Abstract
Arsenic is a metalloid that generates various biological effects on cells and tissues. Depending on the specific tissue exposed and the time and degree of exposure, diverse responses can be observed. In humans, prolonged and/or high dose exposure to arsenic can have a variety of outcomes, including the development of malignancies, severe gastrointestinal toxicities, diabetes, cardiac arrhythmias, and death. On the other hand, one arsenic derivative, arsenic trioxide (As2O3), has important antitumor properties. This agent is a potent inducer of antileukemic responses, and it is now approved by the Food and Drug Administration for the treatment of acute promyelocytic leukemia in humans. The promise and therapeutic potential of arsenic and its various derivatives have been exploited for hundreds of years. Remarkably, research focused on the potential use of arsenic compounds in the treatment of human diseases remains highly promising, and it is an area of active investigation. An emerging approach of interest and therapeutic potential involves efforts to target and block cellular pathways activated in a negative feedback manner during treatment of cells with As2O3. Such an approach may ultimately provide the means to selectively enhance the suppressive effects of this agent on malignant cells and render normally resistant tumors sensitive to its antineoplastic properties.
Arsenic forms complexes with other elements, and it exists in inorganic and organic forms (1–3). The three major inorganic forms of arsenic are arsenic trisulfide (As2S3, yellow arsenic), arsenic disulfide (As2S2, red arsenic), and arsenic trioxide (As2O3, white arsenic) (1–3). There are two different oxidative states of arsenic that correlate with its cytotoxic potential, As(III) and As(V). Among them, As(III) is the most potent form and primarily accounts for its pro-apoptotic and inhibitory effects on target cells and tissues (3). The various forms of arsenic exist in nature primarily in a complex with pyrite (4, 5), although under certain circumstances, arsenic can dissociate from soil and enter natural waters (6), providing a contamination source for humans or animals who ingest such waters. In fact, most associations between long term exposure to arsenic and development of malignancies or other health disorders result from drinking contaminated water, especially in developing countries. Interestingly, pollution of the air with arsenic can also occur under certain circumstances, such as in the case of emissions from coal burning in China (7), providing an additional source of human exposure.
The metabolism of arsenic in humans includes reduction to the trivalent state and oxidative methylation to the pentavalent state (reviewed in Ref. 2). There is also reduction of arsenic acid to the arsenous form and subsequent methylation (2). The generation of inorganic or organic trivalent arsenic forms has important implications with regard to the toxicity of this agent, as such compounds are more toxic to the cells and exhibit more carcinogenic properties (2, 3). Thus, many of the consequences of exposure to arsenic as discussed below are the result of the activities and toxicities of the various metabolic products of arsenic compounds. It should be also noted that arsenic has the ability to bind to reduced thiols, including sulfhydryl groups in some proteins (2). Depending on the cellular context, such protein targeting may explain some of its cellular effects and generation of its toxicities and/or therapeutic effects.
Biological Consequences of Chronic Arsenic Exposure in Humans
Chronic exposure to arsenic produces substantial toxicities and leads to serious and frequently fatal syndromes and disorders. There is evidence that prenatal exposure results in serious short and long term toxicities (reviewed in Ref. 8). Both inorganic arsenic and its methylated metabolites can cross the placenta, and exposure during pregnancy can result in impaired fetal growth or even fetal loss (8). Such exposure can also result in increased post-birth infant mortality, and there is evidence for serious late effects of early exposure to arsenic, including the development of certain malignancies (8).
Beyond the strong association between exposure to arsenic in early life and development of illnesses, there is extensive evidence linking exposure at later stages of life and development of many different syndromes and diseases. Arsenic is a potent carcinogen, and there is a lot of evidence linking arsenic exposure to various types of solid tumors, including lung, prostate, bladder, renal, and skin cancers, as well as other malignancies (9–15). Notably, there are also studies that have shown that, in some parts of the world (Denmark), exposure to low levels of arsenic is not associated with development of malignancies, and on the contrary, it may decrease the incidence of non-melanoma skin cancer (16). Thus, geographical location, genetic factors, and levels of exposure may play important roles in arsenic-associated carcinogenesis.
In addition to the development of malignancies, long term arsenic exposure has been associated with other chronic illnesses and ailments, such as diabetes, hypertension, cardiovascular disease, vascular changes, and neuropathy (17, 18). Moreover, chronic exposure to arsenic can affect long term memory and modify hormonal regulation (18). Thus, a variety of different chronic illnesses and malignancies can be induced by arsenic exposure, underscoring the diversity of its cellular targets and its ability to deregulate important and diverse cell functions.
Antitumor Properties of Arsenic Compounds
Although exposure to arsenic is well documented to lead to the development of tumors in humans, there is also extensive evidence indicating that one form of arsenic, As2O3, exhibits potent antitumor effects in vitro and in vivo (19–21). As2O3 has been approved by the Food and Drug Administration in the United States for the treatment of patients with APL2 that is refractory to ATRA. It is now well established that As2O3 induces complete remissions in ∼80–90% of newly diagnosed patients with APL, as well as in 60–90% of ATRA-refractory patients (19–21). Combinations of As2O3 with ATRA for the treatment of this leukemia are more potent than each agent alone (22), underscoring the differences in the mechanisms of action between the two agents. Although As2O3 is very effective in the treatment of APL, there are associated toxicities with its use, and appropriate precautions should be in place during its administration. Such toxicities include leukocytosis, the ATRA differentiation syndrome, and cardiac arrhythmias associated with prolonged QT interval (20, 21).
As As2O3 has potent effects in vitro against different types of malignant cells beyond APL, there has been substantial interest in its potential clinical development for the treatment of other hematologic malignancies, including MDS and multiple myeloma (23, 24). In addition, As2O3 has potent antileukemic properties against other non-APL subtypes of AML in vitro, and recent studies have suggested important clinical activity when administered to elderly patients with AML in combination with low dose cytarabine (25). There is also an interest in the clinical development of As2O3 for the treatment of various solid tumors, but such potential applications are restricted by the requirement of high toxic doses to induce apoptosis in such cells. It is possible that the spectrum of malignant diseases for which As2O3 is used will widen as new approaches aimed at enhancing the sensitivity of malignant cells to its effects emerge.
Mechanisms of Arsenic-induced Cell Death
As As2O3 has potent cytotoxic and antitumor activities in vitro and in vivo, there has been extensive research focused on the identification of the mechanisms by which it generates its effects on target cells. An important initial cellular event that occurs during treatment of target cells with As2O3 involves elevation of ROS (reviewed in Refs. 1, 2, 26, and 27). Such generation of ROS appears to be regulated, at least in part, by activation of NADPH oxidase and NO synthase isozymes (32). Also, arsenic-containing compounds are potent modulators of the thioredoxin system that includes thioredoxin, thioredoxin reductase, and NADPH (reviewed in Refs. 26 and 27). The thioredoxin system controls, to a large extent, intracellular redox reactions, regulates apoptosis, and protects cells from stress damage (26, 27), and the ability of arsenic-containing compounds to target and block thioredoxin reductase may be important in the induction of its pro-apoptotic effects (26).
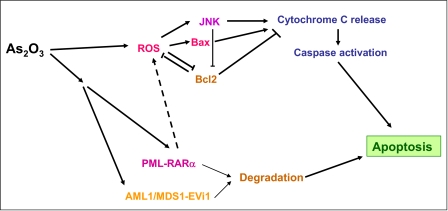
Overproduction of ROS is linked to the induction of apoptosis by As2O3. Accumulation of hydrogen peroxide (H2O2) leads to decreases in the mitochondrial membrane potential, resulting in cytochrome c release and activation of the caspase cascade (27). This appears to be a common mechanism of induction of cell death in diverse cellular backgrounds. There is extensive evidence implicating arsenic-dependent, ROS-mediated activation of caspases in various types of malignant cells. These include cells of APL origin (26), human T cell lymphotrophic virus I-infected T cell lines and primary adult T cell leukemia cells (28), multiple myeloma cells (24), and different types of solid tumor cells (30, 31). However, caspase-independent death pathways have been also reported to be activated by arsenic in myeloma cells and may mediate pro-apoptotic signals (32). Other recent work has implicated the JNK kinase as an essential component of As2O3-dependent apoptosis (Fig. 1) (33). It was demonstrated that activation of JNK occurs in an As2O3-inducible manner in cells of APL origin and that As2O3 resistance correlates with defective activation of the JNK pathway (33). Notably, in these studies, it was also shown that pharmacological inhibition of JNK significantly decreases As2O3-dependent growth inhibition and apoptosis, but it does not protect cells from the effects of chemotherapy (doxorubicin) (33).
FIGURE 1.
Key cellular events associated with induction of arsenic-dependent apoptosis.
As2O3 also activates the pro-apoptotic Bcl-2 family member Bax and induces its translocation from the cytosol to the mitochondria (34). Such Bax engagement plays an important role in apoptosis, whereas its activation is suppressed by the anti-apoptotic protein Bcl-2 via inhibition of mitochondrial ROS generation (34). As2O3 also up-regulates expression of other pro-apoptotic proteins, including the BH3 (Bcl-2 homology domain 3)-only proteins Noxa, Bmf, and Bim (35). On the other hand, it down-regulates Bcl-2 (36, 37) and other anti-apoptotic proteins, including Bcl-1, Bcl-xL, and Mcl-1 (36, 38). An additional mechanism that appears to participate in the induction of apoptosis and/or antitumor effects is down-regulation of NF-κB activity (39), an event that appears to reflect inhibition of the IκB kinase (39). Interestingly, the generation of the effects of arsenite on melanoma cells and the induction of an apoptotic response have been shown to correlate with low nuclear NF-κB activity and endogenous expression of TNFα (40).
There is also evidence that arsenic inhibits the JAK-STAT pathway. Previous studies demonstrated that sodium arsenite inhibits interleukin-6-dependent tyrosine phosphorylation of STAT3 in HepG2 cells via direct suppression of the tyrosine kinase JAK1 (41). Other studies have shown that As2O3 suppresses activation of STAT1, STAT3, and STAT5 in AML cells and inhibits activation of JAK1 and JAK2, which phosphorylate STATs (42), whereas there is also evidence for a synergism between As2O3 and Hsp90 inhibitors in STAT3 activity (43).
It should be also noted that there is evidence that As2O3 enhances the sensitivity of cancer cells to death receptor-induced apoptosis. It was recently shown that As2O3 sensitizes human glioma cells to TRAIL (TNF-related apoptosis-inducing ligand)-induced apoptosis via DR5 up-regulation (44), whereas other studies had shown that As2O3 sensitizes promyelocytic leukemia cell lines to TNFα-induced apoptosis (45).
Cell Type-specific Targets of As2O3
Beyond regulation of common cellular pathways in different types of tumors cells, arsenic compounds frequently target elements and oncogenes selectively expressed in certain malignancies. Previous work has demonstrated that As2O3 induces degradation of the PML-RARα fusion protein in APL cells (27). Interestingly, very recent studies have directly implicated PML-RARα as a mediator of the sensitivity of APL cells to As2O3 via its ability to impair cAMP signaling, resulting in NADPH oxidase activation and enhanced ROS generation (46). These studies strongly suggest that the elevated basal ROS levels in arsenic-sensitive cells result from PML-RARα expression (46) and suggest a mechanism for the unusual sensitivity of APL cells to As2O3. Another recently identified malignancy-specific target of As2O3 is the AML1/MDS1/EVI1 oncoprotein, a product of a fusion gene resulting from the t(3;21)(q26;q22) translocation, which is found in some patients with MDS, AML, or the blast phase of CML (47). Cells expressing AML1/MDS1/EVI1 are sensitive to As2O3 and degraded at therapeutic concentrations, raising the possibility that As2O3 may be effective in the treatment of hematologic malignancies expressing this oncoprotein (47).
Other studies have shown that expression of the BCR-ABL oncoprotein confers sensitivity to As2O3 (48), whereas a recent study demonstrated that the combination of imatinib mesylate and arsenic sulfide exerts more potent antileukemic effects in a BCR-ABL-positive mouse model of CML than each agent alone (49). In that study, evidence was also provided that arsenic targets BCR-ABL via ubiquitination of key lysine residues, leading to its proteasomal degradation (49). Interestingly, As2O3-inducible suppression of PML expression may prove to be of therapeutic relevance in CML, as a recent study demonstrated that PML targeting eradicates BCR-ABL-expressing leukemia-initiating stem cells (50). Moreover, follow-up work subsequently demonstrated that retinoic acid and As2O3 eradicate in a similar manner leukemia-initiating cells in APL via PML-RARα degradation (51). Interestingly, although PML-RARα degradation is not essential for leukemic cell differentiation, it is required for leukemic precursor clearance (51). It remains to be seen whether similar populations of malignant stem cells can be eradicated in other tumors via PML targeting and whether combined use of As2O3 with other agents would be of value for that purpose.
Cellular Pathways and Systems That Negatively Regulate As2O3 Responses
As2O3 has major activity in the treatment of APL, as it can induce differentiation and cell death of APL cells at low concentrations. However, to induce apoptosis of other types of cancer cells, higher toxic concentrations are required. That has been a major limiting factor for its use in other malignancies, as higher doses are associated with dose-limiting toxicities. Such issues have triggered studies aimed at identifying cellular protective mechanisms that limit the induction of arsenic-dependent apoptosis in malignant cells and at designing approaches to target them to overcome resistance.
As generation of ROS and, in particular, H2O2 is of high relevance in the induction of arsenic-mediated cell death, approaches to enhance such ROS production have been explored. GSH in cells has the ability to conjugate arsenic in the form of As(GS)3 complexes or to sequester the ROS induced by arsenic (27), and sensitivity to arsenic correlates with reduced glutathione levels in cells (52). Moreover, depletion of intracellular GSH stores by treatment of cells with ascorbic acid (53) or buthionine sulfoximine (54) has been shown to promote the antitumor effects of As2O3 in vitro. On the other hand, when malignant cells are pretreated with N-acetylcysteine, cellular GSH levels are increased, and the effects of As2O3 are reversed (53). Thus, targeting cellular glutathione stores may be an effective way to enhance the antitumor effects of arsenic trioxide.
There has been also evidence that activation of the p38 MAPK during treatment of malignant cells with As2O3 negatively controls generation of As2O3 responses in APL cells and other malignant hematopoietic and solid tumor cells. It was originally shown that p38 MAPK is activated during As2O3 treatment of cell lines of diverse origin (55). Paradoxically, pharmacological or molecular inhibition of p38 was found to enhance As2O3-dependent leukemic cell differentiation of APL cells and/or generation of growth inhibitory and pro-apoptotic responses (55). These initial studies suggested that the p38 pathway acts as a negative feedback regulator system to control induction of As2O3 responses in malignant cells. Subsequent work led to the identification of Mkk3 and Mkk6 as upstream kinases that regulate arsenic-dependent engagement of p38 and established that different pharmacological inhibitors of p38 enhance the suppressive effects of As2O3 on leukemic granulocyte/macrophage colony-forming unit progenitors from CML patients (56). Other work also demonstrated that pharmacological targeting of p38 promotes As2O3-dependent apoptosis in multiple myeloma cells (57), whereas here has been evidence that pharmacological inhibition of the MEK (mitogen-activated protein kinase/extracellular signal-regulated kinase kinase)-ERK pathway also enhances arsenic-induced malignant cell death (58).
Altogether, it appears that both the p38 and ERK pathways negatively control generation of antitumor responses by As2O3. Efforts to identify specific downstream MAPK-induced signals that suppress As2O3 responses are therefore of interest and may lead to the ultimate development of specific less toxic drug inhibitors that could be used in combination with As2O3. MSK1 (mitogen- and stress-activated kinase 1) is a common effector kinase for the p38 and ERK MAPK pathways and mediates immediate-early gene expression in response to stress. This kinase is activated in response to As2O3 treatment of leukemic cells (59), whereas its pharmacological inhibition enhances antileukemic responses (59). Two other downstream effectors of the p38 MAPK, the kinases Mnk1 and Mnk2, are also activated in an As2O3-inducible manner and regulate phosphorylation of eukaryotic initiation factor 4E at Ser-209 (60). The engagement of these p38 effectors occurs in a negative feedback regulatory manner during treatment of cells with As2O3, as their pharmacological or molecular targeting results in enhanced As2O3-dependent suppressive responses in leukemic lines and primary leukemic progenitors from AML patients (60).
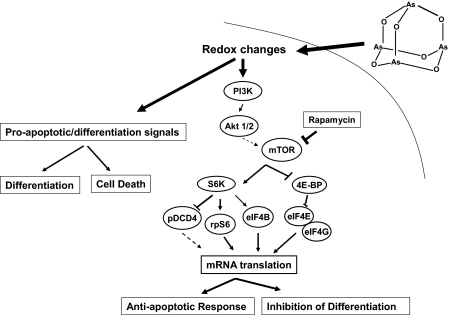
Finally, the Akt-mTOR signaling cascade is also activated during treatment of different types of leukemic cells with As2O3 (Fig. 2) (61, 62). mTOR-generated signals play critical and essential roles in the control of mRNA translation in mammalian cells and mediate important biological responses (29, 63). Recent work demonstrated that Akt, mTOR, and downstream effectors are activated in As2O3-treated leukemic cells (61, 62). Combination studies using knock-out cells for different elements of the mTOR pathway or small interfering RNA-mediated knockdown of mTOR effectors in leukemic progenitors have strongly suggested that targeting this signaling cascade may provide a novel approach to potentiate the effects of arsenic (62). It should be also noted that activation of Akt during treatment of acute leukemia cells with arsenic trioxide suggests that, beyond mTOR, various other anti-apoptotic pathways are activated in a negative feedback regulatory manner, as it is well established that various anti-apoptotic signals are generated during Akt activation (29).
FIGURE 2.
Proposed model for As2O3-dependent activation of the mTOR pathway in a negative feedback regulatory manner in malignant cells. PI3K, phosphatidylinositol 3-kinase; S6K, S6 kinase; 4E-BP, eukaryotic initiation factor 4E-binding protein; rpS6, ribosomal protein S6; eIF, eukaryotic initiation factor.
Conclusions and Future Perspectives
The therapeutic uses and potential of arsenic-containing compounds have been evolving over centuries, starting with the empiric use of arsenic in ancient times up to the current Food and Drug Administration approval of As2O3 for the treatment of APL in humans. Despite the well known toxicities and side effects of arsenic compounds, the prospects for arsenic use in the treatment of human diseases remain high. The evolution of our understanding of how arsenic mediates biological responses over the last decade has led to new studies aimed at establishing conditions for the selective enhancement of its antitumor properties in vitro and in vivo. It is possible that the next phase in the medical use of arsenic compounds will involve selective applications to malignancies with distinct molecular profiles that define arsenic sensitivity and/or combinations with other agents that target cellular pathways that negatively control arsenic responses. The usefulness of such approaches remains to be established over the next several years. Independently of the outcome of such studies and on the basis of historical considerations and the ongoing evolution in the field, one can argue with a degree of certainty that arsenicals will continue to be the focus of intense research investigations in the near and distant future.
Supplementary Material
This is the fifth article of five in the second Thematic Minireview Series on Metals in Biology. This minireview will be reprinted in the 2009 Minireview Compendium, which will be available in January, 2010.
- APL
- acute promyelocytic leukemia
- ATRA
- all-trans-retinoic acid
- MDS
- myelodysplastic syndrome(s)
- AML
- acute myeloid leukemia
- ROS
- reactive oxygen species
- JNK
- c-Jun N-terminal kinase
- TNF
- tumor necrosis factor
- STAT
- signal transducer and activator of transcription
- PML
- promyelocytic leukemia
- RARα
- retinoic acid receptor α
- CML
- chronic myelogenous leukemia
- MAPK
- mitogen-activated protein kinase
- ERK
- extracellular signal-regulated kinase
- mTOR
- mammalian target of rapamycin.
REFERENCES
- 1.Litzow M. R. (2008) Expert Opin. Pharmacother. 9, 1773–1785 [DOI] [PubMed] [Google Scholar]
- 2.Ralph S. J. (2008) Metal-Based Drugs 2008, 1–13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dilda P. J., Hogg P. J. (2007) Cancer Treat. Rev. 33, 542–564 [DOI] [PubMed] [Google Scholar]
- 4.Benbrahim-Tallaa L., Waalkes M. P. (2008) Environ. Health Perspect. 116, 158–164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nordstrom D. K. (2002) Science 21, 2143–2145 [DOI] [PubMed] [Google Scholar]
- 6.Smedley P. L., Kinniburgh D. G. (2002) Appl. Geochem. 17, 517–568 [Google Scholar]
- 7.Millman A., Tang D., Perera F. P. (2008) Pediatrics 122, 620–628 [DOI] [PubMed] [Google Scholar]
- 8.Vahter M. (2008) Basic Clin. Pharmacol. Toxicol. 102, 204–211 [DOI] [PubMed] [Google Scholar]
- 9.Smith A. H., Hopenhayn-Rich C., Bates M. N., Goeden H. M., Hertz-Picciotto I., Duggan H. M., Wood R., Kosnett M. J., Smith M. T. (1992) Environ. Health Perspect. 97, 259–267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chiou H. Y., Hsueh Y. M., Liaw K. F., Horng S. F., Chiang M. H., Pu Y. S., Lin J. S., Huang C. H., Chen C. J. (1995) Cancer Res. 55, 1296–1300 [PubMed] [Google Scholar]
- 11.Tsuda T., Babazono A., Yamamoto E., Kurumatani N., Mino Y., Ogawa T., Kishi Y., Aoyama H. (1995) Am. J. Epidemiol. 141, 198–209 [DOI] [PubMed] [Google Scholar]
- 12.Celik I., Gallicchio L., Boyd K., Lam T. K., Matanoski G., Tao X., Shiels M., Hammond E., Chen L., Robinson K. A., Caulfield L. E., Herman J. G., Guallar E., Alberg A. J. (2008) Environ. Res. 108, 48–55 [DOI] [PubMed] [Google Scholar]
- 13.Yang C. Y., Chang C. C., Chiu H. F. (2008) J. Toxicol. Environ. Health Part A 71, 1559–1563 [DOI] [PubMed] [Google Scholar]
- 14.Radosavljević V., Jakovljević B. (2008) J. Environ. Health 71, 40–42 [PubMed] [Google Scholar]
- 15.Liu J., Waalkes M. P. (2008) Toxicol. Sci. 105, 24–32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Baastrup R., Sørensen M., Balstrøm T., Frederiksen K., Larsen C. L., Tjønneland A., Overvad K., Raaschou-Nielsen O. (2008) Environ. Health Perspect. 116, 231–237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen C. J., Wang S. L., Chiou J. M., Tseng C. H., Chiou H. Y., Hsueh Y. M., Chen S. Y., Wu M. M., Lai M. S. (2007) Toxicol Appl. Pharmacol. 222, 298–304 [DOI] [PubMed] [Google Scholar]
- 18.Kapaj S., Peterson H., Liber K., Bhattacharya P. (2006) J. Environ. Sci. Health Part A Tox. Hazard. Subst. Environ. Eng. 41, 2399–2428 [DOI] [PubMed] [Google Scholar]
- 19.Douer D., Tallman M. S. (2005) J. Clin. Oncol. 23, 2396–2410 [DOI] [PubMed] [Google Scholar]
- 20.Tallman M. S., Nabhan C., Feusner J. H., Rowe J. M. (2002) Blood 99, 759–767 [DOI] [PubMed] [Google Scholar]
- 21.Sanz M. A., Grimwade D., Tallman M. S., Lowenberg B., Fenaux P., Estey E. H., Naoe T., Lengfelder E., Buchner T., Dohner H., Burnett A. K., Lo-Coco F. (2009) Blood 113, 1875–1891 [DOI] [PubMed] [Google Scholar]
- 22.Shen Z. X., Shi Z. Z., Fang J., Gu B. W., Li J. M., Zhu Y. M., Shi J. Y., Zheng P. Z., Yan H., Liu Y. F., Chen Y., Shen Y., Wu W., Tang W., Waxman S., de Thé H., Wang Z. Y., Chen S. J., Chen Z. (2004) Proc. Natl. Acad. Sci. U.S.A. 101, 5328–5335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Schiller G. J., Slack J., Hainsworth J. D., Mason J., Saleh M., Rizzieri D., Douer D., List A. F. (2006) J. Clin. Oncol. 24, 2456–2464 [DOI] [PubMed] [Google Scholar]
- 24.Lunghi P., Giuliani N., Mazzera L., Lombardi G., Ricca M., Corradi A., Cantoni A. M., Salvatore L., Riccioni R., Costanzo A., Testa U., Levrero M., Rizzoli V., Bonati A. (2008) Blood 112, 2450–2462 [DOI] [PubMed] [Google Scholar]
- 25.Roboz G. J., Ritchie E. K., Curcio T., Provenzano J., Carlin R., Samuel M., Wittenberg B., Mazumdar M., Christos P. J., Mathew S., Allen-Bard S., Feldman E. J. (2008) Cancer 113, 2504–2511 [DOI] [PubMed] [Google Scholar]
- 26.Kumagai Y., Sumi D. (2007) Annu. Rev. Pharmacol. Toxicol. 47, 243–262 [DOI] [PubMed] [Google Scholar]
- 27.Miller W. H., Jr., Schipper H. M., Lee J. S., Singer J., Waxman S. (2002) Cancer Res. 62, 3893–3903 [PubMed] [Google Scholar]
- 28.Ishitsuka K., Ikeda R., Utsunomiya A., Uozumi K., Hanada S., Suzuki S., Takeuchi S., Takatsuka Y., Takeshita T., Ohno N., Arima T. (2002) Leuk. Lymphoma 43, 1107–1114 [DOI] [PubMed] [Google Scholar]
- 29.Bhaskar P. T., Hay N. (2007) Dev. Cell 12, 487–502 [DOI] [PubMed] [Google Scholar]
- 30.Li X., Ding X., Adrian T. E. (2004) Cancer Investig. 22, 389–400 [DOI] [PubMed] [Google Scholar]
- 31.Hyun Park W., Hee Cho Y., Won Jung C., Oh Park J., Kim K., Hyuck Im Y., Lee M. H., Ki Kang W., Park K. (2003) Biochem. Biophys. Res. Commun. 300, 230–235 [DOI] [PubMed] [Google Scholar]
- 32.McCafferty-Grad J., Bahlis N. J., Krett N., Aguilar T. M., Reis I., Lee K. P., Boise L. H. (2003) Mol. Cancer Ther. 2, 1155–1164 [PubMed] [Google Scholar]
- 33.Davison K., Mann K. K., Waxman S., Miller W. H., Jr. (2004) Blood 103, 3496–3502 [DOI] [PubMed] [Google Scholar]
- 34.Zheng Y., Yamaguchi H., Tian C., Lee M. W., Tang H., Wang H. G., Chen Q. (2005) Oncogene 24, 3339–3347 [DOI] [PubMed] [Google Scholar]
- 35.Morales A. A., Gutman D., Lee K. P., Boise L. H. (2008) Blood 111, 5152–5162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chen G. Q., Zhu J., Shi X. G., Ni J. H., Zhong H. J., Si G. Y., Jin X. L., Tang W., Li X. S., Xong S. M., Shen Z. X., Sun G. L., Ma J., Zhang P., Zhang T. D., Gazin C., Naoe T., Chen S. J., Wang Z. Y., Chen Z. (1996) Blood 88, 1052–1061 [PubMed] [Google Scholar]
- 37.Akao Y., Mizoguchi H., Kojima S., Naoe T., Ohishi N., Yagi K. (1998) Br. J. Haematol. 102, 1055–1060 [DOI] [PubMed] [Google Scholar]
- 38.Tun-Kyi A., Qin J. Z., Oberholzer P. A., Navarini A. A., Hassel J. C., Dummer R., Döbbeling U. (2008) Ann. Oncol. 19, 1488–1494 [DOI] [PubMed] [Google Scholar]
- 39.Kapahi P., Takahashi T., Natoli G., Adams S. R., Chen Y., Tsien R. Y., Karin M. J. (2000) J. Biol. Chem. 275, 36062–36066 [DOI] [PubMed] [Google Scholar]
- 40.Ivanov V. N., Hei T. K. (2004) J. Biol. Chem. 279, 22747–22758 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cheng H. Y., Li P., David M., Smithgall T. E., Feng L., Lieberman M. W. (2004) Oncogene 23, 3603–3612 [DOI] [PubMed] [Google Scholar]
- 42.Wetzler M., Brady M. T., Tracy E., Li Z. R., Donohue K. A., O'Loughlin K. L., Cheng Y., Mortazavi A., McDonald A. A., Kunapuli P., Wallace P. K., Baer M. R., Cowell J. K., Baumann H. (2006) Clin. Cancer Res. 12, 6817–6825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wetzler M., Earp J. C., Brady M. T., Keng M. K., Jusko W. J. (2007) Clin. Cancer Res. 13, 2261–2270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kim E. H., Yoon M. J., Kim S. U., Kwon T. K., Sohn S., Choi K. S. (2008) Cancer Res. 68, 266–275 [DOI] [PubMed] [Google Scholar]
- 45.Amrán D., Sánchez Y., Fernández C., Ramos A. M., de Blas E., Bréard J., Calle C., Aller P. (2007) Biochim. Biophys. Acta 1773, 1653–1663 [DOI] [PubMed] [Google Scholar]
- 46.Li L., Wang J., Ye R. D., Shi G., Jin H., Tang X., Yi J. (2008) J. Cell. Physiol. 217, 486–493 [DOI] [PubMed] [Google Scholar]
- 47.Shackelford D., Kenific C., Blusztajn A., Waxman S., Ren R. (2006) Cancer Res. 66, 11360–11369 [DOI] [PubMed] [Google Scholar]
- 48.Puccetti E., Güller S., Orleth A., Brüggenolte N., Hoelzer D., Ottmann O. G., Ruthardt M. (2000) Cancer Res. 60, 3409–3413 [PubMed] [Google Scholar]
- 49.Zhang Q. Y., Mao J. H., Liu P., Huang Q. H., Lu J., Xie Y. Y., Weng L., Zhang Y., Chen Q., Chen S. J., Chen Z. (2009) Proc. Natl. Acad. Sci. U.S.A. 106, 3378–3383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ito K., Bernardi R., Morotti A., Matsuoka S., Saglio G., Ikeda Y., Rosenblatt J., Avigan D. E., Teruya-Feldstein J., Pandolfi P. P. (2008) Nature 453, 1072–1078 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nasr R., Guillemin M. C., Ferhi O., Soilihi H., Peres L., Berthier C., Rousselot P., Robledo-Sarmiento M., Lallemand-Breitenbach V., Gourmel B., Vitoux D., Pandolfi P. P., Rochette-Egly C., Zhu J., de Thé H. (2008) Nat. Med. 14, 1333–1342 [DOI] [PubMed] [Google Scholar]
- 52.Yang C. H., Kuo M. L., Chen J. C., Chen Y. C. (1999) Br. J. Cancer 81, 796–799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Grad J. M., Bahlis N. J., Reis I., Oshiro M. M., Dalton W. S., Boise L. H. (2001) Blood 98, 805–813 [DOI] [PubMed] [Google Scholar]
- 54.Gartenhaus R. B., Prachand S. N., Paniaqua M., Li Y., Gordon L. I. (2002) Clin. Cancer Res. 8, 566–572 [PubMed] [Google Scholar]
- 55.Verma A., Mohindru M., Deb D. K., Sassano A., Kambhampati S., Ravandi F., Minucci S., Kalvakolanu D. V., Platanias L. C. (2002) J. Biol. Chem. 277, 44988–44995 [DOI] [PubMed] [Google Scholar]
- 56.Giafis N., Katsoulidis E., Sassano A., Tallman M. S., Higgins L. S., Nebreda A. R., Davis R. J., Platanias L. C. (2006) Cancer Res. 66, 6763–6771 [DOI] [PubMed] [Google Scholar]
- 57.Wen J., Cheng H. Y., Feng Y., Rice L., Liu S., Mo A., Huang J., Zu Y., Ballon D. J., Chang C. C. (2008) Br. J. Haematol. 140, 169–180 [DOI] [PubMed] [Google Scholar]
- 58.Lunghi P., Costanzo A., Salvatore L., Noguera N., Mazzera L., Tabilio A., Lo-Coco F., Levrero M., Bonati A. (2006) Blood 107, 4549–4553 [DOI] [PubMed] [Google Scholar]
- 59.Kannan-Thulasiraman P., Katsoulidis E., Tallman M. S., Arthur J. S., Platanias L. C. (2006) J. Biol. Chem. 281, 22446–22452 [DOI] [PubMed] [Google Scholar]
- 60.Dolniak B., Katsoulidis E., Carayol N., Altman J. K., Redig A. J., Tallman M. S., Ueda T., Watanabe-Fukunaga R., Fukunaga R., Platanias L. C. (2008) J. Biol. Chem. 283, 12034–12042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yoon P., Giafis N., Smith J., Mears H., Katsoulidis E., Sassano A., Altman J., Redig A. J., Tallman M. S., Platanias L. C. (2006) Mol. Cancer Ther. 5, 2815–2823 [DOI] [PubMed] [Google Scholar]
- 62.Altman J. K., Yoon P., Katsoulidis E., Kroczynska B., Sassano A., Redig A. J., Glaser H., Jordan A., Tallman M. S., Hay N., Platanias L. C. (2008) J. Biol. Chem. 283, 1992–2001 [DOI] [PubMed] [Google Scholar]
- 63.Bjornsti M. A., Houghton P. J. (2004) Nat. Rev. Cancer 4, 335–348 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.