To-date, all methods to generate induced pluripotent stem (iPS) cells require the use of genetic materials and/or potentially mutagenic molecules. Here we report the generation of stable iPS cells from human fibroblasts by directly delivering four reprogramming proteins (Oct4, Sox2, Klf4, and c-Myc) fused with a cell penetrating peptide (CPP). These protein-induced human iPS (p-hiPS) cells exhibited similarity to human embryonic stem (hES) cells in morphology, proliferation, and expression of characteristic pluripotency markers. p-hiPS cell lines produced with these recombinant proteins were successfully maintained for more than 35 passages and differentiated into derivatives of all three embryonic germ layers both in vitro and in teratomas. This system eliminates the potential risks associated with the use of viruses, DNA transfection, and potentially harmful chemicals, and in the future could potentially provide a safe source of patient-specific cells for regenerative medicine.
Over a decade ago, Wilmut and colleagues showed that adult somatic cells could be reprogrammed back to an undifferentiated embryonic state using somatic cell nuclear transfer (SCNT)(Wilmut et al., 1997). However, since that time attempts to generate patient-specific cells using SCNT have proven unsuccessful (Chung et al., 2009; French et al., 2008). In 2006, a new and less controversial method of reprogramming somatic cells to pluripotency was reported by viral expression of the transcription factors Oct4, Sox2, Klf4 and c-Myc (Takahashi and Yamanaka, 2006). This and subsequent studies confirmed that mouse and human somatic cells can be reprogrammed to the pluripotent state via viral transduction with the same or similar sets of reprogramming factors (Maherali et al., 2007; Okita et al., 2007; Park et al., 2008; Takahashi et al., 2007; Wernig et al., 2007; Yu et al., 2007). Although the therapeutic potential of iPS cells has been demonstrated in animal models of sickle cell anemia and Parkinson's disease (Hanna et al., 2007; Wernig et al., 2008), these cells contain multiple viral vector integrations that make them unsuitable for human clinical trials. The use of genome-integrating viruses could cause insertional mutagenesis and unpredictable genetic dysfunction (Okita et al., 2007; Yamanaka, 2007).
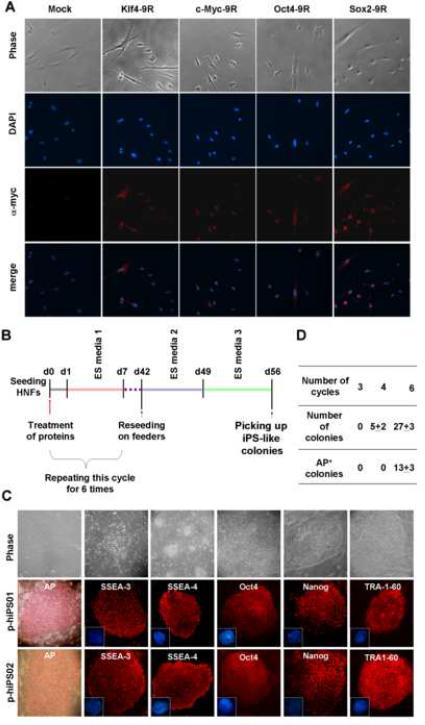
To address whether it is possible to generate human iPS cells without the use of viral or DNA vectors, we attempted to deliver four reprogramming proteins - Oct4, Sox2, Klf4, and c-Myc - directly into somatic cells. A major hurdle for intracellular delivery of macromolecules such as proteins is their limited ability to cross the cellular membrane (Belting et al., 2005). In 1988, Frankel and Pabo found that the human immunodeficiency virus transactivator of transcription (HIV-TAT) protein can overcome this hurdle with a short basic segment residing at amino acid 48-60 that allows this protein to penetrate the cell membrane and activate HIV-specific genes (Frankel et al., 1988; Frankel and Pabo, 1988). This and other naturally occurring peptides capable of overcoming the cell membrane barrier contain a high proportion of basic amino acids (e.g., arginine or lysine) and are known as CPP (El-Sayed et al., 2009; Ziegler et al., 2005). In order to test our hypothesis that CPP-anchored reprogramming proteins may directly reprogram human somatic cells without genetic manipulation and/or chemical treatments, we first examined whether red fluorescent protein (RFP) fused with a 9 arginine (RFP-9R) (Wender et al., 2000) could penetrate into COS7 cells and human newborn fibroblasts (HNFs). RFP-9R was efficiently delivered into both cell types within a few hours, even when in the context of whole cell extracts (Fig. S1). We then generated stable HEK293 cell lines that could express each of the four human reprogramming factors (Oct4, Sox2, Klf4, and c-Myc) fused with 9R and the myc tag. High expression of these proteins was confirmed in HEK 293 cell lines by Western blotting analyses (Fig. S2). When HNFs were treated with cell extracts from the HEK 293 cell lines, efficient intracellular translocation of each recombinant protein was observed within 8 hours (Fig. 1a). Notably, in contrast to RFP-9R, which was translocated to the cytoplasm, it appeared that most recombinant reprogramming proteins were translocated to the nucleus while some remained in the cytoplasm (Fig. 1a and Fig. S1).
Fig. 1. Generation of protein-induced hiPS cell lines by direct delivery of reprogramming proteins fused with 9R as a CPP.
A, HNFs were incubated with HEK 293 extracts expressing each reprogramming protein and subjected to immunocytochemistry using myc antibodies. Nuclei were counterstained with DAPI. B, The schematic protocol depicts a repeated process and the time line for generating p-hiPS cells from HNFs. C, Top panel: starting HNFs (first image); morphology after 3 cycle protein treatments (second image); and increased colony number after 6 cycles (third image). Approximately half of these iPS-like colonies stained positive for AP; early morphology after p-hiPS colonies were transferred to MEF (fourth image); and morphology of established p-hiPS cell line at passage number 10 (p-hiPS01 [fifth image] and p-hiPS02 [sixth image]). Immunostaining of p-hiPS01 (middle panel) and p-hiPS02 (bottom panel) clones show expression of hESC markers, including AP, SSEA-3, SSEA-4, Oct-4, Nanog, and TRA-1-60. Nuclei were stained with DAPI (blue in second and third row panel). D, Efficiency of reprogrammed colony formation with iPS-like morphology and AP-positive staining after different numbers of the protein treatment cycle. This is the summary of three independent experiments with the standard error.
In an initial series of experiments, 5×105 HNFs were treated with combined total extracts of four HEK 293 cell lines for 16 hours (See Protocol 1 in Fig. S3). After washing, cells were incubated for 6 days in ES Media 1 and then transferred onto mouse embryonic feeders (MEF). The transferred cells were incubated with ES Media 2 for up-to 4 weeks. Despite numerous attempts, we did not observe the formation of reprogrammed colonies using this protocol. We next treated with the same total extracts for 16 hours followed by washing and incubation with ES Media 1 for 8 hr/day for 6 days (Protocol 2 in Fig. S3). By day 7, most cells did not survive, and no colonies formed after further incubation on MEF. One potential reason for the lack of success is that, in contrast to virus- or other DNA-based methods, the reprogramming factors were not provided continuously and thus were in short supply. Therefore, we tested whether repeated protein treatment cycles (16 hr protein treatment followed by 6 d incubation in ES Media 1) (Fig. 1b) could yield hiPS cells. Using this approach, after three or four rounds of treatment several colonies with iPS-like morphology were observed (Fig. 1c), although none of these colonies showed alkaline phosphatase (AP) activity, suggesting only rudimentary reprogramming. When this procedure was repeated for further cycles, the number of iPS-like colonies significantly increased and approximately half of the resulting colonies were AP-positive starting from the 6th cycle (Fig. 1d). In contrast, no such colonies were formed at any stage when extracts of naïve HEK 293 cells were used. AP-positive colonies with iPS-like morphology were handpicked and transferred onto MEFs in the presence of ES Media 2 and Media 3 for 7 days each. Five hiPS-like colonies were established, and two of them were maintained and characterized in this study. These two cell lines (p-hiPS01 and p-hiPS02) have been successfully maintained for more than 35 passages and exhibit morphology similar to that of hES cells, characterized by large nuclei and scant cytoplasm (Fig. 1c). Overall, the establishment of these hiPS-like colonies took about 8 weeks, approximately double that seen with viral transduction (Park et al., 2008; Takahashi et al., 2007; Yu et al., 2007). At present, the efficiency of iPS generation is significantly lower using this protein-based protocol (about 0.001% of input cells; Fig. 1d), compared to virus-based protocols (about 0.01% of input cells) (Park et al., 2008; Takahashi et al., 2007; Yu et al., 2007).
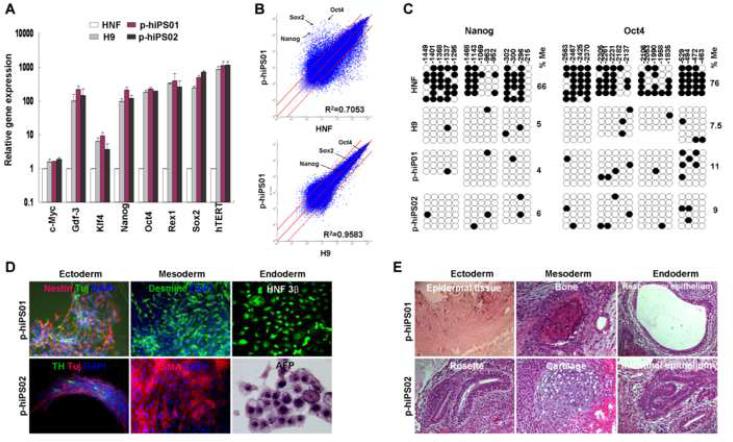
In order to determine whether the p-hiPS cells have hESC-like properties we examined them for expression of markers of pluripotency. As shown in Fig. 1c, both cell lines prominently expressed ES markers, including AP, Oct4, Nanog, tumor-rejection antigen (TRA)1-60, stage-specific embryonic antigen (SSEA) 3 and SSEA 4. Quantitative reverse transcription PCR (qRT-PCR) analysis confirmed that both lines expressed endogenous mRNAs of ES cell markers: Oct4, Nanog, Sox2, reduced expression 1 (Rex1), growth and differentiation factor 3 (Gdf3), and telomerase reverse transcriptase (hTERT) levels were dramatically higher than those of HNF cells (up-to 100-fold, and comparable to hES cells)(Fig. 2a). The expression patterns of ES pluripotency markers were indistinguishable from hES (H9) cells, strongly suggesting that appropriate epigenetic reprogramming had occurred in the p-hiPS cells. Bisulfite sequencing analyses further showed that the promoter regions of the pluripotency genes Nanog and Oct4 were significantly demethylated in both p-hiPS lines and the hES H9 line, whereas the same regions were densely methylated in the parental HNF cells (Fig. 2c). hiPS lines from the starting HNFs were also generated using retroviral vectors expressing the same four reprogramming factors (Park et al., 2008; Takahashi et al., 2007; Yu et al., 2007). These cells displayed similar characteristics and properties as the p-hiPS cells (Fig. S4) and one of them (rv-hiPS01) was used as a control. When global gene expression was compared using the Affymetrix Array U133 Plus 2.0 analyzing over 47,000 human transcripts, both p-hiPS and rv-hiPS01 showed high similarity to hES H9, but not to HNFs (Fig. 2b, Fig. S5). Since hES H9 and rv-hiPS01 were used as control cell lines, it was important to rule out the possibility that the new p-hiPS cells were derived from contaminating cells. RT-PCR analyses detected the all four transgene mRNAs in rv-hiPS01 cells, but not in p-hiPS01 and p-hiPS02 cell lines (Fig. S6). Furthermore, DNA fingerprinting demonstrated that the patterns of both p-hiPS lines and rv-hiPS01 cells were identical to the parental HNF cells, but different from those of the hES (H9) cells and HEK293 cells (Fig. S7), thus confirming that both p-hiPS lines are derived from HNF cells. Both p-hiPS lines exhibited the same karyotype as the starting HNF cells (Fig. S8).
Fig. 2. Characterization of p-hiPS cell lines.
A, Quantitative RT-PCR was performed to assess the expression of c-Myc, Gdf-3, Klf4, Nanog, Oct4, Rex1, Sox2, and hTERT, in p-hiPS01 and p-hiPS02, hES (H9), and HNF cells. Relative gene expression represents fold changes relative to that of HNF cells normalized to β-actin expression. This experiment (repeated twice in triplicate using independently prepared cDNAs) resulted in almost identical patterns. B, The global gene-expression patterns were compared between p-hiPS01 and HNF, and between p-hiPS01 and H9 with Affymetrix microarrays. The red lines indicate the diagonal and 5-fold changes between the paired samples. C, Bisulfite sequencing analysis of the Nanog and Oct4 promoters reveal that almost complete epigenetic reprogramming. Open and closed circles indicate unmethylated and methylated CpG, respectively. Numbers on top show each CpG location. Percentages of CpG methylation (%Me) are shown. D. In vitro differentiation of p-hiPS cells. EBs were made by suspension culture of both p-hiPS lines at day 8 (Far left of the top row panels). Phase contrast (Top row panels) and immunostaining images (second and third row panels) show all three germ layer cells at day 24 including neural (ectodermal), muscle and endothelial-like (mesodermal), and endoderm-like cells (endoderm). E, Teratoma formation in immunodeficiency mice by p-iPS cells. H&E staining were performed for teratomas. The resulting teratomas contained tissues representing all three germ layers: (p-hiPS01, fourth row; and p-hiPS02, bottom row). Ectoderm: epidermal and neural tissue (rosette); mesoderm: bone and cartilage; endoderm: respiratory epithelium and intestinal-like epithelium.
When the p-hiPS cells were allowed to form embryoid bodies (EBs) by suspension culture, they readily differentiated into cells of all three germ layers (Fig. 2d; Fig. S8). After 8 days, well-formed EB structures were observed from both p-iPS clones. When these EB-like structures were incubated on gelatin-coated tissue culture plates in ITSFn media for 15 to 25 days, they differentiated to a wide range of cell types, including neural, muscle, and endodermal cells, among others. Immunocytochemical analyses demonstrated the existence of different cell types positive for hepatocyte necrosis factor 3β (HNF 3β, endoderm marker), alpha-fetoprotein (AFP, endoderm marker), smooth-muscle actin (SMA, mesoderm marker), desmin (mesoderm marker), Tuj1 (ectoderm marker), nestin (ectoderm marker), and tyrosine hydroxylase (TH, ectoderm marker) (Fig. 2d; Fig. S8). In addition, teratoma formation was observed after transplantation of p-hiPS cells under the kidney capsule of nude mice for 6 to 8 weeks. These teratomas contained tissues from all three germ layers including neural tissues (ectoderm), epidermal tissues (ectoderm), striated muscle (mesoderm), adipose tissue (mesoderm), cartilage (mesoderm), respiratory epithelium (endoderm), and intestinal-like epithelial tissues (endoderm) (Fig 2e; Fig. S8), confirming that both p-hiPS clones exhibit pluripotency both in vitro and in vivo.
Protein-based hiPS technology offers a new and potentially safe method for generating patient-specific stem cells that does not require the destruction of ex utero embryos. This system completely eliminates genome manipulation and DNA transfection, resulting in human iPS cells suitable for drug discovery, disease modeling, and future clinical translation. In this regard, the present study demonstrates the “proof-of-concept” that human iPS cells can be generated by direct protein delivery without genetic manipulation. Other studies suggest that it may be possible to replace and/or further reduce the number of factors required for reprogramming (Huangfu et al., 2008; Li et al., 2009; Shi et al., 2008). To minimize/avoid chromosomal disruption, adenovirus and plasmid transfection have been successfully used to generate iPS cells in the mouse system (Kaji et al., 2009; Okita et al., 2008). Also, Thomson and his colleagues reported generation of hiPS cells by transfection with non-integrating episomal vectors (Yu et al., 2009). In addition, piggyBac transposon (Kaji et al., 2009; Woltjen et al., 2009) and Cre-recombinase excisable viruses (Soldner et al., 2009) have been used to generate hiPS cells. While the transgenes can be excised by inducible gene expression once reprogramming is established (Soldner et al., 2009; Stadtfeld et al., 2008; Woltjen et al., 2009), residual sequences and chromosomal disruptions may still result in harmful alterations that could pose clinical risks.
The DNA vector-free, direct protein transduction system described here eliminates limitations that may be caused by viral or any other DNA-based reprogramming methods. However, the generation of p-hiPS cells is very slow and inefficient, and requires further optimization. In particular, the whole protein extracts used in the present study limited the concentrations of factors delivered into the target cells, thus suggesting that p-hiPS cells may be more efficiently generated using purified reprogramming proteins. Recently, Ding and his colleagues reported the generation of mouse iPS cells by combining the use of recombinant reprogramming proteins and the small molecule valproic acid (Zhou et al., 2009). In this study, mouse iPS cells were not generated when only recombinant proteins were used. In contrast, the system described here generated human iPS cells with direct delivery of reprogramming proteins in the absence of any chemical treatment. One possible explanation for these differences is that we used reprogramming proteins expressed in mammalian cells while Ding and colleagues used refolded proteins after expression in E. coli. Since chemicals such as valproic acid and/or genetic manipulation may induce mutations, it has been suggested that whole genomic sequencing would be necessary if such methods are used to generate iPS cells (Yamanaka, 2009). In conclusion, the system described here eliminates the potential risks associated with chromosomal integrations and/or mutations, and may allow the translation of hiPS cell technology into the clinic.
Supplementary Material
ACKNOWLEDGEMENTS
This work was supported by NIH grants MH48866 and DC 006501 and International Grants from the CHA University, Korean Stem Cell Research Center, and Dongyang Corporation Co. in Korea. The authors thank Dr. V. Morgan at the Harvard Partners Center for Genetics and Genomics, Dr. J. Kim (Harvard Medical School) and Dr. J. Lee (Shippensburg University) for microarray analysis, and Ms. J. Johnson (Cell Line Genetics) for karyotyping analysis.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- Belting M, Sandgren S, Wittrup A. Nuclear delivery of macromolecules: barriers and carriers. Adv Drug Deliv Rev. 2005;57:505–527. doi: 10.1016/j.addr.2004.10.004. [DOI] [PubMed] [Google Scholar]
- Chung Y, Bishop CE, Treff NR, Walker SJ, Sandler VM, Becker S, Klimanskaya I, Wun WS, Dunn R, Hall RM, et al. Reprogramming of Human Somatic Cells Using Human and Animal Oocytes. Cloning Stem Cells. 2009 doi: 10.1089/clo.2009.0004. [DOI] [PubMed] [Google Scholar]
- El-Sayed A, Futaki S, Harashima H. Delivery of Macromolecules Using Arginine-Rich Cell-Penetrating Peptides: Ways to Overcome Endosomal Entrapment. Aaps J. 2009 doi: 10.1208/s12248-008-9071-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frankel AD, Bredt DS, Pabo CO. Tat protein from human immunodeficiency virus forms a metal-linked dimer. Science. 1988;240:70–73. doi: 10.1126/science.2832944. [DOI] [PubMed] [Google Scholar]
- Frankel AD, Pabo CO. Cellular uptake of the tat protein from human immunodeficiency virus. Cell. 1988;55:1189–1193. doi: 10.1016/0092-8674(88)90263-2. [DOI] [PubMed] [Google Scholar]
- French AJ, Adams CA, Anderson LS, Kitchen JR, Hughes MR, Wood SH. Development of human cloned blastocysts following somatic cell nuclear transfer with adult fibroblasts. Stem Cells. 2008;26:485–493. doi: 10.1634/stemcells.2007-0252. [DOI] [PubMed] [Google Scholar]
- Hanna J, Wernig M, Markoulaki S, Sun CW, Meissner A, Cassady JP, Beard C, Brambrink T, Wu LC, Townes TM, et al. Treatment of sickle cell anemia mouse model with iPS cells generated from autologous skin. Science. 2007;318:1920–1923. doi: 10.1126/science.1152092. [DOI] [PubMed] [Google Scholar]
- Huangfu D, Osafune K, Maehr R, Guo W, Eijkelenboom A, Chen S, Muhlestein W, Melton DA. Induction of pluripotent stem cells from primary human fibroblasts with only Oct4 and Sox2. Nat Biotechnol. 2008;26:1269–1275. doi: 10.1038/nbt.1502. [DOI] [PubMed] [Google Scholar]
- Kaji K, Norrby K, Paca A, Mileikovsky M, Mohseni P, Woltjen K. Virus-free induction of pluripotency and subsequent excision of reprogramming factors. Nature. 2009 doi: 10.1038/nature07864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W, Wei W, Zhu S, Zhu J, Shi Y, Lin T, Hao E, Hayek A, Deng H, Ding S. Generation of rat and human induced pluripotent stem cells by combining genetic reprogramming and chemical inhibitors. Cell Stem Cell. 2009;4:16–19. doi: 10.1016/j.stem.2008.11.014. [DOI] [PubMed] [Google Scholar]
- Maherali N, Sridharan R, Xie W, Utikal J, Eminli S, Arnold K, Stadtfeld M, Yachechko R, Tchieu J, Jaenisch R, et al. Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell. 2007;1:55–70. doi: 10.1016/j.stem.2007.05.014. [DOI] [PubMed] [Google Scholar]
- Okita K, Ichisaka T, Yamanaka S. Generation of germline-competent induced pluripotent stem cells. Nature. 2007;448:313–317. doi: 10.1038/nature05934. [DOI] [PubMed] [Google Scholar]
- Okita K, Nakagawa M, Hyenjong H, Ichisaka T, Yamanaka S. Generation of mouse induced pluripotent stem cells without viral vectors. Science. 2008;322:949–953. doi: 10.1126/science.1164270. [DOI] [PubMed] [Google Scholar]
- Park IH, Zhao R, West JA, Yabuuchi A, Huo H, Ince TA, Lerou PH, Lensch MW, Daley GQ. Reprogramming of human somatic cells to pluripotency with defined factors. Nature. 2008;451:141–146. doi: 10.1038/nature06534. [DOI] [PubMed] [Google Scholar]
- Shi Y, Desponts C, Do JT, Hahm HS, Scholer HR, Ding S. Induction of pluripotent stem cells from mouse embryonic fibroblasts by Oct4 and Klf4 with small-molecule compounds. Cell Stem Cell. 2008;3:568–574. doi: 10.1016/j.stem.2008.10.004. [DOI] [PubMed] [Google Scholar]
- Soldner F, Hockemeyer D, Beard C, Gao Q, Bell GW, Cook EG, Hargus G, Blak A, Cooper O, Mitalipova M, et al. Parkinson's disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell. 2009;136:964–977. doi: 10.1016/j.cell.2009.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stadtfeld M, Nagaya M, Utikal J, Weir G, Hochedlinger K. Induced pluripotent stem cells generated without viral integration. Science. 2008;322:945–949. doi: 10.1126/science.1162494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- Wender PA, Mitchell DJ, Pattabiraman K, Pelkey ET, Steinman L, Rothbard JB. The design, synthesis, and evaluation of molecules that enable or enhance cellular uptake: peptoid molecular transporters. Proc Natl Acad Sci U S A. 2000;97:13003–13008. doi: 10.1073/pnas.97.24.13003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, Hochedlinger K, Bernstein BE, Jaenisch R. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature. 2007;448:318–324. doi: 10.1038/nature05944. [DOI] [PubMed] [Google Scholar]
- Wernig M, Zhao JP, Pruszak J, Hedlund E, Fu D, Soldner F, Broccoli V, Constantine-Paton M, Isacson O, Jaenisch R. Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson's disease. Proc Natl Acad Sci U S A. 2008;105:5856–5861. doi: 10.1073/pnas.0801677105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilmut I, Schnieke AE, McWhir J, Kind AJ, Campbell KH. Viable offspring derived from fetal and adult mammalian cells. Nature. 1997;385:810–813. doi: 10.1038/385810a0. [DOI] [PubMed] [Google Scholar]
- Woltjen K, Michael IP, Mohseni P, Desai R, Mileikovsky M, Hamalainen R, Cowling R, Wang W, Liu P, Gertsenstein M, et al. piggyBac transposition reprograms fibroblasts to induced pluripotent stem cells. Nature. 2009 doi: 10.1038/nature07863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamanaka S. Strategies and new developments in the generation of patient-specific pluripotent stem cells. Cell Stem Cell. 2007;1:39–49. doi: 10.1016/j.stem.2007.05.012. [DOI] [PubMed] [Google Scholar]
- Yamanaka S. A fresh look at iPS cells. Cell. 2009;137:13–17. doi: 10.1016/j.cell.2009.03.034. [DOI] [PubMed] [Google Scholar]
- Yu J, Hu K, Smuga-Otto K, Tian S, Stewart R, Slukvin II, Thomson JA. Human Induced Pluripotent Stem Cells Free of Vector and Transgene Sequences. Science. 2009 doi: 10.1126/science.1172482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, Nie J, Jonsdottir GA, Ruotti V, Stewart R, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318:1917–1920. doi: 10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- Zhou H, Wu S, Joo JY, Zhu S, Han DW, Lin T, Trauger S, Bien G, Yao S, Zhu Y, et al. Generation of Induced Pluripotent Stem Cells Using Recombinant Proteins. Cell Stem Cell. 2009 doi: 10.1016/j.stem.2009.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler A, Nervi P, Durrenberger M, Seelig J. The cationic cell-penetrating peptide CPP(TAT) derived from the HIV-1 protein TAT is rapidly transported into living fibroblasts: optical, biophysical, and metabolic evidence. Biochemistry. 2005;44:138–148. doi: 10.1021/bi0491604. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.