1A], and choroid plexus cells (Fig. S1B) all stained positive for ITPK1.
Fig. 1.
Characterization of the ITPK1 antibody and ITPK1 protein levels in mice homozygous for the Itpk1–βgal gene trap allele. (A) Western blot analysis of soluble extracts from cadaveric human and mouse brain (6 μg protein/lane). (B and C) Immunoperoxidase staining of wild-type mouse brain showing ITPK1 immunoreactivity in hippocampal (B) and brainstem (C) neurons. (Scale bar = 30 μm.) (D and E) Diagram of the gene trap allele. Alternative mRNA splicing of the gene trap allele produces both a full-length wild-type transcript (D) and Itpk1–βgal fusion transcript (E). (F) Western blot analysis of ITPK1 protein in soluble brain extracts (2.5 μg protein/lane) from wild-type mice and mice either heterozygous or homozygous for the gene trap allele. (G and H) Correlation of Xgal staining and Itpk1 mRNA expression in a mouse heterozygous for the Itpk1–βgal allele. Adjacent cryosections of hippocampus (hc) were subjected to Xgal staining (G) or in situ hybridization for Itpk1 mRNA (H; dark-field microscopy). (Scale bar = 50 μm.)
RT-PCR analysis showed that alternative mRNA splicing of the gene trap allele produced both a full-length wild-type transcript (Fig. 1D) and an Itpk1–βgal fusion transcript (Fig. 1E) (data not shown). Thus, the gene trap results in a hypomorphic rather than a null allele. Western blot analysis of mouse brain extracts probed with the antibody against human ITPK1 was performed using soluble protein extracts from 3 littermates each of wild-type, heterozygote, and homozygote animals. Consistent with the production of full-length enzyme in animals genotypically homozygous for the gene trap allele, all animals produced some ITPK1, but levels varied widely, ranging from 42% to 67% for heterozygotes and 24% to 67% for homozygotes (Fig. 1F).
Itpk1 Expression During Embryonic and Postnatal Development.
To determine the developmental expression pattern of Itpk1, we performed in situ hybridization or Xgal staining on mice heterozygous for the gene trap allele. Adjacent sections of mouse tissue from the hippocampus (Fig. 1 G and H), brown adipose tissue Fig. S2 A and B), and cervical ganglia (Fig. S2 C and D) were subjected to Xgal staining or in situ hybridization; these 2 methods yielded identical results.
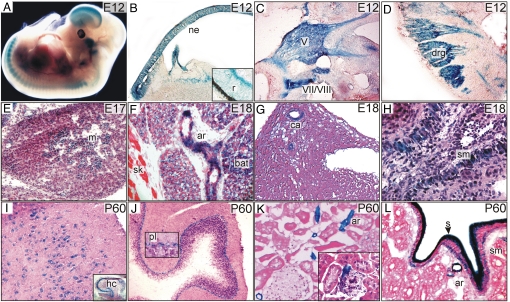
In situ hybridization of sections of embryonic day (E) 7.5 mice showed that Itpk1 mRNA is expressed in decidua but not in the embryo proper (Fig. S3A). Whole-mount Xgal staining of E8.5 mice heterozygous for the gene trap allele revealed expression in paraxial mesoderm but not in the cranial neural folds or anterior intestinal portal (Fig. S3B). By E10.5, Xgal staining was observed in the neural tube as well as in neural crest derivatives, such as the trigeminal ganglion and dorsal root ganglia (Fig. S3 C and D). Whole-mount Xgal staining of an E12.5 mouse showed staining throughout the neural epithelium and neural crest derivatives (Fig. 2A). Analysis of histological sections of Xgal-stained embryos revealed strong Itpk1 expression in neural tissues, including the neural epithelium (Fig. 2 A and B), as well as in the retinal neurons (Fig. 2B, inset), cranial nerve ganglia (Fig. 2C), dorsal root ganglia (Fig. 2D), adrenal medulla (Fig. 2E), and neurons of the lateral temporal lobe, hippocampus, and cerebellum (Fig. 2I and J). Itpk1 expression also was seen in brown adipose tissue, vascular smooth muscle cells (Fig. 2 F, G, and K), and visceral smooth muscle cells in the lung (Fig. 2F) and gut (Fig. 2L). No staining was observed in skeletal muscle (Fig. 2F and Fig. S2 A and B), consistent with enzymatic assays of ITPK1 in bovine tissue extracts demonstrating the lowest activity level in skeletal muscle (Fig. S4).
Fig. 2.
Expression of Itpk1 in the developing mouse. (A) Whole-mount Xgal staining of a mouse embryo heterozygous for the Itpk1–βgal gene trap allele. (B–L) Tissue sections from mice heterozygous for Itpk1–βgal gene trap allele were subjected to Xgal staining. Expression of the transgene was evident in rostal neuroepithelium (ne) and retinal neurons (r) (B, inset); ganglia of cranial nerves V, VII, and VIII (C); dorsal root ganglia (drg) of the lower trunk (D); adrenal medulla (m) (E); brown adipose tissue (bat) and vascular smooth muscle cells in an adjacent artery (ar) but not in skeletal muscle (sk) (F); coronary arteries (ca) (G); airway smooth muscle (sm) (H); neurons of the lateral temporal lobe and hippocampus (hc, inset) (I); neurons in the cerebellum, particularly in the Purkinje cell layer (pl; inset) (J); vascular smooth muscle cells in arterioles (ar) of the kidney (inset shows a juxtaglomerulary arteriole) (K); and serosal cells (s), visceral smooth muscle cells (sm), and arteriolar smooth muscle cells (ar) of the duodenum (L).
NTDs and Skeletal Abnormalities in Itpk1 Hypomorphic Embryos.
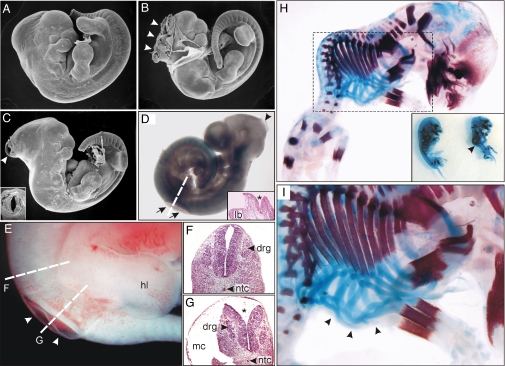
Examination of embryos homozygous for the Itpk1 gene trap allele revealed NTDs in some of the embryos. Scanning electron micrographs of 3 E11.5 homozygous littermates are shown in Fig. 3 A–C. The embryo in Fig. 3A appears normal, whereas the embryos in Fig. 3 B and C exhibit varying degrees of exencephaly. Fig. 3D shows an E10.5 mouse exhibiting both exencephaly and spina bifida. Fig. 3 E–G shows an E11.5 homozygote with a nascent myelomeningocele. The spinal column is often deformed in both human and animal models of NTDs (36). Examination of the skeletons of late gestation Itpk1 hypomorphic mice stained with alizarin red and alcian blue revealed several abnormalities. Fig. 3 H and I shows a skeleton preparation from an E14.5 mouse with severe kyphoscoliosis, a small thorax, and malformed ribs. The inset in Fig. 3H shows this embryo next to a littermate, with an arrow indicating the abnormal spinal curvature; Fig 3I shows a close-up view of the disorganized rib structure.
Fig. 3.
NTDs and axial skeleton malformations in mouse embryos homozygous for the Itpk1–βgal gene trap allele. (A–C) Scanning electron micrographs of a normal-appearing E11.5 mouse (A) and 2 littermates (B and C) with varying degrees of exencephaly (arrowheads). The inset in panel C shows another view of the open neural tube. (D) Xgal-stained E10.5 mouse with both exencephaly (arrowhead) and spina bifida (arrows). The dashed line indicates the orientation of the tissue section shown in the inset. The asterisk highlights the open neural tube. (E) Nascent myelomeningocele (arrowheads) in an E11.5 mouse. (F and G) Dashed lines indicate the orientation of tissue sections through the intact neural tube (F) and open neural tube (G, asterisk). (H and I) E14.5 mouse skeletons stained with alcian blue and alizarin red to visualize cartilage and bone. The inset in panel H shows 2 littermates, one of which has kyphoscoliosis (arrowhead). Higher-magnification views of this embryo are shown in panels H and I. Note the presence of malformed ribs (arrowheads in I). Abbreviations: drg, dorsal root ganglia; hl, hind limb; lb, limb bud; mc, myelocele; ntc, notochord.
The prevalence of NTDs in Itpk1 hypomorphic mice on a mixed C57BL/6 × 129(P2)Ola genetic background is summarized in Table 1. Embryos age E9.5–E12.5 were screened for defects. NTDs were seen at all embryonic days examined and were most pronounced between E9.5 and E11.5. By E12.5, the number of embryos exhibiting NTDs dropped dramatically, consistent with either delayed neural tube closure or embryo resorption. At the earliest embryonic stage examined (E9.5), 6% of the embryos were severely growth-retarded, defined as being <50% the size of their littermates (Fig. S5). But by E12.5, no growth retardation was observed, implying that these embryos are likely resorbed. The overall incidence of birth defects in embryos age E9.5–E12.5 was 44/188 (23%). Examination of wild-type embryos age E9.5–E13.5 from the same mixed genetic background as the Itpk1 hypomorphic mice revealed no NTDs (n = 92; P < .001).
Table 1.
Summary of birth defects in Itpk1 hypomorphic embryos
| E9.5 | E10.5 | E11.5 | E12.5 | |
|---|---|---|---|---|
| Total number of embryos | 36 | 27 | 69 | 56 |
| % with NTD | 17 | 11 | 17 | 4 |
| % resorbed | 0 | 11 | 12 | 11 |
| % growth retarded | 6 | 4 | 1 | 0 |
| % total birth defects | 22 | 26 | 30 | 14 |
Discussion
The inositol metabolic pathway comprises numerous lipid and soluble inositol intermediates containing phosphate moieties on 1 or more of the 6 hydroxyl groups, as well as pyrophosphate bonds (37–39). This dynamic pathway is controlled by the activities of kinases and phosphatases that interconvert these intermediates. One of these kinases, ITPK1, is a multifunctional enzyme that catalyzes the rate-limiting step in the formation of higher phosphorylated forms of inositol in mammalian cells (18).
ITPK1 is expressed throughout the adult nervous system and in the neural epithelium and neural crest derivatives of the developing embryo. Mice homozygous for the gene trap allele all produce some ITPK1 and thus are hypomorphs. Examination of homozygous embryos revealed a subset with NTDs. Between E9.5 and E11.5, 12% of embryos exhibited exencephaly and/or spina bifida. By E12.5, the number of NTDs declined, indicating either that the affected embryos are being resorbed or that the NTDs represent delayed neural tube closure.
Although there are mouse models of NTDs that include mice born with an open neural tube, Pax1/E2α double-mutant mice (40), and mice with an inactive MEKK4 kinase gene (41), the more common event is delayed neural tube closure. The NTDs seen in the Itpk1 hypomorphic mice are consistent with the latter, in which delayed neural tube closure results in modest postnatal defects. These include skeletal defects in curly tail mice (42), grainyhead—like-3 null (Grh3−/−) mice (43), crooked tail mice (44), and TRAF-4–deficient mice (45). Skeletal defects have been observed in some Itpk1 hypomorphic embryos. Embryos presenting with exencephaly represent a small percentage of the NTDs in Itpk1 hypomorphic mice, as is the case for this defect in curly tail mice and in Grh3−/− mice (43, 46), and likely are resorbed before birth.
A link between inositol metabolism and NTDs has been established in several animal models. Rat embryos cultured in inositol-free medium tend to be malformed (32). NTDs are seen in 20% of the offspring of female rats with diabetes, and oral inositol supplementation during pregnancy reduces this number by half (33). The mouse mutants curly tail, Axd, and EphA7 are all susceptible to folate-resistant NTDs (31). Culture of curly tail embryos in inositol-deficient medium increases the incidence of NTDs (34). Intraperitoneal injection of inositol to pregnant curly tail mice reduces the incidence of NTDs by 70% (35). Mice with a null allele for the inositol lipid kinase type I phosphatidylinositol-4-phosphate 5-kinase γ exhibit exencephaly, apparently due to abnormal adhesion junction formation or neuronal cell migration (47).
Only limited data are available for establishing a correlation between inositol and NTDs in humans. One study found that mothers with low serum inositol have a 2.6-fold increased risk of having offspring with spina bifida, and that children with spina bifida have 7% lower serum inositol levels than control children (48). In 2 case reports, women at high risk for having a child with an NTD (defined as having 2 previous pregnancies complicated by an NTD despite folic acid treatment) were given inositol during pregnancy. The first case report described a couple whose third, healthy child was born after inositol and folic acid supplementation (49). The second report described 3 sets of parents at high risk for having a child with an NTD who gave birth to a total of 5 healthy children after inositol and folic acid supplementation (50).
That a perturbation in inositol metabolism is responsible for the NTDs seen in the Itpk1 hypomorphic mice remains to be shown. We speculate that the phenotype is due to impaired production of inositol polyphosphates, which have been shown to be involved in multiple processes required for neural tube closure. The variable penetrance of NTDs in the Itpk1 hypomorphs could result from the highly variable expression of this enzyme in these animals. Mice producing sufficient amounts of ITPK1 may have offspring with no defects, whereas those producing inadequate amounts of ITPK1 to maintain adequate levels of highly phosphorylated inositols may be susceptible to NTDs. Experiments are currently underway to artificially reduce as well as increase the inositol levels in these mice in an effort to exacerbate or reduce the number of NTDs, respectively. In these experiments, the level of each inositol polyphosphate is being measured in hypomorphic embryos metabolically labeled with inositol.
Materials and Methods
Reagents and Chemicals.
Unless noted otherwise, all reagents and chemicals were purchased from Sigma Aldrich.
Generation of Mice Harboring an Itpk1 Gene Trap Allele.
Male chimeric mice generated from the ES cell line DTM066 was obtained from BayGenomics (http://www.genetrap.org/). These animals were inbred until a population was obtained in which the mice were homozygous for the gene trap allele. Detailed genotyping information is provided in SI Materials and Methods.
Embryo Collection.
All animal experiments were approved by Washington University's Animal Studies Committee. See SI Materials and Methods for details on embryo and tissue collection.
Preparation of ITPK1 Antibody and Western Blot Analysis.
Standard methods were used to produce a polyclonal antibody against human ITPK1 (see SI Materials and Methods). Total lysate from brain tissue was assessed by freezing the tissue in liquid nitrogen, followed by homogenization in lysis buffer and Western blot analysis, as described in SI Materials and Methods.
Immunostaining, Xgal Staining, and In Situ Hybridization.
Tissue sections were processed for immunoperoxidase staining (51) and for Xgal staining as described previously (52). Tissue cryosections (10 μm) were subjected to in situ hybridization as described previously (53). Details on the immunostaining, Xgal staining, and in situ hybridization procedures are provided in SI Materials and Methods.
Skeleton Staining.
Skeletal staining of embryos was done using the method of Nomi et al. (54) with minor modifications, as described in SI Materials and Methods.
Scanning Electron Microscopy.
Embryos were prepared and examined using standard techniques, as outlined in SI Materials and Methods (55).
Supplementary Material
Acknowledgments.
We thank Dr. Mike Veith for assisting with the scanning electron microscopy. This work was supported by grants from the Children's Discovery Institute (MD-II-2009–174, to M.P.W.) and the National Institutes of Health (HL-16634–43, to P.W.M.; DK07618, to D.B.W.; and DK52574, to the Histology Core Facility).
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/cgi/content/full/0904172106/DCSupplemental.
References
- 1.York JD, Odom AR, Murphy R, Ives EB, Wente SR. A phospholipase C–dependent inositol polyphosphate kinase pathway required for efficient messenger RNA export. Science. 1999;285:96–100. doi: 10.1126/science.285.5424.96. [DOI] [PubMed] [Google Scholar]
- 2.Feng Y, Wente SR, Majerus PW. Overexpression of the inositol phosphatase SopB in human 293 cells stimulates cellular chloride influx and inhibits nuclear mRNA export. Proc Natl Acad Sci USA. 2001;98:875–879. doi: 10.1073/pnas.021558098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Weirich CS, et al. Activation of the DExD/H-box protein Dbp5 by the nuclear-pore protein Gle1 and its coactivator InsP6 is required for mRNA export. Nat Cell Biol. 2006;8:668–676. doi: 10.1038/ncb1424. [DOI] [PubMed] [Google Scholar]
- 4.Alcazar-Roman AR, Tran EJ, Guo S, Wente SR. Inositol hexakisphosphate and Gle1 activate the DEAD-box protein Dbp5 for nuclear mRNA export. Nat Cell Biol. 2006;8:711–716. doi: 10.1038/ncb1427. [DOI] [PubMed] [Google Scholar]
- 5.Hanakahi LA, Bartlet-Jones M, Chappell C, Pappin D, West SC. Binding of inositol phosphate to DNA-PK and stimulation of double-strand break repair. Cell. 2000;102:721–729. doi: 10.1016/s0092-8674(00)00061-1. [DOI] [PubMed] [Google Scholar]
- 6.Hanakahi LA, West SC. Specific interaction of IP6 with human Ku70/80, the DNA-binding subunit of DNA-PK. EMBO J. 2002;21:2038–2044. doi: 10.1093/emboj/21.8.2038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Odom AR, Stahlberg A, Wente SR, York JD. A role for nuclear inositol 1,4,5-trisphosphate kinase in transcriptional control. Science. 2000;287:2026–2029. doi: 10.1126/science.287.5460.2026. [DOI] [PubMed] [Google Scholar]
- 8.Larsson O, et al. Inhibition of phosphatases and increased Ca2+ channel activity by inositol hexakisphosphate. Science. 1997;278:471–474. doi: 10.1126/science.278.5337.471. [DOI] [PubMed] [Google Scholar]
- 9.Hilton JM, et al. Phosphorylation of a synaptic vesicle-associated protein by an inositol hexakisphosphate–regulated protein kinase. J Biol Chem. 2001;276:16341–16347. doi: 10.1074/jbc.M011122200. [DOI] [PubMed] [Google Scholar]
- 10.Solyakov L, et al. Regulation of casein kinase-2 (CK2) activity by inositol phosphates. J Biol Chem. 2004;279:43403–43410. doi: 10.1074/jbc.M403239200. [DOI] [PubMed] [Google Scholar]
- 11.Hoy M, et al. Inositol hexakisphosphate promotes dynamin I–mediated endocytosis. Proc Natl Acad Sci USA. 2002;99:6773–6777. doi: 10.1073/pnas.102157499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Macbeth MR, et al. Inositol hexakisphosphate is bound in the ADAR2 core and required for RNA editing. Science. 2005;309:1534–1539. doi: 10.1126/science.1113150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Milano SK, Kim YM, Stefano FP, Benovic JL, Brenner C. Nonvisual arrestin oligomerization and cellular localization are regulated by inositol hexakisphosphate binding. J Biol Chem. 2006;281:9812–9823. doi: 10.1074/jbc.M512703200. [DOI] [PubMed] [Google Scholar]
- 14.Wilson MP, Majerus PW. Isolation of inositol 1,3,4-trisphosphate 5/6-kinase, cDNA cloning and expression of the recombinant enzyme. J Biol Chem. 1996;271:11904–11910. doi: 10.1074/jbc.271.20.11904. [DOI] [PubMed] [Google Scholar]
- 15.Fujii M, York JD. A role for rat inositol polyphosphate kinases rIPK2 and rIPK1 in inositol pentakisphosphate and inositol hexakisphosphate production in rat-1 cells. J Biol Chem. 2005;280:1156–1164. doi: 10.1074/jbc.M412006200. [DOI] [PubMed] [Google Scholar]
- 16.Chang SC, Miller AL, Feng Y, Wente SR, Majerus PW. The human homolog of the rat inositol phosphate multikinase is an inositol 1,3,4,6-tetrakisphosphate 5-kinase. J Biol Chem. 2002;277:43836–43843. doi: 10.1074/jbc.M206134200. [DOI] [PubMed] [Google Scholar]
- 17.Verbsky JW, Wilson MP, Kisseleva MV, Majerus PW, Wente SR. The synthesis of inositol hexakisphosphate: Characterization of human inositol 1,3,4,5,6-pentakisphosphate 2-kinase. J Biol Chem. 2002;277:31857–31862. doi: 10.1074/jbc.M205682200. [DOI] [PubMed] [Google Scholar]
- 18.Verbsky JW, Chang SC, Wilson MP, Mochizuki Y, Majerus PW. The pathway for the production of inositol hexakisphosphate in human cells. J Biol Chem. 2005;280:1911–1920. doi: 10.1074/jbc.M411528200. [DOI] [PubMed] [Google Scholar]
- 19.Tan Z, Bruzik KS, Shears SB. Properties of the inositol 3,4,5,6-tetrakisphosphate 1-kinase purified from rat liver: Regulation of enzyme activity by inositol 1,3,4-trisphosphate. J Biol Chem. 1997;272:2285–2290. doi: 10.1074/jbc.272.4.2285. [DOI] [PubMed] [Google Scholar]
- 20.Yang X, et al. Inositol 1,3,4-trisphosphate acts in vivo as a specific regulator of cellular signaling by inositol 3,4,5,6-tetrakisphosphate. J Biol Chem. 1999;274:18973–18980. doi: 10.1074/jbc.274.27.18973. [DOI] [PubMed] [Google Scholar]
- 21.Yang X, Shears SB. Multitasking in signal transduction by a promiscuous human Ins(3,4,5,6)P(4) 1-kinase/Ins(1,3,4)P(3) 5/6-kinase. Biochem J. 2000;351(Part 3):551–555. [PMC free article] [PubMed] [Google Scholar]
- 22.Ho MW, et al. Regulation of Ins(3,4,5,6)P(4) signaling by a reversible kinase/phosphatase. Curr Biol. 2002;12:477–482. doi: 10.1016/s0960-9822(02)00713-3. [DOI] [PubMed] [Google Scholar]
- 23.Miller GJ, Wilson MP, Majerus PW, Hurley JH. Specificity determinants in inositol polyphosphate synthesis: Crystal structure of inositol 1,3,4-trisphosphate 5/6-kinase. Mol Cell. 2005;18:201–212. doi: 10.1016/j.molcel.2005.03.016. [DOI] [PubMed] [Google Scholar]
- 24.Frederick JP, et al. An essential role for an inositol polyphosphate multikinase, Ipk2, in mouse embryogenesis and second messenger production. Proc Natl Acad Sci USA. 2005;102:8454–8459. doi: 10.1073/pnas.0503706102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Verbsky J, Lavine K, Majerus PW. Disruption of the mouse inositol 1,3,4,5,6-pentakisphosphate 2-kinase gene, associated lethality, and tissue distribution of 2-kinase expression. Proc Natl Acad Sci USA. 2005;102:8448–8453. doi: 10.1073/pnas.0503656102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sells CJ. Overview: Neural tube defects. Mental Retard Dev Disabil Res Rev. 1998;4:239–240. [Google Scholar]
- 27.Kirke PN, et al. Maternal plasma folate and vitamin B12 are independent risk factors for neural tube defects. Q J Med. 1993;86:703–708. [PubMed] [Google Scholar]
- 28.Steegers-Theunissen RP, et al. Maternal hyperhomocysteinemia: A risk factor for neural tube defects? Metabolism. 1994;43:1475–1480. doi: 10.1016/0026-0495(94)90004-3. [DOI] [PubMed] [Google Scholar]
- 29.Mills JL, et al. Homocysteine metabolism in pregnancies complicated by neural tube defects. Lancet. 1995;345:149–151. doi: 10.1016/s0140-6736(95)90165-5. [DOI] [PubMed] [Google Scholar]
- 30.MRC Vitamin Study Research Group. Prevention of neural tube defects: Results of the Medical Research Council Vitamin Study. Lancet. 1991;338:131–137. [PubMed] [Google Scholar]
- 31.Copp AJ, Greene ND, Murdoch JN. The genetic basis of mammalian neurulation. Nat Rev Genet. 2003;4:784–793. doi: 10.1038/nrg1181. [DOI] [PubMed] [Google Scholar]
- 32.Cockroft DL. Changes with gestational age in the nutritional requirements of postimplantation rat embryos in culture. Teratology. 1988;38:281–290. doi: 10.1002/tera.1420380312. [DOI] [PubMed] [Google Scholar]
- 33.Reece EA, Khandelwal M, Wu YK, Borenstein M. Dietary intake of myo-inositol and neural tube defects in offspring of diabetic rats. Am J Obstet Gynecol. 1997;176:536–539. doi: 10.1016/s0002-9378(97)70543-x. [DOI] [PubMed] [Google Scholar]
- 34.Cockroft DL, Brook FA, Copp AJ. Inositol deficiency increases the susceptibility to neural tube defects of genetically predisposed (curly tail) mouse embryos in vitro. Teratology. 1992;45:223–232. doi: 10.1002/tera.1420450216. [DOI] [PubMed] [Google Scholar]
- 35.Greene ND, Copp AJ. Inositol prevents folate-resistant neural tube defects in the mouse. Nat Med. 1997;3:60–66. doi: 10.1038/nm0197-60. [DOI] [PubMed] [Google Scholar]
- 36.Tyl RW, Chernoff N, Rogers JM. Altered axial skeletal development. Birth Defects Res B Dev Reprod Toxicol. 2007;80:451–472. doi: 10.1002/bdrb.20134. [DOI] [PubMed] [Google Scholar]
- 37.Berridge MJ, Irvine RF. Inositol phosphates and cell signalling. Nature. 1989;341:197–205. doi: 10.1038/341197a0. [DOI] [PubMed] [Google Scholar]
- 38.Shears SB. How versatile are inositol phosphate kinases? Biochem J. 2004;377:265–280. doi: 10.1042/BJ20031428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Michell RH. Inositol derivatives: Evolution and functions. Nat Rev Mol Cell Biol. 2008;9:151–161. doi: 10.1038/nrm2334. [DOI] [PubMed] [Google Scholar]
- 40.Joosten PH, van Zoelen EJ, Murre C. Pax1/E2a double-mutant mice develop non-lethal neural tube defects that resemble human malformations. Transgenic Res. 2005;14:983–987. doi: 10.1007/s11248-005-2540-9. [DOI] [PubMed] [Google Scholar]
- 41.Abell AN, et al. Ablation of MEKK4 kinase activity causes neurulation and skeletal patterning defects in the mouse embryo. Mol Cell Biol. 2005;25:8948–8959. doi: 10.1128/MCB.25.20.8948-8959.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Copp AJ, Brook FA, Roberts HJ. A cell type–specific abnormality of cell proliferation in mutant (curly tail) mouse embryos developing spinal neural tube defects. Development. 1988;104:285–295. doi: 10.1242/dev.104.2.285. [DOI] [PubMed] [Google Scholar]
- 43.Ting SB, et al. Inositol- and folate-resistant neural tube defects in mice lacking the epithelial-specific factor Grhl-3. Nat Med. 2003;9:1513–1519. doi: 10.1038/nm961. [DOI] [PubMed] [Google Scholar]
- 44.Carter M, et al. Crooked tail (Cd) model of human folate–responsive neural tube defects is mutated in Wnt coreceptor lipoprotein receptor–related protein 6. Proc Natl Acad Sci USA. 2005;102:12843–12848. doi: 10.1073/pnas.0501963102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Regnier CH, et al. Impaired neural tube closure, axial skeleton malformations, and tracheal ring disruption in TRAF4-deficient mice. Proc Natl Acad Sci USA. 2002;99:5585–5590. doi: 10.1073/pnas.052124799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Embury S, Seller MJ, Adinolfi M, Polani PE. Neural tube defects in curly-tail mice, I: Incidence, expression and similarity to the human condition. Proc R Soc Lond B Biol Sci. 1979;206:85–94. doi: 10.1098/rspb.1979.0092. [DOI] [PubMed] [Google Scholar]
- 47.Wang Y, Lian L, Golden JA, Morrisey EE, Abrams CS. PIP5KI gamma is required for cardiovascular and neuronal development. Proc Natl Acad Sci USA. 2007;104:11748–11753. doi: 10.1073/pnas.0700019104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Groenen PM, et al. Maternal myo-inositol, glucose, and zinc status is associated with the risk of offspring with spina bifida. Am J Obstet Gynecol. 2003;189:1713–1719. doi: 10.1016/s0002-9378(03)00807-x. [DOI] [PubMed] [Google Scholar]
- 49.Cavalli P, Copp AJ. Inositol and folate resistant neural tube defects. J Med Genet. 2002;39:e5. doi: 10.1136/jmg.39.2.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cavalli P, Tedoldi S, Riboli B. Inositol supplementation in pregnancies at risk of apparently folate-resistant NTDs. Birth Defects Res A Clin Mol Teratol. 2008;82:540–542. doi: 10.1002/bdra.20454. [DOI] [PubMed] [Google Scholar]
- 51.Bielinska M, Seehra A, Toppari J, Heikinheimo M, Wilson DB. GATA-4 is required for sex steroidogenic cell development in the fetal mouse. Dev Dyn. 2007;236:203–213. doi: 10.1002/dvdy.21004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Narita N, Bielinska M, Wilson DB. Cardiomyocyte differentiation by GATA-4–deficient embryonic stem cells. Development. 1997;124:3755–3764. doi: 10.1242/dev.124.19.3755. [DOI] [PubMed] [Google Scholar]
- 53.Bielinska M, et al. Mouse strain susceptibility to gonadectomy-induced adrenocortical tumor formation correlates with the expression of GATA-4 and luteinizing hormone receptor. Endocrinology. 2003;144:4123–4133. doi: 10.1210/en.2003-0126. [DOI] [PubMed] [Google Scholar]
- 54.Nomi M, et al. Loss of mRor1 enhances the heart and skeletal abnormalities in mRor2-deficient mice: Redundant and pleiotropic functions of mRor1 and mRor2 receptor tyrosine kinases. Mol Cell Biol. 2001;21:8329–8335. doi: 10.1128/MCB.21.24.8329-8335.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bellomo D, Lander A, Harragan I, Brown NA. Cell proliferation in mammalian gastrulation: The ventral node and notochord are relatively quiescent. Dev Dyn. 1996;205:471–485. doi: 10.1002/(SICI)1097-0177(199604)205:4<471::AID-AJA10>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.