Abstract
Survivin is a member of the chromosome passenger complex, which plays an important role in chromosome alignment, separation, and cytokinesis. Although survivin is required for the proliferation and survival of hematopoietic stem and progenitor cells, the extent to which it is necessary for endomitosis of megakaryocytes remains controversial. To determine whether survivin is required for polyploidization, we analyzed mice with a megakaryocyte-specific deletion. PF4-Cre/survivinfl/fl mice harbored normal platelet counts with megakaryocytes that reached ploidy states comparable with those of control littermates. The CD41+ cells within these animals showed little excision but increased annexin V staining, implying that survivin is required for survival of megakaryocyte progenitors in vivo. In contrast, megakaryocytes in which survivin was excised ex vivo showed robust excision and an increased degree of polyploidization. These results demonstrate that survivin is necessary for survival of megakaryocyte progenitors, but is not required for polyploidization of committed megakaryocytes.
Introduction
During a proliferative cell cycle, the chromosome passenger complex (CPC), which includes survivin, aurora B, INCENP, and borealin, plays an essential role in facilitating chromosome alignment, separation, and cytokinesis.1 Cells in which the CPC is disrupted by loss of one of the component proteins show a block in mitosis, indicating that chromosomal passenger proteins are dependent on one another for proper location and function.2 In the absence of survivin, for example, cells display multiple defects, including centrosome dysregulation, multipolar spindles, polyploidy, and subsequent death by mitotic catastrophe.3–6
Megakaryocytes undergo repeated rounds of DNA replication without cytokinesis, resulting in ploidy states as high as 128N. Despite many advances in our knowledge of the endomitotic cell cycle, the precise mechanism by which these cells skip cytokinesis and re-enter G1 is still poorly understood.7 In particular, the role of survivin and the CPC remains controversial. We previously reported that survivin mRNA is present in murine megakaryocytes, but at a level 4-fold lower than that of erythroid cells.8 Ravid and colleagues who also studied mouse cells, which achieve a greater ploidy level in culture than human cells, were unable to detect survivin expression in polyploid murine megakaryocytes by immunofluorescence (Zhang et al9). Moreover, they found that localization of aurora B was abnormal in these cells. In contrast, Geddis and Kaushansky reported that polyploid human megakaryocytes demonstrated proper expression and localization of both aurora B and survivin.10 This difference between survivin protein expression in murine megakaryocytes may reflect the differential sensitivities of survivin antibodies.
To address the controversy of the role of survivin and the CPC in endomitosis, we generated a megakaryocyte-specific deletion of survivin. Our studies reveal that although survivin is required for survival of megakaryocyte progenitors in vivo, it is not required for endomitosis or survival of polyploid megakaryocytes. Thus, although megakaryocytes express components of the CPC, the CPC is dispensable for cells that skip cytokinesis and return to the G0/G1 phase of the cell cycle.
Methods
Animals
Survivin floxed mice were obtained from E. Conway and A. Winoto.11 PF4-Cre transgenic mice12 were obtained from R. Skoda and K. Kaushansky. Genotyping was performed by polymerase chain reaction (PCR) of tail DNA (supplemental Document 1, available on the Blood website; see the Supplemental Materials link at the top of the online article). Animal studies were approved by the Northwestern University IACUC.
Megakaryocyte cultures and retroviral transduction
Lineage-depleted bone marrow progenitors were differentiated into megakaryocytes by culturing in presence of TPO for 3 days as described.13 For the ex vivo excision with Cre, lineage-depleted bone marrow progenitors were infected by spinoculation on days 2 and 3 of expansion with retroviruses harboring MSCV-GFP or MSCV-Cre-GFP prior to differentiation. In some experiments, megakaryocytes were isolated following differentiation on a 1.5% to 3.0% discontinuous bovine serum albumin gradient.14
Flow cytometry
Surface marker expression, DNA content, and apoptosis were measured using a LSR II flow cytometer (BD Biosciences) and data were analyzed with FlowJo software (TreeStar). Details are provided in supplemental Document 1.
Results and discussion
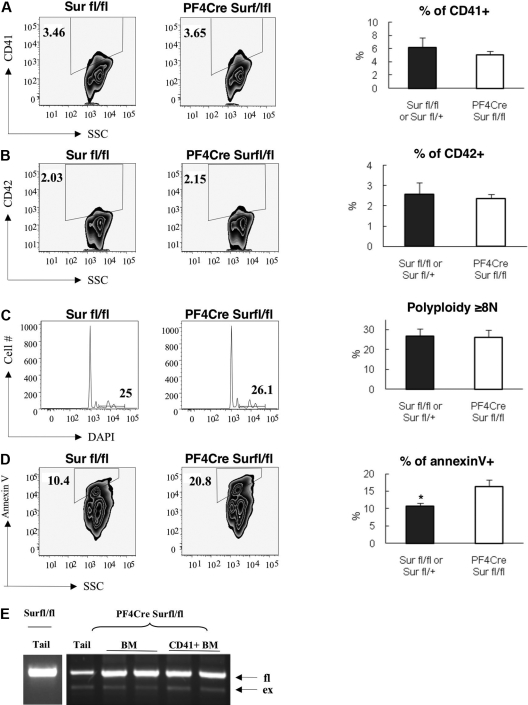
To explore the role of survivin in megakaryopoiesis, we crossed survivin floxed mice (Surfl/fl) to PF4-Cre transgenic mice, which have been used successfully by other investigators to assess megakaryocyte and platelet phenotypes.12,15 Compound mutant animals appeared grossly normal and harbored normal platelet counts (supplemental Table 1). Compared with Surfl/+ and Surfl/fl littermate controls, PF4-Cre/Surfl/fl mice showed no differences in CD41 and CD42 expression or polyploidization of bone marrow megakaryocytes (Figure 1A-C). Since we previously showed that survivin is required for survival of hematopoietic stem and progenitor cells,16 we assayed for apoptosis in the bone marrow compartment. Flow cytometry revealed that there was a significant increase in apoptosis in the CD41+ population in the deleted mice in comparison with control animals (Figure 1D). PCR of DNA extracted from PF4-Cre/Surfl/fl mice revealed that there was very little excision of survivin in bone marrow or in the CD41+ fraction (Figure 1E; averages of 7.2% and 8.4%, respectively). This lack of robust excision coupled with increased apoptosis suggests that megakaryocyte progenitors without survivin undergo apoptosis. Consistent with an underlying defect in survival of megakaryocyte progenitors, PF4-Cre/Surfl/fl mice displayed a reduction in the rebound of platelet counts following antibody-induced thrombocytopenia (supplemental Figure 1).
Figure 1.
Effect of tissue-specific deletion of survivin on polyploidization and differentiation of bone marrow megakaryocytes. Bone marrow cells from control (Surfl/fl and Surfl/+ without Cre) and PF4-Cre/Surfl/fl mice were collected and stained with FITC-labeled anti-CD41, PE-labeled anti-CD42, DAPI, and APC-labeled annexin V and then analyzed by flow cytometry. (A) CD41 expression, (B) CD42 expression, (C) DNA content, and (D) annexin V staining are shown. Representative flow and bar graphs depicting average ± SEM of 5 animals per group are shown. *P = .024. Note that DNA content and apoptosis are shown for only the CD41+ population. (E) Deletion of the floxed alleles was monitored by multiplex PCR using 3 primers to amplify the floxed (fl) and excised (ex) alleles. PCR of tail, bone marrow cells (BM), and fluorescence-activated cell sorting (FACS) purified CD41+ megakaryocytes (CD41+ BM) from 2 representative examples are shown.
The PF4 promoter is up-regulated by thrombopoietin signaling.17 To stimulate PF4-Cre expression and subsequent excision of survivin, we cultured PF4-Cre/Surfl/fl and control bone marrow cells ex vivo with TPO. Under these conditions, deletion of survivin did not lead to changes in the generation of CD41+ and CD42+ cells, or in the extent of polyploidization or apoptosis of CD41+ cells (supplemental Figure 2A-D). Furthermore, the polyploidization of BSA-gradient–purified PF4-Cre/Surfl/fl megakaryocytes was identical to that of Surfl/+ megakaryocytes (supplemental Figure 2E). PCR analysis revealed that excision in megakaryocytes was much greater in vitro, with nearly 70% excision in the BSA-gradient fraction (supplemental Figure 2F lanes 7-8) compared with the less than 10% excision seen in the CD41 population from bone marrow (Figure 1E). The fact that annexin V staining and megakaryocyte ploidy were unaffected, despite the robust deletion of survivin, indicates that survivin is dispensable for both endomitosis and survival of megakaryocytes in vitro.
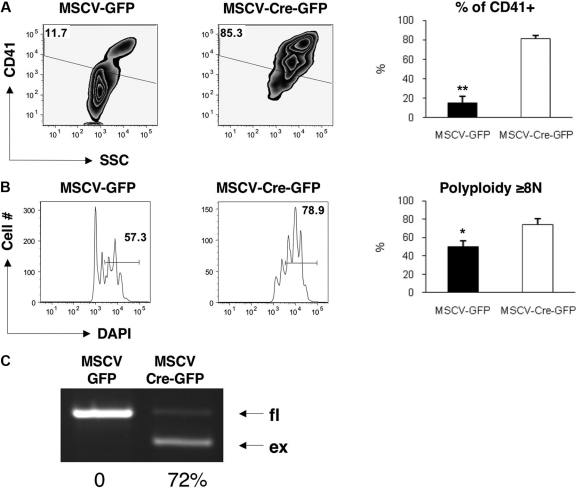
We next infected Surfl/fl or wild-type bone marrow cells with retroviruses expressing either Cre with GFP or GFP alone. The introduction of Cre into Surfl/fl cells led to 2 remarkable phenotypes. First, the proportion of CD41+ cells generated ex vivo was dramatically increased in Surfl/fl cells infected with MSCV-Cre-GFP compared with MSCV-GFP alone (Figure 2A). This difference is likely due to a combination of the death of nonmegakaryocytic cells in the culture and the persistence of survivin-deficient megakaryocytes. Second, the polyploidization of the CD41+ population was significantly elevated in megakaryocytes expressing Cre (Figure 2B). PCR analysis revealed that the extent of deletion within the infected bone marrow was more than 70% (Figure 2C). These results are consistent with those obtained from in vitro culture of PF4-Cre/Surfl/fl bone marrow and serve as further evidence that survivin is not required for the endomitotic cell cycle. Finally, we examined mitosis in GFP or Cre-GFP transduced Surfl/fl cells by confocal microscopy of α-tubulin– and DAPI-stained cells (supplemental Figure 3). We did not detect any differences with respect to chromosome separation in high ploidy cells in the presence of Cre recombinase. In contrast, we observed mitotic defects in low ploidy Cre transduced cells. These findings underscore the requirement for survivin in dividing cells and further suggest that endomitosis proceeds in a grossly similar manner in survivin-deleted cells.
Figure 2.
Ex vivo deletion of survivin in mouse bone marrow cells leads to increased polyploidization of megakaryocytes. Lineage-depleted bone marrow cells from Surfl/fl mice were infected with MSCV-GFP or MSCV-Cre-GFP and cultured with TPO (20 ng/mL) for 3 days. Cells were stained with PE-labeled anti-CD41 and DAPI and analyzed by flow cytometry. CD41 expression (A) and DNA content (B) of the GFP+ population are shown. Representative flow and bar graphs depicting the average ± SEM for 5 to 6 mice per group are shown. *P = .003; **P = .001 (C). Deletion of the floxed region was monitored by multiplex PCR. PCR of DNA extracted from GFP+ sorted MSCV-GFP or MSCV-Cre-GFP transduced cells is shown. Percentage excision (average of 2 experiments) is indicated below the blot.
Why are megakaryocytes less dependent on survivin than other cell types? Loss of survivin in proliferating cells is associated with chromosome misalignment, aberrant separation, failed cytokinesis, and cell death.3–6 Indeed, our studies showed that PF4-Cre–mediated deletion of survivin in vivo led to increased apoptosis of the CD41 population. These results are consistent with a requirement for survivin in proliferating megakaryocyte progenitors. In contrast, it appears that the unique ability of committed megakaryocytes to skip cytokinesis and directly enter G1 spares the cells from the injuries associated with survivin deletion. Along with not requiring the chromosome passenger complex, it is likely that megakaryocytes also tolerate decreased activity of the spindle checkpoint, as mice heterozygous for BubR1, a spindle assembly checkpoint protein, also show a significant increase in megakaryocyte polyploidy.18
Recent studies suggest that megakaryocyte polyploidization is associated with a relatively late failure in mitosis, including the lack of activation of RhoA and lack of accumulation of nonmuscle myosin IIA in the contractile ring19,20 rather than a failure of the CPC to properly localize at the midzone. Although the issue of whether survivin is expressed and properly localized in polyploid megakaryocytes remains controversial, our studies show that survivin is not essential for endomitosis or the survival of polyploid megakaryocytes. Our results also imply that the expression of Cre in the PF4-Cre strain occurs in a proliferating progenitor and is not restricted to terminally differentiating megakaryocytes. Thus, deletion of a gene that is required for survival of megakaryocyte progenitors, such as survivin, leads to selection of cells that have escaped excision. Such selective pressure was not observed for deletion of integrinβ1 or FAK, 2 genes that are not required for megakaryocyte survival.12,15
Acknowledgments
The authors thank Ed Conway and Astar Winoto for the survivin floxed mice and Radek Skoda for the PF4-Cre mice.
This work was supported by the National Institute of Diabetes and Digestive and Kidney Diseases (Bethesda, MD; DK074693). J.D.C. is a scholar of the Leukemia & Lymphoma Society (White Plains, NY).
Footnotes
An Inside Blood analysis of this article appears at the front of this issue.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: Q.W. designed and performed the experiments, interpreted the results, and contributed to writing the paper; J.D.C. designed the experiments, interpreted the results, and contributed to writing the paper; and C.L., Z.H., A.L.R., S.S., and J.D.L. assisted with performing the experiments and interpreting the results.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: John D. Crispino, Division of Hematology/Oncology, Northwestern University, 303 E Superior St, Lurie 5-113, Chicago, IL 60611; e-mail: j-crispino@northwestern.edu.
References
- 1.Ruchaud S, Carmena M, Earnshaw WC. The chromosomal passenger complex: one for all and all for one. Cell. 2007;131:230–231. doi: 10.1016/j.cell.2007.10.002. [DOI] [PubMed] [Google Scholar]
- 2.Gassmann R, Carvalho A, Henzing AJ, et al. Borealin: a novel chromosomal passenger required for stability of the bipolar mitotic spindle. J Cell Biol. 2004;166:179–191. doi: 10.1083/jcb.200404001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Okada H, Mak TW. Pathways of apoptotic and non-apoptotic death in tumour cells. Nat Rev Cancer. 2004;4:592–603. doi: 10.1038/nrc1412. [DOI] [PubMed] [Google Scholar]
- 4.Altieri DC. The case for survivin as a regulator of microtubule dynamics and cell-death decisions. Curr Opin Cell Biol. 2006;18:609–615. doi: 10.1016/j.ceb.2006.08.015. [DOI] [PubMed] [Google Scholar]
- 5.Uren AG, Wong L, Pakusch M, et al. Survivin and the inner centromere protein INCENP show similar cell-cycle localization and gene knockout phenotype. Curr Biol. 2000;10:1319–1328. doi: 10.1016/s0960-9822(00)00769-7. [DOI] [PubMed] [Google Scholar]
- 6.Lens SM, Wolthuis RM, Klompmaker R, et al. Survivin is required for a sustained spindle checkpoint arrest in response to lack of tension. EMBO J. 2003;22:2934–2947. doi: 10.1093/emboj/cdg307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ravid K, Lu J, Zimmet JM, Jones MR. Roads to polyploidy: the megakaryocyte example. J Cell Physiol. 2002;190:7–20. doi: 10.1002/jcp.10035. [DOI] [PubMed] [Google Scholar]
- 8.Gurbuxani S, Xu Y, Keerthivasan G, Wickrema A, Crispino JD. Differential requirements for survivin in hematopoietic cell development. Proc Natl Acad Sci U S A. 2005;102:11480–11485. doi: 10.1073/pnas.0500303102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang Y, Nagata Y, Yu G, et al. Aberrant quantity and localization of Aurora-B/AIM-1 and survivin during megakaryocyte polyploidization and the consequences of Aurora-B/AIM-1-deregulated expression. Blood. 2004;103:3717–3726. doi: 10.1182/blood-2003-09-3365. [DOI] [PubMed] [Google Scholar]
- 10.Geddis AE, Kaushansky K. Megakaryocytes express functional Aurora-B kinase in endomitosis. Blood. 2004;104:1017–1024. doi: 10.1182/blood-2004-02-0419. [DOI] [PubMed] [Google Scholar]
- 11.Xing Z, Conway EM, Kang C, Winoto A. Essential role of survivin, an inhibitor of apoptosis protein, in T cell development, maturation, and homeostasis. J Exp Med. 2004;199:69–80. doi: 10.1084/jem.20031588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tiedt R, Schomber T, Hao-Shen H, Skoda RC. Pf4-Cre transgenic mice allow the generation of lineage-restricted gene knockouts for studying megakaryocyte and platelet function in vivo. Blood. 2007;109:1503–1506. doi: 10.1182/blood-2006-04-020362. [DOI] [PubMed] [Google Scholar]
- 13.Huang Z, Richmond TD, Muntean AG, Barber DL, Weiss MJ, Crispino JD. STAT1 promotes megakaryopoiesis downstream of GATA-1 in mice. J Clin Invest. 2007;117:3890–3899. doi: 10.1172/JCI33010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Drachman JG, Sabath DF, Fox NE, Kaushansky K. Thrombopoietin signal transduction in purified murine megakaryocytes. Blood. 1997;89:483–492. [PubMed] [Google Scholar]
- 15.Hitchcock IS, Fox NE, Prevost N, Sear K, Shattil SJ, Kaushansky K. Roles of focal adhesion kinase (FAK) in megakaryopoiesis and platelet function: studies using a megakaryocyte lineage specific FAK knockout. Blood. 2008;111:596–604. doi: 10.1182/blood-2007-05-089680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Leung CG, Xu Y, Mularski B, Liu H, Gurbuxani S, Crispino JD. Requirements for survivin in terminal differentiation of erythroid cells and maintenance of hematopoietic stem and progenitor cells. J Exp Med. 2007;204:1603–1611. doi: 10.1084/jem.20062395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Oda M, Haruta H, Nagao M, Nagata Y. Isolation and characterization of mouse homolog of the neutrophil activating peptide-2. Biochem Biophys Res Commun. 2002;290:865–868. doi: 10.1006/bbrc.2001.6279. [DOI] [PubMed] [Google Scholar]
- 18.Wang Q, Liu T, Fang Y, et al. BUBR1 deficiency results in abnormal megakaryopoiesis. Blood. 2004;103:1278–1285. doi: 10.1182/blood-2003-06-2158. [DOI] [PubMed] [Google Scholar]
- 19.Lordier L, Jalil A, Aurade F, et al. Megakaryocyte endomitosis is a failure of late cytokinesis related to defects in the contractile ring and Rho/Rock signaling. Blood. 2008;112:3164–3174. doi: 10.1182/blood-2008-03-144956. [DOI] [PubMed] [Google Scholar]
- 20.Geddis AE, Kaushansky K. Endomitotic megakaryocytes form a midzone in anaphase but have a deficiency in cleavage furrow formation. Cell Cycle. 2006;5:538–545. doi: 10.4161/cc.5.5.2537. [DOI] [PubMed] [Google Scholar]