Abstract
In this paper we characterize the metabolic fingerprint and first reported anti-inflammatory activity of Hypericum gentianoides. H. gentianoides has a history of medical use by Native Americans, but it has been studied very little for biological activity. High-performance liquid chromatography (HPLC) and liquid chromatography–electrospray ionization–mass spectrometry (LC-ESI-MS) analyses of a methanol extract show that H. gentianoides contains a family of over nine related compounds that have retention times, mass spectra, and a distinctive UV absorption spectra characteristic of certain acyl-phloroglucinols. These metabolites are abundant relative to other secondary products present in H. gentianoides, accounting for approximately 0.2 g per gram of dry plant tissue. H. gentianoides methanol extracts and a specific semipreparative HPLC fraction from these extracts containing the putative acyl-phloroglucinols reduce prostaglandin E2 synthesis in mammalian macrophages.
Keywords: Hypericum gentianoides, Hypericum perforatum, St. John’s Wort, hyperforin, acyl-phloroglucinols, anti-inflammatory, cytotoxicity, PGE2, RAW264.7
INTRODUCTION
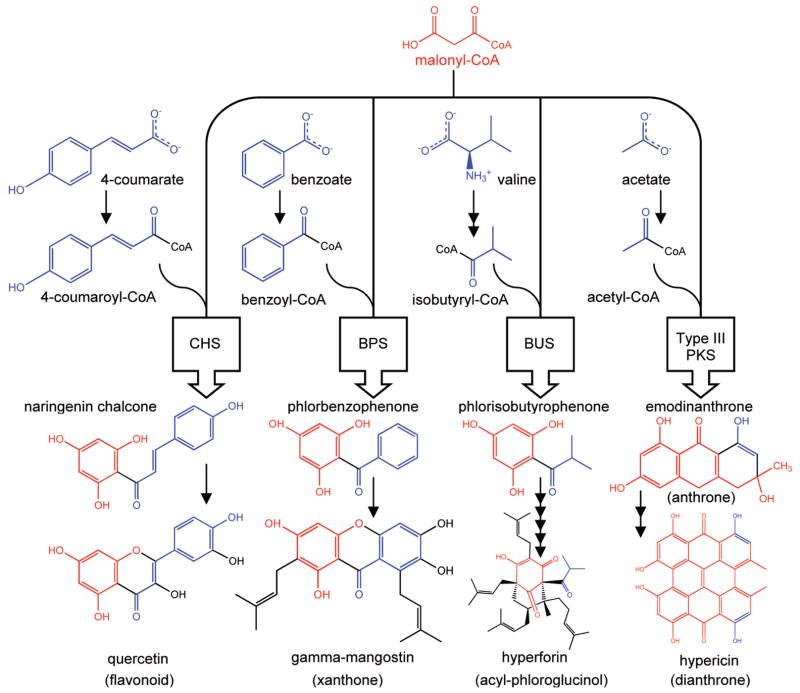
The Hypericum genus contains over 450 species; the 75 to 80 species that have been surveyed for phytochemicals to date are rich in secondary metabolites such as type III polyketides including flavonoids, xanthones, anthrones, dianthrones, and/or acyl-phloroglucinols (1). Type III polyketides result from the condensation of three malonyl-CoA molecules and various precursor substrates including or derived from branch chain amino acids, shikimate-derived phenylpropanoids, benzoic acid, and acetyl-CoA (2) (Figure 1). Divergent type III polyketide synthases (PKS) have evolved to create this wide variety of clade-specific polyketides. Chalcone synthase was the first type III PKS to be characterized, and is the best known (3). The flavonoids observed in Hypericum species, such as hyperoside, rutin, isoquercitrin, quercitrin, and quercetin, are derived from this enzyme’s product, naringenin chalcone. The type III PKS responsible for emodinanthrone biosynthesis is unknown; however, biosynthesis of the dianthrone hypericin from emo-dinanthrone (Figure 1) has been characterized (4). In light of this diversity, the Hypericum genus represents a great reserve for the discovery of new bioactive compounds and advancing our understanding of the underlying mechanisms that cause the reported bioactivities in humans.
Figure 1.
Hypericum species have evolved a wide array of specialized type III polyketide synthases (PKS) that produce polyketides that may interact with various receptors/mechanisms/components of potential pathogens, symbionts, predators, and pollinators. Type III polyketides result from the condensation of three malonyl-CoA molecules and various precursor substrates including branch chain amino acids, shikimate-derived phenylpropanoids, benzoic acid, and acetyl-CoA (20). Sometimes these polyketides are modified by other enzymes, for example, by prenylation, as with γ-mangostin (a xanthone) and hyperforin (an acyl-phloroglucinol). The boxes represent type III PKS’s responsible for specific reactions that form the precursors for secondary metabolites in Hypericum: chalcone synthase (CHS), benzophenone synthase (BPS), and isobutyrophenone synthase (BUS); and the type III PKS responsible for emodinanthrone biosynthesis (not isolated). The biosynthesis of the dianthrone hypericin from emodinanthrone has been characterized (21).
Several Hypericum species, in particular H. perforatum, are used as herbal treatments for anti-inflammatory, antibacterial, antiviral, and antidepressive applications (5–7). The numerous classes of compounds found in this single species may explain these multiple medicinal uses. For example, acyl-phloroglucinols have multiple bioactivities, including antibacterial activity in TLC bioautographic assays (6) and antidepressive properties in humans (5). Hypericum species are particularly rich in acyl-phloroglucinols (8). Acyl-phloroglucinols are derived from the condensation of multiple malonyl-CoA molecules and branch chain amino acid derivatives (9). These acyl-phloroglucinols are also often prenylated, as is the case with hyperforin (10). Acyl-phloroglucinols already identified from Hypericum species include hyperforin and adhyperforin, perforatumone, uliginosin A and B, bromouliginosin B, drummondins A–F, saroaspidins A–C, hyperbrasilols A–C, and numerous sarothralens and japonicines, paglucinol, annulatophenone, hypericophenonoside, hyperjovinol A, otogirin, italidipyrone, chinesins I and II, and sampsonines (6, 8).
Anti-inflammatory activity in mammals and mammalian cells has been reported for more than one species from the Hypericum genus (7, 11–13); however, it is unknown whether this is caused by the same compound(s) in each of these species. This anti-inflammatory activity has been confirmed by both in vivo studies treating mice with Hypericum extracts and measuring pain reduction using the formalin test (11) and in vitro molecular studies on H. patulum showing the down-regulation of NF-κB and the effect of this on PGE2 reduction in LPS-induced C6 rat glioma cells (14). Extracts or compounds from H. reflexum reduce inflammatory responses (11), H. laricifolium moderately reduce cyclooxygenase 1 and 2 activity (12), and H. perforatum reduce inducible nitrous oxide synthase activity in two human epithelial cell lines (13), indicating a direct effect of certain Hypericum metabolite extracts on reducing inflammation.
A few North American species of Hypericum have been used historically for their medical properties. One such species, H. gentianoides (orangegrass), is a small perennial that is native to much of the eastern United States. The Cherokee Native Americans used this plant for the treatment of fever, gastrointestinal disorders, nosebleeds, sores, and venereal disease (15). Despite its use by Native Americans, there has been very little biochemical research on this species. A survey study looking for the presence of several known compounds in 74 Hypericum species found that H. gentianoides did not contain hypericin or hyperforin (16), the two metabolites thought to be the major bioactive constituents of H. perforatum (17, 18). Because our initial survey of multiple Hypericum species indicated unusual metabolites are present in H. gentianoides, and because of the use of this species by Native Americans, we focus on the potential bioactivity of this species.
Here, we describe an efficient, nondestructive extraction method for Hypericum metabolites and a semipreparative separation method for fractionation of the H. gentianoides extracts. Using these protocols, we describe the unique metabolite profile of H. gentianoides and demonstrate the anti-inflammatory activity of this species.
MATERIALS AND METHODS
Extracts
Plant material was collected in October 2004 and September 2005 from two populations of H. gentianoides growing in their native habitats in South Carolina by Dr. Joe-Ann McCoy of the USDA-ARS North Central Regional Plant Introduction Station in Ames, IA. These populations were accessioned in the US National Plant Germplasm System as Ames 27657 and Ames 28015. Descriptive information about these accessions is posted in the Germplasm Resources Information Network (GRIN) database at http://www.ars-grin.gov/npgs/. Voucher specimens of these accessions have been deposited at the Iowa State University Ada Hayden Herbarium. The aboveground portion of mature flowering plants was harvested and allowed to dry naturally in a herbarium press before use.
To maximize the discovery of potentially bioactive metabolites, we developed extraction methods for Hypericum to optimize both the preservation of unstable compounds and the extraction efficiency of a broad range of compounds. Methods such as Soxhlet extraction are efficient; however, such methods can destroy thermally unstable compounds (19). We ground plant material in liquid nitrogen and used sonication to avoid altering the compounds during extraction to minimize the degradation of easily oxidized compounds potentially in H. gentianoides, such as prenylated acyl-phloroglucinols.
Dry plants were ground in liquid nitrogen and 30 g of ground tissue immersed in 100 mL of methanol. The methanol/tissue was sonicated, with the tube in ice, using a Cole-Parmer (Chicago, Ill. 60648) 4710 Series Ultrasonic Homogenizer for 30 s three times. Samples were then centrifuged at 15,000 rcf and the supernatant decanted and filtered through 0.45 μm nylon syringe filters. Solvent was evaporated under nitrogen gas at 40 °C to prevent oxidation. Four grams of dry plant material yielded approximately one gram of extracted methanol-soluble metabolites. The dried extract was dissolved in cell culture grade dimethyl sulfoxide (DMSO) (Sigma, St. Louis, MO).
Extraction with an ethanol–water mixture was avoided, despite efficient extraction, because high temperatures and/or long periods of time in a lyophilizer or evaporator are required to remove the water when the sample is concentrated. Methanol was a more logical choice since that solvent could be evaporated easily and quickly. Furthermore, it effectively extracted a broad range of compounds similar to those extracted with 70% ethanol as the solvent (Supporting Information).
High-Performance Liquid Chromatography (HPLC)
A Beckman Coulter HPLC with a Detector 160 photo diode array (PDA) detector was used for initial chemical profiling. A Synergi Max-RP 4 μm 150 × 4.6 mm column (Phenomenex, Torrance, CA) was used for analytical separation. For the mobile phase, an acetonitrile/methanol 9:1 v/v (solvent B) and 10 mM ammonium acetate (solvent A) gradient was used. The gradient consisted of 87% A/13% B in 10 min to 83% A/17% B, then to 100% B in 25 min and held for 5 min, at 40 °C. The flow rate was 1.0 mL/min (20). All solvents were HPLC grade (Sigma, St. Louis, MO). All standards mentioned where purchased from Chromadex, Irvine, CA.
Liquid Chromatography–Electrospray Ionization–Mass Spectrometry (LC-ESI-MS)
The extracts were also analyzed with an Agilent Technologies Ion Trap 1100 LC-ESI-MS. A Synergi Max-RP 4μm 150 × 4.6 mm column (Phenomenex, Torrance, CA) was used for analytical separation. For the mobile phase an acetonitrile/methanol 9:1 v/v (solvent B) and 10 mM ammonium acetate (solvent A) gradient was used. The gradient was increased from 85%A/15%B over a 10 min time period to 80% A/20% B, then to 100% B over a 25 min time period, and held at 100% B for 5 min, at 40 °C. The flow rate was 0.75 mL/min (20). This method and the HPLC method where chosen for their good separation and short run-time compared to other current methods used for the analysis of Hypericum extracts. All solvents were HPLC grade (Sigma, St. Louis, MO).
Semipreparative HPLC
Semipreparative scale HPLC was used for bioactivity-guided fractionation since it is very reproducible and requires only gram quantities of starting material. A Synergi Max-RP 4 μm 250 × 10 mm column (Phenomenex Torrance, CA) was chosen, because its stationary phase matches that of the analytical method. For the mobile phase, an acetonitrile/methanol 9:1 v/v (solvent B) and 10 mM ammonium acetate (solvent A) gradient elution was used: 13–15% B in 10 min, 15–100% B in 30 min, 100% B for 5 min, at 40 °C. The slow rise from 13% to 15% over 10 min optimized the separation of more polar phenylpropanoids from later eluting flavonoids. While a range of flow rates from 3–5 mL/min were tested, a flow rate of 4.6 mL/min was selected because it optimized separation of compounds while matching retention times with the analytical method within 3 min or less. All solvents were HPLC grade (Sigma, St. Louis, MO).
Cell Cultures and Treatments
RAW264.7 macrophages were cultured as described in Hammer et al. (18). For details see the Supporting Information. Cells were incubated in media containing 0.1% DMSO (Sigma, St. Louis, MO), or media with the inclusion of a H. gentianoides extract or fraction dissolved in DMSO for a final DMSO concentration of 0.1% of the media to match the controls, with or without 1 μg/mL LPS (E. coli 02B:B6) (Sigma, St. Louis, MO). Cell supernatants were collected on ice, and stored in a −70 °C freezer until used in the PGE2 assay (Supporting Information). Four controls were included for each experiment: media alone, media and DMSO, media and LPS, and media and LPS and DMSO. For the PGE2 assays, treatment of cells with 10 μM quercetin was used as a positive control. The H. gentianoides methanol extract, and fractions 1 and 2, were dried under nitrogen gas, weighed, and dissolved in DMSO. The concentration (tested in μg/mL (dry weight)) represents the final concentration of the extract, or fraction, in the media. For cytotoxicity studies (Supporting Information), treatment of cells with 20 μM hypericin was used as a positive control (17).
Statistical Analysis of Bioactivity and Cytotoxicity Data
Anti-inflammatory activity was calculated as the mean percent of control in LPS-induced PGE2 levels (±standard error) as compared to the media + LPS + DMSO control. Statistical significance was determined by an F-protected two-way ANOVA followed by a Tukey-Kramer test for multiple comparisons (21). For cytotoxicity, data were calculated as the mean percent cell viability (±standard error) as compared to the media + DMSO control. Statistical significance was determined by an F-protected two-way ANOVA followed by a Dunnett-Hsu test for multiple comparisons (22). P-values less than 0.05 were considered statistically significant.
RESULTS
Metabolic Profile of H. gentianoides
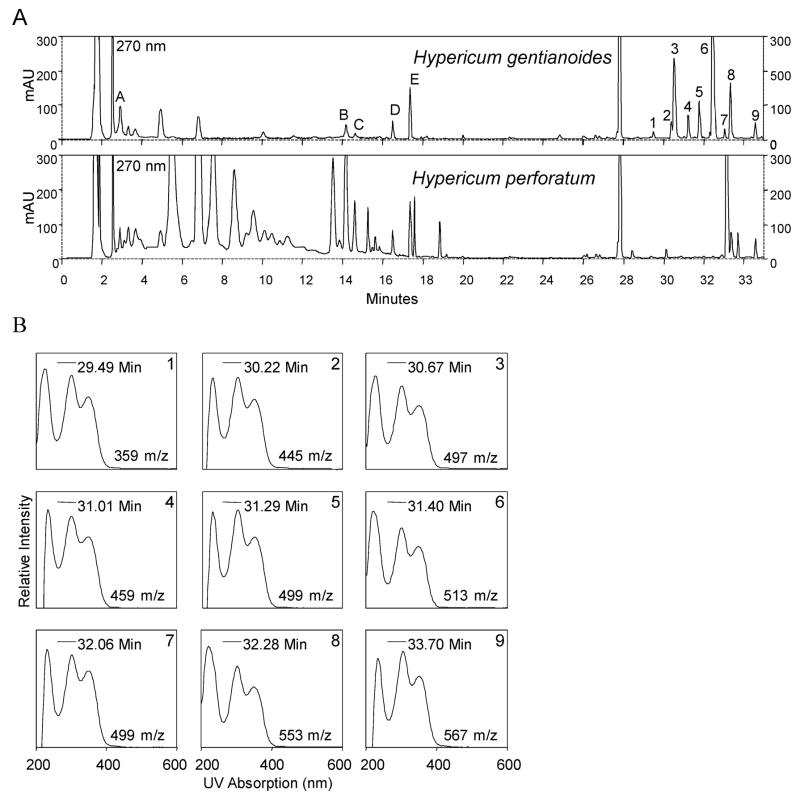
HPLC-PDA analysis of H. gentianoides extracts, using the methanol–low temperature extraction method, revealed a complex chromatographic profile (Figure 2A). A wavelength of 270 nm is used for detection in Hypericum extracts surveyed because a wide range of polyphenolic compounds absorb well at that wavelength. Chlorogenic acid (A), hyperoside (B), isoquercitrin (C), quercitrin (D), and quercetin (E) are present in H. gentianoides as determined by HPLC-PDA with standards (Figure 2A). The presence of A, B, and C are in accordance with ref 16, which used a different H. gentianoides accession.
Figure 2.
H. gentianoides contain a distinct set of metabolites not found in H. perforatum. (A) HPLC chromatography of methanol extracts from H. gentianoides and H. perforatum. Metabolites detected in H. gentianoides and not in H. perforatum are indicated with numbers. Also, many of the abundant secondary metabolites in H. gentianoides are not found in H. perforatum. LC-MS analysis (mass spectra not shown) indicates hypericin and hyperforin, ubiquitous in H. perforatum, are not detected in H. gentianoides. (B) The UV absorption fingerprints of the unknown numbered lipophilic compounds in H. gentianoides are similar, indicating the associated compounds may be related biosynthetically. The mass-to-charge ratio of the molecular ions from complementary LC-MS data are listed next to their associated UV absorption fingerprint as well. See the Supporting Information for the mass spectra of peaks 1–9.
Other than the presence of chlorogenic acid and the four flavonoids listed, H. gentianoides has a very distinct chemical profile compared to that of H. perforatum. Unlike many of the bioactive species in the Hypericum genus (16), neither hypericin (a dianthrone) nor hyperforin (an acyl-phloroglucinol), the two most heavily studied bioactive compounds from St. John’s Wort (17, 18), are present in H. gentianoides. In addition, the ultraviolet (UV) radiation absorption spectra of the HPLC UV-absorbing metabolites reveal a class of related compounds with a retention time from 30 to 45 min; there are at least nine metabolites (Figure 2B) with distinctive UV absorption maxima at 220, 300, and 350 nm or at 226, 287, and 357 nm; the corresponding LC-ESI-MS data are given in the Supporting Information.
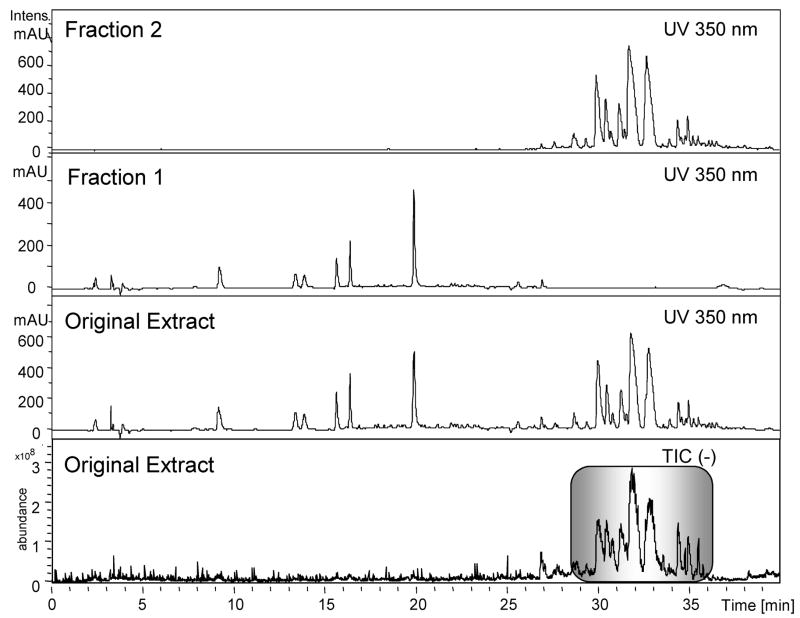
Also, LC-ESI-MS was used to provide additional physical information about compounds in the methanol extract used for fractionation and the anti-inflammatory assays. The total ion chromatograph (TIC) shows that about 79% of the mass in the methanol extract consists of compounds with a retention time between 30 and 35 min, and these compounds comprise 2% of the fresh weight of the plant (Figure 3). This value was calculated by using the combined peak area from 30 to 35 min on the total ion chromatogram (TIC) compared to the total combined peak area from 0 to 45 min, and the extract yield from the original extraction. These nine TIC peaks correlate with the nine UV-absorbing metabolites, as shown with the corresponding mass-to-charge ratios and retention times shown in Figure 2B.
Figure 3.
Semipreparative HPLC separation of H. gentianoides methanol extract. A methanol extract was subjected to semipreparative HPLC and two fractions collected, dividing the more lipophilic constituents (fraction 2) from the more polar constituents, rich in flavonoids such as quercetin (fraction 1), as shown in this analytical LC-UV-MS data. The total ion chromatograph (TIC) from the liquid chromatography–electrospray ionization–mass spectrum analysis shows that the unknown lipophilic compounds (highlighted in the box) based on TIC peak area, comprise approximately 79% of the methanol extract’s mass, accounting for 2% of the plant fresh weight. This high abundance will facilitate purification of enough material for bioactivity-guided fractionation starting with only tens of grams of dry plant material.
Anti-inflammatory Activity of H. gentianoides
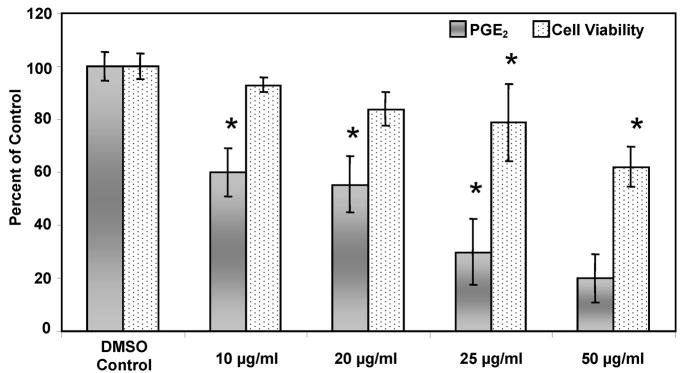
To evaluate the biological activity of H. gentianoides extracts in mammals, we used a RAW264.7 mouse macrophage model system, in which LPS is used to induce an inflammatory response (18). The ability of H. gentianoides extracts to reduce the inflammatory response was evaluated by quantifying PGE2 production in LPS-induced macrophages and comparing to the solvent control (18). Bioassays using extracts prepared from accessions Ames 28015 and Ames 27767 of H. gentianoides both produced a significant reduction of PGE2, compared to the controls. To establish the concentration of extract needed to demonstrate anti-inflammatory activity, macrophages were treated with various concentrations of plant extract from 10 to 50 μg/mL. This revealed a dose response, with decreasing PGE2 accumulation the higher the concentrations of the extract applied (Figure 4).
Figure 4.
H. gentianoides methanol extract significantly reduces PGE2 concentrations in LPS-induced RAW 264.7 macrophages. Cells were treated with 1μg/mL of LPS and the H. gentianoides extract simultaneously for 8 h. Decreased PGE2 indicates increased anti-inflammatory activity. Anti-inflammatory activity is given as the mean percent of control (culture media + DMSO + LPS PGE2 level ± standard error) and cytotoxicity is given as the mean percent of cell viability of control (culture media + DMSO ± standard error) compared to that of H. gentianoides extracts (n) 6 for each). The concentration of the extract tested, in μg/mL dry weight, represents the final concentration of the extract in the media. Addition of LPS to the culture media + DMSO control increased the level of PGE2 11-fold over culture media + DMSO control alone (0.12 ± 0.02 ng/mL for media + DMSO, 1.4 ±0.1 ng/mL for media + DMSO + LPS). Extracts in the culture media without LPS did not affect the concentration of PGE2 as compared to the media +DMSO control. (*) p-value less than 0.05 as compared to the control.
Since H. perforatum contains light-activated compounds, the H. gentianoides extract was tested in dark conditions and after light treatment (17) to determine whether the reduction in PGE2 was dependent on light activation. There was no significant difference in inhibition of PGE2 between the light-activated and dark treatment conditions (data not shown). Therefore, subsequent experiments were performed in light-activated conditions alone. This confirms results shown in Hammer et al. (18) in which H. perforatum extracts also had light-independent anti-inflammatory activity. Also, cytotoxicity was not dependent on light-activation because there was no significant difference between light-activated and dark treatment conditions (Supporting Information). The data validate our hypothesis that H. gentianoides extract, unlike extracts of H. perforatum (23, 24), does not contain any compounds such as hypericin that would cause an increased cytotoxicity following light-activation.
Semipreparative HPLC
To determine which classes of compound(s) are responsible for the bioactivity, and specifically to evaluate the bioactivity of the unidentified metabolites with the distinct UV absorption spectra, a semipreparative HPLC method was developed to fractionate the H. gentianoides extract. This optimized protocol achieves good separation, has a high loading capacity, has the same compound elution order as the analytical separation data, and allows for the isolation of enough fraction material for biological testing in a reasonable amount of time. As an initial test to determine whether the bioactivity in the H. gentianoides extract might be associated with the less polar metabolites with the unique UV spectra, we collected a fraction from 0 to 30 min (fraction 1) and a fraction from 30 to 45 min (fraction 2). Analytical HPLC separation compares the composition of the extract to that of the more polar fraction containing numerous phenylpropanoids and flavonoids (fraction 1) and the less polar fraction (fraction 2) (Figure 3). This effectively separated the compounds with the distinct 230/300/350 UV absorption maxima fingerprint (fraction 2) from the flavonoids and phenylpropanoids to access if they are responsible for the observed anti-inflammatory activity. The UV data for Figure 3 are shown at 350 nm because of the distinct absorption maxima at 350 nm for the compounds of interest in fraction 2.
Anti-inflammatory Activity of H. gentianoides Fractions
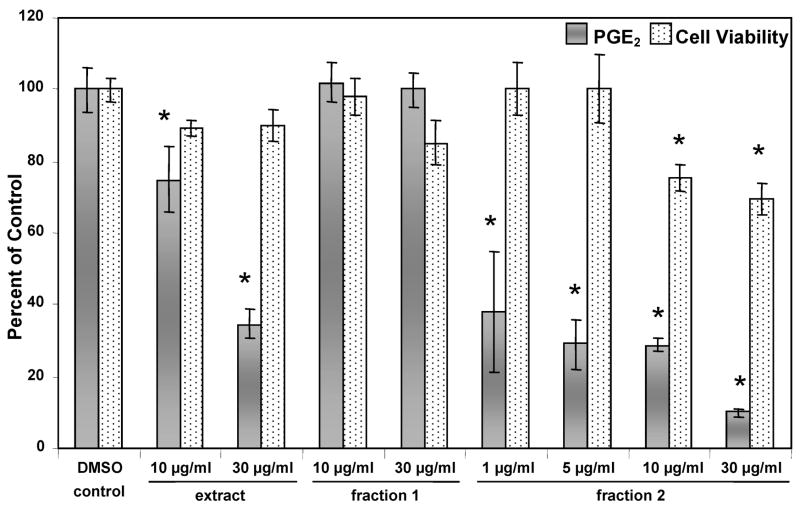
LPS-induced RAW 264.7 macrophages were treated with one of the two HPLC fractions or with the original extract. Fraction 2 significantly reduced the PGE2 concentration in the LPS-induced cells at doses of 30 and 10 μg/mL, while fraction 1 did not reduce the PGE2 concentration at the same doses. This indicates that the active constituents are in fraction 2 (Figure 5). However, fraction 2 has a small amount of significant reduction in cell viability at these concentrations. Further testing revealed that fraction 2 significantly reduces PGE2 concentrations at doses even as low as 1 μg/mL (Figure 5). At 5 and 1 μg/mL, there was no significant reduction in cell viability, suggesting that these compounds have anti-inflammatory activity even at these lower doses in which cytotoxicity is not a concern.
Figure 5.
H. gentianoides fraction 2 significantly reduces PGE2 concentrations in LPS-induced RAW 264.7 macrophages. Decreased PGE2 indicates increased anti-inflammatory activity. Anti-inflammatory activity is given as the mean percent of (media + DMSO + LPS PGE2 level ± standard error) and cytotoxicity is given as the mean percent of (media + DMSO cell viability ± standard error), for fractions 1 and 2, and for extracts of H. gentianoides (n) 4 for each). The concentration of the extract tested, in μg/mL dry weight, represents the final concentration of the extract in the media. Fraction 2 significantly reduced PGE2 concentrations at all doses tested and shows no cytotoxicity at doses of 5 μg/mL or less. (*) p-value less than 0.05 as compared to the control. (**) p-value less than 0.0001 as compared to the control.
DISCUSSION
In recent years metabolomics has accelerated as a field of study (25–27). Not only can we use such tools to understand the biochemistry of an organism and its evolutionary relationship to its environment, but we can also use them to identify the compounds responsible for interorganism biological activities. Recent research on the effect of botanicals on epigenetic events (28) and cell signaling in mammals (29) implicates that numerous small molecules most likely play a large part in the health and life cycle of cells. These findings suggest there are many more unique small molecules that have yet to be discovered, which may play crucial roles in cell signaling. Developing better extraction, separation, and detection methods is crucial for the identification of bioactive constituents not yet discovered.
We developed an extraction method for H. gentianoides designed to minimize degradation of unstable compounds, and a fractionation method for bioactivity-guided separation of the extract. Using these procedures, we determined that extracts from H. gentianoides reduce concentrations of the pro-inflammatory compound PGE2 in LPS-induced RAW 264.7 macrophages, and demonstrated that fraction 2 significantly reduces PGE2 at doses as low as tested, 1 μg/mL, with no significant reduction in cell viability at this dose. This bioactivity is present in extracts from both accessions of H. gentianoides sampled, indicating that the bioactivity is characteristic of at least two populations of this species.
The reduction in PGE2 concentration by the H. gentianoides extract may reflect reduced cyclo-oxygenase activity, since prostaglandin H is the product of this class of enzyme, and is believed to be the substrate for the biosynthesis of PGE2 by prostaglandin E synthase. The mechanism by which this PGE2 reduction is modulated is under investigation.
The characteristic UV absorption fingerprints, retention times, and molecular ions of at least nine HPLC peaks from the bioactive fraction 2, which are described in Figure 2 and in the Supporting Information, indicate that its most abundant constituents may be structurally and/or biosynthetically related, and are consistent with those of certain types of acyl-phloroglucinols (6). Our working hypothesis is thus that the anti-inflammatory compounds of H. gentianoides are acyl-phloroglucinols. These compounds may not have been tested previously for anti-inflammatory properties considering how few molecular cell biology and biochemical studies on North American Hypericum species have been published. This complicates the identification of bioactive constituents given that they must be identified with NMR since there are no standards available. Fraction 2 contains many compounds besides the nine aforementioned peaks of interest, and identification of the bioactive metabolites is in progress, using iterative subfractionation, bioactivity assays, ion trap and quadrapole-time-of-flight (QTOF) LC-MS, and 2D-NMR spectroscopy.
Furthermore, the distinct morphology as well as the potent anti-inflammatory bioactivity and unusual chemical profile of H. gentianoides, relative to other Hypericum species from Eurasia and North America, justifies further investigation. A study comparing the chemical profiles of species in the Section Brathys, to which H. gentianoides belongs, and species in Section Trigynobrathys, since these sections have a similar unusual morphology, would enrich the understanding of evolutionary cohorts of H. gentianoides.
SAFETY
Acetonitrile, methanol, ammonium acetate, and dimethyl sulfoxide are toxic chemicals that should be used in a fume hood.
Acknowledgments
We thank Mark Widrlechner and Joe-Ann McCoy from the USDA-ARS, North Central Regional Plant Introduction Station (112 Plant Introduction Farm, Iowa State University, Ames, IA 50011) for providing plant material. Furthermore, we thank the W. M. Keck Metabolomics Facility, at Iowa State University for providing the analytical instrumentation, including Ann Perera and Director Basil J. Nikolau.
This publication was made possible by grant P01 ES012020 from the National Institute of Environmental Health Sciences (NIEHS) and the Office of Dietary Supplements (ODS), National Institutes of Health (NIH), and grant 9P50AT004155-06 from the National Center for Complementary and Alternative Medicine (NCCAM) and ODS, NIH. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIEHS, NCCAM, or NIH.
ABBREVIATIONS USED
- HPLC
high performance liquid chromatography
- PDA
photo diode array
- LC-ESI-MS
liquid chromatography–electrospray ionization–mass spectrometry
- PGE2
prostaglandin E2
- LPS
lipopolysaccharide
Footnotes
Supporting Information Available: Additional details about previously published cell culture and treatment methods; overlaid HPLC chromatographs of methanol and 70% ethanol extractions of H. gentianoides plant material; data showing no difference in cell viability whether LPS-induced RAW 264.7 macrophages were treated with the H. gentianoides extract in the light or dark; and mass spectrometry data for the numbered peaks in Figure 2A. This material is available free of charge via the Internet at http://pubs.acs.org.
LITERATURE CITED
- 1.Ernst E. Hypericum: The Genus Hypericum. Taylor & Francis CRC Press; London: 2003. p. 196. [Google Scholar]
- 2.Fatland BL, Nikolau BJ, Wurtele ES. Reverse genetic characterization of cytosolic acetyl-CoA generation by ATP-citrate lyase in Arabidopsis. Plant Cell. 2005;17(1):182–203. doi: 10.1105/tpc.104.026211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kreuzaler F, Hahlbrock K. Enzymatic synthesis of aromatic compounds in higher plants: formation of naringenin (5,7,4′-trihydroxyflavonone) from para-coumaroyl coenzyme A and malonyl coenzyme A. FEBS Lett. 1972;28:69–72. doi: 10.1016/0014-5793(72)80679-3. [DOI] [PubMed] [Google Scholar]
- 4.Bais HP, Vepachedu R, Lawrence CB, et al. Molecular and biochemical characterization of an enzyme responsible for the formation of hypericin in St. John’s Wort (Hypericum perforatum L.) J Biol Chem. 2003;278:32413–32422. doi: 10.1074/jbc.M301681200. [DOI] [PubMed] [Google Scholar]
- 5.Mennini T, Gobbi M. The antidepressant mechanism of Hypericum perforatum. Life Sci. 2004;75:1021–1027. doi: 10.1016/j.lfs.2004.04.005. [DOI] [PubMed] [Google Scholar]
- 6.Rocha L, Marston A, Potterat O, Kaplan MAC, Hostettmann K. More phloroglucinols from Hypericum brasiliense. Phytochemistry. 1996;42(1):185–188. doi: 10.1016/0031-9422(95)00507-4. [DOI] [PubMed] [Google Scholar]
- 7.Rabanal RM, Bonkanka CX, Hernandez-Perez M, Sanchez-Mateo CC. Analgesic and topical anti-inflammatory activity of Hypericum canariense L. and Hypericum glandulosum Ait. J Ethnopharmacol. 2005;96:591–596. doi: 10.1016/j.jep.2004.10.013. [DOI] [PubMed] [Google Scholar]
- 8.Singh IP, Bharate SB. Phloroglucinol compounds of natural origin. Nat Prod Rep. 2006;23:558–591. doi: 10.1039/b600518g. [DOI] [PubMed] [Google Scholar]
- 9.Klingauf P, Beuerle T, Mellenthin A, et al. Biosynthesis of the hyperforin skeleton in Hypericum calycinum cell cultures. Phytochemistry. 2005;66:139–145. doi: 10.1016/j.phytochem.2004.11.003. [DOI] [PubMed] [Google Scholar]
- 10.Boubakir Z, Beuerle T, Liu B, Beerhues L. The first prenylation step in hyperforin biosynthesis. Phytochemistry. 2005;66:51–57. doi: 10.1016/j.phytochem.2004.10.020. [DOI] [PubMed] [Google Scholar]
- 11.Sanchez-Mateo CC, Bonkanka CX, Hernandez-Perez M, et al. Evaluation of the analgesic and topical anti-inflammatory effects of Hypericum reflexum L. fil. J Ethnopharmacol. 2006;107(1):1–6. doi: 10.1016/j.jep.2006.01.032. [DOI] [PubMed] [Google Scholar]
- 12.El-Seedi HR, Ringbom T, Torssell K. Constituents of Hypericum laricifolium and their cyclooxygenase (COX) enzyme activities. Chem Pharm Bull. 2003;51:1439–1440. doi: 10.1248/cpb.51.1439. [DOI] [PubMed] [Google Scholar]
- 13.Tedeschi E, Menegazzi M, Margotto D, et al. Anti-Inflammatory actions of St. John’s Wort: Inhibition of human inducible nitric-oxide synthase expression by down-regulating signal transducer and activator of transcription-1α (STAT-1α) activation. J Pharmacol Exp Ther. 2003;307:254–261. doi: 10.1124/jpet.103.054460. [DOI] [PubMed] [Google Scholar]
- 14.Yamakuni T, Aoki K, Nakatani K, et al. Garcinone B reduces prostaglandin E2 release and NF-κB-mediated transcription in C6 rat glioma cells. Neurosci Lett. 2006;394:206–210. doi: 10.1016/j.neulet.2005.10.023. [DOI] [PubMed] [Google Scholar]
- 15.Hamel PB, Chiltoskey MU. Cherokee plants and their uses—A 400 year history. Herald Publishing Co.; Sylva, NC: 1975. p. 72. [Google Scholar]
- 16.Crockett SL, Schaneberg B, Khan IA. Phytochemical profiling of new and old world Hypericum (St. John’s Wort) species. Phytochem Anal. 2005;16:479–485. doi: 10.1002/pca.875. [DOI] [PubMed] [Google Scholar]
- 17.Schmitt LA, Liu Y, Murphy PA, et al. Evaluation of the light-sensitive cytotoxicity of Hypericum perforatum extracts, fractions, and pure compounds. J Agric Food Chem. 2006;54:2881–2890. doi: 10.1021/jf052344k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hammer KDP, Hillwig ML, et al. Inhibition of prostaglandin E2 production by anti-inflammatory Hypericum perforatum extracts and constituents in RAW264.7 mouse macrophage cells. J Agric Food Chem. 2007;55:7323–7331. doi: 10.1021/jf0710074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liu FF, Ang CYW, Springer D. Optimization of extraction conditions for active components in Hypericum perforatum using response surface methodology. J Agric Food Chem. 2000;48:3364–3371. doi: 10.1021/jf991086m. [DOI] [PubMed] [Google Scholar]
- 20.Ganzera MJ, Zhao IA, Khan Hypericum perforatum—Chemical profiling and quantitative results of St. John’s Wort products by an improved high-performance liquid chromatography method. J Pharm Sci. 2002;91:623–630. doi: 10.1002/jps.10057. [DOI] [PubMed] [Google Scholar]
- 21.Snedecor GW. Statistical Methods. Vol. 8. Iowa State University Press; Ames, IA: 1989. [Google Scholar]
- 22.Dunnett CW. A multiple comparison procedure for comparing several treatments with a control. J Am Stat Assoc. 1955;50:1096–1121. [Google Scholar]
- 23.Blank M, Mandel M, Hazan S, Keisari Y, Lavie G. Anti-cancer activities of hypericin in the dark. Photochem Photobiol. 2001;74:120–125. doi: 10.1562/0031-8655(2001)074<0120:acaohi>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 24.Blank M, Lavie G, Mandel M, et al. Antimetastatic activity of the photodynamic agent hypericin in the dark. Int J Cancer. 2004;111:596–603. doi: 10.1002/ijc.20285. [DOI] [PubMed] [Google Scholar]
- 25.Nikolau BJ, Wurtele ES. Concepts in plant metabolomics. Springer London, Limited; London, UK: 2007. p. 297. [Google Scholar]
- 26.Fiehn O. Metabolomics—The link between genotypes and phenotypes. Plant Mol Biol. 2002;48:155–171. [PubMed] [Google Scholar]
- 27.Hall R, Beale M, Feihn O, et al. Plant metabolomics: The missing link in functional genomics strategies. Plant Cell. 2002;14:1437–1440. doi: 10.1105/tpc.140720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shay NF, Banz WJ. Regulation of gene transcription by botanicals: Novel regulatory mechanisms. Annu Rev Nutr. 2005;25:297–315. doi: 10.1146/annurev.nutr.25.050304.092639. [DOI] [PubMed] [Google Scholar]
- 29.Gerisch B, Rottiers V, Li D, et al. A bile acid-like steroid modulates Caenorhabditis elegans lifespan through nuclear receptor signaling. Proc Natl Acad Science, USA. 2007;104:5014–5019. doi: 10.1073/pnas.0700847104. [DOI] [PMC free article] [PubMed] [Google Scholar]