Abstract
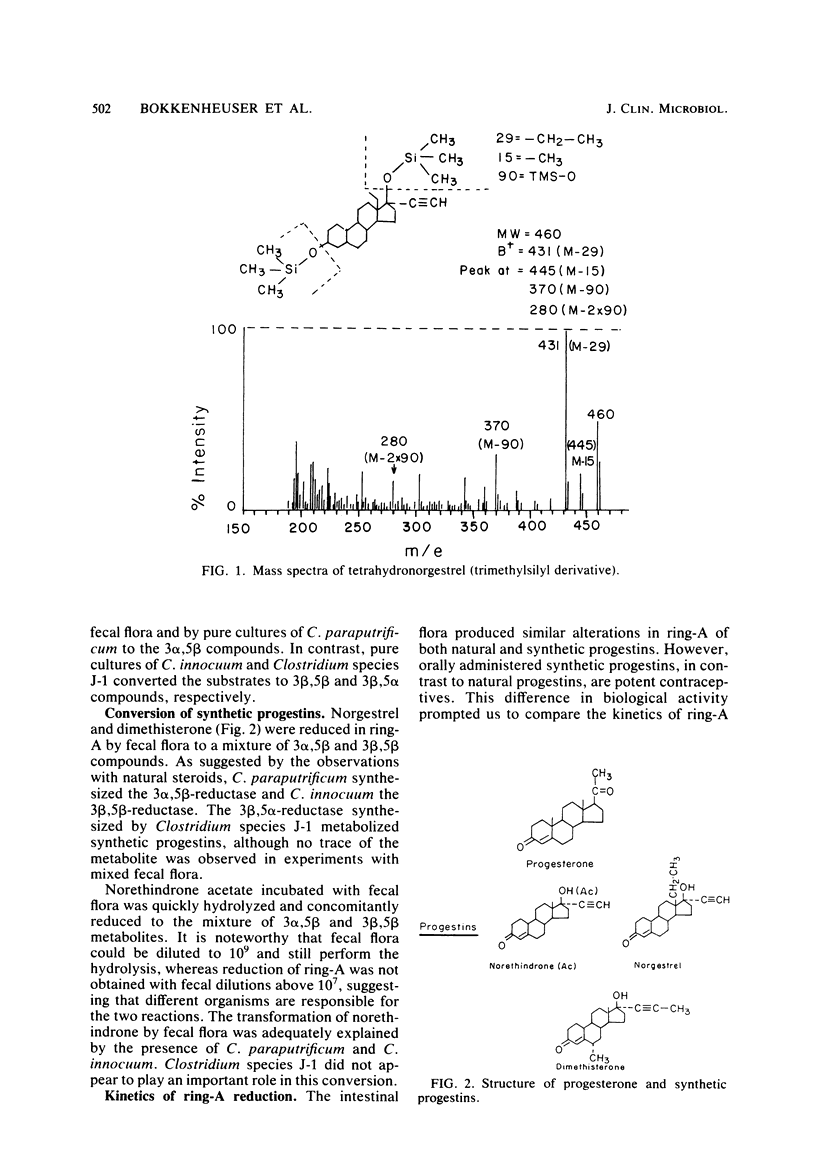
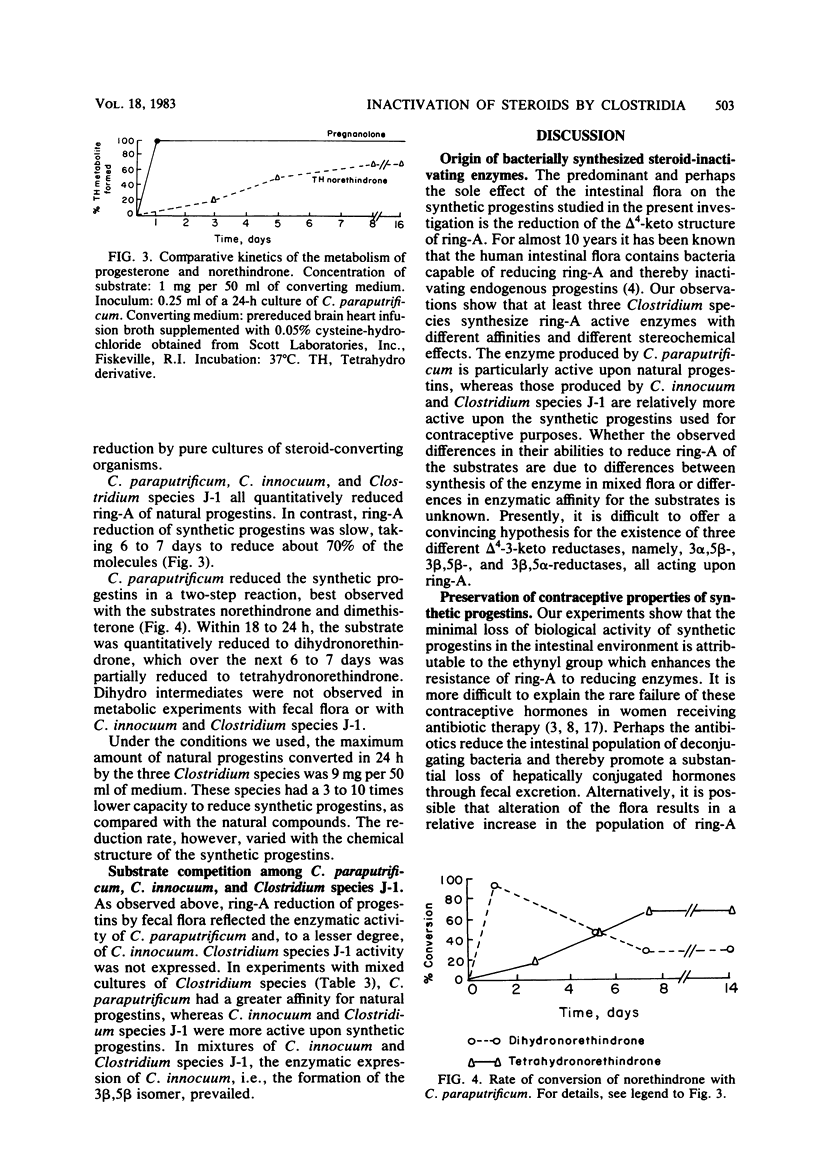
Steroid hormones reduced in ring-A are devoid of hormonal activity. In metabolic experiments we found that human fecal flora reduced the delta 4-3-keto structure of natural progestins to 3 alpha-hydroxy, 5 beta-steroid metabolites (3 alpha,5 beta) and of synthetic progestins to a mixture of 3 alpha,5 beta and 3 beta,5 beta compounds. 3 alpha,5 beta-Reductase was synthesized by Clostridium paraputrificum and had a strong affinity for natural progestins such as progesterone. 3 beta,5 beta-Reductase was synthesized by Clostridium innoculin and had a stronger affinity for synthetic progestins. A third enzyme, 3 beta,5 alpha-reductase, was synthesized by St. Luke's strain 209 (Clostridium species "J-1") but was only observed when pure cultures were used. Ring-A reduction of synthetic progestins was 3 to 10 times slower than that of natural progestins, thus explaining the pharmacological superiority of synthetic progestins over naturally occurring analogs.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adlercreutz H., Martin F., Järvenpä P., Fotsis T. Steroid absorption and enterohepatic recycling. Contraception. 1979 Sep;20(3):201–223. doi: 10.1016/0010-7824(79)90094-5. [DOI] [PubMed] [Google Scholar]
- Back D. J., Breckenridge A. M., Crawford F. E., Cross K. J., Orme M. L., Percival A., Rowe P. H. Reduction of the enterohepatic circulation of norethisterone by antibiotics in the rat: correlation with changes in the gut flora. J Steroid Biochem. 1980 Jan;13(1):95–100. doi: 10.1016/0022-4731(80)90118-1. [DOI] [PubMed] [Google Scholar]
- Bacon J. F., Shenfield G. M. Pregnancy attributable to interaction between tetracycline and oral contraceptives. Br Med J. 1980 Feb 2;280(6210):293–293. doi: 10.1136/bmj.280.6210.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bokkenheuser V. D., Winter J., Dehazya P., de Leon O., Kelly W. G. Formation and metabolism of tetrahydrodeoxycorticosterone by human fecal flora. J Steroid Biochem. 1976 Oct;7(10):837–843. doi: 10.1016/0022-4731(76)90187-4. [DOI] [PubMed] [Google Scholar]
- DECKER H. A., MIGEON C. J., PAUL A. C., SAMUELS L. T., SANDBERG A. A., SMITH D. F. Metabolism of 4-C 14-cortisol in man: body distribution and rates of conjugation. J Clin Endocrinol Metab. 1956 Sep;16(9):1137–1150. doi: 10.1210/jcem-16-9-1137b. [DOI] [PubMed] [Google Scholar]
- DeJongh D. C., Hribar J. D., Littleton P., Fotherby K., Rees R. W., Shrader S., Foell T. J., Smith H. The identification of some human metabolites of norgestrel, a new progestational agent. Steroids. 1968 May;11(5):649–666. doi: 10.1016/s0039-128x(68)80011-x. [DOI] [PubMed] [Google Scholar]
- Fotherby K. Metabolism of synthetic steroids by animals and man. Acta Endocrinol Suppl (Copenh) 1974;185:119–147. doi: 10.1530/acta.0.075s119. [DOI] [PubMed] [Google Scholar]
- Friedman C. I., Huneke A. L., Kim M. H., Powell J. The effect of ampicillin on oral contraceptive effectiveness. Obstet Gynecol. 1980 Jan;55(1):33–37. [PubMed] [Google Scholar]
- Kamyab S., Littleton P., Fotherby K. Metabolism and tissue distribution of norethisterone and norgestrel in rabbits. J Endocrinol. 1967 Nov;39(3):423–435. doi: 10.1677/joe.0.0390423. [DOI] [PubMed] [Google Scholar]
- LAYNE D. S., GOLAB T., ARAI K., PINCUS G. THE METABOLIC FATE OF ORALLY ADMINISTERED 3H-NORETHYNODREL AND 3H-NORETHINDRONE IN HUMANS. Biochem Pharmacol. 1963 Aug;12:905–911. doi: 10.1016/0006-2952(63)90121-7. [DOI] [PubMed] [Google Scholar]
- Laatikainen T. Excretion of neutral steroid hormones in human bile. Ann Clin Res. 1970;2(Suppl):1–28. [PubMed] [Google Scholar]
- Littleton P., Fotherby K., Dennis K. J. Metabolism of [14C]norgestrel in man. J Endocrinol. 1968 Dec;42(4):591–598. doi: 10.1677/joe.0.0420591. [DOI] [PubMed] [Google Scholar]
- MacDonald I. A., Rochon Y. P., Hutchison D. M., Holdeman L. V. Formation of ursodeoxycholic acid from chenodeoxycholic acid by a 7 beta-hydroxysteroid dehydrogenase-elaborating Eubacterium aerofaciens strain cocultured with 7 alpha-hydroxysteroid dehydrogenase-elaborating organisms. Appl Environ Microbiol. 1982 Nov;44(5):1187–1195. doi: 10.1128/aem.44.5.1187-1195.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin F., Järvenpä P., Kosunen K., Somers C., Lindstrom B., Adlercreutz H. Ring-A reduction of medroxyprogesterone acetate [17 alpha-acetoxy-6 alpha-methyl-4-pregnene-3,20-dione (MPA)] in biological systems. J Steroid Biochem. 1980 Jan;12:491–497. doi: 10.1016/0022-4731(80)90312-x. [DOI] [PubMed] [Google Scholar]
- Sisenwine S. P., Kimmel H. B., Liu A. L., Ruelius H. W. Urinary metabolites of DL-norgestrel in women. Acta Endocrinol (Copenh) 1973 May;73(1):91–104. doi: 10.1530/acta.0.0730091. [DOI] [PubMed] [Google Scholar]
- Skolnick J. L., Stoler B. S., Katz D. B., Anderson W. H. Rifampin, oral contraceptives, and pregnancy. JAMA. 1976 Sep 20;236(12):1382–1382. [PubMed] [Google Scholar]
- Taylor W. The excretion of steroid hormone metabolites in bile and feces. Vitam Horm. 1971;29:201–285. doi: 10.1016/s0083-6729(08)60050-3. [DOI] [PubMed] [Google Scholar]
- Winter J., Cerone-McLernon A., O'Rourke S., Ponticorvo L., Bokkenheuser V. D. Formation of 20 beta-dihydrosteroids by anaerobic bacteria. J Steroid Biochem. 1982 Dec;17(6):661–667. doi: 10.1016/0022-4731(82)90568-4. [DOI] [PubMed] [Google Scholar]
- Yamaguchi Y. Enzymic color development of urinary 3 alpha-hydroxysteroids on thin-layer chromatograms. Clin Chem. 1980 Mar;26(3):491–493. [PubMed] [Google Scholar]


