Abstract
TGF-β1 regulates a variety of cellular responses that are dependent on developmental stage and the origins of the cell or tissue. In mature tissues, and especially in tissues of epithelial origin, TGF-β1 is generally considered to be a growth inhibitor that may also promote apoptosis. The ameloblast cells of the enamel organ epithelium are adjacent to and are responsible for the developing enamel layer on unerupted teeth. Once the enamel layer reaches its full thickness, the tall columnar secretory stage ameloblasts shorten and a portion of these maturation stage ameloblasts become apoptotic. Here we ask if TGF-β1 plays a role in apoptosis of the maturation stage ameloblasts. We demonstrate in vitro that ameloblast lineage cells (ALC) are highly susceptible to TGF-β1-mediated growth arrest and are prone to TGF-β1-mediated cell death/apoptosis. We also demonstrate in vivo that TGF-β1 is expressed in the maturation stage enamel organ at significantly higher levels compared to the earlier secretory stage. This increased TGF-β1 expression correlates with an increase in enamel organ immediate-early stress response gene expression and with a decrease in the anti-apoptotic Bcl2/Bax expression ratio. We conclude that TGF-β1 may play an important role in ameloblast apoptosis during the maturation stage of enamel development.
Keywords: TGF-β1, Enamel, Ameloblast, Apoptosis, ALC cells
Introduction
Enamel development progresses through defined stages that can be observed by the changing morphology of the enamel organ that covers the developing tooth. The ameloblasts of the enamel organ are adjacent to the developing enamel and are responsible for organizing and later degrading the extracellular organic enamel matrix so that the enamel can mature into its highly mineralized form. Mature enamel is the hardest substance in the body. Its hardness is intermediate between that of iron and carbon steel, but has higher elasticity (1).
As ameloblasts proceed through the stages of enamel development, the enamel matures from a soft cheese-like substance into its final hardened form. Pre-secretory ameloblasts stop dividing and enlarge into tall columnar secretory ameloblasts that align more or less vertically to the adjacent enamel and are responsible for secreting enamel matrix proteins. During the maturation stage, the ameloblasts become shorter and assist in removal of proteins from the matrix as the enamel hardens (2, 3). Once the enamel is fully mature, ameloblasts regress and become part of the reduced enamel organ that covers and protects the completed enamel surface until the tooth erupts (4). It is during the transition stage, when the ameloblasts shorten from tall secretory cells to short maturation stage cells that the ameloblasts start to undergo apoptosis. Approximately 25% of ameloblasts are lost during the short transition stage and approximately another 25% of ameloblasts are lost throughout the remainder of the maturation stage (5).
Previously, TGF-β1 and/or Smad expression was demonstrated in tissues responsible for tooth and dental enamel development (6–16). Active TGF-β has been characterized as a 25 kDa dimer in which the two polypeptides interact via a disulfide bond and hydrophobic interactions. TGF-β1 initiates signaling by interacting with and complexing two receptor serine/threonine kinases referred to as the type I and type II receptors. This binding initiates sequential kinase activity that results in receptor-regulated Smad phosphorylation/activation. The activated Smad proteins may accumulate in the nucleus where they may collaborate with other transcription factors to regulate gene transcription [reviewed in (17, 18)]. TGF-β1 mediated gene transcription is temporally and spatially regulated and is dependent on the stage of development and on the developmental origins of the affected cell type. Transcriptome analyses have demonstrated that TGF-β1 may lead to the activation or repression of several hundred genes in a given cell type (19–24). A unifying factor is that in general, TGF-β1 is considered to promote growth inhibition. However, genetic evidence from gene ablation studies of TGF-β1 signaling components does not indicate a role for TGF-β1 as a growth inhibitor in early embryogenesis. It is usually later, after the tissues have matured, that TGF-β1 will induce growth arrest and/or cell death (18).
Although several studies have demonstrated TGF-β1 and/or Smad expression in enamel organ cells during dental enamel development, very few of these studies have examined TGF-β1 expression during the maturation stage of enamel formation. One recent study demonstrated that over-expression Smad7, a potent TGF-β1 inhibitor, caused tooth and enamel malformations that appeared to occur during the latter stages of enamel development (13). Another study demonstrated that over-expression of TGF-β1 in mouse teeth caused ameloblasts to detach from forming enamel and to cause the abnormal deposition of maturation stage mineral as “amorphous structures” (10). Even so, few studies have examined TGF-β1 expression at the later stages of enamel formation and no prior study has rigorously compared TGF-β1 expression levels between the secretory and maturation stages of enamel development.
Here we ask if maturation stage ameloblasts, relative to secretory stage ameloblasts, express increased levels of TGF-β1 that may mediate ameloblast apoptosis during the maturation stage of enamel development. Furthermore, we ask if TGF-β1 expression is associated with an increase in stress-response gene expression that may play a role in apoptotic events associated with maturation stage ameloblasts.
Material and methods
Animals
The Animal Care Committee of The Forsyth Institute approved the protocol for the handling, care, and usage of amimals.
Cell growth assay
The mouse ameloblast-lineage cell line (ALC) was cultured in DMEM (Invitrogen, Carlsbad, CA USA) supplemented with 10% fetal bovine serum (FBS), 50 units/ml penicillin, and 50µg/ml streptomycin. MTT assays (Sigma, St. Louis, MO) were performed to assess cell proliferation. MTT is a tetrazolium salt that is turned into a colored dye by mitochondrial dehydrogenase enzymes. The amount of colored dye produced is proportional to the number of living cells. ALC cells were plated in 96-well plates for18 h. TGF-β1 (0.00, 0.10, 0.50, 1.00, or 5.00 ng/ml) was then added to the respective wells. After 24 h, cell growth was determined by measuring MTT reductase activity. Briefly, MTT (0.5 mg/ml final concentration) was added and the resulting absorbance was measured after 3.5 h at 550 nm on a microplate reader (BMG Labtech, POLARstar Optima, Durham, North Carolina). Six wells were analyzed and the mean value was calculated for each TGF-β1 concentration. Experiments were performed in triplicate and were repeated three times.
Cell survival assay
ALC cells in DMEM with 2% FBS were plated at a density of 1000 cells in T-25 cm2 flasks for 18 h and were exposed to concentrations of TGF-β1 from between 0 and 1 ng/ml for a period of 24 h. Cells were washed with PBS and allowed to grow in medium with 2% FBS for approximately 12–14 days with medium change every 3 days. The resulting colonies were stained with 0.5% methylene blue in 50% methanol and counted. Percent cell survival was then calculated (25, 26).
Immunohistochemistry
Immunohistochemical methods were used to identify TGF-β1, Smad2/3, and p-c-Jun in ameloblasts of the continuously erupting mouse incisor enamel organ. Mouse incisors were formalin-fixed, decalcified in a solution of 20% sodium citrate 4% formic acid, paraffin-embedded, and sectioned. Sections were incubated in blocking agent (10% goat serum in PBS) for 1 h, overnight in antisera that detects TGF-β1 (LifeSpan Biosciences, Seattle, WA USA) or Smad2/3 (Santa Cruz Biotechnology. Santa Cruz, CA) or p-c-Jun (Santa Cruz), in peroxidase-conjugated antibody (Vectastain Elite Reagent, Vector Laboratories, Burlingame, CA, USA). Staining was visualized by incubation in Sigma Fast DAB substrate, with counterstaing in 0.1% FastGreen for 2 min. Sections were then examined by light microscopy for the presence of each protein.
Quantitative real-time polymerase chain reaction (qPCR)
Enamel organs were dissected from mandibular first molars from 4 day-old mice (secretory stage) or from 11 day-old mice (maturation stage). Six or seven mice were used per stage per experiment. Total enamel organ RNA was extracted by use of Trizol (Invitrogen, Carlsbad, CA USA) and converted to cDNA by SuperScript III First-Strand Synthesis System (Invitrogen Carlsbad, CA USA) for quantitative real-time PCR (qPCR) analysis. Primer sequence for expression analyses are provided in Table 1. The PCR temperature profile was 3 min 95 °C initial melt then; 20 s 95 °C, 30 s 60–66 °C for 45 cycles then 30 s 95 °C, for 1 cycle; 1 min 55 degrees C followed by stepwise temperature increases from 55 °C to 95 °C to generate the melt curve. Standard curves were generated with each primer set by use of untreated control cDNA preparations and a 10-fold dilution series ranging from 100 ng/ml to 100 pg/ml. PCR efficiencies and relative expression levels of the selected genes as a function of internal reference control gene (eEF1α1) expression were calculated as previously described (27).
Table 1.
qPCR primers for gene expression analysis
| Primer | GenBank acc. no. | Sequence (5’-3’) |
|---|---|---|
| Bax | NM_007527.3 | f: CCGGCGAATTGGAGATGAACTG |
| r: AGCTGCCACCCGGAAGAAGACCT | ||
| Bcl2 | NM_009741.3 | f: TGTCACAGAGGGGCTACGAGT |
| r: TCAGGCTGGAAGGAGAAGATG | ||
| eEF1α1 | NM_010106.2 | f: ATTCCGGCAAGTCCACCACAA |
| r: CATCTCAGCAGCCTCCTTCTCAAAC | ||
| Egr-1 | NM_007913.5 | f: AGGGGAGCCGAGCGAACA |
| r: GAGAAGCGGCCAGTATAGGTGATG | ||
| c-Fos | NM_010234.2 | f: AGCAAAAACAAAACAAAACAAAACAAAA |
| r: CTAAGGAGAAAGAGAAAAGAGACACAGACC | ||
| c-Jun | NM_010591.2 | f: CGACGTCGGGCTGCTCAAG |
| r: GGTGGGGGTCGGTGTAGTGGT | ||
| c-Myc | NM_010849.4 | f: AGACACCGCCCACCACCAG |
| r: AGCCCGACTCCGACCTCTTG | ||
| TGF-β1 | NM_011577.1 | f: AGGACCTGGGTTGGAAGTGGAT |
| r: AAGCGCCCGGGTTGTGTT |
Statistical analyses
Statistical analyses were performed using Prism software (GraphPad Software version 5.0, La Jolla, CA, USA). Statistical significance was evaluated by performing a non-parametric analysis of variance (ANOVA) followed by Bonferroni’s post test.
Results
Assessment of ameloblast lineage cell growth arrest and cell death after TGF-β1 treatment
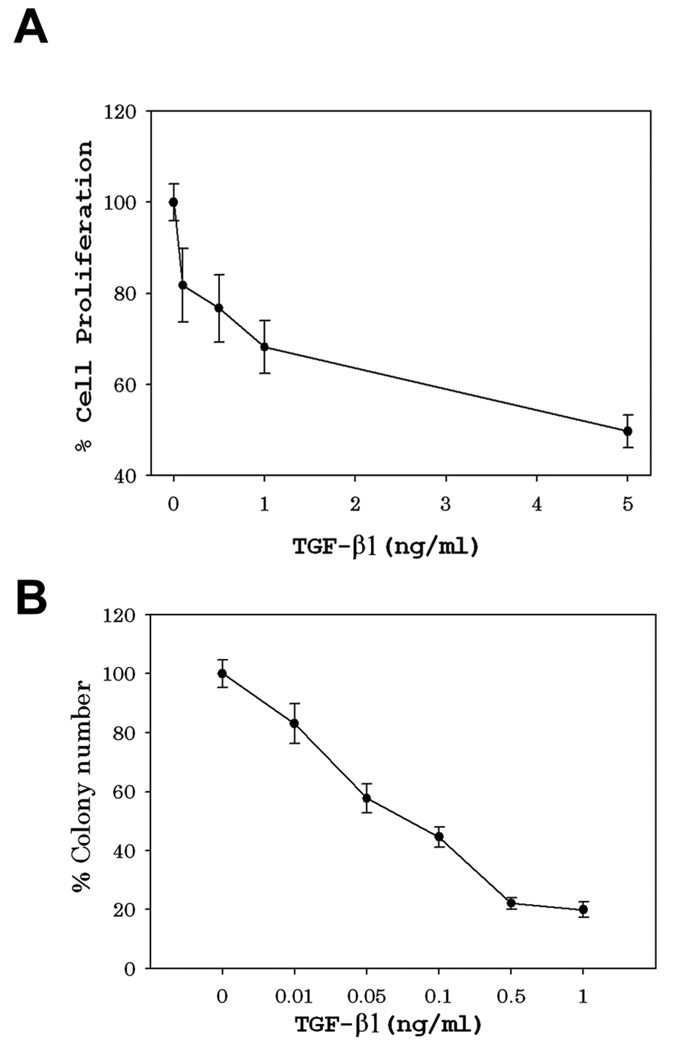
We initially used an in vitro experimental model to determine if ameloblasts are sensitive to the effects of TGF-β1 The ameloblast lineage cell (ALC) line is a spontaneously immortalized epithelial cell line from mouse enamel organ that was previously demonstrated to have alkaline-phosphatase activity and was shown to form calcified nodules in long-term culture (28). It therefore maintains several properties present in normal ameloblasts cells. We utilized the ALC cells to determine if they were sensitive to TGF-β1 treatment.
ALC cells were treated in triplicate with incremental doses of TGF-β1 for 24 h and were then assessed for cell proliferation by use of the MTT assay. As can be observed in Figure 1A, ALC cells were very sensitive to TGF-β1-mediated growth arrest. Cell proliferation was significantly inhibited by treatment with as little as 0.05 ng/ml TGF-β1 and doses above that level resulted in proportionately less cell growth (Fig. 1A). We next asked if TGF-β1 treatment caused ALC cell death. For these experiments ALC cells were again treated with incremental doses of TGF-β1 for 24 h and were then plated at limiting dilutions into small culture flasks. After 12–14 days the number of colonies that grew in each flask were quantified. A decrease in the number of TGF-β1-treated cell colonies relative to the untreated control served as an indication of cell death. Figure 1B demonstrates that TGF-β1 treatment caused a significant amount of ALC cell death even at the lowest dose tested (0.01 ng/ml) with higher doses eliciting progressively increased cell death levels.
Fig. 1.
Antiproliferative and toxic effects of TGF-β treatment on ALC cells. (A) To assess cell proliferation, ALC cells were seeded into 96 well plates and treated for 24 hr with the indicated concentrations of TGF-β. Reduction of MTT to an insoluble formazan dye by mitochondrial enzymes was quantified for each well by OD550 measurements and the results were used to calculate percent cell proliferation (treated OD550/untreated OD550) X 100. Error bars represent the standard error of the mean. (B) To assess cell survival, limiting dilutions of ALC cells were seeded into culture flasks, allowed to adhere for 18 hr, and were treated for 24 hr with the indicated concentrations of TGF-β. After 12–14 days, the resulting colonies were stained, counted, and percent cell survival was calculated (number treated/untreated colonies) x100. Error bars represent the standard error of the mean.
Quantification of Bax/Bcl2 ratios in TGF-β1 treated ALC cells
To determine if the TGF-β1-mediated cell death response in ALC cells was due to apoptosis, we quantified the level of the pro-apoptotic Bax transcripts versus the anti-apoptotic Bcl2 transcripts before and after 24 h TGF-β1 (1 ng/ml) treatment. qPCR with eEF1α1 as the internal reference control mRNA was performed to generate the results presented in Figure 2. The TGF-β1 treatment did significantly (p< 0.01) increase the Bax/Bcl2 ratio indicating that the TGF-β1-mediated ALC cell death was a result of apoptotic events (Fig. 2). Since ALC cells are a spontaneously immortalized cell line, results generated with these cells are not necessarily representative of an in vivo ameloblast response. We, therefore, characterized TGF-β1 and stress response gene expression in mouse secretory and maturation stage enamel organs.
Fig. 2.
Quantification by qPCR of Bax/Bcl2 expression in ALC cells treated with TGF-β for 24 hr. Bax or Bcl2 expression was determined and each were normalized to the expression levels of the internal reference control gene (eEF1α1). The graph shows the Bax to Bcl2 expression ratio with and without 24 hr of 1 ng/ml TGF-β treatment. Six wells were assayed for each experimental treatment and error bars represent the standard error. *, indicates statistical significance (p< 0.01).
Immunohistochemical assessment of TGF-β1 expression in mouse incisors
Because it was demonstrated previously that ameloblasts undergo apoptosis during the transition through maturation stages of enamel development, we wanted to confirm that TGF-β1 is expressed during these developmental stages and we also asked if we could observe higher TGF-β1 expression levels at these stages relative to the earlier secretory stage of enamel development. We performed immunohistochemical experiments on adult demineralized mouse incisors to identify the location of the TGF-β1 protein (Fig. 3). In general, the tooth tissues displayed diffuse staining. However, it did appear that a greater amount of staining for TGF-β1 was present within the cytoplasm of the maturation stage ameloblasts (Fig. 3D) than was present within the cytoplasm of secretory stage ameloblasts (Fig. 3C).
Fig. 3.
Identification of TGF-β in ameloblast cells of the enamel organ as determined by immunohistochemistry. (A) Negative control that lacked the primary antisera treatment. Note that some background staining can be observed above the ameloblasts (am). (B) Staining for TGF-β1 in an adult mouse mandibular incisor [20X magnification]. (C) Magnified image of the indicated secretory stage ameloblasts from panel B. (D) Magnified image of the indicated maturation stage ameloblasts from panel B. Note that staining appeared to be enhanced for TGF-β1 in the ameloblast cytoplasm at the maturation stage as compared to the secretory stage of enamel development. Scale bars represent 120 µm.
Quantification of TGF-β1 expression in mouse enamel organs as a function of developmental stage
To quantify any difference in TGF-β1 abundance, we performed qPCR analysis for TGF-β1 on mouse enamel organ mRNA that was isolated from first molars of 4 day-old mice (predominately secretory stage) and 11 day-old mice (predominately maturation stage). qPCR of TGF-β1 mRNA isolated from kidney epithelia served as the negative control. The amount of TGF-β1 transcripts in the 11 day-old maturation stage enamel organ was almost twice (p< 0.01) that observed in the 4 day-old secretory stage enamel organ (Fig. 4A). By contrast, no difference in TGF-β1 expression was observed between the 4 and 11 day-old kidney (Fig. 4B). Therefore, the ameloblast apoptosis observed in maturation stage of enamel development is consistent with increased levels of TGF-β1 expression.
Fig. 4.
Quantification by qPCR of TGF-β1 expression in mouse enamel organ during the secretory and maturation stages of enamel development. (A) Total RNA from first molar enamel organs was extracted from 4 day-old mice (secretory stage) or was extracted from 11 day-old mice (maturation stage) and was assessed for TGF-β expression levels. (B) Total RNA was also extracted from the kidney of 4 and 11 day-old mice. The kidney served as an epithelial derived negative control for the enamel organ results. Each expression analysis result was normalized to the expression level of the internal reference control gene (eEF1α1). Six different samples were assayed for each tissue and error bars represent the standard error. *, indicates statistical significance (p< 0.01).
Quantification of immediate early stress response gene expression in mouse enamel organs as a function of developmental stage
Because TGF-β1 expression was increased during the maturation stage and because the ameloblasts undergo apoptosis during this stage, we asked if immediate early stress-response genes were also up-regulated from the secretory to maturation stages of development. qPCR expression analysis demonstrated that c-Jun (Fig. 5A), c-Fos (Fig. 5B), Egr-1 (Fig. 5C), but not c-Myc (Fig. 5D) were expressed at significantly higher levels (P< 0.01) during the maturation stage (11 day-old enamel organ) when compared to the secretory stage (4 day-old enamel organ) of enamel development. This suggests that maturation stage ameloblasts are under an increased level of stress as the enamel matures.
Fig. 5.
Quantification by qPCR of immediate early stress response gene expression in mouse enamel organ during the secretory and maturation stages of enamel development. Total RNA extracted from first molar enamel organs from 4 day-old mice (secretory stage) and 11 day-old mice (maturation stage) was assessed for gene expression. (A) c-Jun, (B) c-Fos, (C) Egr-1, (D) c-Myc. Each expression analysis result was normalized to the expression level of the internal reference control gene (eEF1α1). Six samples were assayed for each tissue for each gene and error bars represent the standard error. Note that c-Jun expression increased at the maturation stage to almost six times over that observed in the secretory stage of enamel development. *, indicates statistical significance (p< 0.01).
Identification of increased levels of p-Smad2/3 and p-c-Jun in maturation stage ameloblasts
Here we ask if TGF-β1 likely signals through the TGF-β /activin receptor pathway in the mouse enamel organ. We performed immunohistochemistry to determine if increased levels of Smad2 or Smad3 were present in maturation stage ameloblasts. Ameloblast staining dramatically increased in the maturation stage when compared to the staining observed in ameloblasts of the secretory stage, suggesting that TGF-β1 does signal through this receptor-mediated pathway (Fig. 6).
Fig. 6.
Identification of p-Smad2/3 and p-c-Jun in ameloblast cells of the enamel organ as determined by immunohistochemistry. (Top panels) Staining for p-Smad2/3 in an adult mouse mandibular incisor [40× magnification]. (Bottom panels) Staining for p-c-Jun in an adult mouse mandibular incisor [40× magnification]. Note that staining for Smad2/3 was dramatically increased in the maturation stage ameloblasts and that more p-c-jun nuclear staining was also present in the maturation stage ameloblasts when compared to secretory stage ameloblasts.
Smad3 was previously demonstrated to synergize with c-Jun at AP-1 sites within gene promoters to initiate transcriptional activation by TGF-β1 (29). Because c-Jun is an immediate early response gene that can play a role in several stress response pathways, we sought to confirm that active phosphorylated c-Jun (p-c-Jun) was present within maturation stage ameloblasts. p-c-Jun did locate primarily to the nucleus of manturation stage ameloblasts (Fig. 6) and therefore may synergize with Smad3 to activate transcription of selected genes including stress response genes.
Quantification of Bcl2/Bax ratios in mouse enamel organs as a function of developmental stage
Finally, we asked if TGF-β1 expression in the mouse enamel organ correlates positively with pro-apoptotic Bax expression and negatively with anti-apoptotic Bcl2 expression as occurred for the ALC cells (Fig. 2). Bcl2 expression was reduced significantly (p< 0.01) from the 4 to 11 day-old enamel organ (Fig. 7A) while the expression level of Bax was significantly increased (Fig. 7B). The ratio of enamel organ Bcl2 to Bax expression also displayed a highly significant reduction from the 4 to 11 day-old mice (Fig. 7C), but no significant difference was observed between these age groups in the kidney (Fig. 7D).
Fig. 7.
Quantification by qPCR of Bcl2 and Bax gene expression in mouse enamel organ during the secretory and maturation stages of enamel development. Total RNA extracted from first molar enamel organs from 4 day-old mice (secretory stage) and 11 day-old mice (maturation stage) was assessed by for gene expression. (A) Bcl2, (B) Bax, (C) Bcl2/Bax ratio, (D) The kidney served as an epithelial derived negative control for the enamel organ results for Bcl2/Bax. Each expression analysis result was normalized to the expression level of the internal reference control gene (eEF1α1). Six samples were assayed for each tissue for each gene and error bars represent the standard error. Note that that the greatly decreased ratio of Bcl2 expression to Bax expression in the maturation stage enamel organ favors apoptosis. *, indicates statistical significance (p<0.01).
Taken together, the results are consistent with a role for TGF-β1 in signaling through the TGF-β /activin pathway to induce immediate early stress response genes that lead to a reduction in Bcl2 expression and an induction of Bax expression. Thus, TGF-β1 may be responsible for the observed increase in apoptosis of ameloblasts during the maturation stage of enamel development.
Discussion
TGF-β regulates a wide variety of cellular processes that include cell growth, growth arrest, apoptosis, differentiation, migration and extracellular matrix production [reviewed in (18, 30)]. Here we demonstrate that treatment of the ameloblast lineage cell line with TGF-β1 significantly reduces cell growth and cell survival in a dose-dependent manner (Fig. 1). Furthermore, we show that TGF-β1 increases the Bax/Bcl2 ratio to favor apoptosis in ALC cells. Members of the Bcl2 family bind to Bax to prevent its activation and subsequent cell apoptosis. Bax normally resides in an inactive state within the cytoplasm, but when activated, it inserts into the mitochondrial outer membrane and forms pores that release cytochrome c and other pro-apoptotic factors into the cytoplasm. Therefore, the Bcl2/Bax ratio in a cell may determine its susceptibility to survival or apoptosis [reviewed in (17)]. Previously, it was demonstrated that a decrease in Bcl2 levels was observed in TGF-β1-mediated apoptosis in a variety of cell types including human lens epithelial cells (31), multipotent hematopoietic cell lines (32) and thecal/interstitial cells (33). In addition, TGF-β1-mediated induction of Bax was demonstrated in bovine epithelial BME-UV1 cells (34). Our results demonstrate that in addition to the above listed cell types, TGF-β1 treated ameloblast-lineage cells also display decreased Bcl2 expression and increased Bax expression.
To ascertain the relevance of the results generated in vitro to that which may occur in vivo, we performed TGF-β1 immunohistochemistry on mouse incisors (Fig. 3) and qPCR analysis for gene expression on mouse first molar enamel organs that were either predominantly in the secretory (4 day-old) or maturation (11 day-old) stages of enamel development (Fig. 4, Fig.5, Fig.7). Because a proportion of ameloblasts undergo apoptosis during the maturation stage (5, 35, 36), we asked if TGF-β1 expression was up-regulated during this stage. The TGF-β1 immunohistochemical results suggested it was up-regulated and the TGF-β1 qPCR analysis definitively demonstrated that the maturation stage enamel organ expressed more TGF-β1 than did secretory stage enamel organ (Fig. 4). Prior studies have demonstrated that TGF-β is expressed in the murine enamel organ (6, 13, 14, 16) and a recent study demonstrated that over-expression in epithelial tissues of Smad7, which negatively regulates TGF-β1 signaling, caused malformed tooth and enamel development (13). Our results are the first to use qPCR analysis to show that TGF-β1 expression becomes up-regulated during the maturation stage of enamel development when a proportion of ameloblasts become apoptotic.
Because prior studies have shown that exposure to TGF-β1 may induce the expression of immediate early stress response genes (20–22, 24, 34, 37–39), we sought to determine if selected immediate early genes were induced in the maturation stage enamel organ when compared to the expression levels observed in the secretory stage enamel organ. We found that the expression levels of c-Jun, c-Fos, and Egr-1 were significantly higher in the maturation stage. However, there was no significant difference in c-Myc expression (Fig. 5). c-Fos and c-Jun are members of the AP-1 transcription factor family. They bind to AP-1 sites and may also interact with Smads to activate transcription in response to TGF-β signaling (39). c-Jun was previously shown by immunohistochemical methods, to become more prevalent in rat maturation stage ameloblast nuclei when compared to the secretory stage (40) and analysis of Smad3 null mice found that Smad3 is required for enamel biomineralization (16). Thus, our data support and confirm these findings. Egr-1 is a zinc finger transcription factor early growth response gene. TGF-β1 was demonstrated to induce both protein and mRNA expression of Erg-1 in human skin fibroblasts (37). Interestingly, c-Myc is a proto-oncogene that promotes the cell cycle and simultaneously primes activation of the mitochondria-mediated apoptosis pathway (41). In general, TGF-β1 is an inhibitor of c-Myc (42–44). However, in specific instances, such as in dermal fibroblasts, TGF-β1 will induce c-Myc expression (24). Although we found a trend for increased expression of c-Myc in maturation versus secretory stage enamel organ, the trend was not significant. Taken together, the expression level of the assayed immediate early response genes is consistent with the increased expression of TGF-β1 in the maturation stage enamel organ. This suggests that c-Jun, c-Fos, and Egr-1, and perhaps c-Myc through its apoptosis activity, may play a role in the downstream events leading to maturation stage ameloblast apoptosis.
Lastly, we examined Bcl2 and Bax expression in mouse first molar secretory and maturation stage enamel organ. As for the in vitro TGF-β1 treated ALC cells (Fig. 2), we found that in vivo the maturation stage, with the up-regulated TGF-β1 expression levels (Fig. 4), also had increased expression of Bax and decreased expression of Bcl2 (Fig. 7). This is consistent with the observed increased levels of apoptosis in the maturation stage of enamel development (5, 35, 36). One prior study showed that Bax and Bcl2 are expressed in the developing human enamel organ (45). This study demonstrated that during early stages of enamel development Bcl2 levels predominated over Bax. However, the study did not assess Bax/Bcl2 levels in maturation stage enamel organ. Another study on developing rat enamel organs demonstrated, by TUNEL assays and immunohistochemistry, a positive correlation between Bax/Bcl2 abundance and ameloblast apoptosis (46). When Bax abundance was elevated relative to Bcl2, greater amounts of ameloblast apoptosis were observed. These data support our conclusion that TGF-β1 signaling, perhaps through Smad3 and immediate early stress response genes, increases the ratio of Bax to Bcl2 and plays a role in initiating apoptosis in maturation stage ameloblasts.
In conclusion, we have demonstrated by both in in vitro and, more importantly, in in vivo experimental models that TGF-β1 likely plays a role in ameloblast apoptosis during the maturation stage of enamel development.
Acknowledgment
This work was supported by National Institute of Dental and Craniofacial Research grant DE016276 (to JDB).
References
- 1.Cole AS, Eastoe J. Biochemistry and Oral Biology. London: Butterworth & Co. LTD; 1988. [Google Scholar]
- 2.Bartlett JD, Simmer JP. Proteinases in Developing Dental Enamel. Crit Rev Oral Biol Med. 1999;10:425–441. doi: 10.1177/10454411990100040101. [DOI] [PubMed] [Google Scholar]
- 3.Smith CE. Cellular and Chemical Events During Enamel Maturation. Crit Rev Oral Biol Med. 1998;9:128–161. doi: 10.1177/10454411980090020101. [DOI] [PubMed] [Google Scholar]
- 4.Kallenbach E. Fine Structure of Rat Incisor Enamel Organ During Late Pigmentation and Regression Stages. J Ultrastruct Res. 1970;30:38–63. doi: 10.1016/s0022-5320(70)90063-8. [DOI] [PubMed] [Google Scholar]
- 5.Smith CE, Warshawsky H. Quantitative Analysis of Cell Turnover in the Enamel Organ of the Rat Incisor. Evidence for Ameloblast Death Immediately After Enamel Matrix Secretion. Anat Rec. 1977;187:63–98. doi: 10.1002/ar.1091870106. [DOI] [PubMed] [Google Scholar]
- 6.D'souza RN, Happonen RP, Ritter NM, Butler WT. Temporal and Spatial Patterns of Transforming Growth Factor-Beta 1 Expression in Developing Rat Molars. Arch Oral Biol. 1990;35:957–965. doi: 10.1016/0003-9969(90)90015-3. [DOI] [PubMed] [Google Scholar]
- 7.D'Souza RN, Flanders K, Butler WT. Colocalization of TGF-Beta 1 and Extracellular Matrix Proteins During Rat Tooth Development. Proc Finn Dent Soc. 1992;88 Suppl 1:419–426. [PubMed] [Google Scholar]
- 8.D'Souza rn, Litz M. Analysis of Tooth Development in Mice Bearing a TGF-Beta 1 Null Mutation. Connect Tissue Res. 1995;32:41–46. doi: 10.3109/03008209509013704. [DOI] [PubMed] [Google Scholar]
- 9.Ferguson CA, Tucker AS, Heikinheimo K, Nomura M, Oh P, Li E, Sharpe PT. The Role of Effectors of the Activin Signalling Pathway, Activin Receptors IIA and IIB, and Smad2, in Patterning of Tooth Development. Development. 2001;128:4605–4613. doi: 10.1242/dev.128.22.4605. [DOI] [PubMed] [Google Scholar]
- 10.Haruyama N, Thyagarajan T, Skobe Z, Wright JT, Septier D, Sreenath TL, Goldberg M, Kulkarni AB. Overexpression of Transforming Growth Factor-Beta1 in Teeth Results in Detachment of Ameloblasts and Enamel Defects. Eur J Oral Sci. 2006;114 Suppl 1:30–34. doi: 10.1111/j.1600-0722.2006.00276.x. [DOI] [PubMed] [Google Scholar]
- 11.Ito Y, Sarkar P, Mi Q, Wu N, Bringas P, Jr., Liu Y, Reddy S, Maxson R, Deng C, Chai Y. Overexpression of Smad2 Reveals Its Concerted Action With Smad4 in Regulating TGF-Beta-Mediated Epidermal Homeostasis. Dev Biol. 2001;236:181–194. doi: 10.1006/dbio.2001.0332. [DOI] [PubMed] [Google Scholar]
- 12.Jepsen S, Schiltz P, Strong DD, Scharla SH, Snead ML, Finkelman RD. Transforming Growth Factor-Beta 1 MRNA in Neonatal Ovine Molars Visualized by in Situ Hybridization: Potential Role for the Stratum Intermedium. Arch Oral Biol. 1992;37:645–653. doi: 10.1016/0003-9969(92)90127-t. [DOI] [PubMed] [Google Scholar]
- 13.Klopcic B, Maass T, Meyer E, Lehr HA, Metzger D, Chambon P, Mann A, Blessing M. TGF-Beta Superfamily Signaling Is Essential for Tooth and Hair Morphogenesis and Differentiation. Eur J Cell Biol. 2007;86:781–799. doi: 10.1016/j.ejcb.2007.03.005. [DOI] [PubMed] [Google Scholar]
- 14.Vaahtokari A, Vainio S, Thesleff I. Associations Between Transforming Growth Factor Beta 1 RNA Expression and Epithelial-Mesenchymal Interactions During Tooth Morphogenesis. Development. 1991;113:985–994. doi: 10.1242/dev.113.3.985. [DOI] [PubMed] [Google Scholar]
- 15.Xu X, Jeong L, Han J, Ito Y, Bringas P, Jr., Chai Y. Developmental Expression of Smad1-7 Suggests Critical Function of TGF-Beta/BMP Signaling in Regulating Epithelial-Mesenchymal Interaction During Tooth Morphogenesis. Int J Dev Biol. 2003;47:31–39. [PubMed] [Google Scholar]
- 16.Yokozeki M, Afanador E, Nishi M, Kaneko K, Shimokawa H, Yokote K, Deng C, Tsuchida K, Sugino H, Moriyama K. Smad3 Is Required for Enamel Biomineralization. Biochem Biophys Res Commun. 2003;305:684–690. doi: 10.1016/s0006-291x(03)00806-4. [DOI] [PubMed] [Google Scholar]
- 17.Sanchez-capelo A. Dual Role for TGF-Beta1 in Apoptosis. Cytokine Growth Factor Rev. 2005;16:15–34. doi: 10.1016/j.cytogfr.2004.11.002. [DOI] [PubMed] [Google Scholar]
- 18.Siegel PM, Massague J. Cytostatic and Apoptotic Actions of TGF-Beta in Homeostasis and Cancer. Nat Rev Cancer. 2003;3:807–821. doi: 10.1038/nrc1208. [DOI] [PubMed] [Google Scholar]
- 19.Cao Y, Chen L, Zhang W, Liu Y, Papaconstantinou HT, Bush CR, Townsend CM, Jr., Thompson EA, Ko TC. Identification of Apoptotic Genes Mediating TGF-Beta/Smad3-Induced Cell Death in Intestinal Epithelial Cells Using a Genomic Approach. Am J Physiol Gastrointest Liver Physiol. 2007;292:G28–G38. doi: 10.1152/ajpgi.00437.2005. [DOI] [PubMed] [Google Scholar]
- 20.Coyle B, Freathy C, Gant TW, Roberts RA, Cain K. Characterization of the Transforming Growth Factor-Beta 1-Induced Apoptotic Transcriptome in FaO Hepatoma Cells. J Biol Chem. 2003;278:5920–5928. doi: 10.1074/jbc.M211300200. [DOI] [PubMed] [Google Scholar]
- 21.Hu G, Jain K, Hurle M. Revealing Transforming Growth Factor-Beta Signaling Transduction in Human Kidney by Gene Expression Data Mining. OMICS. 2005;9:266–280. doi: 10.1089/omi.2005.9.266. [DOI] [PubMed] [Google Scholar]
- 22.Levy L, Hill CS. Smad4 Dependency Defines Two Classes of Transforming Growth Factor {Beta} (TGF-{Beta}) Target Genes and Distinguishes TGF-{Beta}-Induced Epithelial-Mesenchymal Transition From Its Antiproliferative and Migratory Responses. Mol Cell Biol. 2005;25:8108–8125. doi: 10.1128/MCB.25.18.8108-8125.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ranganathan P, Agrawal A, Bhushan R, Chavalmane AK, Kalathur RK, Takahashi T, Kondaiah P. Expression Profiling of Genes Regulated by TGF-Beta: Differential Regulation in Normal and Tumour Cells. BMC Genomics. 2007;8:98. doi: 10.1186/1471-2164-8-98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Verrecchia F, Chu ML, Mauviel A. Identification of Novel TGF-Beta /Smad Gene Targets in Dermal Fibroblasts Using a Combined CDNA Microarray/Promoter Transactivation Approach. J Biol Chem. 2001;276:17058–17062. doi: 10.1074/jbc.M100754200. [DOI] [PubMed] [Google Scholar]
- 25.Bartlett JD, Luethy JD, Carlson SG, Sollott SJ, Holbrook NJ. Calcium Ionophore A23187 Induces Expression of the Growth Arrest and DNA Damage Inducible CCAAT/Enhancer-Binding Protein (C/EBP)-Related Gene, Gadd153. Ca2+ Increases Transcriptional Activity and mRNA Stability. J Biol Chem. 1992;267:20465–20470. [PubMed] [Google Scholar]
- 26.Bertrand R, Kerrigan D, Sarang M, Pommier Y. Cell Death Induced by Topoisomerase Inhibitors. Role of Calcium in Mammalian Cells. Biochem Pharmacol. 1991;42:77–85. doi: 10.1016/0006-2952(91)90683-v. [DOI] [PubMed] [Google Scholar]
- 27.Pfaffl MW. A New Mathematical Model for Relative Quantification in Real-Time RT-PCR. Nucleic Acids Res. 2001;29:e45. doi: 10.1093/nar/29.9.e45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nakata A, Kameda T, Nagai H, Ikegami K, Duan Y, Terada K, Sugiyama T. Establishment and Characterization of a Spontaneously Immortalized Mouse Ameloblast-Lineage Cell Line. Biochem Biophys Res Commun. 2003;308:834–839. doi: 10.1016/s0006-291x(03)01467-0. [DOI] [PubMed] [Google Scholar]
- 29.Qing J, Zhang Y, Derynck R. Structural and Functional Characterization of the Transforming Growth Factor-Beta -Induced Smad3/c-Jun Transcriptional Cooperativity. J Biol Chem. 2000;275:38802–38812. doi: 10.1074/jbc.M004731200. [DOI] [PubMed] [Google Scholar]
- 30.Rahimi RA, Leof EB. TGF-Beta Signaling: a Tale of Two Responses. J Cell Biochem. 2007;102:593–608. doi: 10.1002/jcb.21501. [DOI] [PubMed] [Google Scholar]
- 31.Lee JH, Wan XH, Song J, Kang JJ, Chung WS, Lee EH, Kim EK. TGF-Beta-Induced Apoptosis and Reduction of Bcl-2 in Human Lens Epithelial Cells in Vitro. Curr Eye Res. 2002;25:147–153. doi: 10.1076/ceyr.25.3.147.13475. [DOI] [PubMed] [Google Scholar]
- 32.Francis JM, Heyworth CM, Spooncer E, Pierce A, Dexter TM, Whetton AD. Transforming Growth Factor-Beta 1 Induces Apoptosis Independently of P53 and Selectively Reduces Expression of Bcl-2 in Multipotent Hematopoietic Cells. J Biol Chem. 2000;275:39137–39145. doi: 10.1074/jbc.M007212200. [DOI] [PubMed] [Google Scholar]
- 33.Foghi A, Teerds KJ, Van der DH, Moore NC, Dorrington J. Induction of Apoptosis in Thecal/Interstitial Cells: Action of Transforming Growth Factor (TGF) Alpha Plus TGF Beta on Bcl-2 and Interleukin-1 Beta-Converting Enzyme. J Endocrinol. 1998;157:489–494. doi: 10.1677/joe.0.1570489. [DOI] [PubMed] [Google Scholar]
- 34.Gajewska M, Motyl T. IGF-Binding Proteins Mediate TGF-Beta 1-Induced Apoptosis in Bovine Mammary Epithelial BME-UV1 Cells. Comp Biochem Physiol C Toxicol Pharmacol. 2004;139:65–75. doi: 10.1016/j.cca.2004.09.006. [DOI] [PubMed] [Google Scholar]
- 35.Kondo S, Tamura Y, Bawden JW, Tanase S. The Immunohistochemical Localization of Bax and Bcl-2 and Their Relation to Apoptosis During Amelogenesis in Developing Rat Molars. Arch Oral Biol. 2001;46:557–568. doi: 10.1016/s0003-9969(00)00139-4. [DOI] [PubMed] [Google Scholar]
- 36.Nishikawa S, Sasaki F. DNA Localization in Nuclear Fragments of Apoptotic Ameloblasts Using Anti-DNA Immunoelectron Microscopy: Programmed Cell Death of Ameloblasts. Histochem Cell Biol. 1995;104:151–159. doi: 10.1007/BF01451574. [DOI] [PubMed] [Google Scholar]
- 37.Chen SJ, Ning H, Ishida W, Sodin-semrl S, Takagawa S, Mori Y, Varga J. The Early-Immediate Gene EGR-1 Is Induced by Transforming Growth Factor-Beta and Mediates Stimulation of Collagen Gene Expression. J Biol Chem. 2006;281:21183–21197. doi: 10.1074/jbc.M603270200. [DOI] [PubMed] [Google Scholar]
- 38.Levine RA, Hopman T, Guo L, Chang MJ, Johnson N. Induction of Retinoblastoma Gene Expression During Terminal Growth Arrest of a Conditionally Immortalized Fetal Rat Lung Epithelial Cell Line and During Fetal Lung Maturation. Exp Cell Res. 1998;239:264–276. doi: 10.1006/excr.1997.3898. [DOI] [PubMed] [Google Scholar]
- 39.Zhang Y, Feng XH, Derynck R. Smad3 and Smad4 Cooperate With C-Jun/c-Fos to Mediate TGF-Beta-Induced Transcription. Nature. 1998;394:909–913. doi: 10.1038/29814. [DOI] [PubMed] [Google Scholar]
- 40.Nishikawa S. Localization of Transcription Factor AP-1 Family Proteins in Ameloblast Nuclei of the Rat Incisor. J Histochem Cytochem. 2000;48:1511–1520. doi: 10.1177/002215540004801108. [DOI] [PubMed] [Google Scholar]
- 41.Nieminen AI, Partanen JI, Klefstrom J. C-Myc Blazing a Trail of Death: Coupling of the Mitochondrial and Death Receptor Apoptosis Pathways by C-Myc. Cell Cycle. 2007;6:2464–2472. doi: 10.4161/cc.6.20.4917. [DOI] [PubMed] [Google Scholar]
- 42.Feng XH, Liang YY, Liang M, Zhai W, Lin X. Direct Interaction of C-Myc With Smad2 and Smad3 to Inhibit TGF-Beta-Mediated Induction of the CDK Inhibitor P15(Ink4B) Mol Cell. 2002;9:133–143. doi: 10.1016/s1097-2765(01)00430-0. [DOI] [PubMed] [Google Scholar]
- 43.Lim SK, Hoffmann FM. Smad4 Cooperates With Lymphoid Enhancer-Binding Factor 1/T Cell-Specific Factor to Increase C-Myc Expression in the Absence of TGF-Beta Signaling. Proc Natl Acad Sci U S A. 2006;103:18580–18585. doi: 10.1073/pnas.0604773103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pietenpol JA, Stein RW, Moran E, Yaciuk P, Schlegel R, Lyons RM, Pittelkow MR, Munger K, Howley PM, Moses HL. TGF-Beta 1 Inhibition of C-Myc Transcription and Growth in Keratinocytes Is Abrogated by Viral Transforming Proteins With PRB Binding Domains. Cell. 1990;61:777–785. doi: 10.1016/0092-8674(90)90188-k. [DOI] [PubMed] [Google Scholar]
- 45.Hatakeyama S, Tomichi N, Ohara-nemoto Y, Satoh M. The Immunohistochemical Localization of Fas and Fas Ligand in Jaw Bone and Tooth Germ of Human Fetuses. Calcif Tissue Int. 2000;66:330–337. doi: 10.1007/s002230010069. [DOI] [PubMed] [Google Scholar]
- 46.Kondo S, Tamura Y, Bawden JW, Tanase S. The Immunohistochemical Localization of Bax and Bcl-2 and Their Relation to Apoptosis During Amelogenesis in Developing Rat Molars. Arch Oral Biol. 2001;46:557–568. doi: 10.1016/s0003-9969(00)00139-4. [DOI] [PubMed] [Google Scholar]