Abstract
Introduction
Secondhand smoke (SHS) contains respiratory irritants and has the potential to adversely affect adults with chronic obstructive pulmonary disease (COPD), but few studies have evaluated the impact of SHS on COPD.
Methods
We used data from 72 nonsmoking participants in a cohort study of COPD. Urine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) was measured as an indicator of longer term SHS exposure, whereas urine cotinine was assessed as a measure of more recent exposure. The impact of SHS exposure on COPD-related health status was examined using multivariate linear regression (controlling for age, sex, race, educational attainment, and smoking history). Health status was measured using a validated COPD severity score, reported dyspnea, a standard health status measure (Short Form-12), and activity restriction.
Results
The urine NNAL-to-creatinine ratio (per interquartile increment) was associated with greater COPD severity (mean score increase 1.7 points; 95% CI 0.6–2.8; p = .0003). Higher urine NNAL was also related to greater dyspnea, poorer physical health status, and more restricted activity (p ≤ .05 in all cases). When considered simultaneously, longer term exposure (NNAL) had a greater negative impact on COPD status than shorter term exposure (cotinine).
Discussion
Urine NNAL can be used to estimate longer term SHS exposure and negatively affects a number of health outcomes among adults with COPD. Screening for and prevention of SHS exposure among persons with COPD may be beneficial.
Introduction
Secondhand smoke (SHS) contains potent respiratory irritants that are likely to adversely affect persons with obstructive lung disease. Evidence now implicates SHS exposure as a cause of asthma exacerbation among children and, to a lesser extent, adults (Eisner, Klein, et al., 2005; Eisner, Yelin, Katz, Earnest, & Blanc, 2002; Office of Environmental Health Hazard Assessment, California Environmental Protection Agency, 2005). In chronic obstructive pulmonary disease (COPD), there is emerging evidence that SHS exposure may have a negative impact on health status, but the limited available data are inadequate to support definite conclusions (Eisner et al., 2006; Garcia-Aymerich et al., 2003; Osman et al., 2007). Because COPD is a major cause of disability and mortality worldwide, the impact of SHS exposure on the disease has major public health ramifications.
Exposure measurement is a key challenge in studying the effects of SHS exposure. The most widely used biomarker of SHS exposure is cotinine, but it reflects exposure during only a recent 2- to 3-day period (Benowitz, 1999). Survey-based assessment tools can evaluate longer time intervals but are subject to inaccuracy in self-report and recall bias. In this analysis, we measured levels of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) in the urine of persons with COPD. NNAL, which is a metabolite of the tobacco-specific carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK), is highly specific for SHS exposure among nonsmokers (Anderson et al., 2003; Hecht, 2004, 2006; Hecht et al., 2001, 2006; Stark et al., 2007; Tulunay et al., 2005). Because NNAL is persistent in the body (elimination half-life of 45 days), it can serve as a biomarker for longer term SHS exposure (Hecht et al., 1999). In this analysis, we examined the impact of longer term SHS exposure, as measured by NNAL, on health outcomes in COPD.
Methods
Overview
We used data from a population-based cohort study of adults with COPD. The impact of shorter term SHS exposure, as measured by urine cotinine and nicotine badge, on COPD health outcomes has been previously reported for this cohort (Eisner et al., 2006). In the current analysis, longer term SHS exposure was assessed using urine NNAL levels, which reflect exposure for the 1- to 3-month period prior to health status assessment in the cohort. We therefore examined the impact of longer term SHS exposure on health status among persons with COPD. The study was approved by the University of California, San Francisco (UCSF), Committee on Human Research.
Definition of COPD
We used the standard epidemiological approach to define COPD based on subject report of a physician’s diagnosis of chronic bronchitis, emphysema, or COPD (Mannino, 2002; Mannino, Homa, Akinbami, Ford, & Redd, 2002; Sin, Stafinski, Ng, Bell, & Jacobs, 2002). During telephone interviews, subjects were asked whether they had ever received a physician’s diagnosis of any of several chronic respiratory conditions. Those who reported physician’s diagnoses of chronic bronchitis or emphysema were considered to have COPD, along with those who specifically reported a physician’s diagnosis of COPD. We included respondents with COPD who had concomitant asthma because they clinically resemble persons with COPD alone (Eisner, Yelin, Trupin, & Blanc, 2002). As reported previously, we validated the case definition of COPD using spirometry in a subgroup of 47 participants with COPD whose physicians provided spirometry reports (of 386 subjects) and have used this same definition in other reports from the cohort (Blanc et al., 2005; Eisner, Balmes, et al., 2005; Eisner et al., 2006; Eisner, Trupin, et al., 2005; Trupin et al., 2003).
Study recruitment
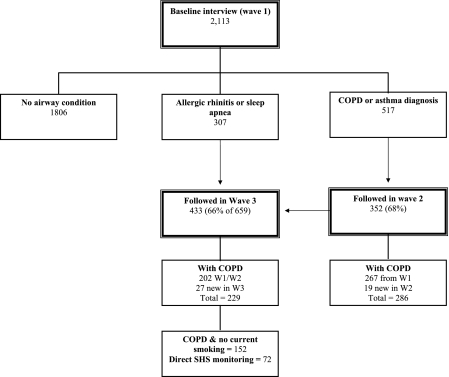
We used data from the third wave of a population-based, multiwave longitudinal cohort study of U.S. adults to elucidate the impact of SHS exposure on COPD health outcomes. Direct measures of SHS exposure were obtained in this wave. Initial recruitment was previously reported in detail (Eisner, Balmes, et al., 2005; Trupin et al., 2003). Briefly, a representative sample of 2,113 adults aged 55–75 years were initially recruited by random digit dialing among residents of the 48 contiguous U.S. states with random oversampling of geographic areas that had the highest published COPD mortality rates (Kim, 1998). The initial overall study participation rate was 53% among households with an eligible respondent present. Details about follow-up of the three survey waves have been previously reported (Blanc et al., 2004; Eisner et al., 2006).
The current analysis was restricted to subjects with COPD who completed interview Wave 3, reported no current smoking, provided a urine sample by a mail-in-protocol, and had urine cotinine levels that were less than 50 ng/ml (a level that is consistent with direct personal smoking; Jarvis, Tunstall-Pedoe, Feyerabend, Vesey, & Saloojee, 1987). In sum, 72 subjects completed direct SHS monitoring with urine NNAL measurements (Figure 1). This compares with 77 subjects from our earlier report of urine cotinine in COPD subjects. Inadequate urine volume was available for NNAL measurement in four subjects, and one subject was excluded for an NNAL level that was more compatible with direct personal smoking (153 pg/ml). There were no statistical differences in sociodemographic characteristics or COPD severity between COPD subjects in whom urine NNAL measurements were available versus all others (p > .15 in all cases).
Figure 1.
Details of sampling and recruitment for baseline through Wave 3. Wave 2 attempted to recruit all subjects who indicated a diagnosis of asthma or chronic obstructive pulmonary disease (COPD) at baseline. Wave 3 attempted to recruit all subjects followed in Wave 2 plus those who indicated a diagnosis of allergic rhinitis or obstructive sleep apnea at baseline. The participants in the current study included subjects with COPD who completed Wave 3 follow-up, were current nonsmokers, and had urine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol measured.
Structured telephone interviews
Participants completed structured telephone interviews that included health history, cigarette smoking, self-reported SHS exposure, sociodemographic characteristics, and health status. Direct personal cigarette smoking was evaluated using standard questions from the National Health Interview Survey (Centers for Disease Control and Prevention, 2002). Based on these responses, subjects were defined as current smokers, ex-smokers, and never-smokers. We also used a validated survey battery to assess SHS exposure during the 7-day period preceding interviews (Eisner, Katz, Yelin, Hammond, & Blanc, 2001).
Direct SHS monitoring
The primary analysis used urine NNAL as a measure of longer term SHS exposure. We also used other direct measurements of SHS exposure for comparison with NNAL levels.
Concentrations of NNAL were determined using liquid chromatography–tandem mass spectrometry (LC-MS/MS; Jacob et al., 2008). Urine specimens were incubated with beta-glucuronidase to cleave the glucuronide conjugates and to free NNAL; NNAL was then extracted using a liquid–liquid extraction procedure. The extracts were treated with hexanoic anhydride to convert NNAL to the hexanoate ester derivative prior to LC-MS/MS analysis. Deuterium-labeled NNAL (NNAL-d3) was used as the internal standard. The mass spectrometer was operated in the positive ion mode using electrospray ionization. Quantitation was achieved using selected reaction monitoring of the transitions m/z 308 to m/z 84 for NNAL hexanoate derivative and m/z 311 to m/z 87 for NNAL-d3 hexanoate. The lower limit of quantitation was 0.25 pg/ml.
We also measured urine cotinine to reflect shorter term exposure. Cotinine, a metabolite of nicotine, is a widely used and specific biomarker of SHS exposure (Benowitz, 1999). Cotinine has a short half-life of 20 hr and reflects shorter term SHS exposure. Urine samples and completed badges were returned to the investigators by mail.
Concentrations of cotinine and trans-3′-hydroxycotinine, which is the proximate metabolite of cotinine and the most abundant metabolite of nicotine present in urine, were determined using LC-MS/MS (Dempsey et al., 2004; Hukkanen, Gourlay, Kenkare, & Benowitz, 2005). The method is similar to a published method for determining cotinine concentrations in serum of nonsmokers (Bernert et al., 1997). Limits of quantification were 0.05 ng/ml for cotinine and 0.1 ng/ml for trans-3′-hydroxycotinine.
Each subject was instructed to wear the personal nicotine badge monitor during regular activities for 7 days. The passive monitor, which has been previously described, samples nicotine from ambient air (Hammond & Leaderer, 1987; Hammond, Sorensen, Youngstrom, & Ockene, 1995). The collected nicotine is analyzed by gas chromatography with nitrogen selective detection. The passive monitors have a limit of detection less than 0.01 μg per filter and a coefficient of variability of .11 for replicate analysis (Hammond & Leaderer).
Study outcome variables: COPD-related health status
We used a combined approach with disease-specific and generic health status measurements to assess COPD-related health status. To measure disease severity, we used a disease-specific COPD severity score that we had previously developed and validated for use in epidemiological and outcomes research (Eisner, Trupin, et al., 2005). Based on survey responses, the COPD severity score comprises five overall aspects of COPD severity: respiratory symptoms, systemic corticosteroid use, other COPD medication use, previous hospitalization or intubation for respiratory disease, and home oxygen use. Each item was weighted based on clinical aspects of the disease and its expected contribution to overall COPD severity. Possible total scores range from 0 to 35, with higher scores reflecting more severe COPD.
Generic physical health status was measured with the Short Form (SF)-12 physical component summary (PCS) score. The SF-12 is derived from the Medical Outcomes Study SF-36 instrument, which is the most widely used measure of generic health status. The SF-36 has been extensively validated in the general population (Ware & Sherbourne, 1992) and among adults with COPD (Benzo, Flume, Turner, & Tempest, 2000). Defined from the eight SF-36 subscales by factor analysis, the PCS score reflects an underlying physical dimension of physical health-related quality of life (Ware, Kosinski, & Keller, 1996). Higher scores reflect more favorable health states.
We used the Airways Questionnaire 20 revised to measure disease-specific quality of life (QOL; Chen, Eisner, Katz, Yelin, & Blanc, 2006; Hajiro et al., 1999). This instrument has been validated, and higher scores correspond to poorer QOL (Alemayehu, Aubert, Feifer, & Paul, 2002; Chen et al., 2006; Hajiro et al., 1999; Jones & Bosh, 1997; Jones, Quirk, Baveystock, & Littlejohns, 1992). Dyspnea was measured using three of five questions from the modified Medical Research Council (MRC) dyspnea scale (Hajiro et al., 1998). Higher scores indicate greater levels of dyspnea. Restricted activity, as a measure of disability due to a respiratory condition, was ascertained using questions modified from the National Health Interview Survey; the number of restricted activity days during the past month was queried (National Center for Health Statistics, n.d.).
Statistical analysis
Statistical analysis was conducted using SAS 9.1 (SAS Institute, Inc., Cary, NC). Bivariate analysis was conducted using the unpaired t test or Wilcoxon test for continuous variables (depending on variable distribution) and likelihood ratio chi-square test for dichotomous variables. The Spearman rank-order correlation was used to examine the association between different SHS measures.
We used linear regression analysis to examine the impact of recent longer term SHS exposure, as measured by NNAL-to-creatinine ratio, and health-related COPD status (COPD severity, generic physical health status, disease-specific QOL, dyspnea, and restricted activity days during the past month attributed to a respiratory condition). We used multivariate linear regression analysis to control for variables that could confound the relationship between SHS exposure and health outcomes, including age, sex, race, educational attainment, and smoking history (all subjects were current nonsmokers; Eisner, Balmes, et al., 2005; Iribarren, Friedman, Klatsky, & Eisner, 2001). Standard linear regression diagnostics indicated adequate model performance. Based on the distribution of NNAL/creatinine, linear regression results were expressed per increment of NNAL/creatinine corresponding to its interquartile range (i.e., 0.043 pmol/mg).
To parse out the health impacts of shorter and longer term SHS exposure, as measured by urine cotinine and NNAL, respectively, we included both in the same linear regression models. Because urine NNAL and cotinine are highly correlated, we developed a residual variable from the regression of urine NNAL/creatinine on cotinine/creatinine (Sternfeld, Ngo, Satariano, & Tager, 2002). The residual variable for NNAL/creatinine (NNAL/creatinineresid) represents that part of longer term SHS exposure (measured by NNAL) not accounted for by shorter term SHS exposure (measured by cotinine). In effect, this approach separates out the longer and shorter term SHS exposure into separate variables; the correlation between NNAL/creatinineresid and cotinine/creatinine ratio is essentially zero.
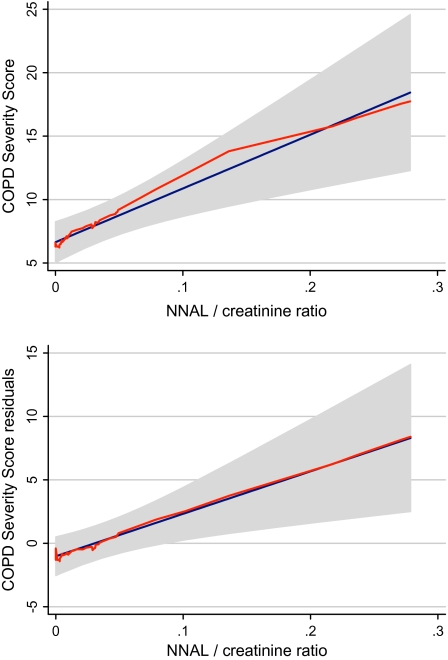
We used the locally weighted regression scatter plot smoother (LOWESS) procedure to graphically depict the relationship between NNAL-to-creatinine ratio and COPD severity score, which fitted a flexible smoothed curve that does not impose a linear relationship (Cleveland, 1979). To adjust the COPD severity score for covariates, we regressed severity score on age, sex, race, educational attainment, and smoking history and calculated the residual values. A LOWESS plot of COPD severity score residuals versus NNAL-to-creatinine ratio depicts the association between the COPD severity, after removing the variability due to the covariates, and the NNAL-to-creatinine ratio. In addition to the LOWESS curve, we superimposed the regression line and the 95% CI.
Results
Subject characteristics and SHS exposure
Characteristics of participants with COPD are shown in Table 1. The prevalence of longer term SHS exposure by any detectable urine NNAL was 65% (95% CI 53–76). Among subjects with any SHS exposure, the range of NNAL-to-creatinine ratio was 0.0012–0.28 pmol/mg (median = 0.024 pmol/mg; 25th–75th interquartile range 0.0055–0.049 pmol/mg). Table 2 shows the correlation of urine NNAL with other direct SHS measures. NNAL correlated most strongly with urine cotinine (r = .75) and hydroxycotinine (r = .74), but less strongly with nicotine measured by diffusion badge and by self-reported SHS exposure for a 7-day period (r = .33 and r = .34, respectively; all p values < .05).
Table 1.
Characteristics of adult nonsmoking participants with COPD
| Characteristics | M (SD) or n (%) |
| Age (years) | 64.9 (6.3) |
| Gender: male (%) | 28 (39%) |
| Race–ethnicity (White, non-Hispanic) | 68 (94%) |
| Educational attainment | |
| High school or less | 32 (44%) |
| Some college | 22 (31%) |
| College graduate or graduate degree | 18 (25%) |
| Smoking history | 46 (63%) |
| COPD severity score | 7.9 (6.6) |
| SF-12 PCS score | 35.4 (11.7) |
| AQ-20 | 8.2 (4.6) |
| Modified MRC dyspnea score | 2.5 (1.2) |
| Restricted activity days due to a respiratory condition (days during the past month) | 7.1 (12.3) |
Note. AQ-20 = Airways Questionnaire 20; COPD = chronic obstructive pulmonary disease; PCS = physical component summary; SF = Short Form. The range of observed scores was 0–24 points (COPD severity), 0–17 points (AQ-20), 1–4 points (MRC), and 14.7–57.2 points (PCS).
Table 2.
Correlation between NNAL levels and other measures of SHS exposure (n = 72)
| Measure | Urine NNAL | Urine NNAL-to-creatinine ratio | Urine cotinine | Urine hydroxycotinine | Nicotine badge | Self-reported SHS exposurea |
| Urine NNAL | .97, p < .0001 | .75, p < .0001 | .74, p < .0001 | .33, p = .004 | .34, p = .003 | |
| Urine NNAL-to-creatinine ratio | .71, p < .0001 | .67, p < .0001 | .31, p = .008 | .29, p = .01 | ||
| Urine cotinine | .73, p < .0001 | .36, p = .002 | .29, p = .01 | |||
| Urine hydroxycotinine | .43, p < .0001 | .25, p = .03 | ||||
| Nicotine badge | .10, p = .37 |
Note. NNAL = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol; SHS = secondhand smoke. Spearman’s rank-order correlation and p values.
Self-reported total hours of exposure during the past 7 days.
Longer term SHS exposure and COPD-related outcomes
Higher SHS exposure, as measured by the urine NNAL-to-creatinine ratio, was associated with greater COPD severity, after controlling for covariates (mean increase in severity score 1.7 points for each interquartile increment of NNAL/creatinine; 95% CI 0.6–2.8 points; Table 3). Higher NNAL/creatinine levels were also related to poorer physical health status, greater dyspnea, and more restricted activity days (Table 3). Figure 2 graphically depicts the approximate linear relationship between NNAL-to-creatinine ratio and COPD severity score.
Table 3.
Longer term secondhand smoke exposure as measured by NNAL and COPD-related health status (n = 72)
| Mean change in outcome | Mean change in outcome | |
| Health outcome measurement | Unadjusted | Adjusteda |
| COPD severity (points) | 1.8 (0.8–2.9), p = .0008 | 1.7 (0.6–2.8), p = .003 |
| Physical health status (points) | −2.3 (−4.2 to −0.4), p = .019 | −1.9 (−3.8 to −0.1), p = .04 |
| Disease-specific health-related quality of life (points) | 0.8 (−0.003 to 1.6), p = .051 | 0.5 (−0.3 to 1.3), p = .19 |
| Dyspnea (points) | 0.24 (0.04–0.4), p = .018 | 0.20 (0–0.39), p = .051 |
| Restricted activity days due to respiratory condition (during past 30 days) | 3.3 (1.4–5.3), p = .001 | 3.2 (1.1–5.3), p = .003 |
Note. COPD = chronic obstructive pulmonary disease; NNAL = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol.
Multivariate analysis controls for age, sex, race, educational attainment, and past smoking (all subjects are current nonsmokers). Results are mean change in health status variable per 0.0.043 pmol/mg increase in NNAL/creatinine ratio (i.e., interquartile range) and 95% CI.
Figure 2.
Longer term secondhand smoke exposure and chronic obstructive pulmonary disease (COPD) severity. (a) Locally weighted regression scatter plot smoother (LOWESS) procedure was used to graphically depict the relationship between 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL)-to-creatinine ratio and COPD severity score in a flexible manner (shown in orange). The blue line is the fitted linear regression line. The gray area shows the 95% CI. (b) LOWESS plot of COPD severity score residuals that adjust for age, sex, race, educational attainment, and smoking history, and NNAL-to-creatinine ratio (orange line). The blue line is the fitted linear regression line. The gray area shows the 95% CI.
Shorter and longer term SHS exposure: Impact on COPD-related outcomes
We included the NNAL and cotinine in the same regression analysis to distinguish the health effects of shorter and longer term SHS exposure. NNAL-to-creatinine residuals (NNAL/creatinineresid) were used to account for colinearity (see Methods section). Higher longer term SHS exposure, as estimated by NNAL/creatinineresid, was associated with worse COPD severity, poorer physical health status, increased dyspnea, and more respiratory-related restricted activity (p < .05 in all cases; Table 4). There was no statistical relationship between shorter term SHS exposure and any of these health outcomes, although the point estimates were in the same direction of adverse effects as for longer term exposure.
Table 4.
Independent impact of shorter and longer term exposure on COPD-related health status (n = 72)
| Health outcome measurement | NNAL/creatinineresida | Cotinine/creatinine |
| COPD severity (points) | 2.0 (0.6–3.4), p = .005 | 0.37 (−0.21 to 0.95), p = .21 |
| Physical health status (points) | −2.4 (−4.8 to −0.02), p = .049 | −0.40 (−1.4 to 0.6), p = .42 |
| Disease-specific health-related quality of life (points) | 0.5 (−0.5 to 1.6), p = .29 | 0.2 (−0.3 to 0.6), p = .43 |
| Dyspnea (points) | 0.27 (0.02–0.52), p = .034 | 0.025 (−0.08 to 0.13), p = .64 |
| Restricted activity days due to respiratory condition (during past 30 days) | 3.6 (0.9–6.2), p = .010 | 0.9 (−0.2 to 2.0), p = .11 |
Note. COPD = chronic obstructive pulmonary disease; NNAL = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol; SHS = secondhand smoke. Multivariate analysis controls for age, sex, race, educational attainment, and past smoking (all subjects are current nonsmokers). Results are mean change in health status variable per interquartile range increase in NNAL/creatinineresid (0.043 pmol/mg) and cotinine/creatinine ratio (0.040 ng/dl) and 95% CI.
Because urine NNAL and cotinine are highly correlated, we developed a residual variable from the regression of urine NNAL/creatinine on cotinine/creatinine. The residual variable for NNAL/creatinine (NNAL/creatinineresid) represents that part of longer term SHS exposure (measured by NNAL) not accounted for by shorter term SHS exposure (measured by cotinine).
Discussion
We previously reported that shorter term SHS exposure, as measured by urine cotinine, was associated with poorer health outcomes in COPD (Eisner et al., 2006). Using a measure of longer term SHS exposure (NNAL), we now report a greater and more consistent negative impact of SHS on a broad array of COPD health status measures, including disease severity, physical health status, and disability. Furthermore, we were able to distinguish between the effects of shorter and longer term SHS exposure; the latter was the most relevant for COPD-related health outcomes.
Previous studies have shown that urine NNAL levels are higher among nonsmokers exposed to SHS, including the home and workplace environments (Anderson et al., 2003; Hecht et al., 2001, 2006; Stark et al., 2007; Tulunay et al., 2005). The strong relationship between NNK, NNAL (its metabolite), and lung carcinogenesis has also been highlighted in previous work (Hecht, 1998; Hecht et al., 1999). The use of NNAL in conjunction with cotinine to estimate the shorter and longer term effects of SHS exposure is a novel use of this biomarker. Our data show that markers of shorter and longer term SHS exposure are correlated but have different associations with COPD-related health outcomes. When studying longer term health outcomes, such as disease severity and QOL in COPD, NNAL appears to be the superior biomarker.
Our study adds to a newly emerging literature that SHS exposure may adversely affect the clinical course of COPD. We previously reported that short-term SHS exposure, as evidenced by urine cotinine, was related to poorer COPD severity and health status measures (Eisner et al., 2006). In a previous cohort study of adults hospitalized for COPD, self-reported SHS exposure was also a risk factor for rehospitalization (Garcia-Aymerich et al., 2003). Another population-based U.S. study found that self-reported SHS exposure was related to a greater risk of “chronic respiratory disease exacerbation,” defined as activity limitation or a physician’s visit due to asthma, chronic bronchitis, emphysema, or chronic sinusitis (Mannino, Siegel, Rose, Nkuchia, & Etzel, 1997). More recently, indoor PM2.5 (particulate matter) levels, a proxy measure of home SHS exposure, was associated with poorer disease-specific QOL among persons with COPD (Osman et al., 2007). The present findings, which link longer term SHS exposure with adverse COPD outcomes, add substantive additional evidence that SHS exposure is deleterious for patients with COPD.
When considered simultaneously, longer term SHS exposure had the greater impact on COPD severity than shorter term exposure. There are several possible explanations. Cotinine, because it reflects SHS exposure during only the past 1–2 days, may miss an important exposure event that causes deterioration of COPD. Moreover, repeated exposures to SHS, which will be better captured by NNAL, may have a greater effect on COPD severity than a single recent exposure. Our previous work showed that shorter term exposure (cotinine) was associated with poorer health outcomes in COPD (Eisner et al., 2006); the current study shows that the longer term exposure measure has an even greater impact on COPD outcomes.
Our study is subject to some limitations. We used the standard epidemiological definition of COPD based on a self-reported physician’s diagnosis of chronic bronchitis, emphysema, or COPD (Mannino, 2002; Mannino et al., 2002; Sin et al., 2002). This survey-based approach enabled us to study a population-based sample of adults who resided throughout the country; this enhanced applicability of the findings. On logistical grounds, conducting spirometry among subjects who reside thousands of miles apart would not be feasible. The use of self-reported physician’s diagnosis, however, may have resulted in some misclassification of disease status. Previous work indicated that a similar survey-based definition of COPD had a high positive predictive value (78%) when validated using a blinded medical record review that included spirometry and radiographic studies (Barr, Herbstman, Speizer, & Camargo, 2002). Other work confirmed that a self-reported history of COPD is a strong predictor of airflow obstruction (Straus, McAlister, Sackett, & Deeks, 2002). Our previous validation study indicated that nearly 9 out of 10 participants who had available spirometry data had objective evidence of airflow obstruction (Eisner, Balmes, et al., 2005). In sum, misclassification of COPD is not likely to bias our results and, if present, it would likely be nondifferential with respect to SHS exposure and diminish effect estimates toward the null value.
Other limitations include the potential confounding effects of direct cigarette smoking. We attempted to reduce this confounding effect by limiting the analysis to persons who indicated no current smoking and had urine cotinine levels below a cutpoint usually associated with active smoking. We also statistically controlled for a history of cigarette smoking in multivariate analysis. Nonetheless, we cannot entirely exclude confounding by occasional or light active smoking. And finally, we cannot completely exclude a bias termed the “healthy passive smoker effect,” a phenomenon in which more severely affected COPD patients might selectively avoid SHS exposure, theoretically attenuating the observed association between exposure and asthma health outcomes. If this bias occurred, it would also be a conservative one. Other sources of selection bias are also possible and cannot be precisely quantified.
In conclusion, longer term SHS exposure appears to negatively influence the health status of adults with COPD. Longer term exposure, as evidenced by urine NNAL, has a more important adverse impact than shorter term exposure. Future studies of respiratory health effects of SHS exposure should consider using urine NNAL to measure exposure. In clinical and public health terms, SHS exposure avoidance may be an important component of COPD management.
Funding
This work was funded by Flight Attendants Medical Research Institute UCSF Bland Lane Center of Excellence in Secondhand Smoke and R01 HL677438 from National Heart, Lung, and Blood Institute/National Institutes of Health.
Declaration of Interests
None declared.
Supplementary Material
Acknowledgments
The authors are grateful to Christopher Havel and Do-Hoon Lee for carrying out the NNAL analyses and Minjiang Duan and Lisa Yu for the cotinine analyses.
References
- Alemayehu B, Aubert RE, Feifer RA, Paul LD. Comparative analysis of two quality-of-life instruments for patients with chronic obstructive pulmonary disease. Value in Health. 2002;5:436–441. doi: 10.1046/J.1524-4733.2002.55151.x. [DOI] [PubMed] [Google Scholar]
- Anderson KE, Kliris J, Murphy L, Carmella SG, Han S, Link C, et al. Metabolites of a tobacco-specific lung carcinogen in nonsmoking casino patrons. Cancer Epidemiology, Biomarkers and Prevention. 2003;12:1544–1546. [PubMed] [Google Scholar]
- Barr RG, Herbstman J, Speizer FE, Camargo CA., Jr. Validation of self-reported chronic obstructive pulmonary disease in a cohort study of nurses. American Journal of Epidemiology. 2002;155:965–971. doi: 10.1093/aje/155.10.965. [DOI] [PubMed] [Google Scholar]
- Benowitz NL. Biomarkers of environmental tobacco smoke exposure. Environmental Health Perspectives. 1999;107(Suppl. 2):349–355. doi: 10.1289/ehp.99107s2349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benzo R, Flume PA, Turner D, Tempest M. Effect of pulmonary rehabilitation on quality of life in patients with COPD: The use of SF-36 summary scores as outcomes measures. Journal of Cardiopulmonary Rehabilitation. 2000;20:231–234. doi: 10.1097/00008483-200007000-00003. [DOI] [PubMed] [Google Scholar]
- Bernert JT, Jr., Turner WE, Pirkle JL, Sosnoff CS, Akins JR, Waldrep MK, et al. Development and validation of sensitive method for determination of serum cotinine in smokers and nonsmokers by liquid chromatography/atmospheric pressure ionization tandem mass spectrometry. Clinical Chemistry. 1997;43:2281–2291. [PubMed] [Google Scholar]
- Blanc PD, Eisner MD, Balmes JR, Trupin L, Yelin EH, Katz PP. Exposure to vapors, gas, dust, or fumes: Assessment by a single survey item compared to a detailed exposure battery and a job exposure matrix. American Journal of Industrial Medicine. 2005;48:110–117. doi: 10.1002/ajim.20187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blanc PD, Eisner MD, Trupin L, Yelin EH, Katz PP, Balmes JR. The association between occupational factors and adverse health outcomes in chronic obstructive pulmonary disease. Occupational and Environmental Medicine. 2004;61:661–667. doi: 10.1136/oem.2003.010058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention; Cigarette smoking among adults—United States, 2000. Morbidity and Mortality Weekly Report. 2002;51:642–645. [PubMed] [Google Scholar]
- Chen H, Eisner MD, Katz PP, Yelin EH, Blanc PD. Measuring disease-specific quality of life in obstructive airway disease: Validation of a modified version of the airways questionnaire 20. Chest. 2006;129:1644–1652. doi: 10.1378/chest.129.6.1644. [DOI] [PubMed] [Google Scholar]
- Cleveland WS. Robust locally weighted regression and smoothing scatterplots. Journal of the American Statistical Association, 1979;74:829–836. [Google Scholar]
- Dempsey D, Tutka P, Jacob P, 3rd, Allen F, Schoedel K, Tyndale RF, et al. Nicotine metabolite ratio as an index of cytochrome P450 2A6 metabolic activity. Clinical Pharmacology and Therapeutics. 2004;76(1):64–72. doi: 10.1016/j.clpt.2004.02.011. [DOI] [PubMed] [Google Scholar]
- Eisner MD, Balmes J, Katz PP, Trupin L, Yelin EH, Blanc PD. Lifetime environmental tobacco smoke exposure and the risk of chronic obstructive pulmonary disease. Environmental Health. 2005;4(1):7. doi: 10.1186/1476-069X-4-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisner MD, Balmes J, Yelin EH, Katz PP, Hammond SK, Benowitz N, et al. Directly measured secondhand smoke exposure and COPD health outcomes. BMC Pulmonary Medicine. 2006;6:12. doi: 10.1186/1471-2466-6-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisner MD, Katz PP, Yelin EH, Hammond SK, Blanc PD. Measurement of environmental tobacco smoke exposure among adults with asthma. Environmental Health Perspectives. 2001;109:809–814. doi: 10.1289/ehp.01109809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisner MD, Klein J, Hammond SK, Koren G, Lactao G, Iribarren C. Directly measured second hand smoke exposure and asthma health outcomes. Thorax. 2005;60:814–821. doi: 10.1136/thx.2004.037283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisner MD, Trupin L, Katz PP, Yelin EH, Earnest G, Balmes J, et al. Development and validation of a survey-based COPD severity score. Chest. 2005;127:1890–1897. doi: 10.1378/chest.127.6.1890. [DOI] [PubMed] [Google Scholar]
- Eisner MD, Yelin EH, Katz PP, Earnest G, Blanc PD. Exposure to indoor combustion and adult asthma outcomes: Environmental tobacco smoke, gas stoves, and woodsmoke. Thorax. 2002;57:973–978. doi: 10.1136/thorax.57.11.973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisner MD, Yelin EH, Trupin L, Blanc PD. The influence of chronic respiratory conditions on health status and work disability. American Journal of Public Health. 2002;92:1506–1513. doi: 10.2105/ajph.92.9.1506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Aymerich J, Farrero E, Felez MA, Izquierdo J, Marrades RM, Anto JM. Risk factors of readmission to hospital for a COPD exacerbation: A prospective study. Thorax. 2003;58:100–105. doi: 10.1136/thorax.58.2.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hajiro T, Nishimura K, Jones PW, Tsukino M, Ikeda A, Koyama H, et al. A novel, short, and simple questionnaire to measure health-related quality of life in patients with chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine. 1999;159:1874–1878. doi: 10.1164/ajrccm.159.6.9807097. [DOI] [PubMed] [Google Scholar]
- Hajiro T, Nishimura K, Tsukino M, Ikeda A, Koyama H, Izumi T. Analysis of clinical methods used to evaluate dyspnea in patients with chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine. 1998;158:1185–1189. doi: 10.1164/ajrccm.158.4.9802091. [DOI] [PubMed] [Google Scholar]
- Hammond SK, Leaderer B. A diffusion monitor to measure exposure to passive smoking. Environmental Science and Technology. 1987;21:494–497. doi: 10.1021/es00159a012. [DOI] [PubMed] [Google Scholar]
- Hammond SK, Sorensen G, Youngstrom R, Ockene JK. Occupational exposure to environmental tobacco smoke [see comments] Journal of the American Medical Association. 1995;274:956–960. [PubMed] [Google Scholar]
- Hecht SS. Biochemistry, biology, and carcinogenicity of tobacco-specific N-nitrosamines. Chemical Research in Toxicology. 1998;11:559–603. doi: 10.1021/tx980005y. [DOI] [PubMed] [Google Scholar]
- Hecht SS. Carcinogen derived biomarkers: Applications in studies of human exposure to secondhand tobacco smoke. Tobacco Control. 2004;13(Suppl. 1):i48–i56. doi: 10.1136/tc.2002.002816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hecht SS. A biomarker of exposure to environmental tobacco smoke (ETS) and Ernst Wynder's opinion about ETS and lung cancer. Preventive Medicine. 2006;43:256–260. doi: 10.1016/j.ypmed.2006.07.020. [DOI] [PubMed] [Google Scholar]
- Hecht SS, Carmella SG, Chen M, Dor Koch JF, Miller AT, Murphy SE, et al. Quantitation of urinary metabolites of a tobacco-specific lung carcinogen after smoking cessation. Cancer Research. 1999;59:590–596. [PubMed] [Google Scholar]
- Hecht SS, Carmella SG, Le KA, Murphy SE, Boettcher AJ, Le C, et al. 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol and its glucuronides in the urine of infants exposed to environmental tobacco smoke. Cancer Epidemiology, Biomarkers and Prevention. 2006;15:988–992. doi: 10.1158/1055-9965.EPI-05-0596. [DOI] [PubMed] [Google Scholar]
- Hecht SS, Ye M, Carmella SG, Fredrickson A, Adgate JL, Greaves IA, et al. Metabolites of a tobacco-specific lung carcinogen in the urine of elementary school-aged children. Cancer Epidemiology, Biomarkers and Prevention. 2001;10:1109–1116. [PubMed] [Google Scholar]
- Hukkanen J, Gourlay SG, Kenkare S, Benowitz NL. Influence of menstrual cycle on cytochrome P450 2A6 activity and cardiovascular effects of nicotine. Clinical Pharmacology and Therapeutics. 2005;77(3):159–169. doi: 10.1016/j.clpt.2004.10.012. [DOI] [PubMed] [Google Scholar]
- Iribarren C, Friedman GD, Klatsky AL, Eisner MD. Exposure to environmental tobacco smoke: Association with personal characteristics and self reported health conditions. Journal of Epidemiology and Community Health. 2001;55:721–728. doi: 10.1136/jech.55.10.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacob P, 3rd, Havel C, Lee DH, Yu L, Eisner MD, Benowitz NL. Subpicogram per milliliter determination of the tobacco-specific carcinogen metabolite 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol in human urine using liquid chromatography-tandem mass spectrometry. Analytical Chemistry. 2008;80:8115–8121. doi: 10.1021/ac8009005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarvis MJ, Tunstall-Pedoe H, Feyerabend C, Vesey C, Saloojee Y. Comparison of tests used to distinguish smokers from nonsmokers. American Journal of Public Health. 1987;77:1435–1438. doi: 10.2105/ajph.77.11.1435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones PW, Bosh TK. Quality of life changes in COPD patients treated with salmeterol. American Journal of Respiratory and Critical Care Medicine. 1997;155:1283–1289. doi: 10.1164/ajrccm.155.4.9105068. [DOI] [PubMed] [Google Scholar]
- Jones PW, Quirk FH, Baveystock CM, Littlejohns P. A self-complete measure of health status for chronic airflow limitation. The St. George's Respiratory Questionnaire. American Review of Respiratory Disease. 1992;145:1321–1327. doi: 10.1164/ajrccm/145.6.1321. [DOI] [PubMed] [Google Scholar]
- Kim J. Atlas of respiratory disease mortality, United States: 1982–1993. Cincinnati, OH: Department of Health and Human Services, National Institute for Occupational Safety and Health; 1998. [Google Scholar]
- Mannino DM. COPD: Epidemiology, prevalence, morbidity and mortality, and disease heterogeneity. Chest. 2002;121(5 Suppl.):121S–126S. doi: 10.1378/chest.121.5_suppl.121s. [DOI] [PubMed] [Google Scholar]
- Mannino DM, Homa DM, Akinbami LJ, Ford ES, Redd SC. Chronic obstructive pulmonary disease surveillance—United States, 1971–2000. Morbidity and Mortality Weekly Report. Surveillance Summaries. 2002;51(6):1–16. [PubMed] [Google Scholar]
- Mannino DM, Siegel M, Rose D, Nkuchia J, Etzel R. Environmental tobacco smoke exposure in the home and worksite and health effects in adults: Results from the 1991 National Health Interview Survey. Tobacco Control. 1997;6:296–305. doi: 10.1136/tc.6.4.296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- National Center for Health Statistics. National Health Interview Survey. Retrieved March 28, 2008, from http://www.cdc.gov/nchs/about/major/nhis/quest_data_related_1997_forward.htm. [Google Scholar]
- Office of Environmental Health Hazard Assessment, California Environmental Protection Agency. Health effects assessment for environmental tobacco smoke. 2005 Retrieved February 24, 2006, from ftp://ftp.arb.ca.gov/carbis/regact/ets2006/app3part%20b.pdf. [Google Scholar]
- Osman LM, Douglas JG, Garden C, Reglitz K, Lyon J, Gordon S, et al. Indoor air quality in homes of patients with chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine. 2007;176:465–472. doi: 10.1164/rccm.200605-589OC. [DOI] [PubMed] [Google Scholar]
- Sin DD, Stafinski T, Ng YC, Bell NR, Jacobs P. The impact of chronic obstructive pulmonary disease on work loss in the United States. American Journal of Respiratory and Critical Care Medicine. 2002;165:704–707. doi: 10.1164/ajrccm.165.5.2104055. [DOI] [PubMed] [Google Scholar]
- Stark MJ, Rohde K, Maher JE, Pizacani BA, Dent CW, Bard R, et al. The impact of clean indoor air exemptions and preemption policies on the prevalence of a tobacco-specific lung carcinogen among nonsmoking bar and restaurant workers. American Journal of Public Health. 2007;97:1457–1463. doi: 10.2105/AJPH.2006.094086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sternfeld B, Ngo L, Satariano WA, Tager IB. Associations of body composition with physical performance and self-reported functional limitation in elderly men and women. American Journal of Epidemiology. 2002;156:110–121. doi: 10.1093/aje/kwf023. [DOI] [PubMed] [Google Scholar]
- Straus SE, McAlister FA, Sackett DL, Deeks JJ. Accuracy of history, wheezing, and forced expiratory time in the diagnosis of chronic obstructive pulmonary disease. Journal of General Internal Medicine. 2002;17:684–688. doi: 10.1046/j.1525-1497.2002.20102.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trupin L, Earnest G, San Pedro M, Balmes JR, Eisner MD, Yelin E, et al. The occupational burden of chronic obstructive pulmonary disease. European Respiratory Journal. 2003;22:462–469. doi: 10.1183/09031936.03.00094203. [DOI] [PubMed] [Google Scholar]
- Tulunay OE, Hecht SS, Carmella SG, Zhang Y, Lemmonds C, Murphy S, et al. Urinary metabolites of a tobacco-specific lung carcinogen in nonsmoking hospitality workers. Cancer Epidemiology, Biomarkers and Prevention. 2005;14:1283–1286. doi: 10.1158/1055-9965.EPI-04-0570. [DOI] [PubMed] [Google Scholar]
- Ware J, Jr., Kosinski M, Keller SD. A 12-Item Short-Form Health Survey: Construction of scales and preliminary tests of reliability and validity. Medical Care. 1996;34:220–233. doi: 10.1097/00005650-199603000-00003. [DOI] [PubMed] [Google Scholar]
- Ware JE, Jr., Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Medical Care. 1992;30:473–483. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.