Abstract
The hypersensitive response (HR) is a cell death phenomenon associated with localized resistance to pathogens. Biphasic patterns in the generation of H2O2, salicylic acid and ethylene have been observed in tobacco during the early stages of the HR. These biphasic models reflect an initial elicitation by pathogen-associated molecular patterns followed by a second phase, induced by pathogen-encoded avirulence gene products. The first phase has been proposed to potentiate the second, to increase the efficacy of plant resistance to disease. This potentiation is comparable to the “priming” of plant defenses which is seen when plants display systemic resistance to disease. The events regulating the generation of the biphasic wave, or priming, remains obscure, however recently we demonstrated a key role for nitric oxide in this process in a HR occurring in tobacco. Here we use laser photoacoustic detection to demonstrate that biphasic ethylene production also occurs during a HR occurring in Arabidopsis. We suggest that ethylene emanation during the HR represents a ready means of visualising biphasic events during the HR and that exploiting the genomic resources offered by this model species will facilitate the development of a mechanistic understanding of potentiating/priming processes.
Key words: hypersensitive response, biphasic patterns, potentiation, defense priming, ethylene, Arabidopsis
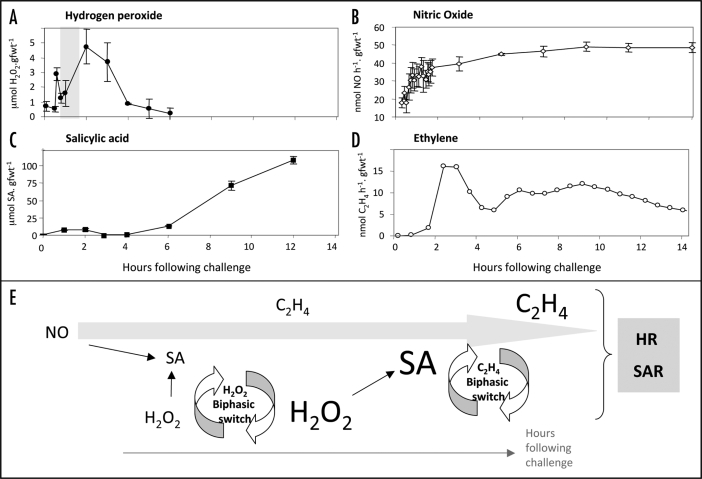
The Hypersensitive Response (HR) is a cell death process which occurs at the site of attempted pathogen attack and which has been associated with host resistance.1 Much work on the regulation of the HR has indicated the importance of H2O2,2 and NO.3 A feature of H2O2 generation during the HR is its biphasic pattern (Fig. 1A). The first rise reflects elicitation by pathogen-associated molecular patterns (PAMPs)4 and the second reflects the interaction between a pathogen-encoded avirulence (avr) gene product with a plant resistance (R) gene. A key aspect of the first rise is the initiation of salicylic acid (SA) synthesis which potentiates the second rise and hence the potency of plant defense and the HR.5
Figure 1.
Patterns of defense signal generation during the Pseudomonas syringae pv. phaseolicola elicited-hypersensitive response in tobacco (Nicotiana tabacum). Generation of (A) H2O2 (●, Mur18); (B) nitric oxide (◇; Mur12 (C) salicylic acid (SA, ■19) and (D) ethylene (○ Mur9) during a HR elicited by Pseudomonas syringae pv. phaseolicola (Psph) in tobacco cv. Samsun NN. In (A) a phase where SA acts to augment the second rise in H2O2—the potentiation phase—is highlighted. The potentiation phase is likely to be similar to defense “priming”.6 Methodological details are contained within the appropriate references. (E) A possible model for biphasic defense signal regulation during the Psph-elicited HR in tobacco. During an initial phase NO and H2O2 act to initiate SA biosynthesis, where SA and NO act to initiate a “H2O2 biphasic switch”. This could initially suppress both SA and the H2O2 generation but subsequently acts to potentiate a second phase of H2O2 generation. This in turn increases SA biosynthesis which could act with NO to initiate the “C2H4 biphasic switch” to potentiate ethylene production. These (and other) signals contribute to initiation of the HR and SAR.
This potentiation mechanism appears to be similar to defense priming; when whole plants display systemic resistance to disease as opposed to a localized resistance against pathogens. Priming can be initiated (the “primary stimulus”) following attack with a necrotizing pathogen (leading to “systemic acquired resistance”, SAR) or non-pathogenic rhizosphere bacteria (to confer “induced systemic resistance”, ISR). In the primed state a plant stimulates a range of plant defense genes, produces anti-microbial phytoalexins and deposits cell wall strengthening molecules, but only on imposition of a “secondary stimulus”.6 Such secondary stimuli include SA3 or PAMPs7 and is likely to be mechanistically similar to the potentiation step in the biphasic pattern of H2O2 generation (shaded in Fig. 1A). Accordingly, the two phases in the biphasic wave represent primary and secondary stimuli in priming.
Highlighting a similarity between local HR-based events and priming, adds further impetus to efforts aiming to describe the underlying mechanism(s), however both phenomena remain poorly understood. Besides SA, both jasmonates and abscisic acid (ABA) have been shown to prime defenses as have a range of non-plant chemicals, with β-aminobutyric acid (BABA) being perhaps most widely used.6,8 Mutants which fail to exhibit BABA-mediated potentiation were defective in either a cyclin-dependent kinase-like protein, a polyphosphoinositide phosphatase or an ABA biosynthetic enzyme.8
We have recently investigated biphasic ethylene production during the HR in tobacco elicited by the nonhost HR-eliciting bacterial pathogen Pseudomonas syringae pv. phaseolicola.9 As with H2O2 generation, this pattern reflected PAMP-and AVR-dependent elicitation events and included a SA-mediated potentiation stage. Crucially, we also showed that NO was a vital component in the SA-potentiation mechanism. When this finding is integrated with our other measurements of defense signal generation in the same host-pathogen system the complexity in the signaling network is revealed (Fig. 1). NO generation (Fig. 1B) appeared to be coincident with the first rise in H2O2 (Fig. 1A) which initiated SA biosynthesis10,11 and together would contribute to the first small, but transient, rise in that hormone (Fig. 1C). In line with established models5 this momentary rise in SA coincides with the potentiation phase (shaded in Fig. 1A) required to augment the second rise in ROS. However, ethylene production seems to be correlated poorly with the patterns of NO, H2O2 and SA (Fig. 1D). Nevertheless, biphasic ethylene production was found to reflect PAMP and AVR-dependent recognition and included a SA-mediated potentiation step.9 Hence, ethylene production could be used as a post-hoc indicator of the potentiation mechanism. Therefore, our discovery that the second wave of ethylene production—a “biphasic switch”—is influenced by NO acting with SA could also be relevant to the H2O2 generation. Significantly, the second phases in both H2O2 and ethylene production occur exactly where SA and NO production coincides; in the case of H2O2 generation 2–4 h post challenge and with ethylene 6 h onwards (Fig. 1E).
Thus, ethylene production represents a readily assayable marker to indicate perturbations in the underlying biphasic and possible priming mechanisms. As we have demonstrated, laser photoacoustic detection (LAPD) is a powerful on-line approach to determine in planta ethylene production in tobacco9,12 but any mechanistic investigations would be greatly facilitated if the genetic resources offered by the model species Arabidopsis could be exploited.
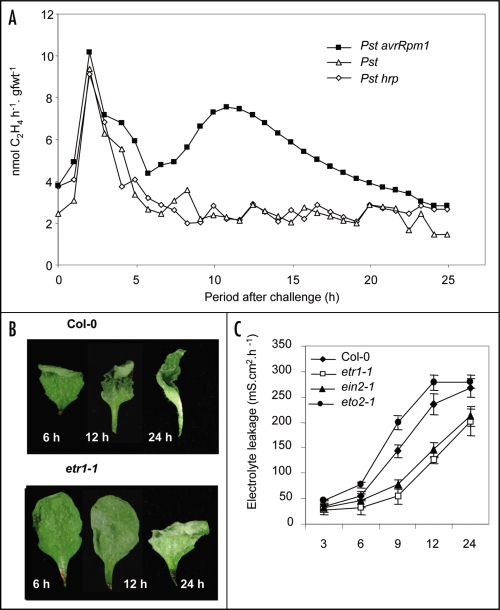
To address this, Arabidopsis Col-0 rosettes were vacuum infiltrated with either Pseudomonas syringae pv. tomato (Pst) avrRpm1 (HR-eliciting), the virulent Pst strain and the non-HR eliciting and non-virulent Pst hrpA strain. Ethylene production was monitored by LAPD (Fig. 2A). Significantly, Pst avrRpm1 initiated a biphasic pattern of ethylene production whose kinetics were very similar to that seen in tobacco (compare Figs. 2A with 1D). Inoculations with Pst and Pst hrpA only displayed the first PAMP-dependent rise in ethylene production. Thus, these data establish that Arabidopsis can be used to investigate biphasic switch mechanism(s) in ethylene production during the HR and possibly defense priming. When considering such mechanisms, it is relevant to highlight the work of Foschi et al.13 who observed that biphasic activation of a monomeric G protein to cause phase-specific activation of different kinase cascades. Interestingly, ethylene has been noted to initiate biphasic activation of G proteins and kinases in Arabidopsis, although differing in kinetics to the phases seen during the HR.14 Further, plant defense priming has been associated with the increased accumulation of MAP kinase protein.6
Figure 2.
Ethylene in the Pseudomonas syringae pv. tomato elicited-hypersensitive response in Arabidopsis thaliana. (A) Ethylene production from 5 week old short day (8 h light 100 µmol.m2.sec−1) grown Arabidopsis rosette leaves which were vacuum infiltrated with bacterial suspensions (2 × 106 colony forming units.ml−1) of Pseudomonas syringae pv. tomato (Pst) strains detected using laser photoacoustic detection (LAPD). Experimental details of the ethylene detection by LAPD are detailed in Mur et al.9 The intercellular spaces in leaves were infiltrated with the HR-eliciting strain Pst avrRpm1, (■), the virulent strain Pst (△) or the non-virulent and non-HR eliciting derivative, Pst hrpA (◇). (B) The appearance of Arabidopsis Col-0 and etr1-1 leaves at various h following injection with 2 × 106 c.f.u.mL−1 with of Pst avrRpm1. (C) Explants (1 cm diameter discs) from Arabidopsis leaf areas infiltrated with suspensions of Pst avrRpm1 were placed in a 1.5 cm diameter well, bathed in 1 mL de-ionized H2O. Changes in the conductivity of the bathing solution, as an indicator of electrolyte leakage from either wild type Col-0 (◆), mutants which were compromised in ethylene signaling; etr1-1 (□), ein2-2 (▲) or which overproduced ethylene; eto2-1 (●) were measured using a conductivity meter. Methodological details are set out in Mur et al.9
A further point requires consideration; the role of ethylene as a direct contributor to plant defense.15 The contribution of ethylene to the HR has been disputed,16 but in tobacco we have observed that altered ethylene production influenced the formation of a P. syringae pv. phaseolicola elicited HR.9 In Arabidopsis, cell death in the ethylene receptor mutant etr1-1 following inoculation with Pst avrRpm1 is delayed compared to wild type (Fig. 2B). When electrolyte leakage was used to quantify Pst avrRpm1 cell death, both etr1-1 and the ethylene insensitive signaling mutant ein2-1 exhibited slower death than wild-type but in the ethylene overproducing mutant eto2, cell death was augmented (Fig. 2C). These data indicate that ethylene influences the kinetics of the HR.
Taking these data together we suggest that the complexity of signal interaction during the HR or in SAR/ISR could be further dissected by combining the genetic resources of Arabidopsis with measurements of ethylene production using such sensitive approaches as LAPD.
Footnotes
Previously published online as a Plant Signaling & Behavior E-publication: http://www.landesbioscience.com/journals/psb/article/8904
References
- 1.Mur LAJ, Kenton P, Lloyd AJ, Ougham H, Prats E. The hypersensitive response; the centenary is upon us but how much do we know? J Exp Bot. 2008;59:501–520. doi: 10.1093/jxb/erm239. [DOI] [PubMed] [Google Scholar]
- 2.Levine A, Tenhaken R, Dixon R, Lamb C. H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell. 1994;79:583–593. doi: 10.1016/0092-8674(94)90544-4. [DOI] [PubMed] [Google Scholar]
- 3.Mur LAJ, Carver TLW, Prats E. NO way to live; the various roles of nitric oxide in plant-pathogen interactions. J Exp Bot. 2006;57:489–505. doi: 10.1093/jxb/erj052. [DOI] [PubMed] [Google Scholar]
- 4.Parker JE. Plant recognition of microbial patterns. Trends Plant Sci. 2003;8:245–247. doi: 10.1016/S1360-1385(03)00105-5. [DOI] [PubMed] [Google Scholar]
- 5.Lamb C, Dixon RA. The oxidative burst in plant disease resistance. Ann Rev Plant Physiol Plant Mol Biol. 1997;48:251–275. doi: 10.1146/annurev.arplant.48.1.251. [DOI] [PubMed] [Google Scholar]
- 6.Goellner K, Conrath U. Priming: it's all the world to induced disease resistance. Eur J Plant Pathol. 2008;121:233–242. [Google Scholar]
- 7.Conrath U, Thulke O, Katz V, Schwindling S, Kohler A. Priming as a mechanism in induced systemic resistance of plants. Eur J Plant Pathol. 2001;107:113–119. [Google Scholar]
- 8.Ton J, Jakab G, Toquin V, Flors V, Iavicoli A, Maeder MN, et al. Dissecting the betaaminobutyric acid-induced priming phenomenon in arabidopsis. Plant Cell. 2005;17:987–999. doi: 10.1105/tpc.104.029728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mur LAJ, Laarhoven LJJ, Harren FJM, Hall MA, Smith AR. Nitric oxide interacts with salicylate to regulate biphasic ethylene production during the hypersensitive response. Plant Physiol. 2008;148:1537–1546. doi: 10.1104/pp.108.124404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chamnongpol S, Willekens H, Moeder W, Langebartels C, Sandermann H, Van Montagu A, et al. Defense activation and enhanced pathogen tolerance induced by H2O2 in transgenic tobacco. Proc Natl Acad Sci USA. 1998;95:5818–5823. doi: 10.1073/pnas.95.10.5818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Durner J, Wendehenne D, Klessig DF. Defense gene induction in tobacco by nitric oxide, cyclic GMP and cyclic ADP-ribose. Proc Natl Acad Sci USA. 1998;95:10328–10333. doi: 10.1073/pnas.95.17.10328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mur LAJ, Santosa IE, Laarhoven LJJ, Holton NJ, Harren FJM, Smith AR. Laser photoacoustic detection allows in planta detection of nitric oxide in tobacco following challenge with avirulent and virulent Pseudomonas syringae pathovars. Plant Physiol. 2005;138:1247–1258. doi: 10.1104/pp.104.055772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Foschi M, Chari S, Dunn MJ, Sorokin A. Biphasic activation of p21(ras) by endothelin-1 sequentially activates the ERK cascade and phosphatidylinositol 3-kinase. EMBO J. 1997;16:6439–6451. doi: 10.1093/emboj/16.21.6439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hall MA, Moshkov IE, Novikova GV, Mur LAJ, Smith AR. Ethylene signal perception and transduction: multiple paradigms? Biol Rev. 2001;76:103–128. doi: 10.1017/s1464793100005649. [DOI] [PubMed] [Google Scholar]
- 15.van Loon LC, Geraats BP, Linthorst HJ. Ethylene as a modulator of disease resistance in plants. Trends Plant Sci. 2006;11:184–191. doi: 10.1016/j.tplants.2006.02.005. [DOI] [PubMed] [Google Scholar]
- 16.Bent AF, Innes RW, Ecker JR, Staskawicz BJ. Disease Development in ethylene-insensitive Arabidopsis thaliana infected with virulent and avirulent Pseudomonas and Xanthomonas pathogens. Molec Plant-Microbe Interact. 1992;5:372–378. doi: 10.1094/mpmi-5-372. [DOI] [PubMed] [Google Scholar]
- 17.Mur LAJ, Brown IR, Darby RM, Bestwick CS, Bi YM, Mansfield JW, Draper J. A loss of resistance to avirulent bacterial pathogens in tobacco is associated with the attenuation of a salicylic acid-potentiated oxidative burst. Plant J. 2000;23:609–621. doi: 10.1046/j.1365-313x.2000.00825.x. [DOI] [PubMed] [Google Scholar]
- 18.Mur LAJ, Kenton P, Draper J. In planta measurements of oxidative bursts elicited by avirulent and virulent bacterial pathogens suggests that H2O2 is insufficient to elicit cell death in tobacco. Plant Cell Environm. 2005;28:548–561. [Google Scholar]