Abstract
Background and Aims
Optimal partitioning theory (OPT) predicts plants will allocate biomass to organs where resources are limiting. Studies of OPT focus on root, stem and leaf mass ratios where roots and stems are often further sub-divided into organs such as fine roots/tap roots or branches/main stem. Leaves, however, are rarely sub-divided into different organs. Heteroblastic species develop juvenile and adult foliage and provide an opportunity of sub-dividing leaf mass ratio into distinct organs. Acacia implexa (Mimosaceae) is a heteroblastic species that develops compound (juvenile), transitional and phyllode (adult) leaves that differ dramatically in form and function. The aims of the present study were to grow A. implexa to examine patterns of plastic development of whole-plant and leaf traits under the OPT framework.
Methods
Plants were grown in a glasshouse under contrasting nutrient, light and water environments in a full factorial design. Allocation to whole-plant and leaf-level traits was measured and analysed with multivariate statistics.
Key Results
Whole-plant traits strongly followed patterns predicted by OPT. Leaf-level traits showed a more complex pattern in response to experimental treatments. Compound leaves on low nutrient plants had significantly lower specific leaf area (SLA) and were retained for longer as quantified by a significantly greater compound leaf mass ratio after 120 d. There was no significant difference in SLA of compound leaves in the light treatment, yet transitional SLA was significantly higher under the low light treatment. The timing of heteroblastic shift from compound to transitional leaves was significantly delayed only in the low light treatment. Therefore, plants in the light treatment responded at the whole-plant level by adjusting allocation to productive compound leaves and at the leaf-level by adjusting SLA. There were no significant SLA differences in the water treatment despite strong trends at the whole-plant level.
Conclusion
Explicitly sub-dividing leaves into different types provided greater insights into OPT.
Key words: Acacia implexa, heteroblastic development, heterophylly, optimal partitioning theory, plasticity, leaf form and function, nutrients, light, water
INTRODUCTION
Optimal partitioning theory (OPT) predicts that plants allocate biomass to above- or below-ground organs to maximize acquisition of the most limiting resources (Thornley, 1972; Bloom et al., 1985) and provides a framework for the study of plasticity in plants. A simple model of a basic plant divides the body into two parts: that above ground and that below ground. The above-ground plant primarily captures light and carbon resources, whereas the below-ground plant captures water and mineral resources necessary for nutrition. When light and/or carbon levels decrease, plants have a relatively high allocation to above-ground biomass. Conversely, when water and/or mineral resources decrease, relatively more biomass is allocated below ground. This is clearly a basic model of how a plant functions, yet it provides a powerful framework for further studies on plant biomass allocation (Tilman, 1988). For example, OPT has provided a context for plant competition for limiting resources (Tilman, 1982; Wedin and Tilman, 1993), community succession (Gleeson and Tilman, 1990, 1994), community diversity (Tilman, 1988; Tilman and Pacala, 1993; Chesson, 2000) and more context-specific examples such as resource allocation in resprouting and seeder species (Bellingham and Sparrow, 2000).
Experiments and studies of OPT often rely on broad categorization of traits into units such as root to shoot ratio (R : S; e.g. Li et al., 1999; Andrews et al., 2001; Zhang et al., 2005; McCarthy and Enquist, 2007). The R : S ratio is relatively simple to measure, and changes in it are straightforward to interpret. However, Poorter and Nagel (2000) argued that better insights into OPT can be gained by sub-dividing R : S into more specific traits. Dividing R : S into root mass ratio, stem mass ratio and leaf mass ratio (or fractions, rather than ratios) provides greater functional insights – particularly into growth analyses and the carbon economy (Poorter and Nagel, 2000). Root, stem and leaf ratios are the bare minimum compartments that should be examined (see Poorter and Nagel, 2000), yet more predictions of OPT could be tested with further sub-divisions of these organs. Previous studies have sub-divided the root mass ratio into fine roots and tap roots (e.g. Wullschleger et al., 2005), and stem mass ratio has been sub-divided into main stem and petioles (e.g. McConnaughay and Bazzaz, 1995). Leaves, on the other hand, are rarely sub-divided since most plants tend to have only a single type of leaf organ or photosynthetic blade.
Leaves are complex organs and are highly variable across species and, sometimes, within species. Different types of leaves have been shown to play various biological and ecological roles (e.g. Boardman, 1977). Therefore, the presence of two or more leaf types undertaking different roles allows for a sub-division of leaf mass ratio into different organs. For instance, rosette and cauline (stem) leaves have been demonstrated to be favoured in contrasting light environments in Arabidopsis thaliana (e.g. Bonser and Geber, 2005) and could be sub-divided into components of leaf mass ratio. Sun leaves and shade leaves on the same plant are often significantly different (Boardman, 1977) and are another example of two different leaf types that can be easily sub-divided within leaf mass ratio.
There are cases of dramatic and sudden changes in leaf form and function that occur in many plant lineages. Heterophylly, where an individual plant exhibits two or more contrasting leaf types (Winn, 1999), defines such plants. Heterophylly is particularly prominent where strong and conflicting selection pressures favour the expression of different leaf types on the same plant. Well known cases of heterophylly involve amphibious or submerged plants where leaf type is plastic according to whether the stem is in an aqueous or terrestrial environment (Sculthorpe, 1967; Bruni et al., 1996; Wells and Pigliucci, 2000; Mommer and Visser, 2005; Mommer et al., 2006). A similar concept is heteroblasty, where a plant may exhibit two or more leaf types during development, which is not necessarily related to a change in environment (Jones, 1999). The shift from one leaf type to another occurs only once and is often used to mark the transition from juvenile to adult development (Goebel, 1898; cited in Wells and Pigliucci, 2000). The key difference between the concepts relies on reversibility, where a heterophyllic species can adjust leaf type many times according to environmental conditions whereas a heteroblastic species is constrained to have a single change in the expression of leaf form through development (Jones, 1999; Wells and Pigliucci, 2000). Nevertheless, a heteroblastic plant can possibly delay or hasten a shift in leaf type given certain environmental conditions, and this constitutes an example of plasticity (Wells and Pigliucci, 2000; Burns, 2005). Studying heterophyllic or heteroblastic species and treating their different leaf types as sub-divisions of leaf mass ratio may improve our capacity to test predictions from OPT similarly to sub-dividing traits within the root mass ratio and stem mass ratio.
Species in the cosmopolitan genus Acacia subgenus Phyllodineae undergo heteroblastic development from double compound leaves, transitional leaves and then to a modified petiole/rachis called a phyllode (Fig. 1; New, 1984). Transitional leaves are unique by having both pinnae of compound leaves and the flattened petiole/rachis of phyllodes. The switch from compound to transitional leaves is heteroblastic in Acacia and its timing is species dependent. The switch from transitional leaves to phyllodes is also heteroblastic in most species; however, juvenile leaves can be retained in certain species, meaning this is a case of heterophylly. Phyllodes are generally considered to be better adapted to hot, dry and high light conditions, whereas compound leaves are more suited to cool, wet and low light (Brodribb and Hill, 1993). It has also been recognized that heteroblastic development is delayed under high rainfall (Farrell and Ashton, 1978) and in forest understoreys (Withers, 1979). Furthermore, the rate of photosynthetic return per unit biomass invested in the leaf is significantly lower in phyllodes than in compound leaves (Brodribb and Hill, 1993). This strategy is known to be used by plants grown in nutrient-impoverished environments and therefore phyllodes are probably better adapted to these environments. Under three contrasting environments of nutrients, light and water, species of Acacia may optimally partition resources to compound leaves, transitional leaves or phyllodes.
Fig. 1.
The three types of ‘leaf’ analysed in this experiment: (A) a double compound leaf; (B) a transitional leaf with two pinnae attached to a flattened rachis/petiole; (C) a modified leaf, the phyllode; a flattened rachis/petiole. Leaves were harvested 4 months after sowing from the fourth, eighth and 13th node, respectively, from a non-experimental plant grown in full sunlight. Scale bar = 10 mm.
The aim of the present study was to examine shifts in biomass allocation in a heteroblastic species, Acacia implexa, under contrasting environments of nutrients, light and water in order to test predictions of biomass allocation under the OPT framework. These three environments have been extensively studied under OPT (e.g. Givnish, 1988; Tilman, 1988; Ryser and Eek, 2000; Navas and Garnier, 2002; Chan et al., 2003; Moriuchi and Winn, 2005); however, studies with heteroblastic species have been comparatively rare. Explicitly, the aims of the study were to: (a) test for differences in the capacity of plants to respond to multiple environmental variables; (b) sub-divide leaf mass ratio into categories of different leaf types (i.e. compound vs. transitional leaves) to determine the capacity of the different leaf types to respond to contrasting environments; and (c) determine whether heteroblasty is a variable trait in A. implexa and under which environment it is most pronounced.
MATERIALS AND METHODS
Acacia implexa is a tree growing between 5 and 12 m tall (Kodela, 2002). It is a relatively fast-growing species primarily inhabiting slopes on shallow, well-drained soils of woodlands and open forest (Maslin and McDonald, 2004). It occurs in a rainfall zone of approx. 400–1000 mm and its natural range extends from northern Queensland to southern Victoria (Maslin and McDonald, 2004). Seeds of A. implexa were obtained from the CSIRO Australian Tree Seed Centre, Canberra, Australia (http://www.csiro.au/content/pt6.html) seedlot number 19770. The seeds were sourced from a population of 12 parents from Bylong New South Wales (32 °37'S, 150 °03'E, mean rainfall 650 mm per annum; average temperature 23 °C).
Experimental design
Seeds were pre-treated in boiling water for 2 min (scarification is necessary for this species as it germinates following heat induced by fire). Six seeds were sown into a single 115 mL pot filled with soil. Soil in each pot consisted of 33 % Australian Native Landscape supply of ‘Organic Garden Mix’ (with a ratio of 50 % black soil, 20 % coarse sand and 30 % organics), 33 % washed river sand and 33 % cocopeat. Previous experience in growing Australian Acacia species found good results using this complex soil mixture. Plants within each pot were randomly thinned to one per pot following the emergence of the first double compound leaf. The first double compound leaf was the second leaf to develop following a single compound leaf that developed after the cotyledons.
All pots were assigned to one of either high or low nutrient, light and water treatments. The high nutrient treatment was established by adding a general fertilizer containing 36 % dolomite, 14 % blood and bone, 14 % ammonium nitrate, 14 % gypsum, 14 % lime and 7 % trace elements to the soil mix. Additionally, a 4 month Osmocote low phosphorus slow-release fertilizer was added with the N:P:K ratio of 17:1·5:8·5. The low nutrient treatment had no additional fertilizer added to the soil mix. Plants in the high light treatment received natural sunlight, whereas the plants in the low light treatment were grown inside a 57 cm cylinder (open ended) of green filter plastic (Lee Filters, Andover, UK; number 121 Lee Green). The light filters simulated a natural canopy where photosynthetic flux density was reduced to 65 % and the red:far red ratio was reduced from 1·0 to 0·2 (Bonser and Geber, 2005). The high water treatment was kept well watered at all times. Plants in the low water treatment were watered after early signs of wilting were shown. Low water plants were typically watered about once per week. Seeds were sown directly into high/low nutrient soil and the light and water treatments began after the emergence of the first double compound leaf.
Plants were grown according to a complete randomized full-factorial design. The size of the experiment was: 2× nutrients, 2× light, 2× water; 25× replicates gave a total sample size of 200 plants. All plants were grown in the School of Biological, Earth and Environmental Sciences glasshouse at the University of New South Wales, Sydney, Australia. The experiment was conducted over 120 d between February and June 2006 with glasshouse temperatures maintained between 19 and 26 °C. Previous experience with growing this species showed most individuals had undergone heteroblastic development by 120 d and it was of interest to determine a ‘snap-shot’ of the plant phenotype under different treatments at a single point in time. A total of 170 plants were harvested while 30 died. Despite efforts to maintain sterility, nine plants were found to have symbiotic rhizobium (which fix atmospheric nitrogen and thus improve fertility and growth), and these were removed from the analysis to give a total sample size of 161 plants. Mortality was largely random across light and water treatments; however, the high nutrient treatment had n = 68 and the low nutrient treatment had n = 93.
The focus of the experiments was on measuring a core set of morphological traits that were classified as either whole-plant traits or those traits associated with heteroblasty (i.e. within module leaf traits). Whole-plant traits included data from all leaves irrespective of leaf type, whereas heteroblastic traits were derived from leaves explicitly divided into different leaf types. A summary of these traits is presented in Table 1. Stem length and diameter at soil level were taken before harvest. At harvest, leaves were separated from the stem and classified as either compound, transitional or phyllode. Leaves were flattened and refrigerated at 4 °C until area measurements were undertaken. This prevented pinnules on the compound leaves from folding and modifying the leaf area. Each individual leaf area was measured using LeafA software (G. Williamson, University of Adelaide, Australia, pers. comm.) with a flatbed scanner. Individual leaves were oven dried at 60 °C for at least 7 d and then the mass was taken. Specific leaf area (SLA) was calculated as fresh leaf area (cm2) divided by dried mass (g) for each leaf and then averages were taken. Soil was carefully removed from roots in a plastic tray to ensure all root material was collected. Stem and roots were oven-dried at 60 °C for at least 7 d and then the mass was taken.
Table 1.
Summary of the traits, abbreviations and statistical transformations that were used in this experiment.
| Trait | Abbreviation | Definition | Transformation |
|---|---|---|---|
| Whole-plant traits | |||
| Root mass ratio (g g−1) | RMR | Ratio of total root mass to sum of stem and leaf mass | log |
| Stem mass ratio (g g−1) | StMR | Ratio of total stem mass to sum of root and leaf mass | log |
| Leaf mass ratio (g g−1) | LMR | Ratio of total leaf mass to sum of stem and root mass | log |
| Leaf area ratio (cm2 g−1) | LAR | Ratio of total leaf area to total biomass | log |
| Height to diameter (mm mm−1) | HtoD | Ratio of stem length to basal stem diameter | log + 1 |
| Internode length (mm) | Internode | Stem length divided by the total number of nodes | log + 1 |
| Specific leaf area (cm2 g−1) | SLA | Ratio of leaf area to leaf mass | log |
| Heteroblastic traits | |||
| Compound leaf mass ratio (g g−1) | CompLMR | Ratio of total compound leaf mass to sum of root, stem and other leaf mass | sqrt |
| Transitional leaf mass ratio (g g−1) | TransLMR | Ratio of total transitional leaf mass to sum of root, stem and other leaf mass | log |
| Compound leaf area ratio (cm2 g−1) | CompLAR | Ratio of total compound leaf area to total biomass | sqrt |
| Transitional leaf area ratio (cm2 g−1) | TransLAR | Ratio of total transitional leaf area to total biomass | log + 1 |
| Compound specific leaf area (cm2 g−1) | CompSLA | Ratio of compound leaf area to compound leaf mass | ln |
| Transitional specific leaf area (cm2 g−1) | TransSLA | Ratio of transitional leaf area to transitional leaf mass | log + 1 |
| Number of compound nodes | CompNodes | Number of nodes which developed a compound leaf before the first transitional leaf | log |
Data analysis
Multivariate analysis of variance (MANOVA: Wilks' λ) was used to test for the effects of nutrient, light and water environment on the following traits: root mass ratio (RMR), stem mass ratio (StMR), leaf mass ratio (LMR), leaf area ratio (LAR), height to diameter (HtoD), internode length (Internode) and SLA (Table 1). Data were checked for normality using the Shapiro–Wilk test and transformed where appropriate (details of transformations are shown in Table 1). The homogeneity of variances in MANOVA was checked with Levene's test of equality (Supplementary Information, available online) and outliers checked with Mahalanobis distances. Multivariate sets of covarying traits were further characterized using discriminant function analysis (DFA). DFA is a classification procedure that can be applied on factorial multivariate data sets to assess whether groups can be uniquely separated (Quinn and Keough, 2002). In this study, interest was focused on whether the eight treatment combinations could be classified into unique groups or whether there were overlaps in groups. Means with 95 % confidence intervals of experimental treatments were plotted against the first and second discriminant functions using procedures outlined in Johnson et al. (2007). Whole-plant and heteroblastic traits were plotted separately. Univariate F-tests on each trait were examined for each environment and interaction, and were ordered from highest to lowest to determine their contribution to the MANOVA (Quinn and Keough, 2002; Monroe and Poore, 2005). In order to examine how heteroblastic traits affect whole-plant traits, the same MANOVA procedure as described above was undertaken. However, in this case, the following traits were analysed: RMR, StMR, compound LMR (CompLMR), transitional LMR (TransLMR), CompLAR, TransLAR, HtoD, Internode, CompSLA and TransSLA. Phyllodes were not included as an individual trait in the multivariate analysis as very few plants produced phyllodes across the experiment. Nevertheless, phyllode mass data were included in overall biomass calculations in those plants that did develop phyllodes. Pairwise comparisons within treatments were sequential Bonferoni corrected to control for multiple comparisons.
A specific examination was undertaken to determine if the developmental shift from a compound leaf to a transitional leaf (i.e. heteroblasty) was a plastic trait in A. implexa. An analysis of covariance (ANCOVA) was performed where the number of nodes which developed a compound leaf (CompNodes – Table 1) was the dependent variable, biomass was the covariate, and nutrient, light and water were fixed factors. All analyses were performed using SPSS 15·0·1 (SPSS Inc., Chicago, IL).
RESULTS
Whole-plant response
Acacia implexa displayed a suite of plastic responses induced by all three experimental treatments, and there were some significant interactions among treatments. MANOVA indicated a strong effect caused by the nutrient and light treatments, with the water treatment having a marginally significant effect (Table 2a). The light × water interaction was also marginally significant; however, there was a strong effect in the three-way nutrient × light × water interaction (Table 2a). In the nutrient treatment, univariate F-tests indicated that the greatest plastic responses were between partitioning of biomass to roots in a low nutrient environment and to leaves in the high nutrient environment (Table 2b, Fig. 2). Only StMR and Internode had no plastic response to nutrients (Table 2b). Conversely, StMR and Internode were highly plastic traits in the light environment, as were HtoD and SLA (Table 2b). These traits were significantly greater in the low light treatment, whereas RMR and LAR were significantly greater in the high light treatment (Fig. 2). There were fewer plastic trait responses in the water treatment (Table 2b); however, RMR and HtoD were higher in the low water treatment, and Internode and LMR were higher in the high water treatment (Fig. 2). It should also be noted that, in spite of significant multivariate interactions in the nutrient × light and nutrient × light × water treatments, these interactions were not attributable to interactions in any single trait as there were no significant univariate interactions (Supplementary Information, available online).
Table 2.
Whole-plant multivariate and univariate analysis
| (a) MANOVA | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Wilks' λ | F-value | P-value | |||||||
| Nutrient | 0·326 | 43·489 | <0·0001 | ||||||
| Light | 0·379 | 34·362 | <0·0001 | ||||||
| Water | 0·906 | 2·170 | 0·040 | ||||||
| N × L | 0·957 | 0·936 | 0·480 | ||||||
| N × W | 0·942 | 1·304 | 0·252 | ||||||
| L × W | 0·908 | 2·125 | 0·044 | ||||||
| N × L × W | 0·881 | 2·829 | 0·009 | ||||||
| (b) ANOVA | |||||||||
| Rank | Nutrient | F-value | P-value | Light | F-value | P-value | Water | F-value | P-value |
| 1 | RMR | 191·396 | <0·0001 | Internode | 159·305 | <0·0001 | RMR | 12·543 | 0·001 |
| 2 | LMR | 71·291 | <0·0001 | HtoD | 153·654 | <0·0001 | HtoD | 7·399 | 0·007 |
| 3 | HtoD | 54·197 | <0·0001 | StMR | 39·505 | <0·0001 | Internode | 5·159 | 0·025 |
| 4 | LAR | 21·035 | <0·0001 | SLA | 39·035 | <0·0001 | LMR | 4·704 | 0·032 |
| 5 | SLA | 11·076 | 0·001 | RMR | 10·035 | 0·002 | LAR | 2·958 | 0·087 |
| 6 | StMR | 2·433 | 0·121 | LAR | 9·975 | 0·002 | StMR | 0·467 | 0·496 |
| 7 | Internode | 0·498 | 0·481 | LMR | 1·881 | 0·172 | SLA | 0·079 | 0·779 |
The univariate ANOVAs have been ranked from highest to lowest F-value as an indication of the influence of traits over the MANOVA model. MANOVA degrees of freedom are 7, 147. ANOVA degrees of freedom are 1, 153. Full ANOVA tables are presented in the Supplementary Information (available online)
Fig. 2.
Estimated marginal means and standard errors of whole-plant traits. Traits and their units of measurement are outlined in Table 1. * P < 0·05; ** P < 0·01; *** P < 0·001; **** P < 0·0001.
Heteroblastic trait responses
A suite of plastic responses were also observed when leaf-related traits were sub-divided into heteroblastic components. MANOVA results indicated that only the nutrient and light treatments had a significant effect, whereas the water treatment had no effect on the multivariate data set (Table 3a). The nutrient × light interaction was highly significant across the heteroblastic traits (Table 3a). Ordering of the F-values in the nutrient treatment showed that RMR had the highest rank, followed by TransLMR, CompLMR, TransLAR and CompLAR (Table 3b). TransLMR and TransLAR were significantly greater in the high nutrient treatment, yet CompLMR and CompLAR were significantly greater in the low nutrient treatment (Fig. 3). The light treatment still produced a strong plastic response in stem traits (StMR, HtoD and Internode; Table 3b); however, plasticity in leaf traits was complex. For example, CompLAR and CompLMR were significantly higher in a low light environment yet there was no difference in TransLAR or TransLMR (Table 3b, Fig. 3). On the other hand, there was no significant difference in CompSLA yet TransSLA was significantly greater in the low light environment (Table 3b).
Table 3.
Heteroblastic multivariate and univariate trait analysis
| (a) MANOVA | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Wilks' λ | F-value | P-value | |||||||
| Nutrient | 0·292 | 15·500 | <0·0001 | ||||||
| Light | 0·421 | 8·808 | <0·0001 | ||||||
| Water | 0·810 | 1·503 | 0·159 | ||||||
| N × L | 0·707 | 2·659 | 0·009 | ||||||
| N × W | 0·898 | 0·724 | 0·699 | ||||||
| L × W | 0·936 | 0·437 | 0·923 | ||||||
| N × L × W | 0·873 | 0·928 | 0·514 | ||||||
| (b) ANOVA | |||||||||
| Rank | Nutrient | F-value | P-value | Light | F-value | P-value | Water | F-value | P-value |
| 1 | TransLMR | 64·611 | <0·0001 | Internode | 64·435 | <0·0001 | RMR | 7·573 | 0·007 |
| 2 | RMR | 59·678 | <0·0001 | HtoD | 41·758 | <0·0001 | Internode | 4·371 | 0·040 |
| 3 | CompLMR | 44·805 | <0·0001 | TransSLA | 24·545 | <0·0001 | HtoD | 3·651 | 0·060 |
| 4 | TransLAR | 38·174 | <0·0001 | StMR | 11·607 | 0·001 | CompSLA | 1·572 | 0·214 |
| 5 | CompLAR | 34·531 | <0·0001 | CompLAR | 5·014 | 0·028 | CompLAR | 0·903 | 0·345 |
| 6 | HtoD | 8·731 | 0·004 | TransLAR | 3·238 | 0·076 | TransLAR | 0·126 | 0·724 |
| 7 | TransSLA | 5·369 | 0·023 | RMR | 3·026 | 0·086 | TransLMR | 0·098 | 0·755 |
| 8 | CompSLA | 4·218 | 0·044 | CompLMR | 1·671 | 0·200 | CompLMR | 0·075 | 0·785 |
| 9 | StMR | 1·348 | 0·249 | CompSLA | 1·216 | 0·274 | StMR | 0·062 | 0·804 |
| 10 | Internode | 0·246 | 0·622 | TransLMR | 4·08 × 10−7 | 0·999 | TransSLA | 0·041 | 0·839 |
The univariate ANOVAs have been ranked from highest to lowest F-value as an indication of the influence of traits over the MANOVA model. MANOVA degrees of freedom are 10, 64. ANOVA degrees of freedom are 1, 73. Full ANOVA tables are presented in the Supplementary Information (available online).
Fig. 3.
Estimated marginal means and standard errors of heteroblastic traits. Traits and their units of measurement are outlined in Table 1. * P < 0·05; ** P < 0·01; *** P < 0·001; **** P < 0·0001.
Discriminant function analysis
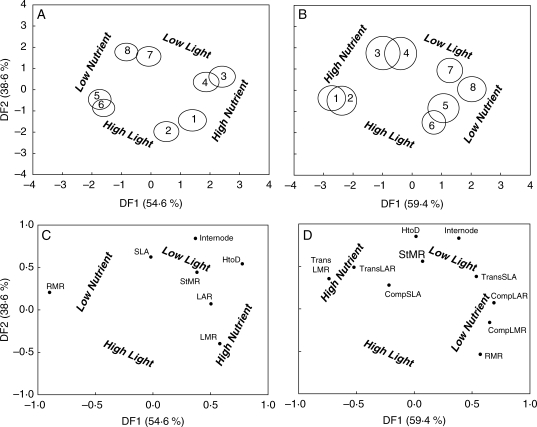
The first two discriminant functions explained 93·2 and 84·9 % of the total variance among whole-plant and heteroblastic traits, respectively. The first discriminant function accounted for the variability among the nutrient treatment, whereas the second discriminant function accounted for the light treatment (Fig. 4). Accordingly, biplots of the whole-plant traits revealed LMR to be positively and RMR to be negatively associated with the first discriminant function, and StMR, SLA, HtoD and Internode were found to be positively associated with the second discriminant function (Fig. 4C). Biplots of the heteroblastic traits revealed a more complex pattern also revealed from the MANOVA and ANOVA analysis. CompLMR and CompLAR were positively associated with the first discriminant function and TransLMR and TransLAR were negatively associated, corresponding to the groupings of the low and high nutrient treatments, respectively (Fig. 4D).
Fig. 4.
Grouping of treatments and traits following discriminant function analysis. Variance explained by the first and second discriminant functions are displayed in parentheses. (A, B) Treatments are mean populations, with circles indicating 95 % confidence intervals for (A) whole-plant and (B) heteroblastic traits. Numbers correspond to treatment group (1, high nutrient, high light, high water; 2, high nutrient, high light, low water; 3, high nutrient, low light, high water; 4, high nutrient, low light, low water; 5 low nutrient, high light, high water; 6, low nutrient, high light, low water; 7, low nutrient, low light, high water; 8, low nutrient, low light, low water). The nutrient and light treatments are highlighted due to significant outcomes following MANOVA. (C, D) Factor loadings of (C) whole-plant and (D) heteroblastic traits across the first and second discriminant functions.
Heteroblastic developmental plasticity
After 120 d of growth not all plants had undergone heteroblastic development, with the least amount occurring in the low nutrient treatment (Supplementary Information). The heteroblastic shift between compound leaves and transitional leaves was significantly different under the light treatment yet there was no difference in the nutrient or water treatment (Fig. 5). None of the interactions between treatments delayed the shift in leaf type. Total biomass had no significant effect on the model as a covariate (Table 4) despite only the light treatment showing no difference in total biomass at point of harvest (Fig. 6).
Fig. 5.
Number of nodes displaying a compound leaf (CompNodes) with standard error bars. Analysis was performed on log data, but untransformed data are presented. Only the light treatment showed a significant difference (**** P < 0·0001). The full ANCOVA table is presented in the Supplementary Information (available online).
Table 4.
ANCOVA table following analysis of CompNodes
| Treatment | d.f. | MSS | F-value | P-value |
|---|---|---|---|---|
| Total biomass | 1 | 0·010 | 1·450 | 0·230 |
| Nutrient | 1 | 0·010 | 1·392 | 0·240 |
| Light | 1 | 0·092 | 12·987 | <0·0001 |
| Water | 1 | 0·002 | 0·224 | 0·636 |
| Nutrient × light | 1 | 0·003 | 0·441 | 0·508 |
| Nutrient × water | 1 | 0·019 | 2·608 | 0·108 |
| Light × water | 1 | 0·017 | 2·436 | 0·121 |
| Nutrient × light × water | 1 | 0·011 | 1·590 | 0·209 |
| Error | 152 | 0·007 | ||
| Total | 161 |
Within-treatment trait means are presented in Fig. 5. Total biomass at point of harvest was chosen as the covariate in order to control for possible differences in plant size on CompNodes.
Fig. 6.
Estimated marginal mean and standard errors of total biomass (g) at point of harvest. Analysis was performed on log data, but untransformed data are presented. The nutrient and water treatments showed a significant difference (**** P < 0·0001). The full ANOVA table is presented in the Supplementary Information (available online).
DISCUSSION
OPT predicts that biomass will be allocated to traits where there are limited below-ground or above-ground resources. Patterns in whole-plant allocation of A. implexa reported here largely support this prediction. For example, a greater allocation to roots and leaves in a low and high nutrient environment, respectively, has been well documented (Reynolds and Dantonio, 1996; Li et al., 1999; Poorter and Nagel, 2000; Navas and Garnier, 2002). It was demonstrated here that the root mass ratio is higher in the low nutrient treatment, and leaf mass ratio and leaf area ratio are greater in the high nutrient treatment (Figs 2 and 4C). Further, OPT predicts a greater allocation to stem traits in a low light environment (Tilman, 1988; Poorter and Nagel, 2000). It was found that stem mass ratio, height to diameter ratio and mean internode length all increased under the low light treatment (Figs 2 and 4C), supporting the prediction of increased allocation to stem traits. OPT predicts that plants in a low water environment will have proportionally greater allocation to roots and less to leaves (Tilman, 1988), and this prediction is supported by significantly greater root mass ratio in the low water treatment and greater leaf mass ratio in the high water treatment (Fig. 2).
In spite of strong conformity of whole-plant traits to predictions made by OPT, SLA seemed to show conflicting results. First, the water treatment had no effect on SLA (Fig. 2) despite much research suggesting aridity should decrease SLA as plants increase structural leaf mass to prevent desiccation (e.g. Cunningham et al., 1999; Specht and Specht, 1999; Zhang et al., 2005). Moreover, leaf area expansion tends to decrease with lack of water as a driving factor. Similarly, nutrient impoverishment should lower SLA as leaves increase structural defences to avoid loss of expensive photosynthetic tissue to herbivory or other damage (e.g. Cunningham et al., 1999; Li et al., 1999; Specht and Specht, 1999; Navas and Garnier, 2002). However, in this study SLA was significantly greater in the low nutrient treatment (Table 3). From a whole-plant perspective, incongruent SLA results are not necessarily conflicting. For example, leaf mass ratio was significantly lower in the low water treatment but leaf area ratio was not significantly different across water treatments (Fig. 2), suggesting that plants produced fewer leaves but maintained a relatively high photosynthetic area. In the high nutrient treatment, plants were significantly larger and developmentally older than plants in the low nutrient treatment (Fig. 6). SLA tends to decrease with age (Thomas and Winner, 2002) as the value of highly productive but short-lived leaves diminishes (Bonser, 2006). The response of SLA in the light treatment was much easier to interpret than the nutrient and water treatments as SLA was significantly larger in low light (Fig. 2). This is a widely accepted pattern allowing plants a greater photosynthetic surface area in a light-limited environment (e.g. Thompson et al., 1988; Ellsworth and Reich, 1992; Dong, 1993, Gunn et al., 1999; James and Bell, 2000; Evans and Poorter, 2001; Poorter et al., 2006; Rozendaal et al., 2006).
Sub-dividing leaf traits into different categories of compound and transitional leaves provided a greater understanding of plant growth strategy and biomass partitioning, particularly in the nutrient treatment. The high nutrient treatment had significantly higher transitional leaf allocation (both transitional leaf mass and area) and the low nutrient treatment had higher allocation to compound leaves (Figs 3 and 4D). This result suggests a case of apparent plasticity where plants in the high nutrient treatment are larger (Fig. 6) and more developmentally advanced than low nutrient plants and therefore would naturally have more transitional leaves. However, results from sub-dividing SLA into compound SLA and transitional SLA suggest that there was a more complex response to the contrasting nutrient treatment – particularly in the development of compound leaves. Compound SLA is significantly larger in the high nutrient than the low nutrient treatment (Fig. 3). Plants in the high nutrient treatment produced cheaply constructed but short-lived compound leaves that led to relatively fast growth of above-ground biomass. Plants in the low nutrient treatment, on the other hand, invested more resources into compound leaves to ensure they were not as cheaply constructed and therefore could obtain a longer lifespan from them. This investment of more resources increased SLA at the expense of further above-ground development. In this way, more resources could be allocated to below-ground biomass.
The light treatment also produced complex patterns and there was evidence of a plastic response of increasing photosynthetic capture area in the low light environment. In compound leaves, plants respond to low light by producing more leaf material on an area basis (Fig. 3). In transitional leaves, plants respond to low light by producing leaves with higher SLA (Fig. 3). Compound leaves are more productive than transitional leaves in a low light environment (Brodribb and Hill, 1993), and therefore there appears to be an adaptive value in developing more of these before the onset of heteroblasty. Yet it is not clear why plants did not also produce compound leaves with relatively higher SLA in the low light treatment as they did with transitional leaves, which is generally known to be the case in shade-avoiding species (e.g. Thompson et al., 1988; Ellsworth and Reich, 1992; Dong, 1993; Gunn et al., 1999; James and Bell, 2000; Evans and Poorter, 2001; Poorter et al., 2006; Rozendaal et al., 2006).
At the point of harvest in this experiment it was not obvious whether having more transitional leaves was adaptive as not all plants had developed phyllodes. The development of phyllodes in this species is not heteroblastic but rather is heterophyllic. Individuals at later stages of development that display only phyllodes can subsequently develop transitional leaves if placed in a shaded environment (M. Forster, pers. obs.). This experiment was run only over 120 d, and running a similar experiment for a longer period will assist in examining patterns of allocation between transitional leaves and phyllodes.
The developmental timing of heteroblasty was only significantly delayed (i.e. developmentally delayed and not the actual timing) in the low light environment (Fig. 5). Maintaining more productive compound leaves in a light-limited environment should allow plants to maintain a high growth rate (Brodribb and Hill, 1993). For example, at the end of the experiment, the low nutrient and low water plants were significantly smaller in overall plant size than their high treatment counterparts, yet plants in the light environment were the same size (Fig. 6). There have also been previous studies documenting no difference in biomass of plants grown in high and low light (Dong, 1993; van Tienderen and van Hinsberg, 1996; van Hinsberg and van Tienderen, 1997). This suggests that some species do not differ in growth according to light environment. Nevertheless, plasticity in heteroblastic development due to differences in the light environment has also been observed in other species (e.g. Day, 1988; James and Bell, 2000; Burns, 2005). In Acacia pycnantha, shaded juveniles retained compound leaves for a longer period than juveniles grown in full sun (Withers, 1979). Farrell (1973) also suggests that compound leaves are shade leaves and phyllodes are sun leaves in Acacia melanoxylon. This would seem an important adaptive strategy in Acacia as members of the genus are pioneer species competing intra- and interspecifically for light as juveniles while adults generally inhabit a relatively higher light environment (New, 1984).
In this experiment, plastic response of traits to contrasting environments largely followed predictions made by OPT. However, it was found that explicitly sub-dividing leaves into different types produced complex plastic responses that would not have been observed if all leaves were treated as a single functional type.
SUPPLEMENTARY DATA
Supplementary data is available at Annals of Botany online and consists of the following. Table S1, complete ANOVA table following analysis of whole-plant traits; Table S2, complete ANOVA table following analysis of heteroblastic traits; Table S3, the number of individuals that had survived and undergone heteroblastic development following 120 d of growth in the experiment; and Table S4, complete ANOVA table following analysis of total biomass.
ACKNOWLEDGEMENTS
We thank Brenton Ladd and Geoff McDonnell for assistance, and Grant Williamson for leaf area software. We also thank Kevin Burns, David Causton and an anonymous reviewer for helpful comments, and gratefully acknowledge funding by a UNSW Faculty Research Grant and Early Career Research grant to S.P.B.
LITERATURE CITED
- Andrews M, Raven JA, Sprent JI. Environmental effects on dry matter partitioning between shoot and root of crop plants: relations with growth and shoot protein concentration. Annals of Applied Biology. 2001;138:57–68. [Google Scholar]
- Bellingham PJ, Sparrow AD. Resprouting as a life history strategy in woody plant communities. Oikos. 2000;89:409–416. [Google Scholar]
- Bloom AJ, Chapin FS, Mooney HA. Resource limitations in plants – an economic analogy. Annual Review of Ecology and Systematics. 1985;16:363–392. [Google Scholar]
- Boardman NK. Comparative photosynthesis of sun and shade plants. Annual Review of Plant Physiology. 1977;28:355–377. [Google Scholar]
- Bonser SP. Form defining function: interpreting leaf functional variability in integrated plant phenotypes. Oikos. 2006;114:187–190. [Google Scholar]
- Bonser SP, Geber MA. Growth form evolution and shifting habitat specialization in annual plants. Journal of Evolutionary Biology. 2005;18:1009–1018. doi: 10.1111/j.1420-9101.2005.00904.x. [DOI] [PubMed] [Google Scholar]
- Brodribb T, Hill RS. A physiological comparison of leaves and phyllodes in Acacia melanoxylon. Australian Journal of Botany. 1993;41:293–305. [Google Scholar]
- Bruni NC, Young JP, Dengler NC. Leaf developmental plasticity of Ranunculus flabellaris in response to terrestrial and submerged environments. Canadian Journal of Botany. 1996;74:823–837. [Google Scholar]
- Burns KC. Plastic heteroblasty in beach groundsel (Senecio lautus) New Zealand Journal of Botany. 2005;43:665–672. [Google Scholar]
- Chan SS, Radosevich SR, Grotta AT. Effects of contrasting light and soil moisture availability on the growth and biomass allocation of Douglas-fir and red alder. Canadian Journal of Forestry Research. 2003;33:106–117. [Google Scholar]
- Chesson P. Mechanisms of maintenance of species diversity. Annual Review of Ecology and Systematics. 2000;31:343–366. [Google Scholar]
- Cunningham SA, Summerhayes B, Westoby M. Evolutionary divergences in leaf structure and chemistry, comparing rainfall and soil nutrient gradients. Ecological Monographs. 1999;69:569–588. [Google Scholar]
- Day JS. Light conditions and the evolution of heteroblasty (and the divaricate form) in New Zealand. New Zealand Journal of Ecology. 1998;22:43–54. [Google Scholar]
- Dong M. Morphological plasticity in the clonal herb Lamiastrum galeobdolon (L.) Ehrend. & Polatschek in response to partial shading. New Phytologist. 1992;124:291–300. doi: 10.1111/j.1469-8137.1993.tb03819.x. [DOI] [PubMed] [Google Scholar]
- Ellsworth DS, Reich PB. Leaf mass per area, nitrogen content, and photosynthetic carbon gain in Acer saccharum seedlings in contrasting forest light environments. Functional Ecology. 1992;6:423–435. [Google Scholar]
- Evans JR, Poorter H. Photosynthetic acclimation of plants to growth irradiance: the relative importance of specific leaf area and nitrogen partitioning in maximizing carbon gain. Plant, Cell and Environment. 2001;24:755–767. [Google Scholar]
- Farrell TP. The ecology of Acacia melanoxylon. Melbourne, Australia: University of Melbourne; 1973. MSc thesis. [Google Scholar]
- Farrell TP, Ashton DH. Population studies of Acacia melanoxylon. I. Variation in seed and vegetation characteristics. Australian Journal of Botany. 1978;26:365–379. [Google Scholar]
- Givnish TJ. Adaptation to sun and shade: a whole-plant perspective. Australian Journal of Plant Physiology. 1988;15:63–92. [Google Scholar]
- Gleeson SK, Tilman D. Allocation and the transient dynamics of succession on poor soils. Ecology. 1990;71:1144–1155. [Google Scholar]
- Gleeson SK, Tilman D. Plant allocation, growth rate and successional status. Functional Ecology. 1994;8:543–550. [Google Scholar]
- Goebel K. Organographie der Pflanzen. Jena: Fischer; 1898. [Google Scholar]
- Gunn S, Farrar JS, Collis BE, Nason M. Specific leaf area in barley: individual leaves versus whole plants. New Phytologist. 1999;143:45–51. [Google Scholar]
- van Hinsberg A, van Tienderen P. Variation in growth form in relation to spectral light quality (red/far-red ratio) in Plantago lanceolata L. in sun and shade populations. Oecologia. 1997;111:452–459. doi: 10.1007/s004420050258. [DOI] [PubMed] [Google Scholar]
- James SA, Bell DT. Influence of light availability on leaf structure and growth of two Eucalyptus globulus ssp. globulus provenances. Tree Physiology. 2000;20:1007–1018. doi: 10.1093/treephys/20.15.1007. [DOI] [PubMed] [Google Scholar]
- Johnson HE, Llyod AJ, Mur LAJ, Smith AR, Causton DR. The application of MANOVA to analyse Arabidopsis thaliana metabolomic data from factorially designed experiments. Metabolomics. 2007;3:517–530. [Google Scholar]
- Jones CS. An essay on juvenility, phase change, and heteroblasty in seed plants. International Journal of Plant Sciences. 1999;160:S105–S111. doi: 10.1086/314215. [DOI] [PubMed] [Google Scholar]
- Kodela PG. Acacia. In: Harden GJ, editor. Flora of New South Wales. Kensington: University of New South Wales; 2002. pp. 381–476. [Google Scholar]
- Li B, Suzuki J, Hara T. Competitive ability of two Brassica varieties in relation to biomass allocation and morphological plasticity under varying nutrient availability. Ecological Research. 1999;14:255–266. [Google Scholar]
- Maslin BR, McDonald MW. AcaciaSearch: evaluation of Acacia as a woody crop option for southern Australia. Canberra: Rural Industries Research and Development Corportation; 2004. [Google Scholar]
- McCarthy MC, Enquist BJ. Consistency between an allometric approach and optimal partitioning theory in global patterns of plant biomass allocation. Functional Ecology. 2007;21:713–720. [Google Scholar]
- McConnaughay KDM, Bazzaz FA. The occupation and fragmentation of space – consequences of neighboring shoots. Functional Ecology. 1992;6:711–718. [Google Scholar]
- Mommer L, Visser EJW. Underwater photosynthesis in flooded terrestrial plants: a matter of leaf plasticity. Annals of Botany. 2005;96:581–589. doi: 10.1093/aob/mci212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mommer L, Pons TL, Visser JW. Photosynthetic consequences of phenotypic plasticity in response to submergence: Rumex palustris as a case study. Journal of Experimental Biology. 2006;57:283–290. doi: 10.1093/jxb/erj015. [DOI] [PubMed] [Google Scholar]
- Monroe K, Poore AGB. Light quantity and quality induce induce shade-avoiding plasticity in a marine macroalga. Journal of Evolutionary Biology. 2005;18:426–435. doi: 10.1111/j.1420-9101.2004.00826.x. [DOI] [PubMed] [Google Scholar]
- Moriuchi KS, Winn AA. Relationships among growth, development and plastic response to environment quality in a perennial plant. New Phytologist. 2005;166:149–158. doi: 10.1111/j.1469-8137.2005.01346.x. [DOI] [PubMed] [Google Scholar]
- Navas M, Garnier E. Plasticity of whole plant and leaf traits in Rubia peregrina in response to light, nutrient and water availability. Acta Oecologica. 2002;23:375–383. [Google Scholar]
- New TR. A biology of Acacias. Melbourne: Oxford University Press; 1984. [Google Scholar]
- Poorter H, Nagel O. The role of biomass allocation in the growth response of plants to different levels of light, CO2, nutrients and water: a quantitative review. Australian Journal of Plant Physiology. 2000;27:595–607. [Google Scholar]
- Poorter H, Pepin S, Rijkers T, Jong Yd, Evans JR, Korner C. Construction costs, chemical composition and payback time of high- and low-irradiance leaves. Journal of Experimental Biology. 2006;57:355–371. doi: 10.1093/jxb/erj002. [DOI] [PubMed] [Google Scholar]
- Quinn GP, Keough MJ. Experimental design and data analysis for biologists. Port Melbourne: Cambridge University Press; 2002. [Google Scholar]
- Reynolds HL, Dantonio C. The ecological significance of plasticity in root weight ratio in response to nitrogen. Plant and Soil. 1996;185:75–97. [Google Scholar]
- Rozendaal DMA, Hurtado VH, Poorter L. Plasticity in leaf traits of 38 tropical tree species in response to light; relationships with light demand and adult stature. Functional Ecology. 2006;20:207–216. [Google Scholar]
- Ryser P, Eek L. Consequences of phenotypic plasticity vs. interspecific differences in leaf and root traits for acquisition of aboveground and belowground resources. American Journal of Botany. 2000;87:402–411. [PubMed] [Google Scholar]
- Sculthorpe CD. The biology of aquatic vascular plants. London: Edward Arnold; 1967. [Google Scholar]
- Specht RL, Specht A. Australian plant communities: dynamics of structure, growth and biodiversity. South Melbourne: Oxford University Press; 1999. [Google Scholar]
- Thomas SC, Winner WE. Photosynthetic differences between saplings and adult trees: an integration of field results by meta-analysis. Tree Physiology. 2002;22:117–127. doi: 10.1093/treephys/22.2-3.117. [DOI] [PubMed] [Google Scholar]
- Thompson WA, Stocker GC, Kriedemann PE. Growth and photosynthetic response to light and nutrients in Flindersia brayleyana F. Muell., a rainforest tree with broad tolerance to sun and shade. Australian Journal of Plant Physiology. 1988;15:299–315. [Google Scholar]
- Thornley JHM. A balanced quantitative model for root:shoot ratios in vegetative plants. Annals of Botany. 1972;36:431–441. [Google Scholar]
- van Tienderen PH, van Hinsberg A. Phenotypic plasticity in growth habit in Plantago lanceolata: how tight is a suite of correlated characters? Plant Species Biology. 1996;11:87–96. [Google Scholar]
- Tilman D. Resource competition and community structure. Princeton: Princeton University Press; 1982. [PubMed] [Google Scholar]
- Tilman D. Plant strategies and the dynamics and structure of plant communities. Princeton, NJ: Princeton University Press; 1988. [Google Scholar]
- Tilman D, Pacala S. The maintenance or species richness in plant communities. In: Ricklefs RE, Schluter D, editors. Species diversity in ecological communities. Chicago: University of Chicago Press; 1993. pp. 13–25. [Google Scholar]
- Wedin DA, Tilman D. Competition among grasses along a nitrogen gradient: initial conditions and mechanisms of competition. Ecological Monographs. 1993;63:199–229. [Google Scholar]
- Wells CL, Pigliucci M. Adaptive phenotypic plasticity: the case of heterophylly in aquatic plants. Perspectives in Plant Ecology, Evolution and Systematics. 2000;3:1–18. [Google Scholar]
- Winn AA. The functional significance and fitness consequences of heterophylly. International Journal of Plant Sciences. 1999;160:S113–S121. doi: 10.1086/314222. [DOI] [PubMed] [Google Scholar]
- Withers JR. Studies on the status of unburnt Eucalyptus woodland at Ocean Grove, Victoria. IV. The effect of shading on seedling establishment. Australian Journal of Botany. 1979;27:47–66. [Google Scholar]
- Wullschleger SD, Yin TM, Fazio SPD, Tschaplinski TJ, Gunter LE, Davis MF, Tuskan GA. Phenotypic variation in growth and biomass distribution for two advanced-generation pedigrees of hybrid poplar. Canadian Journal of Forestry Research. 2005;35:1779–1789. [Google Scholar]
- Zhang X, Wu N, Li C. Physiological and growth responses of Populus davidiana ecotypes to different soil water contents. Journal of Arid Environments. 2005;60:567–579. [Google Scholar]