Abstract
Background
Evolutionary analyses of enterohemorrhagic Escherichia coli (EHEC) have identified two distantly related clonal groups: EHEC 1, including serotype O157:H7 and its inferred ancestor O55:H7; and EHEC 2, comprised of several serogroups (O26, O111, O118, etc.). These two clonal groups differ in their virulence and global distribution. Although several fully annotated genomic sequences exist for strains of serotype O157:H7, much less is known about the genomic composition of EHEC 2. In this study, we analyzed a set of 24 clinical EHEC 2 strains representing serotypes O26:H11, O111:H8/H11, O118:H16, O153:H11 and O15:H11 from humans and animals by comparative genomic hybridization (CGH) on an oligoarray based on the O157:H7 Sakai genome.
Results
Backbone genes, defined as genes shared by Sakai and K-12, were highly conserved in EHEC 2. The proportion of Sakai phage genes in EHEC 2 was substantially greater than that of Sakai-specific bacterial (non-phage) genes. This proportion was inverted in O55:H7, reiterating that a subset of Sakai bacterial genes is specific to EHEC 1. Split decomposition analysis of gene content revealed that O111:H8 was more genetically uniform and distinct from other EHEC 2 strains, with respect to the Sakai O157:H7 gene distribution. Serotype O26:H11 was the most heterogeneous EHEC 2 subpopulation, comprised of strains with the highest as well as the lowest levels of Sakai gene content conservation. Of the 979 parsimoniously informative genes, 15% were found to be compatible and their distribution in EHEC 2 clustered O111:H8 and O118:H16 strains by serotype. CGH data suggested divergence of the LEE island from the LEE1 to the LEE4 operon, and also between animal and human isolates irrespective of serotype. No correlation was found between gene contents and geographic locations of EHEC 2 strains.
Conclusion
The gene content variation of phage-related genes in EHEC 2 strains supports the hypothesis that extensive modular shuffling of mobile DNA elements has occurred among EHEC strains. These results suggest that EHEC 2 is a multiform pathogenic clonal complex, characterized by substantial intra-serotype genetic variation. The heterogeneous distribution of mobile elements has impacted the diversification of O26:H11 more than other EHEC 2 serotypes.
Background
Enterohemorrhagic Escherichia coli (EHEC), the intersection of Shiga toxin producing E. coli (STEC) and attaching and effacing E. coli (AEEC), comprise a group of pathogenic E. coli that cause a variety of human and animal illnesses ranging from diarrhea to hemorrhagic colitis (HC), and the multifactorial hemolytic uremic syndrome (HUS) [1]. Intimate adherence to the intestinal epithelium resulting in characteristic attaching and effacing (A/E) lesions, and the destruction of capillary walls via production of phage borne Shiga toxins (Stx 1, 2, and variants) are hallmarks of EHEC pathogenesis. A/E lesion formation is dependent upon a type three secretion system (TTSS), which is encoded on the laterally acquired locus of enterocyte effacement (LEE) [2].
E. coli O157:H7 is the dominant EHEC serotype in the United States, Argentina, Great Britain, and Japan [3,4]. However, multiple reports have shown that other EHEC, including serogroups O26, O111, O103, and O118, frequently cause sporadic cases of human illness [5-12], and have been implicated in numerous outbreaks [13-17]. In Australia and parts of Europe, infections with serogroups O26 and O111 are prevailing while the incidence of O157:H7-associated disease appears to be declining [18-21]. In contrast to E. coli O157:H7, EHEC serogroups O26, O111, O118, O103, and O5 are commonly linked to outbreaks and sporadic cases of calf diarrhea (scours) and HC [22-28], which has been validated from experimental infections in calves [29-32]. In Germany and Belgium, for example, EHEC O118 is the most prevalent type of STEC associated with diarrhea in calves [33], with evidence for zoonotic transmission [8,34].
Phylogenetic analyses of conserved metabolic genes have revealed some of the basis for the variation among EHEC strains. Multilocus enzyme electrophoresis [35] and partial sequencing of 13 housekeeping genes [36] classified EHEC into two distantly related clonal groups: EHEC 1 includes serotype O157:H7 and its inferred ancestor O55:H7, whereas EHEC 2 includes numerous serogroups (e.g., O26, O111, O118). The key virulence factors shared between EHEC 1 and EHEC 2 clonal complexes were postulated to have been introduced through multiple and parallel acquisitions of mobile elements [37]. A comparison of E. coli O157:H7 genomes has also revealed the extent and significant impact of horizontal transfer on the evolution of virulence [38,39]. Furthermore, array comparative genomic hybridizations (CGH) have shown that the divergence in gene content among closely related O157 strains is ~140 times greater than the divergence at the nucleotide sequence level [40]. Although recent evidence indicates the emergence of highly virulent lineages among non-O157 EHEC, notably the O26 serogroup [19,41], little is known about the gene content, genetic diversity and evolution of virulence in members of the EHEC 2 group.
The function of ancillary virulence determinants is somewhat characterized in O157:H7 [2,42], however, the relevance as well as the distribution of these factors in EHEC 2 is not clear. To systematically investigate the gene content variations within the EHEC 2 clonal group. we analyzed a set of 24 clinical EHEC 2 strains representing serotypes O26:H11, O111:H8/H11, O118:H16, O153:H11 and O15:H11 from humans and animals using array-based CGH. Because there are no EHEC 2 genome sequences available, a multi-genome spotted oligoarray containing probes for 5,978 ORFs from O157:H7 Sakai, O157:H7 EDL933, and K-12 MG1655 was used to examine the distribution of these E. coli genes in our collection of EHEC 2 strains. The findings of this study shed light on the diversification of horizontally acquired elements in a group of pathogens that represent recent evolutionary branches of EHEC clonal groups.
Results
Sequence types (STs) and stx profiles of EHEC 2 strains
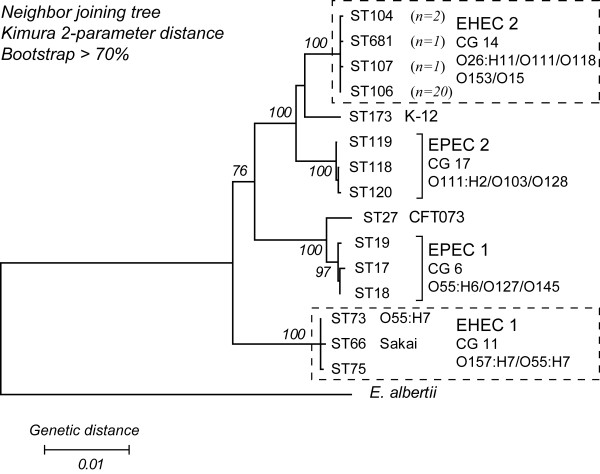
Phylogenetic analyses of multi locus sequence typing (MLST) data grouped the 24 EHEC 2 strains (Table 1) into four STs. The most common was ST 106, which was found in 20 strains, while the remaining three STs each differed from ST 106 by a single nucleotide polymorphism (SNP) in almost 4,000 bp of the concatenated MLST sequence. MLST data revealed a lack of nucleotide sequence diversity in house keeping genes among these EHEC 2 strains. The neighbor-joining phylogeny based on concatenated MLST allelic sequences grouped the EHEC 2 strains into a distinct cluster, with 100% bootstrap support, which was more closely related to the EPEC 2 group (100% bootstrap support) than to members of EHEC 1 (Figure 1). Most of these EHEC 2 strains (n = 17) were PCR positive for only stx1, whereas four strains had both stx1 and stx2, and three strains were negative for both stx genes (Table 1).
Table 1.
Properties of strains used in this study sorted by serotype.
| Straina | Serotypeb | Host | Clinicalc | Location | Dated | stx | STe | Sourcef, Reference |
| DEC 9f | O26: [h11] | Human | diarrhea | USA, S. D. | 1974 | - | 106 | CDC, [80] |
| DEC 10e | O26:H11 | Calf | scours | USA, S. D. | 1989 | 1 | 106 | Francis, D., [81] |
| F5863 | O26:H11 | Human | diarrhea | USA, Nebr. | 1998 | 1 | 106 | Fey, P., [6] |
| 97–3250 | O26:H11 | Human | HUS | USA, Idaho | 1997 | 1,2 | 104 | O'Brien, [82] |
| 413/89-1 | O26: [h11] | Calf | diarrhea | Germany | 1998 | 1 | 106 | Wieler, L., [83] |
| DA-22 | O26: [h11] | Human | diarrhea | USA, D.C. | 1999 | 1 | 106 | Acheson, D. W. |
| 03-ST-296 | O26:H11 | Human | b. d. | USA, Mich. | 2003 | 1 | 106 | MDCH, [84] |
| CB 7505 | O26:H11 | Calf | no data | Germany | 1998 | 1 | 106 | Beutin, L., [85] |
| DEC 8c | O111: [h11] | Calf | scours | USA, S. D. | 1986 | 1 | 107 | Francis, D., [80] |
| DEC 8d | O111:H11 | Human | diarrhea | Cuba | 1953 | - | 106 | Orskov, F., [86] |
| C408 | O111: [h8] | Calf | diarrhea | Scotland | 1993 | 1 | 106 | Hart, C. A., [87] |
| BCL71 | O111: [h8] | Calf | diarrhea | USA, Calif. | 1993 | 1,2 | 106 | Love, B.C. |
| ML178190 | O111: [h8] | Human | diarrhea | USA, Nebr. | 1998 | 1,2 | 106 | Fey, P., [6] |
| W29104 | O111:H8 | Human | diarrhea | USA, Nebr. | 1998 | 1,2 | 106 | Fey, P., [6] |
| EK34 | O111: [h8] | Human | diarrhea | USA, Wash. | 1999 | 1 | 106 | Tarr, P., [88] |
| EK35 | O111:H8 | Human | diarrhea | USA, Wash. | 2001 | 1 | 106 | Tarr, P., [88] |
| RW2030 | O118: [h16] | Calf | diarrhea | Germany | 1994 | 1 | 106 | Wieler, L., [33] |
| RW1302 | O118: [h16] | Calf | diarrhea | Germany | 1994 | 1 | 106 | Wieler, L., [33] |
| 666/89 | O118:H16 | Calf | diarrhea | Germany | 1989 | 1 | 106 | Wieler, L., [33] |
| 05482 | O118:H16 | Human | HUS | Germany | 1996 | 1 | 106 | Beutin, L., [89] |
| EK36 | O118:H16 | Human | diarrhea | USA, Wash. | 2001 | 1 | 106 | Tarr, P., [88] |
| EK37 | O118:H16 | Human | diarrhea | USA, Wash. | 2000 | 1 | 106 | Tarr, P., [88] |
| RDEC-1 | O15: [h11] | Rabbit | diarrhea | USA, S.C. | 1970s | - | 681 | ECRC, [81] |
| 02–3751 | O153: [h11] | Rabbit | HUS | USA, Mass. | 2002 | 1 | 104 | Fox, J., [90] |
| 97–3256 | O55:H7 | Human | diarrhea | USA, Mich. | 1997 | 2 | 73 | O'Brien, [82] |
a. designations assigned to strains deposited in the STEC Reference Center
b. [h] – flagellar allele determined by fliC gene sequencing; H – expression of flagellar type confirmed by reaction to antisera. To avoid confusion in text, flagellar type will be denoted as H, regardless whether it was determined by sequencing or serologic typing.
c. b. d. – bloody diarrhea; HUS – hemolytic uremic syndrome; scours – neonatal calf diarrhea.
d. Year of isolation.
e. ST – sequence type based on MLST of 7 housekeeping genes (aspC, clpX, fadD, icdA, lysP, mdh, and uidA).
f. MDCH – Michigan Dept. of Community Health.
Figure 1.
Phylogenetic relationships of EHEC and EPEC sequence types. The sequence types (STs) of EHEC 2 belong to a clonal group (CG 14), which is more closely related to EPEC 2 (CG 17), than EHEC 1 STs (CG 11). The phylogenetic tree was constructed using the Neighbor-joining algorithm based on the Kimura 2-parameter distance matrix of nucleotide substitution. Bootstrap confidence values were based on 1000 replicates. Only those higher than 70% are shown.
Gene content of EHEC 2 strains
Binary classification of genes as present or divergent/absent, inferred by GACK analyses of the CGH data, was used to determine the gene content of all 24 EHEC 2 strains (Table 2) and of each individual strain (Table 3). Because all CGH experiments were performed with Sakai as the reference strain, our analyses focused on probes targeting genes present in the Sakai genome. The oligo probes were classified to represent backbone genes (shared by Sakai and K-12), and Sakai-specific genes (note that the term "Sakai-specific" is used here only in comparison to K-12). The Sakai-specific genes were further classified in Sakai phage genes (phage-related genes present in Sakai but absent in K-12) and Sakai bacterial genes (non-phage-related genes present in Sakai but absent in K-12) [38]. Of the 3,696 backbone genes, 80.9% were shared by all EHEC 2 strains, whereas only 5.8% of the Sakai phage genes (n = 814) and 6.5% of the Sakai bacterial genes (n = 434) were found in every tested EHEC 2 strain. While 84.7% of the Sakai phage genes were found in at least one of the 24 EHEC 2 strains, a whole 53% of the Sakai bacterial genes were not found in any of the these strains (Table 2).
Table 2.
Percentage of Sakai genes that are present, divergent/absent or variably absent or present (VAP) in all 24 EHEC 2 strains.
| Backbone (shared with K-12) n = 3696 | Sakai-specific | ||
| phage-related n = 814 | Bacterial n = 434 | ||
| Present | 80.9% | 5.8% 6.5% | |
| Divergent/absent | 1.1% | 9.5% 53.0% | |
| VAPa | 18.0% | 84.7% 40.5% | |
a – genes that were detected in at least one of the 24 EHEC 2 strains, but not in all EHEC 2 strains.
Table 3.
Percentages of Sakai genes found in tested strains sorted by serotype.
| Serotype | Strain | Sakai genes on array n = 4,944 | Backbone (shared with K-12) n = 3,696 | Sakai-specific | |
| phage-related n = 814 |
Bacterial n = 434 |
||||
| O26: [h11] | DEC 9f | 78% | 94% | 30% | 20% |
| O26:H11 | DEC 10e | 82% | 96% | 51% | 22% |
| O26:H11 | F5863 | 84% | 97% | 56% | 25% |
| O26:H11 | 97–3250 | 85% | 97% | 65% | 23% |
| O26: [h11] | 413/89-1 | 83% | 96% | 56% | 20% |
| O26: [h11] | DA-22 | 84% | 97% | 57% | 24% |
| O26:H11 | 03-ST-296 | 84% | 97% | 54% | 22% |
| O26:H11 | CB 7505 | 84% | 96% | 59% | 30% |
| O26:H11 | Average | 83% | 96% | 54% | 23% |
| Stan. Dev.a | 2.2% | 1% | 10.3% | 3.2% | |
| O111: [h11] | DEC 8c | 82% | 94% | 55% | 19% |
| O111:H11 | DEC 8d | 77% | 93% | 31% | 21% |
| O111: [h8] | C408 | 82% | 95% | 49% | 24% |
| O111: [h8] | BCL71 | 83% | 95% | 58% | 24% |
| O111: [h8] | ML178190 | 82% | 95% | 52% | 23% |
| O111:H8 | W29104 | 81% | 95% | 48% | 23% |
| O111: [h8] | EK34 | 81% | 95% | 47% | 24% |
| O111:H8 | EK35 | 80% | 94% | 49% | 23% |
| O111:H8 | Average | 82% | 95% | 51% | 24% |
| Stan. Dev. | 1% | 0.4 | 4% | 0.5% | |
| O118: [h16] | RW2030 | 84% | 96% | 58% | 23% |
| O118: [h16] | RW1302 | 82% | 95% | 56% | 19% |
| O118:H16 | 666/89 | 83% | 95% | 57% | 21% |
| O118:H16 | 05482 | 82% | 96% | 53% | 22% |
| O118:H16 | EK36 | 83% | 96% | 53% | 21% |
| O118:H16 | EK37 | 84% | 97% | 55% | 25% |
| O118:H16 | Average | 83% | 96% | 55% | 22% |
| Stan. Dev. | 0.9% | 0.8% | 2.1% | 2% | |
| O153: [h11] | 02–3751 | 84% | 97% | 60% | 24% |
| O15: [h11] | RDEC-1 | 80% | 94% | 42% | 22% |
| O55:H7 | 97–3256 | 84% | 97% | 33% | 70% |
a – Stan. Dev., standard deviation.
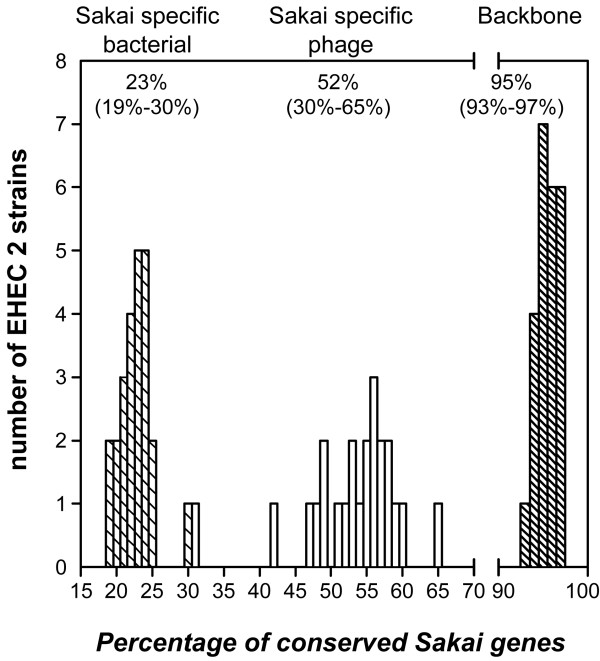
In each individual EHEC 2 strain, approximately 95% of the 3,696 backbone genes were found (Table 3, Figure 2), with little variation (95.5% ± 1.2%, range 93% – 97%). In contrast, about 52% of the Sakai phage genes were found, but with a much greater variability across EHEC 2 strains (52.1% ± 8.2%, range 30% – 65%). This may be an over estimation of Sakai phage gene distribution in EHEC 2, as 231 of the 814 phage gene probes analyzed had multiple phage gene targets in the Sakai genome, based on in silico analysis of probe specificity. Sakai bacterial genes were found less frequently in EHEC 2 strains (22.7% ± 2.3%, range 19% – 30%). Serotype O26:H11 showed the most interstrain variation, whereas O111:H8 and O118:H16 were more uniform with respect to Sakai gene distribution. The O55:H7 representative also had a high percentage of backbone genes (96.6%). Furthermore, 33% of the 814 Sakai phage genes and 70% of the 434 Sakai bacterial genes were conserved in O55:H7, suggesting an inverse trend relative to that observed in EHEC 2 strains (Table 3).
Figure 2.
Distribution of Sakai genes among EHEC 2 clinical strains. The three histograms represent distribution trends of three Sakai gene groups in EHEC 2 strains: Sakai bacterial genes (left histogram – hatched bars), Sakai phage genes (middle histogram, open bars), and backbone genes (right histogram – hatched bars). The levels of Sakai gene content conservation were calculated for each EHEC 2 strain by dividing the number of Sakai genes, from a particular gene group, found in a strain by the total number of Sakai genes from the respective gene group, represented on the oligoarray; these values were expressed as percentages. Each bar represents the number of EHEC 2 strains that were found to have the same percentage of Sakai gene content conservation. Each strain is represented on each histogram and the bars in each histogram add up to 24, the total number of strains investigated. One exception is the bar representing Sakai phage gene content conservation in strain DEC9f, which is hidden by the hatched bar representing the Sakai bacterial gene content conservation in strain CB7505. As can be seen in Table 3, strain DEC9f has 30% of Sakai phage genes and strain CB7505 has 30% of Sakai bacterial genes, causing the bars to overlap. Numbers above each plot represent the average for each group of genes and the range of the distribution is given in parentheses.
Identification of potential EHEC-specific genes
From the 1,248 Sakai-specific genes represented on the microarray, 152 (12.2%) were conserved in 23 of the 24 EHEC 2 strains; 102 of these were phage-related. Sixty-four genes encode hypothetical proteins of unknown function, and the remainder consisted mostly of genes responsible for various prophage and other mobile element functions. Nucleotide sequences of these 152 genes were compared against five non-EHEC pathogenic E. coli (536, APEC O1, B171, CFT073, UTI89) and six Shigella (Sf2a 2457T, Sf2a 301, Sf5 8401, Ss046, Sb227, Sd197) published genomes, using BLAST. With a minimum of 80% nucleotide sequence identity in a minimum of 80% query coverage as the cutoff value to identify conserved genes, 26 of the 152 genes were not found in any of the 11 queried non-EHEC genome sequences. The 26 gene sequences were then "BLASTed" against the entire GenBank database with the same cutoff value. Only three of these 26 genes were not found in any other organisms and therefore could be considered as specific to EHEC strains: ECs1561 (Sakai prophage (Sp) 6); ECs1763, and ECs1822 (Sp 9). All three genes encode hypothetical proteins of unknown function.
Genomic relatedness of EHEC 2 strains
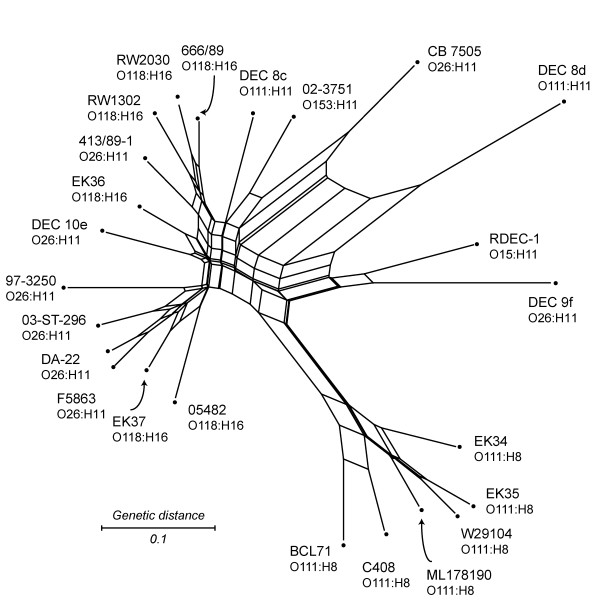
We used the split decomposition method to infer the strain relatedness based on gene content data. We first analyzed all the 4,800 genes whose probe intensities were higher than those for negative controls. As expected, the analysis showed a network like phylogeny (Figure 3), in which the parallel edges reflected incompatible signals in the data that were indicative of parallel gene gain/loss due to multiple transduction events or past recombination. All O111:H8 strains were clustered closely and branched away from the remaining EHEC 2 strains, which formed a loose cluster without any recognizable concordance to serotypes, hosts, or locations (Figure 3). The pairwise homoplasy index (PHI) [43], generated in Splitstree, confirmed that there was significant evidence of recombination (p-value = 0.0).
Figure 3.
Split decomposition analysis of Sakai genes in 24 EHEC 2 strains. The network was generated based on the presence/absence of 4800 Sakai genes among 24 EHEC 2 strains. 144 genes were excluded because their probe intensities were below those of randomized negative controls in the various Sakai/EHEC 2 hybridizations. Node labels refer to strain names (listed in Table 1). Parallel edges represent phylogenetic incompatibilities in the data set, which are indicative of parallel gene gain/loss by multiple transduction events. The network was generated in Splitstree 4.3, using neighbor net with the uncorrected p distance. Scale bar represents number of gene differences (present or divergent/absent) per gene site.
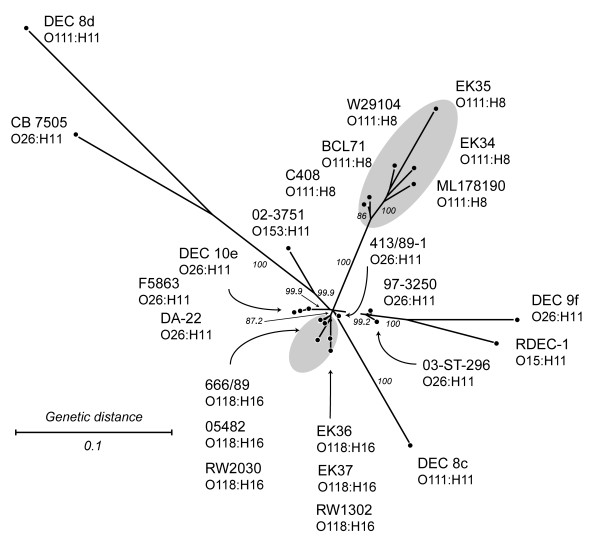
Among the 4,800 genes whose probe intensities were higher than those for negative controls, 70.8% were found to be either present or divergent/absent in all 24 strains, and therefore, phylogenetically uninformative. Compatibility analysis of the 979 parsimoniously informative (PI) genes identified 147 PI genes to be phylogenetically compatible with each other, but not compatible with the rest of the PI genes (the distribution of these genes is shown in Additional file 1). For the second split decomposition analysis, these 147 genes were combined with 421 singleton genes (genes found present or divergent/absent in only one of the 24 EHEC 2 strains). Singletons were added to generate terminal edges of the network and to help distinguish strain-specific changes. The analysis with this set of genes showed a more tree like phylogeny with a better separation of EHEC 2 strains (Figure 4). Six O111:H8 strains and six O118:H16 strains formed two tight and distinct clusters, while the twelve O26:H11, O111:H11, O153:H11, and O15:H11 strains were dispersed throughout the network. The O111:H8 cluster was visibly distinct from the rest, reiterating its particular pattern of gene content conservation across all 4,800 genes (Figure 3). The two O111:H11 strains did not cluster with O111:H8 strains, which is not unusual since the O111 serogroup has been suggested to include several lineages [44]. In this analysis, the O118:H16 strains appear to be more closely related to most of the O26:H11 strains than any other EHEC 2 serotype. Nonetheless, there was a short edge separating the O118:H16 serotype from O26:H11, followed by strain-specific splits within O118:H16 that were based on singleton genes. The eight O26:H11 strains did not cluster together, suggesting that strains of this serotype are considerably more diverse than O111:H8 and O118:H16 strains.
Figure 4.
Split decomposition analysis of compatible parsimony informative genes and singleton genes in 24 EHEC 2 strains. Gray ovals encompass serotype-specific clusters of O118:H16 and O111:H8 strains. Node labels refer to strain names (listed in Table 1). The network was generated in Splitstree 4.3, using neighbor net with the uncorrected p distance. Scale bar represents number of gene differences (present or divergent/absent) per gene site. Percent bootstrap confidence values based on 1000 replicates are shown for selected edges.
Prophages
To visualize gene content of the 814 Sakai phage genes within the EHEC 2 clonal group, we classified these genes by Sakai phage groups (Sakai prophages Sp1–18, and prophage-like elements SpLE1–6) and sorted the genes in each group by chromosomal order (based on ECs numbers). This classification does not necessarily infer that these genes are present in EHEC 2 within the same phage or order as they are in Sakai, but simply allows an assessment of gene content variation of laterally acquired genes known to be linked in the Sakai chromosome. Dendrograms based on pairwise comparison of gene content were used to identify EHEC 2 strains with similar gene content (Figure 5 and Additional file 2). Overall, there was no common pattern of gene distribution for all phage groups (Figure 5), which was also implied by additional split decomposition networks (data not shown). Some similarity was detected among O111:H8 strains for Sp5, Sp15 and Sp8 genes, with more Sp5 and Sp15 genes being conserved in the O111:H8 serotype than in other EHEC 2 strains. Conversely, Sp8 was well-conserved in all but the O111:H8 strains (data not shown), in which Sp8 genes were virtually absent except for two short gene segments, ECs1638–43 and ECs1656–63, which encode tail and hypothetical proteins, respectively.
Figure 5.
Gene content of Sakai phage genes and the LEE island in EHEC 2 strains. Sakai phage genes inferred as present or divergent/absent were grouped and sorted according to the Sakai annotation. Colormaps, with dendrograms, of individual phages were generated in R software (v 2.4.0.), using the 'gplots' package (v 2.3.2). Present genes are depicted as black, absent/divergent as white. Gray squares symbolize genes that have been classified as present after the cutoff was relaxed for 20%, representing a 'low' level of gene divergence. Dendrogram labels refer to strain names (Table 1). Labels with asterisks in the Sp15 and Sp5 colormaps refer to strains that were positive for stx1 and stx2 genes, respectively. Labels with open boxes in the LEE colormap represent animal strains. Arrows and numerals atop the LEE colormap represent operons and the direction of their transcription. The ECs numbers for the phage genes depicted, and the distribution of these genes, are provided in Additional file 2. Sp – Sakai prophage, SpLE – Sakai prophage-like element, TAI – tellurite resistance and adherence island.
Stx converting prophages
The CGH data confirmed the stx1/stx2 profile of the EHEC 2 strains determined by PCR. In Sp15 (stx1-prophage), a block of genes at the beginning of the phage (ECs2940–2952) was conserved in most strains (Figure 5). These genes encode tail proteins and the putative outer membrane protein Lom precursor (ECs2942). Adjacent is a group of genes (ECs2953–2963) encoding two tail proteins, a putative terminase large subunit and several unknown proteins, which are fully conserved in O111:H8 strains but almost completely divergent/absent in the rest. Two regions in the Sp15 phage, ECs2984–2988 and ECs2998–3006, were well conserved in all strains positive for the stx1 gene, except in O111:H8 strains. Excisionase and integrase genes (ECs3012 and ECs3013) were divergent/absent in most of the EHEC 2 strains. Overall, the gene content of Sp15 in strains negative for the stx1 gene was different from those in stx1 positive strains (Figure 5).
Strains positive for the stx2 gene, mostly representing serotype O111:H8, had more Sp5 (stx2-phage) genes. Integrase and excisionase genes (ECs1160 and ECs1161), and the block of genes at the beginning of the phage, ECs1160–1187, were missing from most strains. The rest of Sp5 genes, which encode replication proteins O and P, NinE and NinG, Shiga toxin 2, antirepressor proteins, antitermination protein Q, outer membrane precursor proteins, terminases, tail proteins, and a number of hypothetical proteins, were present in five of the six O111:H8 strains as well as in the O26:H11 strain containing both stx1 and stx2 (Figure 5).
Locus of enterocyte effacement (LEE) island
Of the 41 genes in the Sakai LEE island that are located on SpLE4, all except escU were present in the O55:H7 strain. This includes genes that were categorized as present after the initial GACK cutoff was relaxed by 20%. Since dye-swap genomic microarrays represent competitive hybridizations between two populations of DNA, there were instances when a small difference in the nucleotide sequence of the tested strain resulted in weaker probe signal intensity. For example, both of the two known SNPs present between the variable regions of γ intimin in O55:H7 and O157:H7 [45] are located in the middle region of the 70-mer probe for eae. Hence the signal intensity for this gene was just below the cutoff (gray shading in Figure 5). Based on the level of divergence of EHEC 2 LEE genes from O157 LEE genes, strains clustered into two major groups (Figure 5). The top group of the dendrogram is composed of human strains, which have a high level of similarity to O157 LEE genes, whereas the bottom cluster represents 11 animal and 3 human strains that have a lower level of similarity to the O157 LEE genes. The level of divergence was also found to be heterogeneous between LEE operons (Table 4). The genes that encode the type III secretion system (TTSS), escRSTUCJVNDF, were detected in 14 to 24 strains, with the exception of escR and escC, which were found in 11 and 5 strains, respectively. The needle filament gene, espA, was present in 23 strains, whereas espB and espD were divergent/absent in all. The tir and γ intimin genes were also divergent/absent in EHEC 2; the γ intimin was conserved only in the O55:H7 representative, an expected result because the 70-mer probe was designed to detect the variable (allele-specific) part of eae.
Table 4.
Conservation of O157 LEE operons in a set of 24 EHEC 2 strains.
| LEE1 (9)a | LEE2 (6) | LEE3 (7) | LEE5 (3) | LEE4 (8) | |
| Humanb | 8.8 ± 0.6 | 3.1 ± 1.7 | 4.8 ± 1.1 | 1.0 ± 0.0 | 4.4 ± 0.5 |
| Animalc | 3.2 ± 1.2 | 0.6 ± 0.9 | 2.7 ± 0.9 | 0.1 ± 0.2 | 3.8 ± 0.9 |
a. The number of genes in each operon is given in parentheses.
b. Refers to the 10 human isolates in the top cluster of the dendrogram in the LEE image in Figure 5, not including O55:H7. Values represent average number of genes in an operon, with standard deviation.
c. Refers to the 11 animal and 3 human isolates in the bottom cluster of the dendrogram in the LEE image in Figure 5. Values represent average number of genes in an operon, with standard deviation.
Other phage gene groups
Most genes from SpLE1, which encodes the tellurite resistance and adherence island (TAI), were divergent/absent from two EHEC 2 strains and from the O55:H7 representative, but present in the rest of the EHEC 2 strains (Figure 5). The diverse trend in retention or loss of laterally acquired genes was emphasized by the arrangement of Sp10 genes. CGH data inferred three patterns of Sp10 gene content conservation in EHEC 2 (Figure 5). In the first 14 strains (top to bottom), Sp10 genes were found to be present or divergent/absent in an en bloc fashion. The middle branch of the dendrogram represents six strains in which virtually all Sp10 genes were present. In the remaining five strains, Sp10 genes appeared to have a mosaic structure with individual genes present or divergent/absent. In contrast, Sp18 was either entirely divergent/absent or nearly completely present. There was no correlation between the distribution of Sakai phage genes in EHEC 2 and geographic location of the EHEC 2 isolates.
Non-LEE encoded effectors
The gene content of non-LEE encoded effectors, which are predicted to be secreted by the LEE-encoded TTSS [42] in EHEC 2, varied from totally divergent/absent to present in every strain (Additional file 3). Genes espY1, nleD, espX2, espY4, espL3', espX3', espL4, and nleB2-1 were divergent/absent from EHEC 2, whereas a set of 15 genes (espX1, espX5, espX6, espY3, espK, nleA, nleE, nleG, nleG2-2, nleG6-1, espM1, espM2, espR1, espL1, and espW) were present in at least 22 EHEC 2 strains. The nleG7 gene, which was recently found to be conserved in a group of non-O157 EHEC strains [46], was also divergent/absent in all EHEC 2 examined in this study.
Discussion
Comparative analysis of genomes from 17 commensal and pathogenic E. coli strains has revealed a diverse species 'pan-genome', while the E. coli 'core conserved' genome was calculated to be about one-half of the genome of a given E. coli isolate [47]. Although EHEC utilize similar virulence mechanisms, this pathotype is comprised of phylogenetically distinct lineages that vary in their ability to cause disease in both humans and animals. Clearly, the genome of a single strain cannot reflect how the genomic diversity among EHEC strains influences pathogenesis of the EHEC population. Because no strains from the EHEC 2 clonal group have been sequenced, the genetic variability of 24 EHEC 2 strains were examined in relation to the distribution of genes from O157:H7 Sakai, which belongs to the EHEC 1 clonal group. The Sakai genome was used in this study, as its annotation is suggested to include more strain-specific genes compared to EDL933 [47]. Genes specific to the EHEC 2 group have yet to be described. Some genes shared with Sakai might have been missed in our study, if the gene sequence had diverged to a point where the 70-mer oligonucleotide probes and the stringency of competitive hybridization preclude detection. Although this study allowed screening of known genes only, the gene content data still offered new insight on strain relatedness and the distribution and subsequent diversification of mobile elements within the EHEC 2 clonal group.
The CGH data presented here indicate that there are two distinct trends, which reflect the bacterial (vertical) and phage (lateral) origin of genes, impacting the genomic divergence of EHEC 2. Virtually the entire set of backbone genes was present within the EHEC 2 clonal group (Tables 2 and 3). CGH inferences pertaining to the distribution of backbone genes can vary depending on array type, sample size, and strain diversity [46]. For example, Anjum et al. have proposed that the O26 serogroup exhibits greater genetic homogeneity than was observed in our study [48]; however, the microarray platform used in that study was limited to the genome of K-12 MG1655. Despite these differences, the degree of conservation among backbone genes in this CGH investigation was similar in previous studies [46,49,50]. The distribution of Sakai-specific genes in EHEC 2 was, not surprisingly, noticeably lower than that of the backbone, which restates established findings about intraspecies genomic variability [40,51,52]. The conservation of Sakai phage genes was, however, found to be more than 2-fold higher when compared to Sakai bacterial genes (Figure 2 and Table 3). In O55:H7, the inferred ancestor of O157:H7 [53], the proportion of Sakai phage to bacterial gene conservation was opposite from the proportion observed in EHEC 2; this suggests that Sakai bacterial genes have been vertically acquired from the O55:H7 progenitor and are not disseminated among the EHEC 2 clone. Cursory assessment of K-12-specific genes suggests a homogenous distribution in EHEC 2, with less than half of the genes present; most K-12 phage-related genes were found to be uniformly divergent/absent from the entire EHEC 2 population (Additional file 4). Assessing the conservation of K-12 specific genes was, however, beyond the scope of this study, as K-12 MG1655 is a non-pathogenic laboratory-derived strain that is distantly related to EHEC (Figure 1).
The increased presence of Sakai phage genes in the EHEC 2 group compared to Sakai bacterial genes reveals independent acquisition and exchange of similar mobile elements. For example, of the 152 Sakai-specific genes present in EHEC 2, only 26 genes were not found in 11 completed non-EHEC E. coli and Shigella spp. genomes. About one-half of the 26 "EHEC only" genes were found in stx1-encoding phages BP-4795 and CP-1639 from STEC O84:H11 and O111:H-, respectively [54,55]. Sakai genes identified by BLASTN as present on BP-4795 are disseminated on phages Sp6, 9, 10, and 12, which is in agreement with the evidence for recombination between phages [56]. Although the number of phage genes shared by all tested strains was low, the percentage of those that were VAP was high (Table 2), which may reflect sequence heterogeneity in prophage genomes with similar modular structures [54,56,57], and not true absence of genes.
Phylogenetic network analysis implied a serotype-specific uniformity of O111:H8 strains, unlike other EHEC 2 strains (Figure 3), which can also be inferred from the arrangement of Sakai phage genes in O111:H8 strains (Figure 5). Interestingly, these six EHEC 2 representatives are the only strains with the θ intimin allele while the remaining eighteen EHEC 2 strains had β intimin, as determined by PCR-based RFLP typing of eae; the method for eae typing was described previously [58]. By contrast, members of the EHEC 1 clonal group (i.e., O157:H7 and O55:H7) typically had the γ allele. Although intimin θ has been found in an atypical EPEC O55:H7 and a non-EHEC 2 strain (GenBank Acc. No. AJ833638 and AF253561), O111:H8 is, to our knowledge, the only EHEC 2 serotype with this intimin allele, providing further support for the hypothesis that O111:H8 represents a distinct grouping.
Based on the distinguishing distribution of Sakai genes (Figures 3 and 4), serotype O26:H11 appears to be considerably more diverse compared to the distinct and more uniform O111:H8. This suggests that the genetic make-up of O26:H11 is such that it allows more frequent lateral exchange of DNA elements, which can result in acquisition of novel fitness and virulence genes by O26:H11 more commonly than by other EHEC 2. For example, O26:H11 possess the Yersinia spp. high pathogenicity island (HPI) that encodes the iron-uptake siderophore yersiniabactin and the pesticin receptor, whereas other EHEC serotypes, including O157:H7, O111:H-, O103:H2, and O145:H-, do not have this HPI [59]. The diversity of O26:H11/H- has also been implied with other methods [60].
A proportion of the EHEC 2 hybridization data (15% of the PI genes) were identified as genes that are phylogenetically compatible with each other, i.e., having no homoplasy. Although this represents a small number of genes, it is remarkable that the distribution pattern grouped EHEC 2 O111:H8 and O118:H16 strains by serotype (Figure 4). The pathogenic E. coli used in this study represent tips of phylogenetic branches, where high frequencies of recombination strongly impact the shaping of genomic content [61] and eventually lead to erosion of the phylogenetic signal between clonal complexes [62]. Thus, the set of genes shared with EHEC 1 O157:H7 whose pattern of presence and absence in EHEC 2 infers compatibility and is not random, but coincides with serotype, warrants further investigation.
The heterogeneity of Stx phages has been demonstrated [57,63], even within the O157:H7 lineage itself [64,65], so it is not unexpected to find such variation between different EHEC 2 strains. In addition, Ogura et al. propose that Stx phages have alternative integration sites in EHEC 2 [46]; this may explain our lack of detection of integrase genes, as integration site specificity is dependent on the alignment of the phage integrase with the attachment sequence in the bacterial chromosome [66]. Strains that were stx negative in our study were, nevertheless, found to carry genes from the Sp15 and Sp5 phages, which is a common effect of frequent modular shuffling of sequences between phages of related enteric hosts [56,67,68]. The significance of the unique conservation patterns of Sp10 and Sp18 phage genes is not clear. Sp10 is perhaps more conserved as it harbors non-LEE effector genes [42], all 3 of which were detected in at least 22 out of 24 EHEC 2 strains. Absence of the entire Sp18 was also detected among O157:H7 strains [65], one of which belongs to a hyper-virulent lineage of the O157:H7 population [69].
Incongruent divergence of LEE operons has been previously suggested. Studies indicate that this island is a dynamic region [70], and that different selective pressures act on different parts of the LEE [71]. The sequence diversity of the LEE, both at the nucleotide and amino acid level, increases along the length of the island from the LEE1 to the LEE4 operon [71,72]. A comparable trend can be observed in the CGH data presented here, as there was greater conservation of the content of genes that encode the secretion apparatus (LEE1–3). However, differences in the content of O157:H7 Sakai LEE genes between human and animal EHEC 2 strains of the same serotype (Figure 5 and Table 4) suggest that the LEE has diverged between EHEC 2 strains in a host dependent manner, possibly due to host species adaptive pressure. This result was not expected and its implications are not supported by the current literature. Multiple, parallel acquisitions of the LEE by different clonal groups have been inferred [37,73-75].
Muniesa et al. suggest that the LEE genes associated with serogroup O26 are present more commonly in STEC than the LEE genes associated with EHEC O157:H7 or EPEC O127:H6 [76]. Yet, there is no clear evidence to support the hypothesis that LEE divergence within a lineage results from positive adaptive pressure in different host species. In fact, when several LEE genes from strain RDEC-1 were compared to those from other AEEC, the variation appeared to be associated with evolutionary lineage and not host specificity [77]. Even so, given the heterogeneous diversification of this island and the recent inference about host-specific expression of espA and eae in O157:H7 [78], it would be interesting to compare complete LEE sequences from a larger sample of EHEC 2 strains of human and animal origin.
Conclusion
Here, we present an assessment of the gene content of a set of EHEC 2 clinical strains of animal and human origin, isolated from the USA and Europe. The small subset of phylogenetically compatible genes represent potential markers that will aid in the investigation of the relatedness and cladogenesis of the EHEC 2 clonal group. In this study, serotype O26:H11, the most frequent EHEC 2 serotype associated with overt disease, represented the most diverse EHEC 2 population. Compared to the more homogeneous O111:H8 strains, O26:H11 strains may have an increased propensity to laterally exchange DNA, which may ultimately give rise to hyper-virulent lineages within EHEC 2 O26:H11. Furthermore, the identification of several EHEC-specific genes could potentially be used as novel genetic markers to identify strains belonging to this pathotype.
Methods
Bacterial strains and DNA isolation
Since genome sequences for tested strains are not available, two-color hybridizations between sequenced strains of E. coli O157:H7 RIMD 0509952 (Sakai) [38] and K-12 MG1655 [79] were used as references. A total of 24 EHEC 2 strains including serotypes O26:H11 (n = 8), O111:H8 (n = 6), O111:H11 (n = 2), O118:H16 (n = 6), O153:H- (n = 1), and O15:H11 (n = 1), originally isolated from human and animal cases of STEC-associated disease, were used in this study and were selected based on the serotype and source (Table 1) [6,33,80-90]. The study also included an EHEC 1 O55:H7 strain, isolated from a human diarrhea case. Bacterial DNA was prepared from overnight LB cultures grown at 37°C using the Puregene genomic DNA isolation kit (Gentra Systems, Minneapolis, MN).
Multilocus sequence typing (MLST) and Shiga toxin (Stx) genes
The detailed MLST protocol and multiplex PCR conditions for characterizing the Stx genes (stx1/stx2) can be found at the STEC Reference Center website http://www.shigatox.net. Briefly, MLST was performed on seven conserved housekeeping genes (aspC, clpX, fadD, icdA, lysP, mdh, and uidA), and sequence type (ST) assignments were made based on phylogenetic analyses of the concatenated sequences.
Oligonucleotide arrays
The Qiagen (Valencia, Calif.) spotted multi-genome arrays containing probes specific for 5,978 ORFs from E. coli K-12 MG1655, O157:H7 Sakai and EDL933 were utilized. Of these probes, a total of 5,943 were 70-mer oligonucleotides and 35 ranged from 41–69 bp. The probes were printed in duplicate on UltraGaps glass slides (Corning Inc., NY) at the Research Technology Support Facility at Michigan State University. The array also contained 384 spots representing 12 randomized negative control 70-mer probes. All probes were assigned ORF designations (b- = MG1655, ECs- = Sakai, or Z- = EDL933 numbers) or intergenic region labels based on the RefSeq database available on the National Center for Biotechnology Information (NCBI) website [91].
In silico analysis of microarray probe specificity
To verify the probes with the up-to-date genome annotations, we compared all 5,990 probe sequences against the three E. coli genomes (MG1655, Sakai, and EDL933) by BLASTN available on NCBI, and recorded the two highest hits for every probe (top hit and second hit) for each genome. A probe was considered to be specific for a target when the top hit demonstrated ≥ 80% identity to the probe sequence stretch in the strain. Probes with nonspecific hybridization and multiple target hybridizations within MG1655 or Sakai DNA were excluded from the data analysis of MG1655 and Sakai hybridizations. These included probes that had multiple top hits with 75% overall identity or probes that had multiple top hits between 50% and 75% of overall identity with alignments containing a stretch of nucleotides with 100% identity, in which the stretch was 20% of the probe length. With respect to the MG1655 and Sakai genomes, out of 5,978 probes, 12 had no target (EDL933 specific), 731 showed nonspecific hybridization or had multiple targets, and 5,235 matched single genome targets. Of these, 3,803 targeted both genomes, with 1,002 targeting only Sakai and 430 targeting only K-12.
DNA labeling and microarray hybridization
Genomic DNA was sheared into 500 to 5,000 bp fragments in a cup sonicator (Heat Systems Ultrasonics W-225, 20 KHz, 200 W) and 250 ng of sheared DNA was labeled with aminoallyl-dUTP (Sigma, St. Louis, Mo.) using the Invitrogen (Carlsbad, Calif.) DNA labeling system, as previously described [40]. Equal amounts of DNA from Sakai and test strains were suspended and combined in a final volume of 44 μL of SlydeHyb Buffer #1 (Ambion, Inc., Austin, TX). Qiagen E. coli spotted oligoarrays were hybridized and washed according to the manufacturer's instructions for hybridization using coverslips. Test strains were hybridized twice with Sakai as a reference: once with the Cy5 labeled test strain and Cy3 labeled Sakai and once with the Cy3 labeled test strain and Cy5 labeled Sakai to correct for dye incorporation bias.
Data collection and analyses
Arrays were scanned with the Genepix 4000B array scanner (Axon Instruments, Union City, Calif.) and probe intensities (median pixel intensities) were retrieved using Genepix 6.0 (Axon Instruments). Data quality was assessed by viewing plots of M versus A [M = log2 (test/reference); A = log2 (test × reference)] and by checking for spatial effects with Genepix 6.0 and GeneTraffic (Iobion, La Jolla, Calif.) as described previously [40]. Because genome sequences of tested strains were not available, microarray data were not normalized to avoid biasing the gene content of tested strains. Instead, microarray images showing spatial bias were discarded and hybridizations were repeated until control parameters were appropriate. Duplicate probes for each gene were averaged prior to analyses. Probes with median pixel intensities higher than the median of the randomized negative controls were analyzed as the distribution of the two-color signal ratios using the "GACK" program [92]. Analysis of the log2 (test strain/reference strain) distribution (GACK1) as well as of the reciprocal ratio, log2 (reference strain/test strain) (GACK2), were performed for Sakai versus MG1655 hybridizations to determine a cutoff. Genes with a GACK1 value of ≥ 0.1 were classified as present, whereas genes with a GACK1 value of < 0.1 were classified as divergent/absent. At this cutoff, maximum sensitivity (98.8%) and specificity (96%) were achieved for the MG1655/Sakai dye-swap hybridizations, and therefore, this cutoff was used to interpret the data from Sakai versus EHEC 2 hybridizations. The term 'present' is used to indicate that a gene was detected by CGH, and does not necessarily imply that the whole gene is conserved or functional; likewise, the term 'divergent/absent' indicates that a gene was not detected by CGH.
Phylogenetic analyses
Strains were assigned to clonal groups based on STs and bootstrap analyses as described previously [36,93]. A neighbor-joining tree of the concatenated MLST sequences was constructed using the Kimura 2-parameter distance method with 1000 bootstrap replications in MEGA 3.1 [94]. The tree includes other enteropathogenic E. coli (EPEC) and EHEC STs as well as the lab-derived K-12 (ST173) and the uropathogenic E. coli CFT073 (ST27) for comparison; an E. albertii strain was used as the outgroup. For phylogenetic analyses of the microarray data, a total of 144 genes (from all array hybridizations) with probe intensities below those of negative controls were excluded from the set of 4,944 genes. Neighbor-net phylogenies highlighting the distribution of Sakai genes in EHEC 2 strains, for which the presence or absence of genes was coded as 0 (divergent/absent) or 1 (present), were constructed using the uncorrected p distance in Splitstree 4.3 [95]. The number of Sakai genes whose distribution in EHEC 2 was parsimoniously informative were determined in MEGA 3.1 [94], and the set of Sakai genes in EHEC 2 whose distribution was compatible with a single phylogeny was identified using the clique module of PHYLIP [96].
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
GSA designed the study, collected and analyzed CGH data, and drafted the manuscript. DWL performed intimin and stx typing, phylogenetic analyses and helped to draft the manuscript. LMW participated in design and analysis of CGH data and WQ performed in silico verification of microarray probe specificity. TSW participated in the design and coordination of the study, conducted the phylogenetic analyses, and helped draft the manuscript. The first four authors read and approved the final manuscript; TSW had approved an earlier draft of the manuscript (deceased, December 5, 2008).
Supplementary Material
Distribution of phylogenetically compatible genes in EHEC 2, determined with the clique program in the PHYLIP package. Conserved genes have a value of 1 and divergent/absent have a value of 0.
Genes in EHEC 2 whose distribution was used to generate colormaps in Figure 5. Conserved genes have a value of 1, divergent/absent genes have a value of 0 and genes that have a value of 0.5 were inferred as conserved after the GACK cutoff was relaxed by 20%.
Distribution of 49 non-LEE effector genes in EHEC 2. Conserved genes have a value of 1, divergent/absent genes have a value of 0 and genes that have a value of 0.5 were inferred as conserved after the GACK cutoff was relaxed by 20%.
Distribution of K-12-specific genes in EHEC 2. Conserved genes have a value of 1 and divergent/absent genes have a value of 0.
Acknowledgments
Acknowledgements
The authors thank Shannon Manning, James Riordan, Sivapriya Kailasan Vanaja, Linda Mansfield, Martha Mulks, and Jillian Tietjen for critically reviewing earlier versions of the manuscript; Lindsey Ouellette for technical assistance with MLST; and those investigators who supplied strains for use in the study. This project was funded in part by the MSU foundation and the NIAID, NIH, DHHS, under NIH research contract N01-AI-30058 (TSW), which supports the STEC Center. The authors wish to dedicate this work to the memory of Dr. Thomas S. Whittam.
Contributor Information
Galeb S Abu-Ali, Email: abualiga@cvm.msu.edu.
David W Lacher, Email: David.Lacher@fda.hhs.gov.
Lukas M Wick, Email: lukas.wick@biosynth.ch.
Weihong Qi, Email: weihong.qi@fgcz.ethz.ch.
Thomas S Whittam, Email: abualiga@cvm.msu.edu.
References
- Donnenberg MS. Introduction. In: Donnenberg M, editor. Escherichia coli, Virulence Mechanisms of a Versatile Pathogen. San Diego: Academic Press; 2002. pp. xxi–xxv. [Google Scholar]
- Kaper JB, Nataro JP, Mobley HL. Pathogenic Escherichia coli. Nat Rev Microbiol. 2004;2:123–140. doi: 10.1038/nrmicro818. [DOI] [PubMed] [Google Scholar]
- FoodNet Surveillance Report for 2004 (Final Report) FoodNet Foodborne Diseases Active Surveillance Network CDC's Emerging Infections Program. 2004. http://www.cdc.gov/foodnet/annual/2004/Report.pdf accessed 2008.
- Thorpe CM, Ritchie JM, Acheson DWK. EHEC and other Shiga Toxin producing Escherichia coli. In: Donnenberg M, editor. Escherichia coli: Virulence mechanisms of a versatile pathogen. San Diego: Academic Press; 2002. pp. 119–154. [Google Scholar]
- Allerberger F, Friedrich AW, Grif K, Dierich MP, Dornbusch HJ, Mache CJ, Nachbaur E, Freilinger M, Rieck P, Wagner M, et al. Hemolytic-uremic syndrome associated with enterohemorrhagic Escherichia coli O26:H infection and consumption of unpasteurized cow's milk. Int J Infect Dis. 2003;7:42–45. doi: 10.1016/S1201-9712(03)90041-5. [DOI] [PubMed] [Google Scholar]
- Fey PD, Wickert RS, Rupp ME, Safranek TJ, Hinrichs SH. Prevalence of non-O157:H7 shiga toxin-producing Escherichia coli in diarrheal stool samples from Nebraska. Emerg Infect Dis. 2000;6:530–533. doi: 10.3201/eid0605.000513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karch HH, Hans-Iko , Bockemühl J, Schmidt H, Schwarzkopf A, Lissner R. Shiga Toxin-Producing Escherichia coli Infections in Germany. Journal of Food Protection. 1997;60:1454–1457. doi: 10.4315/0362-028X-60.11.1454. [DOI] [PubMed] [Google Scholar]
- Beutin L, Bulte M, Weber A, Zimmermann S, Gleier K. Investigation of human infections with verocytotoxin-producing strains of Escherichia coli (VTEC) belonging to serogroup O118 with evidence for zoonotic transmission. Epidemiol Infect. 2000;125:47–54. doi: 10.1017/S0950268899004094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beutin L, Zimmermann S, Gleier K. Human infections with Shiga toxin-producing Escherichia coli other than serogroup O157 in Germany. Emerg Infect Dis. 1998;4:635–639. doi: 10.3201/eid0404.980415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tozzi AE, Caprioli A, Minelli F, Gianviti A, De Petris L, Edefonti A, Montini G, Ferretti A, De Palo T, Gaido M, et al. Shiga toxin-producing Escherichia coli infections associated with hemolytic uremic syndrome, Italy, 1988–2000. Emerg Infect Dis. 2003;9:106–108. doi: 10.3201/eid0901.020266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerber A, Karch H, Allerberger F, Verweyen HM, Zimmerhackl LB. Clinical course and the role of shiga toxin-producing Escherichia coli infection in the hemolytic-uremic syndrome in pediatric patients, 1997–2000, in Germany and Austria: a prospective study. J Infect Dis. 2002;186:493–500. doi: 10.1086/341940. [DOI] [PubMed] [Google Scholar]
- Sayers G, McCarthy T, O'Connell M, O'Leary M, O'Brien D, Cafferkey M, McNamara E. Haemolytic uraemic syndrome associated with interfamilial spread of E. coli O26:H11. Epidemiol Infect. 2006;134:724–728. doi: 10.1017/S0950268805005455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Escherichia coli O111:H8 outbreak among teenage campers – Texas, 1999. MMWR Morb Mortal Wkly Rep. 2000;49:321–324. [PubMed] [Google Scholar]
- McCarthy TA, Barrett NL, Hadler JL, Salsbury B, Howard RT, Dingman DW, Brinkman CD, Bibb WF, Cartter ML. Hemolytic-Uremic Syndrome and Escherichia coli O121 at a Lake in Connecticut, 1999. Pediatrics. 2001;108:E59. doi: 10.1542/peds.108.4.e59. [DOI] [PubMed] [Google Scholar]
- Misselwitz J, Karch H, Bielazewska M, John U, Ringelmann F, Ronnefarth G, Patzer L. Cluster of hemolytic-uremic syndrome caused by Shiga toxin-producing Escherichia coli O26:H11. Pediatr Infect Dis J. 2003;22:349–354. doi: 10.1097/01.inf.0000059338.38673.ae. [DOI] [PubMed] [Google Scholar]
- Brooks JT, Bergmire-Sweat D, Kennedy M, Hendricks K, Garcia M, Marengo L, Wells J, Ying M, Bibb W, Griffin PM, et al. Outbreak of Shiga toxin-producing Escherichia coli O111:H8 infections among attendees of a high school cheerleading camp. Clin Infect Dis. 2004;38:190–198. doi: 10.1086/380634. [DOI] [PubMed] [Google Scholar]
- Caprioli A, Luzzi I, Rosmini F, Resti C, Edefonti A, Perfumo F, Farina C, Goglio A, Gianviti A, Rizzoni G. Community-wide outbreak of hemolytic-uremic syndrome associated with non-O157 verocytotoxin-producing Escherichia coli. J Infect Dis. 1994;169:208–211. doi: 10.1093/infdis/169.1.208. [DOI] [PubMed] [Google Scholar]
- Zoonotic non-O157 Shiga-toxin producing Escherichia coli (STEC) World Health Organization Scientific Working Group, 23–26 June: 1998; Berlin, Germany. World Health Organization, Geneva, Switzerland; 1998. Zoonotic non-O157 Shiga-toxin producing Escherichia coli (STEC) pp. 1–30. [Google Scholar]
- Bielaszewska M, Zhang W, Mellmann A, Karch H. Enterohaemorrhagic Escherichia coli O26:H11/H-: a human pathogen in emergence. Berl Munch Tierarztl Wochenschr. 2007;120:279–287. [PubMed] [Google Scholar]
- Eklund M, Scheutz F, Siitonen A. Clinical isolates of non-O157 Shiga toxin-producing Escherichia coli : serotypes, virulence characteristics, and molecular profiles of strains of the same serotype. J Clin Microbiol. 2001;39:2829–2834. doi: 10.1128/JCM.39.8.2829-2834.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elliott EJ, Robins-Browne RM, O'Loughlin EV, Bennett-Wood V, Bourke J, Henning P, Hogg GG, Knight J, Powell H, Redmond D. Nationwide study of haemolytic uraemic syndrome: clinical, microbiological, and epidemiological features. Arch Dis Child. 2001;85:125–131. doi: 10.1136/adc.85.2.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leomil L, Aidar-Ugrinovich L, Guth BE, Irino K, Vettorato MP, Onuma DL, de Castro AF. Frequency of Shiga toxin-producing Escherichia coli (STEC) isolates among diarrheic and non-diarrheic calves in Brazil. Vet Microbiol. 2003;97:103–109. doi: 10.1016/j.vetmic.2003.08.002. [DOI] [PubMed] [Google Scholar]
- Gunning RF, Wales AD, Pearson GR, Done E, Cookson AL, Woodward MJ. Attaching and effacing lesions in the intestines of two calves associated with natural infection with Escherichia coli O26:H11. Vet Rec. 2001;148:780–782. doi: 10.1136/vr.148.25.780. [DOI] [PubMed] [Google Scholar]
- Pearson GR, Bazeley KJ, Jones JR, Gunning RF, Green MJ, Cookson A, Woodward MJ. Attaching and effacing lesions in the large intestine of an eight-month-old heifer associated with Escherichia coli O26 infection in a group of animals with dysentery. Vet Rec. 1999;145:370–373. doi: 10.1136/vr.145.13.370. [DOI] [PubMed] [Google Scholar]
- Wieler LH, Bauerfeind R, Baljer G. Characterization of Shiga-like toxin producing Escherichia coli (SLTEC) isolated from calves with and without diarrhoea. Zentralbl Bakteriol. 1992;276:243–253. doi: 10.1016/s0934-8840(11)80011-3. [DOI] [PubMed] [Google Scholar]
- Mainil JG, Duchesnes CJ, Whipp SC, Marques LR, O'Brien AD, Casey TA, Moon HW. Shiga-like toxin production and attaching effacing activity of Escherichia coli associated with calf diarrhea. Am J Vet Res. 1987;48:743–748. [PubMed] [Google Scholar]
- Mercado EC, Gioffre A, Rodriguez SM, Cataldi A, Irino K, Elizondo AM, Cipolla AL, Romano MI, Malena R, Mendez MA. Non-O157 Shiga toxin-producing Escherichia coli isolated from diarrhoeic calves in Argentina. J Vet Med B Infect Dis Vet Public Health. 2004;51:82–88. doi: 10.1111/j.1439-0450.2004.00729.x. [DOI] [PubMed] [Google Scholar]
- Hall GA, Dorn CR, Chanter N, Scotland SM, Smith HR, Rowe B. Attaching and effacing lesions in vivo and adhesion to tissue culture cells of Vero-cytotoxin-producing Escherichia coli belonging to serogroups O5 and O103. J Gen Microbiol. 1990;136:779–786. doi: 10.1099/00221287-136-4-779. [DOI] [PubMed] [Google Scholar]
- Stordeur P, China B, Charlier G, Roels S, Mainil J. Clinical signs, reproduction of attaching/effacing lesions, and enterocyte invasion after oral inoculation of an O118 enterohaemorrhagic Escherichia coli in neonatal calves. Microbes Infect. 2000;2:17–24. doi: 10.1016/S1286-4579(00)00290-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoonderwoerd M, Clarke RC, van Dreumel AA, Rawluk SA. Colitis in calves: natural and experimental infection with a verotoxin-producing strain of Escherichia coli O111:NM. Can J Vet Res. 1988;52:484–487. [PMC free article] [PubMed] [Google Scholar]
- Moxley RA, Francis DH. Natural and experimental infection with an attaching and effacing strain of Escherichia coli in calves. Infect Immun. 1986;53:339–346. doi: 10.1128/iai.53.2.339-346.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naylor SW, Gally DL, Low JC. Enterohaemorrhagic E. coli in veterinary medicine. Int J Med Microbiol. 2005;295:419–441. doi: 10.1016/j.ijmm.2005.07.010. [DOI] [PubMed] [Google Scholar]
- Wieler LH, Schwanitz A, Vieler E, Busse B, Steinruck H, Kaper JB, Baljer G. Virulence properties of Shiga toxin-producing Escherichia coli (STEC) strains of serogroup O118, a major group of STEC pathogens in calves. J Clin Microbiol. 1998;36:1604–1607. doi: 10.1128/jcm.36.6.1604-1607.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen EM, Jensen C, Baggesen DL. Evidence of transmission of verocytotoxin-producing E. coli O111 from a cattle stable to a child. Clin Microbiol Infect. 2005;11:767–770. doi: 10.1111/j.1469-0691.2005.01213.x. [DOI] [PubMed] [Google Scholar]
- Whittam TS. Evolution of E. coli O157:H7 and other Shiga toxin-producing E. coli strains. In: Kaper JB, O'Brien AD, editor. E coli O157:H7 and other Shiga Toxin-Producing E coli Strains. Washington: American Society for Microbiology; 1998. pp. 195–212. [Google Scholar]
- Tarr CL, Large TM, Moeller CL, Lacher DW, Tarr PI, Acheson DW, Whittam TS. Molecular characterization of a serotype O121:H19 clone, a distinct Shiga toxin-producing clone of pathogenic Escherichia coli. Infect Immun. 2002;70:6853–6859. doi: 10.1128/IAI.70.12.6853-6859.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid SD, Herbelin CJ, Bumbaugh AC, Selander RK, Whittam TS. Parallel evolution of virulence in pathogenic Escherichia coli. Nature. 2000;406:64–67. doi: 10.1038/35017546. [DOI] [PubMed] [Google Scholar]
- Hayashi T, Makino K, Ohnishi M, Kurokawa K, Ishii K, Yokoyama K, Han CG, Ohtsubo E, Nakayama K, Murata T, et al. Complete genome sequence of enterohemorrhagic Escherichia coli O157:H7 and genomic comparison with a laboratory strain K-12. DNA Res. 2001;8:11–22. doi: 10.1093/dnares/8.1.11. [DOI] [PubMed] [Google Scholar]
- Perna NT, Plunkett G, 3rd, Burland V, Mau B, Glasner JD, Rose DJ, Mayhew GF, Evans PS, Gregor J, Kirkpatrick HA, et al. Genome sequence of enterohaemorrhagic Escherichia coli O157:H7. Nature. 2001;409:529–533. doi: 10.1038/35054089. [DOI] [PubMed] [Google Scholar]
- Wick LM, Qi W, Lacher DW, Whittam TS. Evolution of genomic content in the stepwise emergence of Escherichia coli O157:H7. J Bacteriol. 2005;187:1783–1791. doi: 10.1128/JB.187.5.1783-1791.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks JT, Sowers EG, Wells JG, Greene KD, Griffin PM, Hoekstra RM, Strockbine NA. Non-O157 Shiga toxin-producing Escherichia coli infections in the United States, 1983–2002. J Infect Dis. 2005;192:1422–1429. doi: 10.1086/466536. [DOI] [PubMed] [Google Scholar]
- Tobe T, Beatson SA, Taniguchi H, Abe H, Bailey CM, Fivian A, Younis R, Matthews S, Marches O, Frankel G, et al. An extensive repertoire of type III secretion effectors in Escherichia coli O157 and the role of lambdoid phages in their dissemination. Proc Natl Acad Sci USA. 2006;103:14941–14946. doi: 10.1073/pnas.0604891103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruen TC, Philippe H, Bryant D. A simple and robust statistical test for detecting the presence of recombination. Genetics. 2006;172:2665–2681. doi: 10.1534/genetics.105.048975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campos LC, Whittam TS, Gomes TA, Andrade JR, Trabulsi LR. Escherichia coli serogroup O111 includes several clones of diarrheagenic strains with different virulence properties. Infect Immun. 1994;62:3282–3288. doi: 10.1128/iai.62.8.3282-3288.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGraw EA, Li J, Selander RK, Whittam TS. Molecular evolution and mosaic structure of alpha, beta, and gamma intimins of pathogenic Escherichia coli. Mol Biol Evol. 1999;16:12–22. doi: 10.1093/oxfordjournals.molbev.a026032. [DOI] [PubMed] [Google Scholar]
- Ogura Y, Ooka T, Asadulghani , Terajima J, Nougayrede JP, Kurokawa K, Tashiro K, Tobe T, Nakayama K, Kuhara S, et al. Extensive genomic diversity and selective conservation of virulence-determinants in enterohemorrhagic Escherichia coli strains of O157 and non-O157 serotypes. Genome Biol. 2007;8:R138. doi: 10.1186/gb-2007-8-7-r138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasko DA, Rosovitz MJ, Myers GS, Mongodin EF, Fricke WF, Gajer P, Crabtree J, Sebaihia M, Thomson NR, Chaudhuri R, et al. The pangenome structure of Escherichia coli : comparative genomic analysis of E. coli commensal and pathogenic isolates. J Bacteriol. 2008;190:6881–6893. doi: 10.1128/JB.00619-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anjum MF, Lucchini S, Thompson A, Hinton JC, Woodward MJ. Comparative genomic indexing reveals the phylogenomics of Escherichia coli pathogens. Infect Immun. 2003;71:4674–4683. doi: 10.1128/IAI.71.8.4674-4683.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobrindt U, Agerer F, Michaelis K, Janka A, Buchrieser C, Samuelson M, Svanborg C, Gottschalk G, Karch H, Hacker J. Analysis of genome plasticity in pathogenic and commensal Escherichia coli isolates by use of DNA arrays. J Bacteriol. 2003;185:1831–1840. doi: 10.1128/JB.185.6.1831-1840.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukiya S, Mizoguchi H, Tobe T, Mori H. Extensive genomic diversity in pathogenic Escherichia coli and Shigella Strains revealed by comparative genomic hybridization microarray. J Bacteriol. 2004;186:3911–3921. doi: 10.1128/JB.186.12.3911-3921.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lan R, Reeves PR. Intraspecies variation in bacterial genomes: the need for a species genome concept. Trends Microbiol. 2000;8:396–401. doi: 10.1016/S0966-842X(00)01791-1. [DOI] [PubMed] [Google Scholar]
- Welch RA, Burland V, Plunkett G, 3rd, Redford P, Roesch P, Rasko D, Buckles EL, Liou SR, Boutin A, Hackett J, et al. Extensive mosaic structure revealed by the complete genome sequence of uropathogenic Escherichia coli. Proc Natl Acad Sci USA. 2002;99:17020–17024. doi: 10.1073/pnas.252529799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng P, Lampel KA, Karch H, Whittam TS. Genotypic and phenotypic changes in the emergence of Escherichia coli O157:H7. J Infect Dis. 1998;177:1750–1753. doi: 10.1086/517438. [DOI] [PubMed] [Google Scholar]
- Creuzburg K, Kohler B, Hempel H, Schreier P, Jacobs E, Schmidt H. Genetic structure and chromosomal integration site of the cryptic prophage CP-1639 encoding Shiga toxin 1. Microbiology. 2005;151:941–950. doi: 10.1099/mic.0.27632-0. [DOI] [PubMed] [Google Scholar]
- Creuzburg K, Recktenwald J, Kuhle V, Herold S, Hensel M, Schmidt H. The Shiga toxin 1-converting bacteriophage BP-4795 encodes an NleA-like type III effector protein. J Bacteriol. 2005;187:8494–8498. doi: 10.1128/JB.187.24.8494-8498.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brussow H, Canchaya C, Hardt WD. Phages and the evolution of bacterial pathogens: from genomic rearrangements to lysogenic conversion. Microbiol Mol Biol Rev. 2004;68:560–602. doi: 10.1128/MMBR.68.3.560-602.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Recktenwald J, Schmidt H. The nucleotide sequence of Shiga toxin (Stx) 2e-encoding phage phiP27 is not related to other Stx phage genomes, but the modular genetic structure is conserved. Infect Immun. 2002;70:1896–1908. doi: 10.1128/IAI.70.4.1896-1908.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lacher DW, Steinsland H, Whittam TS. Allelic subtyping of the intimin locus (eae) of pathogenic Escherichia coli by fluorescent RFLP. FEMS Microbiol Lett. 2006;261:80–87. doi: 10.1111/j.1574-6968.2006.00328.x. [DOI] [PubMed] [Google Scholar]
- Karch H, Schubert S, Zhang D, Zhang W, Schmidt H, Olschlager T, Hacker J. A genomic island, termed high-pathogenicity island, is present in certain non-O157 Shiga toxin-producing Escherichia coli clonal lineages. Infect Immun. 1999;67:5994–6001. doi: 10.1128/iai.67.11.5994-6001.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang WL, Bielaszewska M, Liesegang A, Tschape H, Schmidt H, Bitzan M, Karch H. Molecular characteristics and epidemiological significance of Shiga toxin-producing Escherichia coli O26 strains. J Clin Microbiol. 2000;38:2134–2140. doi: 10.1128/jcm.38.6.2134-2140.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guttman DS, Dykhuizen DE. Clonal Divergence in Escherichia-Coli as a Result of Recombination, Not Mutation. Science. 1994;266:1380–1383. doi: 10.1126/science.7973728. [DOI] [PubMed] [Google Scholar]
- Feil EJ. Small change: keeping pace with microevolution. Nat Rev Microbiol. 2004;2:483–495. doi: 10.1038/nrmicro904. [DOI] [PubMed] [Google Scholar]
- Herold S, Karch H, Schmidt H. Shiga toxin-encoding bacteriophages – genomes in motion. Int J Med Microbiol. 2004;294:115–121. doi: 10.1016/j.ijmm.2004.06.023. [DOI] [PubMed] [Google Scholar]
- Gamage SD, Patton AK, Hanson JF, Weiss AA. Diversity and host range of Shiga toxin-encoding phage. Infect Immun. 2004;72:7131–7139. doi: 10.1128/IAI.72.12.7131-7139.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohnishi M, Terajima J, Kurokawa K, Nakayama K, Murata T, Tamura K, Ogura Y, Watanabe H, Hayashi T. Genomic diversity of enterohemorrhagic Escherichia coli O157 revealed by whole genome PCR scanning. Proc Natl Acad Sci USA. 2002;99:17043–17048. doi: 10.1073/pnas.262441699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serra-Moreno R, Jofre J, Muniesa M. Insertion site occupancy by stx2 bacteriophages depends on the locus availability of the host strain chromosome. J Bacteriol. 2007;189:6645–6654. doi: 10.1128/JB.00466-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hendrix RW, Hatfull GF, Smith MC. Bacteriophages with tails: chasing their origins and evolution. Res Microbiol. 2003;154:253–257. doi: 10.1016/S0923-2508(03)00068-8. [DOI] [PubMed] [Google Scholar]
- Hendrix RW, Smith MC, Burns RN, Ford ME, Hatfull GF. Evolutionary relationships among diverse bacteriophages and prophages: all the world's a phage. Proc Natl Acad Sci USA. 1999;96:2192–2197. doi: 10.1073/pnas.96.5.2192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manning SD, Motiwala AS, Springman AC, Qi W, Lacher DW, Ouellette LM, Mladonicky JM, Somsel P, Rudrik JT, Dietrich SE, et al. Variation in virulence among clades of Escherichia coli O157:H7 associated with disease outbreaks. Proc Natl Acad Sci USA. 2008;105:4868–4873. doi: 10.1073/pnas.0710834105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandner L, Eguiarte LE, Navarro A, Cravioto A, Souza V. The elements of the locus of enterocyte effacement in human and wild mammal isolates of Escherichia coli : evolution by assemblage or disruption? Microbiology. 2001;147:3149–3158. doi: 10.1099/00221287-147-11-3149. [DOI] [PubMed] [Google Scholar]
- Castillo A, Eguiarte LE, Souza V. A genomic population genetics analysis of the pathogenic enterocyte effacement island in Escherichia coli : the search for the unit of selection. Proc Natl Acad Sci USA. 2005;102:1542–1547. doi: 10.1073/pnas.0408633102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frankel G, Phillips AD, Rosenshine I, Dougan G, Kaper JB, Knutton S. Enteropathogenic and enterohaemorrhagic Escherichia coli : more subversive elements. Mol Microbiol. 1998;30:911–921. doi: 10.1046/j.1365-2958.1998.01144.x. [DOI] [PubMed] [Google Scholar]
- Tauschek M, Strugnell RA, Robins-Browne RM. Characterization and evidence of mobilization of the LEE pathogenicity island of rabbit-specific strains of enteropathogenic Escherichia coli. Mol Microbiol. 2002;44:1533–1550. doi: 10.1046/j.1365-2958.2002.02968.x. [DOI] [PubMed] [Google Scholar]
- Jores J, Rumer L, Kiessling S, Kaper JB, Wieler LH. A novel locus of enterocyte effacement (LEE) pathogenicity island inserted at pheV in bovine Shiga toxin-producing Escherichia coli strain O103:H2. FEMS Microbiol Lett. 2001;204:75–79. doi: 10.1111/j.1574-6968.2001.tb10866.x. [DOI] [PubMed] [Google Scholar]
- Perna NT, Mayhew GF, Posfai G, Elliott S, Donnenberg MS, Kaper JB, Blattner FR. Molecular evolution of a pathogenicity island from enterohemorrhagic Escherichia coli O157:H7. Infect Immun. 1998;66:3810–3817. doi: 10.1128/iai.66.8.3810-3817.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muniesa M, Schembri MA, Hauf N, Chakraborty T. Active genetic elements present in the locus of enterocyte effacement in Escherichia coli O26 and their role in mobility. Infect Immun. 2006;74:4190–4199. doi: 10.1128/IAI.00926-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu C, Agin TS, Elliott SJ, Johnson LA, Thate TE, Kaper JB, Boedeker EC. Complete nucleotide sequence and analysis of the locus of enterocyte Effacement from rabbit diarrheagenic Escherichia coli RDEC-1. Infect Immun. 2001;69:2107–2115. doi: 10.1128/IAI.69.4.2107-2115.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rashid RA, Tabata TA, Oatley MJ, Besser TE, Tarr PI, Moseley SL. Expression of putative virulence factors of Escherichia coli O157:H7 differs in bovine and human infections. Infect Immun. 2006;74:4142–4148. doi: 10.1128/IAI.00299-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blattner FR, Plunkett G, 3rd, Bloch CA, Perna NT, Burland V, Riley M, Collado-Vides J, Glasner JD, Rode CK, Mayhew GF, et al. The complete genome sequence of Escherichia coli K-12. Science. 1997;277:1453–1474. doi: 10.1126/science.277.5331.1453. [DOI] [PubMed] [Google Scholar]
- Herbelin CJ, Chirillo SC, Melnick KA, Whittam TS. Gene conservation and loss in the mutS-rpoS genomic region of pathogenic Escherichia coli. J Bacteriol. 2000;182:5381–5390. doi: 10.1128/JB.182.19.5381-5390.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid SD, Betting DJ, Whittam TS. Molecular detection and identification of intimin alleles in pathogenic Escherichia coli by multiplex PCR. J Clin Microbiol. 1999;37:2719–2722. doi: 10.1128/jcm.37.8.2719-2722.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gansheroff LJ, Wachtel MR, O'Brien AD. Decreased adherence of enterohemorrhagic Escherichia coli to HEp-2 cells in the presence of antibodies that recognize the C-terminal region of intimin. Infect Immun. 1999;67:6409–6417. doi: 10.1128/iai.67.12.6409-6417.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deibel C, Kramer S, Chakraborty T, Ebel F. EspE, a novel secreted protein of attaching and effacing bacteria, is directly translocated into infected host cells, where it appears as a tyrosine-phosphorylated 90 kDa protein. Mol Microbiol. 1998;28:463–474. doi: 10.1046/j.1365-2958.1998.00798.x. [DOI] [PubMed] [Google Scholar]
- Manning SD, Madera RT, Schneider W, Dietrich SE, Khalife W, Brown W, Whittam TS, Somsel P, Rudrik JT. Surveillance for Shiga toxin-producing Escherichia coli, Michigan, 2001–2005. Emerg Infect Dis. 2007;13:318–321. doi: 10.3201/eid1302.060813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leomil L, Pestana de Castro AF, Krause G, Schmidt H, Beutin L. Characterization of two major groups of diarrheagenic Escherichia coli O26 strains which are globally spread in human patients and domestic animals of different species. FEMS Microbiol Lett. 2005;249:335–342. doi: 10.1016/j.femsle.2005.06.030. [DOI] [PubMed] [Google Scholar]
- Jeter C, Matthysse AG. Characterization of the binding of diarrheagenic strains of E. coli to plant surfaces and the role of curli in the interaction of the bacteria with alfalfa sprouts. Mol Plant Microbe Interact. 2005;18:1235–1242. doi: 10.1094/MPMI-18-1235. [DOI] [PubMed] [Google Scholar]
- Fletcher JN, Embaye HE, Getty B, Batt RM, Hart CA, Saunders JR. Novel invasion determinant of enteropathogenic Escherichia coli plasmid pLV501 encodes the ability to invade intestinal epithelial cells and HEp-2 cells. Infect Immun. 1992;60:2229–2236. doi: 10.1128/iai.60.6.2229-2236.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein EJ, Stapp JR, Clausen CR, Boster DR, Wells JG, Qin X, Swerdlow DL, Tarr PI. Shiga toxin-producing Escherichia coli in children with diarrhea: a prospective point-of-care study. J Pediatr. 2002;141:172–177. doi: 10.1067/mpd.2002.125908. [DOI] [PubMed] [Google Scholar]
- Durso LM, Bono JL, Keen JE. Molecular serotyping of Escherichia coli O26:H11. Appl Environ Microbiol. 2005;71:4941–4944. doi: 10.1128/AEM.71.8.4941-4944.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia A, Bosques CJ, Wishnok JS, Feng Y, Karalius BJ, Butterton JR, Schauer DB, Rogers AB, Fox JG. Renal injury is a consistent finding in Dutch belted rabbits experimentally infected with enterohemorrhagic Escherichia coli. Journal of Infectious Diseases. 2006;193:1125–1134. doi: 10.1086/501364. [DOI] [PubMed] [Google Scholar]
- Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. Basic local alignment search tool. J Mol Biol. 1990;215:403–410. doi: 10.1016/S0022-2836(05)80360-2. [DOI] [PubMed] [Google Scholar]
- Kim CC, Joyce EA, Chan K, Falkow S. Improved analytical methods for microarray-based genome-composition analysis. Genome Biol. 2002;3:RESEARCH0065. doi: 10.1186/gb-2002-3-11-research0065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lacher DW, Steinsland H, Blank TE, Donnenberg MS, Whittam TS. Molecular evolution of typical enteropathogenic Escherichia coli : clonal analysis by multilocus sequence typing and virulence gene allelic profiling. J Bacteriol. 2007;189:342–350. doi: 10.1128/JB.01472-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar S, Tamura K, Nei M. MEGA3: Integrated software for Molecular Evolutionary Genetics Analysis and sequence alignment. Brief Bioinform. 2004;5:150–163. doi: 10.1093/bib/5.2.150. [DOI] [PubMed] [Google Scholar]
- Huson DH, Bryant D. Application of phylogenetic networks in evolutionary studies. Mol Biol Evol. 2006;23:254–267. doi: 10.1093/molbev/msj030. [DOI] [PubMed] [Google Scholar]
- Felsenstein J. PHYLIP – Phylogeny Inference Package (Version 3.2) Cladistics. 1989;5:164–166. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Distribution of phylogenetically compatible genes in EHEC 2, determined with the clique program in the PHYLIP package. Conserved genes have a value of 1 and divergent/absent have a value of 0.
Genes in EHEC 2 whose distribution was used to generate colormaps in Figure 5. Conserved genes have a value of 1, divergent/absent genes have a value of 0 and genes that have a value of 0.5 were inferred as conserved after the GACK cutoff was relaxed by 20%.
Distribution of 49 non-LEE effector genes in EHEC 2. Conserved genes have a value of 1, divergent/absent genes have a value of 0 and genes that have a value of 0.5 were inferred as conserved after the GACK cutoff was relaxed by 20%.
Distribution of K-12-specific genes in EHEC 2. Conserved genes have a value of 1 and divergent/absent genes have a value of 0.