Abstract
The depletion of limbal stem cells due to various diseases leads to corneal opacification and visual loss. The unequivocal identification and isolation of limbal stem cells may be a considerable advantage because long-term, functional recovery of corneal epithelium is linked to graft constructs that retain viable stem cell populations. As specific markers of limbal stem cells, the ATP-binding cassette, sub-family G, member2 (ABCG2), a member of the multiple drug-resistance (MDR) family of membrane transporters which leads to a side population phenotype, and transcription factor p63 were proposed recently. Conventional corneal transplantation is not applicable for patients with limbal stem cells deficiency, because the conventional allograft lacks limbal stem cells. The introduction of limbal epithelial cell transplantation was a major advance in the therapeutic techniques for reconstruction of the corneal surface. Limbal epithelial cell transplantation is clinically conducted when cultured allografts as well as autografts are available; however, allografts have a risk of immunologic rejection and autografts are hardly available for patients with bilateral ocular surface disorders. Embryonic stem (ES) cells are characterized by their capacity to proliferate indefinitely and to differentiate into any cell type. We induced corneal epithelial cells from ES cells by culturing them on type IV collagen or alternatively, by introduction of the pax6 gene into ES cells. Recent advances in our study supports the possibility of their clinical use as a cell source for reconstruction of the damaged corneal surface. This review summarizes the recent advances in corneal regeneration therapies and the possible application of ES cell-derived corneal epithelial cells.
Keywords: corneal epithelial cell, limbal stem cell, transplantation, embryonic stem cell
Introduction
Limbal stem cell deficiencies cause conjunctival epithelial ingrowth, neovascularization, chronic inflammation, recurrent epithelial erosions and defects, destruction of the basement membrane, and fibrous tissue ingrowth, leading to severe functional impairment (Tsai et al 1990; Tsubota et al 1995; Kruse and Reinhard 2001). The pathology includes Stevens-Johnson syndrome, ocular cicatricial pemphigoid, chemicals and thermal burns and radiation injury.
Proper visual function requires an intact ocular surface. The integrity of the corneal surface is maintained by two specialized epithelia, the conjunctival epithelium and the limbal corneal epithelium. Corneal epithelial stem cells reside in the palisades of Vogt, located in the basal layer of the limbus, coinciding with the transitional zone between the cornea and the bulbar conjunctiva (Schermer et al 1986; Cotsarelis et al 1989). Limbal stem cells produce undifferentiated progeny with limited proliferative potential that migrate centripetally from the periphery of the corneal epithelium to replace cells desquamating during normal cell turnover (Kinoshita et al 1981; Buck 1985; Kruse 1994; Beebe and Masters 1996; Lehrer et al 1998; Collinson et al 2003; Nagasaki and Zhao 2003).
Conventional corneal transplantation, which replaces the central part of the cornea with the same part from a donor cornea, is used to treat diseases such as keratoconus with intact limbal stem cells; however, conventional corneal transplantation is not applicable for patients with limbal stem cell deficiency. In those cases, the donor cornea is rejected and the cornea is covered by cells migrating from the recipient’s conjunctival epithelium. A series of events leads to functional blindness. To prevent this conjunctival invasion of the corneal surface, restoration of the limbus is required. Recently, corneal regeneration therapy made dramatic progress and is already in clinical practice.
In this article, we summarize recent advances in corneal regeneration therapies and report the possible application of epithelial cells derived from embryonic stem (ES) cells for surface reconstruction of the damaged cornea.
Efforts to identify corneal epithelial stem cells
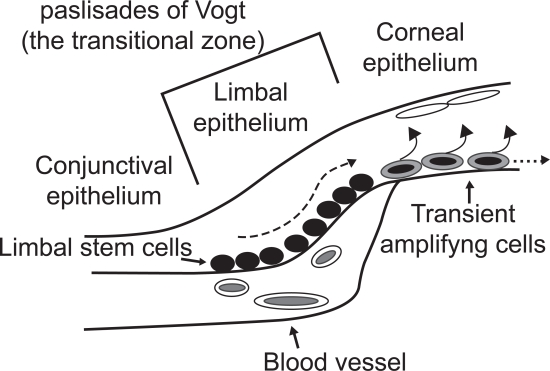
The integrity of the ocular surface is maintained by the centripetal migration of transient amplifying cells derived from limbal stem cells. Transient amplifying cells are present in the corneal epithelia and are committed to epithelial differentiation with limited proliferative potential (Figure 1). The rapidly proliferating transient amplifying cells undergo terminal differentiation accompanied by loss of their proliferative potential.
Figure 1.
Schematic representation of corneal epithelial cells. The ocular surface is composed of three epithela, conjunctival, limbal and corneal. Limbal stem cells are located in the palisades of Vogt, the transitional zone between the cornea and the conjunctiva. Limbal stem cells are close to blood vessels. They generate transient amplifying cells that terminally differentiate after a discrete number of cell divisions to corneal epithelial cells and undergo both centripetal migration and vertical migration.
Limbal stem cells are normally slow cycling cells. When activated by wounding or by in vitro culture conditions, they grow and regenerate the tissue with high proliferative potential (Thoft and Friend 1983; Buck 1985; Cotsarelis et al 1989). Limbal stem cells lack corneal epithelial differentiation-associated markers, such as cytokeratin 3 (Schermer et al 1986), and have highly mitotic ability in vitro (Ebato et al 1987).
Cytokeratins are intermediate filamentous proteins expressed by epithelial cells. A large number of cytokeratin proteins have been identified, each with a specific expression pattern in different epithelial cells. Cytokeratin 12 and cytokeratin 3 are expressed in differentiated and stratified corneal epithelium, although the relative expression of each appears to vary between species. Conjunctiva epithelia express mainly cytokeratin 5 and cytokeratin 14 in their basal layers. On the ocular surface, it is important to distinguish the corneal epithelia from conjunctival epithelia, because, during the healing of corneal epithelial wounds with limbal cell deficiency, conjunctival epithelium often migrates across the denuded limbus to cover the corneal surface (Shapiro et al 1981; Tsai et al 1990; Dua 1998), causing a pronounced decrease in visual acuity.
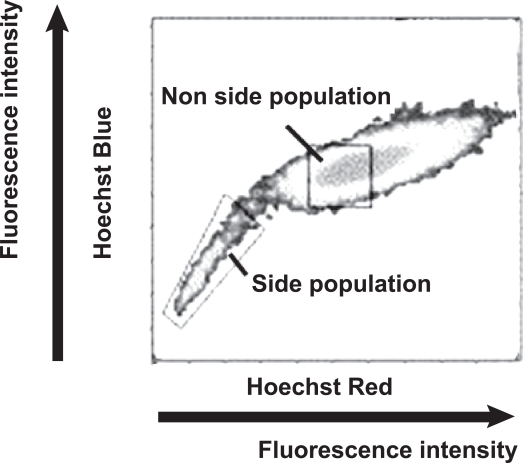
Specific markers for limbal stem cells have not been identified. Recently, many types of tissue-specific stem cells have been shown to exhibit a side population phenotype, defined by their unique ability to efflux the DNA binding dye Hoechst 33342 (Goodell et al 1997; Storms et al 2000). Side population cells are small size and exclude Hoechst 33342 dye with blue and red fluorescence from the cell body. Thus, side population cells lack blue and red fluorescence when analyzed by flow cytometer (Figure 2). The side population phenotype is largely determined by expression of the ATP-binding cassette, sub-family G, and member2 (ABCG2), a member of the multiple drug resistance (MDR) family of membrane transporters (Zhou et al 2001; Lechner et al 2002). ABCG2 is reported to contribute to the side population phenotype in cells from various cell sources, including hematopoietic stem cells. Using fluorescence-activated cell sorting with Hoechst 33342 dye staining, approximately 0.3%–0.5% of limbal epithelial cells exhibit the side population phenotype in humans, whereas no side population cells have been identified in the central corneal epithelium (Watanabe et al 2004, Wolosin et al 2004); therefore, it was suggested that ABCG2-positive limbal epithelial cells may include putative limbal stem cells (Watanabe et al 2004).
Figure 2.
Schematic representation of the side population cells analyzed by flow cytometry. Rat limbal stem cells were treated with Hoechst 33342 dye and were analyzed by flow cytometry. A population of cells with low Hoechst 33342 blue/red fluorescence was isolated as side population cells in the limbus. Non-side population cells accompanying high Hoechst 33342 blue/red fluorescence formed a distinct cell population.
Transcription factor p63 is a member of the p53 family, and is essential for epithelial development and differentiation. p63 has been considered a potential marker of stem/progenitor cells in ocular tissue (Pellegrini et al 2001). It has been hypothesized that p63 may be an earlier marker of limbal stem cells. p63 is highly expressed in the nuclei of limbal basal cells but not in the corneal epithelium (Parsa et al 1999). In addition, it has been demonstrated that p63 expression is gradually reduced from basal cells to terminally differentiated corneal epithelial cells (De et al 2000; Pellegrini et al 2001). These findings suggested that p63-positive cells may include limbal stem cells. In any event, the identification and isolation of limbal stem cells may contribute largely to reconstruction of the ocular surface because long-term, functional recovery of the corneal epithelium is linked to graft constructs that retain viable stem cell populations.
Development of corneal regeneration therapies
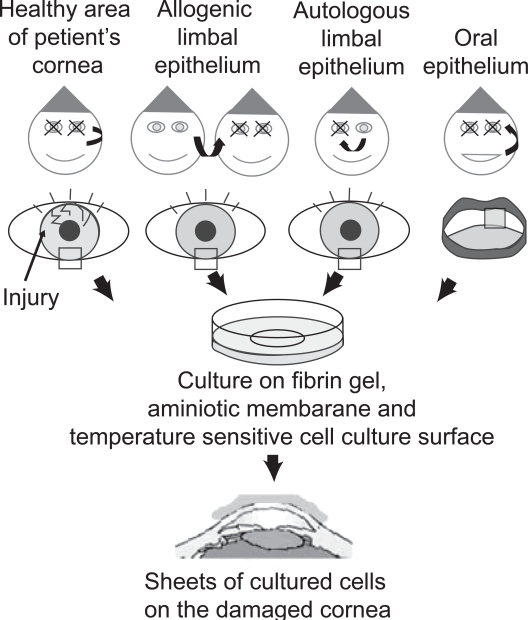
Several ophthalmologists have succeeded in the clinical transplantation of grafts generated from expanded limbal stem cells to treat patients with limbal stem cell deficiencies (Pellegrini et al 1997; Tsai et al 2000; Koizumi et al 2001; Rama et al 2001; Nishida 2003). The methods involve the following two major approaches, allograft transplantation and autologous limbal transplantation (Figure 3).
Figure 3.
Corneal regeneration by using cell sheets made of cultured corneal epithelial cells. Several cell sources which include limbal stem cells are applicable to form epithelial cell sheets for treating patients with limbal stem cell deficiencies. Limbal stem cells are obtained from a healthy area of a patient’s injured cornea (autologous limbal epithelium). Allogeneic limbal epithelium and autologous limbal epithelium of a patient’s contralateral normal cornea are similarly applicable. After culturing on fibrin gel, the amniotic membrane and a temperature-sensitive cell culture surface, these cells formed cell sheets, which were then transplanted onto the damaged cornea. Successful application of a patient’s oral mucous membrane was reported. Oral epithelium is used as an alternative to the limbal epithelium when the patient has severe bilateral limbal stem cell deficiency.
Allograft transplantation is performed in patients with unilateral or bilateral deficiencies (Koizumi et al 2001). The limbal stem cells obtained by biopsy were forced to proliferate sufficiently in the ex vivo culture and the cell sheet made of the expanded limbal stem cells was transplanted to the damaged cornea. Transient amplifying cells are supplied continuously from the donor limbus, and thus corneal limbal epithelium is formed largely by transient amplifying cells in the recipient cornea (Lehrer et al 1998; Tsubota et al 1999); however, this procedure carries risks of immunologic rejection and infection of unidentified organisms. Graft failure usually occurs, even with continuous immunosuppression, owing to serious pre-operative conditions such as persistent inflammation of the ocular surface, severe dry eye and lid related abnormalities (Solomon et al 2002).
Transplantation of autologous limbal stem cell sources is an alternative to allograft transplantation and does not require immunosuppression (Pellegrini et al 1997; Nishida 2003). Autologous limbal transplantation is a method of surface reconstruction of the cornea in patients with unilateral total deficiency of limbal stem cells (Kenyon and Tseng 1989; Tsai and Tseng 1994; Morgan and Murray 1996); however, this procedure requires a large limbal graft from the healthy eye (usually 30%–40% of the donor limbs) (Chen and Tseng 1990). Corneal cell sheets used as grafts were prepared from ex vivo culture of the autologous limbal stem cells; however, grafts lacking a structural basement membrane and extracellular matrix system were fragile and had problems with the decrease of cell to matrix adhesion. Therefore, several researchers have developed autologous cultivated corneal epithelial stem cell sheets using the amniotic membrane, fibrin gel and a temperature-sensitive cell culture surface as a carrier of the cell sheets. (Tsubota et al 1996; Holland and Schwartz 1999; Schwab et al 2000; Tsai et al 2000; Koizumi et al 2001; Rama et al 2001; Shimazaki et al 2004; Nakamura et al 2004; Nishida et al 2004a). These procedures use biological materials and synthetic polymers to cultivate transplantable sheets. The amniotic membrane supports the proliferation and differentiation of limbal stem cells (Kim and Tseng 1995). Cells cultured on the amniotic membrane showed significantly reduced vascularization and inflammation by comparison with other carriers of sheets. Immunoreaction may be suppressed by using the amniotic membrane, because the amniotic membrane expresses incomplete HLA-A, B, C and DR antigens, which may account for the fact that immunological rejection after transplantation has not been observed in this method (Akle et al 1981). Collectively, cells cultured on the amniotic membrane are suitable for transplantation and are easy to handle. These results revealed that the amniotic membrane is applicable as a carrier of corneal epithelial cell sheets. This method (autologous cultivated corneal epithelial stem cell sheets using the amniotic membrane) can not be applied for bilateral total limbal stem cell deficiency such as Stevens-Johnson and ocular cicatricial pemphigoid. In addition, there is a possibility of infection by an unidentified microorganism.
Recently, autologous epithelial cell sheets were developed by culturing a patient’s oral mucous membrane on temperature-sensitive culture surfaces and were successfully transplanted to the damaged cornea (Nishida et al 2004b). Temperature-sensitive culture surfaces allow oral mucousal epithelial cells to interact directly with a recipient’s ocular surface without interference from cell carriers such as fibrin gel and the amniotic membrane. After transplantation and adaptation to the recipient’s microenvironment, the grafted oral mucosal epithelial cells expressed cytokeratin 3; however, they did not express cytokeratin 12. These findings suggest that oral mucousal epithelial cells do not differentiate fully to authentic corneal epithelial cells. If oral mucosa membranes are damaged in systematic diseases such as Stevens-Johnson syndrome, it is hard to recover oral mucosal epithelial cells to make cell sheets.
The most important key to realizing the reconstruction is a sufficient supply of the cells/materials needed for transplantation; therefore, we have focused our attention on ES cells, because ES cells proliferate indefinitely and may provide sufficient numbers of epithelial cells for corneal transplantation.
ES cells as a new cell source of corneal epithelial cell transplantation
Stem cells are the primary cells common to all multicellular organisms that maintain the ability of renewal through cell division, and differentiate into a wide range of specialized cell types. Recent stem cell researches indicated their huge potential as a source of tissue for regenerative medicine. Stem cells may be obtained from embryonic tissues, umbilical cord blood, and some differentiated adult tissues; however, adult stem cells preferentially generate differentiated cells of the same lineage as their tissue of origin.
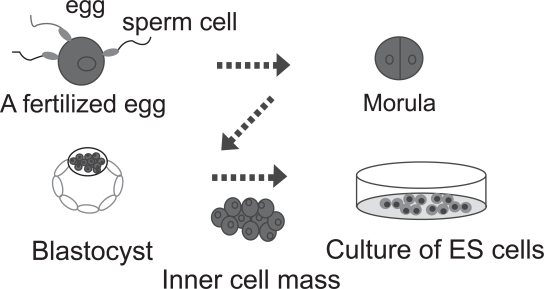
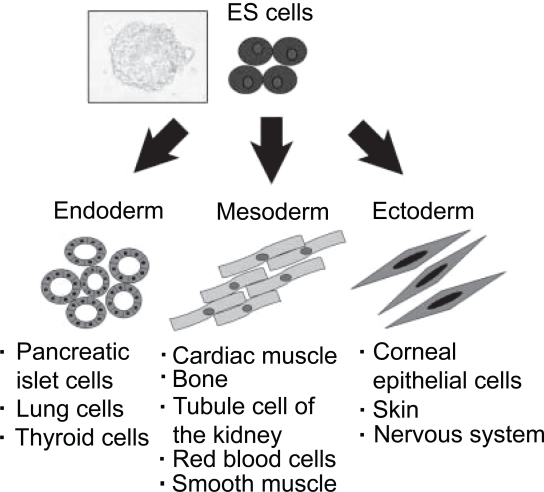
The most primitive cell is the fertilized egg. The fertilized egg and the descendants of the first two cell divisions are totipotent, and are able to form the embryo and the trophoblasts of the placenta. After about four days, these totipotent cells begin to differentiate into blastocysts (Figure 4). ES cells are cultures of cells derived from epiblast tissue of the inner cell mass (ICM) of a blastocyst (Alison et al 2002; Czyz et al 2003). ES cells are isolated by transferring the inner cell mass into a plastic culture dish that contains a nutrient culture medium. After propagation culture, ES cells were established. ES cells are pluripotent cells; they have the ability to differentiate into any cell type of all three germ layers as ectoderm, mesoderm, and endoderm cells, including corneal epithelial progenitors.
Figure 4.
Establishment of ES cells from fertilized egg. ES cells are derived from embryos developed from eggs that have been fertilized in vitro. Human ES cells are typically four or five days old and are a hollow microscopic ball of cells called a blastocyst. The inner cell mass of the blastocyst is collected and cultured on a feeder cell layer. After several passages, the cells are established as ES cells.
ES cells develop into each of the more than 200 cell types of the adult body when given sufficient and necessary stimulation for specific cell-type differentiation (Figure 5). When ES cells are given no stimuli for differentiation, they continue to divide in vitro for a long time and each daughter cell remains pluripotent; therefore, ES cells are a valuable cell source for regenerative medicine and tissue replacement after injury and disease (Kim and Auerbach 2002; Blyszczuk and Czyz 2003). After transplantation, ES-derived cells have the potential to replace host cells lost during injury and disease. Alternatively, ES-derived cells supply host tissues with therapeutic factors leading to a functional benefit. Medical researchers who are studying ES cell differentiation anticipate treating a wide variety of diseases including leukemia, Parkinson’s disease, spinal cord injuries, and muscle damage, amongst a number of other impairments and conditions (Lindvall 2003; Goldman and Windrem 2006). Mouse ES cells are commonly used in basic research field, because they are easy to transfect with a specific gene and perform gene knock out.
Figure 5.
Differentiation of ES cells which give rise to several descendants. ES cells are pluripotent, and give rise during development to all derivatives of the three primary germ layers: endoderm, mesoderm and ectoderm.
In 1998, Dr. Thomson reported the culture of human ES cells, and the production of all kinds of cells and tissues derived from ES cells for transplantation has logically become a reality (Thomson et al 1998); however, ES cell research is ethically controversial, because an ES cell line is made from an extra fertilized ovum used in infertility therapy. Many nations currently have a moratorium on either ES cell research or the production of new ES cell lines.
Previously, we have reported induction of the differentiation of mouse ES cells into corneal epithelial progenitor cells by culturing them on type IV collagen and successful reconstruction of the damaged cornea by transplantation of ES cell-derived epithelial progenitor cells (Homma et al 2004). In general, the presence of substrates or basement membranes is necessary to support normal differentiation of corneal epithelial cells. Type IV collagen is the basic structural component of all basement membranes and forms the backbone to which other basement membrane components attach (Cleutjens et al 1990). The cornea is initially covered with an epithelium that attaches to the normal basement membrane during eye development (Cleutjens et al 1990); therefore, we used type IV collagen as a substitution substrate of the basement membrane. Undifferentiated ES cells were collected from the maintenance culture consisting of leukemia inhibitor factor and mouse embryonic fibroblasts as feeder cells. ES cells were cultured on cell culture dishes for four days to make embryoid bodies. The embryoid bodies were transferred to dishes coated with type IV collagen. Cells cultured on type IV collagen began to form an epithelium-like monolayer at day 8 of culture. These cells expressed pax6 and cytokeratin 12, both of which are markers of corneal epithelial cell differentiation (Liu et al 1994; Moyer et al 1996). They did not express cytokeratin 14, a marker of conjunctival epithelial cells. This suggested that pax6 and other signals necessary for the appropriate differentiation of corneal epithelial cells from ES cells were provided by their interaction with type IV collagen.
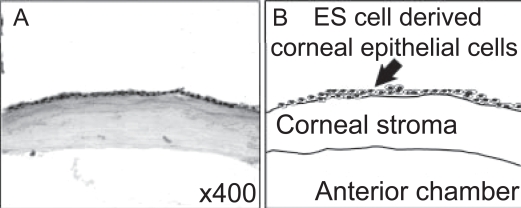
Next, we transplanted these cells. After corneal surface injury with n-heptanol, these cells were transplanted to the injured cornea (Cintron et al 1979; Tsai et al 1990). At 24 hours after transplantation, histological examination of the eyes disclosed that the grafted cells adhered tight to the recipient corneal stroma and completely covered the damaged corneal surface (Figure 6). We have not noted the development of teratoma. We concluded that ES cell-derived epithelial progenitor cells were applicable for corneal epithelial cell transplantation.
Figure 6.
Transplantation of ES cell-derived corneal epithelial cells onto injured cornea. Mouse cornea was denuded by n-heptanol treatment. ES cell-derived epithelial progenitor cells were transplanted to the injured cornea. Histologic analysis was conducted. (A) At 24 hours after transplantation, the eyes were enucleated. Cryostat sections were stained with hematoxylin and eosin staining. (B) Schematic representation of panel A.
With our method, approximately 20% of undifferentiated ES cells cultured on type IV collagen differentiated into corneal epithelial progenitor cells. This result means that the purity of the corneal epithelial cells induced by our culture protocol was not satisfactory. To yield purified corneal epithelial cells, transfer of a gene that promotes corneal epithelial cell differentiation into ES cells is a promising technology. The pax6 transcription factor is a member of the pax gene transcription factor family (Walther and Gruss 1991) and is known to function during early embryogenesis to control key steps in ocular system development (Collinson et al 2003). In particular, pax6 is required not only for embryonic development but also for postnatal development and maintenance of the adult cornea (Davis et al 2003). Pax6 protein is localized to the nuclei of epithelium, but neither stromal nor endothelial cells of the cornea (Koroma et al 1997). Pax6 has been shown to activate corneal gene promoters for cytokeratin 12 (Liu et al 1999; Shiraishi et al 1998); thus, pax6 is considered a master regulator of eye development.
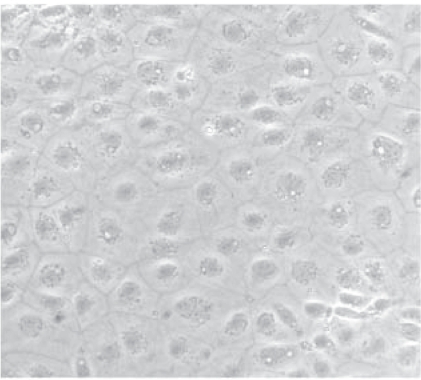
To obtain highly purified corneal epithelial cells, we introduced the pax6 gene to mouse ES cells (Kayama et al 2006). The pax6 gene in combination with green fluorescence protein (GFP) and neomycin-resistant gene was electrotransfected to ES cells, and the transfected cells were cultivated with antibiotic neomycin for 14 days. Neomycin selection culture enables us to purify cells which have taken up the pax6 gene. After neomycin selection, pax6-transfected cells formed a monolayer of cells and demonstrated an epithelial cell-like appearance. We found that the efficiency of inducing corneal epithelial cells with the pax6 transfection method is higher than that by culturing on type IV collagen. We then transplanted pax6-transfected cells onto damaged cornea (Ueno et al 2006). The pax6-transfected cells formed bilayers of epithelial cells that were GFP-positive 24 hours after transplantation. The injury-induced detachment of corneal epithelial layers caused swelling of the corneal stroma, whereas transplantation of pax6-transfected cells reduced the swelling and inflammatory cell infiltration of the corneal stroma was subsided. The pax6-transfected cells expressed cytokeratin 12 at the grafted site. Contamination of other germ cells and teratoma were not detected. Thus, the highly purified corneal epithelial cells derived from pax6-transfected ES cells survived on the injured cornea. The cells restored the damaged cornea histologically and functioned as native corneal epithelial cells because they suppressed corneal epithelial damage-induced inflammation. As the next step to clinical application, we have started to culture cynomolgus monkey ES cells for the induction of corneal epithelial cells. Cynomolgus monkey ES cells share many characteristics with human ES cells, and resemble human ES cells more than mouse ES cells. Cynomolgus monkey ES cells preferentially differentiated into corneal epithelial-like cells in vitro (Figure 7).
Figure 7.
Corneal epithelial progenitor cells differentiated from cynomolgus monkey ES cells in vitro. Inverted microscopic view. ES cells were cultured on cell culture dishes for 4 days to form embryoid bodies, and the embryoid bodies were cultured on type IV collagen for 4 more days. The adhering cells emerged from the embryoid bodies had an epithelial cell-like appearance.
In summary, we have shown the establishment of corneal epithelial cell differentiation of mouse ES cells and cynomolgus monkey ES cells. We hope that human ES-derived epithelial cells may become applicable for surface reconstruction of damaged corneas in the near future.
Acknowledgments
The authors are grateful to our collaborators, Dr. R. Homma, Dr. Y. Kumagai, Prof. S. Ueno (St. Marianna University School of Medicine, Japan) and Prof. K. Tsubota (Keio University School of Medicine, Japan).
This study was supported in part by a Grant for Translational Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan, and a Grant from St. Marianna University School of Medicine, Kawasaki, Japan.
References
- Akle CA, Adinolfi M, Welsh KI, et al. Immunogenicity of human amniotic epithelial cells after transplantation into volunteers. Lancet. 1981;2:1003–5. doi: 10.1016/s0140-6736(81)91212-5. [DOI] [PubMed] [Google Scholar]
- Alison MR, Poulsom R, Forbes S, et al. An introduction to stem cells. J Pathol. 2002;197:419–23. doi: 10.1002/path.1187. [DOI] [PubMed] [Google Scholar]
- Beebe DC, Masters BR. Cell lineage and the differentiation of corneal epithelial cells. Invest Ophthalmol Vis Sci. 1996;37:1815–25. [PubMed] [Google Scholar]
- Blyszczuk P, Czyz J. Expression of Pax4 in embryonic stem cells promotes differentiations of nestin-positive progenitor and insulin-producing cells. Proc Natl Acad Sci USA. 2003;100:998–1003. doi: 10.1073/pnas.0237371100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buck RC. Measurement of centripetal migration of normal corneal epithelial cells in the mouse. Invest Ophthalmol Vis Sci. 1985;26:1296–9. [PubMed] [Google Scholar]
- Chen JJ, Tseng SC. Corneal epithelial wound healing in partial limbal deficiency. Invest Ophthalmol Vis Sci. 1990;31:1301–14. [PubMed] [Google Scholar]
- Cintron C, Hassinger L, Kublin CL, et al. A simple method for the removal of rabbit corneal epithelium utilizing n-heptanol. Ophthalmic Res. 1979;11:90–6. [Google Scholar]
- Cleutjens JP, Havenith MG, Kasper M, et al. Absence of type IV collagen in the centre of the corneal epithelial basement membrane. Histochem J. 1990;22:688–94. doi: 10.1007/BF01047454. [DOI] [PubMed] [Google Scholar]
- Collinson JM, Quinn JC, Hill RE, et al. The roles of Pax6 in the cornea, retina and olfactory epithelium of the developing mouse embryo. Dev Biol. 2003;255:303–12. doi: 10.1016/s0012-1606(02)00095-7. [DOI] [PubMed] [Google Scholar]
- Cotsarelis G, Cheng SZ, Dong G, et al. Existence of slow-cycling limbal epithelial basal cells that can be preferentially stimulated to proliferate: implications on epithelial stem cells. Cell. 1989;57:201–9. doi: 10.1016/0092-8674(89)90958-6. [DOI] [PubMed] [Google Scholar]
- Czyz J, Wiese C, Rolletschek A, et al. Potential of embryonic and adult stem cells in vitro. Biol Chem. 2003;384:1391–409. doi: 10.1515/BC.2003.155. [DOI] [PubMed] [Google Scholar]
- Davis J, Duncan MK, Robison WG, et al. Requirement for Pax6 in corneal morphogenesis: a role in adhesion. J Cell Sci. 2003;116:2157–67. doi: 10.1242/jcs.00441. [DOI] [PubMed] [Google Scholar]
- De LV, Rossi A, Terrinoni A, et al. p63 and p73 transactivate differentiation gene promoters in human keratinocytes. Biochem Biophys Res Commun. 2000;273:342–6. doi: 10.1006/bbrc.2000.2932. [DOI] [PubMed] [Google Scholar]
- Dua HS. The conjunctiva in corneal epithelial wound healing. Br J Ophthalmol. 1998;82:1407–11. doi: 10.1136/bjo.82.12.1407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebato B, Friend J, Thoft RA. Comparison of central and peripheral human corneal epithelium in tissue culture. Invest Ophthalmol Vis Sci. 1987;28:1450–6. [PubMed] [Google Scholar]
- Goldman S, Windrem M. Cell replacement therapy in neurological disease. Philos Trans R Soc Lond B Biol Sci. 2006;361:1463–75. doi: 10.1098/rstb.2006.1886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodell MA, Rosenzweig M, Kim H, et al. Dye efflux studies suggest that hematopoietic stem cells expressing low or undetectable levels of CD34 antigen exist in multiple species. Nat Med. 1997;3:1337–45. doi: 10.1038/nm1297-1337. [DOI] [PubMed] [Google Scholar]
- Holland EJ, Schwartz GS. Epithelial stem-cell transplantation for severe ocular-surface disease. N Engl J Med. 1999;340:1752–3. doi: 10.1056/NEJM199906033402208. [DOI] [PubMed] [Google Scholar]
- Homma R, Yoshikawa H, Takeno M, et al. Induction of epithelial progenitors in vitro from mouse embryonic stem cells and application for reconstruction of damaged cornea in mice. Invest Ophthalmol Vis Sci. 2004;45:4320–6. doi: 10.1167/iovs.04-0044. [DOI] [PubMed] [Google Scholar]
- Kayama M, Ueno H, Homma R, et al. 2006In vitro corneal epithelial cell Induction with high purity by Pax6 gene transfection of mouse embryonic stem cells Invest Ophthalmol Vis Sci 47E-Abstract 1110. [Google Scholar]
- Kenyon KR, Tseng SCG. Limbal autograft transplantation for ocular surface disorders. Ophthalmology. 1989;96:709–22. doi: 10.1016/s0161-6420(89)32833-8. [DOI] [PubMed] [Google Scholar]
- Kim JC, Tseng SC. Transplantation of preserved human amniotic membrane for surface reconstruction in severely damaged rabbit corneas. Cornea. 1995;14:473–84. [PubMed] [Google Scholar]
- Kim JH, Auerbach JM. Dopamine neurons derived from embryonic stem cells function in an animal model of Parkinson’s disease. Nature. 2002;418:50–6. doi: 10.1038/nature00900. [DOI] [PubMed] [Google Scholar]
- Kinoshita S, Friend J, Thoft RA. Sex chromatin of donor corneal epithelium in rabbits. Invest Ophthalmol Vis Sci. 1981;21:434–41. [PubMed] [Google Scholar]
- Koizumi N, Inatomi T, Suzuki T, et al. Cultivated corneal epithelial stem cell transplantation in ocular surface disorders. Ophthalmology. 2001;108:1569–74. doi: 10.1016/s0161-6420(01)00694-7. [DOI] [PubMed] [Google Scholar]
- Koroma BM, Yang JM, Sundin OH. The Pax-6 homeobox gene is expressed throughout the corneal and conjunctival epithelia. Invest Ophthalmol Vis Sci. 1997;38:108–20. [PubMed] [Google Scholar]
- Kruse FE. Stem cells and corneal epithelial regeneration. Eye. 1994;8:170–83. doi: 10.1038/eye.1994.42. [DOI] [PubMed] [Google Scholar]
- Kruse FE, Reinhard T. Limbus transplantation for reconstruction of the ocular surface. Ophthalmologe. 2001;98:818–31. doi: 10.1007/s003470170057. [DOI] [PubMed] [Google Scholar]
- Lechner A, Leech CA, Abraham EJ, et al. Nestin-positive progenitor cells derived from adult human pancreatic islets of Langerhans contain side population (SP) cells defined by expression of the ABCG2 (BCRP1) ATP-binding cassette transporter. Biochem Biophys Res Commun. 2002;293:670–4. doi: 10.1016/S0006-291X(02)00275-9. [DOI] [PubMed] [Google Scholar]
- Lehrer MS, Sun T-T, Lavker RM. Strategies of epithelial repair: modulation of stem cell and transient amplifying cell proliferation. J Cell Sci. 1998;111:2867–75. doi: 10.1242/jcs.111.19.2867. [DOI] [PubMed] [Google Scholar]
- Lindvall O. Stem cells for cell therapy in Parkinson’s disease. Pharmacol Res. 2003;47:279–87. doi: 10.1016/s1043-6618(03)00037-9. [DOI] [PubMed] [Google Scholar]
- Liu CY, Zhu G, Converse R, et al. Characterization and chromosomal localization of the cornea-specific murine keratin gene Krt1. 12. J Biol Chem. 1994;269:24627–36. [PubMed] [Google Scholar]
- Liu JJ, Kao WW, Wilson SE. Corneal epithelium-specific mouse keratin K12 promoter. Exp Eye Res. 1999;68:295–301. doi: 10.1006/exer.1998.0593. [DOI] [PubMed] [Google Scholar]
- Morgan S, Murray A. Limbal autotransplantation in the acute and chronic phases of severe chemical injuries. Eye. 1996;10:349–54. doi: 10.1038/eye.1996.72. [DOI] [PubMed] [Google Scholar]
- Moyer PD, Kaufman AH, Zhang Z, et al. Conjuctival epithelial cells can resurface denuded cornea, but do not transdifferentiate to express cornea-specific keratin12 following removal of limbal epithelium in mouse. Differentiation. 1996;60:31–8. doi: 10.1046/j.1432-0436.1996.6010031.x. [DOI] [PubMed] [Google Scholar]
- Nagasaki T, Zhao J. Centripetal migration of corneal epithelial cells in the normal adult mouse. Invest Ophthalmol Vis Sci. 2003;44:558–66. doi: 10.1167/iovs.02-0705. [DOI] [PubMed] [Google Scholar]
- Nakamura T, Yoshitani M, Rigby H, et al. Sterilized, freeze-dried amniotic membrane-A useful substrate for ocular surface reconstruction. Invest Ophthalmol Vis Sci. 2004;45:93–9. doi: 10.1167/iovs.03-0752. [DOI] [PubMed] [Google Scholar]
- Nishida K. Tissue engineering of the cornea. Cornea. 2003;22:28–34. doi: 10.1097/00003226-200310001-00005. [DOI] [PubMed] [Google Scholar]
- Nishida K, Yamamoto M, Hayashida Y, et al. Functional bioengineered corneal epithelial sheet grafts from corneal stem cells expanded ex vivo on a temperature-responsive cell culture surface. Transplantation. 2004a;77:379–85. doi: 10.1097/01.TP.0000110320.45678.30. [DOI] [PubMed] [Google Scholar]
- Nishida K, Yamamoto M, Hayashida Y, et al. Corneal reconstruction using tissue-engineered cell sheets comprising autologous oral mucosal epithelium. N Engl J Med. 2004b;351:1187–96. doi: 10.1056/NEJMoa040455. [DOI] [PubMed] [Google Scholar]
- Parsa R, Yang A, McKeon F, et al. Association of p63 with proliferative potential in normal and neoplastic human keratinocytes. J Invest Dermatol. 1999;113:1099–105. doi: 10.1046/j.1523-1747.1999.00780.x. [DOI] [PubMed] [Google Scholar]
- Pellegrini G, Traverso CE, Franzi AT, et al. Long-term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet. 1997;349:990–3. doi: 10.1016/S0140-6736(96)11188-0. [DOI] [PubMed] [Google Scholar]
- Pellegrini G, Dellambra E, Golisano O, et al. p63 identifies keratinocyte stem cells. Proc Natl Acad Sci USA. 2001;98:3156–61. doi: 10.1073/pnas.061032098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rama P, Bonini S, Lambiase A, et al. Autologous fibrin-cultured limbal stem cells permanently restore the corneal surface of patients with total limbal stem cell deficiency. Transplantation. 2001;72:1478–85. doi: 10.1097/00007890-200111150-00002. [DOI] [PubMed] [Google Scholar]
- Schermer A, Galvin S, Sun TT. Differentiation-related expression of a major 64K corneal keratin in vitro and in culture suggests limbal location of corneal epithelial stem cells. J Cell Biol. 1986;103:49–62. doi: 10.1083/jcb.103.1.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwab IR, Reyes M, Isseroff RR. Successful transplantation of bioengineered tissue replacements in patients with ocular surface disease. Cornea. 2000;19:421–6. doi: 10.1097/00003226-200007000-00003. [DOI] [PubMed] [Google Scholar]
- Shapiro MS, Friend J, Thoft RA. Corneal re-epithelialization from the conjunctiva. Invest Ophthalmol Vis Sci. 1981;21:135–42. [PubMed] [Google Scholar]
- Shimazaki J, Shimmura S, Tsubota K. Donor source affects the outcome of ocular surface reconstruction in chemical or thermal burns of the cornea. Ophthalmology. 2004;111:38–44. doi: 10.1016/j.ophtha.2003.02.003. [DOI] [PubMed] [Google Scholar]
- Shiraishi A, Converse RL, Liu CY, et al. Identification of the cornea-specific keratin 12 promoter by in vivo particle-mediated gene transfer. Invest Ophthalmol Vis Sci. 1998;39:2554–61. [PubMed] [Google Scholar]
- Solomon A, Ellies P, Anderson DF, et al. Long-term outcome of keratolimbal allograft with or without penetrating keratoplasty for total limbal stem cell deficiency. Ophthalmology. 2002;109:1159–66. doi: 10.1016/s0161-6420(02)00960-0. [DOI] [PubMed] [Google Scholar]
- Storms RW, Goodell MA, Fisher A, et al. Hoechst dye efflux reveals a novel CD7(+) CD34(–) lymphoid progenitor in human umbilical cord blood. Blood. 2000;96:2125–33. [PubMed] [Google Scholar]
- Thoft RA, Friend J. The X, Y, Z hypothesis of corneal epithelial maintenance. Invest Ophthalmol Vis Sci. 1983;24:1442–3. [PubMed] [Google Scholar]
- Thomson JA, Itskovitz-Eldor J, Shapiro SS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–7. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- Tsai RJ, Sun TT, Tseng SC. Comparison of limbal and conjunctival autograft transplantation in corneal surface reconstruction in rabbits. Ophthalmology. 1990;97:446–55. doi: 10.1016/s0161-6420(90)32575-7. [DOI] [PubMed] [Google Scholar]
- Tsai RJ, Tseng SC. Human allograft limbal transplantation for corneal surface reconstruction. Cornea. 1994;13:389–400. doi: 10.1097/00003226-199409000-00003. [DOI] [PubMed] [Google Scholar]
- Tsai RJ, Li LM, Chen JK. Reconstruction of damaged corneas by transplantation of autologous limbal epithelial cells. N Engl J Med. 2000;343:86–93. doi: 10.1056/NEJM200007133430202. [DOI] [PubMed] [Google Scholar]
- Tsubota K, Toda I, Saito H, et al. Reconstruction of the corneal epithelium by limbal allograft transplantation for severe ocular surface disorders. Ophthalmology. 1995;102:1486–96. doi: 10.1016/s0161-6420(95)30841-x. [DOI] [PubMed] [Google Scholar]
- Tsubota K, Satake Y, Ohyama M, et al. Surgical reconstruction of the ocular surface in advanced ocular cicatricial pemphigoid and Stevens-Johnson syndrome. Am J Ophthalmol. 1996;122:38–52. doi: 10.1016/s0002-9394(14)71962-2. [DOI] [PubMed] [Google Scholar]
- Tsubota K, Satake Y, Kaido M, et al. Treatment of severe ocular surface disorders with corneal epithelial stem-cell transplantation. N Engl J Med. 1999;340:1697–1703. doi: 10.1056/NEJM199906033402201. [DOI] [PubMed] [Google Scholar]
- Ueno H, Kayama K, Homma R, et al. 2006Experimental transplantation of corneal epithelial cells derived from mouse embryonic stem (ES) cells transfected with Pax6 gene Invest Ophthalmol Vis Sci 47E–Abstract 1109. [Google Scholar]
- Walther C, Gruss P. Pax-6, a murine paired box gene, is expressed in the developing CNS. Development. 1991;113:1435–49. doi: 10.1242/dev.113.4.1435. [DOI] [PubMed] [Google Scholar]
- Watanabe K, Nishida K, Yamato M, et al. Human limbal epithelium contains side population cells expressing the ATP-binding cassette transporter ABCG2. FEBS Lett. 2004;565:6–10. doi: 10.1016/j.febslet.2004.03.064. [DOI] [PubMed] [Google Scholar]
- Wolosin JM, Budak MT, Akinci MA. Ocular surface epithelial and stem cell development. Int J Dev Biol. 2004;48:981–91. doi: 10.1387/ijdb.041876jw. [DOI] [PubMed] [Google Scholar]
- Zhou S, Schuetz JD, Bunting KD, et al. The ABC transporter Bcrp1/ABCG2 is expressed in a wide variety of stem cells and is a molecular determinant of the side-population phenotype. Nat Med. 2001;7:1028–34. doi: 10.1038/nm0901-1028. [DOI] [PubMed] [Google Scholar]