Abstract
Background
Treatment of severe sepsis is expensive, often encompassing a number of discretionary modalities. The objective of the present study was to assess intercenter variation in resource and therapeutic modality use in patients with severe sepsis.
Methods
We conducted a prospective cohort study of 1028 adult admissions with severe sepsis from a stratified random sample of patients admitted to eight academic tertiary care centers. The main outcome measures were length of stay (LOS; total LOS and LOS after onset of severe sepsis) and total hospital charges.
Results
The adjusted mean total hospital charges varied from $69 429 to US$237 898 across centers, whereas the adjusted LOS after onset varied from 15.9 days to 24.2 days per admission. Treatments used frequently after the first onset of sepsis among patients with severe sepsis were pulmonary artery catheters (19.4%), ventilator support (21.8%), pressor support (45.8%) and albumin infusion (14.4%). Pulmonary artery catheter use, ventilator support and albumin infusion had moderate variation profiles, varying 3.2-fold to 4.9-fold, whereas the rate of pressor support varied only 1.92-fold across centers. Even after adjusting for age, sex, Charlson comorbidity score, discharge diagnosis-relative group weight, organ dysfunction and service at onset, the odds for using these therapeutic modalities still varied significantly across centers. Failure to start antibiotics within 24 hours was strongly correlated with a higher probability of 28-day mortality (r2 = 0.72).
Conclusion
These data demonstrate moderate but significant variation in resource use and use of technologies in treatment of severe sepsis among academic centers. Delay in antibiotic therapy was associated with worse outcome at the center level.
Keywords: bacteremia, cohort study, costs, resource utilization, sepsis, severe sepsis, variation
Introduction
Each year approximately 750 000 patients in the USA suffer from sepsis. Treatment for this disease costs $16.7 billion annually [1]. Despite advances in supportive intensive care and use of appropriate antibiotics, the mortality associated with sepsis remains high, especially among those who develop hemodynamic shock [2-4]. Such patients frequently progress to multiple organ dysfunction syndrome, which results in a much higher mortality rate than among patients who do not develop such complications [3,5-7]. This life-threatening syndrome is largely attributed to the cardiovascular abnormalities associated with septic shock, requiring supportive therapy (e.g. mechanical ventilation, fluid resuscitation, and vasopressors) with volume loading, oxygen delivery, and regional perfusion [8]. However, there are still many controversies regarding choice of fluids [9-12], vasopressors [13-15], hemodynamic end-points for resuscitation [16-20], and monitoring techniques. Findings from a national survey of intensive care unit (ICU) utilization showed differences in types of procedures performed between unit types and hospital sizes [21].
The goals of the present study were to examine systematically the variations in overall resource use, therapeutic modality use, and outcome in patients with severe sepsis across academic medical centers, and to evaluate relationships between therapeutic modality use and variation in resource use.
Methods
Patient population
All participating centers were members of the Academic Medical Center Consortium, which sponsored the study. All eight centers were large tertiary care academic medical centers with approximately 18 800–43 700 admissions annually [22]. Patient enrollment occurred between January 1993 and April 1994.
Patients surveyed represented a stratified random sample of patients in ICUs and outside ICUs for whom blood cultures were either positive or negative, as previously described [22]. Among non-ICU patients with blood cultures that were negative at 48 hours, sampling at all centers was fixed at 10%. Among ICU patients and patients with a blood culture positive for a pathogen within 48 hours, the fraction of patients sampled was established at each center and varied from 30% to 60%. In addition, all patients who died in an emergency department or an ICU and all patients who received a novel therapy for sepsis syndrome were surveyed. The ICUs involved included both open and closed units.
Although previous reports included 1342 episodes of sepsis among 1166 unique patients with 1190 hospital admissions [22,23], the results presented here from the same database were based on the 1011 (87%) patients, representing 1028 (86%) admissions with 1173 (87%) episodes of sepsis syndrome, for whom billing information was available. When patients had more than one episode in an admission, only the first episode in that admission was evaluated.
Definitions
Sepsis syndrome was defined as described by Sands and coworkers [22], using a modification of the criteria developed by Bone [24], and involved assessment of the presence of both screening criteria and confirmatory criteria for sepsis syndrome. Either of the following were required for a patient to satisfy the screening criteria: all four of temperature greater than 38.3°C or less than 35.6°C rectally, respirations greater than 20/min or mechanical ventilation, heart rate greater than 90 beats/min, and clinical evidence of infection; or one or more blood cultures positive for a pathogen at 48 hours following admission. Presence of any of the following seven features, without an alternative cause, was required for satisfaction of the confirmatory criteria: ratio of partial arterial oxygen tension to inspired fractional oxygen below 280 (intubated) or 40% face mask in use (nonintubated); arterial pH below 7.30; urine output below 30 ml/h; systolic blood pressure below 90 mmHg or a fall in systolic blood pressure by more than 40 mmHg sustained for 2 hours despite fluid challenge; systemic vascular resistance below 800 dynes·s/cm5; prothrombin time or partial thromboplastin time greater than normal or platelets below 100.0 × 109/l or platelets decreased to less than 50% or most recent measurement before current day; and documentation of deterioration in mental status within 24 hours. In addition, all patients who received a form of novel therapy for sepsis syndrome were enrolled in the sepsis syndrome group. Because all patients with sepsis syndrome also meet the criteria for 'severe sepsis' (the term that is most popular now), below we use the term 'severe sepsis'.
Acute respiratory distress syndrome (ARDS) was defined as partial arterial oxygen tension below 50 mmHg despite fractional inspired oxygen in excess of 50%, and bilateral pulmonary infiltrates without signs of heart failure. Disseminated intravascular coagulation (DIC) was defined as a positive D-dimer or elevated fibrin degradation products plus a falling platelet count of 25% of less of baseline, and elevation of either prothrombin time or partial prothrombin time, or clinical evidence of bleeding. Central nervous system (CNS) dysfunction was defined as a Glasgow Coma Scale score below 15 in patients with normal results at baseline neurologic examination, or at least 1 point lower than baseline in patients with a previously abnormal examination result. Renal failure was defined as an increase in serum creatinine of at least 2.0 mg/dl during the sepsis episode if creatinine was below 1.5 mg/dl at baseline, or an absolute increase of 1.0 mg/dl or more if baseline creatinine was at least 1.5. Liver failure was defined as present if total bilirubin was 2.0 mg/dl or greater, and either the alkaline phosphatase or a transaminase level was greater than twice normal, in the absence of confounding disease. Shock (after onset of sepsis) was defined as either at least 1 hour of decreased systolic blood pressure by at least 40 mmHg, or systolic blood pressure of 90 mmHg or less after adequate volume replacement, and in the absence of antihypertensive agents, or continuous infusion of pressors. Uncontrolled hemorrhage was defined as active and live-threatening bleeding, requiring transfusions of more than 4 units within 24 hours of onset of sepsis. Moderate or severe liver disease was defined as cirrhosis with portal hypertension, or hepatic failure with coma or encephalopathy within the preceding 6 months.
Data collection
Surveyors recorded the presence of all screening criteria for every day that a patient was a valid member of the surveillance group. If screening criteria were met, then the patient was enrolled as a case and detailed information, including demographic, historical, clinical and laboratory data, were abstracted from the medical record into a standardized data collection form for which all variables and criteria were defined. Additional data, including information on antibiotic use, treatment modalities (pulmonary artery catheter [PAC] use, ventilator support, pressor support, albumin infusion, het-astarch, and dextran), complications (ARDS, renal failure, liver failure, DIC, and shock), and associated outcomes, were abstracted 28 days after the first onset of severe sepsis or at the time of death or hospital discharge, whichever came first. Hospital length of stay (LOS) before and after the onset of severe sepsis and total hospital charge were obtained from the unified Academic Medical Center Consortium hospital billing database.
We calculated the patient's Charlson comorbidity score [25]; a higher score reflects greater comorbidity. We also obtained information on discharge diagnosis-relative groups (DRGs) and DRG weight. The current generation of DRGs was originally based on ICD-9-CM and takes into consideration operations, complications, and comorbidities. Each DRG was preassigned an average LOS and reimbursement rate by the US Health Care Financing Administration. The higher the DRG weight, the higher the level of reimbursement.
Analyses
Because the sampling fractions from different strata varied [22], individual cases carried different weight [23] and all calculations, including means and proportions, were performed using appropriate case weights.
We compared the proportion of sepsis patients receiving each intervention therapy and then the proportion with complications among eight academic centers, using weighted χ2 statistics. We also calculated the ratio of the highest to lowest proportion for each intervention and complication. Multivariate comparisons were made using generalized linear modeling, with post-onset LOS and hospital charges as dependent variables and study center (coded as dummy variables) as the main covariate, adjusting for age, sex, Charlson comorbidity score, discharge DRG weight, organ dysfunction (ARDS, uncontrolled hemorrhage, moderate or severe liver disease, or acute renal failure), and service (surgical versus medical) at onset of sepsis. The same adjustment of variables was applied in logistic regression models to compare the likelihood that a sepsis patient at a particular center received an intervention and experienced a complication, arbitrarily choosing center #1 as the reference center. Because patients within the same center could share some common characteristics, generalized estimating equation regression models were used to perform the analyses on the comparisons of resource use between patients with and without use of modalities. The r2 value was calculated for each model. All analyses were performed using the SAS statistical package (SAS Institute, Inc., Cary, NC, USA) [26].
Results
The study cohort included 1028 admissions with severe sepsis distributed across the eight participating centers, ranging from 91 to 261 admissions with severe sepsis episodes per study center (Table 1). The study patients were predominantly males in six of the eight centers, and the mean ages ranged from 55 to 60 years across the centers. The mean Charlson score and discharge DRG weight were 2.6 (range 2.3–2.9) and 4.8 (range 3.7–6.0), respectively. Almost half of the patients with severe sepsis underwent surgery during their hospital stay. The percentage undergoing organ transplantation varied from 0.4–11.3% across centers. The proportion of patients who had an infectious etiology for their primary discharge DRG varied from 6.1% to 23.5% among the participating centers.
Table 1.
Patient characteristics by participating center
| Center | |||||||||
| C1 | C2 | C3 | C4 | C5 | C6 | C7 | C8 | All | |
| Admissions with episode (% multiple episodes) | 261 (9.3) | 143 (5.6) | 97 (7.2) | 109 (13.2) | 104 (6.5) | 91 (5.5) | 97 (9.3) | 126 (9.9) | 1028 (8.5) |
| Patients (n) | 253 | 142 | 97 | 109 | 100 | 91 | 96 | 123 | 1011 |
| Episodes (n) | 298 | 156 | 111 | 139 | 113 | 104 | 107 | 145 | 1173 |
| Age (mean ± SD) | 55 ± 19 | 60 ± 20 | 57 ± 17 | 56 ± 19 | 59 ± 20 | 60 ± 16 | 60 ± 19 | 59 ± 20 | 58 ± 19 |
| Males (%) | 58.9 | 64.6 | 62.7 | 46.2 | 65.1 | 57.7 | 41.2 | 66.4 | 58.2 |
| On surgical service at onset (%) | 38.8 | 38.3 | 41.4 | 38.0 | 37.5 | 32.3 | 35.8 | 35.7 | 37.5 |
| Charlson score (mean ± SD) | 2.3 ± 2.6 | 2.5 ± 2.9 | 2.8 ± 2.6 | 2.8 ± 3.1 | 2.7 ± 2.8 | 2.9 ± 2.9 | 2.9 ± 3.1 | 2.3 ± 2.3 | 2.6 ± 2.8 |
| Self-pay or Medicaid (%) | 10.0 | 17.4 | 21.8 | 20.4 | 20.0 | 16.2 | 10.2 | 11.9 | 15.0 |
| In ICU at onset of sepsis (%) | 57.0 | 39.4 | 62.7 | 41.0 | 37.0 | 62.7 | 57.1 | 54.1 | 50.8 |
| Discharge DRG weight (mean ± SD) | 5.0 ± 6.1 | 3.9 ± 5.7 | 5.5 ± 5.6 | 4.9 ± 6.4 | 3.7 ± 4.6 | 4.4 ± 5.1 | 6.0 ± 8.2 | 5.4 ± 6.4 | 4.8 ± 6.1 |
| Primary discharge DRG (%) | |||||||||
| Surgery | 48.3 | 41.1 | 56.7 | 50.7 | 43.0 | 39.3 | 41.6 | 48.3 | 46.1 |
| Cancer | 7.0 | 5.5 | 5.8 | 1.4 | 0.8 | 3.5 | 9.1 | 7.4 | 5.4 |
| Transplant | 3.0 | 1.0 | 0.4 | 3.5 | 1.9 | 5.2 | 11.3 | 2.1 | 3.3 |
| AIDS | 5.9 | 0 | 3.4 | 14.2 | 7.7 | 6.0 | 0 | 4.9 | 5.2 |
| Infectious | 15.1 | 23.5 | 11.1 | 6.1 | 19.2 | 20.9 | 14.0 | 21.0 | 16.6 |
| Other | 20.7 | 28.9 | 22.6 | 24.1 | 27.5 | 25.1 | 24.0 | 16.3 | 23.4 |
| APACHE III chance of 28-day survival (mean ± SD) | 0.75 ± 0.24 | 0.73 ± 0.25 | 0.75 ± 0.20 | 0.72 ± 0.21 | 0.71 ± 0.24 | 0.69 ± 0.23 | 0.68 ± 0.28 | 0.63 ± 0.26 | 0.71 ± 0.24 |
| Organ dysfunction at onset (%) | |||||||||
| ARDS | 2.2 | 7.6 | 6.5 | 3.0 | 7.3 | 6.2 | 1.1 | 9.0 | 5.0 |
| Uncontrolled hemorrhage | 1.0 | 3.0 | 4.3 | 1.4 | 0.6 | 1.0 | 1.1 | 2.1 | 1.7 |
| Moderate or severe liver disease | 1.3 | 5.0 | 3.4 | 8.2 | 4.3 | 4.1 | 13.6 | 2.8 | 4.8 |
| Acute renal failure | 5.1 | 16.3 | 12.2 | 14.8 | 10.5 | 11.3 | 0 | 11.4 | 9.8 |
APACHE, Acute Physiology and Chronic Health Evaluation; ARDS, acute respiratory distress syndrome; C, center; DRG, diagnosis-related group; ICU, intensive care unit; SD, standard deviation.
Variation in resource use
There was significant variation in mean total charges among admissions with severe sepsis among the study centers, varying from $65 162 (median $42 802) to $244 293 (median $181 758; Table 2). The mean post-onset LOS varied from 16.1 days to 24.8 days (median range 9–16 days) among centers. Other measures of resource utilization including total LOS and LOS in the ICU were also significantly different across centers, with means of 22.7–36.9 days and 5.0–17.8 days, respectively. After adjusting for age, sex, Charlson comorbidity score, discharge DRG weight, organ dysfunction, and service at onset of sepsis, the variations in mean total charges and post-onset LOS remained significant, ranging from $69 429 ± $9562 to $237 898 ± $12 129, and from 15.9 ± 2.0 days to 24.2 ± 1.8 days, respectively (Table 3).
Table 2.
Resource utilization among patients with severe sepsis in eight centers
| Site Total Charges | Total LOS | Post-onset LOS | LOS in ICU | |
| C1 (n = 261) | ||||
| Mean ± SD | 92,201 ± 120,457 | 28.0 ± 29.0 | 19.9 ± 22.6 | 12.2 ± 22.4 |
| Median (25th, 75th percentile) | 68,001 (29,220, 132,236) | 21 (11, 35) | 14 (8, 25) | 7 (2, 18) |
| C2 (n = 143) | ||||
| Mean ± SD | 67,240 ± 107,234 | 22.7 ± 30.5 | 17.1 ± 25.1 | 5.0 ± 11.8* |
| Median (25th, 75th percentile) | 42,219 (13,733, 84,361) | 15 (7, 30) | 11 (5, 21) | 1 (0, 7) |
| C3 (n = 97) | ||||
| Mean ± SD | 244,293 ± 217,219* | 30.0 ± 24.3 | 21.4 ± 23.3 | 17.8 ± 21.1 |
| Median (25th, 75th percentile) | 181,758 (90,044, 374,095) | 28 (10, 42) | 17 (4, 32) | 11 (3, 33) |
| C4 (n = 109) | ||||
| Mean ± SD | 65,162 ± 84,024 | 30.6 ± 30.0 | 21.0 ± 24.0 | 14.1 ± 24.9 |
| Median (25th, 75th percentile) | 42,802 (24,415, 100,715) | 24 (14, 39) | 16 (9, 25) | 8 (3, 22) |
| C5 (n = 104) | ||||
| Mean ± SD | 72,970 ± 129,237 | 24.4 ± 33.6 | 19.7 ± 31.6 | 9.1 ± 20.7 |
| Median (25th, 75th percentile) | 44,846 (22,095, 112,674) | 17.5 (9, 36) | 13.5 (5, 25.5) | 4 (0, 18) |
| C6 (n = 91) | ||||
| Mean ± SD | 88,638 ± 93,098 | 24.4 ± 20.7 | 17.1 ± 17.6 | 12.1 ± 15.0 |
| Median (25th, 75th percentile) | 61,982 (26,340, 128,770) | 19 (11, 37) | 13 (5, 26) | 9 (2, 21) |
| C7 (n = 97) | ||||
| Mean ± SD | 194,986 ± 261,132* | 25.4 ± 34.3 | 16.1 ± 21.2 | 12.6 ± 18.0 |
| Median (25th, 75th percentile) | 92,714 (34 143, 209,007) | 14 (7, 28) | 9 (4, 18) | 6 (0, 13) |
| C8 (n = 126) | ||||
| Mean ± SD | 86,478 ± 89,866 | 36.9 ± 36.4* | 24.8 ± 29.4* | 13.0 ± 20.9 |
| Median (25th, 75th percentile) | 61,946 (25,698, 113,446) | 22.5 (13, 49) | 16 (7, 31) | 7 (1, 20) |
| All (n = 1028) | ||||
| Mean ± SD | 103,529 ± 153,692 | 27.7 ± 30.6 | 19.6 ± 24.7 | 11.5 ± 20.3 |
| Median (25th, 75th percentile) | 63,496 (26,366, 137,046) | 20 (10, 37) | 13 (6, 25) | 6 (1, 18) |
C, center; ICU, intensive care unit; length of stay. *P < 0.05, versus C1.
Table 3.
Adjusted mean hospital charges and post-onset length of stay in patients with severe sepsis
| Total charges ($) | Post-onset LOS (days) | |||
| Site | Adjusted mean ± SE | Difference ± SE | Adjusted mean ± SE | Difference ± SE |
| C1 | 96,272 ± 6493 | Reference | 20.1 ± 1.3 | Reference |
| C2 | 84,514 ± 8221 | -11,758 ± 10,537 | 19.7 ± 1.6 | -0.4 ± 2.0 |
| C3 | 237,898 ± 12,129 | 141,626 ± 13,758* | 20.9 ± 2.3 | 0.8 ± 2.7 |
| C4 | 69,429 ± 9562 | -26,843 ± 11,561* | 22.0 ± 1.8 | 1.9 ± 2.2 |
| C5 | 92,778 ± 10,084 | -3494 ± 12,001 | 22.4 ± 1.9 | 2.3 ± 2.3 |
| C6 | 100,461 ± 11,565 | 4189 ± 13,283 | 18.8 ± 2.2 | -1.3 ± 2.6 |
| C7 | 188,835 ± 10,385 | 92,563 ± 12,244 * | 15.9 ± 2.0 | -4.2 ± 2.4 |
| C8 | 83,043 ± 9484 | -13,229 ± 11,516 | 24.2 ± 1.8 | 4.1 ± 2.2 |
| r2 = 0.46, P < 0.001 | r2 = 0.22, P < 0.001 | |||
Adjusted means and r2 were calculated in the generalized linear models, including age, sex, Charlson comorbidity score, discharge diagnosis-related group weight, organ dysfunction, and service at onset of sepsis. C, center; LOS, length of stay; SE, standard error. * P < 0.05, versus C1.
Variation in therapeutic modality use
Interventions used frequently after the onset of severe sepsis included PACs (19.4%), ventilator support (21.8%), pressor support (45.8%), and albumin infusion (14.4%; Table 4). Although there were variations across medical centers for each therapeutic modality, variation profiles differed substantially among the modalities. Modalities with moderate variation profiles among the eight participating centers included PACs (varied 4.0-fold, 95% confidence interval [CI] of 2.6-fold to 6.4-fold; range 8.7% to 35.1%), ventilator support (varied 3.2-fold, 95% CI 2.1-fold to 4.9-fold; range 11.5% to 36.4%), and albumin infusion within 24 hours after sepsis onset (varied 4.9-fold, 95% CI 2.2-fold to 10.9-fold; range 6.0% to 29.1%). In contrast, use of pressor support varied only 1.9-fold (95% CI 1.4-fold to 2.6-fold) across centers, ranging from 31.4% to 60.4%, and antibiotics given within 24 hours after onset had the least variation, of 1.1-fold (95% CI 1.0-fold to 1.1-fold] and ranging from 87.0% to 98.5%. Use of hetastarch and use of dextran within 24 hours after onset in sepsis patients were the least common therapeutic modalities among the eight centers, with ranges of 0–6.8% and 0–6.2%, respectively. Of patients receiving pressors within 24 hours after sepsis onset, 28.3% received low-dose dopamine only (<5 μg/kg per min); the proportion varied 7-fold (95% CI 2.4-fold to 20.8-fold) across centers, ranging from 6.4% in center 5 to 44.8% in center 1 (data not shown).
Table 4.
Variation in therapeutic modalities use by center in patients with severe sepsis
| Center | ||||||||||
| Modality | C1 | C2 | C3 | C4 | C5 | C6 | C7 | C8 | All | P |
| PAC (%) | 8.7 | 10.3 | 25.7 | 30.6 | 25.1 | 24.2 | 15.5 | 35.1 | 19.4 | 0.001 |
| Ventilator support (%) | 11.5 | 19.6 | 36.4 | 34.1 | 29.3 | 20.1 | 12.9 | 28.3 | 21.8 | 0.001 |
| Pressor support after onset (%) | 41.8 | 31.4 | 53.7 | 51.8 | 47.6 | 42.5 | 60.4 | 50.7 | 45.8 | 0.001 |
| On albumin within 24 hours after onset (%) | 8.2 | 16.7 | 13.1 | 15.4 | 6.0 | 13.4 | 18.1 | 29.1 | 14.4 | 0.001 |
| On hetastarch within 24 hours after onset (%) | 0.6 | 0.5 | 0 | 6.3 | 0 | 0 | 0.3 | 6.8 | 1.8 | 0.001 |
| On dextran within 24 hours after onset (%) | 2.2 | 0 | 0 | 1.4 | 0 | 6.2 | 0 | 0.7 | 1.3 | 0.001 |
| Antibiotic given within 24 hours after onset (%) | 98.5 | 95.9 | 87.0 | 93.9 | 92.6 | 93.0 | 95.5 | 92.3 | 94.7 | 0.001 |
Data were collected at 28 days or discharge after onset of first sepsis episode. Percentages were calculated using number of admission with severe sepsis in each center as the denominator; P value for test % variation across sites. C, center; PAC, pulmonary artery catheter.
The results of models adjusting for age, sex, Charlson comorbidity score, discharge DRG weight, organ dysfunction, and service at onset of sepsis are shown in Fig. 1. Centers that had rates significantly higher than the reference center were centers 3, 4, 5, 6, and 8 for PAC use; centers 2, 3, 4, 5, and 8 for ventilator support; center 7 for pressor support; and centers 2, 7, and 8 for albumin infusion. In contrast, significantly lower rates for antibiotic use within 24 hours after onset were identified in centers 3, 4, 5, 6, 7, and 8.
Figure 1.
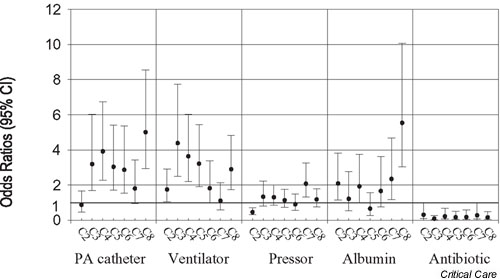
Variation in odds ratios (ORs) for therapeutic modalities. C1 was the reference center. ORs were calculated after adjusting for age, sex, Charlson comorbidity score and discharge diagnosis-related group weight, organ dysfunction, and service at onset of sepsis. Albumin, on albumin within 24 hours after onset; Antibiotic, antibiotic given within 24 hours after onset; CI, confidence interval; PA, pulmonary artery; Pressor, pressor support; Ventilator, ventilator support.
Variation in outcomes
Among all patients with severe sepsis, frequent complications included shock (45.4%), renal failure (19.7%), CNS dysfunction (18.9%), liver failure (12.2%), DIC (10.3%), ARDS (9.1%), and death (30.0%) within 28 days after sepsis onset (Table 5). There was significant variation in each type of complication across the eight centers, with ranges of 23.9–70.0% for shock, 9.5–35.0% for renal failure, 0.3–37.5% for CNS dysfunction, 4.1–27.9% for liver failure, 5.5–15.0% for DIC, 2.8–21.8% for ARDS, and 19.3–49.1% for 28-day mortality rate.
Table 5.
Outcomes among patients with severe sepsis
| Center | ||||||||||
| Outcome | C1 | C2 | C3 | C4 | C5 | C6 | C7 | C8 | All | P |
| ARDS (%) | 5.5 | 10.7 | 13.2 | 7.3 | 8.4 | 6.6 | 2.8 | 21.8 | 9.1 | 0.001 |
| Renal failure (%) | 9.5 | 15.2 | 24.5 | 35.0 | 32.5 | 14.8 | 21.9 | 20.4 | 19.7 | 0.001 |
| Liver failure (%) | 11.0 | 9.6 | 9.9 | 27.9 | 12.7 | 4.1 | 5.0 | 15.7 | 12.2 | 0.001 |
| DIC (%) | 9.4 | 14.6 | 14.0 | 15.0 | 10.4 | 7.2 | 5.9 | 5.5 | 10.3 | 0.024 |
| Shock (%) | 23.9 | 37.2 | 45.6 | 70.0 | 52.8 | 42.7 | 57.8 | 63.8 | 45.4 | 0.001 |
| CNS dysfunction (%) | 0.3 | 15.2 | 33.8 | 36.1 | 35.8 | 22.4 | 0.8 | 37.5 | 18.9 | 0.001 |
| Death (%) | 19.3 | 26.0 | 49.1 | 31.0 | 28.3 | 40.2 | 36.6 | 34.0 | 30.0 | 0.001 |
Data were collected at 28 days or discharge after onset of first sepsis episode. Percentages were calculated using number of admission with severe sepsis in each center as the denominator; P value for test % variation across sites. ARDS, acute respiratory distress syndrome; C, center; CNS, central nervous system; DIC, disseminated intravascular coagulation.
Figure 2 shows the results of differences in rates of sepsis complications adjusted for age, sex, Charlson comorbidity score, discharge DRG weight, organ dysfunction, and service at onset of sepsis. Analysis of the 28-day in-hospital adjusted mortality rates revealed 3-fold variability among centers. Compared with the reference center, higher rates were found for ARDS among one center (C8), for renal failure among five centers (C3, C4, C5, C7, and C8), for liver failure among one center (C4), for shock among six centers (C3, C4, C5, C6, C7, and C8), and for 28-day mortality among four centers (C3, C6, C7, and C8).
Figure 2.
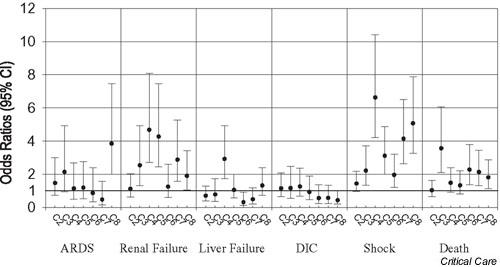
Variation in odds ratios (ORs) for outcomes. C1 was the reference center. ORs were calculated after adjusting for age, sex, Charlson comorbidity score, and discharge diagnosis-related group weight, organ dysfunction, and service at onset of sepsis. ARDS, acute respiratory distress syndrome; CI, confidence interval; DIC, disseminated intravascular coagulation.
Also, a significant inverse correlation was seen between antibiotic use within 24 hours after onset of episode and the 28-day mortality rates (r2 = 0.72; P = 0.01; Fig. 3). This correlation remained significant even after excluding patients with 'do not resuscitate' orders (r2 = 0.63; P = 0.02). Significant associations were not found between 28-day mortality and the use of other modalities across the eight centers (data not shown).
Figure 3.
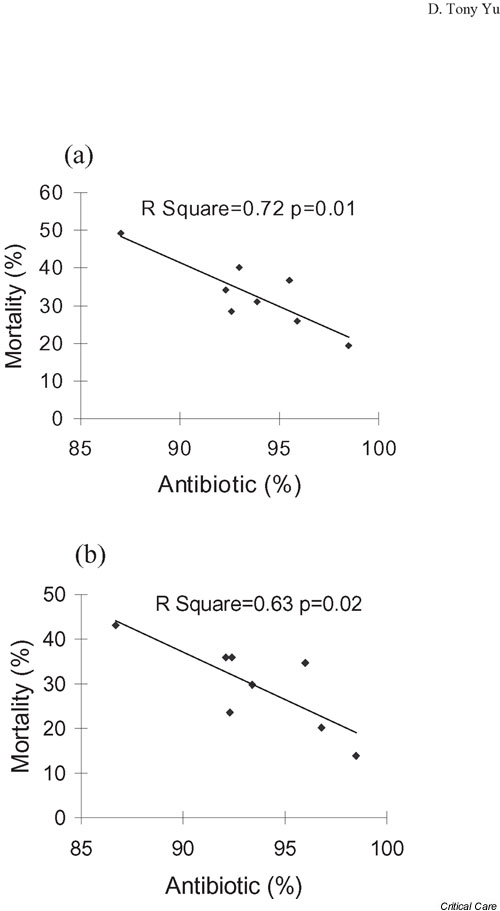
Relationship between mortality rate and antibiotic use. This plot displays the mortality rate versus whether an antibiotic was given within 24 hours after sepsis onset in the eight centers. The results were significant for (a) all patients (n = 1028) and (b) for the subgroup (n = 924) after excluding those with a 'do not resuscitate' order (n = 104).
Relation between resource use and modalities
Comparisons of resource use among patients treated with specific modalities and those who were not showed that administration of antibiotics within 24 hours after sepsis onset had no impact on any of the LOS categories, but was associated with total and daily hospital charges (Table 6). Use of other modalities (including albumin infusion) was associated with increased resource use for daily hospital charges. Total hospital charges and LOS in the ICU were significantly higher among patients who were given pressor support and/or albumin infusion after onset of sepsis as compared with those who were not. Increased post-onset LOS and LOS in the ICU were associated with use of PACs. No associations were found between total LOS and the modalities studied.
Table 6.
Comparison of resource use in sepsis patients with and without use of specific modalities
| Modalities | |||||||||||||||
| PAC | Ventilator support | Pressor support | On albumin | Antibiotic within 24 hours | |||||||||||
| Resources | No | Yes | r2 | No | Yes | r2 | No | Yes | r2 | No | Yes | r2 | No | Yes | r2 |
| Total charges (× $1000) | 114 | 131 | 0.35 | 116 | 122 | 0.35 | 107 | 146* | 0.37 | 114 | 141* | 0.35 | 80 | 117* | 0.35 |
| Hospital charges/day (× $1000) | 2.8 | 3.7* | 0.14 | 2.8 | 4.3* | 0.16 | 2.6 | 4.6* | 0.23 | 2.9 | 4.0* | 0.14 | 2.4 | 2.9* | 0.13 |
| Post-onset LOS (days) | 19.8 | 23.6* | 0.22 | 20.3 | 21.4 | 0.22 | 20.1 | 21.0 | 0.21 | 19.9 | 23.9 | 0.22 | 20.0 | 20.6 | 0.21 |
| LOS in ICU (days) | 7.1 | 11.1* | 0.41 | 7.5 | 9.3 | 0.40 | 8.4 | 13.1* | 0.42 | 7.4 | 13.0* | 0.41 | 6.3 | 7.4 | 0.40 |
| LOS (days) | 28.3 | 32.0 | 0.26 | 29.3 | 28.6 | 0.25 | 28.9 | 29.4 | 0.25 | 28.6 | 32.1 | 0.25 | 27.2 | 29.2 | 0.25 |
Mean and r2 values were calculated from the generalized estimating equation regression models adjusting for age, sex, Charlson comorbidity score, discharge diagnosis-related group weight, organ dysfunction and service at onset of sepsis. Center was a subject-effect variable. *Significant difference from the group without using modality.
Discussion
Overall, we found moderate variation in resource and therapeutic modality use among patients with severe sepsis in academic centers. In general, this variation had little relationship to clinical outcomes, although clinical outcomes varied substantially between the centers. The exception is that, even adjusting for potential confounders, early antibiotic therapy was associated with a lower 28-day mortality rate. Increased use of some of the therapeutic modalities was associated with greater resource utilization.
Prior studies have shown variability in the use of therapeutic modalities (including PACs, ventilator, and intravenous vasoactive and inotropic agents) among ICU patients [21,27] and different impacts on outcomes and use of hospital resources associated with these treatments [16-18,28,29]. In general, with the exception of the recent trial demonstrating benefit of early goal-directed treatment involving oxygenation (including ventilatory support if necessary) and blood pressure (including intravenous pressors if necessary) [30], few data are available that demonstrate that these modalities improve outcomes.
Colloids and crystalloid are equally effective in restoring tissue perfusion in patients with septic shock [31]. However, the choice of fluid has considerable cost implications: colloids cost more for volume replacement. Largely inappropriate use of colloids was found in a survey of academic health centers despite guidelines from a US hospital consortium recommending that colloids be used in hemorrhagic shock only until blood products become available, and in nonhemorrhagic shock only if an initial infusion with crystalloid is insufficient [32,33]. A recent systematic review of randomized controlled trials comparing colloids and crystalloid solutions for volume replacement found that resuscitation with colloids in critically ill patients was actually associated with an increased risk of mortality [10].
In addition, vasopressors have been recommended to achieve end-points of hemodynamic, and normal or supranormal oxygen transport variables in sepsis patients who remain hypotensive despite adequate volume therapy. However, after reviewing the literature on the use of vasopressors in patients with sepsis syndrome, Rudis and colleagues [13] found that catecholamine therapy resulting in increased hemodynamic and oxygen transport measures did not change the overall mortality, with the exception of two instances in which epinephrine (adrenaline) and norepinephrine (noradrenaline) were given alone after volume repletion. The results from a recent trial of low-dose dopamine in critically ill patients, who had systemic inflammatory response syndrome and were at risk for renal failure, did not show benefit in renal protection and survival from the treatment [34]. However, a very recent trial of early goal-directed therapy in treatment of sepsis did demonstrate substantial improvement in outcome [30].
Another technology is monitoring central pressures by PAC, which is often used in ICUs to assess the effect of pharmacotherapy on the cardiac index in patients with septic shock. This is done even though the efficacy of the PAC has never been demonstrated convincingly in a large randomized controlled trial. A number of randomized controlled trials have examined the effectiveness of PACs or PAC-guided strategies in sepsis patients [16-20,35,36] and other specific patient groups [29,37-43], with conflicting results. A recent case mix adjusted observational study of a large sample of ICU patients found that PAC use was associated with increased risk for mortality and resource use [28].
Mechanical ventilation in patients with ARDS has been reported to be associated with pneumonia and lung injury [44-47]. Properly constructed trials are still needed to define the best use of mechanical ventilation in sepsis, although early ventilatory support was a component of the recent early goal-directed intervention by Rivers and coworkers [30].
Use of some therapeutic modalities does appear correlated with organizational characteristics. For example, results from a national ICU survey conducted by Greoger and colleagues [21] showed that more technologies were used in surgical units as compared with other units, as well as in larger versus smaller hospitals, and in university-affiliated facilities. In addition, a large prospective multicenter study evaluated the differences in ICU characteristics and performance among teaching and nonteaching hospitals [48]. The results of that study revealed more frequent use of monitoring and therapeutic interventions, and greater resource utilization in teaching hospital ICUs. Also, a recent report from a large retrospective database study of ICU patients [27] confirmed that organizational characteristics of ICUs were associated with variation in PAC use. In addition, that report indicated that economic incentives and insurance coverage, as well as clinical variables, were associated with PAC use. Similarly, much of the variation in resource use and modality use across centers in the present study may not be due to patient-related factors, but rather to organizational factors and physician beliefs.
We did find a number of relationships between modality use and resource utilization. In particular, there were associations between LOS in the ICU and use of PACs, pressor support, and albumin infusion. These may probably be accounted for in part by the fact that patients with longer LOS in the ICU were more likely to be treated with supportive technologies. In general, however, modalities with higher variation profiles reflect areas of greater disagreement in terms of treatment decision-making. Such areas may be fertile for additional investigation.
It is striking that delay in antibiotic therapy was associated with a higher mortality rate and that some delays were present in this very ill population. Delays are common in American medicine [49], even in urgent situations such as treatment of life-threatening laboratory abnormalities [50]. Although we do not know the causes of delays in antibiotic initiation, they appeared more common in some institutions than in others. We speculate that some may have occurred because of problems related to crowding; for example, emergency rooms or ICUs might have been full, resulting in delayed transfer, or the clinical importance of changes in vital signs might not have been recognized. Approaches such as protocols for early recognition and treatment of severe sepsis, and facilitation of medication orders that are really needed urgently may be helpful. Guidelines for the management of severe sepsis are probably most applicable for certain treatment modalities, such as PACs, albumin, dextran, and hetastarch. Even use of organ failure treatment modalities such as ventilator support and renal replacement therapies might be different if such guidelines were developed.
An important underlying issue is why variation occurs [51,52]. It should not be surprising that it is present in this domain, because the pioneering work of Wennberg [52] identified variation across a broad array of domains. One of the major causes of variation is probably physician uncertainty regarding what interventions are truly beneficial, and clinician beliefs, where training occurred, and regional practices probably also play roles. Uncertainty may be especially problematic in conditions such as sepsis, in which mortality is high, and clinicians strongly want to do everything possible. Variation is likely to diminish as more evidence becomes available and is brought to the point of care. Analysis of variation can be very useful for identifying areas of high uncertainty [52].
This study has a number of limitations. We had information only on hospital charges, rather than hospital costs. We did not have information on organizational variables such as organizational setting, staffing, or leadership of practices, and the study included only academic centers. Also, our data did not include the indications for use of therapeutic modalities.
In conclusion, significant variations were present in hospital resource use and patient outcome among sepsis patients across eight academic medical centers. In general, variation in therapeutic modality use did not correlate with clinical outcomes, suggesting that some use of these modalities may be of limited value, and that further evaluation of these modalities is warranted. The exception was that delay in giving antibiotics to sepsis patients was associated with a higher 28-day mortality rate. Approaches to eliminate these delays may improve outcomes.
Competing interests
None declared.
Key messages
• Moderate variation in resource use and use of technologies was present in treatment of severe sepsis among academic centers
• This variation presisted even after adjusting for potential confounders
• Increased use of some therapeutic modalities such as albumin infusion was associated with greater resource utilization
• Among therapeutic modalities, only early antibiotic use was associated with lower 28-day mortality
• Taken together, these data suggest that some therapeutic modalities may be of limited value in sepsis treatment and that further evaluation is warranted
Abbreviations
ARDS = acute respiratory distress syndrome; CI = confidence interval; CNS = central nervous system; DIC = disseminated intravascular coagulation; DRG = diagnosis-relative group; ICU = intensive care unit; LOS = length of stay; PAC = pulmonary artery catheter.
See related Commentary http://ccforum.com/content/7/3/211
References
- Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–1310. doi: 10.1097/00003246-200107000-00002. [DOI] [PubMed] [Google Scholar]
- Niederman MS, Fein AM. Sepsis syndrome, the adult respiratory distress syndrome, and nosocomial pneumonia. A common clinical sequence. Clin Chest Med. 1990;11:633–656. [PubMed] [Google Scholar]
- Rangel-Frausto MS, Pittet D, Costigan M, Hwang T, Davis CS, Wenzel RP. The natural history of the systemic inflammatory response syndrome (SIRS). A prospective study. JAMA. 1995;273:117–123. [PubMed] [Google Scholar]
- Wenzel RP. Anti-endotoxin monoclonal antibodies: a second look. N Engl J Med. 1992;326:1151–1153. doi: 10.1056/NEJM199204233261710. [DOI] [PubMed] [Google Scholar]
- Pittet D, Thievent B, Wenzel RP, Li N, Gurman G, Suter PM. Importance of pre-existing co-morbidities for prognosis of septicemia in critically ill patients. Intensive Care Med. 1993;19:265–272. doi: 10.1007/BF01690546. [DOI] [PubMed] [Google Scholar]
- Pittet D, Rangel-Frausto S, Li N, Tarara D, Costigan M, Rempe L, Jebson P, Wenzel RP. Systemic inflammatory response syndrome, sepsis, severe sepsis and septic shock: incidence, morbidities and outcomes in surgical ICU patients. Intensive Care Med. 1995;21:302–309. doi: 10.1007/BF01705408. [DOI] [PubMed] [Google Scholar]
- Brun-Buisson C, Doyon F, Carlet J, Dellamonica P, Gouin F, Lepoutre A, Mercier JC, Offenstadt G, Regnier B. Incidence, risk factors, and outcome of severe sepsis and septic shock in adults. A multicenter prospective study in intensive care units. French ICU Group for Severe Sepsis. JAMA. 1995;274:968–974. [PubMed] [Google Scholar]
- Reinhart K, Sakka SG, Meier-Hellmann A. Haemodynamic management of a patient with septic shock. Eur J Anaesthesiol. 2000;17:6–17. doi: 10.1046/j.1365-2346.2000.00575.x. [DOI] [PubMed] [Google Scholar]
- Roberts I. Human albumin administration in critically ill patients: systematic review of randomised controlled trials. Cochrane Injuries Group Albumin Reviewers. BMJ. 1998;317:235–240. doi: 10.1136/bmj.317.7153.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schierhout G, Roberts I. Fluid resuscitation with colloid or crystalloid solutions in critically ill patients: a systematic review of randomised trials. BMJ. 1998;316:961–964. doi: 10.1136/bmj.316.7136.961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisonni RS, Holtgrave DR, Lawler F, Marley DS. Colloids versus crystalloids in fluid resuscitation: an analysis of randomized controlled trials. J Fam Pract. 1991;32:387–390. [PubMed] [Google Scholar]
- Velanovich V. Crystalloid versus colloid fluid resuscitation: a meta-analysis of mortality. Surgery. 1989;105:65–71. [PubMed] [Google Scholar]
- Rudis MI, Basha MA, Zarowitz BJ. Is it time to reposition vasopressors and inotropes in sepsis? Crit Care Med. 1996;24:525–537. doi: 10.1097/00003246-199603000-00026. [DOI] [PubMed] [Google Scholar]
- Shama VK, Dellinger RP. The International Sepsis Forum's controversies in sepsis: my initial vasopressor agent in septic shock is norepinephrine rather that dopamine. Crit Care. 2003;7:3–5. doi: 10.1186/cc1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vincent JL, de Backer D. The International Sepsis Forum's controversies in sepsis: my initial vasopressor agent in septic shock is dopamine rather that norepinephrine. Crit Care. 2003;7:6–8. doi: 10.1186/cc1851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu M, Takanishi D, Myers SA, Takiguchi SA, Severino R, Hasaniya N, Levy MM, McNamara JJ. Frequency of mortality and myocardial infarction during maximizing oxygen delivery: a prospective, randomized trial. Crit Care Med. 1995;23:1025–1032. doi: 10.1097/00003246-199506000-00006. [DOI] [PubMed] [Google Scholar]
- Gattinoni L, Brazzi L, Pelosi P, Latini R, Tognoni G, Pesenti A, Fumagalli R. A trial of goal-oriented hemodynamic therapy in critically ill patients. SvO2 Collaborative Group. N Engl J Med. 1995;333:1025–1032. doi: 10.1056/NEJM199510193331601. [DOI] [PubMed] [Google Scholar]
- Hayes MA, Timmins AC, Yau EH, Palazzo M, Hinds CJ, Watson D. Elevation of systemic oxygen delivery in the treatment of critically ill patients. N Engl J Med. 1994;330:1717–1722. doi: 10.1056/NEJM199406163302404. [DOI] [PubMed] [Google Scholar]
- Yu M, Levy MM, Smith P, Takiguchi SA, Miyasaki A, Myers SA. Effect of maximizing oxygen delivery on morbidity and mortality rates in critically ill patients: a prospective, randomized, controlled study. Crit Care Med. 1993;21:830–838. doi: 10.1097/00003246-199306000-00009. [DOI] [PubMed] [Google Scholar]
- Tuchschmidt J, Fried J, Astiz M, Rackow E. Elevation of cardiac output and oxygen delivery improves outcome in septic shock. Chest. 1992;102:216–220. doi: 10.1378/chest.102.1.216. [DOI] [PubMed] [Google Scholar]
- Groeger JS, Guntupalli KK, Strosberg M, Halpern N, Raphaely RC, Cerra Fkaye W. Descriptive analysis of critical care units in the United States: patient characteristics and intensive care unit utilization. Crit Care Med. 1993;21:279–291. doi: 10.1097/00003246-199302000-00022. [DOI] [PubMed] [Google Scholar]
- Sands KE, Bates DW, Lanken PN, Graman PS, Hibberd PL, Kahn KL, Parsonnet J, Panzer R, Orav EJ, Snydman DR. Epidemiology of sepsis syndrome in eight academic medical centers. JAMA. 1997;278:234–240. [PubMed] [Google Scholar]
- Bates DW, Sands K, Miller E, Lanken PN, Hibberd PL, Graman PS, Schwartz JS, Kahn K, Snydman DR, Parsonnet J, Moore R, Black E, Johnson BL, Jha A, Platt R. Predicting bacteremia in patients with sepsis syndrome. J Infect Dis. 1997;176:1538–1551. doi: 10.1086/514153. [DOI] [PubMed] [Google Scholar]
- Bone RC. Sepsis, the sepsis syndrome, multi-organ failure: a plea for comparable definitions. Ann Intern Med. 1991;114:332–333. doi: 10.7326/0003-4819-114-4-332. [DOI] [PubMed] [Google Scholar]
- Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal populations: development and validation. J Chronic Dis. 1987;40:373–383. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- SAS Procedures Guide, Release 612 Edition. Cary, NC: SAS Institute Inc; 1996. SAS Institute, Inc. [Google Scholar]
- Rapoport J, Teres D, Steingrub J, Higgins T, McGee W, Lemeshow S. Patient characteristics and ICU organizational factors that influence frequency of pulmonary artery catheterization. JAMA. 2000;283:2559–2567. doi: 10.1001/jama.283.19.2559. [DOI] [PubMed] [Google Scholar]
- Connors AF, Jr, Speroff T, Dawson NV, Thomas C, Harrell FE, Jr, Wagner D, Desbiens N, Goldman L, Wu AW, Califf RM, Fulkerson WJ, Jr, Vidaillet H, Broste S, Bellamy P, Lynn J, Knaus WA. The effectiveness of right heart catheterization in the initial care of critically ill patients. SUPPORT Investigators. JAMA. 1996;276:889–897. doi: 10.1001/jama.276.11.889. [DOI] [PubMed] [Google Scholar]
- Bishop MH, Shoemaker WC, Appel PL, Meade P, Ordog GJ, Wasserberger J, Wo CJ, Rimle DA, Kram HB, Umali R, et al. Prospective, randomized trial of survivor values of cardiac index, oxygen delivery, and oxygen consumption as resuscitation endpoints in severe trauma. J Trauma. 1995;38:780–787. doi: 10.1097/00005373-199505000-00018. [DOI] [PubMed] [Google Scholar]
- Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B, Peterson E, Tomlanovich M, for the Early Goal-Directed Therapy Collaborative Group Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med. 2001;345:1368–1377. doi: 10.1056/NEJMoa010307. [DOI] [PubMed] [Google Scholar]
- Rackow EC, Falk JL, Fein IA, Siegel JS, Packman MI, Haupt MT, Kaufman BS, Putnam D. Fluid resuscitation in circulatory shock: a comparison of the cardiorespiratory effects of albumin, hetastarch, and saline solutions in patients with hypovolemic and septic shock. Crit Care Med. 1983;11:839–850. [PubMed] [Google Scholar]
- Yim JM, Vermeulen LC, Erstad BL, Matuszewski KA, Burnett DA, Vlasses PH. Albumin and nonprotein colloid solution use in US academic health centers. Arch Intern Med. 1995;155:2450–2455. [PubMed] [Google Scholar]
- Vermeulen LCJ, Ratko TA, Erstad BL, Brecher ME, Matuszewski KA. A paradigm for consensus. The University Hospital Consortium guidelines for the use of albumin, nonprotein colloid, and crystalloid solutions. Arch Intern Med. 1995;155:373–379. doi: 10.1001/archinte.155.4.373. [DOI] [PubMed] [Google Scholar]
- Bellomo R, Chapman M, Finfer S, Hickling K, Myburgh J. Low-dose dopamine in patients with early renal dysfunction: a placebo-controlled randomised trial. Australian and New Zealand Intensive Care Society (ANZICS) Clinical Trials Group. Lancet. 2000;356:2139–2143. doi: 10.1016/s0140-6736(00)03495-4. [DOI] [PubMed] [Google Scholar]
- Durham RM, Neunaber K, Mazuski JE, Shapiro MJ, Baue AE. The use of oxygen consumption and delivery as endpoints for resuscitation in critically ill patients. J Trauma. 1996;41:32–39. doi: 10.1097/00005373-199607000-00007. [DOI] [PubMed] [Google Scholar]
- Boyd O, Grounds RM, Bennett ED. A randomized clinical trial of the effect of deliberate perioperative increase of oxygen delivery on mortality in high-risk surgical patients. JAMA. 1993;270:2699–2707. [PubMed] [Google Scholar]
- Valentine RJ, Duke ML, Inman MH, Grayburn PA, Hagino RT, Kakish HB, Clagett GP. Effectiveness of pulmonary artery catheters in aortic surgery: a randomized trial. J Vasc Surg. 1998;27:203–211. doi: 10.1016/s0741-5214(98)70351-9. [DOI] [PubMed] [Google Scholar]
- Bender JS, Smith-Meek MA, Jones CE. Routine pulmonary artery catheterization does not reduce morbidity and mortality of elective vascular surgery: results of a prospective, randomized trial. Ann Surg. 1997;226:229–236. doi: 10.1097/00000658-199709000-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleming A, Bishop M, Shoemaker W, Appel P, Sufficool W, Kuvhenguwha A, Kennedy F, Wo CJ. Prospective trial of supra-normal values as goals of resuscitation in severe trauma. Arch Surg. 1992;127:1175–1179. doi: 10.1001/archsurg.1992.01420100033006. [DOI] [PubMed] [Google Scholar]
- Guyatt G. A randomized control trial of right-heart catheterization in critically ill patients. Ontario Intensive Care Study Group. J Intensive Care Med. 1991;6:91–95. doi: 10.1177/088506669100600204. [DOI] [PubMed] [Google Scholar]
- Berlauk JF, Abrams JH, Gilmour IJ, O'Connor SR, Knighton DR, Cerra FB. Preoperative optimization of cardiovascular hemodynamics improves outcome in peripheral vascular surgery. A prospective, randomized clinical trial. Ann Surg. 1991;214:289–297. doi: 10.1097/00000658-199109000-00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joyce WP, Provan JL, Ameli FM, McEwan MM, Jelenich S, Jones DP. The role of central haemodynamic monitoring in abdominal aortic surgery. A prospective randomised study. Eur J Vasc Surg. 1990;4:633–636. doi: 10.1016/s0950-821x(05)80820-5. [DOI] [PubMed] [Google Scholar]
- Isaacson IJ, Lowdon JD, Berry AJ, Smith RB, Knos GB, Weitz FI, Ryan K. The value of pulmonary artery and central venous monitoring in patients undergoing abdominal aortic reconstructive surgery: a comparative study of two selected, randomized groups. J Vasc Surg. 1990;12:754–760. doi: 10.1067/mva.1990.24456. [DOI] [PubMed] [Google Scholar]
- Anonymous International consensus conferences in intensive care medicine. Ventilator-associated lung injury in ARDS. American Thoracic Society, European Society of Intensive Care Medicine, Societe de Reanimation Langue Francaise. Intensive Care Med. 1999;25:1444–1452. [PubMed] [Google Scholar]
- Morehead RS, Pinto SJ. Ventilator-associated pneumonia. Arch Intern Med. 2000;160:1926–1936. doi: 10.1001/archinte.160.13.1926. [DOI] [PubMed] [Google Scholar]
- Markowicz P, Wolff M, Djedaini K, Cohen Y, Chastre J, Delclaux C, Merrer J, Herman B, Veber B, Fontaine A, Dreyfuss D. Multi-center prospective study of ventilator-associated pneumonia during acute respiratory distress syndrome. Incidence, prognosis, and risk factors. ARDS Study Group. Am J Respir Crit Care Med. 2000;161:1942–1948. doi: 10.1164/ajrccm.161.6.9909122. [DOI] [PubMed] [Google Scholar]
- Livingston DH. Prevention of ventilator-associated pneumonia. Am J Surg. 2000;179(suppl):12S–17S. [PubMed] [Google Scholar]
- Zimmerman JE, Shortell SM, Knaus WA, Rousseau DM, Wagner DP, Gillies RR, Draper EA, Devers K. Value and cost of teaching hospitals: a prospective, multicenter, inception cohort study. Crit Care Med. 1993;21:1432–1442. doi: 10.1097/00003246-199310000-00009. [DOI] [PubMed] [Google Scholar]
- Institute of Medicine . Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, DC: National Academy Press; 2001. [PubMed] [Google Scholar]
- Kuperman GJ, Boyle D, Jha A, Rittenberg E, Ma'Luf N, Tanasijevic MJ, Teich JM, Winkelman J, Bates DW. How promptly are inpa-tients treated for critical laboratory results? J Am Med Inform Assoc. 1998;5:112–119. doi: 10.1136/jamia.1998.0050112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McPherson K, Wennberg JE, Hovind OB, Clifford P. Small-area variation in the use of common surgical procedures: an international comparison of New England, England and Norway. N Engl J Med. 1982;307:1310–1314. doi: 10.1056/NEJM198211183072104. [DOI] [PubMed] [Google Scholar]
- Wennberg JE. Understanding geographic variation in health care delivery. N Engl J Med. 1999;340:52–53. doi: 10.1056/NEJM199901073400111. [DOI] [PubMed] [Google Scholar]


