Abstract
Dengue is the most common arthropod-borne infection worldwide, affecting at least 50 million people every year and endemic in more than 100 countries. The dengue virus is a single-stranded RNA virus with four major serotypes. Infection with one serotype confers homotypic immunity but not heterologous immunity, and secondary infection with another serotype may lead to more severe disease. The major route of transmission occurs through the Aedes aegypti mosquito vector, but dengue has also been transmitted through blood transfusion and organ transplantation. Infection results in a spectrum of clinical illness ranging from asymptomatic infection, undifferentiated fever, dengue fever, dengue haemorrhagic fever (DHF) to dengue shock syndrome (DSS). Dengue is spreading rapidly to new areas and with increasing frequency of major outbreaks. A trend has also been observed towards increasing age among infected patients. This will impact blood supply availability as more blood donors are deferred because of dengue infection or exposure to infection. The risk of transmission through transfusion of blood from asymptomatic viraemic donors will also increase. Although screening tests for dengue and effective pathogen reduction processes are now available for the blood supply, the value of implementing these costly measures needs to be carefully considered. Demand for platelets and fresh frozen plasma will rise with increasing number of DHF/DSS. Evidence-based guidelines for the clinical use of these blood components in the management of patients with DHF/DSS have not been well established, and inappropriate use will contribute to the challenges faced by blood services.
Keywords: blood supply, dengue fever, dengue virus
The World Health Organization classifies dengue as a major international public health concern. Some 2·5 billion people or two-fifths of the world’s population are at risk of infection. Dengue haemorrhagic fever (DHF) is endemic in more than 100 countries in Africa, the Americas, Eastern Mediterranean, South-East Asia and the Western Pacific regions (World Health Organization, 2008).
Worldwide, it is estimated that 50 million dengue infections occur every year with 500 000 requiring hospitalization; 2·5% of those affected may die (World Health Organization, 2008). These figures are likely to be underestimated as the numbers reported are influenced by different surveillance and reporting systems as well as varying interpretations of case definitions and the presence of missed and silent infections (Guha-Sapir & Schimmer, 2005).
EPIDEMIOLOGY
Prior to the 1940s, dengue was characterized by epidemics that occurred relatively infrequently, and generally, each location involved a single viral serotype. When sufficient numbers of vectors and susceptible hosts for the particular serotype were reached, a sudden sharp rise in transmission occurred. In contrast, the predominant pattern of global transmission today is hyperendemic dengue, where there is continuous circulation of multiple viral serotypes within a constant pool of susceptible hosts and vectors.
The first known epidemic of DHF in Asia occurred in Manila, Philippines in 1953–1954, to be followed by Bangkok, Thailand in 1958 and Malaysia, Singapore and Vietnam in the 1960s. With the economic boom and associated urbanization, it rapidly spread to involve most of Asia. By mid-1970s, dengue had overtaken malaria to become the leading cause of hospitalization and death among children in the region. Increased spread is also seen into suburban and rural areas (Gubler, 2002).
In the Americas, with the eradication of A. aegypti vector to control yellow fever, only sporadic cases were reported until the early 1980s when a large outbreak occurring in Cuba marked the start of epidemic spread to the Pacific and the American tropics. Dengue has been prevalent in tropical Africa, although DHF is rare. It occurred occasionally in the temperate regions of North Africa and the Mediterranean regions until the 1990s when epidemics were reported in the Comoros. Dengue continues to expand its territory, and the only continents that currently do not experience dengue transmission are Europe and Antarctica.
The factors that have contributed to the dramatic expansion of dengue include population growth, urbanization, inadequate water management, lack of effective mosquito control and convenient global travel. In the last few years, changes in weather patterns may have contributed to the expansion in the habitat range of the mosquito vector. Increased epidemic activity may also increase the rate of viral genetic change and emergence of strains or genotypes with greater epidemic potential (Gubler, 1998; Messer et al., 2003; Dash et al., 2006).
Although DHF is primarily an illness of children, surveillance studies in recent years in several countries have reported increasing age among infected patients (Liu et al., 2003; Gupta et al., 2006; Khan et al., 2007; Ooi et al., 2006). In Singapore, this has been ascribed to lowered herd immunity in the population as a result of reduced dengue in the 1970s and 1980s. Studies have shown that A. aegypti bites adults more frequently than children (De Benedictis et al., 2003), and together, these could explain the higher incidence rate among young adults in Singapore.
DENGUE VIRUS AND THE MOSQUITO VECTOR
The dengue virus is a single-stranded RNA virus belonging to the Flaviviridae family. The viral genome is approximately 11 kb in length and is surrounded by an icosahedral nucleocapsid covered by a lipid envelope. The mature virion has three structural (core, membrane-associated and envelope) and seven non-structural (NS1, NS2a, NS2b, NS3, NS4a, NS4b and NS5) proteins.
The envelope protein is involved in the main biological functions and is responsible for binding and transport into host cells. It is also associated with the induction of neutralizing antibodies and development of protective immune response in the host. The non-structural proteins are expressed as both membrane-associated and secreted forms and have been implicated in the pathogenesis of severe disease (Gubler, 1998).
There are four serotypes classified according to their immunological properties – DEN-1, DEN-2, DEN-3 and DEN-4. Infection with one dengue serotype confers lifelong immunity against that serotype but only transient protection against infection by other serotypes. All four serotypes have been associated with outbreaks, having seen DEN-2 as the predominant serotype of primary infection during the outbreak in 2005 (Savage et al., 1998; Messer et al., 2003; Gupta et al., 2006; Ooi et al., 2006). Recent data have also demonstrated that the various genotypes within each serotype possess varying epidemic potential (Anderson & Rico-Hesse, 2006).
The primary vector for the dengue virus is the A. aegypti mosquito, although the virus may be transmitted by the Aedes albopictus and Aedes polynesiensis as well. Infected humans are the main carriers and amplification host of the dengue virus. Female mosquitoes acquire the virus by biting infected humans in the viraemic phase and become infective after an extrinsic incubation period of 7–14 days. Subsequently, the mosquito may transmit the virus during every feeding. The length of the extrinsic incubation period is dependent on the ambient temperature and the virus involved, both of which affect the replication rate of the virus in the vector.
Aedes aegypti is a highly domesticated mosquito that breeds in artificial containers such as water storage tanks, subterranean pits, flowerpot trays and other ornamental containers. The vector is known to prefer to rest indoor, although studies have shown that they may seek oviposition outdoors. Peak biting activity is at dawn and dusk. The multiple feeding behaviour of A. aegypti and its preference for human hosts are believed to contribute to the explosive spread of dengue virus, even in the presence of a low A. aegypti population (Scott et al., 1997).
LABORATORY DIAGNOSIS
Laboratory diagnosis of dengue can be performed by viral isolation, serological tests, dengue antigen tests and molecular detection.
Virus isolation for dengue is performed by inoculation of the sample into live mice, live mosquitoes or cell cultures. Successful detection of virus may be affected by the presence of interfering antibodies and the heat-labile nature of the virus. It is normally used as a confirmatory test, being impractical for diagnosis or screening on a large scale.
Dengue-specific immunoglobulin M (IgM) and immunoglobulin G (IgG) enzyme-linked immunosorbent assay (ELISA) is widely used in diagnosis as it is relatively inexpensive, has good sensitivity and is quick and easy to perform. IgM is detectable at 3–5 days after infection, peaks at about 2 weeks and declines to undetectable levels over 2–3 months. IgG becomes elevated after 9–10 days and persists at detectable levels for life. During secondary infection, IgG increases rapidly to much higher levels within 1–2 days after infection. Because the virus shares antigenic epitopes with other flaviviruses, the presence of cross-reactive antibodies may interfere with serological diagnosis (World Health Organization, 1997).
The dengue NS1 antigen-capture ELISA is useful for detection of dengue early in the disease. The test sensitivity has been demonstrated to be significantly higher in primary dengue infection (97·3%) than in secondary dengue infection (70·0%), with a positive predictive value of 100% and negative predictive value of 97·3% (Kumarasamy et al., 2007). Although it is useful in the first week of disease and provides evidence of presence of the virus, its effectiveness in screening blood donors has not been established yet.
Like the NS1 test, the dengue RNA test detects viral material that is typically present in the first 5 days of disease. The advantages of the test are good sensitivity and specificity, and the ability to rapidly detect minute quantities of dengue virus material in serum. The disadvantages are the relatively high cost and expertise needed, particularly proper quality control to avoid false positives.
Real-time reverse transcription–polymerase chain reaction (RT-PCR), using either a universal dengue oligonucleotide primer pair or a combination of the four serotype-specific oligonucleotide primers, is widely used for clinical diagnosis and public health surveillance. It has established detection limits of 0·1 plaque-forming units (PFU) mL−1 for DEN-1 and DEN-2, 1 PFU mL−1 for DEN-3 and 0·01 PFU mL−1 for DEN-4 and 88% correlation with virus isolation (Lai et al., 2006).
Group-specific one-step PCR using universal dengue oligonucleotide primer pairs and SYBR green I is widely used for population surveillance as it is fast and cost-effective for mass screening, with a detection limit of 10–4·1 PFU mL−1 (Shu et al., 2003; Lai et al., 2006).
A prototype dengue RNA transcription-mediated amplification (TMA) assay (Gen-Probe, Inc., San Diego, CA, USA) was developed for use in large-scale screening of blood donor samples and uses target genomic sequences that are highly conserved across all four serotypes. The analytical sensitivity of the assay has been established at a detection of 14·9 copies mL−1 at 95% detection limit and 3·5 copies mL−1 at 50% detection limit for DEN-1, with comparable sensitivity for all four serotypes, and with a specificity of 99·91% (Linnen et al., 2008).
CLINICAL FEATURES
Dengue infection can produce a spectrum of clinical illness – undifferentiated fever, dengue fever (DF), DHF and dengue shock syndrome (DSS). In infants and children younger than 15 years, the patient is usually either asymptomatic or has a mild undifferentiated febrile illness with maculopapular rash.
DF is characterized by the sudden onset of high fever, severe headache (especially in the retro-orbital area), arthralgia, myalgia, nausea, vomiting and rash. Infants and younger children tend to present with an undifferentiated febrile disease, often with rash. The acute febrile illness lasts approximately 2–7 days.
DHF is clinically defined by high fever, haemorrhagic manifestations, thrombocytopenia and evidence of plasma leakage (Table 1). Four grades of severity have been defined (Table 2), where grades III and IV are considered to be DSS. The presence of thrombocytopenia with concurrent haemoconcentration differentiates grades I and II DHF from DF (World Health Organization, 1997). Clinical deterioration usually occurs towards the end of the febrile phase when the patient progresses to the phase of plasma leakage.
Table 1.
World Health Organization case definition for DHF and DSS*
| Case definition for DHF |
| The following must be present: |
| Fever or history of acute fever lasting 2–7 days, occasionally biphasic |
| Haemorrhagic tendencies, evidenced by at least one of the following: |
| A positive tourniquet test |
| Petechiae, ecchymoses or purpura |
| Bleeding from the mucosa, gastrointestinal tract, injection sites or other locations |
| Haematemesis or melaena |
| Thrombocytopenia (100 000 cells mm−3 or less) |
| Evidence of plasma leakage because of increased vascular permeability, manifested by at least one of the following: |
| A rise in the haematocrit equal to or greater than 20% above average for age, sex and population |
| A drop in the haematocrit following volume replacement treatment equal to or greater than 20% of baseline |
| Signs of plasma leakage such as pleural effusion, ascites and hypoproteinaemia |
| Case definition for DSS |
| All the above four criteria for DHF must be present plus evidence of circulatory failure manifested by: |
| Rapid and weak pulse |
| Narrow pulse pressure [<20 mmHg (2·7 kPa)] |
| Or manifested by: |
| Hypotension for age |
| Cold, clammy skin and restlessness |
Table 2.
World Health Organization grading severity of DHF*
| Grade I | Fever accompanied by nonspecific constitutional symptoms; the only haemorrhagic manifestation is a positive tourniquet test and/or easy bruising |
| Grade II | Spontaneous bleeding in addition to the manifestations of grade I patients, usually in the forms of skin or other haemorrhages |
| Grade III | Circulatory failure manifested by a rapid, weak pulse and narrowing of pulse pressure or hypotension, with the presence of cold, clammy skin and restlessness |
| Grade IV | Profound shock with undetectable blood pressure or pulse |
Plasma leakage manifests as tachycardia, hypotension, pleural effusions and ascites develop. Bleeding from the gastrointestinal tract or epistaxis may occur, with menorrhagia in females. Epigastric discomfort, myalgia, vomiting, diarrhoea and abdominal pain are common in adults. Tender hepatomegaly is observed in all patients and splenomegaly in some. Shock, plasma leakage and marked thrombocytopenia are more common in children, whereas internal haemorrhage is more frequent as age increases (Hammond et al., 2005).
In DHF, the decline in platelet counts tends to precede plasma leakage, and platelet counts and packed cell volume are often used to monitor for impending deterioration (Tai et al., 1999). Haemoconcentration may be difficult to ascertain in areas where a significant proportion of the population carries the thalassaemia genetic trait or where there is a high degree of iron deficiency (Khan et al., 2007).
DSS is a medical emergency associated with very high mortality. Severe plasma leakage results in prolonged shock, accompanied by metabolic acidosis, which in turn precipitates disseminated intravenous coagulation (DIC). Massive haemorrhage or encephalopathy may develop, the former requiring intensive blood transfusion support.
Severe dengue infections can result in complications such as liver failure, DIC, encephalopathy, myocarditis, acute renal failure and haemolytic-uraemic syndrome. These are generally rare and usually seen in patients with DHF grades III and IV (Malavige, 2006).
There are no specific drugs against the dengue virus, and management is symptomatic and supportive. DF is typically a self-limiting disease with a mortality rate of less than 1%, although the convalescent phase may sometimes be prolonged in adults. Successful management of DHF lies in proper management of fluid balance. Early identification of the plasma leakage phase with prompt resuscitation has been shown to reduce complications and improve outcome. When adequately treated, the mortality rate for DHF can be as low as 1%, but if untreated, it can exceed 20% (World Health Organization, 1997, 2008).
PATHOPHYSIOLOGY
The primary pathophysiology seen in DHF is an acute increase in vascular permeability that leads to leakage of plasma into the extravascular compartment, resulting in haemoconcentration and decreased blood pressure. Increased vascular permeability is shown to result from endothelial gaps in the peripheral vascular bed. No necrotic or inflammatory lesions are seen, suggesting that the changes are functional and likely to be caused by a short-acting mediator.
The principal mechanisms by which dengue virus infection causes DHF are still not clearly elucidated and are believed to be a result of viral and host factors that involve the activation of T cells, production of cytokines/chemokines and disturbance of the haemostatic system. These result in the release of other cytokines that mediate the systemic effects of plasma leakage and circulatory insufficiency. In addition, there is evidence for increased apoptosis and endothelial cell dysfunction (Gubler, 1998; Guglani & Kabra, 2005).
More severe infection is observed to occur in secondary infection than in primary infection (Pancharoen et al., 2001). Various theories have been put forward to explain this observation, including the phenomenon of antibody-dependent enhancement and more recently the theory of original antigenic sin (Mongkolsapaya et al., 2003).
During the early stage of infection, thrombocytopenia occurs as a result of bone marrow hypocellularity resulting from direct dengue infection of haematopoietic progenitor cells and stromal cells (Nakao et al., 1989). As the fever settles, bone marrow hypercellularity is seen, and increased destruction from immune-mediated clearance of platelets becomes the primary mechanism for thrombocytopenia (Mitrakul et al., 1977).
RISK FACTORS FOR DHF
There are age-related differences in susceptibility to DHF/DSS. Although children infected with dengue virus tend to manifest milder clinical symptoms than adults, they have higher risk of developing DHF/DSS (Hammond et al., 2005; Ooi et al., 2006). It is postulated that children have a greater propensity for vascular leakage under normal physiological conditions than adults, and this results in less ability to accommodate additional factors that increase vascular permeability.
Hospitalization and death rates for severe and very severe dengue are highest in those younger than 15 years and those older than 60 years (Guzmán et al., 2002).
Elderly patients older than 65 years are more likely to develop severe illness, with higher risk of hospitalization and death when infected with dengue virus than youths and younger adults (García-Rivera & Rigau-Pérez, 2003). Age above 65 years, history of dengue infection, diabetes mellitus, hypertension and renal insufficiency have been identified as being significantly associated with DHF/DSS (Lee et al., 2006).
Genetic influences, including human leucocyte antigen allele associations, may play a part in disease susceptibility and severity (Wagenaar et al., 2004). In Haiti, very few DHF cases are recorded despite a transmission rate of 30% and evidence showing the presence of all four serotypes in the population (Halstead et al., 2001). There is similar paucity of DHF cases in Africa despite the presence of dengue virus in the population, suggesting the presence of a dengue resistance gene.
VIRAEMIA AND TRANSMISSION
The primary mode of transmission for the dengue virus occurs through the mosquito vector. After the bite of an infected mosquito, the dengue virus enters the body and replicates within the cells of the mononuclear phagocyte lineage. There is usually an incubation period of 3–14 days (average 4–7 days). The period of viraemia roughly corresponds to the period of fever, peaking at the time of or shortly after onset of illness and remaining detectable for various periods (typically 2–7 days). There is no correlation between the duration of viraemia and serotype. Some studies have reported correlation between viraemia and disease severity, whereas others report no correlation (Gubler et al., 1981; Vaughn et al., 1997; Murgue et al., 2000).
Infectivity during the incubation period has not been well defined, partly because of the lack of an animal model for dengue. In studies involving human volunteers during the 1920s, clinically apparent DF resulted in 80 and 25% of biting experiments at 1 and 2 days before the onset of fever, indicating that infected persons could transmit virus as early as or even earlier than 2 days before symptoms develop (Nishiura & Halstead, 2007).
Vertical transmission has been reported, with intrapartum transmission in at least two cases where the onset of neonatal illness developed on the first day of life (Sirinavin et al., 2004) and one case involving a mother who was diagnosed with DHF just prior to delivery (Rigau-Pérez et al., 2001).
Nosocomial transmission through needlestick injury and mucocutaneous exposure has also been reported. The case of mucocutaneous transmission involved a healthcare worker who developed dengue infection after blood from a febrile-dengue-infected traveller splashed onto her face (Chen & Wilson, 2005).
Transmission of dengue infection has been reported from donor to recipient in one case of living donor renal transplant (Tan et al., 2005). The clinical presentation and course of illness was similar to that of an immunocompetent patient, except for prolonged course of illness (19 days) and duration of thrombocytopenia (12 days).
Transmission during a bone marrow transplant was reported in one instance during a dengue epidemic in Puerto Rico in 1994 (Rigau-Pérez et al., 2001). The bone marrow recipient developed fever 4 days after the transplant and subsequently died. The donor developed fever and headache 2 days after the marrow was harvested.
There are only two reported instances of transmission through blood transfusion. The first involved a patient in Hong Kong who developed fever 3 days after a blood transfusion, associated with moderate neutropenia, severe thrombocytopenia and hypotension responsive to fluid therapy. The donor was asymptomatic at the time of donation but developed mild symptoms of DF 1 day after blood donation. An archived sample from the donation also tested positive for dengue virus by RT-PCR (Chuang et al., 2008; C. K. Lin, Hong Kong Red Cross Blood Transfusion Service, Hong Kong, pers. comm.).
The second involved the transmission of dengue from an asymptomatic blood donor who developed an acute febrile illness the day after donating blood. Look back investigation confirmed dengue infection in the recipients of the three blood products from his donation. Two recipients had DF with some evidence of capillary leakage, whereas the platelet recipient had asymptomatic seroconversion. All recovered without sequelae. A stored serum sample from the donation tested positive for DEN-2 by RT-PCR (Tambyah et al., 2008).
The small number of reports of transfusion transmission could be because of the fact that it is difficult to differentiate between non-mosquito transmission and mosquito-borne infection in endemic areas where the vector is widespread. Many infections may also result in mild or asymptomatic illness that is not recognized as transfusion-acquired infection, and diagnostic laboratories to document infections and their sources are often not available in many endemic countries (Chen & Wilson, 2005).
ASYMPTOMATIC INFECTION
Various studies have shown the presence of asymptomatic or subclinical infection, which can range from 0·77 to 87% depending on the population studied (Ooi et al., 2006). It is estimated that for every one symptomatic case, there can be 6·7 cases that are asymptomatic (Chen & Wilson, 2005). There is no correlation with total dengue incidence, hospitalization rates, age or sex, although one study found silent transmission to occur more commonly in the 15- to 40-year-old age group.
In a study of 329 healthy volunteers in a province in Thailand with high rate of dengue infection, 29 (8·8%) had a serum sample positive for dengue IgM, of which two samples tested positive for viral RNA (Poblap et al., 2006). Cluster sampling studies around index cases in Indonesia detected eight post-enrolment asymptomatic dengue infections of 785 volunteers over a 2-year period, of which two demonstrated viraemia by RT-PCR (Beckett et al., 2005). Virus was isolated in 215 of 3189 (6·7%) persons in a study evaluating the dynamics of transmission of dengue virus in a dengue epidemic area of Colombia; most of whom were asymptomatic (Méndez et al., 2006).
Two recent studies reported viraemia in blood donations collected from four countries experiencing high dengue transmission rates. In the first study, 12 (0·07%) of 16 521 blood donations collected in Puerto Rico tested positive using the dengue-specific TMA nucleic acid amplification test (NAT) (Mohammed et al., 2008). Supplemental testing using RT-PCR was positive in four samples (viral loads 2 × 103 to 8 × 107 copies mL−1), and live virus was recovered from three of the PCR-positive samples.
In the second study, samples from asymptomatic blood donors in Honduras, Brazil and Australia were obtained during periods of clinical dengue outbreaks and screened using the dengue-specific TMA assay (Linnen et al., 2008). Nine (0·30%) of 2994 Honduran samples tested positive, of which 8 were confirmed by RT-PCR (viral loads 3 × 104 to 4·2 × 104 copies mL−1) and 4 samples yielded infectious viruses. Three (0·06%) of 4858 Brazilian samples tested positive, of which 2 were RT-PCR positive (viral loads 12 and 294 copies mL−1); viral isolation was not performed for these samples.
IMPACT ON BLOOD DONORS
Dengue is spreading rapidly into new areas, and major outbreaks are occurring with greater frequency. More than half the world is now endemic for dengue, and there is increasing likelihood that blood donors in these countries will be infected or exposed to infection. As the numbers continue to increase, the effect on blood donor attendance will reach levels sufficient enough to impact significantly on the blood supply.
In most countries, donors who have been infected with dengue virus are deferred for periods of up to 6 months (Table 3) or even longer if they have received blood transfusions. Blood donors who donate regularly will be particularly affected, including apheresis donors who may donate as often as 12 or 13 times a year. Healthy uninfected donors whose family members or working colleagues are infected with dengue are also likely to be deferred from donating blood. These donors would also have less time available to donate blood, as they would need to care for sick family members or take on increased work responsibilities as a result of more workplace illness.
Table 3.
Dengue and donor deferral
| Country | Donor deferral measures for dengue |
|---|---|
| Singapore* | 6 months deferral for history of dengue infection |
| 3 weeks deferral for history of fever | |
| No travel-related deferral for dengue | |
| Hong Kong* | 6 months deferral for history of dengue infection |
| 2 weeks deferral for history of fever | |
| No travel-related deferral for dengue | |
| Sri Lanka* | No specific deferral for history of dengue infection |
| 2 weeks deferral for history of fever | |
| No travel-related deferral for dengue | |
| Australia† | 4 weeks deferral for history of dengue infection |
| No travel-related deferral for dengue | |
| New Zealand‡ | 4 weeks deferral for history of dengue infection |
| No travel-related deferral for dengue | |
| UK‡ | 2 weeks deferral for history of dengue infection |
| No travel-related deferral for dengue | |
| United States‡ | 4 weeks deferral for history of dengue infection |
| No travel-related deferral for dengue |
Endemic for dengue.
Non-endemic except parts of Northern Australia.
Non-endemic.
In the past, dengue affected mainly children who do not donate blood. As this trend reverses and the modal age of infection increases, dengue will increasingly affect the segment of population that do donate blood – youths and young adults – further reducing blood donor availability. The annual incidence of dengue infection in Singapore was 180·6 per 100 000 population in 2007, with highest incidence in the segment of population older than 15 years. It is estimated that dengue infection has reduced the blood donor pool by 0·2%; this excludes donors deferred for symptoms related to dengue and exposure to dengue, which is estimated to lose a further 1·1% of donors in 2007.
RISK OF TRANSFUSION-TRANSMITTED DENGUE
Increased prevalence in the population increases the risk that blood will be collected from a viraemic donor during the asymptomatic or subclinical phase of infection. Transmission of dengue through blood collected from asymptomatic donors has been demonstrated in the two reports from Hong Kong and Singapore, both occurring during the height of epidemics in these countries (Chuang et al., 2008; Tambyah et al., 2008). Blood services in countries experiencing dengue epidemics will need to decide whether stronger measures are needed to protect the blood supply.
During infectious disease outbreaks such as dengue, severe acute respiratory syndrome (SARS) and chikungunya disease, most countries have adopted risk reduction strategies that focus on excluding donors who may be at higher risk of infection or who may be exhibiting early symptoms of infection. In the case of dengue, this has relied mainly on questioning the donor for a recent history of travel to outbreak areas and for symptoms of fever, rash or malaise.
The relatively low sensitivity and specificity of blood safety measures based on donor history has long been debated. Travel-related deferrals in particular lead to high rates of unnecessary donor deferral. In addition, there is often a negative effect on the donor of being deferred, and many donors do not return even if the deferral is temporary (Halperin et al., 1998). Introducing such measures during outbreaks of dengue is likely to exacerbate the problems of dwindling donor attendance and decreasing blood collection described earlier. For example, in Singapore, the SARS outbreak in 2003 and chikungunya outbreak in 2008 resulted in donor deferral rates of 4 and 3%, respectively.
In a population where dengue is endemic, exclusion on the basis of symptoms also fails to address the risk of collecting blood from asymptomatic viraemic donors. Studies so far suggest that there is at least a 1–2 days period of infectivity prior to the development of symptoms. The presence of dengue viraemia in asymptomatic persons, including blood donors, is well documented during dengue epidemics.
The efficiency of transmission depends on a combination of factors: amount and stability of virus, volume of viraemic blood transfused and immune status of the recipient. Although dengue viraemia titres in vertebrate hosts are usually in the range of 105–109 copies mL−1 (Chen & Wilson, 2005), it is likely that lower levels and shorter duration of viraemia occur during asymptomatic infection compared with DF or DHF. Mohammed et al. (2008) and Linnen et al. (2008) demonstrated the presence of viral loads of at least 2 × 103 to 8 × 107 copies mL−1 and 12 to 4·2 × 104 copies mL−1, respectively, in healthy blood donors.
Experience with West Nile Virus (WNV) suggests that not all positive donations are infectious. In these two studies, live virus was recovered from 7 of 21 positive donations, indicating that these donations were capable of transmitting infection to recipients. The viral loads present were in the order of 4·4 × 102 copies mL−1 to 8·12 × 107 copies mL−1. In contrast, cell cultures were negative in five donations with viral loads between 78 and 7·4 × 103 copies mL−1. Although this suggests that lower viral loads are associated with lower risk of infectivity, WNV has been transmitted from transfusions with estimated viral loads as low as 0·06 PFU mL−1 or 24 copies mL−1. It is therefore possible that larger volumes involved during blood transfusion could still transmit infection despite lower viral load concentrations.
The risk of infection will also depend on the size of population exposed to the infection and immunity because of prior infection. Infection may not occur if the majority of individuals have previously been infected and developed protective antibodies.
Studies on the prototype dengue NAT system showed sufficient sensitivity and early detection to enable interdiction of infective donations. However, it is likely that individual sample NAT rather than mini-pool NAT will be required for dengue screening because of the relatively low levels of dengue RNA (Mohammed et al., 2008). At the time of writing, at least two dengue NAT systems for blood supply screening are in development from Gen-Probe Ltd. and Roche Diagnostics, Ltd., and it is possible that these will be commercially available within the next few years. While the cost of such systems is not available as yet, it is unlikely that they will be less than the current WNV-NAT or hepatitis B virus NAT (HBV-NAT).
An alternative strategy to testing is pathogen reduction, which has the added advantage that it is effective against other infections that can be transmitted through blood, such as the chikungunya virus and WNV. Preliminary studies using the amotosalen-HCl and long-wavelength ultraviolet light (UVA) system on apheresis platelets have shown effectiveness against the dengue virus (Lam et al., 2007). Pathogen reduction technology is not yet available for red cells, and this has somewhat limited its impact. Nonetheless, as more vector-transmitted infections emerge to threaten the blood supply, pathogen reduction of platelets and plasma coupled with selective red cell inventory hold may be useful in reducing transmission risks without unnecessary donor deferral.
Regardless of whether a new screening assay or pathogen reduction method is selected, the recent experience with human immunodeficiency virus NAT (HIV-NAT) and hepatitis C virus NAT (HCV-NAT) suggests that this will involve high costs and expensive technical expertise (Marshall et al., 2004). There is little doubt that transfusion-transmitted infections will represent only a small fraction of total infections during an epidemic. In countries with high dengue prevalence, the introduction of a screening test or pathogen reduction process is therefore likely to prevent infections through blood transfusion, but the proportion would be low in comparison to the total number of infections (Table 4).
Table 4.
Options for minimizing dengue risk in the blood supply
| Strategies | Endemic countries | Non-endemic countries |
|---|---|---|
| No specific measures taken for dengue | Risk of transfusion-transmitted dengue increased, dependent on prevalence in donor population and proportion of donors with asymptomatic infection | Risk of transfusion-transmitted dengue low, dependent on proportion of donor population who may recently be exposed to dengue infection through travel |
| No direct cost to blood service, but indirect cost from patient morbidity from transfusion-transmitted infection and loss of confidence in blood supply safety | No direct cost to blood service, but indirect cost from loss of confidence in blood supply safety in event of a transfusion-transmitted infection occurring | |
| Donor qualification – deferral of at-risk donors, e.g. symptoms of fever, travel history, exposure to dengue patients, etc. | Deferral based on exposure not feasible when disease is endemic, unable to exclude early and asymptomatic infection | Deferral based on exposure feasible, able to reduce risk of accepting donations from early and asymptomatic infected donors |
| Nonspecific, leads to high donor loss | Low donor loss, dependent on proportion of donor population likely to travel to endemic countries | |
| Low cost-effectiveness | Cost-effective | |
| NAT testing of donations for dengue | Able to detect asymptomatic infection | Able to detect asymptomatic infection |
| Donor loss dependent on specificity of test system | Donor loss dependent on specificity of test system | |
| Expensive | Expensive | |
| Cost-effectiveness depends on prevalence of asymptomatic infected donors | Low cost-effectiveness | |
| Pathogen reduction | Able to reduce transmission risks | Able to reduce transmission risks |
| Expensive and only available for platelets and plasma currently. May result in reduced product yields | Expensive and only available for platelets and plasma currently. May result in reduced product yields | |
| Low cost-effectiveness for dengue alone | Low cost-effectiveness for dengue alone | |
| Increased cost-effectiveness depends on ability to reduce risks of other transfusion-transmitted diseases as well | Increased cost-effectiveness depends on ability to reduce risks of other transfusion-transmitted diseases as well |
Dengue carries a high economic burden on society, in terms of medical costs and control measures, as well as reduced workforce productivity (Gubler, 2002; Guha-Sapir & Schimmer, 2005). In developing countries with limited resources, the decision to implement a costly risk reduction strategy into the blood supply must take into consideration its cost-effectiveness in reducing disease morbidity in relation to the overall health situation in the country.
Cost-effectiveness of risk reduction strategies for transfusion-transmitted diseases depends on the clinical consequences on recipient health, and whether infection acquired through blood transfusion results in more serious disease. There are few studies reported on the clinical effects of superimposed dengue infection on patients. A retrospective analysis in Brazil of primary dengue infection in 27 renal transplant recipients showed similar clinical picture and outcome as in the general population (Azevedo et al., 2007). A report from French Guiana of 22 pregnant women who had DF during pregnancy found no abnormality in their infants but observed a higher foetal death rate than usual, suggesting that dengue infection might increase the risk for foetal mortality (Carles et al., 1999).
Superimposed dengue may subject the patient to additional risk. Tan et al. (2005) noted in their case report that the occurrence of DHF early postoperatively posed potential danger to the transplant patient as a result of persistent haemorrhage from cut tissue surfaces from the associated bleeding diathesis. Most recipients of blood transfusion are in the older age group and will therefore be at increased risk of DHF and complications following dengue infection. The higher risks to patients involving secondary infection with a different serotype have also not been studied.
Case studies suggest that dengue acquired through blood transfusion will not result in more severe illness than that acquired through the usual vector routes of transmission. The four patients who acquired infection through blood transfusion in Hong Kong and Singapore experienced relatively benign course of illness and uneventful recovery with no serious sequelae. This could be because of the lower viral loads present in the blood donations collected. Nevertheless, more data will be needed to make a firm conclusion.
DENGUE AND PLATELET TRANSFUSIONS
Blood transfusion is only indicated in cases where there is significant clinical bleeding. The exact platelet counts at which platelet transfusions should be given in dengue infection are still not well defined, although most clinical guidelines recommend that platelet transfusions are given to patients who develop serious haemorrhagic manifestations or have very low platelet counts – platelet counts falling below 10–20 × 109 L−1 without haemorrhage or 50 × 109 L−1 with bleeding or haemorrhage.
In children, there is little correlation between platelet count and bleeding manifestations or between platelet count and disease severity (Malavige, 2006). A study of risk factors for haemorrhage in 114 paediatric patients with DSS showed no correlation between bleeding and platelet count, and prolonged duration of shock was in fact the strongest risk factor for haemorrhage (Lum et al., 2002).
In adults, a platelet count of 5 × 109 L−1 and packed cell volume >50 are significantly associated with the presence of bleeding manifestations (Malavige, 2006). However, in uncomplicated dengue patients, one study of 120 patients found no difference in morbidity between those with platelet counts above or below 50 × 109 L−1 with or without fever (Lye et al., 2008).
Another study of 245 dengue patients showed no correlation between clinical bleeding and platelet count, and 81 non-bleeding patients had counts of less than 20 × 109 L−1 (Chaudhary et al., 2006). In contrast, another study of 225 dengue patients suggested that bleeding occurred more often in patients with platelet counts below 20 × 109 L−1 (Makroo et al., 2007).
In a study of 106 paediatric patients with DSS with thrombocytopenia and coagulopathy, there was no significant difference in haemorrhage between patients who received preventive transfusions compared with those who did not. Patients who received transfusion had higher frequency of development of pulmonary oedema and increased length of hospitalization (Lum et al., 2003).
Corticosteroids have been shown to be no more effective than placebo or no treatment for reducing the number of deaths, the need for blood transfusion or the number of serious complications (Panpanich et al., 2006). Randomized controlled study of two treatment groups treated with or without intravenous immunoglobulin also showed no effect in hastening recovery of platelet counts in patients with secondary dengue infection (Dimaano et al., 2007).
In contrast, interim data from two randomized placebo-controlled trials in 47 patients with DHF and thrombocytopenia have shown increase in platelet count with anti-D immune globulin (De Castro et al., 2007). The use of recombinant activated factor VII (rFVIIa) may also be useful in DHF with massive bleeding. Effective response was reported in 8/15 (53%) Thai children with grades III and IV DHF and life-threatening bleeding who were treated with rFVIIa (Chuansumrit et al., 2005).
IMPACT ON BLOOD SUPPLY
Explosive outbreaks of DHF are often associated with an increased need for blood components, especially platelets. When platelets are transfused based on commonly accepted consensus guidelines, about 8% of patients with DF and DHF will require platelet transfusions (Lye et al., 2008). During a large epidemic, this will translate to a significant increase in platelet requirements. During the height of the dengue epidemic in Singapore in 2005, a significant increase in platelet transfusions was recorded (Fig. 1), due to increased requirements from DHF patients.
Fig. 1.
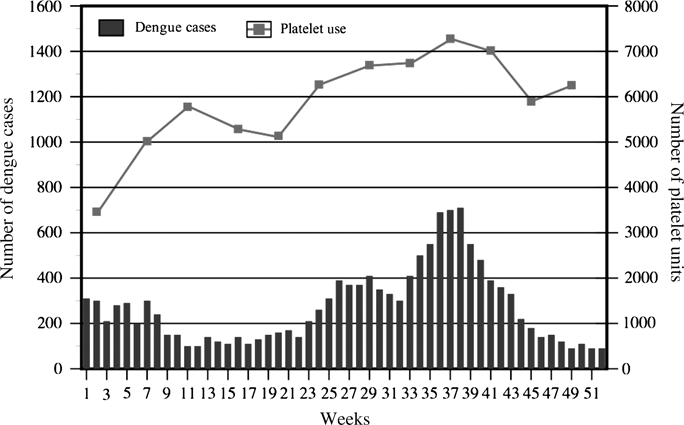
Comparison of platelet usage with number of dengue cases in Singapore in 2005.
Inappropriate ordering and use of platelets aggravate these increased requirements and, additionally, expose the patients to unnecessary transfusion risks. Chaudhary et al. (2006) report in their study that 35% of patients received unnecessary prophylactic transfusions, and inappropriate doses were given in 89% of transfusion episodes during a DF epidemic. In another study, 31 of 97 patients still received inappropriate transfusions despite the presence of platelet transfusion guidelines (Makroo et al., 2007).
In areas where blood services are less well developed and there is lack of transfusion specialists, lack of knowledge becomes a major factor leading to inappropriate use of blood and blood products. Lum et al. (2003) recommend that prevention of haemorrhage in patients with DHF is better achieved through early detection and management of circulatory imbalance and shock than through transfusions. The development of clear and specific guidelines for platelet and fresh frozen plasma transfusion in dengue may be useful in reducing unnecessary use of blood components particularly in areas with limited resources.
Wider access to medical care from health providers with knowledge of DHF can reduce death rates to less than 1% (World Health Organization, 2008). Unfortunately, in many developing countries, rural areas lack such access, and blood supply facilities are often very basic and poorly resourced. As dengue spreads to rural areas, the challenge of providing sufficient supplies of safe blood and blood components to treat DHF/DSS-associated medical emergencies will place a heavy demand on blood services in these countries.
REFERENCES
- Anderson JR, Rico-Hesse R. Aedes aegypti vectorial capacity is determined by the infecting genotype of dengue virus. American Journal of Tropical Medicine and Hygiene. 2006;75:886–892. [PMC free article] [PubMed] [Google Scholar]
- Azevedo LS, Carvalho DB, Matuck T, et al. Dengue in renal transplant patients: a retrospective analysis. Transplantation. 2007;84:792–794. doi: 10.1097/01.tp.0000280547.91617.25. [DOI] [PubMed] [Google Scholar]
- Beckett CG, Kosasih H, Faisal I, et al. Early detection of dengue infections using cluster sampling around index cases. American Journal of Tropical Medicine and Hygiene. 2005;72:777–782. [PubMed] [Google Scholar]
- Carles G, Peiffer H, Talarmin A. Effects of dengue fever during pregnancy in French Guiana. Clinical Infectious Diseases. 1999;28:637–640. doi: 10.1086/515144. [DOI] [PubMed] [Google Scholar]
- Chaudhary R, Khetan D, Sinha S, et al. Transfusion support to dengue patients in a hospital based blood transfusion service in north India. Transfusion and Apheresis Science. 2006;35:239–244. doi: 10.1016/j.transci.2006.08.007. [DOI] [PubMed] [Google Scholar]
- Chen LH, Wilson ME. Non-vector transmission of dengue and other mosquito-borne flaviviruses. Dengue Bulletin. 2005;29:18–30. [Google Scholar]
- Chuang VW, Wong TY, Leung YH, et al. Review of dengue fever cases in Hong Kong during 1998 to 2005. Hong Kong Medical Journal. 2008;14:170–177. [PubMed] [Google Scholar]
- Chuansumrit A, Wangruangsatid S, Lektrakul Y, Chua MN, Zeta Capeding MR, Bech OM, Dengue Study Group Control of bleeding in children with dengue hemorrhagic fever using recombinant activated factor VII: a randomized, double-blind, placebo-controlled study. Blood Coagulation & Fibrinolysis. 2005;16:549–555. doi: 10.1097/01.mbc.0000186837.78432.2f. [DOI] [PubMed] [Google Scholar]
- Dash PK, Parida MM, Saxena P, et al. Reemergence of dengue virus type-3 (subtype-III) in India: implications for increased incidence of DHF & DSS. Virology Journal. 2006;3 doi: 10.1186/1743-422X-3-55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Benedictis J, Chow-Schaffer E, Costero A, Clark GC, Edman JD, Scott TW. Identification of the people from whom engorged Aedes aegypti took blood meals in Florida, Puerto Rico, using polymerase chain reaction-based DNA profiling. American Journal of Tropical Medicine and Hygiene. 2003;68:437–446. [PubMed] [Google Scholar]
- De Castro RA, de Castro JA, Barez MY, Frias MV, Dixit J, Genereux M. Thrombocytopenia associated with dengue hemorrhagic fever responds to intravenous administration of anti-D (Rh(0)-D) immune globulin. American Journal of Tropical Medicine and Hygiene. 2007;76:737–742. [PubMed] [Google Scholar]
- Dimaano EM, Saito M, Honda S, et al. Lack of efficacy of high-dose intravenous immunoglobulin treatment of severe thrombocytopenia in patients with secondary dengue virus infection. American Journal of Tropical Medicine and Hygiene. 2007;77:1135–1138. [PubMed] [Google Scholar]
- García-Rivera EJ, Rigau-Pérez JG. Dengue severity in the elderly in Puerto Rico. Pan American Journal of Public Health. 2003;13:362–368. doi: 10.1590/s1020-49892003000500004. [DOI] [PubMed] [Google Scholar]
- Gubler DJ. Dengue and dengue hemorrhagic fever. Clinical Microbiology Reviews. 1998;11:480–496. doi: 10.1128/cmr.11.3.480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gubler DJ. Epidemic dengue/dengue hemorrhagic fever as a public health, social and economic problem in the 21st century. Trends in Microbiology. 2002;10:100–103. doi: 10.1016/s0966-842x(01)02288-0. [DOI] [PubMed] [Google Scholar]
- Gubler DJ, Suharyono W, Tan R, Abidin M, Sie A. Viraemia in patients with naturally acquired dengue infection. Bulletin of the World Health Organization. 1981;59:623–630. [PMC free article] [PubMed] [Google Scholar]
- Guglani L, Kabra S. T cell immunopathogenesis of dengue virus infection. Dengue Bulletin. 2005;29:58–69. [Google Scholar]
- Guha-Sapir D, Schimmer B. Dengue fever: new paradigms for a changing epidemiology. Emerging Themes in Epidemiology. 2005;2 doi: 10.1186/1742-7622-2-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta E, Dar L, Kapoor G, Broor S. The changing epidemiology of dengue in Delhi, India. Virology Journal. 2006;3 doi: 10.1186/1743-422X-3-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guzmán MG, Kouri G, Bravo J, Valdes L, Vazquez S, Halstead SB. Effect of age on outcome of secondary dengue 2 infections. International Journal of Infectious Diseases. 2002;6:118–124. doi: 10.1016/s1201-9712(02)90072-x. [DOI] [PubMed] [Google Scholar]
- Halperin D, Baetens J, Newman B. The effect of short-term, temporary deferral on future blood donation. Transfusion. 1998;38:181–183. doi: 10.1046/j.1537-2995.1998.38298193102.x. [DOI] [PubMed] [Google Scholar]
- Halstead SB, Streit TG, Lafontant JG, et al. Haiti: absence of dengue hemorrhagic fever despite hyperendemic dengue virus transmission. American Journal of Tropical Medicine and Hygiene. 2001;65:180–183. doi: 10.4269/ajtmh.2001.65.180. [DOI] [PubMed] [Google Scholar]
- Hammond SN, Balmaseda A, Pérez L, et al. Differences in dengue severity in infants, children, and adults in a 3-year hospital-based study in Nicaragua. American Journal of Tropical Medicine and Hygiene. 2005;73:1063–1070. [PubMed] [Google Scholar]
- Khan E, Siddiqui J, Shakoor S, Mehraj V, Jamil B, Hasan R. Dengue outbreak in Karachi, Pakistan, 2006: experience at a tertiary care center. Transactions of the Royal Society of Tropical Medicine and Hygiene. 2007;101:1114–1119. doi: 10.1016/j.trstmh.2007.06.016. [DOI] [PubMed] [Google Scholar]
- Kumarasamy V, Chua SK, Hassan Z, Wahab AH, Chem YK, Mohamad M, Chua KB. Evaluating the sensitivity of a commercial dengue NS1 antigen-capture ELISA for early diagnosis of acute dengue virus infection. Singapore Medical Journal. 2007;48:669–673. [PubMed] [Google Scholar]
- Lai Y, Chung Y, Tan H, Yap H, Yap G, Ooi E, Ng L. Cost-effective real-time reverse transcriptase PCR (RT-PCR) to screen for dengue virus followed by rapid single-tube multiplex RT-PCR for serotyping of the virus. Journal of Clinical Microbiology. 2006;45:935–941. doi: 10.1128/JCM.01258-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lam S, Tan HC, Tan LK, Ng LC, Teo D, Koh M. Efficacy of INTERCEPT treatment for the inactivation of dengue virus in single-donor platelet concentrate. Transfusion. 2007;47 [Google Scholar]
- Lee MS, Hwang KP, Chen TC, Lu PL, Chen TP. Clinical characteristics of dengue and dengue hemorrhagic fever in a medical center of southern Taiwan during the 2002 epidemic. Journal of Microbiology, Immunology and Infection. 2006;39:121–129. [PubMed] [Google Scholar]
- Linnen J, Vinelli E, Sabino E, et al. Dengue viremia in blood donors from Honduras, Brazil, and Australia. Transfusion. 2008;48:1355–1362. doi: 10.1111/j.1537-2995.2008.01772.x. [DOI] [PubMed] [Google Scholar]
- Liu J, Khor B, Lee C, Lee I, Chen R, Yang KD. Dengue haemorrhagic fever in Taiwan. Dengue Bulletin. 2003;27:19–24. [Google Scholar]
- Lum LC, Goh AY, Chan PW, El-Amin AL, Lam SK. Risk factors for hemorrhage in severe dengue infections. Journal of Pediatrics. 2002;140:629–631. doi: 10.1067/mpd.2002.123665. [DOI] [PubMed] [Google Scholar]
- Lum LC, Abdel-Latif MA, Goh AY, Chan PW, Lam SK. Preventive transfusion in dengue shock syndrome-is it necessary? Journal of Pediatrics. 2003;143:682–684. doi: 10.1067/s0022-3476(03)00503-1. [DOI] [PubMed] [Google Scholar]
- Lye DC, Chan M, Lee VJ, Leo YS. Do young adults with uncomplicated dengue fever need hospitalisation? A retrospective analysis of clinical and laboratory features. Singapore Medical Journal. 2008;49:476–479. [PubMed] [Google Scholar]
- Makroo RN, Raina V, Kumar P, Kanth RK. Role of platelet transfusion in the management of dengue patients in a tertiary care hospital. Asian Journal of Transfusion Science. 2007;1:4–7. doi: 10.4103/0973-6247.28065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malavige G. Patterns of disease among adults hospitalized with dengue infections. QJM. 2006;99:299–305. doi: 10.1093/qjmed/hcl039. [DOI] [PubMed] [Google Scholar]
- Marshall DA, Kleinman SH, Wong JB, AuBuchon JP, Grima DT, Kulin NA, Weinstein MC. Cost-effectiveness of nucleic acid test screening of volunteer blood donations for hepatitis B, hepatitis C and human immunodeficiency virus in the United States. Vox Sanguinis. 2004;86:28–40. doi: 10.1111/j.0042-9007.2004.00379.x. [DOI] [PubMed] [Google Scholar]
- Méndez F, Barreto M, Arias JF, Rengifo G, Muñoz J, Burbano ME, Parra B. Human and mosquito infections by dengue viruses during and after epidemics in a dengue-endemic region of Colombia. American Journal of Tropical Medicine and Hygiene. 2006;74:678–683. [PubMed] [Google Scholar]
- Messer WB, Gubler DJ, Harris E, Sivananthan K, de Silva AM. Emergence and global spread of a dengue serotype 3, subtype III virus. Emerging Infectious Diseases. 2003;9:800–809. doi: 10.3201/eid0907.030038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitrakul C, Poshyachinda M, Futrakul P, Sangkawibha N, Ahandrik S. Hemostatic and platelet kinetic studies in dengue hemorrhagic fever. American Journal of Tropical Medicine and Hygiene. 1977;26:975–984. doi: 10.4269/ajtmh.1977.26.975. [DOI] [PubMed] [Google Scholar]
- Mohammed H, Linnen J, Muoz-Jordn J, Tomashek K, Foster G, Broulik A, Petersen L, Stramer S. Dengue virus in blood donations, Puerto Rico, 2005. Transfusion. 2008;48:1348–1354. doi: 10.1111/j.1537-2995.2008.01771.x. [DOI] [PubMed] [Google Scholar]
- Mongkolsapaya J, Dejnirattisai W, Xu XN, et al. Original antigenic sin and apoptosis in the pathogenesis of dengue hemorrhagic fever. Nature Medicine. 2003;9:921–927. doi: 10.1038/nm887. [DOI] [PubMed] [Google Scholar]
- Murgue B, Roche C, Chungue E, Deparis X. Prospective study of the duration and magnitude of viraemia in children hospitalised during the 1996-1997 dengue-2 outbreak in French Polynesia. Journal of Medical Virology. 2000;60:432–438. doi: 10.1002/(sici)1096-9071(200004)60:4<432::aid-jmv11>3.0.co;2-7. [DOI] [PubMed] [Google Scholar]
- Nakao S, Lai CJ, Young NS. Dengue virus, a flavivirus, propagates in human bone marrow progenitors and hematopoietic cell lines. Blood. 1989;74:1235–1240. [PubMed] [Google Scholar]
- Nishiura H, Halstead S. Natural history of dengue virus (DENV)-1 and DENV-4 infections: reanalysis of classic studies. Journal of Infectious Disease. 2007;195:1007–1013. doi: 10.1086/511825. [DOI] [PubMed] [Google Scholar]
- Ooi EE, Goh KT, Gubler DJ. Dengue prevention and 35 years of vector control in Singapore. Emerging Infectious Diseases. 2006;12:887–893. doi: 10.3201/10.3201/eid1206.051210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pancharoen C, Mekmullica J, Thisyakorn U. Primary dengue infection: what are the clinical distinctions from secondary infection? Southeast Asian Journal of Tropical Medicine and Public Health. 2001;32:476–480. [PubMed] [Google Scholar]
- Panpanich P, Somchai P, Kanjanaratanakorn K. Corticosteroids for treating dengue shock syndrome. Cochrane Database of Systemic Reviews. 2006;3 doi: 10.1002/14651858.CD003488.pub2. online. [DOI] [PubMed] [Google Scholar]
- Poblap T, Nitatpattana N, Chaimarin A, Barbazan P, Chauvancy G, Yoksan S, Gonzalez JP. Silent transmission of virus during a dengue epidemic, Nakhon Pathom Province, Thailand 2001. Southeast Asian Journal of Tropical Medicine & Public Health. 2006;37:899–903. [PubMed] [Google Scholar]
- Rigau-Pérez JG, Vorndam AV, Clark GG. The dengue and dengue hemorrhagic fever epidemic in Puerto Rico, 1994-1995. American Journal of Tropical Medicine and Hygiene. 2001;64:67–74. doi: 10.4269/ajtmh.2001.64.67. [DOI] [PubMed] [Google Scholar]
- Savage HM, Fritz CL, Rutstein D, Yolwa A, Vorndam V, Gubler DJ. Epidemic of dengue-4 virus in Yap State, Federated States of Micronesia, and implication of Aedes hensilli as an epidemic vector. American Journal of Tropical Medicine and Hygiene. 1998;58:519–524. doi: 10.4269/ajtmh.1998.58.519. [DOI] [PubMed] [Google Scholar]
- Scott TW, Naksathit A, Day JF, Kittayapong P, Edman JD. A fitness advantage for Aedes aegypti and the viruses it transmits when females feed only on human blood. American Journal of Tropical Medicine and Hygiene. 1997;57:235–239. doi: 10.4269/ajtmh.1997.57.235. [DOI] [PubMed] [Google Scholar]
- Shu PY, Chang SF, Kuo YC, Yueh YY, Chien LJ, Sue CL, Lin TH, Huang JH. Development of group- and serotype-specific one-step SYBR green I-based real-time reverse transcription-PCR assay for dengue virus. Journal of Clinical Microbiology. 2003;41:2408–2416. doi: 10.1128/JCM.41.6.2408-2416.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sirinavin S, Nuntnarumit P, Supapannachart S, Boonkasidecha S, Techasaensiri C, Yoksarn S. Vertical dengue infection. Pediatric Infectious Disease Journal. 2004;23:1042–1047. doi: 10.1097/01.inf.0000143644.95692.0e. [DOI] [PubMed] [Google Scholar]
- Tai DY, Chee YC, Chan KW. The natural history of dengue illness based on a study of hospitalised patients in Singapore. Singapore Medical Journal. 1999;40:238–242. [PubMed] [Google Scholar]
- Tambyah P, Koay E, Poon M, Lin R, Ong B. Dengue hemorrhagic fever transmitted by blood transfusion. New England Journal of Medicine. 2008;359:1526–1527. doi: 10.1056/NEJMc0708673. [DOI] [PubMed] [Google Scholar]
- Tan FL-S, Loh DLSK, Prabhakaran K. Dengue haemorrhagic fever after living donor renal transplantation. Nephrology Dialysis Transplantation. 2005;20:447–448. doi: 10.1093/ndt/gfh601. [DOI] [PubMed] [Google Scholar]
- Vaughn DW, Green S, Kalayanarooj S, et al. Dengue in the early febrile phase: viremia and antibody responses. Journal of Infectious Disease. 1997;176:322–330. doi: 10.1086/514048. [DOI] [PubMed] [Google Scholar]
- Wagenaar J, Mairuhu A, Gorp Ev. Genetic influences on dengue virus infections. Dengue Bulletin. 2004;28 [Google Scholar]
- World Health Organization . Dengue Haemorrhagic Fever – Diagnosis, Treatment, Prevention and Control. 2nd edn. Geneva: WHO; 1997. [Google Scholar]
- World Health Organization . Dengue and Dengue Haemorrhagic Fever. Geneva: WHO; 2008. WHO Fact Sheet. [Google Scholar]


