Abstract
Expression of the immediate-early gene c-fos, a marker of neuronal activation, was employed to localize brainstem neuronal populations functionally related to the expiration reflex (ER). Twelve spontaneously breathing, non-decerebrate, pentobarbital anesthetized cats were used. The level of Fos-like immunoreactivity (FLI) in 6 animals with repetitive ERs mechanically induced from the glottis (296±9 ERs) was compared to FLI in 6 control non-stimulated cats. Respiratory rate, arterial blood pressure, and end tidal CO2 concentration remained stable during the experiment.
In the medulla, increased FLI was found in the region of nucleus tractus solitarii (p<0.001), in the ventrolateral medulla along with the lateral tegmental field (p<0.01), and in the vestibular nuclei (p<0.01). In the pons, increased FLI was detected in the caudal extensions of the lateral parabrachial and Kölliker-Fuse nuclei (p<0.05). Within the rostral mesencephalon FLI was enhanced in the midline area (p<0.05). A lower level of ER-related FLI compared to control animals was detected in the pontine raphe region (p<0.05) and the lateral division of mesencephalic periaqueductal gray (p<0.05).
The results suggest that the ER is coordinated by a complex long loop of medullary-pontine-mesencephalic neuronal circuits, some of which may differ from those of other respiratory reflexes. The FLI related to the expulsive behavior ER differs from that induced by laryngeal stimulation and laryngeal adductor responses, particularly in ventrolateral medulla and mesencephalon.
Keywords: c-fos, brainstem, expulsion, expiration reflex, cough, cat
1. Introduction
The expiration reflex (ER) is a defensive reflex of the airways characterized by a solitary brief and intense expiratory effort without a preceding inspiration. It is a significant aspiration prevention behavior restricting penetration of foreign bodies into the lower respiratory tract and expelling them by a fast expiratory airflow (Korpas and Tomori, 1979). The reflex is frequent in many animal species as well as in humans from the very early postnatal period (Korpas and Tomori, 1979; Nishino et al., 1990). ER is easily produced by mechanical stimulation of the medial margin of the vocal folds (Korpas and Tomori, 1979; Korpas and Jakus, 2000). Sensory afferents from laryngeal mucosa project to the solitary tract nucleus containing second order neurons of defensive airway reflexes such as cough or ER (Kalia and Mesulam, 1980; Dyachenko, 1988). The primary efferent pathway begins in the region of the caudal ventral respiratory group (VRG). The expiratory pre-motor neurons in the caudal VRG drive spinal thoracic and abdominal pools of expiratory motoneurons during active expiration resulting in strong expiratory motor output during ER, cough, or sneeze (see review of Iscoe, 1998). The neuronal network that integrates the powerful expulsions during these reflexes is poorly understood. Unlike cough and sneeze, ER is a model of a simple expulsive expiratory activation with no preparatory inspiration. Previous electrophysiological studies reported alterations in the neuronal activities of both the respiratory and non-respiratory modulated neurons in ER, predominantly in the medullary lateral and gigantocellular tegmental fields (Dyachenko, 1990). Kainic acid microinjections into the neurons of the medullary raphe (Jakus et al., 1998), the lateral tegmental field (FTL) of medulla (Jakus et al., 2000) and the rostral dorsolateral pons (Poliacek et al., 2004) were shown to preclude the motor signs of the reflex. Similarly, focal cold block in the rostral VRG led to the reversible suppression of ER (Jakus et al., 1996). Recent studies confirmed that the activity of the rostral and caudal respiratory neurons of VRG are markedly modified during the fictive ER (Baekey et al., 2004), however, these studies are not sufficient to identify all brainstem components of the neurogenic mechanisms responsible for the ER.
The Fos method is based on induced expression of an immediate early gene c-fos, by neuronal depolarization during extra-cellular synaptic activation (Dragunow and Robertson, 1988; Morgan and Curran, 1991). The products of c-fos can be detected by an immunohistochemical method (Dragunow and Faull, 1989). The method was successfully used in the study of functional neural pathways in e.g. laryngeal stimulation (Tanaka et al., 1995), cardiac sympathoexcitatory reflexes (Guo et al., 2002a; 2002b), sneezing (Wallois et al., 1995), coughing (Gestreau et al., 1997), aspiration reflex (AR; Jakus et al., 2004a).
The present study was designed to determine the neuronal populations and the brainstem areas involved in ER using the Fos method. We hypothesized that the frequent ERs would increase the Fos-related expression in several areas of the medulla and pons mostly involved in production of forceful expulsions. Activation of motor pathways involved in ER would presumably expose differences in Fos-like immunoreactivity (FLI) distribution compared to laryngeal stimuli that did not induce ER (Tanaka et al., 1995; Ambalavanar et al., 1999; 2004). Furthermore, we expected to detect differences in ER-related distribution of FLI relative to that found in coughing (Gestreau et al., 1997), sneezing (Wallois et al., 1995), and in the AR (Jakus et al., 2004a; 2004b).
2. Results
The stimulation (and ERs) had little effect on breathing. A slight shortening of the expiratory phase was usually observed (Fig. 1). The respiratory rate (ResR) returned to the baseline within 5–10 s after a series of ERs. As shown in Table 1, there was no significant difference in the mean ResR, blood pressure (BP), and end tidal CO2 concentration (ETCO2) during and post-stimulation referred to the baseline and compared to the control, sham-operated animals.
Figure 1.
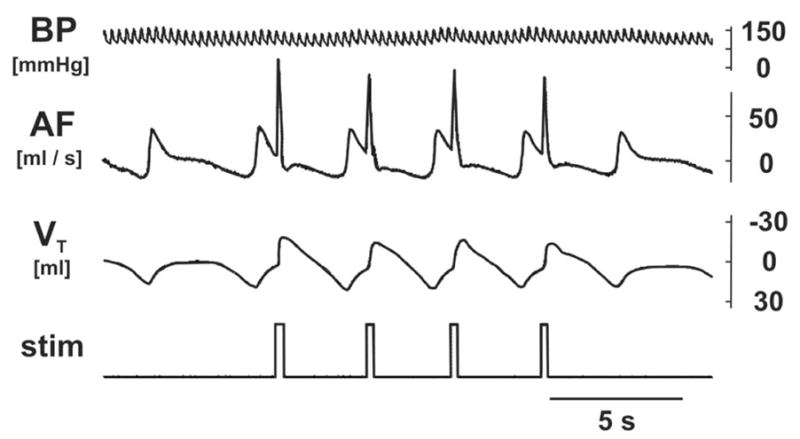
An original record of blood pressure (BP), airflow (AF; expiration up) and tidal volume (VT; expiratory volume up) during expiration reflexes induced by mechanical stimulation (stim) of the glottis.
Table 1.
Cardio-respiratory parameters in ER-induced and sham-operated animals.
| ER induced | sham-operated | ||||
|---|---|---|---|---|---|
| period | control | stim | survival | control | survival |
| ResR [cycles/min] (range) | 17.5±1.8 (13–25) | 18.0±1.9 (14–27) | 20.2±2.5 (14–29) | 18.0±1.8 (13–23) | 19.0±0.7 (17–21) |
| BP systolic [mmHg] (range) | 147±15 (120–190) | 162±13 (125–190) | 151±11 (120–180) | 165±15 (120–190) | 161±15 (120–190) |
| BP diastolic [mmHg] (range) | 97±8 (65–120) | 109±8 (80–120) | 104±8 (80–130) | 112±5 (95–120) | 119±7 (95–130) |
| ETCO2 [%] (range) | 4.7±0.2 (4.0–5.3) | 4.3±0.2 (3.5–4.8) | 4.1±0.3 93.6–4.8) | 4.8±0.3 (4.1–5.3) | 4.3±0.4 (3.5–5.3) |
Respiratory rate (ResR), systolic and diastolic blood pressure (BP), and end tidal CO2 (ETCO2) before stimulation (control), at the intervals of repetitive glottal stimulations (stim), and during survival period (survival) in group of stimulated animals (ER induced). The same parameters analyzed in the early (control) and late (survival) period in a group of sham-operated, non-stimulated cats.
Fos-like expression was detected as a dark-brown staining of variable intensity in inspected brainstem slices. The quantitative analysis of the number of immunolabeled neurons at the brainstem regions with significant differences between the groups of stimulated and control cats is shown in Table 2. The control and ER-related FLI (ER FLI) distribution within the medulla oblongata and pons Varoli (at 1, 5, and 10 mm rostral to the obex) as well as at the mesencephalon (at 13 and 17 mm rostral to the obex) are compared on diagrammatic reconstructions of transverse brainstem sections (Fig. 2). Examples of significantly enhanced or diminished FLI in some medullary and mesencephalic structures (stimulated animal) compared non-stimulated one are shown on Fig. 3.
Table 2.
Unilateral Fos-like immunoreactivity neuronal counts in brainstem regions.
| LEVEL | NUCLEI AND REGIONS | AGN/H | |
|---|---|---|---|
| rostral to the obex | Control | ER | |
|
MEDULLA OBLONGATA | |||
| 0.5 – 1.5 mm | NTS and adjacent border of 5SP | 15 ± 2 (8–20)*** | 50 ± 4 (39–66) |
| 0.5 – 1.5 mm | LRN, NA, NPA, FTL | 22 ± 7 (6–52)** | 73 ± 11 (30–105) |
| 2 – 2.5 mm | LRN, NA, NPA, and adjacent FTL | 13 ± 4 (5–28)** | 39 ± 7 (21–58) |
| 3 – 4 mm | FTL, medial 5SP and IFT, NA, NPA, RFN | 14 ± 3 (9–23)** M | 52 ± 11 (32–105) |
| 4.5 – 5 mm | VN, dorsal part of FTL | 19 ± 4 (7–30)** | 42 ± 4 (32–55) |
| 4.5 – 5.5 mm | RFN, LRN, ventral FTL, caudal VII & RTN | 11 ± 4 (0–27)*** | 59 ± 7 (35–80) |
|
| |||
|
PONS VAROLI | |||
| 7 – 8 mm | Raphe | 8 ± 2 (4–15)* | 2 ± 2 (0–8) |
| 10 mm | NPBL, KF | 10 ± 2 (7–15)* | 19 ± 3 (11–30) |
|
| |||
|
MESENCEPHALON | |||
| 12.5 – 13.5 mm | PAG lateral to Aqueduct | 78 ± 15 (37–120)* W | 38 ± 6 (10–48) |
| 17 – 18 mm | Midline area | 76 ± 10 (40–102)** | 131 ± 11 (98–153) |
The average group number of FLI neurons in a particular area/hemisection (AGN/H), the range of the counts in brackets, and significant differences (*, p<0.05; **, p<0.01; ***, p<0.001) between the non-stimulated (Control) and stimulated group of cats (ER) are shown. Unpaired t-test, Welch corrected unpaired t-test (W), and Mann-Whitney test (M) were used for statistical processing. 5SP, parvocellular division of the alaminar spinal trigeminal ncl; FTL, medullary lateral tegmental fields; IFT, infratrigeminal ncl; KF, Kölliker-Fuse ncl; LRN, lateral reticular ncl; NA, ambigual ncl; NPA, paraambigual ncl; NPBL, lateral parabrachial ncl; NTS, solitary tract ncl; PAG, periaqueductal grey; RFN, retrofacial ncl; RTN, retrotrapezoid ncl; VII, facial ncl; VN, vestibular nuclei.
Figure 2.
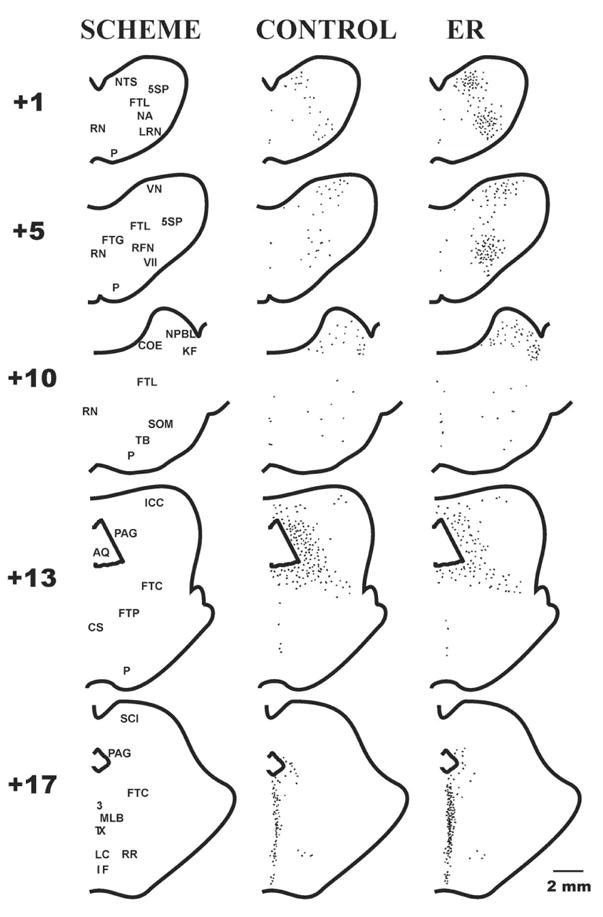
Diagrammatic reconstructions of transverse medullary (+1 and +5), pontine (+10), and mesencephalic (+13 and +17) hemisections with the map of some brainstem structures (SCHEME), control levels of FLI in non-stimulated cats (CONTROL), and the ER-related immunoreactivity in animals with expiration reflex (ER). +1, +5, +10, +13, and +17 represent the positions of the transections (in mm) rostral to the obex. 3, oculomotor ncl; 5SP, parvocellular division of the alaminar spinal trigeminal ncl; VII, facial ncl; AQ, aqueduct; COE, ncl coeruleus; CS, superior central ncl; FTC, central tegmental field; FTG, gigantocellular tegmental field; FTL, medullary and pontine lateral tegmental fields; FTP, paralemniscal tegmental field; ICC, central ncl of the inferior colliculus; IF, interfascicular ncl; KF, Kölliker-Fuse ncl; LC, central linear ncl; LRN, lateral reticular ncl; MLB, medial longitudinal bundle; NA, ambigual ncl; NPBL, lateral parabrachial ncl; NTS, solitary tract ncl; P, pyramidal tract; PAG, periaqueductal grey; RFN, retrofacial ncl; RN, raphe nuclei; RR, retrorubral ncl; SCI, superior colliculus, intermediate layer; SOM, medial ncl of the superior olive; TB, trapezoid body; TX, tegmental decussations; VN, vestibular nuclei.
Figure 3.
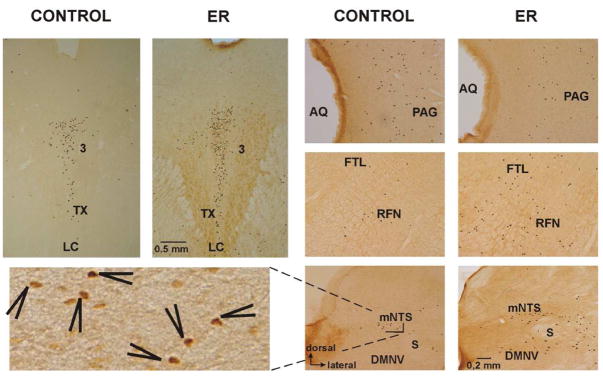
Examples of Fos immunoreactive neurons (dark dots) in cats with expiration reflexes (ER) compared to the control FLI in animals without ER (CONTROL). The level of immunoreactivity decreased in the lateral division of the caudal mesencephalic PAG (right top), but it was enhanced at the rostral ventrolateral medulla (right middle), in the midline area of the rostral mesencephalon (left top), and in the NTS (right bottom). The inset high power photograph of mNTS (left bottom) shows the dark brown staining in Fos positive neurons (arrows). A few weakly stained neurons appear in the high power inset, but these neurons were not clearly visible at low power and as such did not pass our criteria for counting. 3, oculomotor ncl; AQ, aqueduct; DMNV, dorsal motor nucleus of vagus; FTL, lateral tegmental field; LC, central linear ncl; mNTS, medial subnucleus of the solitary tract nucleus; PAG, periaqueductal grey; RFN, retrofacial ncl; S, solitary tract; TX, tegmental decussations..
In control sham-operated non-stimulated cats
FLI was scattered throughout the brainstem in the sensory and motor areas related mostly to the respiratory, cardiac, vasomotor, vestibular, and nociceptive control.
In the medulla oblongata, FLI was detected in relatively few neurons in the caudal medulla (at −4 to −2 mm) mostly confined to the retroambigual nucleus and the adjacent parvocellular reticular formation (the Berman’s FTL) where an average group number of immunoreactive neurons at particular area/hemisection (AGN/H) was 2±1. In the caudal and ventral divisions of the commissural, cuneate, and gracile nuclei, and in the laminar spinal trigeminal nucleus AGN/H was 11±2. Higher AGN/H = 98±32 was found in control animals at the level of obex and slightly caudal to it (to −1 mm) distributed in extended areas through the gracile nucleus, the solitary tract nucleus (NTS), FTL, and the region of lateral reticular nucleus (LRN). At the level 0.5 – 1.5 mm rostral to the obex. (Table 2, Fig. 2) FLI was found in the region of NTS (Fig. 3) and an adjacent border of the alaminar spinal trigeminal nucleus with AGN/H = 15±2, also the region of FTL (AGN/H = 9±2), and the area of ventrolateral medulla (VLM; the ambigual and paraambigual nuclei, LRN) with AGN/H = 13±6. At the rostral medulla discrete FLI labeling was still present in the VLM (the ambigual and paraambigual nuclei, the retrofacial nucleus, the infratrigeminal nucleus, the caudal edge of facial and retrotrapezoid nucleus) and within the area of FTL, with AGN/H 14±3 and 11±4 at levels 3 – 4 mm and up to 5.5 mm rostral to the obex, respectively (Table 2, Fig. 2 and 3). Low FLI was detected in the midline raphe area (AGN/H = 2±1 and 4±2 at +2 till +5 mm and at +5 till +6 mm, respectively). The AGN/H = 19±4 was found in the vestibular nuclei and the adjacent FTL (+4 till +6 mm, Table 2, Fig. 2).
In the lower and middle pons (+6.5 till +8 mm), FLI positive cells were found in the raphe region (AGN/H = 8±2, Table 2) and in the medial and superior vestibular nuclei (AGN/H = 11±3). In the middle and rostral pons within a large area, which includes lateral parts of the gigantocellular and paragigantocellular nuclei, the facial nucleus, FTL, the preolivary nucleus and the medial nucleus of the superior olivae, the AGN/H was 12±3 (10±2 in rostral extension). The highest basal density of immunoreactive neurones was found in the rostral dorsolateral pons (+10.5 till +11 mm), particularly in the lateral parabrachial (NPBL) and the Kölliker-Fuse nuclei (KF) with AGN/H = 84±8. A lower number of immunostained neurones was detected in the brachium conjunctivum, the medial parabrachial nucleus, and the nucleus coeruleus (AGN/H = 24±4). At caudal extension of those areas (approximately at level +10 mm) the number of labeled neurons dramatically dropped (Table 2, Fig. 2).
Within the caudal mesencephalon an intense FLI was detected in the subcuneiform nucleus of the central tegmental field (AGN/H = 60±7 at +12 till +13 mm, Fig. 2) and in the periaqueductal gray (PAG) with AGN/H = 100±10 (+12 till +15 mm, Fig. 2 and 3). In the rostral mesencephalon (+16 till +18 mm) high distribution of FLI was found in midline and adjacent structures with AGN/H = 76±10 (Table 2, Fig. 2 and 3). At this rostral extension the AGN/H within PAG dropped to 24±6.
In stimulated cats with induced ERs, the density of ER FLI was significantly different from that found in control animals. There were no significant differences found between left and right hemisections in any of animals.
At the medulla oblongata ER FLI was significantly enhanced at all rostral locations (+0.5 till +5.5 mm) of VLM (including VRG) and FTL (Table 2, Fig. 2 and 3). At the level around 1 mm rostral to the obex ER FLI was higher within NTS (primarily in the dorsal, interstitial, and ventrolateral subnuclei), and in adjacent medullary structures (Table 2, Fig. 2 and 3). In addition, in rostral medulla enhanced ER FLI was found within a region of the vestibular nuclei (Table 2, Fig. 2). Although higher AGN/H = 10±4 was found in the areas of caudal FTL and the caudal VRG (at −4 till −2 mm) in the group of stimulated animals (compared to 2±1 in control non-stimulated cats) the difference was not statistically significant (p>0.075).
Within the caudal pons (+7 till +8 mm) in the region of raphe nuclei a lower density of ER FLI was found compared to the control FLI (Table 2). Within the rostral dorsolateral pons a significant increase of ER FLI was detected only in the caudal extension of NPBL and KF (at the level approximately 10 mm rostral to the obex; Table 2, Fig. 2). At the rostral part of that region (+10.5 till +11 mm) the ER FLI was not significantly enhanced.
Within the mesencephalon at level approximately 13 mm rostral to the obex a decrease of ER FLI was detected in the area of PAG just lateral to the aqueduct (Table 2, Fig. 2 and 3). At the most rostral extension of the mesencephalic midline area (around +17.5 mm) the density of ER FLI increased (Table 2, Fig. 2 and 3).
3. Discussion
The study presents neuronal activation induced by ER, identified in the brainstem of the cat by the FLI technique. Several nuclei and regions of the medulla, pons, and mesencephalon expressed significant differences in the number of FLI positive neurons in the group of stimulated cats compared to the control non-stimulated animals.
The immunocytochemical detection of immediate early gene c-fos expression is considered a sensitive technique for mapping stimulated neurons (Dragunow and Faull, 1989; Morgan and Curran, 1991). However, the method does not expose the entire neuronal circuit because not all neurons activated during a particular motor behavior may express Fos (Dampney and Horiuchi, 2003). We saw in control cats only a limited number of Fos positive neurons in respiratory active areas of the medulla, which was similar to the results of others (e.g. Wallois et al., 1995). Relatively strong neuronal activation enhances the induction of c-fos (Morgan and Curran, 1991). It has been shown in cats, that gentle insertion of catheter into the nostril (when respiration was slightly modified by few weak sneeze reflexes) did not modify the basal FLI. The substantial increase in FLI followed repetitive sneezing produced by much more efficient method of air puff stimulation (Wallois et al., 1995). Based on these findings we do not believe that in our experiments induction of FLI may have resulted from the alterations in ResR, ETCO2, and BP. These parameters were not significantly changed during the experiment (either inside the groups, or between the groups of stimulated and control cats). We also believe that nociceptive stimulation did not affect our FLI counts. The period of time required for FLI related to non-specific (e.g. nociceptive) stimulation to decline to minimal levels differs depending on the type, intensity, and duration of the stimuli, and also on anesthetic conditions (Bullitt et al., 1992). Pentobarbital anesthesia is known as a suppressant of c-fos expression (Morgan et al., 1987). However, this anesthetic has been utilized for decades in the cat to study airway reflexes such cough or ER and this species reliably respond to glottal stimulation with ER under pentobarbital anesthesia (Korpas and Tomori, 1979; Jakus et al., 2004b, Poliacek et al. 2004). In pentobarbital anesthetized cats, marked reductions of FLI induced by vigorous and repetitive sneezing were observed after a 3 hour period from the stimulation to the perfusion (Wallois et al., 1995). We shortened the period between the surgical preparation and the stimulation because prolonged of anesthesia can reduce an excitability of ER. Our preliminary experiments did not reveal prominent differences in basal FLI between 3 and 6 hour time periods from the surgical preparation to the perfusion. In control cats we found low levels of baseline FLI in most brainstem areas and these findings correspond well with the results of e.g. Wallois et al. (1995) and Gestreau et al. (1997). Limited manipulation of the animal and surgery, the use of local anesthetic in the wound margins leading to a reduction in nociception-related FLI (Lykkegaard et al., 2005), and suppression of c-fos expression by pentobarbital anesthesia (Morgan et al., 1987) all could account for our observations. We found only a weak baseline FLI in the vestibular nuclei and spinal trigeminal nucleus (compared to the studies of Miller and Ruggiero (1994) or Gestreau et al. (1997) reporting robust FLI in those structures, probably as a result of extensive surgical preparations in decerebrated animals), similarly as in other pain-related areas such the medullary and pontine raphe, caudal pontine reticular formation, olivary nuclei, superior colliculi (Bullitt, 1990; Liu et al., 1998). Taken together, our control animals expressed FLI levels that were in line with other studies in the cat that employed longer post surgical times (Wallois et al., 1995; Gestreau et al., 1997; Guo et al., 2002a; 2002b; Ambalavanar et al., 2004). However, we revealed a larger baseline FLI in the rostral dorsolateral pons, and also in several regions of the mesencephalon, probably indicating their complex control and integrative role in somatosensory, alimentary, cardiovascular, and respiratory functions, in arousal and behavioral reactions, vocalization, etc. (Bandler and Shipley, 1994; Bianchi et al., 1995; Davis et al., 1996; Guo et al., 2002b). Anesthesia itself may be considered as an effective stimulus that requires appropriate neuronal responses in order to maintain optimal cardio-respiratory functions. A lower density of basal FLI in the rostral dorsolateral pons in decerebrate (Gestreau et al., 1997) and conscious animals (Hirooka et al., 1997) may support such explanation.
In ER, the afferent information from laryngeal sensory receptors projects primarily to the medullary areas of the interstitial, commissural, and lateral subnuclei of the NTS (Kalia and Mesulam, 1980; Dyachenko, 1988). Hence, the higher level of FLI found in the NTS was expected in our animals. We found a few labeled neurons also at the dorsomedial edge of the spinal trigeminal nucleus near the region of NTS, described as the site of the projections of superior laryngeal nerve (SLN) as well (Nomura and Mizuno, 1983).
The dense expression of ER FLI was found within all VLM rostral to the obex (Table 2). Since pools of expiratory units (mainly from the Bötzinger complex) might provide the excitatory drive to premotor and motor neurons during the cough and ER (Bongianni et al., 1998; Baekey et al., 2004) we assume that the higher levels of ER FLI at the most rostral VLM may reflect the activation of these expiratory neurons. In contrast, such intense FLI was never observed following the AR (Jakus et al., 2004a) and laryngeal stimulation with laryngeal adductor responses (LAR; Tanaka et al., 1995; Ambalavanar et al., 2004) induced no Fos labeling in this region. The VRG from the level of obex up to 3 mm rostral to it is associated mainly with the ambigual and paraambigual nuclei characterized by a functional dominance of the inspiratory neurons (see e.g. Bianchi et al., 1995). Since all inspiratory activity during ER is generally inhibited (Korpas and Jakus, 2000) and expiratory neurons in this region are scarce, it is possible that some of the ER FLI detected in this area reflects units recruited at the time of ER. That includes the labeling in the area of ambigual nucleus (containing laryngeal motoneurons) reported to be related to the LAR (Tanaka et al., 1995; Ambalavanar et al., 2004), which is an inseparable part of the ER (Poliacek et al., 2003). However, FLI induced by laryngeal stimulation and LAR (Tanaka et al., 1995; Ambalavanar et al., 2004) was concentrated to the ambigual area. In contrast, ER FLI (similarly to FLI related to other reflex motor behaviors e.g. cough) was diffusely spread through all structures of VLM (Fig. 2; Table 2). In particular, ER FLI was detected in LRN, retrofacial nucleus, and also in the adjacent ventral and lateral reticular formation. Increased ER FLI in the region of caudal VRG were not statistically significant in the current experiments. The caudal VRG is characterized by a high concentration of expiratory premotor neurons involved in the transmission of excitatory drive to the spinal motoneurons (Iscoe, 1998), particularly during the expulsive reflex behaviors. Significantly increased FLI at this expiratory area was found in sneezing (Wallois et al., 1995), in fictive laryngeal cough (Gestreau et al., 1997), tracheobronchial cough (our preliminary data), and even unexpectedly in the AR (Jakus et al., 2004a). At variance with coughing or sneezing, in ER the number of recruited expiratory units at the caudal VRG might have been not as high as expected (Baekey et al., 2004). The longer-lasting and differently shaped pattern of expiratory activity during coughing compared to ER is likely to be more efficient in the production of Fos. Wallois et al. (1995) raised the still unanswered question of whether spontaneously active medullary expiratory premotor units with axonal projections to the spinal cord are able to express FLI. The extent to which our results represent the expression of c-fos in primarily recruited neurons or in both spontaneously active expiratory premotor as well as recruited neurons is unknown (Bolser et al., 2006).
The expression of ER FLI in the rostral medullary FTL could be amplified by labeling of respiratory, non-respiratory, as well as recruited neurons (Dyachenko, 1990). FTL has been shown before to contain higher densities of FLI in various reflex motor behaviors as in the AR (Jakus et al., 2004a), fictive laryngeal cough (Gestreau et al., 1997), sneezing (Wallois et al., 1995), and fictive vomiting (Miller and Ruggiero, 1994). Significant increases in number of labelled cells at FTL were reported after the SLN stimulation (Ambalavanar et al., 2004) but not after the tactile stimulation of laryngeal vestibulum (Tanaka et al., 1995), both stimulations inducing LAR. The vestibular areas of the brainstem have not displayed increases in FLI due to fictive laryngeal cough (Gestreau et al., 1997), SLN (Ambalavanar et al., 2004) or mechanically induced LAR (Tanaka et al., 1995), fictive vomiting (Miller and Ruggiero, 1994), or sneezing (Wallois et al., 1995), and showed small increase in AR (Jakus et al., 2004a). The role of neurons labeled at caudal vestibular nuclei during ER is as yet unknown. The scattered Fos labeling in the medullary raphe, the significant region for the production of cough and ER in cats (Jakus et al., 1998; Baekey et al., 2003), was presumably caused by the pentobarbital anesthesia, resulting in the suppression of neuronal excitability. Moreover, tonically active neurons (including tonic respiratory units) in the raphe (Baekey et al., 2003) may contribute to the cough and ER by modulation of neuronal activity not effective enough to induce FLI. Sneezing (Wallois et al., 1995), fictive laryngeal cough (Gestreau et al., 1997), and AR (Jakus et al., 2004a) also did not enhance Fos staining in medullary raphe. The only one exception was fictive vomiting (Miller and Ruggiero, 1994).
The labeled areas in the rostral dorsolateral pons are involved in manifestation of cough and ER (Poliacek et al., 2004; Shannon et al., 2004). Fos labeling following fictive laryngeal cough (Gestreau et al., 1997), sneezing (Wallois et al., 1995), and the AR (Jakus et al., 2004a) was markedly enhanced in this area. Lower ER FLI within the entire region of the rostral dorsolateral pons that we found may result from insufficient neuronal activation for producing FLI during ER. Significant alterations in neuronal activity during the defensive airway responses in this area (that induced FLI) could be primarily related to forceful inspirations in cough, sneeze, and AR that is absent in ER. At the pontine raphe region the ER FLI was significantly reduced. This is at variance with previous c-fos study on the AR showing a significant increase in Fos labeling at the pontine raphe midline (Jakus et al., 2004a).
As previously reported (Bolser, 1991; Baekey et al., 2004; Shannon et al., 2004), decerebrate animals showed reliable parameters of coughing and ER, indicating that both the mesencephalic and diencephalic structures are probably not crucially involved in control of these reflexes. However, our ER c-fos study and that on AR (Jakus et al., 2004a) revealed significant increases in the number of FLI positive neurons at the midbrain level. The caudal area of the mesencephalic PAG and the central tegmental field showed an enhanced level of FLI induced by AR (Jakus et al., 2004a). Ambalavanar et al. (1999) in chloralose anesthetized cats reported an increase of FLI related to SLN stimulation, predominantly at the lateral subdivision of PAG. In our experiments with ER, the regions of PAG lateral to the aqueduct expressed less intense ER FLI compared to the control. The reason for this disparity is not clear. Perhaps some of neuronal populations vigorously activated during the expulsive expirations could compensate higher basal Fos expression in PAG, e.g. by an inhibitory action on PAG neurons, since reduced Fos labeling was detected in the lateral PAG also after the coughing (our preliminary data). In the rostral part of the mesencephalic midline area we found the ER FLI markedly enhanced, whereas no such effect was observed either due to SLN stimulation (Ambalavanar et al., 1999) or in the AR (Jakus et al., 2004a). The role of midbrain neurons in the control of airway reflexes remains obscure.
In conclusion, comparison of FLI distribution typical for ER and for other airway reflexes (cough, sneeze, AR) indicated that neuronal populations in the rostral VLM and perhaps also in the rostral mesencephalic midline contribute solely to expiratory expulsions. Some brainstem locations, particularly the caudal VLM and rostral dorsolateral pons, expressed ER FLI that was markedly different from those relevant to the AR, cough, and sneeze. The ER FLI differed also from the Fos labeling related to laryngeal stimulation and LAR, in particular, within the VLM, medullary vestibular nuclei, and the mesencephalon. The ER FLI found in the midbrain suggests a possible modulatory role of the mesencephalic structures in ER and in expulsive expirations.
4. Experimental Procedure
General procedure
Experiments were performed on 12 adult cats of either sex. The animals were divided in the two groups, 6 animals (3.03±0.22 kg) were stimulated in order to induce repetitive ERs, the other 6 animals (3.00±0.26 kg) were non-stimulated and they represent a control (sham operated) group. Cats were anaesthetized with pentobarbital (Vetbutal, Polfa) by an initial dose of 35–40 mg/kg i.p. right in animal facility and then transported to the laboratory. A surgical level of anesthesia was maintained by supplemental doses of anesthetic given intravenously (1–3 mg/kg/hour) depending on an appearance of the palpebral reflex, eye blink reflex, and a jaw tone. Skin and muscle infiltrations by local anesthetics (Mesocaine, Zentiva) were performed at the sites of surgical interventions to minimize induction of c-fos being related to a stimulation of nociceptors (Lykkegaard et al., 2005).
The trachea was cannulated in both the caudal and rostral parts allowing the animal to breathe and for mechanical stimulation of the glottis, respectively. Catheters were inserted into the right femoral vein and artery to inject supplemental doses of anesthetic and for monitoring of the arterial BP, respectively. Histological processing was restricted to animals that had maintained systolic/diastolic values of BP within the range of 120–190/65–130 mmHg. Rectal temperature was kept at 37–38 °C using a heating lamp and a pad.
Recording and mechanical stimulation
We continuously monitored arterial BP, airflow, tidal volume, and ETCO2. The mean values of ResR, systolic and diastolic BP and ETCO2 were taken in the pre-stimulation control, stimulation, and survival time periods (for non-stimulated cats in early - control and late - survival periods; Table 1, ANOVA). At the minimum 3 measurements were taken in each period (the time gap 5 min. in pre-stimulation and stimulation periods, 20 min. or more in the survival period).
Frequent ERs were evoked by gentle repetitive mechanical stimulation of the glottis with a nylon fiber (diameter 0.2–0.35 mm) or nylon fiber loop (Korpas and Tomori, 1979) through the rostral tracheal cannula. Several brief stimuli were applied in the stimulation trial to the glottal area (Korpas and Tomori, 1979), usually one during each expiratory phase of rhythmic respiration, resulting in several ERs per trial period (Fig. 1). Inter-trial intervals of 30–60s allowed for complete recovery of breathing. We paid special attention to the intensity and timing (early expiration, Korpas and Tomori, 1979) of our stimuli so the number of “miss-stimulations” without ER or with other responses induced by laryngeal stimulation such as cough or swallow was very low (below 5%). Animals with higher rates of “miss-stimulations” were excluded from the study. The ER was characterized by a single, short-lasting and strong expiratory effort presenting as a narrow and sharp positive swing of airflow (Fig. 1). In the series of stimulated animals 296±9 ERs (270–320 ERs) were induced in the 25–35 min period of stimulation (i.e. 10–20 ERs per 1 min). Control cats were only monitored during this period.
Perfusion, fixation and immunohistochemical tissue procedure
We did not detect differences in baseline FLI in our control animals with 3 or 6 hours post-surgical periods. Thus, following the preparatory period (approximately 1 hour from the induction of anesthesia to the completion of surgery), the ER protocol was initiated 30 min later. After the stimulation period (25–35 min.), in order to maximize the Fos accumulation within the stimulated neurons (Herdegen et al., 1991; Orendacova et al., 2000) both groups of cats had the same 2 hour survival time, so the perfusion of all animals occurred 3 hours after the completion of the surgical preparation. Animals were then deeply anaesthetized (by an additional dose of 10 mg/kg of pentobarbital i.v.) and perfused transcardially with a 2500 ml bolus of saline containing heparin (1000U/100ml), followed by a solution of 1500 ml of paraformaldehyde 4% in 0.1 M phosphate buffer (PBS) at pH 7.4.
Immediately after perfusion, the brainstem was removed and post-fixed in the same fixative solution for 6 hours. Tissue was then processed in solutions containing 10, 20, and 30% sucrose at 4°C during a period of 48–72 hours. Transverse frozen tissue sections (40μm) were prepared using a cryostat and collected in PBS (0.9% NaCl in 0.1 M phosphate buffer, pH 7.4). Immunohistochemistry was performed using standard procedures. In order to block peroxidase activity, free-floating sections were incubated (30 min. at room temperature) in PBS containing 0.3% H2O2. The slices were washed twice in PBS. To prevent non-specific binding, the sections were incubated for 3 hrs in 0.1 M PBS containing 1% bovine albumin, 3% normal goat serum and 0.2% Triton X-100. An incubation in polyclonal Fos rabbit antibody (c-fos Ab-5 PC 38, Calbiochem, diluted 1:7000) at 4°C followed for 24 hrs. This primary antibody recognizes Fos and Fos-related proteins. The sections were then rinsed twice in PBS and incubated for 2 hours in biotinylated goat-anti rabbit antiserum (BA 100, Vector Laboratories, CA, diluted 1:600 in PBS) and washed twice again after incubation. An avidin-biotin-horseradish peroxidase procedure (1:100, 1hr, Peroxidase Vectastain Elite ABC kit VC-PK 6100, Vector Laboratories, CA) with diaminobenzidine was used to visualize c-Fos staining. All the sections were mounted and dehydrated with graded alcohol cleared with xylene and cover-slipped in Canadian balsam. Adjacent sections were counterstained with Hematoxylin-eosin staining to delineate the location of the neural structures.
Data analysis and statistical evaluation
Sections processed immunocytochemically for Fos-like proteins were drawn by camera lucida. Anatomical landmarks for detection of brainstem structures were established using adjacent counterstained sections. The criteria used to select specific sections for quantification included: (i) identification of the nuclei and structures under investigation, (ii) the lack of artifacts, and (iii) the same rostro-caudal level for sections from different animals. The reconstructions of representative sections were performed according to the cytoarchitectonic atlas (Berman, 1968). The subdivisions of the solitary tract nucleus are referred according to Kalia and Mesulam (1980), and that of Berman (1968), and to Petrovicky (1980) for other brainstem structures. Every fifth brainstem section was used to identify and to count FLI neurons. The distribution and the number of stained neurons were determined with an optical Leitz microscope and video-scanning system (CCD Philips) connected to a PC. The counted cells were inspected (and confirmed) for a presence of Fos staining under the high magnification of the microscope (Fig. 3). Software Ellipse (ViDiTo) was used to evaluate the grain density (Jakus et al., 2004a) in addition to manual counting of Fos positive cells. Individual data were collected for comparison between the right and left sides of the medulla and for averaging of the counts in each area of the brainstem (at the minimum 2 sections in every inspected structure and each rostro-caudal range).
One way Analysis of Variance (ANOVA) with Student-Newman-Keuls post tests was used for statistical processing of cardio-respiratory data (Table 1). Unpaired t-test, Welch corrected unpaired t-test, or Mann-Whitney test (GraphPad, InStat) were applied as appropriate to evaluate the statistical significance of the FLI counts (Table 2). All results shown are means ± S.E.M and their range. In all cases p<0.05 was considered significant.
Acknowledgments
We gratefully acknowledge the technical assistance of Eva Frolova, Peter Machac and Roman Kubizna as well as significant comments on the manuscript of prof. Malgorzata Szereda-Przestaszewska. This study was supported by grant No. 1/2274/05 (VEGA) from the Grant Agency for Science of the Slovak Republic to J. Jakus and NIH R01 07125 to D.C. Bolser.
Abbreviations
- AGN/H
average group number of immunoreactive neurons at particular area/hemisection
- AR
aspiration reflex
- BP
blood pressure
- ER
expiration reflex
- ETCO2
end tidal CO2 concentration
- ER FLI
expiration reflex-related FLI
- FLI
Fos-like immunoreactivity
- FTL
lateral tegmental field
- KF
Kölliker-Fuse nucleus
- LAR
laryngeal adductor response
- LRN
lateral reticular nucleus
- NPBL
lateral parabrachial nucleus
- NTS
solitary tract nucleus
- PAG
periaqueductal gray
- PBS
phosphate buffer
- ResR
respiratory rate
- SLN
superior laryngeal nerve
- VLM
ventrolateral medulla
- VRG
ventral respiratory group
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ambalavanar R, Tanaka Y, Damirjian M, Ludlow CL. Laryngeal Afferent Stimulation Enhances Fos Immunoreactivity in Periaqueductal Gray in the Cat. J Comp Neurol. 1999;409:411–423. [PubMed] [Google Scholar]
- Ambalavanar R, Tanaka Y, Selbie WS, Ludlow CL. Neuronal activation in the medulla oblongata during selective elicitation of the laryngeal adductor response. J Neurophysiol. 2004;92:2920–2932. doi: 10.1152/jn.00064.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baekey DM, Morris KF, Nuding SC, Segers LS, Lindsey BG, Shannon R. Medullary raphe neuron activity is altered during fictive cough in the decerebrate cat. J Appl Physiol. 2003;94:93–100. doi: 10.1152/japplphysiol.00341.2002. [DOI] [PubMed] [Google Scholar]
- Baekey DM, Morris KF, Nuding SC, Segers LS, Lindsey BG, Shannon R. Ventrolateral medullary respiratory network participation in the expiration reflex in the cat. J Appl Physiol. 2004;96:2057–2072. doi: 10.1152/japplphysiol.00778.2003. [DOI] [PubMed] [Google Scholar]
- Bandler R, Shipley MT. Columnar organization in the midbrain periaqueductal gray: modules for emotional expression? Trends Neurosci. 1994;17:379–389. doi: 10.1016/0166-2236(94)90047-7. [DOI] [PubMed] [Google Scholar]
- Berman AL. A cytoarchitectonic atlas with stereotaxic coordinates. The University of Wisconsin press; Madison: 1968. The brainstem of the cat. [Google Scholar]
- Bianchi AL, Denavit-Saubie M, Champagnat J. Central control of breathing in Mammals: Neuronal circuitry, membrane properties, and neurotransmitters. Physiol Rev. 1995;75:1–45. doi: 10.1152/physrev.1995.75.1.1. [DOI] [PubMed] [Google Scholar]
- Bolser DC. Fictive cough in the cat. J Appl Physiol. 1991;71:2325–2331. doi: 10.1152/jappl.1991.71.6.2325. [DOI] [PubMed] [Google Scholar]
- Bolser DC, Poliacek I, Jakus J, Fuller DD, Davenport PW. Neurogenesis of cough, other airway defensive behaviors and breathing: a holarchical system? Respir Physiol Neurobiol. 2006;152:255–265. doi: 10.1016/j.resp.2006.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bongianni F, Mutolo D, Fontana GA, Pantaleo T. Discharge patterns of Bötzinger complex neurons during cough in the cat. Am J Physiol. 1998;274 doi: 10.1152/ajpregu.1998.274.4.R1015. [DOI] [PubMed] [Google Scholar]; Regulatory Integrative Comp Physiol. 43:R1015–R1024. doi: 10.1152/ajpregu.00198.2005. [DOI] [PubMed] [Google Scholar]
- Bullitt E. Expression of c-fos-like protein as a marker for neuronal activity following noxious stimulation in the rat. J Comp Neurol. 1990;296(4):517–530. doi: 10.1002/cne.902960402. [DOI] [PubMed] [Google Scholar]
- Bullitt E, Lee CL, Light AR, Willcockson H. The effect of stimulus duration on noxious-stimulus induced c-fos expression in the rodent spinal cord. Brain Res. 1992;580:172–179. doi: 10.1016/0006-8993(92)90941-2. [DOI] [PubMed] [Google Scholar]
- Dampney RAL, Horiuchi J. Functional organisation of central cardiovascular pathways: studies using c-fos gene expression. Prog Neurobiol. 2003;71:359–384. doi: 10.1016/j.pneurobio.2003.11.001. [DOI] [PubMed] [Google Scholar]
- Davis PJ, Zhang SP, Winkworth A, Bandler R. Neural control of vocalization: respiratory and emotional influences. J Voice. 1996;10:23–38. doi: 10.1016/s0892-1997(96)80016-6. [DOI] [PubMed] [Google Scholar]
- Dragunow M, Robertson HA. Localization and induction of c-fos proteine-like immunoreactive material in the nuclei of adult mammalian neurons. Brain Res. 1988;440:252–260. doi: 10.1016/0006-8993(88)90993-6. [DOI] [PubMed] [Google Scholar]
- Dragunow M, Faull R. The use of c-fos as a metabolic marker in neuronal pathway tracing. J Neurosci Methods. 1989;29:261–265. doi: 10.1016/0165-0270(89)90150-7. [DOI] [PubMed] [Google Scholar]
- Dyachenko YE. Neurophysiological study of bulbar projections of different groups of superior laryngeal afferents. Neirofiziologija. 1988;20:81–90. [PubMed] [Google Scholar]
- Dyachenko YE. Spontaneous and evoked activity of bulbar respiratory and nonrespiratory neurons during the expiration reflex. Neirofiziologija. 1990;22:670–680. [PubMed] [Google Scholar]
- Gestreau C, Bianchi AL, Grelot L. Differential brainstem Fos-like immunoreactivity after laryngeal-induced coughing and its reduction by codeine. J Neurosci. 1997;17:9340–9352. doi: 10.1523/JNEUROSCI.17-23-09340.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo ZL, Lai HC, Longhurst JC. Medullary pathways involved in cardiac sympathoexcitatory reflexes in the cat. Brain Res. 2002a;925:55–66. doi: 10.1016/s0006-8993(01)03259-0. [DOI] [PubMed] [Google Scholar]
- Guo ZL, Li P, Longhurst JC. Central pathways in the pons and midbrain involved in cardiac sympathoexcitatory reflexes in cats. Neuroscience. 2002b;113:435–447. doi: 10.1016/s0306-4522(02)00173-2. [DOI] [PubMed] [Google Scholar]
- Herdegen T, Kovary K, Leah J, Bravo R. Specific temporal and spatial distribution of JUN, FOS and KROX-24 proteins in spinal neurons following noxious transsynaptic stimulation. J Comp Neurol. 1991;313:178–191. doi: 10.1002/cne.903130113. [DOI] [PubMed] [Google Scholar]
- Hirooka Y, Polson JW, Potts PD, Dampney RAL. Hypoxia-induced Fos expression in neurons projecting to the pressor region in the rostral ventrolateral medulla. Neurosci. 1997;80:1209–1224. doi: 10.1016/s0306-4522(97)00111-5. [DOI] [PubMed] [Google Scholar]
- Iscoe S. Control of abdominal muscles. Prog Neurobiol. 1998;56:433–506. doi: 10.1016/s0301-0082(98)00046-x. [DOI] [PubMed] [Google Scholar]
- Jakus J, Tomori Z, Stransky A. Activity of bulbar respiratory neurones during cough and other respiratory tract reflexes in cats. Physiol Bohemoslov. 1985;34:127–136. [PubMed] [Google Scholar]
- Jakus J, Stransky A, Poliacek I, Barani H. Laryngeal patency and expiration reflex following focal cold block of the medulla in the cat. Physiol Res. 1996;45:107–116. [PubMed] [Google Scholar]
- Jakus J, Stransky A, Poliacek I, Barani H, Boselova L. Effects of medullary midline lesions on cough and other airway reflexes in anaesthetized cats. Physiol Res. 1998;47:203–213. [PubMed] [Google Scholar]
- Jakus J, Stransky A, Poliacek I, Barani H, Boselova L. Kainic acid lesions to the lateral tegmental field of medulla: effects on cough, expiration and aspiration reflexes in anesthetized cats. Physiol Res. 2000;49:387–398. [PubMed] [Google Scholar]
- Jakus J, Halasova E, Poliacek I, Tomori Z, Stransky A. Brainstem areas involved in the aspiration reflex: c-fos study in anaesthetized cats. Physiol Res. 2004a;53:703–717. [PubMed] [Google Scholar]
- Jakus J, Tomori Z, Stransky A. Neuronal Determinants of Breathing, Coughing and Related Motor Behaviours: Basics of Nervous Control and Reflex Mechanisms. Wist, Martin; 2004b. [Google Scholar]
- Kalia M, Mesulam MM. Brain stem projections of sensory and motor components of the vagus complex in cat. II Laryngeal, tracheobronchial, pulmonary, cardiac, and gastrointestinal branches. J Comp Neurol. 1980;193:467–508. doi: 10.1002/cne.901930211. [DOI] [PubMed] [Google Scholar]
- Korpas J, Jakus J. The expiration reflex from the vocal folds. Acta Physiol Hung. 2000;87:201–215. doi: 10.1556/APhysiol.87.2000.3.1. [DOI] [PubMed] [Google Scholar]
- Korpas J, Tomori Z. Cough and other respiratory reflexes. New York: S Karger, Basel; 1979. [Google Scholar]
- Liu RJ, Qiang M, Qiao JT. Nociceptive c-fos expression in supraspinal areas in avoidance of descending suppression at the spinal relay station. Neurosci. 1998;85(4):1073–1087. doi: 10.1016/s0306-4522(97)00662-3. [DOI] [PubMed] [Google Scholar]
- Lykkegaard K, Lauritzen B, Tessem L, Weikop P, Svendsen O. Local anaesthetics attenuates spinal nociception and HPA-axis activation during experimental laparotomy in pigs. Res Vet Sci. 2005;79:245–251. doi: 10.1016/j.rvsc.2004.11.017. [DOI] [PubMed] [Google Scholar]
- Miller AD, Ruggiero D. Emetic reflex arc revealed by expression of the immediate-early gene c-Fos in the cat. J Neurosci. 1991;14:871–888. doi: 10.1523/JNEUROSCI.14-02-00871.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan JI, Cohen DR, Hempstead JL, Curran T. Mapping patterns of c-fos expression in the central nervous system after seizure. Science. 1987;237(4811):192–197. doi: 10.1126/science.3037702. [DOI] [PubMed] [Google Scholar]
- Morgan JI, Curran T. Stimulus-transcription coupling in the nervous system: involvement of the inducible proto-oncogenes fos and jun. Annu Rev Neurosci. 1991;14:421–451. doi: 10.1146/annurev.ne.14.030191.002225. [DOI] [PubMed] [Google Scholar]
- Nishino T, Hiraga K, Yokokawa N. Laryngeal and respiratory responses to tracheal irritation at different depth of enflurane anesthesia in humans. Anesthesiology. 1990;73:46–51. doi: 10.1097/00000542-199007000-00008. [DOI] [PubMed] [Google Scholar]
- Nomura S, Mizuno N. Central distribution of efferent and afferent components of the cervical branches of the vagus nerve. Anat Embryol. 1983;166:1–18. doi: 10.1007/BF00317941. [DOI] [PubMed] [Google Scholar]
- Orendacova J, Marsala M, Sulla I, Kafka J, Jalc P, Cizova D, Taira Y, Marsala J. Incipient cauda equina syndrome as a model of somatovisceral pain in dogs: Spinal cord structures involved as revealed by the expression of c-fos and NADPH diaphorase activity. Neurosci. 2000;95:543–557. doi: 10.1016/s0306-4522(99)00429-7. [DOI] [PubMed] [Google Scholar]
- Petrovicky P. Cytoarchitectonics with comparative aspects. Acta Univ. Carolinae Med; Monographia XLIX, Prague: 1980. Reticular formation and its raphe system I. [PubMed] [Google Scholar]
- Poliacek I, Stransky A, Jakus J, Barani H, Tomori Z, Halasova E. Activity of the Laryngeal Abductor and Adductor Muscles during Cough, Expiration and Aspiration Reflexes in Cats. Physiol Res. 2003;52(6):749–762. [PubMed] [Google Scholar]
- Poliacek I, Jakus J, Stransky A, Barani H, Halasova E, Tomori Z. Cough, expiration and aspiration reflexes following kainic acid lesions to the pontine respiratory group in anesthetized cats. Physiol Res. 2004;53:155–163. [PubMed] [Google Scholar]
- Shannon R, Baekey DM, Morris KF, Nuding SC, Segers LS, Lindsey BG. Pontine respiratory group neuron discharge is altered during fictive cough in the decerebrate cat. Respir Physiol Neurobiol. 2004;142:43–54. doi: 10.1016/j.resp.2004.05.002. [DOI] [PubMed] [Google Scholar]
- Tanaka Y, Yoshida Y, Hirano M. Expression of Fos-protein activated by tactile stimulation on the laryngeal vestibulum in the cat’s lower brain stem. J Laryng Otol. 1995;109:39–44. doi: 10.1017/s0022215100129196. [DOI] [PubMed] [Google Scholar]
- Wallois F, Gros F, Masmoudi K, Larnicol N. C-Fos-like immunoreactivity in the cat brainstem evoked by sneeze-inducing air puff stimulation of the nasal mucosa. Brain Res. 1995;687:143–154. doi: 10.1016/0006-8993(95)00487-b. [DOI] [PubMed] [Google Scholar]