Abstract
Chronic exposure to the copper-chelating agent, cuprizone (CPZ), is an increasingly popular model for producing demyelination. More importantly, cessation of cuprizone exposure allows for full remyelination, which represents a window of opportunity for determining the influence of environmental factors on regenerative processes. In the present study, CPZ-treated animals were assessed for functional status of systemic and central cytokine responsiveness to LPS, as well as assessment for signs of body weight changes. Exposure of male C57BL/6J mice to 5 weeks of 0.2% CPZ in the diet was optimal in producing demyelination and microglial activation, as measured by myelin basic protein, CD11b, and CD45 immunohistochemistry. Acute challenge with LPS at the end of 5 weeks CPZ treatment did not alter IL-1β, IL-6, nor TNFα responses in the spleen and corpus callosum. Similarly, repeated exposure to LPS during the remyelination phase (CPZ removal) did not influence these measures to LPS. Plasma corticosterone was unaffected following acute challenge of CPZ-pretreated animals, but after repeated LPS treatment, there was a significant augmentation of the corticosterone response in CPZ-pretreated mice. Interestingly, the basal concentration of IL-1β in the corpus callosum of CPZ treated animals was significantly increased, which was in keeping with the increase in activated microglial cells. In conclusion, the cuprizone model of demyelination and remyelination does not appear to influence the systemic nor central IL-1, IL-6 and TNF responses to acute nor repeated LPS. This opens up the possibility for studying the contribution of systemic inflammatory processes on remyelination after cessation of CPZ treatment.
1. Introductions
Multiple Sclerosis (MS) is a progressive, neurodegenerative disease of the central nervous system (CNS). It occurs most often in a relapsing/remitting form, in which a period of demyelination is followed by a period of functional recovery. The stage of recovery involves the generation of new oligodendrocytes, but as the disease progresses the ability to regenerate these cells fails and there is continuous pathology with loss of function (Keirstead & Blakemore, 1999). The etiology of this disease remains unclear, although infectious agents such as Epstein-Barr virus (EBV), human herpes virus 6 (HHV-6), and Chlamydia pneumoniae have been linked to MS (Hunter and Hafler, 2000), in addition to genetic susceptibility and cell-mediated autoimmunity (Oksenberg & Barcellos 2005; Huseby et al., 2001; Ewing & Bernard 1998).
The most popular model for investigating MS and/or demyelinating disease is experimental allergic encephalomyelitis (EAE), wherein the focus is on autoreactive T lymphocytes, CNS infiltrating leukocytes and cytokine production (Vipin 2004). However, while EAE focuses on immunologically-induced demyelination and neurological impairment, alternative neurotoxicant-based models have been utilized to induce demyelination and, upon removal of the neurotoxicant, initiate a process of oligdendrocyte regeneration and axon remyelination (Matushima & Morell 2001; Blakemore 2005; Crang et al., 2004; Franklin and Goldman 2004). Although pathology in these latter models does not involve an autoimmune etiology, the contribution of cytokines to demyelination and remyelination has been well demonstrated. For example, using the neurotoxicant cuprizone (bis-cyclohexanone oxaldihydrazone), it was shown that demyelination and remyelination are dependent on tumor necrosis factor (TNFα) and interferon-λ (IFNλ) (Arnett et al., 2001; Mason et al., 2003), and that the conditional inhibition of suppressor of cytokine signaling – 3 (SOCS-3) protects against cuprizone-induced demyelination (Emery et al., 2006). This demonstrates that the cuprizone model provides a useful approach to studying the basic cytokine mechanisms underlying demyelination and remyelination, as well as the impact of environmental factors on these processes.
Cuprizone-induced demyelination occurs after several weeks of ingesting a cuprizone-containing diet. Interestingly, demyelination is regionally selective, with appreciable loss of myelin confined to anterior extent of the corpus callosum, as well as the fornix (Hiremath et al., 1998). The reasons for this are not understood. Similarly, the mechanism of action by which demyelination occurs is still unclear, but may rely on the copper-chelating properties of cuprizone (Matushima & Morell 2001). Alternatively, in vitro addition of cuprizone fails to be deleterious to cultured oligodendrocytes in the absence of cytokine-producing microglial cells (Pasquini et al., In Press), suggesting that the oligodendrocyte-targeted toxicity of cuprizone relies on local inflammatory processes. Perhaps the most promising aspect of this model is that demyelination is reversible, since removal of short term exposure to cuprizone from the diet leads to the full restoration of a mature oligodendrocyte population (Mason et al., 2004). However, during this period of recovery it is likely that the CNS is particularly vulnerable to environmental stressors that might influence the success of myelin restoration and functional recovery.
It is well known that immunological stimulation provokes a series of behavioral symptoms referred to collectively as “sickness behavior,” which is characterized by anorexia, lethargy, anhedonia, and alterations in sleep and sexual patterns (Danzer 2001). As such the immune response can be viewed as a stressor that in addition to these behavioral adjustments produces a range of neurochemical and endocrine alterations typically induced by psychogenic stressors (Anisman et al., 2002). A particularly potent immunologic stressor is lipopolysaccharide (LPS), which strongly stimulates circulating monocytes and tissue macrophages to produce a variety of proinflammatory cytokines, including interleukin-1β (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor α (TNFα). These cytokines mediate sickness behavior by increasing neurotransmitter release, immediate early gene induction, and activating the hypothalamic-pituitary-adrenal (HPA) axis (MohanKumar et al., 1999; Wan et al., 1994; Dunn et al., 1999)
Exposure to LPS has also been shown to exacerbate neuropathology in a number of disease models (Castano et al., 1998; Cunningham et al., 2005; Brugg et al., 1995). For example, it has been hypothesized that activated brain microglia are primed during disease states to respond to further insult with immune stressors such as LPS (Perry 2004). This has been demonstrated in animal models of prion disease as well as Alzheimer’s disease, in which systemic LPS augmented brain levels of IL-1, relative to healthy animals challenged with LPS (Cunningham et al., 2005; Sly et al., 2001). Similarly, in demyelinating diseases such as MS, exacerbated symptoms and prolonged impairment is associated with systemic infection (Buljevac et al., 2002; review Perry et al., 2004). This may result in central inflammatory events that exacerbate the level of damage during the early relapsing remitting phase of MS and affect prognosis (Bitsch et al., 2000). However, the mechanisms by which infection and the presence of inflammatory cells may influence CNS lesions in demyelinating diseases is not clear, although the possibility of molecular mimicry and/or bystander effects in the presence of inflammatory mediators remains a viable hypothesis (Martino et al, 2000; McCoy et al, 2006).
The foregoing observations involve inductive models of antigen-specific immune responses. However, there are few models of systemic inflammatory effects on neuropathology induced by non-immunogenic models of demyelination. To this end we chose to use the cuprizone model of demyelination and tested the feasibility of using LPS as a model of systemic inflammation. Little information exists on the cytokine-inducing effects of LPS in cuprizone treated animals, a critical gap in knowledge that needs to be addressed prior to implementing studies of whether peripheral inflammatory processes induced by LPS may impact the remyelinating phase of recovery from a demyelinated state. That is, such approaches would not be possible in the face of possible cuprizone-induced suppression of cytokine production. Therefore, the current study subjected cuprizone-fed and unfed C57BL/6J mice to acute and repeated regimens of systemic LPS exposure and assessed for illness-related body weight changes, activation of the HPA axis, and splenic and brain production of IL-1β, IL-6 and TNFα. The results showed little impairment of the proinflammatory response to LPS both systemically and in the brain. However, cuprizone-fed animals displayed significantly elevated corticosterone responses to a regimen of treatment that otherwise produced LPS tolerance in animals not treated with cuprizone.
2. Materials and Methods
2.1. Animals
Male C57BL/6J mice were purchased from The Jackson Laboratory (Bar Harbor, ME) at 5–6 weeks of age and housed four per cage under 12:12 hr light:dark illumination (lights on 0600h). Animals were fed for five weeks ad libitum milled mouse chow with or without 0.2% cuprizone. All experiments were conducted in accordance with the guidelines of the Animal Care and Use Committee of Rutgers University
2.2. Reagents
Lipopolysaccharide (LPS: from strain 055:B5) and cuprizone were purchased from Sigma-Aldrich (St. Louis, MO). Biotinylated rat anti-mouse CD11b antibody was used at a dilution of 1:1000 (Serotec, Raleigh, NC), rat anti-mouse CD45 was used at a 1:2000 dilution (Serotec, Raleigh, NC), and anti-rat MBP antibody was used at a dilution of 1:1000 (Abcam, Cambridge, MA).
2.3. MBP, CD11b, and CD45 Immunohistochemistry
2.3.1. Perfusion and Sectioning for Immunochemistry
To validate that the CPZ model did indeed produce demyelination, MBP was assessed after 3 and 5 weeks continuous exposure to CPZ, as well as 2 wks after removal of CPZ that had been given for 5 weeks (2 week off group) (N=3–4/group). Additionally, to confirm microglial activation, the number of CD45+ and CD11b+ cells was measured at the end of 5 weeks of CPZ treatment, and 1 and 2 weeks after removal (1 and 2 week off groups) (N= 3–4/group). For perfusion, animals were lethally anesthetized with 50 mg/Kg sodium pentobarbital and sacrificed via transcardial perfusion for 5-minutes with isotonic saline, followed by 10 minutes with 4% paraformaldehyde, and a 5-minute wash with isotonic saline. Brains were post-fixed overnight in 4% paraformaldehyde and then placed in 30% sucrose solution until sectioning. Coronal brain sections of 30 μm thickness were made on a freezing microtome and cryopreserved at −20°C as free-floating sections. Sections were restricted to the fornix region of the corpus callosum, using guidance from a mouse stereotaxic atlas (Franklin & Paxinos, 1997).
2.3.2. Immunocytochemistry
Tissue sections were incubated initially for 48 hours with rabbit anti-rat MBP (1:1000), 72 hrs for rat anti-mouse CD11b (1:1000) and 72 hrs for rat anti-mouse CD45 (1:2000) at 4°C in 0.4%Triton X-100 in KPBS, pH7.2. For verification of the staining specificity of the MBP antibody, Rabbit IgG (Sigma, St. Louis, MO) was used at 1:1000 dilution to serve as an isoptype control. Rat IgG (Sigma, Louis, MO) was used to verify staining specificity for CD11b (1:1000) and CD45 (1:2000). After rinsing in Potassium phosphate buffered saline (KPBS), the tissues were incubated for 2h at room temperature in KPBS/0.4% Triton X-100 containing a 1:500 final dilution of biotinylated goat anti-rabbit IgG or goat anti-rat IgG (Vector Laboratories, Burlingame, CA). After an additional rinse, tissues were treated for 1h at room temperature with a avidin–biotin–peroxidase complex solution from the Vector Elite ABC kit (Vector Laboratories), followed by sequential washes in PBS and 0.175 M sodium acetate (NaOAc). The enzyme–substrate reaction was subsequently generated by the addition of a 3,3-diaminobenzadine (DAB) substrate solution consisting of 0.175 M NaOAc containing 25 mg/ml NiSO4, 0.2 mg/ml DAB, and 0.28% H2O2. For CD11b detection, NiSO4 was added to allow for dark blue to black stains, whereas for MBP immunocytochemistry, it was excluded to allow for a brown reaction. Termination of the enzyme-substrate reaction was achieved by rinsing the tissues in 0.175 M NaOAc and then in KPBS, after which the tissues was mounted on Superfrost Plus slides (Fisher Scientific), dehydrated in a graded series of alcohols (or 1-butanol for MBP), and clarified and coverslipped using Histoclear and Histomount (VWR Scientific, Westchester, PA). The sections stained for CD11b were counterstained with 1% neutral red while the sections stained for MBP were counterstained with methyl green. For quantitation of immunoreactive cells, stained slides were examined under a Nikon Eclipse E400 microscope equipped with a high resolution CCD camera. Neuroanatomically distinct regions, as defined by the mouse atlas of Franklin and Paxinos (1997), were digitally captured, and immunopositive cells were enumerated using the NIH Image software program. The region of interest for analysis of immunopositve cells was always the midline of the corpus callosum. Sections corresponded to Bregma −0.46 mm to −1.46 mm. This represented a 1mm area. Four sections per animal were counted, with each representing points 0.25 mm apart along the anterior to posterior extent from these bregma points. On each slide, counting was restricted to the region located 0.5 mm lateral from the midline and 1.75 mm ventral. The minimum pixel size for a cell to be counted was 5 pixels and the maximum size was 35 pixels. The threshold for each image was determined separately for each individual image. Each section was counted 3 times and a total of four sections per animal were counted and averaged. Because thresholding the image was different for each image, sections were randomly hand counted for verification. One section from each of six animals was randomly chosen for hand counting and correspondence to the software was established. Confirmation of software accuracy was conducted by two observers, blind to treatment, hand-counting selected regions of interest. Variation in number between human and software counts were 1–2%.
2.4. LPS Injection protocol
At the end of 5 weeks of CPZ treatment, animals were given either an acute IP injection of LPS (N = 10–14/group) or repeated IP injections of LPS every three days up to the point of sacrifice on day 14 post-CPZ removal (2 wks off) (9–14/group) (see Fig. 1). Animals treated with acute LPS received 10 μg of LPS while control injections consisted of physiological saline. For animals repeatedly treated with LPS, the first injection was 10 μg per mouse, since this dose induces sickness behavior, cytokine expression in the brain, and microglial activation (unpublished data). The second dose was increased to 15 μg on days 3 and 6, and finally on days 9 and 12 the dose increased to 20 μg per mouse. The progressive increase was designed to counteract the likelihood of observing LPS tolerance (Ziegler-Heitbrock 1995; Beishuizen & Thijs 2003). To ensure LPS efficacy, animals were weighed every day to assess physical consequences of sickness behavior. Animals were sacrificed by decapitation 2 hours after the last injection of LPS (or 2 hrs after the acute LPS injection) and trunk blood and tissues collected. This time point was previously shown to be optimal for increased pituitary-adrenal activation (Kawashima & Kusnecov, 2002). Tissue was flash frozen in 2-methylbutane and stored at −70 °C until dissection and protein extraction.
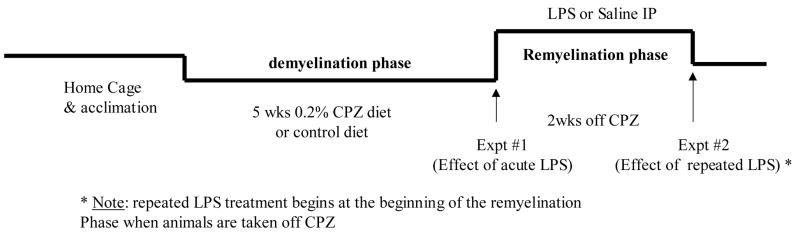
Fig. 1.
Summary of experimental design. Animals were given 0.2% cuprizone (CPZ) or control feed for 5 weeks to induce demyelination. After that point, animals were either sacrificed after an acute injection of LPS or saline (Expt 1), or allowed to remyelinate for a two week period throughout which they were given injections of LPS or saline every three days (Expt 2). Arrows represent days of sacrifice, which was 2 hrs after injection.
2.5. Protein extraction and quantification
The corpus callosum (CC) and spleen were dissected and placed in 0.5 ml (for CC) or 1 ml (for spleen) of 1mM Phenylmethanesulfonyl fluoride (PMSF) in 0.1M phosphate buffer to inhibit protease activity. The tissue was homogenized and centrifuged at 4000 RPM for 30 minutes after which the supernatant was collected. Total protein was quantified using the BCA protein assay kit (Pierce, Rockford, IL). Absorbance was read at 562 nm and concentrations were calculated off a standard curve generated using bovine serum albumin (BSA). Total protein was expressed as μg/ml.
2.6. Cytokine enzyme-linked immunosorbent assay (ELISA)
Spleen homogenates were centrifuged and the supernatants assayed for immunoreactive IL-6, TNFα, and IL-1β using OptEIA ELISA kits according to the manufacturer’s instructions (BD Biosciences, San Diego, CA). All standards and samples were run in duplicate. The absorbance was read at 450 nm using an EL800 universal BioTek microplate reader. Concentrations were calculated off the standard curve using KC Junior software (Biotek). The cytokine data were expressed as a ratio of total protein.
2.7. Corticosterone Radioimmunoassay (RIA)
Trunk blood was collected by rapid decapitation into heparin-treated vacutainer tubes (Becton Dickinson, Rutherford, NJ). The blood was centrifuged immediately at 2000 rpm for 15 min, and the plasma was collected and stored at −70°C. Corticosterone was measured by ImmunChem™ Double Antibody Corticosterone 125I kit following the manufacturer’s instructions (MP Biomedicals, Irvine, CA). All standards and samples were run in duplicate and counted using a Cobra II Auto Gamma counter. The corticosterone data were expressed as ng/ml.
2.8. Statistical Analysis
Most experiments conformed to a factorial design and were therefore analyzed by ANOVA using Statview, a statistical software package. In the event of significant main effects, appropriate post hoc comparisons were conducted with the inbuilt protection for Type I error. Comparisons were conducted using the Fisher’s least significant difference test when the omnibus F achieved a 0.05 level of significance. In some cases, in which a priori predictions based on previous data were warranted, an unpaired t test was conducted.
3. Results
Figure 1 provides a summary of the design for the current study. Experiment 1 assessed the impact of an acute injection of LPS delivered at the end of 5 weeks of continuous exposure to CPZ in the diet. At this time, demyelination is maximal in the corpus callosum (see Fig. 2). However, upon CPZ removal affected regions of the brain begin to remyelinate showing significant recovery at 1 week off (see Fig. 2), and signs of full remyelination by 2 weeks off CPZ (see Fig. 2). Therefore, Experiment 2 involved repeated LPS injections during the period of recovery from CPZ treatment (see Fig. 1).
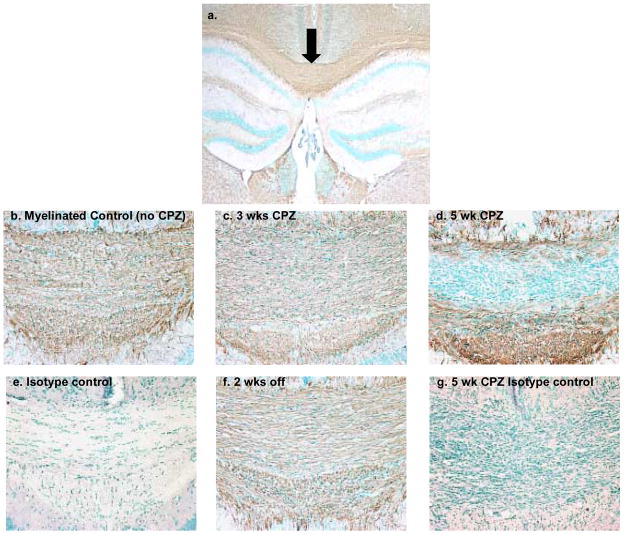
Fig. 2.
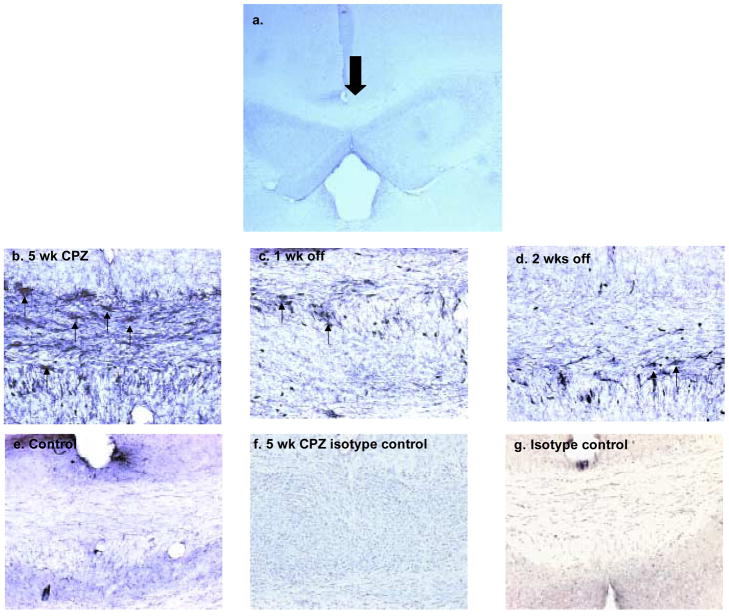
Immunohistochemistry for Myelin Basic Protein (MBP). (a). Representative overview panel of the corpus callosum (CC) at 4X magnification. The large arrow points to the region of the CC assessed in this study. (b–g). High magnification (20X) insets of the CC. (b) Figure 2b shows extensive MBP immunoreactivity, as indicated by the brown staining in control animals not fed CPZ. (c) After three weeks on a CPZ-containing diet, MBP staining is still evident, but is dramatically diminished after five weeks on the CPZ diet (see panel d). However, once CPZ was removed from the diet, MBP staining in 5-week fed animals reappeared (see panel f), being comparable to the control. (e and g) isotype controls for nonspecific immunoreactivity; note the darker staining of the green counterstain for the 5 wk CPZ treated isotype control. This is due to hypercellularity of the CC during demyelination, and probably due to microglial/macrophage accumulation at the site of injury (eg., Figures 4 and 5).
3.1. Weight
Body weight data were obtained at the end of the 5 week CPZ feeding period when LPS and saline injections were initiated. Consistent with previous literature CPZ animals weighed less than control animals (t (22) = −21.429, p = <.0001). While animals in Experiment 1 were sacrificed after a single LPS or Saline injection, weights continued to be monitored for animals in Experiment 2, and these data are summarized in Figure 3. Figure 3a presents the percentage weight over the 2 weeks from starting weight at the end of CPZ treatment. Overall, CPZ pretreated animals showed a dramatic increase in weight gain, irrespective of whether they were treated with LPS or saline (F (1, 20) = 209.159, p = <.0001) (see Fig. 3a).
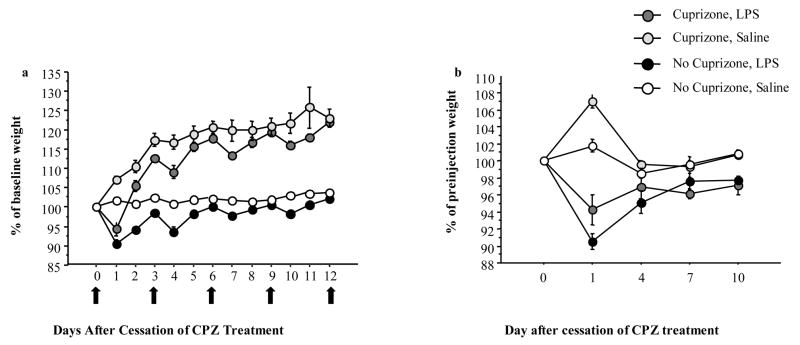
Fig. 3.
(a) Percent of baseline weight. Animals that received LPS showed a decline in the percent of baseline body weight throughout the experiment compared to their starting weight at day 0 (N = 6/group). Arrows indicate day of LPS treatment. (b) Percent of body weight measured at the time of each LPS or Saline injection. The CPZ-fed and control animals that received LPS continued to display a reduction in weight after each injection (N = 6/group). Error bars represent standard error of the mean.
Importantly, acute body weight changes were monitored after each LPS or Saline injection. Analysis of weight after each LPS injection showed a decline for both control and cuprizone fed animals relative to their weight from each previous day of injection (F (1,20) = 128.486, p = <.0001). Figure 3b presents this data as a percentage of weight from the day before an injection of LPS or Saline. After the initial injection of LPS, there was a dramatic overnight decline in weight for both CPZ pretreated and control fed animals (see Day 1, Fig. 3b). Interestingly, the percentage drop in weight was more marked for the control animals given LPS, when compared to similarly challenged CPZ pretreated mice. While this initial injection produced a decline in overnight body weight, this was significantly attenuated for both CPZ and control animals after subsequent injections with LPS, in spite of progressive increases in LPS dose (see Days 4, 7, and 10 Fig. 3b). Of note was the significant increase in body weight for the CPZ fed animals injected with saline on Day 0. The gain in weight was significantly greater than in control-fed animals similarly handled and injected with saline. This may reflect a differential sensitivity to handling and stress in the two groups.
3.2. Expression of CD11b and CD45
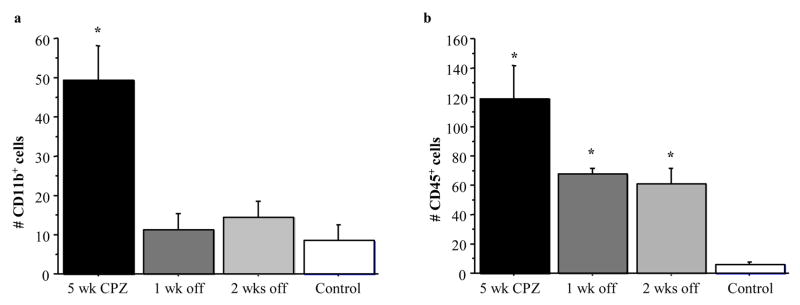
Coincident with the loss of myelin in the corpus callosum, this study also determined whether CPZ treatment increased expression of CD11b, a marker that allows for the differentiation of activated and quiescent microglial cells (Rock et al., 2004). As shown in Figures 4 and 6, CD11b+ activated microglia were significantly more prominent at the end of the 5 week CPZ treatment period, but by 1 and 2 weeks after removal of CPZ, the presence of activated morphology and CD11b over-expression was diminished (F (3,10) = 12.445, p = .0010). Figures 5 and 6 present the immunohistochemical and quantitation results for CD45 expression, another marker of microglial cells, as well as most hematopoietic cells (Carson et al., 1998), which was also highly evident in CPZ treated animals (F (3,10) = 13.806, p .0007). However, this did not diminish over the remyelination period, since at 1 and 2 weeks after CPZ removal there was still an abundant presence of CD45 immunohistochemistry (see Fig. 5 and 6). It should be noted that the CD11b and CD45 positive cells appeared to congregate at the site of most extensive demyelination (midline of the corpus callosum). Examination of regions more lateral to the area chosen in this study revealed that the myelin loss is less detectable (see Fig. 2). Therefore, microglial cells tend to aggregate where the lesion is most pronounced.
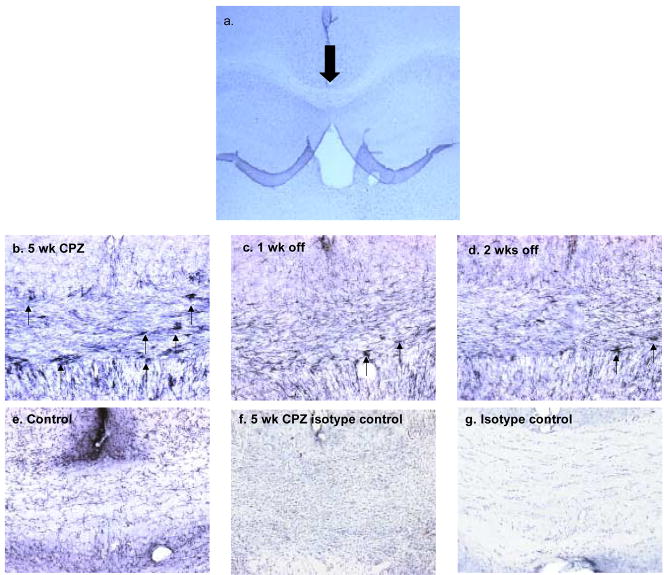
Fig. 4.
Representative images of immunohistochemistry for CD11b. (a) Representative overview panel of the CC at 4X magnification. The large arrow shows area of detail where numbers of heavily stained CD11b+ cells were observed and quantified (see Figure 6). The panels show CD 11b immunoreactive cells (arrows) in the CC of animals fed CPZ for 5 weeks (b) or taken off CPZ at the end of 5 weeks feeding and perfused after one (c) or two (d) weeks. (e) Representative immunohistochemistry conducted on brains from mice fed a diet that did not contain CPZ. (f and g) Isotype controls for nonspecific immunoreactivity.
Fig. 6.
Numbers of CD11b+ and CD45+ cells in the corpus callosum. Area of detail where counts were obtained corresponds to that shown in panel A of Figures 4 and 5. N = 3–4/group; *p < 0.05. Error bars represent standard error of the mean.
Fig. 5.
Representative images of immunohistochemistry for CD45. See Figure 4 for details.
3.3. Splenic Cytokines
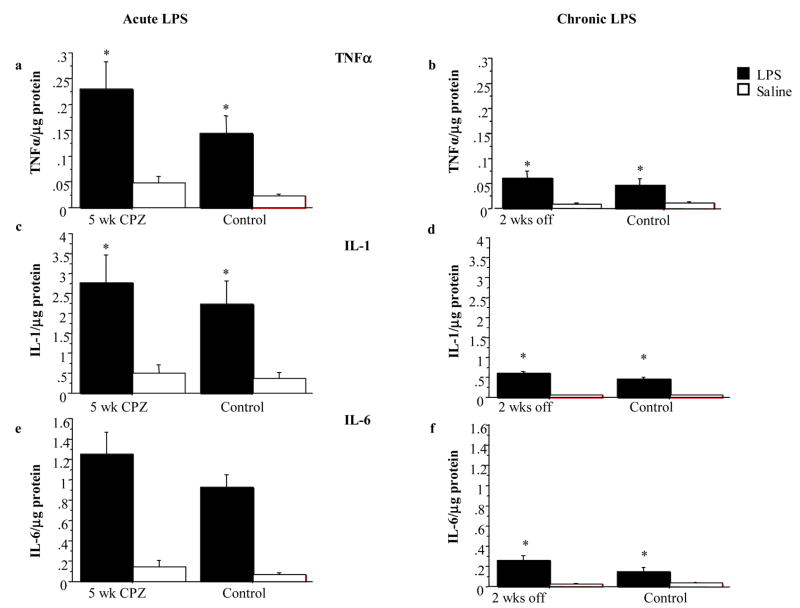
At the time of CPZ removal (Experiment 1 in Figure 1), acute LPS exposure significantly increased splenic TNFα when compared to saline-injected mice (F (1, 41) = 16.378, p < .0002). This was observed for both CPZ and control fed animals and did not differ in magnitude as revealed by a lack of a significant interaction effect (see Fig. 7a). This elevation in TNFα was markedly reduced after repeated exposure to LPS, and occurred equally for both control fed and CPZ pretreated animals (F (1, 36) = 17.821, p = .0002) (see Fig. 7b).
Fig. 7.
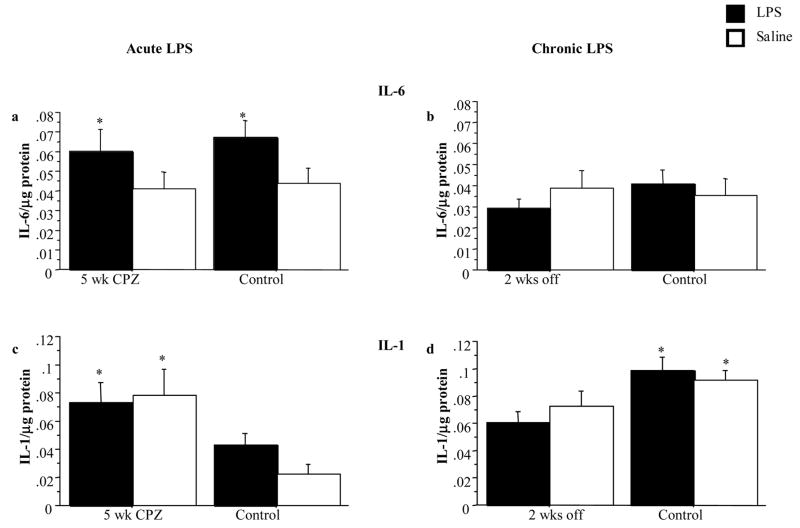
Mean splenic TNFα, IL-1β, and IL-6 concentrations (+/− SE) after acute (a, c, and e) or chronic (b, d, f) LPS and Saline injections. N= 10–14/group for acute LPS Expt.; N= 9–12/group for chronic Expt. See Materials and Methods, as well as Figure 1, for details. *p < 0.05.
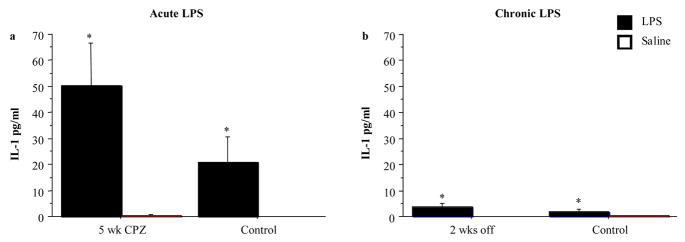
This pattern of results was repeated for measurement of IL-1β and IL-6 production after acute LPS injection (IL-1β: F (1, 41) = 15.690, p = .0003; IL-6: F (1, 40) = 40.827, p = .0001) (see Fig. 7c and 7e), there being no difference between the CPZ and control groups. Similarly, after repeated LPS injection, significant reductions in IL-1β and IL-6 were observed, although the concentration of splenic cytokine was still higher than saline injected animals (IL-1β: F (1, 36) = 150.239, p = .0001; IL-6: F (1, 35) = 19.101, p < .0001) (see Fig. 7d and 7f). Increased levels of plasma IL-1 after acute and chronic treatment with LPS confirmed that splenic concentrations include extracellular levels of cytokine that can be measured in plasma (Acute: F (1,41) = 5.382, p = .0254; Chronic: F (1, 36) = 5.561, p = .0239) (see Fig. 8a–8b).
Fig. 8.
Mean plasma IL-1β concentration (+/− SE) after acute (a) or chronic (b) LPS and Saline injections. Acute LPS Expt (N= 6–18/group); Chronic LPS Expt (N = 9–12/group). *p < 0.05
3.4. Corpus Callosum Cytokine profile
Since microglial activation had taken place in the corpus callosum of CPZ treated animals, it was important to determine whether the levels of proinflammatory cytokines IL-1β, IL-6 and TNFα were differentially altered after systemic acute or repeated LPS administration. To this end, the corpus callosum was dissected and assayed for total cytokine concentration. Measurements of TNFα did not reveal any differences in response to acute LPS treatment (data not shown), and therefore no assays were conducted on brains from animals given repeated LPS treatment. However, for IL-6, a significant elevation was observed after acute LPS challenge in both CPZ and control animals (F (1,27) = 5.187, p = .0309) (see Fig. 9a). This however was not observed after repeated LPS treatment (see Fig. 9b).
Fig 9.
Mean concentration (+/− SE) of IL-1β and IL-6 in the corpus callosum of mice after acute (a) or chronic (b) LPS and Saline injections. For further details see Fig. 8 *p < 0.05
In contrast, while IL-1β concentrations did not change in response to a single LPS injection given at the end of the 5 week CPZ treatment regimen, there nevertheless was a significant effect of CPZ treatment alone (F (1,41) = 5.502, p = .0239) (see Fig. 9c). That is, these animals had higher concentrations of IL-1β when compared to control-fed mice (see Fig 9c). The latter also failed to show an IL-1 response to LPS. Alternatively, after a regimen of repeated LPS or saline injections, the CPZ pretreated animals had lower levels of IL-1β than control-fed animals, irrespective of LPS or saline treatment (F (1,36) = 9.782, p = .0035) (see Fig. 9d).
3.5. Plasma Corticosterone
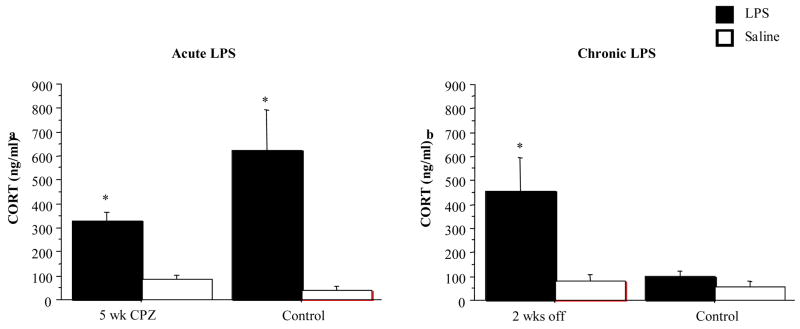
Acute exposure to LPS increased the concentration of cortiocosterone in both the five week CPZ group and controls (F (1, 66) = 8.957, p < .0001) (see Fig. 10a). Although not statistically significant, an unpaired t test suggested that the five wk CPZ animals showed an attenuated corticosterone response after acute LPS exposure (p = .0677). Conversely, when taken off CPZ and subjected to the repeated LPS treatment regimen, there was an increase in corticosterone compared to control-fed animals that received LPS (F (1, 66) = 4.807, p = .0043 (see Fig. 10b).
Fig. 10.
Mean plasma corticosterone concentration after (a) acute, and (b) chronic LPS and Saline injections. (N = 9–12/group for all LPS groups; N= 4–9 for all saline groups). *p < 0.05
4. Discussion
The present study examined the impact of LPS on murine central and peripheral cytokine production during CNS remyelination following cessation of treatment with the demyelinating agent cuprizone. Because this study was interested in both acute and repeated exposure to LPS, body weight was measured every day to ensure that the particular paradigm employed continued to be efficacious and did not produce a reduction in CNS reactivity due to the phenomenon of LPS tolerance (Zeigler-Heitbrock 1995). Both CPZ and control animals displayed a reduction in body weight after repeated injections of LPS. Therefore, the period of remyelination and repair after a neuropathological neurotoxic demyelinating insult, did not appear to alter the hypophagic response to LPS.
To ensure that any effects in the brain were not due to peripheral alterations of cytokine production due to cuprizone, splenic cytokine levels were measured in saline and LPS-treated animals. The results showed no differences in the splenic levels of IL-1β, TNFα nor IL-6 between CPZ and control animals acutely challenged with LPS. Similarly, although splenic cytokine responses to repeated LPS treatment were markedly attenuated, there were no differences between the CPZ and control groups. These results provided evidence that 5 weeks of CPZ treatment did not significantly affect the functional capacity of splenic macrophages. Moreover, these data suggest that any effects of LPS treatment on brain and behavior are not due to the differential effects of CPZ on peripheral inflammatory cytokine responses. Finally, it was of interest to note that the development of LPS tolerance (Zeigler-Heitbrock 1995) remained unaffected by cuprizone treatment.
Neuropathological investigations have confirmed that the brains of MS patients present with extensive inflammation and microglial activation (Antel 2006). Therefore, the risk of systemic infection inducing relapse in MS patients may be mediated by the induction of neuroinflammatory events in an already compromised brain. In the present study, it was confirmed that at five wks of CPZ treatment there is a significant increase in the number of CD11b+ and CD45 +cells at the site of the lesion in the corpus callosum, which is consistent with other observations (Hiremath et al., 1998). Nonetheless, due to the fact that CD45 is constitutively expressed on all hematopoietic cells, there remains the possibility that the blood-brain barrier (BBB) may be impaired by CPZ treatment, thereby allowing parenchymal accumulation of peripheral macrophages. Alternatively, Kondo et al (1987) failed to observe disruption in the BBB due to cuprizone treatment. Whether through an impaired BBB or other mechanisms involving cell adhesion molecules, peripheral macrophages do accumulate in the brain during cuprizone induced demyelination (McMahon et al., 2002), although they represent a proportionately smaller percentage of the total population, which appears to be comprised mostly of resident microglial cells (McMahon et al., 2002). Given this literature, therefore, it remains to be determined whether the CD11b+ and CD45+ cells that are present in the corpus callosum after cuprizone treatment are largely activated local cells recruited by neuropathological events related to demyelination.
These issues notwithstanding, it was still reasonable to hypothesize that evidence of possibly activated microglial cells might be associated with an exaggerated synthesis of proinflammatory cytokines in the corpus callosum subsequent to systemic LPS exposure. However, as reported here, analysis of cellular lysates from the callosal region of CPZ-treated animals showed that the IL-1β, TNFα and IL-6 cytokine responses to acute LPS exposure did not differ from animals that were fed a CPZ-free diet. Moreover, after repeated LPS treatment over the course of the remyelination period, there was no evidence for an LPS-induced increase in cytokine levels in corpus callosum tissue.
Interestingly, there was no increase in the concentration of IL-1β in the corpus callosum after acute nor repeated LPS treatment. However, it was observed that IL-1β levels in the CPZ-treated animals were significantly higher at the 5 week time point of CPZ treatment (when CPZ was actually removed from the diet) than in control animals that were not fed with CPZ. The increase in IL-1β concentration at this 5 week time point may be explained by the fact that it represented the peak time for demyelination and microglial activation. Interestingly, we did not see an exaggerated IL-1 response after LPS treatment in the 5 wk CPZ animals, which might be explained by a refractoriness to further IL-1β production. However, non-CPZ animals did appear to show an increased callosal concentration of IL-1β after an acute LPS injection, although this failed to reach statistical significance. Other studies, focusing mainly on mRNA changes, have observed that IL-1β transcriptional activity is elevated in a number of regions of the brain after LPS treatment of C57BL/6 mice (Danzer et al., 1998). Therefore, the trend reported in the present study for actual IL-1β protein is consistent with this previous literature.
It is interesting that there was no change in cytokine release due to the abundant number of suspected activated microglial cells. The disparity may be due to the magnitude of cytokine release, as well as the types of cytokines. Immunocytochemical detection of cytokines might be more informative, and future studies should aim at characterizing this. In terms of not seeing an elevation in IL-1, perhaps the dose of LPS was insufficient. However, it was behaviorally active on the basis of the weight loss reported in other animals not sacrificed (eg., see Figure 3), as well as other studies in our lab showing anorexic and taste aversion effects in normal non-CPZ exposed mice (data not shown). Moreover, there may be changes in IL-1 expression in brain regions known to produce elevated cytokine levels after LPS exposure, such as the hypothalamaus and hippocampus, which were not looked at in this particular study (Laye et al., 1994).
It was of interest to note that after two weeks of remyelination, the level of IL-1β in LPS and saline-treated animals remained unchanged from that observed at the end of the CPZ treatment period (i.e. the 5 week time-point). However, for animals that had received the control diet, IL-1β levels increased substantially, irrespective of whether mice were treated with LPS or saline. Since neuropathological processes were unlikely to account for this increase, it is possible that this elevation was driven by the stress like conditions of the repeated injection schedule to which these particular animals had been subjected. For example, it has been demonstrated that psychological stressors can activate microglial cells and increase proinflammatory cytokines in the brain (Frank et al., 2006; Nair & Bonneau 2006; Nguyen et al., 1998; O’Connor et al., 2003). Consequently, the marked change in the concentration of the callosal IL-1β concentration may have been due to the repeated handling required to provide the intraperitoneal injection, as well as to measure the daily body weight over the two-week period. Although this hypothesis remains to be tested, it is interesting that the previously CPZ-treated animals failed to show this increase in IL-1β concentration. An explanation for this is not readily forthcoming, although it might reflect a resistance to IL-1β modulation during a period of myelin repair.
The present study also examined the neuroendocrine response to acute and repeated LPS challenge. Consistent with the cytokine responses to acute LPS, there was no difference in the corticosterone responses to LPS between CPZ and control fed animals. However, after repeated LPS treatment, the corticosterone response to the final LPS injection was significantly elevated in the CPZ group; in contrast, for control fed animals, there was a poor corticosterone response, which was consistent with the LPS tolerance observed for splenic cytokine levels. Interestingly, a tolerance effect was observed for the cytokine response to repeated LPS in the CPZ animals. Therefore, the augmented corticosterone response in this group suggests a possible shift in sensitivity to peripheral cytokine elevations during remyelination or perhaps a direct effect of LPS within areas of poor blood-brain barrier integrity. The explanation for this augmented corticosterone response requires further investigation, although it may suggest differential sensitivity of the HPA axis and upstream elements of the limbic system during periods of remyelination. These findings are consistent with other data (unpublished) from our lab showing that two weeks off cuprizone animals display more anxiety-like behavior when exposed to the open field, as well as a heightened c-fos response in the paraventricular nucleus of the hypothalamus (unpublished data).
Given the anti-inflammatory properties of glucocorticoids (Webster et al., 2002), the rise in corticosterone may be intended as protection in an already compromised brain. It has been demonstrated that both oligodendrocyte progenitor cells (OPCs) and mature oligodendrocytes are vulnerable to proinflammatory cytokines (Benveniste & Benos 1995; Lehnardt et al., 2002; Melcangi et al., 2000). However, when oligodendrocytes were exposed to corticosteroids, they were protected from apoptosis induced by cytokines (Melcangi et al., 2000). Therefore, corticosterone may be upregulated more so in the remyelinating CPZ animals in order to protect the OPCs from chronic exposure to proinflammatory cytokines induced by LPS. Additionally, it has been suggested that during development, glucocorticoids can induce proliferation and differentiation of OPCs, further supporting the hypothesis of a possible protective role for corticosterone in CPZ treated animals undergoing remyelination (Preston & McMorris 1984; Warringa et al., 1987).
An alternative view is that the augmented corticosterone in the face of repeated LPS treatment is detrimental to remyelination. Elevated levels of corticosterone have been shown to inhibit cell proliferation and neurogenesis and induce dendritic atrophy in the hippocampus (Yu et al., 2004; Stranahan et al., 2006; Sapolsky, et al., 1999). In addition, it has been reported that repeated treatment with glucocorticoids inhibits the proliferation of oligodendrocyte progenitor cells (OPC) and delays remyelination in the brain (Alonso 2000; Chari et al., 2006). It has also been suggested that a mechanism for corticosterone induced antiproliferative effects in the brain is downregulation of neurotrophins such as BDNF and IGF (Yu et al., 2004; Anderson et al., 2002). While the focus of BDNF has traditionally been on neural development in the hippocampus, BDNF has also been shown to be important for the myelination of the optic nerve and differentiation of oligodendrocyte progenitor cells in the basal forebrain (Binder & Scharfman 2004; Cellerino, et al., 1997; Du et al., 2003; Du et al., 2006). Insulin-like growth factor type 1 has also been shown to be important for remyelination, acting as a proliferation, differentiation, and survival factor for oligodendrocytes and oligodendrocyte progenitor cells (Frederick & Wood 2004; Mason et al., 2003). Therefore, the downregulation of these important neurotrophins by corticosterone may also be detrimental to remyelination. Future studies should aim at determining if corticosterone induced down regulation of these factors influence the rate and degree of remyelination in the brain.
In conclusion, this study demonstrated that cuprizone may be used as a potential model to explore the effects of peripheral inflammatory stimuli, such as LPS, on remyelination and recovery in the brain. In the periphery, both CPZ and control animals showed similar TNFα, IL-1, and IL-6 profiles after treatment with LPS. Therefore, the peripheral effects of CPZ were not interfering with splenic production of cytokines and the cytokine levels in the brain were not mediated by cytokine changes in the periphery. The 5 wk CPZ animals all showed an increase in the level of IL-1 regardless of treatment. Interestingly, the remyelinating animals receiving a stressful paradigm of repeated injections, maintained the same level of IL-1 as the 5 wk CPZ group without showing an elevated IL-1 response as seen in the control-fed animals. It remains to be determined whether the excessive corticosterone response after repeated LPS treatment of remyelinating animals may have restrained a further increase in IL-1 to maintain the levels within an optimal range that serves to allow for proper regeneration of oligodendrocytes.
Acknowledgments
The authors wish to express their gratitude to Dr. Kenneth Reuhl for his advice and Beth E. Crowell for her assistance in some of the experiments.
Support Contributed By: Grants MH60706, DA141186, NIEHS P30 ES05022 and NIEHS Graduate Training grant 5T32 E507148
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Alonso G. Prolonged corticosterone treatment of adult rats inhibits the proliferation of oligodendrocyte progenitors present throughout white and gray matter regions of the brain. Glia. 2000;31:219–231. doi: 10.1002/1098-1136(200009)31:3<219::aid-glia30>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- Anderson MF, Aberg MAI, Nilsson M, Eriksson PS. Insulin-like growth factor-I and neurogensis in the adult mammalian brain. Brain Res Dev Brain Res. 2002;134:115–122. doi: 10.1016/s0165-3806(02)00277-8. [DOI] [PubMed] [Google Scholar]
- Anisman H, Hayley S, Turrin N, Merali Z. Cytokines as a stressor: implications for depressive illness. Int J Neurosychopharmacol. 2002;5:357–373. doi: 10.1017/S1461145702003097. [DOI] [PubMed] [Google Scholar]
- Antel J. Oligodendrocyte/myelin injury and repair as a function of the central nervous system environment. Clin Neurol Neurosurg. 2006;108:245–249. doi: 10.1016/j.clineuro.2005.11.013. [DOI] [PubMed] [Google Scholar]
- Arnett HA, Mason J, Marino M, Suzuki K, Matsushima GK, Ting JPY. TNFa promotes proliferation of oligodendrocyte progenitors and remyelination. Nat Neurosci. 2001;4:1116–1122. doi: 10.1038/nn738. [DOI] [PubMed] [Google Scholar]
- Beishuizen A, Thijs LG. Endotoxin and the hypothalamopituitary-adrenal (HPA) axis. J Endotoxin Res. 2003;9:3–24. doi: 10.1179/096805103125001298. [DOI] [PubMed] [Google Scholar]
- Benveniste EN, Benos DJ. TNF-alpha and IFN-gamma-mediated signal transduction pathways: effects on glial cell gene expression and function. FASEB J. 1995;9:1577–1584. doi: 10.1096/fasebj.9.15.8529837. [DOI] [PubMed] [Google Scholar]
- Binder DK, Scharfman HE. Brain-derived neurotrophic factor. Growth Factors. 2004;22:123–131. doi: 10.1080/08977190410001723308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bitsch A, Schuchardt J, Bunkowski S, Kuhlmann T, Bruck W. Acute axonal injury in multiple sclerosis. Correlation with demyelination and inflammation. Brain. 2002;123:1174–1183. doi: 10.1093/brain/123.6.1174. [DOI] [PubMed] [Google Scholar]
- Blakemore WF. The case for a central nervous system (CNS) origin for the Schwann cells that remyelinate CNS axons following concurrent loss of oligodendrocytes and astrocytes. Neuropathol Appl Neurobio. 2005;31:1–10. doi: 10.1111/j.1365-2990.2005.00637.x. [DOI] [PubMed] [Google Scholar]
- Brugg B, Dubreuil YL, Huber G, Wollman EE, Delhaye-Bouchaud N, Mariani J. Inflammatory processes induce beta-amyloid precursor protein changes in mouse brain. Proc Natl Acad Sci. 1995;92:3032–3035. doi: 10.1073/pnas.92.7.3032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buljevac D, Flach HZ, Hop WC, Hijdra D, Laman JD, Savelkoul HF, van Der Meche FG, van Doorn PA, Hintzen RQ. Prospective study on the relationship between infections and multiple sclerosis exacerbations. Brain. 2002;125:952–960. doi: 10.1093/brain/awf098. [DOI] [PubMed] [Google Scholar]
- Carson MJ, Reilly CR, Sutcliffe JG, Lo D. Mature microglial resemble immature antigen-presenting cells. Glia. 1998;22:72–85. doi: 10.1002/(sici)1098-1136(199801)22:1<72::aid-glia7>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- Castano A, Herrera AJ, Cano J, Machado A. Lipopolysaccharide intranigral injection induces inflammatory reaction and damage in nigrostriatal dopaminergic system. J Neurochem. 1998;70:1584–1592. doi: 10.1046/j.1471-4159.1998.70041584.x. [DOI] [PubMed] [Google Scholar]
- Cellerino A, Carroll P, Thoenen H, Barde YA. Reduced size of retinal ganglion cell axons and hypomeylination in mice lacking brain-derived neurotrophic factor. Mol Cell Neurosci. 1997;9:397–408. doi: 10.1006/mcne.1997.0641. [DOI] [PubMed] [Google Scholar]
- Chari DM, Zhao C, Kotter MR, Blakemore WF, Franklin RJM. Corticosteroids Delay Remyelination of Experimental Demyelination in the Rodent Central Nervous System. J Neurosci. 2006;83:594–605. doi: 10.1002/jnr.20763. [DOI] [PubMed] [Google Scholar]
- Crang AJ, Gilson JM, Li WW, Blakemore WF. The remyelinating potential and in vitro differentiation of MOG-expressing oligodendrocyte precursors isolated from the adult rat CNS. Eur J Neurosci. 2004;20:1445–1460. doi: 10.1111/j.1460-9568.2004.03606.x. [DOI] [PubMed] [Google Scholar]
- Cunningham C, Wilcockson DC, Campion S, Lunnon K, Perry H. Central and systemic endotoxin challenge exacerbate the local inflammatory response and increase neuronal death during chronic neurodegeneration. J Neurosci. 2005;25:9275–9284. doi: 10.1523/JNEUROSCI.2614-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dantzer R, Bluthe RM, Laye S, Bret-Dibat JL, Parnet P, Kelley KW. Cytokines and sickness behavior. Ann NY Acad Sci. 1998;840:586–590. doi: 10.1111/j.1749-6632.1998.tb09597.x. [DOI] [PubMed] [Google Scholar]
- Danzer R. Cytokine-induced sickness behavior: mechanisms and implications. Ann NY Acad Sci. 2001;933:222–234. doi: 10.1111/j.1749-6632.2001.tb05827.x. [DOI] [PubMed] [Google Scholar]
- Du Y, Fischer TZ, Lee LN, Lercher LD, Dreyfus CF. Regionally specific effects of BDNF on oligodendrocytes. Dev Neurosci. 2003;25:116–126. doi: 10.1159/000072261. [DOI] [PubMed] [Google Scholar]
- Du Y, Fischer TZ, Clinton-Luke P, Lercher LD, Dreyfus CF. Distinct 0effects of p75 in mediating actions of neurotrophins on basal forebrain oligodendrocytes. Mol Cell Neurosci. 2006;31:366–375. doi: 10.1016/j.mcn.2005.11.001. [DOI] [PubMed] [Google Scholar]
- Dunn AJ, Wang J, Ando T. Effects of cytokines on cerebral neurotransmission. Comparison with the effects of stress. Adv Exp Med Biol. 1999;461:117–127. doi: 10.1007/978-0-585-37970-8_8. [DOI] [PubMed] [Google Scholar]
- Emery B, Cate HS, Marriott M, Merson T, Binder MD, Snell C, Soo PY, Murray S, Croker B, Zhang JG, Alexander WS, Cooper H, Butzkueven H, Kilpatrick TJ. Suppressor of cytokine signaling 3 limits protection of leukemia inhibitory factor receptor signaling against central demyelination. PNAS. 2006;103:7859–7864. doi: 10.1073/pnas.0602574103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ewing C, Bernard CC. Insights into the aetiology and pathogenesis of multiple sclerosis. Immunol Cell Biol. 1998;76:47–54. doi: 10.1046/j.1440-1711.1998.00718.x. [DOI] [PubMed] [Google Scholar]
- Frank GM, Baratta MV, Sprunger DB, Watkins LR, Maier SF. Microglia serve as a neuroimmune substrate for stress-induced potentiation of CNS pro-inflammatory cytokine responses. Brain Behav Immun. 2006 doi: 10.1016/j.bbi.2006.03.005. In Press. [DOI] [PubMed] [Google Scholar]
- Franklin KBJ, Paxinos G. The mouse brain in stereotaxic coordinates. San Diego: Academic; 1997. [Google Scholar]
- Franklin RJM, Goldman JE. Remyelination by endogenous glia. In: Lazzarini RA, editor. Myelin biology and disorders. Vol. 1. Elsevier; San Diego: 2004. pp. 173–196. [Google Scholar]
- Frederick TJ, Wood TL. IGF-1 and FGF-2 coordinately enhance cyclin D1 and cyclin E-cdk2 association and activity to promote G1 progression in oligodendrocyte progenitor cells. Mol Cell Neurosci. 2004;25:480–492. doi: 10.1016/j.mcn.2003.11.015. [DOI] [PubMed] [Google Scholar]
- Hiremath MM, Saito Y, Knapp GW, Tin JP, Suzuki K, Matsushima GK. Microglial/macrophage accumulation during cuprizone-induced demyelination in C57BL/6 mice. J Neuroimmunol. 1998;92:38–49. doi: 10.1016/s0165-5728(98)00168-4. [DOI] [PubMed] [Google Scholar]
- Hunter SF, Hafler DA. Ubiquitous pathogens: links between infection and autoimmunity in MS? Neurology. 2000;55:164–165. doi: 10.1212/wnl.55.2.164. [DOI] [PubMed] [Google Scholar]
- Huseby ES, Liggitt D, Brabb T, Schnabel B, Ohlen C, Goverman J. A pathogenic role for myelin-specific CD8(+) T cells in a model for multiple sclerosis. J Exp Med. 2001;194:669–676. doi: 10.1084/jem.194.5.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawashima N, Kusnecov AW. Effects of staphylococcal enterotoxin A on pituitary-adrenal activation and neophobic behavior in the C57BL/6 mouse. J Immunol. 2002;123:41–49. doi: 10.1016/s0165-5728(01)00486-6. [DOI] [PubMed] [Google Scholar]
- Keirstead HS, Blakemore WF. The role of oligodendrocytes and oligodendrocyte progenitors in CNS remyelination. Adv Exp Med Biol. 1999;468:183–197. doi: 10.1007/978-1-4615-4685-6_15. [DOI] [PubMed] [Google Scholar]
- Kondo A, Nakano T, Suzuki K. Blood-brain barrier permeability to horseradish peroxidase in twitcher and cuprizone-intoxicated mice. Brain Res. 1987;425:186–190. doi: 10.1016/0006-8993(87)90499-9. [DOI] [PubMed] [Google Scholar]
- Laye S, Parnet P, Goujon E, Danzer R. Peripheral administration of lipopolysaccharide induces the expression of cytokine transcripts in the brain and pituitary of mice. Brain Res Mol Brain Res. 1994;27:157–162. doi: 10.1016/0169-328x(94)90197-x. [DOI] [PubMed] [Google Scholar]
- Lehnardt S, Lachance C, Patrizi S, Lefebvre S, Follett PL, Jensen FE, Rosenberg PA, Volpe JJ, Vartanian T. The toll-like receptor TLR4 is necessary for lipopolysaccharide-induced oligodendrocyte injury in the CNS. J Neurosci. 2002;22:2478–2486. doi: 10.1523/JNEUROSCI.22-07-02478.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martino G, Furlan R, Brambilla E, Bergami A, Ruffini F, Gironi M, Poliani PL, Grimaldi LM, Comi G. Cytokines and immunity in multiple sclerosis: the dual signal hypothesis. J Neuroimmunol. 2000;109(1):3–9. doi: 10.1016/s0165-5728(00)00295-2. [DOI] [PubMed] [Google Scholar]
- Mason JL, Xuan S, Dragatsis I, Efstratiadis A, Goldman JE. Insulin-like growth factor (IGF) signaling through type 1 IGF receptor plays an important role in remyelination. J Neurosci. 2003;23:7710–7718. doi: 10.1523/JNEUROSCI.23-20-07710.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason JL, Toews A, Hostettler JD, Morell P, Suzuki K, Goldman JE, Matsushima GK. Oligodendrocytes and progenitors become progressively depleted within chronically demyelinated lesions. Am J Pathol. 2004;164:1673–1682. doi: 10.1016/S0002-9440(10)63726-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matushima GK, Morell P. The neurotoxicant, cuprizone, as a model to study demyelination and remyelination in the central nervous system. Brain Pathol. 2001;11:107–116. doi: 10.1111/j.1750-3639.2001.tb00385.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melcangi RC, Cavarretta I, Magnaghi V, Ciusani E, Salmaggi A. Corticosteroids protect oligodendrocytes from cytokine-induced cell death. NeuroReport. 2000;11:3969–3972. doi: 10.1097/00001756-200012180-00013. [DOI] [PubMed] [Google Scholar]
- McMahon EJ, Suzuki K, Matsushima GK. Peripheral macrophage recruitment in cuprizone-induced CNS demyelination despite an intact blood-brain barrier. J Neuroimmunol. 2002;130:32–45. doi: 10.1016/s0165-5728(02)00205-9. [DOI] [PubMed] [Google Scholar]
- McCoy L, Tsunoda I, Fujinami RS. Multiple sclerosis and virus induced immune responses: autoimmunity can be primed by molecular mimicry and augmented by bystander activation. 2006;39(1):9–19. doi: 10.1080/08916930500484799. [DOI] [PubMed] [Google Scholar]
- MohanKumar SMJ, MohanKumar PS, Quadri SK. Lipopolysaccharide-induced changes in monoamines in specific areas of the brain: blockade by interleukin-1 receptor antagonist. Brain Res. 1999;824:232–237. doi: 10.1016/s0006-8993(99)01206-8. [DOI] [PubMed] [Google Scholar]
- Nair A, Bonneau RH. Stress-induced elevation of glucocorticoids increases microglia proliferation through NMDA receptor activation. J Neuroimmunol. 2006;171:72–85. doi: 10.1016/j.jneuroim.2005.09.012. [DOI] [PubMed] [Google Scholar]
- Nguyen KT, Deak T, Owens SM, Kohno T, Fleshner M, Watkins LR, Maier SF. Exposure to acute stress induces brain interleukin-1beta protein in the rat. J Neurosci. 1998;18:2239–2246. doi: 10.1523/JNEUROSCI.18-06-02239.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Connor KA, Johnson JD, Hansen MK, Wieseler JL, Maksimova FE, Watkins LR, Maier SF. Peripheral and central proinflammatory cytokine response to a severe acute stressor. Brain Res. 2003;991:123–132. doi: 10.1016/j.brainres.2003.08.006. [DOI] [PubMed] [Google Scholar]
- Oksenberg JR, Barcellos AF. Multiple Sclerosis genetics: leaving no stone unturned. Genes Immun. 2005;6:375–387. doi: 10.1038/sj.gene.6364237. [DOI] [PubMed] [Google Scholar]
- Pasquini LA, Calatayud CA, Bertone Una AL, Millet V, Pasquini JM, Soto EF. The Neurotoxic Effect of Cuprizone on Oligodendrocytes Depends on the Presence of Pro-inflammatory Cytokines Secreted by Microglia. Neurochem Res. 2006 doi: 10.1007/s11064-006-9165-0. In Press. [DOI] [PubMed] [Google Scholar]
- Perry VH. The influence of systemic inflammation on inflammation in the brain: implications for chronic neurodegenerative disease. Brain Behav Immun. 2004;18:407–413. doi: 10.1016/j.bbi.2004.01.004. [DOI] [PubMed] [Google Scholar]
- Preston SC, McMoriss FA. Adrenalectomy of rats results in hypomyelination of the central nervous system. J Neuochem. 1984;42:262–267. doi: 10.1111/j.1471-4159.1984.tb09727.x. [DOI] [PubMed] [Google Scholar]
- Rock RB, Gekker G, Hu S, Sheng WS, Cheeran M, Lokensgard JR, Peterson PK. Role of microglial in central nervous system infections. Clin Microbiol. 2004;17:942–964. doi: 10.1128/CMR.17.4.942-964.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sapolsky RM. Glucocorticoids, stress, and their adverse neurological effects: relevance to aging. Exp Gerontal. 1999;34:721–732. doi: 10.1016/s0531-5565(99)00047-9. [DOI] [PubMed] [Google Scholar]
- Sly LM, Krzesicki RF, Brashler JR, Buhl AE, Mckinley DD, Carter DB, Chin JE. Endogenous brain cytokine mRNA and inflammatory responses to lipopolysaccharide are elevated in the Tg2576 transgenic mouse model of Alzheimer’s disease. Brain Res Bull. 2001;56:581–588. doi: 10.1016/s0361-9230(01)00730-4. [DOI] [PubMed] [Google Scholar]
- Stranahan AM, Khalil D, Gould E. Social isolation delays the positive effects of running on adult neurogenesis. Nat Neurosci. 2006;9:526–533. doi: 10.1038/nn1668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Asensio VC, Campbell IL. Cytokines and chemokines as mediators of protection and injury in the central nervous system assessed in transgenic mice. Curr Top Microbiol Immunol. 2002;265:23–48. doi: 10.1007/978-3-662-09525-6_2. [DOI] [PubMed] [Google Scholar]
- Warringa RA, Hoeben RC, Koper JW, Sykes JE, van Golde LM, Lopes-Cardozo M. Hydrocortisone stimulates the development of oligodendrocytes in primary glial cultures and affects glucose metabolism and lipid synthesis in these cultures. Brain Res. 1987;431:79–86. doi: 10.1016/0165-3806(87)90197-0. [DOI] [PubMed] [Google Scholar]
- Webster JI, Tonelli L, Sternberg EM. Neuroendocrine regulation of immunity. Annu Rev Immunol. 2002;20:125–163. doi: 10.1146/annurev.immunol.20.082401.104914. [DOI] [PubMed] [Google Scholar]
- Vipin K. Homeostatic control of immunity by TCR peptide–specific Tregs. J Clin Invest. 2004;114:1222–1226. doi: 10.1172/JCI23166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu IT, Lee SH, Lee YS, Son H. Differential effects of corticosterone and dexamethasone on hippocampal neurogenesis in vitro. Biochem Biophys Res Commun. 2004;317:484–490. doi: 10.1016/j.bbrc.2004.03.071. [DOI] [PubMed] [Google Scholar]
- Ziegler-Heitbrock HW. Molecular mechanism in tolerance to lipopolysaccharide. J Inflamm. 1995;45:13–26. [PubMed] [Google Scholar]