Abstract
Acute ischemic injury and chronic cardiomyopathies damage healthy heart tissue. Dead cells are gradually replaced by a fibrotic scar, which disrupts the normal electromechanical continuum of the ventricular muscle and compromises its pumping capacity. Recent studies in animal models of ischemic cardiomyopathy suggest that transplantation of various stem cell preparations can improve heart recovery after injury. The first clinical trials in patients produced some encouraging results, showing modest benefits. Most of the positive effects are probably because of a favorable paracrine influence of stem cells on the disease microenvironment. Stem cell therapy attenuates inflammation, reduces apoptosis of surrounding cells, induces angiogenesis, and lessens the extent of fibrosis. However, little new heart tissue is formed. The current challenge is to find ways to improve the engraftment, long-term survival and appropriate differentiation of transplanted stem cells within the cardiovascular tissue. Hence, there has been a surge of interest in pluripotent stem cells with robust cardiogenic potential, as well as in the inherent repair and regenerative mechanisms of the heart. Recent discoveries on the biology of adult stem cells could have relevance for cardiac regeneration. Here, we discuss current developments in the field of cardiac repair and regeneration, and present our ideas about the future of stem cell therapy.
The clinical problem
Ischemic and non-ischemic cardiomyopathies eventually lead to left ventricular dysfunction and heart failure. Heart failure impedes quality of life, decreases life expectancy and increases medical costs dramatically, and thus constitutes a major health problem (McMurray and Pfeffer, 2005). As the population ages, the prevalence of heart failure will increase. Current therapy provides some survival benefit and improvement of symptoms, but cannot reverse the condition of cardiac tissue from a diseased to a healthy state. Recent developments in stem cell biology and regenerative medicine may offer ways to manage heart failure by replacing damaged or lost cardiac muscle with healthy tissue, and thus improve the quality of life and survival in patients with various cardiomyopathies.
The disease process of ischemic cardiomyopathy
Myocardial infarction usually results from coronary artery occlusion owing to acute atherosclerotic plaque rupture and platelet aggregation, which leads to thrombosis within the vessel (Antman and Braunwald, 2001). Severe ischemia downstream from occluded arteries causes cardiomyocytic apoptosis within minutes. The widespread injury and cell death, often exacerbated by reoxygenation following reperfusion, floods the region with reactive oxygen species and toxic agents (Yellon and Hausenloy, 2007). In response, cells in and around the affected areas upregulate and secrete cytokines and chemokines such as tumor necrosis factor-α (TNF-α), monocyte chemoattractant protein-1 (MCP-1), interleukin (Il)-1β, Il-6 or Il-8, which trigger an immediate and massive infiltration of circulating leukocytes into the ischemic core (Frangogiannis, 2008). Adjacent endothelial cells bolster the recruitment of pro-inflammatory immune cells by upregulating their expression of cell adhesion molecules such as E-selectin, intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1). The initial wave of infiltrating cells is followed by monocytes that home to the damaged area and mature into macrophages. The immune cells gradually clear out cellular debris and matrix degradation products in the injury site, leaving behind sparse tissue with enlarged capillaries.
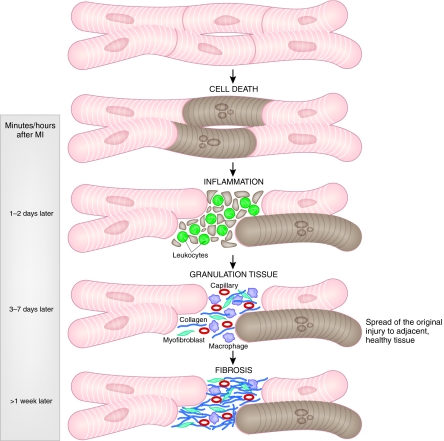
After cellular debris is removed from the injured area, the gap left behind fills with granulation tissue. This process begins with the arrival of myofibroblasts a few days after the initial injury (Frangogiannis, 2008). Granulation tissue is composed mainly of blood vessels, macrophages and myofibroblasts, the latter depositing collagen and other extracellular matrix proteins. A week after the infarction, the granulation tissue starts to develop into a dense scar with collagen deposits intermingled with myofibroblasts (Fig. 1).
Fig. 1.
Schematic representation of cellular events after cardiac ischemic injury. Severe ischemia downstream from an occluded coronary artery results in cardiomyocytic apoptosis within minutes after the supplying blood vessel is closed. Cell death and the release of toxic products trigger a massive inflammatory response. After the cellular debris is cleared, the injury site fills with granulation tissue that is composed mainly of enlarged capillaries, macrophages and myofibroblasts, which deposit collagen and other extracellular matrix proteins to build a dense scar. The disease milieu affects neighboring cardiomyocytes, leading to a gradual spread of the original injury to relatively healthy tissue.
The ischemic area is rich in inflammatory cytokines and protease activity, which harms surrounding healthy cells (Vanhoutte et al., 2006). The extra mechanical burden on the remaining ventricular cells, further compromises the integrity of the cardiac tissue. Thus, the initial localized injury creates a ripple effect that spreads slowly to larger areas of the heart. The loss of functional tissue and subsequent remodeling eventually causes ventricular dysfunction and electrical instability, leading to heart failure and malignant arrhythmias (Pfeffer and Braunwald, 1990; Sun, 2009).
Non-ischemic cardiomyopathies (caused by genetic mutations, viral infections, drug abuse, chemotherapy, etc.) can also destroy cardiomyocytes leading to inflammation, scar formation, ventricular remodeling and heart failure (Schönberger and Seidman, 2001).
Stem cells repair damaged cardiac tissue
Until a few years ago, damage to cardiac tissue and the gradual deterioration towards heart failure were regarded as irreparable. A series of studies in animal models, showing that transplantation of various progenitor cell preparations of bone marrow, skeletal, or embryonic origin improve ventricular function after injury, are challenging this view (Segers and Lee, 2008). Some reports document significant rates of stem cell differentiation to cardiovascular tissue, whereas other studies do not. Thus, there is some controversy about the benefits of stem cell transplantation. It is likely that the positive outcome of stem cell application depends on two factors: (1) the ability of transplanted progenitor cells to differentiate, even with low frequency, to cardiac and vascular cells; and (2) the secretion of survival factors by progenitor cells to stimulate tissue recovery after ischemic injury and minimize the infarct size (Kupatt et al., 2005a; Kupatt et al., 2005b; Gnecchi et al., 2005; Uemura et al., 2006; Gnecchi et al., 2008). Recent studies show that products of transplanted progenitor cells such as β4 thymosin, which promotes wound healing, or the Wnt antagonist secreted frizzled-related protein 2 (sFRP2), have favorable effects on cell survival and preservation of cardiac function after myocardial infarction (Mirotsou et al., 2007; Hinkel et al., 2008; Alfaro et al., 2008).
Cardiac repair using exogenous progenitor cells
As the isolation and characterization of embryonic and adult stem cells increased over the last decade, it became evident that many cell types have cardiogenic potential in vitro, either as part of the natural course of their differentiation program (e.g. embryonic stem cells) (Doetschman et al., 1985) or under specific inductive conditions (e.g. mesenchymal stem cells) (Reinecke et al., 2008). These findings led to numerous experimental studies in animal models of cardiovascular disease to assess the potential of progenitor cell populations to replace lost ventricular tissue and improve cardiac function. The most prominent cell types now considered for cardiac repair are discussed briefly below.
Mesenchymal stem cells
Mesenchymal stem cells (MSCs) are found in the bone marrow stroma and adipose tissue, as well as in other organs (Uccelli et al., 2008). They can be isolated readily and expanded in culture to obtain large numbers that are adequate for cell therapy. MSCs are multipotent and give rise to adipocytes, chondrocytes, osteoblasts and endothelial cells (Pittenger et al., 1999; Jiang et al., 2002). In culture, they can also differentiate into contracting cardiomyocytes, but only with low efficiency and specialized culture conditions (Makino et al., 1999; Tomita et al., 1999; Shiota et al., 2007). An interesting aspect of MSCs is that they appear to have low immunogenicity, which promotes their use in allogeneic recipients (Aggarwal and Pittenger, 2005; Amado et al., 2005; Dai et al., 2005).
Transplantation of MSCs in animal models of myocardial infarction and dilated cardiomyopathy significantly improves left ventricular cardiac function (Tomita et al., 1999; Toma et al., 2002; Kudo et al., 2003; Amado et al., 2005; Dai et al., 2005; Silva et al., 2005; Grauss et al., 2008). Besides functional improvement, mortality also decreases in animals after cell transplantation (Miyahara et al., 2006). However, despite enhanced cardiac function, the differentiation of MSCs to cardiomyocytes is limited (Fazel et al., 2005; Silva et al., 2005; Noiseux et al., 2006; Rose et al., 2008). The beneficial effects of MSCs are postulated to come from their influence on neovascularization of the ischemic tissue and their protection of resident cells (Caplan and Dennis, 2006; Gnecchi et al., 2008). This ‘cell-help-cell’ effect is enhanced further when MSCs are engineered to express the pro-survival kinase Akt1 (Mangi et al., 2003; Gnecchi et al., 2005; Noiseux et al., 2006). Akt1-expressing MSCs secrete sFRP2 that protects cardiomyocytes from hypoxia-induced apoptosis (Mirotsou et al., 2007; Zhang, Z. et al., 2009).
The positive effects of transplanted MSCs, and the ability to genetically modify them, promotes their use as a cellular vehicle to deliver angiogenic proteins [e.g. angiopoietin-1 (Ang1)] (Sun, L. et al., 2007), survival factors [insulin-like growth factor-1 (IGF-1)] (Haider et al., 2008), chemokines [stromal cell-derived factor-1 (SDF-1)] (Zhang et al., 2007) or Wnt antagonists (sFRP2) (Alfaro et al., 2008) to further enhance the recovery of injured myocardium. However, it should be noted that MSC transplantation after myocardial infarction produced calcification and ossification islands inside the ventricular tissue in some mice (Breitbach et al., 2007). In one study, this was also observed in approximately 28% of rats analyzed, raising concerns about the direct use of MSCs in cardiovascular cell therapy (Yoon et al., 2004).
Skeletal myoblasts
Skeletal myoblasts, or satellite cells, are found in the basal membrane of muscle fibers and maintain the homeostasis of skeletal tissue (Shi and Garry, 2006; Buckingham and Montarras, 2008). Myoblasts are easy to isolate from small muscle biopsies as they can proliferate and expand substantially in culture. Obvious similarities between skeletal and cardiac muscle tissue suggest that satellite cells may adopt a cardiomyocytic fate once inside ventricular tissue. Moreover, they are resistant to hypoxiainduced apoptosis, providing another potential advantage for them in repopulating the ischemic myocardium (Menasché, 2007).
Animal studies show that myoblasts that are injected into cardiac tissue after ischemic injury cause global and regional functional improvements (Murry et al., 1996; Taylor et al., 1998; Ghostine et al., 2002). In a chronic heart failure dog model, transplantation of myoblasts also led to a significant recovery in left ventricular hemodynamics (He et al., 2005). In similar fashion, intramyocardial delivery of myoblasts in rabbits with an acute myocardial infarction demonstrated an increase in regional left ventricular wall thickness by magnetic resonance imaging and a decrease in the deleterious effects of post-infarction cardiac remodeling (van den Bos et al., 2005). However, even though myoblasts seem to incorporate into the infarct site and develop contraction-like characteristics, they generally fail to form intercalated disks and appropriate gap junctions with resident cardiomyocytes (Reinecke et al., 2002). The lack of electromechanical coupling with the surrounding host cardiac tissue caused arrhythmias in a number of cases (Leobon et al., 2003).
Although straightforward application of skeletal myoblasts may have limited future use in cardiovascular cell therapy, satellite cells could offer an ample source of cellular material that is similar to cardiac progenitor cells and that might be reprogrammed with cardiac-specific regulatory factors. Skeletal myoblasts may also be engineered to form connections with resident cardiomyocytes by expressing appropriate gap junction proteins before transplantation (Roell et al., 2007).
Bone marrow-derived progenitor cells
Bone marrow (BM)-derived progenitor cells have been used widely in animal models to enhance the repair of injured myocardium. This area was fueled by results which show that the hearts of female patients, who received male BM, contain Y-chromosome-positive cardiomyocytes, indicating that the new cardiac cells originated from BM (Deb et al., 2003; Bayes-Genis et al., 2007). Transplants of female hearts in male recipients were also found to contain Y-chromosome-positive cardiomyocytes, suggesting an extracardial source of new cells (Laflamme et al., 2002; Bayes-Genis et al., 2002; Quaini et al., 2002; Müller et al., 2002). Moreover, certain subpopulations of BM cells, or of circulating BM-derived peripheral blood progenitor cells, can differentiate into cardiomyocytes, endothelial cells or smooth muscle cells under certain conditions in culture (Badorff et al., 2003; Yeh et al., 2003; Belema Bedada et al., 2005; Koyanagi et al., 2005; Koyanagi et al., 2007; Pallante et al., 2007; Flaherty et al., 2008). Consistent with these in vitro results, some transplantation studies suggest extensive differentiation of donor BM-derived cells to both vascular and cardiac cells in the host (Orlic et al., 2001a; Orlic et al., 2001b; Yeh et al., 2003; Kajstura et al., 2005; Rota et al., 2007). By contrast, other studies find that transplanted BM cells only make a significant contribution to vascular structures (Jackson et al., 2001), or show that there is very little, if any, differentiation of BM cells to cardiovascular cell types in vivo (Balsam et al., 2004; Murry et al., 2004). Future work may resolve the controversy surrounding the regenerative potential of BM cells for cardiac repair.
Despite the discrepancies regarding the differentiation of BM cells into cardiovascular tissue in vivo, most experimental studies demonstrate that transplanted BM cells have beneficial effects, including a robust stimulation of angiogenesis (Zhao et al., 2008). The pioneering work of T. Asahara and the late J. Isner first showed that CD34+ cells from the vascular circulation could differentiate into endothelial cells in culture and promote neovascularization in vivo (Asahara et al., 1997). It is possible that the CD34+/CD133+/VEGFR2+ subpopulation of BM cells, also described as endothelial progenitor cells (EPCs), contributes directly or indirectly to neovascularization, thus promoting angiogenesis and reperfusion of ischemic tissue (Kocher et al., 2001; Kawamoto et al., 2003; Young et al., 2007; Lamparter and Hatzopoulos, 2007). The potential of EPCs for cell therapy of vascular disease has been reviewed recently (Gulati and Simari, 2009).
A second possible explanation for the positive action of BM-derived cells on cardiac regeneration is that BM isolates often contain MSCs, which, as discussed already, produce beneficial effects on their own. Also, monocytic cells are part of the BM mononuclear cell population and may promote arteriogenesis and growth of collateral vessels (Kamihata et al., 2001; Kinnaird et al., 2004). Thus, BM mononuclear cells may improve the perfusion index of ischemic areas, which shrinks the infarct territory and improves cardiac function. Although BM cells might have limited regenerative capacity on their own, they could be useful as an accessory transplant in combination cell therapies, together with stem cells of high cardiomyocytic potential, to enhance angiogenesis and promote stem cell survival in the ischemic environment.
Embryonic stem cells
Embryonic stem (ES) cells originate from the inner cell mass of pre-implantation blastocysts. The first ES cells were isolated from mice at around day 3.5 of development (Evans and Kaufman, 1981; Martin, 1981). Since then, ES cell lines have been derived from other mammalian species, including humans (Thomson et al., 1998). Although in mice the inner cell mass consists of only about 15–20 cells, these few cells replicate almost indefinitely without differentiating in culture, providing an almost inexhaustible source of stem cells. Injection of culture-expanded mouse ES cells into blastocysts proved that they are pluripotent; that is, they are able to give rise to all embryonic lineages including germ cells.
Mouse and human ES cells can differentiate into a broad variety of organ-specific cells types in vitro, offering a rich source of cells for regenerative purposes (Doetschman et al., 1985; Keller, 2005; Murry and Keller, 2008), including cardiovascular cell lineages (Kehat et al., 2001; Xu et al., 2002; Sachinidis et al., 2003; Kattman et al., 2006; Moretti et al., 2006; Wu et al., 2006; Yang, L. et al., 2008).
After transplantation, ES cells and ES cell-derived cardiac cells have been shown to integrate into heart tissue and improve cardiac function in ischemic or cryoinjury models (Klug et al., 1996; Kehat et al., 2001; Kehat et al., 2004; Behfar et al., 2002; Min et al., 2002; Roell et al., 2002; Hodgson et al., 2004; Kofidis et al., 2005; Laflamme et al., 2005; Laflamme et al., 2007; Yan et al., 2009). In myocardial infarction mouse models, ES cells that were injected directly into the infarcted myocardium differentiated into cardiomyocytes, vascular smooth muscle cells and endothelial cells. The ES cell transplant caused a significant increase in left ventricular systolic function and a decrease in cardiac tissue remodeling, suggesting that the size of the injury was reduced (Singla et al., 2006).
ES cell therapy might also be beneficial in non-ischemic heritable cardiomyopathies. In Kir6.2-knockout mice, which lack functional KATP channels (recapitulating human dilated cardiomyopathy 10), ES cell transplant results in an improvement in systolic dysfunction and electrical synchronization; a decrease in left ventricular remodeling; and an increase in survival (Yamada et al., 2008).
It appears that the differentiation rate of ES cells into new cardiovascular tissues is the most efficient among the stem cell types tested so far. Still, there are several disadvantages that may complicate their use in clinical investigations. First, ES cells have the propensity to develop teratomas, tumors that contain a wide array of different cell types (Nussbaum et al., 2007). Studies show that the number of ES cells used in treatments may need to be titrated carefully to avoid uncontrolled tumor growth. Interestingly, the tumorogenicity is curtailed in mice that overexpress TNF-α in the heart (Behfar et al., 2007). Second, ES cells differentiate into heterogeneous populations. This means that the number of cardiac cells created under normal differentiation conditions is rather low (usually less than, or around, 1% of the total cells in the culture). Elaborate protocols may be needed to increase the production of cardiac cells so that they can be selected from cells of other lineages. So far, a number of ways have been described to manipulate the differentiation process of ES cells in order to increase cardiac cell output (Yuasa et al., 2005; Qyang et al., 2007; Ueno et al., 2007; Hao et al., 2008; Yan et al., 2009). The next challenge is to direct ES-derived cardiac progenitors to monotypic cultures of specialized cell types, such as ventricular and atrial myocytes, or to pacemaker and conduction system cells (Behfar et al., 2008; Chien et al., 2008). Third, transplantation of allograft ES cells can create an immunologic response and cell rejection (Swijnenburg et al., 2005). These technical difficulties are in addition to the ethical issues voiced by some people, which further preclude the straightforward application of ES cells in clinical studies. New developments regarding the possibility to generate ES-like cells, called inducible pluripotent stem (iPS) cells, by reprogramming adult somatic cells (Takahashi et al., 2007; Wernig et al., 2007; Okita et al., 2007) or by adapting adult spermatogonia (Guan et al., 2006), may bypass some of these technical and ethical issues.
Inducible pluripotent stem cells
iPS cells are generated by expressing specific pluripotency transcription factors in somatic cells, such as fibroblasts or epithelial cells, using viral vectors (Takahashi et al., 2007; Wernig et al., 2007; Okita et al., 2007). Pluripotency factors include proteins that are crucial for the maintenance of the stem cell phenotype, for example Oct3/4, Sox2, Klf4, Nanog or c-Myc. The pluripotency factors gradually suppress the expression of genes that are associated with somatic cell function, shutting down differentiation programs and activating the expression of endogenous stem cell pluripotency factors (Jaenisch and Young, 2008). Within several weeks, the altered gene expression reprograms the somatic cells to acquire similar morphological, molecular and functional characteristics to ES cells. iPS cells can generate mice after blastocyst injection and can also differentiate into various lineages in culture, including cardiovascular cells (Schenke-Layland et al., 2008; Narazaki et al., 2008; Mauritz et al., 2008; Zhang, J. et al., 2009).
The same technical challenges of ES cell therapy (low cardiogenic output, heterogeneous differentiation, limited differentiation to specialized cell types, and tumorogenicity) must be resolved before clinical applications of iPS cells are realized fully. Already, studies show that reprogramming can be achieved with only two pluripotency factors, eliminating the need for oncogenic c-Myc, or without viral integration into the genome, which can lead to pathological complications (Huangfu et al., 2008; Stadtfeld et al., 2008).
Clinical studies
The experimental success of using progenitor cells to improve heart recovery after ischemic damage in animals led clinicians to test the safety and efficacy of cell therapy in patients with myocardial damage following myocardial infarction (Dimmeler and Zeiher, 2008; Segers and Lee, 2008; Burt et al., 2008). Randomized clinical trials with a variety of cell preparations, including BM-derived cells (Wollert et al., 2004; Ruan et al., 2005; Assmus et al., 2006; Lunde et al., 2006; Schächinger et al., 2006a; Janssens et al., 2006; Ge et al., 2006; Hendrikx et al., 2006), circulating progenitor cells (Erbs et al., 2005; Assmus et al., 2006; Kang et al., 2006), skeletal myoblasts (Menasché et al., 2008) and MSCs (Chen et al., 2004; Chen et al., 2006), with intracoronary or intramuscular delivery, suggest that these approaches are safe and may improve ventricular function (Table 1). Although some trials failed to demonstrate a significant increase in cardiac function after cell transplantation when compared with controls, several other randomized trials showed measurable improvements that were comparable to established therapeutic regimes (Reffelmann et al., 2009). Non-randomized, smaller-scale trials also produced variable results, ranging from no significant changes in left ventricular ejection fraction to a significant improvement, of up to a 14% increase, during the follow-up period (Strauer et al., 2002; Perin et al., 2004; Fernandez-Aviles et al., 2004; Ince et al., 2004; Strauer et al., 2005; Katritsis et al., 2005; Bartunek et al., 2005; Mocini et al., 2006; Gavira et al., 2006; Ahmadi et al., 2007; Choi et al., 2007; Klein et al., 2007; Li et al., 2007; Stamm et al., 2007; Tatsumi et al., 2007).
Table 1.
Stem cell and progenitor cell therapies: randomized clinical trials
| Study | Year | Clinical setting | n | Cell type | Method of cell transplantation | Cell number transplanted | Mean follow-up (months) | % Change in LVEF compared with control |
|---|---|---|---|---|---|---|---|---|
| Menasché et al. (MAGIC) | 2008 | ICMP | 97 | SMB | Intramyocardial† | 4×108 or 8×108 | 6 | −1.0% or +0.8%; NS |
| Meluzin et al. | 2008 | AMI | 60 | BMMNC | Intracoronary | 1×108 | 12 | +7.0%; P=0.03 |
| Meyer et al. (BOOST) | 2006 | AMI | 60 | BMMNC | Intracoronary | 2.5×109 | 18 | +2.8%; NS |
| Assmus et al. (TOPCARE-CHD) | 2006 | ICMP | 51 | BMMNC | Intracoronary | 2×108 | 3 | +4.1%; P<0.001 |
| Schächinger et al. (REPAIR-AMI) | 2006 | AMI | 204 | BMMNC | Intracoronary | 2.4×108 | 12 | * |
| Schächinger et al. (REPAIR-AMI) | 2006 | AMI | 204 | BMMNC | Intracoronary | 2.4×108 | 4 | +2.5%; P=0.01 |
| Ge et al. (TCT-STAMI) | 2006 | AMI | 20 | BMMNC | Intracoronary | 4×107 | 6 | +6.7%; NS |
| Hendrikx et al. | 2006 | ICMP | 20 | BMMNC | Intramyocardial† | 6×107 | 4 | +2.5%; NS |
| Janssens et al. | 2006 | AMI | 67 | BMMNC | Intracoronary | 1.7×108 | 4 | +1.2%; NS |
| Lunde et al. (ASTAMI) | 2006 | AMI | 100 | BMMNC | Intracoronary | 8.7×107 | 6 | −3.0%; P=0.05 |
| Ruan et al. | 2005 | AMI | 20 | BMC | Intracoronary | Not available | 6 | +9.2%; P<0.05 |
| Chen et al. | 2006 | ICMP | 45 | MSC | Intracoronary | 5×106 | 12 | −3.0%; NS |
| Chen et al. | 2004 | AMI | 69 | MSC | Intracoronary | 6×1010 | 6 | +12.0%; P=0.01 |
| Assmus et al. (TOPCARE-CHD) | 2006 | ICMP | 47 | CPC | Intracoronary | 2.2×107 | 3 | +0.8%; NS |
| Kang et al. | 2006 | AMI/ICMP | 82 | CPC | Intracoronary | 1.4×109 | 6 | −0.2%; NS |
| Erbs et al. | 2005 | ICMP | 26 | CPC | Intracoronary | 7×107 | 3 | +7.2%; NS |
| Losordo et al. | 2007 | ICMP | 24 | CD34+ | Intramyocardial‡ | 5×104, 1×105 or 5×105 | 6 | ** |
AMI, acute myocardial infarction; BMC, bone marrow cells; BMMNC, bone marrow mononuclear cells; CPC, circulating progenitor cells; LVEF, left ventricular ejection fraction; ICMP, ischemic cardiomyopathy; MSC, mesenchymal stem cells; NS, non-significant; SMB, skeletal myoblasts.
Intramyocardial through surgical transepicardial approach.
Intramyocardial through percutaneous transendocardial approach.
BMMNC therapy resulted in significant decrease in death, myocardial infarction and revascularization at 12 months, whereas LVEF was not included in the pre-specified cumulative endpoint.
In CCS class III or IV angina patients, CD34+ cell therapy resulted in a trend towards a decrease in the frequency of angina and improvement in CCS class, exercise times and nitroglycerin use.
To date, the largest clinical trial to assess the benefit of skeletal myoblast therapy is the MAGIC (Myoblast Autologous Grafting in Ischemic Cardiomyopathy) trial, which randomized patients to receive either stem cell injection or culture medium. Although early fears of severe arrhythmias did not materialize, the results have been disappointing, showing no significant benefit with skeletal myoblast cell implantation (Menasché et al., 2008). By contrast, a meta-analysis of 18 randomized and non-randomized trials involving a total of 999 acute myocardial infarction or chronic ischemic cardiomyopathy patients found that transplantation of adult BM-derived stem cells improved left ventricular ejection fraction by 5.40% (P<0.001); decreased infarct scar size by 5.49% (P=0.003); and lowered left ventricular end-systolic volume by 4.80 mls (P=0.006) (Abdel-Latif et al., 2007). It is also encouraging that a multicenter, randomized clinical trial of 204 patients with acute myocardial infarction showed that intracoronary delivery of BM cells (at 3 to 7 days post-reperfusion therapy) decreased the incidence of myocardial infarction and death, or revascularization, at a 12-month follow-up when compared with placebo (P=0.009) (Schächinger et al., 2006b).
Cell therapy also shows promise to relieve chronic angina symptoms. In patients with Canadian Cardiovascular Society (CCS) class III or IV angina, intramyocardial transplantation of autologous CD34+ stem cells decreased the frequency of angina, resulting in an improvement in CCS class, exercise times and nitroglycerin use when compared with patients who did not receive CD34+ cells (Losordo et al., 2007).
Perhaps the inconsistent results in clinical trials reflect both the variety of stem cell types used and the route of administration, as well as the quantity and quality of the injected cells. For example, mononuclear BM cell transplantation improves left ventricular ejection fraction in post-myocardial infarction patients when a higher number of cells are injected (i.e. 108 cells compared with 107 cells) (Meluzín et al., 2006). In addition, the ability of BM-derived cells to migrate, or induce neovascularization, might be impaired in heart disease patients, which would affect the efficacy of autologous cell therapy (Heeschen et al., 2004; Walter et al., 2005). The timing of left ventricular assessment after cell transplantation in patients may further explain some of the discrepancies in ventricular function since a number of benefits appear to be transient (Meyer et al., 2006; Meluzín et al., 2008), consistent with findings in animal studies of ES cell transplantation (van Laake et al., 2008). Finally, the portion of stem cells retained at the area of interest may be low, thereby diminishing the beneficial effects, as discussed in the next section.
Methods of stem cell delivery
In the clinical setting, stem cells have been delivered by intracoronary injection; intravenous infusion; by direct injection into cardiac muscle through the endocardium by a percutaneous approach; or through the epicardium during open-heart surgery.
The infusion of stem cells into the coronary arterial circulation is relatively straightforward. In this situation, the cells are surrounded by nutrients and oxygen, which creates a favorable environment. However, the number of stem cells retained in the ventricular area of interest might be low. For example, in patients who sustained an ST-segment myocardial infarction and received a coronary stent, it was estimated that only 1.2–3.6% of BM cells that were delivered through an intracoronary route remained within the myocardium (Hofmann et al., 2005).
The homing of progenitor cells requires interaction with the vascular wall and transendothelial migration towards injured or ischemic areas, which may involve similar mechanisms to those involved in the recruitment of immune system cells to sites of inflammation (Vajkoczy et al., 2003; Chavakis et al., 2005; Chavakis et al., 2008). Thus, activation of adhesion molecules in the host vasculature and donor progenitor cells before cell delivery may stimulate homing efficiency. For example, adenosine increases the adhesion of EPCs to the vascular wall in experimental models (Ryzhov et al., 2008). Adenosine can be delivered directly into the coronary circulation in humans without significant adverse events and it has a short half-life in the bloodstream; thus, it may increase progenitor cell retention locally in a clinically feasible way (Leesar et al., 1997; Shen and DiCorleto, 2008).
Direct injection of stem cells into injured myocardium ensures that cells are placed in the area of interest. Nevertheless, ischemic conditions along with inflammation and scar tissue make a less than ideal environment for cell survival. Moreover, mechanical loss further diminishes the engraftment of injected cells. Estimates suggest that, during open-heart surgery, the leakage of injected microspheres is 33% and 89% in non-beating and beating porcine hearts, respectively (Teng et al., 2006). Low retention efficiency (approximately 3%) was also recorded for EPCs that were injected directly into the myocardium of rats (Aicher et al., 2003), or for BM cells in infarcted hearts of sheep (Bel et al., 2003); moreover, only a small percentage of myoblasts (approximately 7%) that were injected directly into infarcted mouse hearts survived for longer than 72 hours (Suzuki et al., 2004). Thus, intraventricular delivery may not improve cell homing compared with intracoronary injection in all cases.
The modest, short-term cardioprotective effects of transplanted cells underscore the need to maximize delivery and differentiation of stem cells in order to produce functional cardiovascular tissue with long-term survival and engraftment within diseased tissue. Current roadblocks to the use of exogenous cells are renewing research interest in the inherent regenerative and reparative mechanisms of cardiac tissue in order to develop novel tools and strategies to enhance the efficacy of current cell therapy protocols.
Endogenous stem cells in cardiac regeneration
Classically, the heart is thought of as a terminally differentiated, postmitotic organ without intrinsic mechanisms to replace lost cardiomyocytes. However, a number of recent studies show that the adult heart contains cell populations with stem cell characteristics and that cardiac homeostasis is maintained by replenishing damaged cells (Ellison et al., 2007; Kajstura et al., 2008). Genetic fate-mapping studies show that a considerable number of new cardiomyocytes form in the adult mouse heart after injury (Hsieh et al., 2007), and incorporation of 14C (which did not exist naturally before nuclear tests) into the DNA of human cardiomyocytes estimates that about 50% of adult cardiomyocytes are exchanged during a normal life span, suggesting a renewal mechanism (Bergmann et al., 2009). The properties of cardiac progenitor cells in the adult heart are discussed briefly below.
Side population cells
A typical property of some stem cell populations is the exclusion of the vital dyes Hoechst 33342 and rhodamine 123. The cells that do not take up the dyes are called the side population (SP) (Goodell et al., 1996). Dye exclusion in SP cells is the result of high efflux rates mediated by ATP-binding cassette transporters such as ABCG2 and MDR1 (Challen and Little, 2006). SP cells are multipotent and have been identified in various organs including the BM, skeletal muscle and adipose tissue (Challen and Little, 2006).
The dissociation of heart tissue and dye stains show that the adult heart also has a resident pool of SP cells (Hierlihy et al., 2002; Martin et al., 2004). Isolated SP cells from heart tissue can be induced to express cardiac-specific genes in vitro when co-cultured with cardiomyocytes or when treated with agents such as oxytocin or the histone deacetylase inhibitor trichostatin A (Pfister et al., 2005; Oyama et al., 2007). The cardiogenic potential is higher in the subgroup of SP cells that express the stem cell antigen-1 (Sca-1+), but are negative for the endothelial marker CD31 (Pfister et al., 2005).
The number of SP cells in the mouse heart changes after myocardial infarction through proliferation of resident SP cells, as well as homing of BM-derived SP cells (Mouquet et al., 2005), but this might depend on the injury model used (Oyama et al., 2007). There is limited information about the regenerative potential of SP cells in vivo. One study showed homing of SP cells to the injured heart and differentiation of these cells into cardiomyocytes, endothelial cells and smooth muscle cells (Oyama et al., 2007). In another report, isolated SP cells that were allowed to form cardiospheres in culture displayed features of neural crest (NC) progenitor cells in vitro and in vivo, with the potential to differentiate into glia, neurons, smooth muscle and cardiomyocytes, suggesting that at least a portion of SP cells are of neural crest origin (Tomita et al., 2005).
c-Kit+ progenitor cells
A second resident progenitor population is the c-Kit+ cells, which are located in small clusters within the ventricles and atria of the adult heart (Beltrami et al., 2003). Isolated c-Kit+ cells do not differentiate fully into cardiovascular cell types in culture, but show impressive regenerative potential after transplantation in the injured rat heart, where they give rise to cardiomyocytes, endothelial cells and smooth muscle cells (Beltrami et al., 2003; Dawn et al., 2005; Rota et al., 2008). Cardiac c-Kit+ stem cells improve left ventricular ejection fraction after myocardial infarction by 11% compared with control rats that did not receive cell transplantation at a 20-day follow-up (Beltrami et al., 2003). An equivalent c-Kit+ cell population has been isolated from small samples of human myocardium (Bearzi et al., 2007).
Sca-1+ progenitor cells
A third reported stem cell type in the heart expresses Sca-1, but is c-Kit− (Oh et al., 2003). Sca-1+ cells express some of the early regulatory factors of cardiogenesis, such as GATA4, and they can be induced to express Nkx2.5 and sarcomeric proteins after 5-azacytidine (5-Aza-C) treatment. When given intravenously in mice, they home to infarcted myocardium and yield cardiomyocytes around the injury area, although a degree (about 50%) of cell fusion with resident myocytes was noted (Oh et al., 2003). A rare Sca-1+/c-Kit+ cardiac cell type that differentiates into cardiomyocytic cells upon oxytocin treatment was also identified in mice (Matsuura et al., 2004). Finally, a Sca-1+/CD31− cell population was isolated and induced to differentiate to both cardiomyocytes and endothelial cells using combinations of fibroblast growth factor (FGF) proteins, the Wnt antagonist Dkk-1, and 5-Aza-C treatment (Wang et al., 2006). Transplantation of Sca-1+/CD31− cells after myocardial infarction in mice improved cardiac function and enhanced neovascularization. However, the benefits seem to be the result of favorable paracrine effects of the transplanted cells on new blood vessel formation and resident cardiomyocytes (Wang et al., 2006).
Cardiosphere-derived progenitor cells
An elaborate technique for isolation of cardiac progenitors from mouse hearts and human biopsies was described following gentle enzymatic digestion to release round cells that form so-called cardiospheres (CS) in suspension (Messina et al., 2004; Smith et al., 2007). CS-derived cells express endothelial and stem cell markers; show contractile activity in culture; and can differentiate into cardiomyocytes, endothelial cells and smooth muscle cells. Transplantation of these cells improved ventricular function in mice and swine owing to the formation of vascular and cardiac cells, and positive paracrine effects (Smith et al., 2007; Takehara et al., 2008). However, other attempts to obtain cardiac progenitor cells following a similar approach, but without cardiosphere formation, failed to recapitulate the differentiation and regenerative potential of these cells (Shenje et al., 2008).
Adult cardiac homeostasis and repair
The isolation of various cardiac tissue stem cell types with novel characteristics suggests that they represent either distinct cell populations or different developmental stages of a single cell lineage. Much of the origin and biological properties of cardiac stem cells remain poorly defined. It is unclear whether the cardiogenic stem cells home from the BM, reside in specialized niches, are remnants of embryonic cell populations, or are artifacts of the isolation procedures (Slack, 2008). These possibilities are not mutually exclusive.
Epithelial- and endothelial-to-mesenchymal transitions generate cardiovascular progenitor cells during embryogenesis
It is reasonable to expect that the mechanisms that give rise to cardiac progenitor cells in the adult heart share similar pathways with the specification and differentiation of embryonic cardioblasts. During development, a key process that generates progenitor cells that differentiate into various tissues is the epithelial-to-mesenchymal transition (EMT). EMT produces mesodermal cells from primitive ectoderm during gastrulation and neural crest stem cells from neuroepithelium (Nakaya and Sheng, 2008; Sauka-Spengler and Bronner-Fraser, 2008). Although cardiac development is a complex morphogenetic operation (van den Berg and Moorman, 2009; Perez-Pomares et al., 2009) involving the well-orchestrated action of scores of genes (Brand, 2003; Olson, 2006), it can also be divided into a series of EMT events, each generating a distinct set of cardiovascular progenitor cells that differentiate into the cellular components of the mature heart.
The first EMT that gives rise to cardiovascular progenitor cells takes place during gastrulation, when epithelial cells of the epiblast begin to delaminate, adopt mesenchymal characteristics, and migrate to form the mesoderm layer that occupies the space between the two outer epithelia of endoderm and ectoderm (Nakaya and Sheng, 2008). The timing and location of epithelial cell delamination within the primitive streak determines the destination of the migrating mesodermal/mesenchymal cells in the developing embryo and this specifies their subsequent fate (Abu-Issa and Kirby, 2007). Mesodermal cells in the lateral plate adjacent to the foregut differentiate into progenitor cells that express early cardiac regulatory genes, such as Nkx2–5, Mef2c and Gata4 (Brand, 2003; Moorman et al., 2007). Early cardioblasts are divided into two types representing the primary and secondary cardiac fields (Buckingham et al., 2005).
The cardiac stem cells of the primary field form the original heart tube that surrounds the endocardial vascular layer, contributing mainly to the future left ventricle; the secondary field cells then migrate and wrap around the primitive heart and contribute to the formation of the atria, the right ventricle, and part, or most, of the left ventricle (Cai et al., 2003; Yang et al., 2006; Sun, Y. et al., 2007; Prall et al., 2007; Ma et al., 2008). The role of the primary heart field progenitors may be limited to the formation of the original cardiac tube during embryogenesis, whereas the secondary field cells, marked by Isl1+ expression, are maintained throughout development, contribute to the neonatal heart growth, and remain present, albeit in small numbers, in the adult heart (Laugwitz et al., 2008). Isolated Isl1+ cells can give rise to cardiomyocytes, smooth muscle and endothelial cells (Laugwitz et al., 2005; Moretti et al., 2006).
The second mesenchymal transformation that shapes the heart takes place when a subpopulation of endocardial cells (the endothelial cells of the original inner cardiac tube) in the atrioventricular canal area undergo endothelial-to-mesenchymal transition (EndMT), migrate into the adjacent cardiac jelly, and build the endocardial cushions that develop into the heart valves (Person et al., 2005; Norris et al., 2008). There is evidence that EndMT continues in the adult valves, supplying cells to maintain and repair the valvular leaflets (Yang, J. et al., 2008).
The third EMT occurs in the epithelium formed by the epicardial cells at the outer surface of the heart. Pre-epicardial tissue, which appears as a cauliflower structure from splachnopleuric mesoderm during development, attaches to the exterior surface of the heart and spreads out over the entire organ in a single epithelial cell layer called the epicardium (Winter and Gittenberger-de Groot, 2007). Shortly thereafter, the epicardial epithelium undergoes EMT, generating a mesenchymal stem cell population named the epicardial-derived progenitor cells or EPDCs. EPDCs invade the cardiac tissue and differentiate into interstitial fibroblasts, perivascular fibroblasts and smooth muscle cells of the developing coronary blood vessels (Reese et al., 2002; Wessels and Pérez-Pomares, 2004). Cell-lineage-tracing experiments using Cre-recombinase technology show a considerable contribution of epicardium to ventricular myocytes, suggesting a more substantial role of EPDCs in heart tissue formation than was thought previously (Cai et al., 2008; Zhou et al., 2008). Transplantation of embryonic EPDCs improved cardiac function after myocardial infarction in mice, but the cells did not differentiate into cardiovascular cells (Winter et al., 2007).
The fourth EMT takes place in the neural tube and generates the cardiac NC progenitor cells, between the cranial and trunk neural crest, that migrate to the heart and take part in the remodeling of the aortic arch arteries and the septation of the common outflow tract into the aortic and pulmonary arteries (Hutson and Kirby, 2007; Snider et al., 2007). Lineage-tracing experiments, using transgenic mice with gene markers that are under the control of NC-specific promoter elements or Cre recombinase technology, suggest that NC cells also contribute to the conduction system and epicardium, but this remains controversial (Stottmann et al., 2004; Stoller and Epstein, 2005; Poelmann et al., 2004; Nakamura et al., 2006). Recent reports show that nestin+ stem cells of neural crest origin reside in the adult heart and take part in de novo blood vessel formation and reparative fibrosis after ischemic injury (El-Helou et al., 2008).
EMT and EndMT contribute to cardiac repair and regeneration after injury
Although the importance of EMT in tissue development during embryogenesis is well documented, the contribution of EMT in regenerative processes in the adult organism is not. EMT has been implicated in the emergence of tumor cells that are highly proliferative (or carcinogenic) and motile (or metastatic), suggesting that it plays a key role in the generation and spreading of tumor stem cells (Hollier et al., 2009). A recent study indicates that EMT also yields adult cells with stem cell characteristics (Mani et al., 2008). Therefore, one could predict that, in line with their role during heart development, EMT and EndMT contribute to the pool of cardiovascular progenitor cells to maintain cardiac homeostasis.
Besides the connection of mesenchymal transition to the emergence of stem cells, both EMT and EndMT are recognized as important mechanisms in the generation of the myofibroblasts that take part in fibrosis (Kalluri and Neilson, 2003; Wynn, 2008). Therefore, it is possible that EMT and EndMT contribute to both cardiac regeneration and fibrosis after injury. In the last few years a number of studies have provided supportive evidence for this notion.
For example, EndMT in the adult mouse heart gives rise to myofibroblasts that migrate and produce scar tissue in mouse models of pressure overload and chronic allograft rejection, recapitulating pathways that take place during formation of the atrioventricular cushions in the embryonic heart (Zeisberg et al., 2007). Moreover, isolated adult epicardial cells can undergo EMT in culture, differentiating to smooth muscle and endothelial cells (van Tuyn et al., 2007; Smart et al., 2007). Epicardial cells that express stem cell markers are induced after injury, migrate to the infarct, and contribute to both cardiac and vascular cell types (Limana et al., 2007). These experiments suggest a function of epicardial cells that echoes their role in the regeneration of the adult zebrafish heart after partial resection (Lepilina et al., 2006). Therefore, both endothelial and epicardial cells become activated after injury and give rise to cardiac, vascular and myofibroblast/smooth muscle cells; this seems similar to their capacity during development.
Wnt signaling, stem cells and fibrosis
The experimental evidence outlined above links EMT to the production of stem cells and fibrosis. Although these two processes are controlled by complex regulatory networks (Neilson, 2006; Lowry and Richter, 2007), a common molecular thread that has been studied extensively is the canonical Wnt signaling pathway (Thiery and Sleeman, 2006; Nusse, 2008). The canonical Wnt pathway has been implicated in the generation, maintenance and growth of all stem cell types that have been investigated so far, including neuronal stem cells, hematopoietic stem cells, gut epithelium stem cells, muscle satellite cells, hepatic stem cells, lung stem cells and others (Kalani et al., 2008; Luis et al., 2009; Haegebarth and Clevers, 2009; Otto et al., 2008; Yang, W. et al., 2008; Zhang et al., 2008). The canonical Wnt pathway is also crucial for the formation of embryonic cardiac stem cells in vitro and in vivo (Nakamura et al., 2003; Kwon et al., 2007; Lin et al., 2007; Qyang et al., 2007; Cohen et al., 2008; Laugwitz et al., 2008). Conversely, blockade of canonical, and activation of non-canonical, Wnt signaling is crucial for the differentiation of embryonic and adult progenitor cells into cardiomyocytes (Koyanagi et al., 2005; Flaherty et al., 2008; Cohen et al., 2008; David et al., 2008).
The canonical Wnt pathway inactivates glycogen synthase kinase-3β (GSK-3β) leading to dephosphorylation and stabilization of cytoplasmic β-catenin, which then translocates to the nucleus where it activates a host of target gene loci. Among them are the transcriptional repressors Slug and Snail that shut down the expression of the cell adhesion protein E-cadherin to loosen the junctions between epithelial cells (Thiery and Sleeman, 2006; Nusse, 2008). Liberated epithelial cells quickly rearrange their cytoskeletal structure and adopt a highly proliferative phenotype with a mesenchymal morphology; this is regulated by β-catenin, which induces genes such as those encoding cyclin D and c-Myc (Clevers, 2006). Thus, Wnt signaling is a central regulatory knot that controls two of the landmark events in the biology of stem cells, EMT and cell cycle regulation. Interestingly, Wnt signaling also regulates fibrosis, controlling the generation of mesenchymal cells by EMT as well as their proliferation and motility (Cheon et al., 2002; Bowley et al., 2007; Alfaro et al., 2008). Collectively, these data suggest that proper modulation of Wnt signaling is key in balancing cardiac fibrosis and regeneration after injury.
Reparative and regenerative cells: two sides of the same coin?
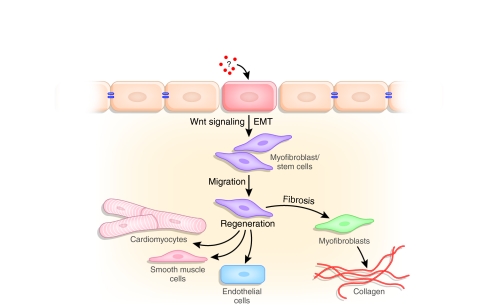
The parallels between the cellular and molecular biology of reparative (myofibroblasts) and regenerative (stem) cells raise the possibility that the two cell phenotypes are connected intrinsically. Moreover, similar to stem cells, myofibroblasts have been shown to derive from multiple sources, such as the BM and resident MSCs, as well as by EMT and EndMT (Hinz et al., 2007). It is possible that mesenchymal cells with stem cell properties follow a default pathway after injury that steers them towards producing scar tissue in order to seal off the necrotic areas and save the surrounding healthy penumbra. These same cells might differentiate to tissue-specific cell types when the conditions favor replacement of malfunctioning or dying cells during organ homeostasis (Fig. 2). This model is consistent with recent findings in skeletal muscle where stem cells switch from a regenerative to a pro-fibrotic phenotype with aging, owing to increased Wnt signaling (Brack et al., 2007).
Fig. 2.
A model of cardiac homeostasis and repair. Normal wear and tear, as well as acute ischemic injury, induces unknown factors (?) that stimulate Wnt signaling-mediated epithelial-to-mesenchymal transition (EMT), generating mesenchymal cells with dual stem cell and myofibroblast characteristics. Favorable conditions allow stem cells to differentiate to various cardiovascular cell types, whereas the disease environment favors a switch to a pro-fibrotic phenotype leading to scar formation.
Too many stem cells, too little regeneration?
Our current knowledge suggests that a multicellular army of progenitor cells, of BM, endothelial, epicardial and neural crest origin, respond to cardiac tissue injury and take part in the repair process. Why is there a need for so many different cell types? Does a particularly crucial situation, resulting from a catastrophic event, mobilize all of the available forces at the disposal of the organ? Or, do the different cell types perform distinct functions, such as replenishing specific cell types, including cardiomyocytes, endothelial cells, smooth muscle cells, conduction system cells and neurons, that have been destroyed during ischemic injury?
If progenitor cells are mobilized after injury, why does the function of myofibroblasts and fibrosis predominate? This is particularly disappointing, since the various stem cells that have been analyzed are able to produce cardiomyocytes, smooth muscle cells and endothelial cells in vitro. It is plausible that stem cells perform their respective roles under normal conditions to maintain cardiac homeostasis, but assume a reparative or pro-fibrotic phenotype when confronted with inflammatory proteins, toxic products of apoptotic cells, ischemia and a disintegrating extracellular matrix. Consistent with this, MSCs adopt different fates depending on extracellular matrix tension and other environmental factors (Engler et al., 2006). It is also conceivable that only myofibroblasts survive in the hostile disease environment.
Over the last few years, we have learned a great deal about the individual pieces in the reparative and regenerative puzzle of the heart, but it is unclear how the different components will fit together. Although the findings point to a complex healing process that relies on many different types of repair/stem cells, they reflect the natural program of cardiopoiesis, where a variety of progenitor cells contribute to new cardiac tissue at distinct stages of embryonic development. Our current challenge is to isolate and study the different stem cell-like populations in the heart and identify their individual roles during cardiac repair and regeneration. It is also important to understand how the mission of progenitor cells is adjusted during a severe injury such as a myocardial infarction. If progenitor cells in the heart are confronted with a choice between tissue regeneration and scar formation, it is crucial to recognize the intrinsic molecular mechanisms and environmental factors that determine the outcome of this decision. This information will help devise novel strategies to fortify the endogenous regenerative capacities of the adult heart and to optimize the cardiovascular differentiation of transplanted stem cells.
Acknowledgments
Work in the authors’ laboratory was supported by an American Heart Association Fellowship to K.D.B. and by NIH grants to A.K.H. Deposited in PMC for release after 12 months.
Footnotes
COMPETING INTERESTS
The authors declare no competing financial interests.
This Perspective is part of an ongoing series on stem cells. Please see the related articles: ‘Cell therapy for the diseased liver:from stem cell biology to novel models for hepatotropic human pathogens’ in DMM Volume 1, Issue 2/3, pages 113-130; ‘Progenitor cell therapies for traumatic brain injury: barriers and opportunities in translation’ in DMM Volume 2, Issue 1/2, pages 23-38; and ‘Defining the potential for cell therapy for vascular disease using animal models’ in DMM Volume 2, Issue3/4, pages 130-137.
REFERENCES
- Abdel-Latif A, Bolli R, Tleyjeh IM, Montori VM, Perin EC, Hornung CA, Zuba-Surma EK, Al-Mallah M, Dawn B. (2007). Adult bone marrow-derived cells for cardiac repair: a systematic review and meta-analysis. Arch Intern Med. 167, 989–997 [DOI] [PubMed] [Google Scholar]
- Abu-Issa R, Kirby ML. (2007). Heart field: from mesoderm to heart tube. Annu Rev Cell Dev Biol. 23, 45–68 [DOI] [PubMed] [Google Scholar]
- Aggarwal S, Pittenger MF. (2005). Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 105, 1815–1822 [DOI] [PubMed] [Google Scholar]
- Ahmadi H, Baharvand H, Ashtiani SK, Soleimani M, Sadeghian H, Ardekani JM, Mehrjerdi NZ, Kouhkan A, Namiri M, Madani-Civi M, et al. (2007). Safety analysis and improved cardiac function following local autologous transplantation of CD133(+) enriched bone marrow cells after myocardial infarction. Curr Neurovasc Res. 4, 153–160 [DOI] [PubMed] [Google Scholar]
- Aicher A, Brenner W, Zuhayra M, Badorff C, Massoudi S, Assmus B, Eckey T, Henze E, Zeiher AM, Dimmeler S. (2003). Assessment of the tissue distribution of transplanted human endothelial progenitor cells by radioactive labeling. Circulation 107, 2134–2139 [DOI] [PubMed] [Google Scholar]
- Alfaro MP, Pagni M, Vincent A, Atkinson J, Hill MF, Cates J, Davidson JM, Rottman J, Lee E, Young PP. (2008). The Wnt modulator sFRP2 enhances mesenchymal stem cell engraftment, granulation tissue formation and myocardial repair. Proc Natl Acad Sci USA 105, 18366–18371 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amado LC, Saliaris AP, Schuleri KH, St John M, Xie JS, Cattaneo S, Durand DJ, Fitton T, Kuang JQ, Stewart G, et al. (2005). Cardiac repair with intramyocardial injection of allogeneic mesenchymal stem cells after myocardial infarction. Proc Natl Acad Sci USA 102, 11474–11479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antman EM, Braunwald E. (2001). Acute myocardial infarction. In Harrison’s Principles of internal medicine (ed. Fauci AS, Braunwald E, Isselbacher KJ, Wilson JD, Martin JB, Kasper DL, Hauser SL, Longo DL, Harrison TR.), pp. 1386–1399 New York, USA: McGraw-Hill [Google Scholar]
- Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T, Witzenbichler B, Schatteman G, Isner JM. (1997). Isolation of putative progenitor endothelial cells for angiogenesis. Science 275, 964–967 [DOI] [PubMed] [Google Scholar]
- Assmus B, Honold J, Schächinger V, Britten MB, Fischer-Rasokat U, Lehmann R, Teupe C, Pistorius K, Martin H, Abolmaali ND, et al. (2006). Transcoronary transplantation of progenitor cells after myocardial infarction. N Engl J Med. 355, 1222–1232 [DOI] [PubMed] [Google Scholar]
- Badorff C, Brandes RP, Popp R, Rupp S, Urbich C, Aicher A, Fleming I, Busse R, Zeiher AM, Dimmeler S. (2003). Transdifferentiation of blood-derived human adult endothelial progenitor cells into functionally active cardiomyocytes. Circulation 107, 1024–1032 [DOI] [PubMed] [Google Scholar]
- Balsam LB, Wagers AJ, Christensen JL, Kofidis T, Weissman IL, Robbins RC. (2004). Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature 428, 668–673 [DOI] [PubMed] [Google Scholar]
- Bartunek J, Vanderheyden M, Vandekerckhove B, Mansour S, De Bruyne B, De Bondt P, Van Haute I, Lootens N, Heyndrickx G, Wijns W. (2005. Intracoronary injection of CD133-positive enriched bone marrow progenitor cells promotes cardiac recovery after recent myocardial infarction: feasibility and safety. Circulation 112 Suppl. 9, I178–I183 [DOI] [PubMed] [Google Scholar]
- Bayes-Genis A, Salido M, Solé Ristol F, Puig M, Brossa V, Campreciós M, Corominas JM, Marinoso ML, Baró T, Vela MC, et al. (2002). Host cell-derived cardiomyocytes in sex-mismatch cardiac allografts. Cardiovasc Res. 56, 404–410 [DOI] [PubMed] [Google Scholar]
- Bayes-Genis A, Roura S, Prat-Vidal C, Farré J, Soler-Botija C, de Luna AB, Cinca J. (2007. Chimerism and microchimerism of the human heart: evidence for cardiac regeneration. Nat. Clin. Pract. Cardiovasc. Med. 4 Suppl. 1, S40–S45 [DOI] [PubMed] [Google Scholar]
- Bearzi C, Rota M, Hosoda T, Tillmanns J, Nascimbene A, De Angelis A, Yasuzawa-Amano S, Trofimova I, Siggins RW, Cascapera S, et al. (2007). Human cardiac stem cells. Proc Natl Acad Sci USA 104, 14068–14073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behfar A, Zingman LV, Hodgson DM, Rauzier JM, Kane GC, Terzic A, Pucéat M. (2002). Stem cell differentiation requires a paracrine pathway in the heart. FASEB J. 16, 1558–1566 [DOI] [PubMed] [Google Scholar]
- Behfar A, Perez-Terzic C, Faustino RS, Arrell DK, Hodgson DM, Yamada S, Puceat M, Niederländer N, Alekseev AE, Zingman LV, et al. (2007). Cardiopoietic programming of embryonic stem cells for tumor-free heart repair. J Exp Med. 204, 405–420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behfar A, Faustino RS, Arrell DK, Dzeja PP, Perez-Terzic C, Terzic A. (2008). Guided stem cell cardiopoiesis: discovery and translation. J Mol Cell Cardiol. 45, 523–529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bel A, Messas E, Agbulut O, Richard P, Samuel JL, Bruneval P, Hagège AA, Menasché P. (2003. Transplantation of autologous fresh bone marrow into infarcted myocardium: a word of caution. Circulation 108 Suppl. 1, II247–II252 [DOI] [PubMed] [Google Scholar]
- Belema Bedada F, Technau A, Ebelt H, Schulze M, Braun T. (2005). Activation of myogenic differentiation pathways in adult bone marrow-derived stem cells. Mol Cell Biol. 25, 9509–9519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beltrami AP, Barlucchi L, Torella D, Baker M, Limana F, Chimenti S, Kasahara H, Rota M, Musso E, Urbanek K, et al. (2003). Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 114, 763–776 [DOI] [PubMed] [Google Scholar]
- Bergmann O, Bhardwaj RD, Bernard S, Zdunek S, Barnabé-Heider F, Walsh S, Zupicich J, Alkass K, Buchholz BA, Druid H, et al. (2009). Evidence for cardiomyocyte renewal in humans. Science 324, 98–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowley E, O’Gorman DB, Gan BS. (2007). Beta-catenin signaling in fibroproliferative disease. J Surg Res. 138, 141–150 [DOI] [PubMed] [Google Scholar]
- Brack AS, Conboy MJ, Roy S, Lee M, Kuo CJ, Keller C, Rando TA. (2007). Increased Wnt signaling during aging alters muscle stem cell fate and increases fibrosis. Science 317, 807–810 [DOI] [PubMed] [Google Scholar]
- Brand T. (2003). Heart development: molecular insights into cardiac specification and early morphogenesis. Dev Biol. 258, 1–19 [DOI] [PubMed] [Google Scholar]
- Breitbach M, Bostani T, Roell W, Xia Y, Dewald O, Nygren JM, Fries JW, Tiemann K, Bohlen H, Hescheler J, et al. (2007). Potential risks of bone marrow cell transplantation into infarcted hearts. Blood 110, 1362–1369 [DOI] [PubMed] [Google Scholar]
- Buckingham M, Montarras D. (2008). Skeletal muscle stem cells. Curr Opin Genet Dev. 18, 330–336 [DOI] [PubMed] [Google Scholar]
- Buckingham M, Meilhac S, Zaffran S. (2005). Building the mammalian heart from two sources of myocardial cells. Nat Rev Genet. 6, 826–835 [DOI] [PubMed] [Google Scholar]
- Burt RK, Loh Y, Pearce W, Beohar N, Barr WG, Craig R, Wen Y, Rapp JA, Kessler J. (2008). Clinical applications of blood-derived and marrow-derived stem cells for nonmalignant diseases. JAMA 299, 925–936 [DOI] [PubMed] [Google Scholar]
- Cai CL, Liang X, Shi Y, Chu PH, Pfaff SL, Chen J, Evans S. (2003). Isl1 identifies a cardiac progenitor population that proliferates prior to differentiation and contributes a majority of cells to the heart. Dev Cell 5, 877–889 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai CL, Martin JC, Sun Y, Cui L, Wang L, Ouyang K, Yang L, Bu L, Liang X, Zhang X, et al. (2008). A myocardial lineage derives from Tbx18 epicardial cells. Nature 454, 104–108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caplan AI, Dennis JE. (2006). Mesenchymal stem cells as trophic mediators. J Cell Biochem. 98, 1076–1084 [DOI] [PubMed] [Google Scholar]
- Challen GA, Little MH. (2006). A side order of stem cells: the SP phenotype. Stem Cells 24, 3–12 [DOI] [PubMed] [Google Scholar]
- Chavakis E, Aicher A, Heeschen C, Sasaki K, Kaiser R, El Makhfi N, Urbich C, Peters T, Scharffetter-Kochanek K, Zeiher AM, et al. (2005). Role of beta2-integrins for homing and neovascularization capacity of endothelial progenitor cells. J Exp Med. 201, 63–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chavakis E, Urbich C, Dimmeler S. (2008). Homing and engraftment of progenitor cells: a prerequisite for cell therapy. J Mol Cell Cardiol. 45, 514–522 [DOI] [PubMed] [Google Scholar]
- Chen SL, Fang WW, Ye F, Liu YH, Qian J, Shan SJ, Zhang JJ, Chunhua RZ, Liao LM, Lin S, et al. (2004). Effect on left ventricular function of intracoronary transplantation of autologous bone marrow mesenchymal stem cell in patients with acute myocardial infarction. Am J Cardiol. 94, 92–95 [DOI] [PubMed] [Google Scholar]
- Chen S, Liu S, Tian S, Zhang J, Yei F, Duan B, Zhu Z, Lin S, Kwan TW. (2006). Intracoronary transplantation of autologous bone marrow mesenchymal stem cells for ischemic cardiomyopathy due to isolated chronic occluded left anterior descending artery. J Invasive Cardiol. 18, 552–556 [PubMed] [Google Scholar]
- Cheon SS, Cheah AY, Turley S, Nadesan P, Poon R, Clevers H, Alman BA. (2002). β-Catenin stabilization dysregulates mesenchymal cell proliferation, motility, and invasiveness and causes aggressive fibromatosis and hyperplastic cutaneous wounds. Proc Natl Acad Sci USA 99, 6973–6978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chien KR, Domian IJ, Parker KK. (2008). Cardiogenesis and the complex biology of regenerative cardiovascular medicine. Science 322, 1494–1497 [DOI] [PubMed] [Google Scholar]
- Choi JH, Choi J, Lee WS, Rhee I, Lee SC, Gwon HC, Lee SH, Choe YH, Kim DW, Suh W, et al. (2007). Lack of additional benefit of intracoronary transplantation of autologous peripheral blood stem cell in patients with acute myocardial infarction. Circ J. 71, 486–494 [DOI] [PubMed] [Google Scholar]
- Clevers H. (2006). Wnt/beta-catenin signaling in development and disease. Cell 127, 469–480 [DOI] [PubMed] [Google Scholar]
- Cohen ED, Tian Y, Morrisey EE. (2008). Wnt signaling: an essential regulator of cardiovascular differentiation, morphogenesis and progenitor self-renewal. Development 135, 789–798 [DOI] [PubMed] [Google Scholar]
- Dai W, Hale SL, Martin BJ, Kuang JQ, Dow JS, Wold LE, Kloner RA. (2005). Allogeneic mesenchymal stem cell transplantation in postinfarcted rat myocardium: short- and long-term effects. Circulation 112, 214–223 [DOI] [PubMed] [Google Scholar]
- David R, Brenner C, Stieber J, Schwarz F, Brunner S, Vollmer M, Mentele E, Müller-Höcker J, Kitajima S, Lickert H, et al. (2008). MesP1 drives vertebrate cardiovascular differentiation through Dkk-1-mediated blockade of Wnt-signalling. Nat Cell Biol. 10, 338–345 [DOI] [PubMed] [Google Scholar]
- Dawn B, Stein AB, Urbanek K, Rota M, Whang B, Rastaldo R, Torella D, Tang XL, Rezazadeh A, Kajstura J, et al. (2005). Cardiac stem cells delivered intravascularly traverse the vessel barrier, regenerate infarcted myocardium, and improve cardiac function. Proc Natl Acad Sci USA 102, 3766–3771 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deb A, Wang S, Skelding KA, Miller D, Simper D, Caplice NM. (2003). Bone marrow-derived cardiomyocytes are present in adult human heart: A study of gender-mismatched bone marrow transplantation patients. Circulation 107, 1247–1249 [DOI] [PubMed] [Google Scholar]
- Dimmeler S, Zeiher AM. (2008). Cell therapy of acute myocardial infarction: open questions. Cardiology 113, 155–160 [DOI] [PubMed] [Google Scholar]
- Doetschman TC, Eistetter H, Katz M, Schmidt W, Kemler R. (1985). The in vitro development of blastocyst-derived embryonic stem cell lines: formation of visceral yolk sac, blood islands and myocardium. J Embryol Exp Morphol. 87, 27–45 [PubMed] [Google Scholar]
- El-Helou V, Beguin PC, Assimakopoulos J, Clement R, Gosselin H, Brugada R, Aumont A, Biernaskie J, Villeneuve L, Leung TK, et al. (2008). The rat heart contains a neural stem cell population; role in sympathetic sprouting and angiogenesis. J Mol Cell Cardiol. 45, 694–702 [DOI] [PubMed] [Google Scholar]
- Ellison GM, Torella D, Karakikes I, Nadal-Ginard B. (2007. Myocyte death and renewal: modern concepts of cardiac cellular homeostasis. Nat. Clin. Pract. Cardiovasc. Med. 4 Suppl. 1, S52–S59 [DOI] [PubMed] [Google Scholar]
- Engler AJ, Sen S, Sweeney HL, Discher DE. (2006). Matrix elasticity directs stem cell lineage specification. Cell 126, 677–689 [DOI] [PubMed] [Google Scholar]
- Erbs S, Linke A, Adams V, Lenk K, Thiele H, Diederich KW, Emmrich F, Kluge R, Kendziorra K, Sabri O, et al. (2005). Transplantation of blood-derived progenitor cells after recanalization of chronic coronary artery occlusion: first randomized and placebo-controlled study. Circ Res. 97, 756–762 [DOI] [PubMed] [Google Scholar]
- Evans MJ, Kaufman MH. (1981). Establishment in culture of pluripotential cells from mouse embryos. Nature 292, 154–156 [DOI] [PubMed] [Google Scholar]
- Fazel S, Chen L, Weisel RD, Angoulvant D, Seneviratne C, Fazel A, Cheung P, Lam J, Fedak PW, Yau TM, et al. (2005). Cell transplantation preserves cardiac function after infarction by infarct stabilization: augmentation by stem cell factor. J Thorac Cardiovasc Surg. 130, 1310.e1–1310.e10 [DOI] [PubMed] [Google Scholar]
- Fernandez-Aviles F, San Roman JA, Garcia-Frade J, Fernandez ME, Penarrubia MJ, de la Fuente L, Gomez-Bueno M, Cantalapiedra A, Fernandez J, Gutierrez O, et al. (2004). Experimental and clinical regenerative capability of human bone marrow cells after myocardial infarction. Circ Res. 95, 742–748 [DOI] [PubMed] [Google Scholar]
- Flaherty MP, Abdel-Latif A, Li Q, Hunt G, Ranjan S, Ou Q, Tang XL, Johnson RK, Bolli R, Dawn B. (2008). Noncanonical Wnt11 signaling is sufficient to induce cardiomyogenic differentiation in unfractionated bone marrow mononuclear cells. Circulation 117, 2241–2252 [DOI] [PubMed] [Google Scholar]
- Frangogiannis NG. (2008). The immune system and cardiac repair. Pharmacol Res. 58, 88–111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gavira JJ, Herreros J, Perez A, Garcia-Velloso MJ, Barba J, Martin-Herrero F, Canizo C, Martin-Arnau A, Martí-Climent JM, Hernández M, et al. (2006). Autologous skeletal myoblast transplantation in patients with nonacute myocardial infarction: 1-year follow-up. J Thorac Cardiovasc Surg. 131, 799–804 [DOI] [PubMed] [Google Scholar]
- Ge J, Li Y, Qian J, Shi J, Wang Q, Niu Y, Fan B, Liu X, Zhang S, Sun A, et al. (2006). Efficacy of emergent transcatheter transplantation of stem cells for treatment of acute myocardial infarction (TCT-STAMI). Heart 92, 1764–1767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghostine S, Carrion C, Souza LC, Richard P, Bruneval P, Vilquin JT, Pouzet B, Schwartz K, Menasché P, Hagège AA. (2002. Long-term efficacy of myoblast transplantation on regional structure and function after myocardial infarction. Circulation 106 Suppl. 1, I131–I136 [PubMed] [Google Scholar]
- Gnecchi M, He H, Liang OD, Melo LG, Morello F, Mu H, Noiseux N, Zhang L, Pratt RE, Ingwall JS, et al. (2005). Paracrine action accounts for marked protection of ischemic heart by Akt-modified mesenchymal stem cells. Nat Med. 11, 367–368 [DOI] [PubMed] [Google Scholar]
- Gnecchi M, Zhang Z, Ni A, Dzau VJ. (2008). Paracrine mechanisms in adult stem cell signaling and therapy. Circ Res. 103, 1204–1219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodell MA, Brose K, Paradis G, Conner AS, Mulligan RC. (1996). Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. J Exp Med. 183, 1797–1806 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grauss RW, van Tuyn J, Steendijk P, Winter EM, Pijnappels DA, Hogers B, Gittenberger-de Groot AC, van der Geest R, van der Laarse A, de Vries AA, et al. (2008). Forced myocardin expression enhances the therapeutic effect of human mesenchymal stem cells after transplantation in ischemic mouse hearts. Stem Cells 26, 1083–1093 [DOI] [PubMed] [Google Scholar]
- Guan K, Nayernia K, Maier LS, Wagner S, Dressel R, Lee JH, Nolte J, Wolf F, Li M, Engel W, et al. (2006). Pluripotency of spermatogonial stem cells from adult mouse testis. Nature 440, 1199–1203 [DOI] [PubMed] [Google Scholar]
- Gulati R, Simari RD. (2009). Defining the potential for cell therapy for vascular disease using animal models. Dis Model Mech. 2, 130–137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haegebarth A, Clevers H. (2009). Wnt signaling, lgr5, and stem cells in the intestine and skin. Am J Pathol. 174, 715–721 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haider HK, Jiang S, Idris NM, Ashraf M. (2008). IGF-1-overexpressing mesenchymal stem cells accelerate bone marrow stem cell mobilization via paracrine activation of SDF-1alpha/CXCR4 signaling to promote myocardial repair. Circ Res. 103, 1300–1308 [DOI] [PubMed] [Google Scholar]
- Hao J, Daleo MA, Murphy CK, Yu PB, Ho JN, Hu J, Peterson RT, Hatzopoulos AK, Hong CC. (2008). Dorsomorphin, a selective small molecule inhibitor of BMP signaling, promotes cardiomyogenesis in embryonic stem cells. PLoS ONE 3, e2904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He KL, Yi GH, Sherman W, Zhou H, Zhang GP, Gu A, Kao R, Haimes HB, Harvey J, Roos E, et al. (2005). Autologous skeletal myoblast transplantation improved hemodynamics and left ventricular function in chronic heart failure dogs. J Heart Lung Transplant. 24, 1940–1949 [DOI] [PubMed] [Google Scholar]
- Heeschen C, Lehmann R, Honold J, Assmus B, Aicher A, Walter DH, Martin H, Zeiher AM, Dimmeler S. (2004). Profoundly reduced neovascularization capacity of bone marrow mononuclear cells derived from patients with chronic ischemic heart disease. Circulation 109, 1615–1622 [DOI] [PubMed] [Google Scholar]
- Hendrikx M, Hensen K, Clijsters C, Jongen H, Koninckx R, Bijnens E, Ingels M, Jacobs A, Geukens R, Dendale P, et al. (2006. Recovery of regional but not global contractile function by the direct intramyocardial autologous bone marrow transplantation: results from a randomized controlled clinical trial. Circulation 114 Suppl. 1, I101–I107 [DOI] [PubMed] [Google Scholar]
- Hierlihy AM, Seale P, Lobe CG, Rudnicki MA, Megeney LA. (2002). The post-natal heart contains a myocardial stem cell population. FEBS Lett. 530, 239–243 [DOI] [PubMed] [Google Scholar]
- Hinkel R, El-Aouni C, Olson T, Horstkotte J, Mayer S, Müller S, Willhauck M, Spitzweg C, Gildehaus F-G, Münzing W, et al. (2008). Thymosin β4 is an essential paracrine factor of embryonic endothelial progenitor cell mediated cardioprotection. Circulation 117, 2232–2240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinz B, Phan SH, Thannickal VJ, Galli A, Bochaton-Piallat ML, Gabbiani G. (2007). The myofibroblast: one function, multiple origins. Am J Pathol. 170, 1807–1816 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodgson DM, Behfar A, Zingman LV, Kane GC, Perez-Terzic C, Alekseev AE, Pucéat M, Terzic A. (2004). Stable benefit of embryonic stem cell therapy in myocardial infarction. Am J Physiol Heart Circ Physiol. 287, H471–H479 [DOI] [PubMed] [Google Scholar]
- Hofmann M, Wollert KC, Meyer GP, Menke A, Arseniev L, Hertenstein B, Ganser A, Knapp WH, Drexler H. (2005). Monitoring of bone marrow cell homing into the infarcted human myocardium. Circulation 111, 2198–2202 [DOI] [PubMed] [Google Scholar]
- Hollier BG, Evans K, Mani SA. (2009). The epithelial-to-mesenchymal transition and cancer stem cells: a coalition against cancer therapies. J Mammary Gland Biol Neoplasia 14, 29–43 [DOI] [PubMed] [Google Scholar]
- Hsieh PC, Segers VF, Davis ME, MacGillivray C, Gannon J, Molkentin JD, Robbins J, Lee RT. (2007). Evidence from a genetic fate-mapping study that stem cells refresh adult mammalian cardiomyocytes after injury. Nat Med. 13, 970–974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huangfu D, Osafune K, Maehr R, Guo W, Eijkelenboom A, Chen S, Muhlestein W, Melton DA. (2008). Induction of pluripotent stem cells from primary human fibroblasts with only Oct4 and Sox2. Nat Biotechnol. 26, 1269–1275 [DOI] [PubMed] [Google Scholar]
- Hutson MR, Kirby ML. (2007). Model systems for the study of heart development and disease. Cardiac neural crest and conotruncal malformations. Semin Cell Dev Biol. 18, 101–110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ince H, Petzsch M, Rehders TC, Chatterjee T, Nienaber CA. (2004). Transcatheter transplantation of autologous skeletal myoblasts in postinfarction patients with severe left ventricular dysfunction. J Endovasc Ther. 11, 695–704 [DOI] [PubMed] [Google Scholar]
- Jackson KA, Majka SM, Wang H, Pocius J, Hartley CJ, Majesky MW, Entman ML, Michael LH, Hirschi KK, Goodell MA. (2001). Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. J Clin Invest. 107, 1395–1402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaenisch R, Young R. (2008). Stem cells, the molecular circuitry of pluripotency and nuclear reprogramming. Cell 132, 567–582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janssens S, Dubois C, Bogaert J, Theunissen K, Deroose C, Desmet W, Kalantzi M, Herbots L, Sinnaeve P, Dens J, et al. (2006). Autologous bone marrow-derived stem-cell transfer in patients with ST-segment elevation myocardial infarction: double-blind, randomised controlled trial. Lancet 367, 113–121 [DOI] [PubMed] [Google Scholar]
- Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR, Reyes M, Lenvik T, Lund T, Blackstad M, et al. (2002). Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418, 41–49 [DOI] [PubMed] [Google Scholar]
- Kajstura J, Rota M, Whang B, Cascapera S, Hosoda T, Bearzi C, Nurzynska D, Kasahara H, Zias E, Bonafé M, et al. (2005). Bone marrow cells differentiate in cardiac cell lineages after infarction independently of cell fusion. Circ Res. 96, 127–137 [DOI] [PubMed] [Google Scholar]
- Kajstura J, Urbanek K, Rota M, Bearzi C, Hosoda T, Bolli R, Anversa P, Leri A. (2008). Cardiac stem cells and myocardial disease. J Mol Cell Cardiol. 45, 505–513 [DOI] [PubMed] [Google Scholar]
- Kalani MY, Cheshier SH, Cord BJ, Bababeygy SR, Vogel H, Weissman IL, Palmer TD, Nusse R. (2008). Wnt-mediated self-renewal of neural stem/progenitor cells. Proc Natl Acad Sci USA 105, 16970–16975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalluri R, Neilson EG. (2003). Epithelial-mesenchymal transition and its implications for fibrosis. J Clin Invest. 112, 1776–1784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamihata H, Matsubara H, Nishiue T, Fujiyama S, Tsutsumi Y, Ozono R, Masaki H, Mori Y, Iba O, Tateishi E, et al. (2001). Implantation of bone marrow mononuclear cells into ischemic myocardium enhances collateral perfusion and regional function via side supply of angioblasts, angiogenic ligands, and cytokines. Circulation 104, 1046–1052 [DOI] [PubMed] [Google Scholar]
- Kang H, Lee H, Na S, Chang SA, Park KW, Kim HK, Kim SY, Chang HJ, Lee W, Kang WJ, et al. (2006. Differential effect of intracoronary infusion of mobilized peripheral blood stem cells by granulocyte colony stimulating factor on left ventricular function and remodeling in patients with acute myocardial infarction versus old myocardial infarction: the MAGIC Cell-3-DES randomized, controlled trial. Circulation 114 Suppl. 1, I145–I151 [DOI] [PubMed] [Google Scholar]
- Katritsis DG, Sotiropoulou PA, Karvouni E, Karabinos I, Korovesis S, Perez SA, Voridis EM, Papamichail M. (2005). Transcoronary transplantation of autologous mesenchymal stem cells and endothelial progenitors into infarcted human myocardium. Catheter Cardiovasc Interv. 65, 321–329 [DOI] [PubMed] [Google Scholar]
- Kattman SJ, Huber TL, Keller GM. (2006). Multipotent flk-1+ cardiovascular progenitor cells give rise to the cardiomyocyte, endothelial, and vascular smooth muscle lineages. Dev Cell 11, 723–732 [DOI] [PubMed] [Google Scholar]
- Kawamoto A, Tkebuchava T, Yamaguchi J, Nishimura H, Yoon YS, Milliken C, Uchida S, Masuo O, Iwaguro H, Ma H, et al. (2003). Intramyocardial transplantation of autologous endothelial progenitor cells for therapeutic neovascularization of myocardial ischemia. Circulation 107, 461–468 [DOI] [PubMed] [Google Scholar]
- Kehat I, Kenyagin-Karsenti D, Snir M, Segev H, Amit M, Gepstein A, Livne E, Binah O, Itskovitz-Eldor J, Gepstein L. (2001). Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J Clin Invest. 108, 407–414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kehat I, Khimovich L, Caspi O, Gepstein A, Shofti R, Arbel G, Huber I, Satin J, Itskovitz-Eldor J, Gepstein L. (2004). Electromechanical integration of cardiomyocytes derived from human embryonic stem cells. Nat Biotechnol. 22, 1282–1289 [DOI] [PubMed] [Google Scholar]
- Keller G. (2005). Embryonic stem cell differentiation: emergence of a new era in biology and medicine. Genes Dev. 19, 1129–1155 [DOI] [PubMed] [Google Scholar]
- Kinnaird T, Stabile E, Burnett MS, Epstein SE. (2004). Bone-marrow-derived cells for enhancing collateral development: mechanisms, animal data, and initial clinical experiences. Circ Res. 95, 354–363 [DOI] [PubMed] [Google Scholar]
- Klein HM, Ghodsizad A, Marktanner R, Poll L, Voelkel T, Mohammad Hasani MR, Piechaczek C, Feifel N, Stockschlaeder M, Burchardt ER, et al. (2007). Intramyocardial implantation of CD133+ stem cells improved cardiac function without bypass surgery. Heart Surg Forum 10, E66–E69 [DOI] [PubMed] [Google Scholar]
- Klug MG, Soonpaa MH, Koh GY, Field LJ. (1996). Genetically selected cardiomyocytes from differentiating embronic stem cells form stable intracardiac grafts. J Clin Invest. 98, 216–224 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kocher AA, Schuster MD, Szabolcs MJ, Takuma S, Burkhoff D, Wang J, Homma S, Edwards NM, Itescu S. (2001). Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med. 7, 430–436 [DOI] [PubMed] [Google Scholar]
- Kofidis T, Lebl DR, Martinez EC, Hoyt G, Tanaka M, Robbins RC. (2005. Novel injectable bioartificial tissue facilitates targeted, less invasive, large-scale tissue restoration on the beating heart after myocardial injury. Circulation 112 Suppl. 9, I173–I177 [DOI] [PubMed] [Google Scholar]
- Koyanagi M, Haendeler J, Badorff C, Brandes RP, Hoffmann J, Pandur P, Zeiher AM, Kühl M, Dimmeler S. (2005). Non-canonical Wnt signaling enhances differentiation of human circulating progenitor cells to cardiomyogenic cells. J Biol Chem. 280, 16838–16842 [DOI] [PubMed] [Google Scholar]
- Koyanagi M, Bushoven P, Iwasaki M, Urbich C, Zeiher AM, Dimmeler S. (2007). Notch signaling contributes to the expression of cardiac markers in human circulating progenitor cells. Circ Res. 101, 1139–1145 [DOI] [PubMed] [Google Scholar]
- Kudo M, Wang Y, Wani MA, Xu M, Ayub A, Ashraf M. (2003). Implantation of bone marrow stem cells reduces the infarction and fibrosis in ischemic mouse heart. J Mol Cell Cardiol. 35, 1113–1119 [DOI] [PubMed] [Google Scholar]
- Kupatt C, Horstkotte J, Vlastos GA, Pfosser A, Lebherz C, Semisch M, Thalgott M, Büttner K, Browarzyk C, Mages J, et al. (2005a). Embryonic endothelial progenitor cells expressing a broad range of pro-angiogenic and remodeling factors enhance vascularization and tissue recovery in acute and chronic ischemia. FASEB J. 19, 1576–1578 [DOI] [PubMed] [Google Scholar]
- Kupatt C, Hinkel R, Lamparter M, von Brühl M-L, Pohl T, Horstkotte J, Beck H, Müller S, Delker S, Gildehaus F-J, et al. (2005b. Retroinfusion of embryonic endothelial progenitor cells attenuates ischemia-reperfusion injury in pigs: Role of PI3-Kinase/AKT. Circulation 112 Suppl. 9, I117–I122 [DOI] [PubMed] [Google Scholar]
- Kwon C, Arnold J, Hsiao EC, Taketo MM, Conklin BR, Srivastava D. (2007). Canonical Wnt signaling is a positive regulator of mammalian cardiac progenitors. Proc Natl Acad Sci USA 104, 10894–10899 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laflamme MA, Myerson D, Saffitz JE, Murry CE. (2002). Evidence for cardiomyocyte repopulation by extracardiac progenitors in transplanted human hearts. Circ Res. 90, 634–640 [DOI] [PubMed] [Google Scholar]
- Laflamme MA, Gold J, Xu C, Hassanipour M, Rosler E, Police S, Muskheli V, Murry CE. (2005). Formation of human myocardium in the rat heart from human embryonic stem cells. Am J Pathol. 167, 663–671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laflamme MA, Chen KY, Naumova AV, Muskheli V, Fugate JA, Dupras SK, Reinecke H, Xu C, Hassanipour M, Police S, et al. (2007). Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nat Biotechnol. 25, 1015–1024 [DOI] [PubMed] [Google Scholar]
- Lamparter M, Hatzopoulos AK. (2007). The biology of embryonic and adult endothelial progenitor cells. In Therapeutic Neovascularization – Quo Vadis? (ed. Deindl E, Kupatt C.), pp. 197–213 Dordrecht, The Netherlands: Springer [Google Scholar]
- Laugwitz KL, Moretti A, Lam J, Gruber P, Chen Y, Woodard S, Lin LZ, Cai CL, Lu MM, Reth M, et al. (2005). Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature 433, 647–653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laugwitz KL, Moretti A, Caron L, Nakano A, Chien KR. (2008). Islet1 cardiovascular progenitors: a single source for heart lineages? Development 135, 193–205 [DOI] [PubMed] [Google Scholar]
- Leesar MA, Stoddard M, Ahmed M, Broadbent J, Bolli R. (1997). Preconditioning of human myocardium with adenosine during coronary angioplasty. Circulation 95, 2500–2507 [DOI] [PubMed] [Google Scholar]
- Leobon B, Garcin I, Menasche P, Vilquin JT, Audinat E, Charpak S. (2003). Myoblasts transplanted into rat infarcted myocardium are functionally isolated from their host. Proc Natl Acad Sci USA 100, 7808–7811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lepilina A, Coon AN, Kikuchi K, Holdway JE, Roberts RW, Burns CG, Poss KD. (2006). A dynamic epicardial injury response supports progenitor cell activity during zebrafish heart regeneration. Cell 127, 607–619 [DOI] [PubMed] [Google Scholar]
- Li ZQ, Zhang M, Jing YZ, Zhang WW, Liu Y, Cui LJ, Yuan L, Liu XZ, Yu X, Hu TS. (2007). The clinical study of autologous peripheral blood stem cell transplantation by intracoronary infusion in patients with acute myocardial infarction (AMI). Int J Cardiol. 115, 52–56 [DOI] [PubMed] [Google Scholar]
- Limana F, Zacheo A, Mocini D, Mangoni A, Borsellino G, Diamantini A, De Mori R, Battistini L, Vigna E, Santini M, et al. (2007). Identification of myocardial and vascular precursor cells in human and mouse epicardium. Circ Res. 101, 255–265 [DOI] [PubMed] [Google Scholar]
- Lin L, Cui L, Zhou W, Dufort D, Zhang X, Cai CL, Bu L, Yang L, Martin J, Kemler R, et al. (2007). Beta-catenin directly regulates Islet1 expression in cardiovascular progenitors and is required for multiple aspects of cardiogenesis. Proc Natl Acad Sci USA 104, 9313–9318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Losordo DW, Schatz RA, White CJ, Udelson JE, Veereshwarayya V, Durgin M, Poh KK, Weinstein R, Kearney M, Chaudhry M, et al. (2007). Intramyocardial transplantation of autologous CD34+ stem cells for intractable angina: a phase I/IIa double-blind, randomized controlled trial. Circulation 115, 3165–3172 [DOI] [PubMed] [Google Scholar]
- Lowry WE, Richter L. (2007). Signaling in adult stem cells. Front Biosci. 12, 3911–3927 [DOI] [PubMed] [Google Scholar]
- Luis TC, Weerkamp F, Naber BA, Baert MR, de Haas EF, Nikolic T, Heuvelmans S, De Krijger RR, van Dongen JJ, Staal FJ. (2009). Wnt3a deficiency irreversibly impairs hematopoietic stem cell self-renewal and leads to defects in progenitor cell differentiation. Blood 113, 546–554 [DOI] [PubMed] [Google Scholar]
- Lunde K, Solheim S, Aakhus S, Arnesen H, Abdelnoor M, Egeland T, Endresen K, Ilebekk A, Mangschau A, Fjeld JG, et al. (2006). Intracoronary injections of mononuclear bone marrow cells in acute myocardial infarction. N Engl J Med. 355, 1199–1209 [DOI] [PubMed] [Google Scholar]
- Ma Q, Zhou B, Pu WT. (2008). Reassessment of Isl1 and Nkx2-5 cardiac fate maps using a Gata4-based reporter of Cre activity. Dev Biol. 323, 98–104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makino S, Fukuda K, Miyoshi S, Konishi F, Kodama H, Pan J, Sano M, Takahashi T, Hori S, Abe H, et al. (1999). Cardiomyocytes can be generated from marrow stromal cells in vitro. J Clin Invest. 103, 697–705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mangi AA, Noiseux N, Kong D, He H, Rezvani M, Ingwall JS, Dzau VJ. (2003). Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts. Nat Med. 9, 1195–1201 [DOI] [PubMed] [Google Scholar]
- Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, et al. (2008). The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133, 704–715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin GR. (1981). Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci USA 78, 7634–7638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin CM, Meeson AP, Robertson SM, Hawke TJ, Richardson JA, Bates S, Goetsch SC, Gallardo TD, Garry DJ. (2004). Persistent expression of the ATP-binding cassette transporter, Abcg2, identifies cardiac SP cells in the developing and adult heart. Dev Biol. 265, 262–275 [DOI] [PubMed] [Google Scholar]
- Matsuura K, Nagai T, Nishigaki N, Oyama T, Nishi J, Wada H, Sano M, Toko H, Akazawa H, Sato T, et al. (2004). Adult cardiac Sca-1-positive cells differentiate into beating cardiomyocytes. J Biol Chem. 279, 11384–11391 [DOI] [PubMed] [Google Scholar]
- Mauritz C, Schwanke K, Reppel M, Neef S, Katsirntaki K, Maier LS, Nguemo F, Menke S, Haustein M, Hescheler J, et al. (2008). Generation of functional murine cardiac myocytes from induced pluripotent stem cells. Circulation 118, 507–517 [DOI] [PubMed] [Google Scholar]
- McMurray JJ, Pfeffer MA. (2005). Heart failure. Lancet 365, 1877–1889 [DOI] [PubMed] [Google Scholar]
- Meluzín J, Mayer J, Groch J, Janousek S, Hornácek I, Hlinomaz O, Kala P, Panovsky R, Prásek J, Kamínek M, et al. (2006). Autologous transplantation of mononuclear bone marrow cells in patients with acute myocardial infarction: the effect of the dose of transplanted cells on myocardial function. Am Heart J. 152, e9–e15 [DOI] [PubMed] [Google Scholar]
- Meluzín J, Janousek S, Mayer J, Groch L, Hornácek I, Hlinomaz O, Kala P, Panovsky R, Prásek J, Kamínek M, et al. (2008). Three-, 6-, and 12-month results of autologous transplantation of mononuclear bone marrow cells in patients with acute myocardial infarction. Int J Cardiol. 128, 185–192 [DOI] [PubMed] [Google Scholar]
- Menasché P. (2007). Skeletal myoblasts as a therapeutic agent. Prog Cardiovasc Dis. 50, 7–17 [DOI] [PubMed] [Google Scholar]
- Menasché P, Alfieri O, Janssens S, McKenna W, Reichenspurner H, Trinquart L, Vilquin JT, Marolleau JP, Seymour B, Larghero J, et al. (2008). The Myoblast Autologous Grafting in Ischemic Cardiomyopathy (MAGIC) trial: first randomized placebo-controlled study of myoblast transplantation. Circulation 117, 1189–1200 [DOI] [PubMed] [Google Scholar]
- Messina E, De Angelis L, Frati G, Morrone S, Chimenti S, Fiordaliso F, Salio M, Battaglia M, Latronico MV, Coletta M, et al. (2004). Isolation and expansion of adult cardiac stem cells from human and murine heart. Circ Res. 95, 911–921 [DOI] [PubMed] [Google Scholar]
- Meyer GP, Wollert KC, Lotz J, Steffens J, Lippolt P, Fichtner S, Hecker H, Schaefer A, Arseniev L, Hertenstein B, et al. (2006). Intracoronary bone marrow cell transfer after myocardial infarction: eighteen months’ follow-up data from the randomized, controlled BOOST (BOne marrow transfer to enhance ST-elevation infarct regeneration) trial. Circulation 113, 1287–1294 [DOI] [PubMed] [Google Scholar]
- Min JY, Yang Y, Converso KL, Liu L, Huang Q, Morgan JP, Xiao YF. (2002). Transplantation of embryonic stem cells improves cardiac function in postinfarcted rats. J Appl Physiol. 92, 288–296 [DOI] [PubMed] [Google Scholar]
- Mirotsou M, Zhang Z, Deb A, Zhang L, Gnecchi M, Noiseux N, Mu H, Pachori A, Dzau V. (2007). Secreted frizzled related protein 2 (Sfrp2) is the key Akt-mesenchymal stem cell-released paracrine factor mediating myocardial survival and repair. Proc Natl Acad Sci USA 104, 1643–1648 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyahara Y, Nagaya N, Kataoka M, Yanagawa B, Tanaka K, Hao H, Ishino K, Ishida H, Shimizu T, Kangawa K, et al. (2006). Monolayered mesenchymal stem cells repair scarred myocardium after myocardial infarction. Nat Med. 12, 459–465 [DOI] [PubMed] [Google Scholar]
- Mocini D, Staibano M, Mele L, Giannantoni P, Menichella G, Colivicchi F, Sordini P, Salera P, Tubaro M, Santini M. (2006). Autologous bone marrow mononuclear cell transplantation in patients undergoing coronary artery bypass grafting. Am Heart J. 151, 192–197 [DOI] [PubMed] [Google Scholar]
- Moorman AF, Christoffels VM, Anderson RH, van den Hoff MJ. (2007). The heart-forming fields: one or multiple? Philos Trans R Soc Lond B Biol Sci. 362, 1257–1265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moretti A, Caron L, Nakano A, Lam JT, Bernshausen A, Chen Y, Qyang Y, Bu L, Sasaki M, Martin-Puig S, et al. (2006). Multipotent embryonic isl1+ progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 127, 1151–1165 [DOI] [PubMed] [Google Scholar]
- Mouquet F, Pfister O, Jain M, Oikonomopoulos A, Ngoy S, Summer R, Fine A, Liao R. (2005). Restoration of cardiac progenitor cells after myocardial infarction by self-proliferation and selective homing of bone marrow-derived stem cells. Circ Res. 97, 1090–1092 [DOI] [PubMed] [Google Scholar]
- Müller P, Pfeiffer P, Koglin J, Schäfers HJ, Seeland U, Janzen I, Urbschat S, Böhm M. (2002). Cardiomyocytes of noncardiac origin in myocardial biopsies of human transplanted hearts. Circulation 106, 31–35 [DOI] [PubMed] [Google Scholar]
- Murry CE, Keller G. (2008). Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development. Cell 132, 661–680 [DOI] [PubMed] [Google Scholar]
- Murry CE, Wiseman RW, Schwartz SM, Hauschka SD. (1996). Skeletal myoblast transplantation for repair of myocardial necrosis. J Clin Invest. 98, 2512–2523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murry CE, Soonpaa MH, Reinecke H, Nakajima H, Nakajima HO, Rubart M, Pasumarthi KB, Virag JI, Bartelmez SH, Poppa V, et al. (2004). Haematopoietic stem cells do not transdifferentiate into cardiac myocytes in myocardial infarcts. Nature 428, 664–668 [DOI] [PubMed] [Google Scholar]
- Nakamura T, Sano M, Songyang Z, Schneider MD. (2003). A Wnt- and beta-catenin-dependent pathway for mammalian cardiac myogenesis. Proc Natl Acad Sci USA 100, 5834–5839 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura T, Colbert MC, Robbins J. (2006). Neural crest cells retain multipotential characteristics in the developing valves and label the cardiac conduction system. Circ Res. 98, 1547–1554 [DOI] [PubMed] [Google Scholar]
- Nakaya Y, Sheng G. (2008). Epithelial to mesenchymal transition during gastrulation: an embryological view. Dev Growth Differ. 50, 755–766 [DOI] [PubMed] [Google Scholar]
- Narazaki G, Uosaki H, Teranishi M, Okita K, Kim B, Matsuoka S, Yamanaka S, Yamashita JK. (2008). Directed and systematic differentiation of cardiovascular cells from mouse induced pluripotent stem cells. Circulation 118, 498–506 [DOI] [PubMed] [Google Scholar]
- Neilson EG. (2006). Mechanisms of disease: fibroblasts – a new look at an old problem. Nat Clin Pract Nephrol. 2, 101–108 [DOI] [PubMed] [Google Scholar]
- Noiseux N, Gnecchi M, Lopez-Ilasaca M, Zhang L, Solomon SD, Deb A, Dzau VJ, Pratt RE. (2006). Mesenchymal stem cells overexpressing Akt dramatically repair infarcted myocardium and improve cardiac function despite infrequent cellular fusion or differentiation. Mol Ther. 14, 840–850 [DOI] [PubMed] [Google Scholar]
- Norris RA, Moreno-Rodriguez RA, Sugi Y, Hoffman S, Amos J, Hart MM, Potts JD, Goodwin RL, Markwald RR. (2008). Periostin regulates atrioventricular valve maturation. Dev Biol. 316, 200–213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nussbaum J, Minami E, Laflamme MA, Virag JA, Ware CB, Masino A, Muskheli V, Pabon L, Reinecke H, Murry CE. (2007). Transplantation of undifferentiated murine embryonic stem cells in the heart: teratoma formation and immune response. FASEB J. 21, 1345–1357 [DOI] [PubMed] [Google Scholar]
- Nusse R. (2008). Wnt signaling and stem cell control. Cell Res. 18, 523–527 [DOI] [PubMed] [Google Scholar]
- Oh H, Bradfute SB, Gallardo TD, Nakamura T, Gaussin V, Mishina Y, Pocius J, Michael LH, Behringer RR, Garry DJ, et al. (2003). Cardiac progenitor cells from adult myocardium: homing, differentiation, and fusion after infarction. Proc Natl Acad Sci USA 100, 12313–12318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okita K, Ichisaka T, Yamanaka S. (2007). Generation of germ-line competent induced pluripotent stem cells. Nature 448, 313–317 [DOI] [PubMed] [Google Scholar]
- Olson EN. (2006). Gene regulatory networks in the evolution and development of the heart. Science 313, 1922–1927 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orlic D, Kajstura J, Chimenti S, Jakoniuk I, Anderson SM, Li B, Pickel J, McKay R, Nadal-Ginard B, Bodine DM, et al. (2001a). Bone marrow cells regenerate infarcted myocardium. Nature 410, 701–705 [DOI] [PubMed] [Google Scholar]
- Orlic D, Kajstura J, Chimenti S, Limana F, Jakoniuk I, Quaini F, Nadal-Ginard B, Bodine DM, Leri A, Anversa P. (2001b). Mobilized bone marrow cells repair the infarcted heart, improving function and survival. Proc Natl Acad Sci USA 98, 10344–10349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otto A, Schmidt C, Luke G, Allen S, Valasek P, Muntoni F, Lawrence-Watt D, Patel K. (2008). Canonical Wnt signalling induces satellite-cell proliferation during adult skeletal muscle regeneration. J Cell Sci. 121, 2939–2950 [DOI] [PubMed] [Google Scholar]
- Oyama T, Nagai T, Wada H, Naito AT, Matsuura K, Iwanaga K, Takahashi T, Goto M, Mikami Y, Yasuda N, et al. (2007). Cardiac side population cells have a potential to migrate and differentiate into cardiomyocytes in vitro and in vivo. J Cell Biol. 176, 329–341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pallante BA, Duignan I, Okin D, Chin A, Bressan MC, Mikawa T, Edelberg JM. (2007). Bone marrow Oct3/4+ cells differentiate into cardiac myocytes via age-dependent paracrine mechanisms. Circ Res. 100, e1–e11 [DOI] [PubMed] [Google Scholar]
- Perez-Pomares JM, Gonzalez-Rosa JM, Munoz-Chapuli R. (2009. Building the vertebrate heart – an evolutionary approach to cardiac development. Int. J. Dev. Biol. Jan 23 [Epub ahead of print] [doi: 10.1387/ijdb.072409jp] [DOI] [PubMed] [Google Scholar]
- Perin EC, Dohmann HF, Borojevic R, Silva SA, Sousa AL, Silva GV, Mesquita CT, Belém L, Vaughn WK, Rangel FO, et al. (2004. Improved exercise capacity and ischemia 6 and 12 months after transendocardial injection of autologous bone marrow mononuclear cells for ischemic cardiomyopathy. Circulation 110 Suppl. 1, II213–II218 [DOI] [PubMed] [Google Scholar]
- Person AD, Klewer SE, Runyan RB. (2005). Cell biology of cardiac cushion development. Int Rev Cytol. 243, 287–335 [DOI] [PubMed] [Google Scholar]
- Pfeffer MA, Braunwald E. (1990). Ventricular remodeling after myocardial infarction. Experimental observations and clinical implications. Circulation 81, 1161–1172 [DOI] [PubMed] [Google Scholar]
- Pfister O, Mouquet F, Jain M, Summer R, Helmes M, Fine A, Colucci WS, Liao R. (2005). CD31- but not CD31+ cardiac side population cells exhibit functional cardiomyogenic differentiation. Circ Res. 97, 52–61 [DOI] [PubMed] [Google Scholar]
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. (1999). Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147 [DOI] [PubMed] [Google Scholar]
- Poelmann RE, Jongbloed MR, Molin DG, Fekkes ML, Wang Z, Fishman GI, Doetschman T, Azhar M, Gittenberger-de Groot AC. (2004). The neural crest is contiguous with the cardiac conduction system in the mouse embryo: a role in induction? Anat Embryol (Berl) 208, 389–393 [DOI] [PubMed] [Google Scholar]
- Prall OW, Menon MK, Solloway MJ, Watanabe Y, Zaffran S, Bajolle F, Biben C, Mcbride JJ, Robertson BR, Chaulet H, et al. (2007). An Nkx2-5/Bmp2/Smad1 negative feedback loop controls heart progenitor specification and proliferation. Cell 128, 947–959 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quaini F, Urbanek K, Beltrami AP, Finato N, Beltrami CA, Nadal-Ginard B, Kajstura J, Leri A, Anversa P. (2002). Chimerism of the transplanted heart. N Engl J Med. 346, 5–15 [DOI] [PubMed] [Google Scholar]
- Qyang Y, Martin-Puig S, Chiravuri M, Chen S, Xu H, Bu L, Jiang X, Lin L, Granger A, Moretti A, et al. (2007). The renewal and differentiation of Isl1+ cardiovascular progenitors are controlled by a Wnt/beta-catenin pathway. Cell Stem Cell 1, 165–179 [DOI] [PubMed] [Google Scholar]
- Reese DE, Mikawa T, Bader DM. (2002). Development of the coronary vessel system. Circ Res. 91, 761–768 [DOI] [PubMed] [Google Scholar]
- Reffelmann T, Könemann S, Kloner RA. (2009). Promise of blood- and bone marrow-derived stem cell transplantation for functional cardiac repair: putting it in perspective with existing therapy. J Am Coll Cardiol. 53, 305–308 [DOI] [PubMed] [Google Scholar]
- Reinecke H, Poppa V, Murry CE. (2002). Skeletal muscle stem cells do not transdifferentiate into cardiomyocytes after cardiac grafting. J Mol Cell Cardiol. 34, 241–249 [DOI] [PubMed] [Google Scholar]
- Reinecke H, Minami E, Zhu WZ, Laflamme MA. (2008). Cardiogenic differentiation and transdifferentiation of progenitor cells. Circ Res. 103, 1058–1071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roell W, Fan Y, Xia Y, Stoecker E, Sasse P, Kolossov E, Bloch W, Metzner H, Schmitz C, Addicks K, et al. (2002). Cellular cardiomyoplasty in a transgenic mouse model. Transplantation 73, 462–465 [DOI] [PubMed] [Google Scholar]
- Roell W, Lewalter T, Sasse P, Tallini YN, Choi BR, Breitbach M, Doran R, Becher UM, Hwang SM, Bostani T, et al. (2007). Engraftment of connexin 43-expressing cells prevents post-infarct arrhythmia. Nature 450, 819–824 [DOI] [PubMed] [Google Scholar]
- Rose RA, Jiang H, Wang X, Helke S, Tsoporis JN, Gong N, Keating SC, Parker TG, Backx PH, Keating A. (2008). Bone marrow-derived mesenchymal stromal cells express cardiac-specific markers, retain the stromal phenotype, and do not become functional cardiomyocytes in vitro. Stem Cells 26, 2884–2892 [DOI] [PubMed] [Google Scholar]
- Rota M, Kajstura J, Hosoda T, Bearzi C, Vitale S, Esposito G, Iaffaldano G, Padin-Ireguas ME, Gonzalez A, Rizzi R, et al. (2007). Bone marrow cells adopt the cardiomyogenic fate in vivo. Proc Natl Acad Sci USA 104, 17783–17788 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rota M, Padin-Iruegas ME, Misao Y, De Angelis A, Maestroni S, Ferreira-Martins J, Fiumana E, Rastaldo R, Arcarese ML, Mitchell TS, et al. (2008). Local activation or implantation of cardiac progenitor cells rescues scarred infarcted myocardium improving cardiac function. Circ Res. 103, 107–116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruan W, Pan C, Huang G, Li Y, Ge JB, Shu XH. (2005). Assessment of left ventricular segmental function after autologous bone marrow stem cells transplantation in patients with acute myocardial infarction by tissue tracking and strain imaging. Chin Med J (Engl) 118, 1175–1181 [PubMed] [Google Scholar]
- Ryzhov S, Solenkova NV, Goldstein AE, Lamparter M, Fleenor Y, Young PP, Greelish JP, Byrne JG, Vaughan DE, Biaggioni I, et al. (2008). Adenosine receptor-mediated adhesion of endothelial progenitors to cardiac microvascular endothelial cells. Circ Res. 102, 356–363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sachinidis A, Fleischmann BK, Kolossov E, Wartenberg M, Sauer H, Hescheler J. (2003). Cardiac specific differentiation of mouse embryonic stem cells. Cardiovasc Res. 58, 278–291 [DOI] [PubMed] [Google Scholar]
- Sauka-Spengler T, Bronner-Fraser M. (2008). A gene regulatory network orchestrates neural crest formation. Nat Rev Mol Cell Biol. 9, 557–568 [DOI] [PubMed] [Google Scholar]
- Schächinger V, Erbs S, Elsasser A, Haberbosch W, Hambrecht R, Holschermann H, Yu J, Corti R, Mathey DG, Hamm CW, et al. (2006a). Intracoronary bone marrow-derived progenitor cells in acute myocardial infarction. N Engl J Med. 355, 1210–1221 [DOI] [PubMed] [Google Scholar]
- Schächinger V, Erbs S, Elsässer A, Haberbosch W, Hambrecht R, Hölschermann H, Yu J, Corti R, Mathey DG, Hamm CW, et al. (2006b). Improved clinical outcome after intracoronary administration of bone-marrow-derived progenitor cells in acute myocardial infarction: final 1-year results of the REPAIR-AMI trial. Eur Heart J. 27, 2775–2783 [DOI] [PubMed] [Google Scholar]
- Schenke-Layland K, Rhodes KE, Angelis E, Butylkova Y, Heydarkhan-Hagvall S, Gekas C, Zhang R, Goldhaber JI, Mikkola HK, Plath K, et al. (2008). Reprogrammed mouse fibroblasts differentiate into cells of the cardiovascular and hematopoietic lineages. Stem Cells 26, 1537–1546 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schönberger J, Seidman CE. (2001). Many roads lead to a broken heart: the genetics of dilated cardiomyopathy. Am J Hum Genet. 69, 249–260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segers VF, Lee RT. (2008). Stem-cell therapy for cardiac disease. Nature 451, 937–942 [DOI] [PubMed] [Google Scholar]
- Shen J, DiCorleto PE. (2008). Adenosine prompts the heart to recruit endothelial progenitors. Circ Res. 102, 280–282 [DOI] [PubMed] [Google Scholar]
- Shenje LT, Field LJ, Pritchard CA, Guerin CJ, Rubart M, Soonpaa MH, Ang KL, Galinanes M. (2008). Lineage tracing of cardiac explant derived cells. PLoS ONE 3, e1929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi X, Garry DJ. (2006). Muscle stem cells in development, regeneration, and disease. Genes Dev. 20, 1692–1708 [DOI] [PubMed] [Google Scholar]
- Shiota M, Heike T, Haruyama M, Baba S, Tsuchiya A, Fujino H, Kobayashi H, Kato T, Umeda K, Yoshimoto M, et al. (2007). Isolation and characterization of bone marrow-derived mesenchymal progenitor cells with myogenic and neuronal properties. Exp Cell Res. 313, 1008–1023 [DOI] [PubMed] [Google Scholar]
- Silva GV, Litovsky S, Assad JA, Sousa AL, Martin BJ, Vela D, Coulter SC, Lin J, Ober J, Vaughn WK, et al. (2005). Mesenchymal stem cells differentiate into an endothelial phenotype, enhance vascular density, and improve heart function in a canine chronic ischemia model. Circulation 111, 150–156 [DOI] [PubMed] [Google Scholar]
- Singla DK, Hacker TA, Ma L, Douglas PS, Sullivan R, Lyons GE, Kamp TJ. (2006). Transplantation of embryonic stem cells into the infarcted mouse heart: formation of multiple cell types. J Mol Cell Cardiol. 40, 195–200 [DOI] [PubMed] [Google Scholar]
- Slack JM. (2008). Origin of stem cells in organogenesis. Science 322, 1498–1501 [DOI] [PubMed] [Google Scholar]
- Smart N, Risebro CA, Melville AA, Moses K, Schwartz RJ, Chien KR, Riley PR. (2007). Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization. Nature 445, 177–182 [DOI] [PubMed] [Google Scholar]
- Smith RR, Barile L, Cho HC, Leppo MK, Hare JM, Messina E, Giacomello A, Abraham MR, Marbán E. (2007). Regenerative potential of cardiosphere-derived cells expanded from percutaneous endomyocardial biopsy specimens. Circulation 115, 896–908 [DOI] [PubMed] [Google Scholar]
- Snider P, Olaopa M, Firulli AB, Conway SJ. (2007). Cardiovascular development and the colonizing cardiac neural crest lineage. ScientificWorldJournal 7, 1090–1113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stadtfeld M, Nagaya M, Utikal J, Weir G, Hochedlinger K. (2008). Induced pluripotent stem cells generated without viral integration. Science 322, 945–949 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stamm C, Kleine HD, Choi YH, Dunkelmann S, Lauffs JA, Lorenzen B, David A, Liebold A, Nienaber C, Zurakowski D, et al. (2007). Intramyocardial delivery of CD133+ bone marrow cells and coronary artery bypass grafting for chronic ischemic heart disease: safety and efficacy studies. J Thorac Cardiovasc Surg. 133, 717–725 [DOI] [PubMed] [Google Scholar]
- Stoller JZ, Epstein JA. (2005). Cardiac neural crest. Semin Cell Dev Biol. 16, 704–715 [DOI] [PubMed] [Google Scholar]
- Stottmann RW, Choi M, Mishina Y, Meyers EN, Klingensmith J. (2004). BMP receptor IA is required in mammalian neural crest cells for development of the cardiac outflow tract and ventricular myocardium. Development 131, 2205–2218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauer BE, Brehm M, Zeus T, Köstering M, Hernandez A, Sorg RV, Kögler G, Wernet P. (2002). Repair of infarcted myocardium by autologous intracoronary mononuclear bone marrow cell transplantation in humans. Circulation 106, 1913–1918 [DOI] [PubMed] [Google Scholar]
- Strauer BE, Brehm M, Zeus T, Bartsch T, Schannwell C, Antke C, Sorg RV, Kögler G, Wernet P, Müller HW, et al. (2005). Regeneration of human infarcted heart muscle by intracoronary autologous bone marrow cell transplantation in chronic coronary artery disease: the IACT Study. J Am Coll Cardiol. 46, 1651–1658 [DOI] [PubMed] [Google Scholar]
- Sun L, Cui M, Wang Z, Feng X, Mao J, Chen P, Kangtao M, Chen F, Zhou C. (2007). Mesenchymal stem cells modified with angiopoietin-1 improve remodeling in a rat model of acute myocardial infarction. Biochem Biophys Res Commun. 357, 779–784 [DOI] [PubMed] [Google Scholar]
- Sun Y. (2009). Myocardial repair/remodelling following infarction: roles of local factors. Cardiovasc Res. 81, 482–490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Y, Liang X, Najafi N, Cass M, Lin L, Cai CL, Chen J, Evans SM. (2007). Islet 1 is expressed in distinct cardiovascular lineages, including pacemaker and coronary vascular cells. Dev Biol. 304, 286–296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki K, Murtuza B, Beauchamp JR, Smolenski RT, Varela-Carver A, Fukushima S, Coppen SR, Partridge TA, Yacoub MH. (2004). Dynamics and mediators of acute graft attrition after myoblast transplantation to the heart. FASEB J. 10, 1153–1155 [DOI] [PubMed] [Google Scholar]
- Swijnenburg RJ, Tanaka M, Vogel H, Baker J, Kofidis T, Gunawan F, Lebl DR, Caffarelli AD, de Bruin JL, Fedoseyeva EV, et al. (2005. Embryonic stem cell immunogenicity increases upon differentiation after transplantation into ischemic myocardium. Circulation 112 Suppl. 9, I166–I172 [DOI] [PubMed] [Google Scholar]
- Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. (2007). Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 [DOI] [PubMed] [Google Scholar]
- Takehara N, Tsutsumi Y, Tateishi K, Ogata T, Tanaka H, Ueyama T, Takahashi T, Takamatsu T, Fukushima M, Komeda M, et al. (2008). Controlled delivery of basic fibroblast growth factor promotes human cardiosphere-derived cell engraftment to enhance cardiac repair for chronic myocardial infarction. J Am Coll Cardiol. 52, 1858–1865 [DOI] [PubMed] [Google Scholar]
- Tatsumi T, Ashihara E, Yasui T, Matsunaga S, Kido A, Sasada Y, Nishikawa S, Hadase M, Koide M, Nakamura R, et al. (2007). Intracoronary transplantation of non-expanded peripheral blood-derived mononuclear cells promotes improvement of cardiac function in patients with acute myocardial infarction. Circ J. 71, 1199–1207 [DOI] [PubMed] [Google Scholar]
- Taylor DA, Atkins BZ, Hungspreugs P, Jones TR, Reedy MC, Hutcheson KA, Glower DD, Kraus WE. (1998). Regenerating functional myocardium: improved performance after skeletal myoblast transplantation. Nat Med. 4, 929–933 [DOI] [PubMed] [Google Scholar]
- Teng CJ, Luo J, Chiu RC, Shum-Tim D. (2006). Massive mechanical loss of microspheres with direct intramyocardial injection in the beating heart: implications for cellular cardiomyoplasty. J Thorac Cardiovasc Surg. 132, 628–632 [DOI] [PubMed] [Google Scholar]
- Thiery JP, Sleeman JP. (2006). Complex networks orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell Biol. 7, 131–142 [DOI] [PubMed] [Google Scholar]
- Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. (1998). Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 [DOI] [PubMed] [Google Scholar]
- Toma C, Pittenger MF, Cahill KS, Byrne BJ, Kessler PD. (2002). Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation 105, 93–98 [DOI] [PubMed] [Google Scholar]
- Tomita S, Li RK, Weisel RD, Mickle DA, Kim EJ, Sakai T, Jia ZQ. (1999. Autologous transplantation of bone marrow cells improves damaged heart function. Circulation 100 Suppl. 19, II247–II256 [DOI] [PubMed] [Google Scholar]
- Tomita Y, Matsumura K, Wakamatsu Y, Matsuzaki Y, Shibuya I, Kawaguchi H, Ieda M, Kanakubo S, Shimazaki T, Ogawa S, et al. (2005). Cardiac neural crest cells contribute to the dormant multipotent stem cell in the mammalian heart. J Cell Biol. 170, 1135–1146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uccelli A, Moretta L, Pistoia V. (2008). Mesenchymal stem cells in health and disease. Nat Rev Immunol. 8, 726–736 [DOI] [PubMed] [Google Scholar]
- Uemura R, Xu M, Ahmad N, Ashraf M. (2006). Bone marrow stem cells prevent left ventricular remodeling of ischemic heart through paracrine signaling. Circ Res. 98, 1414–1421 [DOI] [PubMed] [Google Scholar]
- Ueno S, Weidinger G, Osugi T, Kohn AD, Golob JL, Pabon L, Reinecke H, Moon RT, Murry CE. (2007). Biphasic role for Wnt/beta-catenin signaling in cardiac specification in zebrafish and embryonic stem cells. Proc Natl Acad Sci USA 104, 9685–9690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vajkoczy P, Blum S, Lamparter M, Mailhammer R, Erber R, Engelhardt B, Vestweber D, Hatzopoulos AK. (2003). Multistep nature of microvascular recruitment of ex vivo expanded embryonic endothelial progenitor cells during tumor angiogenesis. J Exp Med. 197, 1755–1765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Berg G, Moorman AF. (2009). Concepts of cardiac development in retrospect. Pediatr Cardiol. 30, 580–587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Bos EJ, Thompson RB, Wagner A, Mahrholdt H, Morimoto Y, Thomson LE, Wang LH, Duncker DJ, Judd RM, Taylor DA. (2005). Functional assessment of myoblast transplantation for cardiac repair with magnetic resonance imaging. Eur J Heart Fail. 7, 435–443 [DOI] [PubMed] [Google Scholar]
- van Laake LW, Passier R, Doevendans PA, Mummery CL. (2008). Human embryonic stem cell-derived cardiomyocytes and cardiac repair in rodents. Circ Res. 102, 1008–1010 [DOI] [PubMed] [Google Scholar]
- van Tuyn J, Atsma DE, Winter EM, van der Velde-van Dijke I, Pijnappels DA, Bax NA, Knaän-Shanzer S, Gittenberger-de Groot AC, Poelmann RE, van der Laarse A, et al. (2007). Epicardial cells of human adults can undergo an epithelial-to-mesenchymal transition and obtain characteristics of smooth muscle cells in vitro. Stem Cells 25, 271–278 [DOI] [PubMed] [Google Scholar]
- Vanhoutte D, Schellings M, Pinto Y, Heymans S. (2006). Relevance of matrix metalloproteinases and their inhibitors after myocardial infarction: a temporal and spatial window. Cardiovasc Res. 69, 604–613 [DOI] [PubMed] [Google Scholar]
- Walter DH, Haendeler J, Reinhold J, Rochwalsky U, Seeger F, Honold J, Hoffmann J, Urbich C, Lehmann R, Arenzana-Seisdesdos F, et al. (2005). Impaired CXCR4 signaling contributes to the reduced neovascularization capacity of endothelial progenitor cells from patients with coronary artery disease. Circ Res. 97, 1142–1151 [DOI] [PubMed] [Google Scholar]
- Wang X, Hu Q, Nakamura Y, Lee J, Zhang G, From AH, Zhang J. (2006). The role of the sca-1+/CD31– cardiac progenitor cell population in postinfarction left ventricular remodeling. Stem Cells 24, 1779–1788 [DOI] [PubMed] [Google Scholar]
- Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, Hochedlinger K, Bernstein BE, Jaenisch R. (2007). In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 448, 318–324 [DOI] [PubMed] [Google Scholar]
- Wessels A, Pérez-Pomares JM. (2004). The epicardium and epicardially derived cells (EPDCs) as cardiac stem cells. Anat Rec A Discov Mol Cell Evol Biol. 276, 43–57 [DOI] [PubMed] [Google Scholar]
- Winter EM, Gittenberger-de Groot AC. (2007). Epicardium-derived cells in cardiogenesis and cardiac regeneration. Cell Mol Life Sci. 64, 692–703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winter EM, Grauss RW, Hogers B, van Tuyn J, van der Geest R, Lie-Venema H, Steijn RV, Maas S, DeRuiter MC, deVries AA, et al. (2007). Preservation of left ventricular function and attenuation of remodeling after transplantation of human epicardium-derived cells into the infarcted mouse heart. Circulation 116, 917–927 [DOI] [PubMed] [Google Scholar]
- Wollert KC, Meyer GP, Lotz J, Ringes-Lichtenberg S, Lippolt P, Breidenbach C, Fichtner S, Korte T, Hornig B, Messinger D, et al. (2004). Intracoronary autologous bone-marrow cell transfer after myocardial infarction: the BOOST randomised controlled clinical trial. Lancet 364, 141–148 [DOI] [PubMed] [Google Scholar]
- Wu SM, Fujiwara Y, Cibulsky SM, Clapham DE, Lien CL, Schultheiss TM, Orkin SH. (2006). Developmental origin of a bipotential myocardial and smooth muscle cell precursor in the mammalian heart. Cell 127, 1137–1150 [DOI] [PubMed] [Google Scholar]
- Wynn TA. (2008). Cellular and molecular mechanisms of fibrosis. J Pathol. 214, 199–210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu C, Police S, Rao N, Carpenter MK. (2002). Characterization and enrichment of cardiomyocytes derived from human embryonic stem cells. Circ Res. 91, 501–508 [DOI] [PubMed] [Google Scholar]
- Yamada S, Nelson TJ, Crespo-Diaz RJ, Perez-Terzic C, Liu XK, Miki T, Seino S, Behfar A, Terzic A. (2008). Embryonic stem cell therapy of heart failure in genetic cardiomyopathy. Stem Cells 26, 2644–2653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan P, Nagasawa A, Uosaki H, Sugimoto A, Yamamizu K, Teranishi M, Matsuda H, Matsuoka S, Ikeda T, Komeda M, et al. (2009). Cyclosporin-A potently induces highly cardiogenic progenitors from embryonic stem cells. Biochem Biophys Res Commun. 379, 115–120 [DOI] [PubMed] [Google Scholar]
- Yang JH, Wylie-Sears J, Bischoff J. (2008). Opposing actions of Notch1 and VEGF in post-natal cardiac valve endothelial cells. Biochem Biophys Res Commun. 374, 512–516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L, Cai CL, Lin L, Qyang Y, Chung C, Monteiro RM, Mummery CL, Fishman GI, Cogen A, Evans S. (2006). Isl1Cre reveals a common Bmp pathway in heart and limb development. Development 133, 1575–1585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L, Soonpaa MH, Adler ED, Roepke TK, Kattman SJ, Kennedy M, Henckaerts E, Bonham K, Abbott GW, Linden RM, et al. (2008). Human cardiovascular progenitor cells develop from a KDR+ embryonic-stem-cell-derived population. Nature 453, 524–528 [DOI] [PubMed] [Google Scholar]
- Yang W, Yan HX, Chen L, Liu Q, He YQ, Yu LX, Zhang SH, Huang DD, Tang L, Kong XN, et al. (2008). Wnt/beta-catenin signaling contributes to activation of normal and tumorigenic liver progenitor cells. Cancer Res. 68, 4287–4295 [DOI] [PubMed] [Google Scholar]
- Yeh ET, Zhang S, Wu HD, Körbling M, Willerson JT, Estrov Z. (2003). Transdifferentiation of human peripheral blood CD34-Enriched cell population into cardiomyocytes, endothelial cells, and smooth muscle cells in vivo. Circulation 108, 2070–2073 [DOI] [PubMed] [Google Scholar]
- Yellon DM, Hausenloy DJ. (2007). Myocardial reperfusion injury. N Engl J Med. 357, 1121–1135 [DOI] [PubMed] [Google Scholar]
- Yoon YS, Park JS, Tkebuchava T, Luedeman C, Losordo DW. (2004). Unexpected severe calcification after transplantation of bone marrow cells in acute myocardial infarction. Circulation 109, 3154–3157 [DOI] [PubMed] [Google Scholar]
- Young PP, Vaughan DE, Hatzopoulos AK. (2007). Biologic properties of endothelial progenitor cells and their potential for cell therapy. Prog Cardiovasc Dis. 49, 421–429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuasa S, Itabashi Y, Koshimizu U, Tanaka T, Sugimura K, Kinoshita M, Hattori F, Fukami S, Shimazaki T, Ogawa S, et al. (2005). Transient inhibition of BMP signaling by Noggin induces cardiomyocyte differentiation of mouse embryonic stem cells. Nat Biotechnol. 23, 607–611 [DOI] [PubMed] [Google Scholar]
- Zeisberg EM, Tarnavski O, Zeisberg M, Dorfman AL, McMullen JR, Gustafsson E, Chandraker A, Yuan X, Pu WT, Roberts AB, et al. (2007). Endothelial-to-mesenchymal transition contributes to cardiac fibrosis. Nat Med. 13, 952–961 [DOI] [PubMed] [Google Scholar]
- Zhang J, Wilson GF, Soerens AG, Koonce CH, Yu J, Palecek SP, Thomson JA, Kamp TJ. (2009). Functional cardiomyocytes derived from human induced pluripotent stem cells. Circ Res. 104, e30–e41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang M, Mal N, Kiedrowski M, Chacko M, Askari AT, Popovic ZB, Koc ON, Penn MS. (2007). SDF-1 expression by mesenchymal stem cells results in trophic support of cardiac myocytes after myocardial infarction. FASEB J. 21, 3197–3207 [DOI] [PubMed] [Google Scholar]
- Zhang Y, Goss AM, Cohen ED, Kadzik R, Lepore JJ, Muthukumaraswamy K, Yang J, DeMayo FJ, Whitsett JA, Parmacek MS, et al. (2008). A Gata6-Wnt pathway required for epithelial stem cell development and airway regeneration. Nat Genet. 40, 862–870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Z, Deb A, Zhang Z, Pachori A, He W, Guo J, Pratt R, Dzau VJ. (2009). Secreted frizzled related protein 2 protects cells from apoptosis by blocking the effect of canonical Wnt3a. J Mol Cell Cardiol. 46, 370–377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao TC, Tseng A, Yano N, Tseng Y, Davol PA, Lee RJ, Lum LG, Padbury JF. (2008). Targeting human CD34+ hematopoietic stem cells with anti-CD45 x anti-myosin light chain bispecific antibody preserves cardiac function in myocardial infarction. J Appl Physiol. 104, 1793–1800 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou B, Ma Q, Rajagopal S, Wu SM, Domian I, Rivera-Feliciano J, Jiang D, von Gise A, Ikeda S, Chien KR, et al. (2008). Epicardial progenitors contribute to the cardiomyocyte lineage in the developing heart. Nature 454, 109–113 [DOI] [PMC free article] [PubMed] [Google Scholar]