Abstract
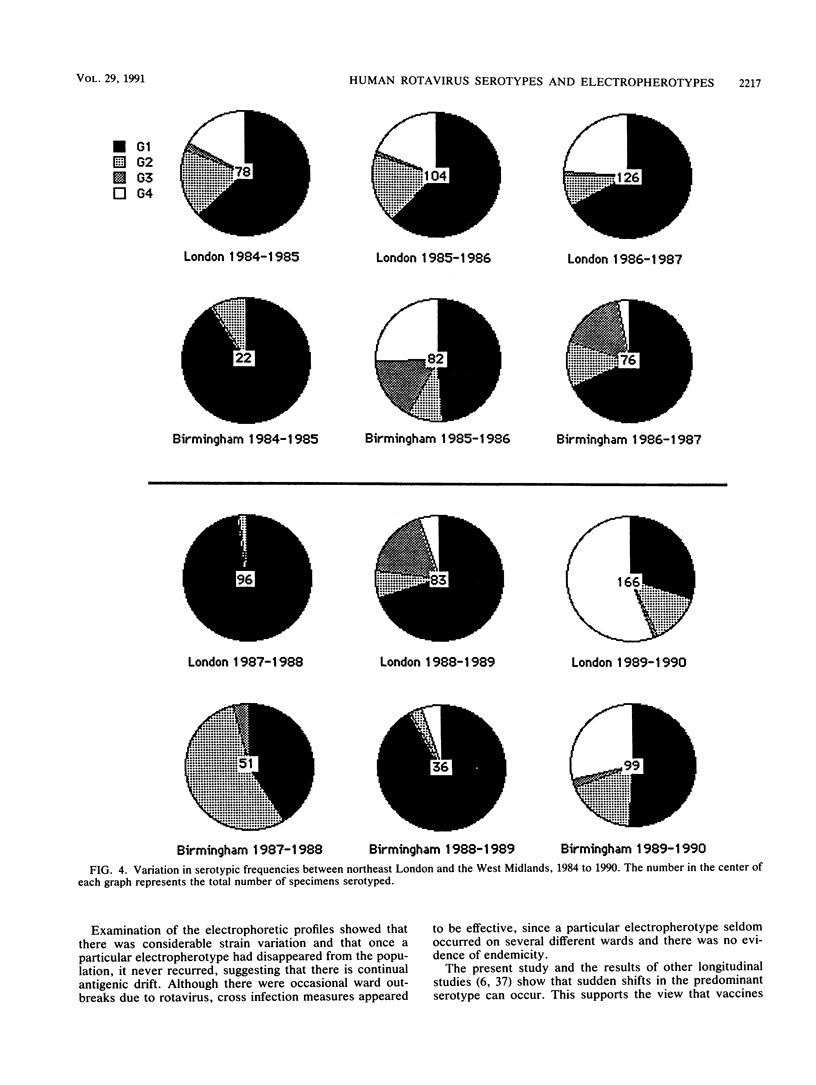
A retrospective and prospective survey was carried out to determine the relative frequency of rotavirus serotypes infecting children with diarrhea or vomiting or both who were admitted to the Hospitals for Sick Children in London during a 6-year period from 1984 to 1990. The results were compared with data for the same period from a study in Birmingham, United Kingdom. The serotype of rotaviruses infecting 1,019 children was ascertained by enzyme immunoassay with VP7-specific monoclonal antibodies. In London, serotype G1 accounted for 60% of the cases, serotype G4 accounted for 24%, serotype G2 accounted for 11%, G3 accounted for 3%, and coinfections accounted for 2%. Considerable differences in the relative prevalence of serotypes were seen when data from London and Birmingham were compared. A major shift from serotype G1 to G4 was observed in London in the 1989 to 1990 season, and a lesser shift was seen in Birmingham. Examination of the electrophoretic profiles of 611 rotaviruses from London showed that there were at least 108 different profiles. Continuous variation occurred throughout the 6-year period, and the same electropherotype never recurred once it had disappeared from the population. None of the electrophoretic profiles were characteristic of group B or group C rotaviruses. There was no evidence that any strain of rotavirus had become endemic in either of the children's hospitals in London.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aboudy Y., Shif I., Zilberstein I., Gotlieb-Stematsky T. Use of polyclonal and monoclonal antibodies and analysis of viral RNA in the detection of unusual group A human rotaviruses. J Med Virol. 1988 Jul;25(3):351–359. doi: 10.1002/jmv.1890250312. [DOI] [PubMed] [Google Scholar]
- Beards G. M., Desselberger U., Flewett T. H. Temporal and geographical distributions of human rotavirus serotypes, 1983 to 1988. J Clin Microbiol. 1989 Dec;27(12):2827–2833. doi: 10.1128/jcm.27.12.2827-2833.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beards G. M. Serotyping of rotavirus by NADP-enhanced enzyme-immunoassay. J Virol Methods. 1987 Nov;18(2-3):77–85. doi: 10.1016/0166-0934(87)90113-3. [DOI] [PubMed] [Google Scholar]
- Birch C. J., Heath R. L., Gust I. D. Use of serotype-specific monoclonal antibodies to study the epidemiology of rotavirus infection. J Med Virol. 1988 Jan;24(1):45–53. doi: 10.1002/jmv.1890240107. [DOI] [PubMed] [Google Scholar]
- Bishop R. F., Davidson G. P., Holmes I. H., Ruck B. J. Virus particles in epithelial cells of duodenal mucosa from children with acute non-bacterial gastroenteritis. Lancet. 1973 Dec 8;2(7841):1281–1283. doi: 10.1016/s0140-6736(73)92867-5. [DOI] [PubMed] [Google Scholar]
- Bishop R. F., Unicomb L. E., Barnes G. L. Epidemiology of rotavirus serotypes in Melbourne, Australia, from 1973 to 1989. J Clin Microbiol. 1991 May;29(5):862–868. doi: 10.1128/jcm.29.5.862-868.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown D. W., Mathan M. M., Mathew M., Martin R., Beards G. M., Mathan V. I. Rotavirus epidemiology in Vellore, south India: group, subgroup, serotype, and electrophoretype. J Clin Microbiol. 1988 Nov;26(11):2410–2414. doi: 10.1128/jcm.26.11.2410-2414.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coulson B. S., Unicomb L. E., Pitson G. A., Bishop R. F. Simple and specific enzyme immunoassay using monoclonal antibodies for serotyping human rotaviruses. J Clin Microbiol. 1987 Mar;25(3):509–515. doi: 10.1128/jcm.25.3.509-515.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flewett T. H., Bryden A. S., Davies H. Letter: Virus particles in gastroenteritis. Lancet. 1973 Dec 29;2(7844):1497–1497. doi: 10.1016/s0140-6736(73)92760-8. [DOI] [PubMed] [Google Scholar]
- Flores J., Taniguchi K., Green K., Perez-Schael I., Garcia D., Sears J., Urasawa S., Kapikian A. Z. Relative frequencies of rotavirus serotypes 1, 2, 3, and 4 in Venezuelan infants with gastroenteritis. J Clin Microbiol. 1988 Oct;26(10):2092–2095. doi: 10.1128/jcm.26.10.2092-2095.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Georges-Courbot M. C., Beraud A. M., Beards G. M., Campbell A. D., Gonzalez J. P., Georges A. J., Flewett T. H. Subgroups, serotypes, and electrophoretypes of rotavirus isolated from children in Bangui, Central African Republic. J Clin Microbiol. 1988 Apr;26(4):668–671. doi: 10.1128/jcm.26.4.668-671.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerna G., Sarasini A., Arista S., di Matteo A., Giovannelli L., Parea M., Halonen P. Prevalence of human rotavirus serotypes in some European countries 1981-1988. Scand J Infect Dis. 1990;22(1):5–10. doi: 10.3109/00365549009023112. [DOI] [PubMed] [Google Scholar]
- Gerna G., Sarasini A., Zentilin L., Di Matteo A., Miranda P., Parea M., Battaglia M., Milanesi G. Isolation in Europe of 69 M-like (serotype 8) human rotavirus strains with either subgroup I or II specificity and a long RNA electropherotype. Arch Virol. 1990;112(1-2):27–40. doi: 10.1007/BF01348983. [DOI] [PubMed] [Google Scholar]
- Greenberg H., McAuliffe V., Valdesuso J., Wyatt R., Flores J., Kalica A., Hoshino Y., Singh N. Serological analysis of the subgroup protein of rotavirus, using monoclonal antibodies. Infect Immun. 1983 Jan;39(1):91–99. doi: 10.1128/iai.39.1.91-99.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanlon P., Hanlon L., Marsh V., Byass P., Shenton F., Hassan-King M., Jobe O., Sillah H., Hayes R., M'Boge B. H. Trial of an attenuated bovine rotavirus vaccine (RIT 4237) in Gambian infants. Lancet. 1987 Jun 13;1(8546):1342–1345. doi: 10.1016/s0140-6736(87)90649-0. [DOI] [PubMed] [Google Scholar]
- Heath R., Birch C., Gust I. Antigenic analysis of rotavirus isolates using monoclonal antibodies specific for human serotypes 1, 2, 3 and 4, and SA11. J Gen Virol. 1986 Nov;67(Pt 11):2455–2466. doi: 10.1099/0022-1317-67-11-2455. [DOI] [PubMed] [Google Scholar]
- Hoshino Y., Sereno M. M., Midthun K., Flores J., Kapikian A. Z., Chanock R. M. Independent segregation of two antigenic specificities (VP3 and VP7) involved in neutralization of rotavirus infectivity. Proc Natl Acad Sci U S A. 1985 Dec;82(24):8701–8704. doi: 10.1073/pnas.82.24.8701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalica A. R., Greenberg H. B., Espejo R. T., Flores J., Wyatt R. G., Kapikian A. Z., Chanock R. M. Distinctive ribonucleic acid patterns of human rotavirus subgroups 1 and 2. Infect Immun. 1981 Sep;33(3):958–961. doi: 10.1128/iai.33.3.958-961.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi N., Lintag I. C., Urasawa T., Taniguchi K., Saniel M. C., Urasawa S. Unusual human rotavirus strains having subgroup I specificity and "long" RNA electropherotype. Arch Virol. 1989;109(1-2):11–23. doi: 10.1007/BF01310514. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lanata C. F., Black R. E., del Aguila R., Gil A., Verastegui H., Gerna G., Flores J., Kapikian A. Z., Andre F. E. Protection of Peruvian children against rotavirus diarrhea of specific serotypes by one, two, or three doses of the RIT 4237 attenuated bovine rotavirus vaccine. J Infect Dis. 1989 Mar;159(3):452–459. doi: 10.1093/infdis/159.3.452. [DOI] [PubMed] [Google Scholar]
- Linhares A. C., Gabbay Y. B., Mascarenhas J. D., Freitas R. B., Flewett T. H., Beards G. M. Epidemiology of rotavirus subgroups and serotypes in Belem, Brazil: a three-year study. Ann Inst Pasteur Virol. 1988 Jan-Mar;139(1):89–99. doi: 10.1016/s0769-2617(88)80009-1. [DOI] [PubMed] [Google Scholar]
- Nakagomi O., Nakagomi T., Hoshino Y., Flores J., Kapikian A. Z. Genetic analysis of a human rotavirus that belongs to subgroup I but has an RNA pattern typical of subgroup II human rotaviruses. J Clin Microbiol. 1987 Jul;25(7):1159–1164. doi: 10.1128/jcm.25.7.1159-1164.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakagomi O., Oyamada H., Kuroki S., Kobayashi Y., Ohshima A., Nakagomi T. Molecular identification of a novel human rotavirus in relation to subgroup and electropherotype of genomic RNA. J Med Virol. 1989 Jul;28(3):163–168. doi: 10.1002/jmv.1890280311. [DOI] [PubMed] [Google Scholar]
- Nakagomi T., Akatani K., Ikegami N., Katsushima N., Nakagomi O. Occurrence of changes in human rotavirus serotypes with concurrent changes in genomic RNA electropherotypes. J Clin Microbiol. 1988 Dec;26(12):2586–2592. doi: 10.1128/jcm.26.12.2586-2592.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nonoyama M., Watanabe Y., Graham A. F. Defective virions of reovirus. J Virol. 1970 Aug;6(2):226–236. doi: 10.1128/jvi.6.2.226-236.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taniguchi K., Hoshino Y., Nishikawa K., Green K. Y., Maloy W. L., Morita Y., Urasawa S., Kapikian A. Z., Chanock R. M., Gorziglia M. Cross-reactive and serotype-specific neutralization epitopes on VP7 of human rotavirus: nucleotide sequence analysis of antigenic mutants selected with monoclonal antibodies. J Virol. 1988 Jun;62(6):1870–1874. doi: 10.1128/jvi.62.6.1870-1874.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taniguchi K., Morita Y., Urasawa T., Urasawa S. Cross-reactive neutralization epitopes on VP3 of human rotavirus: analysis with monoclonal antibodies and antigenic variants. J Virol. 1987 May;61(5):1726–1730. doi: 10.1128/jvi.61.5.1726-1730.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taniguchi K., Urasawa T., Morita Y., Greenberg H. B., Urasawa S. Direct serotyping of human rotavirus in stools by an enzyme-linked immunosorbent assay using serotype 1-, 2-, 3-, and 4-specific monoclonal antibodies to VP7. J Infect Dis. 1987 Jun;155(6):1159–1166. doi: 10.1093/infdis/155.6.1159. [DOI] [PubMed] [Google Scholar]
- Tursi J. M., Albert M. J., Bishop R. F. Production and characterization of neutralizing monoclonal antibody to a human rotavirus strain with a "super-short" RNA pattern. J Clin Microbiol. 1987 Dec;25(12):2426–2427. doi: 10.1128/jcm.25.12.2426-2427.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unicomb L. E., Coulson B. S., Bishop R. F. Experience with an enzyme immunoassay for serotyping human group A rotaviruses. J Clin Microbiol. 1989 Mar;27(3):586–588. doi: 10.1128/jcm.27.3.586-588.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urasawa S., Urasawa T., Taniguchi K., Wakasugi F., Kobayashi N., Chiba S., Sakurada N., Morita M., Morita O., Tokieda M. Survey of human rotavirus serotypes in different locales in Japan by enzyme-linked immunosorbent assay with monoclonal antibodies. J Infect Dis. 1989 Jul;160(1):44–51. doi: 10.1093/infdis/160.1.44. [DOI] [PubMed] [Google Scholar]
- Vesikari T. Clinical and immunological studies of rotavirus vaccines. Southeast Asian J Trop Med Public Health. 1988 Sep;19(3):437–447. [PubMed] [Google Scholar]
- Zheng B. J., Lam W. P., Yan Y. K., Lo S. K., Lung M. L., Ng M. H. Direct identification of serotypes of natural human rotavirus isolates by hybridization using cDNA probes derived from segment 9 of the rotavirus genome. J Clin Microbiol. 1989 Mar;27(3):552–557. doi: 10.1128/jcm.27.3.552-557.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]