Abstract
Treatment of patients with allergic asthma using low doses of peptides containing T cell epitopes from Fel d 1, the major cat allergen, reduces allergic sensitization and improves surrogate markers of disease. Here, we demonstrate a key immunological mechanism, linked epitope suppression, associated with this therapeutic effect. Treatment with selected epitopes from a single allergen resulted in suppression of responses to other (“linked”) epitopes within the same molecule. This phenomenon was induced after peptide immunotherapy in human asthmatic subjects and in a novel HLA-DR1 transgenic mouse model of asthma. Tracking of allergen-specific T cells using DR1 tetramers determined that suppression was associated with the induction of interleukin (IL)-10+ T cells that were more abundant than T cells specific for the single-treatment peptide and was reversed by anti–IL-10 receptor administration. Resolution of airway pathophysiology in this model was associated with reduced recruitment, proliferation, and effector function of allergen-specific Th2 cells. Our results provide, for the first time, in vivo evidence of linked epitope suppression and IL-10 induction in both human allergic disease and a mouse model designed to closely mimic peptide therapy in humans.
In previous clinical studies, intradermal (i.d.) injection of allergen-derived peptides resulted in modulation of surrogate markers of disease in allergic asthma; early- and late-phase skin responses to allergen were reduced (Oldfield et al., 2002), along with nasal symptoms (Alexander et al., 2005a) and airway hyperreactivity (AHR; Alexander et al., 2005b). Peptide treatment suppressed allergen-specific T cell proliferation and production of IL-4, IL-13, and IFN-γ, whereas IL-10 production was enhanced (Oldfield et al., 2001; Oldfield et al., 2002). The immunological mechanisms responsible have yet to be defined, but may include clonal deletion, induction of anergy (for review see Schwartz 2003), and/or active regulation (Sundstedt et al., 2003; Apostolou and Von Boehmer, 2004). We recently demonstrated induction of functional allergen-specific regulatory T cells after peptide therapy (Verhoef et al., 2005).
Our clinical studies have focused on peripheral blood responses to allergen before and after therapy. A limitation of studying asthmatic human subjects is that it is not possible to directly examine local allergen-specific T cell responses within lung tissue. To address this issue, we have developed a model of Fel d 1 (the major allergen of the domestic cat, Felis domesticus)–induced allergic lung inflammation in a mouse transgenic for the human MHC class II molecule (HLA-DR1; DRA/DRB1*0101) and lacking endogenous mouse MHC class II (H2-Aβo). We were thus able to investigate mechanisms of peptide therapy–induced immunomodulation using the same peptides and route of treatment as that used in human therapy. Low-dose (1 µg) peptide therapy was used, reflecting our current clinical practice, and was substantially lower than previously used in mouse models. Immunological mechanisms were investigated by analyzing T cell responses to treatment peptides and nontreatment peptides from the same allergen molecule and using MHC class II (HLA-DR1) tetramers.
Here, we provide evidence for the induction of linked epitope suppression in both human and mouse responses, together with substantial induction of IL-10 in T cells of mice, which was not limited to cells specific for the treatment peptide. In mice, reduced Th2 cell recruitment to the lung and suppressed effector cell function resulted in IL-10–dependent resolution of allergic lung inflammation, directly demonstrating treatment efficacy of peptide therapy in this model. The common responses to cat peptide therapy in humans and mice, as well as clear reduction in allergic inflammation in the mouse model, provide evidence that peptide therapy suppresses allergic asthma via induction of IL-10 and linked epitope suppression.
RESULTS
Peptide immunotherapy induces linked epitope suppression of antigen-specific responses in allergic asthmatic subjects
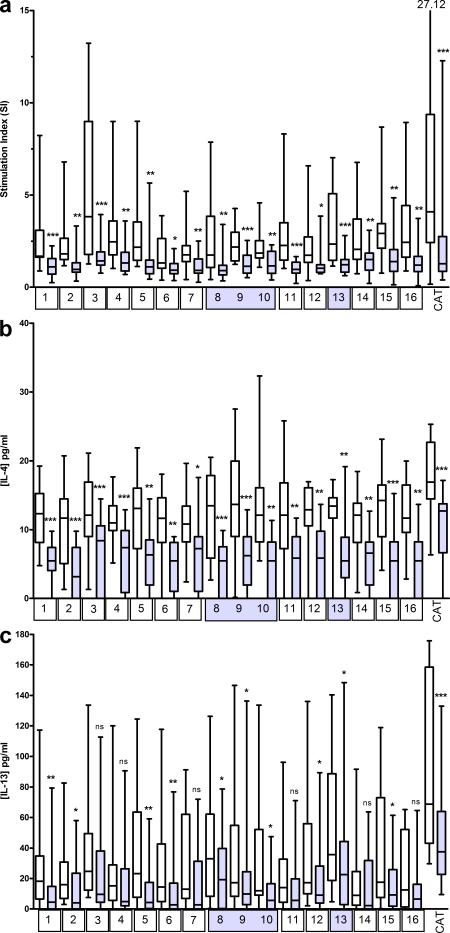
To determine the effect of Fel d 1 peptide therapy on responses to Fel d 1 peptides not included in the therapeutic cocktail, we examined ex vivo peptide-specific proliferative responses and cytokine production from PBMCs of peptide-treated allergic asthmatics. PBMC proliferative responses to each of 16 Fel d 1 peptides (12 peptides from a therapeutic peptide vaccine; “treatment peptides,” and 4 Fel d 1 peptides not included in the vaccine; “non-treatment peptides”) were assessed in a randomized, double-blind, placebo-controlled trial in 24 cat-allergic asthmatic subjects. Clinical outcomes are reported elsewhere (Oldfield et al., 2002). Responses to all 12 treatment peptides were significantly reduced in the 16 subjects receiving active treatment, but not in the 8 subjects receiving placebo. Strikingly, responses to the four nontreatment peptides were significantly reduced in the active treatment group only (Fig. 1 a). Cytokine secretion followed a similar pattern. IL-4 production to all peptides (treatment and nontreatment) was significantly reduced in the active treatment, but not the placebo group (Fig. 1 b). IL-13 responses were more variable, with those to 6/12 treatment peptides significantly reduced, together with all nontreatment peptides (Fig. 1 c). IFN-γ responses were generally low but variable, with those to 9/12 treatment peptides significantly reduced, together with all nontreatment peptides (unpublished data). No significant changes in proliferative or cytokine responses to the control recall antigen (in a routinely vaccinated population) purified protein derivative of Mycobacterium tuberculosis were observed.
Figure 1.
Peptide immunotherapy in cat allergic asthmatic subjects induces linked epitope suppression. PBMCs were isolated before and after a randomized, double-blind, placebo-controlled trial of peptide immunotherapy and frozen. Cells were thawed and cultured with individual peptides and with cat allergen extract for 6 d before quantification of proliferation and cytokine release. Assays were performed once on both pre- and posttreatment samples. Peptides shown by number on the x axis correspond to amino acid sequences in chain 1 (peptides 1–7) and chain 2 (peptides 8–16) of Fel d 1 sequence as follows: 1:1–17, 2:12–28, 3:23–38, 4:29–45, 5:39–55, 6:48–63, 7:54–69, 8:1–16, 9:7–23, 10:20–35, 11:29–44, 12:40–55, 13:48–63, 14:56–71, 15:67–82, 16:77–92. (a) Proliferation data were normalized by conversion to stimulation index (SI; cpm for peptide/allergen culture divided by cpm of medium alone culture). Peptide immunotherapy resulted in statistically significant reductions in proliferative responses to cat allergen extract and to treatment peptides and nontreatment peptides (peptide number shown in shaded boxes). The response to each peptide is represented in paired bars. Pretreatment, open bars; posttreatment, filled bars. IL-4 (b) and IL-13 (c) production to individual peptides and cat allergen extract was reduced as indicated by statistical significance. No significant changes in proliferative or cytokine responses to the control antigen–purified protein derivative (PPD) of Mycobacterium tuberculosis were observed. Data are median, range, and interquartiles (box-and-whisker plot). *, P < 0.05; **, P < 0.01; ***, P < 0.001.
Peptide immunotherapy reduces lung inflammation and improves lung function in sensitized and rechallenged DR1-tg/Aβo mice
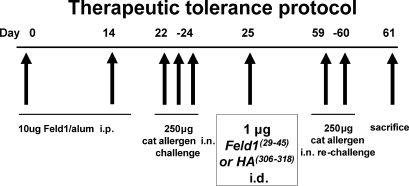
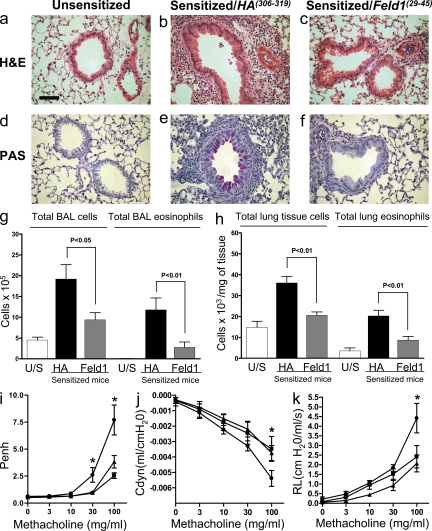
To investigate the cellular and molecular mechanisms leading to immunosuppression of allergic responses after peptide immunotherapy, we have developed a specific mouse model. We generated mice transgenic for human HLA DR1, but deficient in mouse MHC class II, thus MHC class II–mediated antigen presentation in the DR1-tg/Aβo model was restricted to HLA-DR1–binding peptides. We chose a dosing schedule that was similar to that used in our human study and used a single peptide (residues 29–45 of chain 1 of the cat allergen Fel d 1, referred to here as Feld1[29–45], which was also a component of the clinical vaccine) to induce tolerance in allergen-primed animals to subsequent inhaled challenge with an aqueous extract containing Fel d 1 and other cat proteins (Fig. 2; sensitization and treatment protocol). We have previously shown that peptide immunotherapy in allergic asthmatic subjects improves surrogate clinical markers of airway inflammation (Alexander et al., 2005a; Alexander et al., 2005b). Therefore, we determined whether the peptide immunotherapy schedule had a similar effect on lung inflammation and function in our mouse model. Mice that were sham sensitized but challenged with allergen did not exhibit any pulmonary inflammation (Fig. 3 a). Animals treated with the control HA(306–318) (residues 306–318 of influenza hemagglutinin) peptide showed extensive peribronchial and perivascular eosinophilic inflammatory infiltrates comparable to those seen in mice sensitized and challenged with cat allergen (Fig. 3 b and not depicted). In contrast, mice treated with Feld1(29–45) had substantially reduced airway inflammation (Fig. 3 c) compared with HA(306–318)-treated animals. Importantly, we could assess airway inflammation in both the lumen (by lavage) and lung tissue in the mouse model. Compared with unsensitized mice (Fig. 3 d), exposure to cat allergen in sensitized mice was marked by goblet cell hyperplasia and mucus hypersecretion (Fig. 3 e). This was reduced after treatment with Feld1(29–45) (Fig. 3 f). The effect of Feld1(29–45) treatment was reflected in relative levels of leukocytes in the lung lumen. Eosinophils made up >60% of the BAL inflammatory infiltrate, the remaining cells being macrophages, lymphocytes, and a few neutrophils. Total BAL leukocytes and eosinophils were significantly reduced in Feld1(29–45) versus HA(306–318)-treated mice (Fig. 3 g). Both total leukocytes and eosinophils were also significantly reduced in digested lung tissue after Feld1(29–45) treatment (Fig. 3 h). Thus, a single therapeutic i.d. administration of Feld1(29–45) was able to significantly reduce allergic lung inflammation in both airway lumen and tissue after allergen rechallenge weeks later.
Figure 2.
Schematic protocol flow chart for cat allergen-induced airway disease and therapeutic treatment with Fel d 1 peptide. Airway inflammation was induced in DR1-tg/Aβo mice sensitized with 10 µg recombinant Fel d 1 in alum (days 0 and 14) and challenged i.n. with 250 µg cat allergen extract (days 22, 23, and 24). To induce tolerance, a single i.d. injection of 1 µg of Feld1(29–45) (or HA(306–318) as a DR1-binding control) was administered (day 25) after establishment of eosinophilic inflammation. Mice were rechallenged with cat allergen extract (days 59 and 60), lung function was analyzed, and the mice were sacrificed (day 61) for tissue harvest. Unsensitized control mice received PBS in alum and were challenged in the same manner as sensitized animals.
Figure 3.
Peptide immunotherapy reduces allergic lung inflammation and improves lung function. MHC II (DR1-tg/Aβ0) mice were sensitized with PBS (unsensitized control) or rFel d 1 in alum and allergic inflammation localized to the lung by repeated i.n. challenge with cat allergen extract. Mice were treated by i.d. injection of HA(306–318) (control peptide) or Feld1(29–45) (treatment peptide) and rechallenged with cat allergen extract (i.n.). Representative lung histology from unsensitized (a and d), allergen-sensitized control peptide-treated (b and e), and allergen-sensitized allergen peptide-treated (c and f) mice. Mice were sacrificed 24 h after the final cat allergen challenge. Paraffin-embedded lung was sectioned (4 µM) and stained with hematoxylin and eosin (a-c) or periodic acid schiff for detection of mucin (d–f). Representative sections are shown for each treatment group. Bar, 50 µm. Original magnification, 400×. (g) Airways were lavaged with PBS via a tracheal cannula. BAL cells were transferred to slides by cytocentrifugation and differential cell counts were performed by counting eight high power fields (40× magnification; total area, 0.5 mm2 per area randomly selected) and dividing this number by the total number of cells per high power field. To obtain absolute numbers of each leukocyte subtype, percentages were multiplied by the total number of cells obtained in the lavage fluid. (h) Single lobes of lung tissue were digested with collagenase and DNase as described in the Materials and methods. Differential counts were performed on cytocentrifuge preparations as for BAL. (i) AHR was measured at day 60 by whole-body plethysmography in response to inhaled methacholine (3–100 mg/ml) for 1 min. Penh data were confirmed by direct measurement of airway resistance (RL; j) and lung dynamic compliance (Cdyn; k) in anesthetized and tracheotomized mice in response to inhaled methacholine (3–100 mg/ml). For i and j, Unsensitized mice are represented by squares, sensitized/HA(306–318) by circles, and sensitized/Feld1(29–45) by triangles. All data are expressed as mean ± SEM. n = 6 mice/group. Two independent experiments were performed. *, P < 0.05 for sensitized HA(306–318) versus DR1Feld1-treated mice, Mann-Whitney U test.
To determine whether the reduction in bronchial inflammation was associated with improved lung function in cat allergen–sensitized DR1-tg/Aβo mice, we measured AHR to methacholine by whole body plethysmography 24 h after the first allergen rechallenge (d59) in addition to direct measurements of lung airway resistance and dynamic compliance in mechanically ventilated mice 24 h after the second allergen rechallenge (d60). Cat allergen sensitization and challenge resulted in airway inflammation and a methacholine dose-dependent increase in enhanced pause (Penh) at day 24 (unpublished data). Animals were subsequently treated with HA(306–318) or Feld1(29–45) and rechallenged with allergen 4 wk later. i.d. treatment with Feld1(29–45) significantly reduced Penh responses at 30 and 100 mg/ml methacholine doses to levels observed in unsensitized mice (Fig. 3 i). These data were confirmed by lung resistance and compliance measurements at the 100 mg/ml methacholine dose (Fig. 3, j and k). Thus, the reduction in cellular and tissue inflammation after Feld1(29–45) therapy was associated with a significant improvement in lung function.
Peptide immunotherapy induces linked epitope suppression of antigen-specific T cell responses in the mouse lung
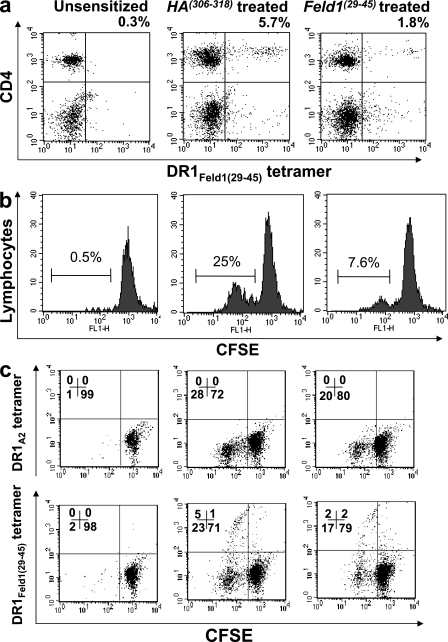
To investigate the mechanisms behind the effect of peptide immunotherapy on allergic pathophysiology, we studied specific T cell responses in the lungs of treated and untreated mice. Because we were using DR1tg mice we could use an HLA-DR tetramer incorporating the treatment peptide (DR1Feld1[29–45]tetramer) to track allergen-specific cells in the lung and to study specific responses to allergen. The number of tetramer+ cells in the lungs was small. Therefore, to generate a detectable amount of cells, samples within each group were pooled and restimulated with recombinant Fel d 1 (rFel d 1) protein in vitro. Cells were labeled with CFSE to evaluate antigen-specific proliferative responses in Feld1(29–45)-treated versus HA(306–318) (control peptide)-treated mice. Representative data plots of lung tissue digest cultures stained with CD4 and DR1Feld1(29–45)tetramer demonstrate that Feld1(29–45) treatment reduced the number of CD4+DR1Feld1(29–45)tetramer+ cells (Fig. 4 a). Feld1(29–45) treatment reduced antigen-driven lymphocyte proliferation (Fig. 4 b). Cells from unsensitized mice did not proliferate in response to rFel d 1. In contrast, cells from sensitized, challenged, and control peptide–treated mice demonstrated substantial proliferation to rFel d 1, which was markedly reduced after Feld1(29–45) treatment.
Figure 4.
Peptide immunotherapy reduces proliferative responses to Fel d 1. Mice were sacrificed at day 61 and cells from collagenase digests of lung tissue (cells pooled/treatment group of 6 mice) were labeled with CFSE and restimulated with Fel d 1 for 6 d. Cells were stained with HLA-DR1 tetramers specific for the treatment peptide (DR1Feld1[29–45]tetramer) or a control peptide (DR1A2tetramer) and analyzed by flow cytometry. (a) DR1Feld1(29–45)tetramer and CD4 double staining of cells from unsensitized (left), sensitized and treated with control peptide HA(306–318) (middle), and sensitized mice treated with Feld1(29–45) (right). (b) CFSE dilution analysis of total lymphocyte population (defined by scatter) in cells from unsensitized (left), sensitized control peptide-treated (middle), and Feld1(29–45)-treated mice (right). (c) CFSE dilution analysis of treatment peptide Feld1(29–45)–specific cells. (top row) Proliferation of CFSE-labeled lung digest cells labeled with control tetramer (DR1A2tetramer). (bottom row) Proliferation of CFSE-labeled lung digest cells labeled with DR1Feld1(29–45)tetramer. Representative plots are shown from three independent experiments. n = 6/group.
To determine a mechanistic link between amelioration of cat allergy and Feld1(29–45) treatment, we examined relative populations of Feld1(29–45)+ and Feld1(29–45)neg cells (i.e., T cells specific for the treatment peptide and T cells specific for other Fel d 1 epitopes, respectively) in ex vivo cell cultures stained with CFSE and stimulated with whole rFel d 1. The combination of CFSE and tetramer staining of allergen-stimulated cells allowed us to identify allergen-specific cycling cells that were treatment peptide-specific (tetramer+) and those specific for other Fel d 1 DR1-restricted epitopes (tetramerneg). We were thus able to visualize the effect of treatment with a single epitope on the T cell response to other epitopes in the same molecule (linked epitopes). Treatment with Feld1(29–45) decreased numbers of DR1Feld1(29–45)tetramer+ cells and DR1Feld1(29–45)tetramerneg cells proliferating to other DR1-restricted epitopes of Fel d 1 (Fig. 4 c). No staining of cells was observed with control tetramer (DR1A2tetramer, which contained a DR1-binding peptide from HLA-A2; Fig. 4 c, top) or in unsensitized mice (Fig. 4 c, left). Staining of cells with DR1Feld1(29–45)tetramer demonstrated that a proportion of proliferating cells in control peptide-treated mice were specific for Feld1(29–45). Their numbers were unaffected by control peptide treatment. After treatment with Feld1(29–45), the number of DR1Feld1(29–45)tetramer+ cells was reduced (Fig. 4 c, bottom right versus middle). Furthermore, the proliferative response of DR1Feld1(29–45)tetramerneg cells was also reduced (Fig. 4 c, bottom). This is indicative of active regulation through the induction of linked suppression (intramolecular tolerance) because treatment targeting T cells specific for one epitope of a molecule down-regulated the response to other epitopes.
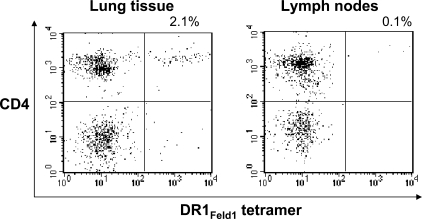
Another important observation from this experiment is that no increase in DR1Feld1(29–45)tetramer+ noncycling cells was observed, suggesting that induction of anergy in airway T cells was not a contributing mechanism in this model. The possibility that these cells had trafficked to draining lymph nodes was investigated by tetramer staining of cell suspensions. Strikingly, there were very few DR1Feld1(29–45)tetramer+ cells in restimulated draining lymph node cell cultures compared with lung tissue cell cultures (Fig. 5). Collectively, these data in DR1 transgenic mice provide evidence of Feld1(29–45) treatment–induced linked epitope suppression associated with largely resolved lung inflammation and improved lung function.
Figure 5.
Lung tissue digests, but not draining lymph nodes, contain T cells specific for the treatment peptide. Mice were sacrificed at day 61 and cells from draining lymph nodes and collagenase digests of lung tissue were restimulated with Fel d 1 for 6 d. Cells were stained with HLA-DR1 tetramers specific for the treatment peptide (DR1Feld1[29–45]tetramer) or a control peptide (DR1A2tetramer; not depicted) and analyzed by flow cytometry. Representative plots are shown from three independent experiments. n = 6 mice/group.
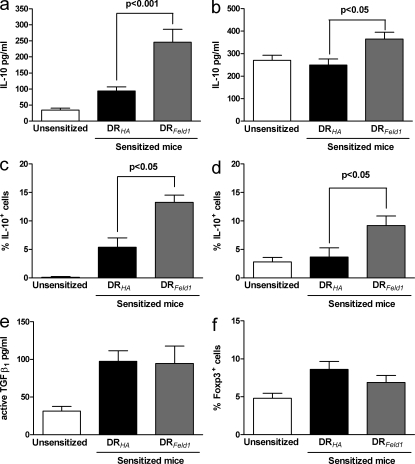
Cat allergen–sensitized and –challenged DR1-tg/Aβo mice demonstrate increased IL-10 after peptide immunotherapy, but no change in TGF-β or Foxp3
Previous studies have implicated IL-10 and TGF-β as important mediators in the regulation of in vivo immune responses; therefore, we investigated whether IL-10 or TGF-β expression were associated with tolerance in our mouse model of cat allergy. We have previously reported induction of IL-10 in human peptide immunotherapy (Oldfield et al., 2002; Tarzi et al., 2006). Feld1(29–45) treatment in allergic mice was associated with a significant increase in IL-10 protein in bronchoalveolar lavage (BAL) and homogenates of lung tissue (Fig. 6, a and b). As IL-10 can be produced by many cell types, we determined whether allergen-specific CD4+ T cells contributed to increased IL-10 production. Lung tissue digest cells were cultured with rFel d 1 in vitro for 6 d, and then restimulated. Mice that had been treated with Feld1(29–45) had a significantly higher proportion of IL-10+ CD4+ cells and IL-10+ lymphocytes than control peptide-treated mice (Fig. 6, c and d). A substantial proportion of all CD4+ cells and lymphocytes stained positive for IL-10 after Feld1(29–45) treatment (>10%), indicating that Feld1(29–45) treatment induced a population of IL-10–producing CD4+ T cells that was much larger than the pool of cells specific for the treatment peptide (∼2%; Fig. 4).
Figure 6.
Peptide immunotherapy is associated with increased IL-10 production, but no role is evident for TGF-β or Foxp3. Mice were sacrificed at day 61 and subjected to BAL. Individual lobes of lung tissue were enzymatically digested to release inflammatory cells. (a) IL-10 levels in BAL fluid were quantified by ELISA. (b) IL-10 levels in lung digest homogenates (100 mg of tissue) were quantified by ELISA. (c) The percentage of CD4+ cells within lung tissue digests expressing IL-10 was detected by flow cytometry. (d) The percentage of total lymphocytes within lung tissue digests expressing IL-10. (e) Levels of active TGF-β1 were measured in BAL by ELISA. (f) Foxp3 expression in CD4+ lung tissue digest cells was measured by flow cytometry. For all graphs, white bars represent responses of cells from unsensitized mice and black bars and gray bars represent responses of sensitized HA(306–318)-treated and sensitized Feld1(29–45)-treated mice, respectively. P values were determined using Mann-Whitney U test. Mean data ± SEM from three independent experiments each with five mice per group.
Active TGF-β was measured by ELISA in BAL and lung tissue homogenates. Feld1(29–45) treatment had no effect on active TGF-β levels (Fig. 6 e; BAL data shown). TGF-β was not detectable by flow cytometry in or on CD4+ T cells (not depicted). Foxp3 has been described as marker of regulatory T cells, and we were indeed able to detect intracellular Foxp3 expression in lung tissue digest, BAL, and peribronchial lymph node CD4+ cells. However, there was no significant increase in numbers of Foxp3+ CD4+ cells from Feld1(29–45)-treated compared with HA(306–318)-treated mice (Fig. 6 f; data for lung tissue cells shown).
Pulmonary and systemic Th2 responses are reduced in peptide-treated mice
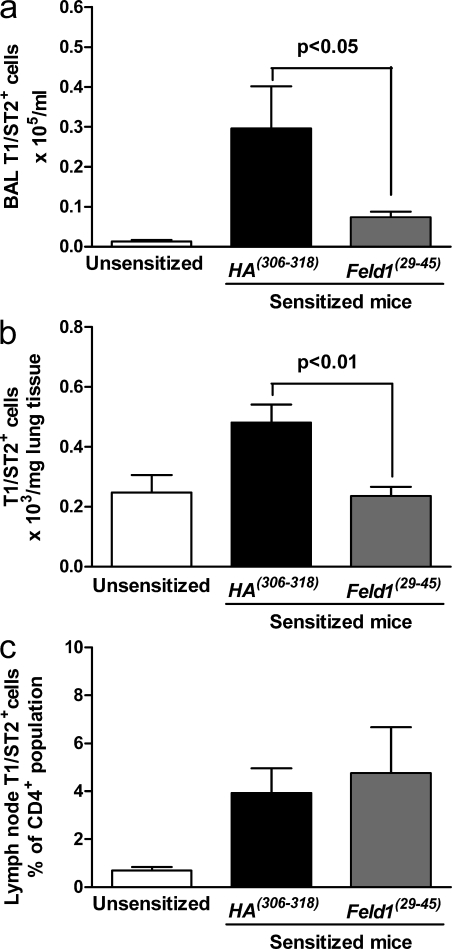
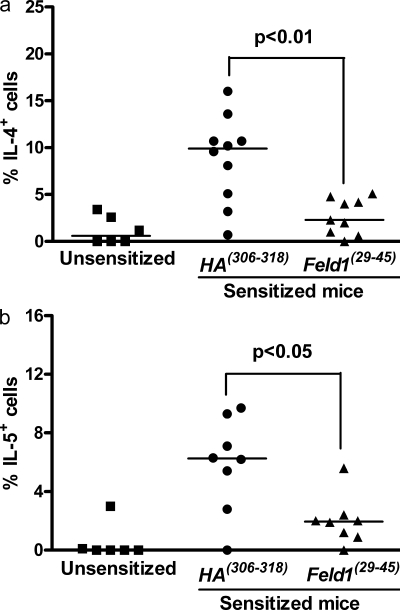
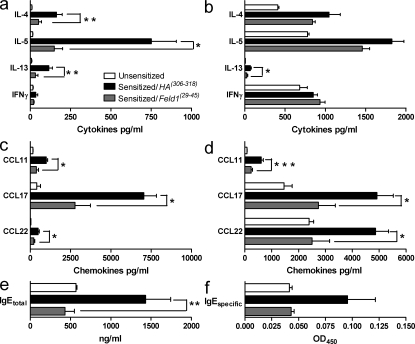
Peptide immunotherapy was associated with an improvement in allergen-driven inflammation and lung function, both of which are dependent on Th2 cells (Coyle et al., 1999). Therefore, we assessed the effect of peptide treatment on Th2 function and recruitment. Peptide immunotherapy was associated with significantly decreased recruitment of Th2 cells in BAL and digested lung tissue (Fig. 7), although recruitment to draining lymph nodes was unaffected. Numbers of CD4+ T cells synthesizing IL-4 or IL-5 were significantly reduced in lung tissue digest cells after therapy (Fig. 8). Interestingly, no recruitment of these cells was seen in draining lymph nodes after allergen sensitization and challenge. We measured Th2 cytokines (IL-4, IL-5, and IL-13) and the Th1 cytokine IFN-γ in BAL and in digested lung homogenates of unsensitized and sensitized mice treated with HA(306–318) or Feld1(29–45) before inhaled allergen rechallenge (Fig. 2). Allergen sensitization resulted in significant elevation of Th2 cytokines. Levels of IL-4, IL-5, and IL-13 were elevated in sensitized/HA(306–318)-treated mice. Feld1(29–45) administration substantially reduced Th2 cytokine levels in the BAL with more modest reductions in lung tissue homogenates (Fig. 9, a and b). IFN-γ levels were only marginally elevated in this model and not significantly modulated by Feld1(29–45) treatment. The chemokines CCL11/eotaxin, CCL17/TARC and CCL22/MDC are produced chiefly by epithelial cells and attract CCR3 (receptor for CCL11) and CCR4 (receptor for CCL17 and 22) expressing eosinophils and Th2 cells, respectively (Daugherty et al., 1996; Sallusto et al., 1997; Lloyd et al., 2000). Hence, we determined by ELISA whether these chemokines were altered in mice treated with DR1Feld1. CCL11, CCL17, and CCL22 were elevated in sensitized and challenged mice. i.d. treatment with Feld1(29–45) significantly reduced levels of these chemokines in the BAL and in lung tissue homogenates compared with HA(306–318) treatment (Fig. 9, c and d). Elevated systemic IgE is also associated with allergic airways disease. In our DR1-tg/Aβo model, total serum IgE and Fel d 1–specific IgE were increased in sensitized and challenged mice. Treatment with Feld1(29–45) but not HA(306–318) significantly reduced levels of IgE in the serum (Fig. 9, e and f). Thus, i.d. treatment with Feld1(29–45) reduced both systemic and local lung Th2 responses to cat allergen.
Figure 7.
Peptide immunotherapy reduces Th2 cell recruitment to the lung. T1ST2+ (a Th2 cell phenotypic marker) cells were measured in the BAL (a), lung tissue digest (b), and peri-bronchial lymph nodes (c) by flow cytometry. Mice were sacrificed at day 61, and cells were isolated and stained with anti–mouse CD4 and anti–mouse T1ST2 as described in the Materials and methods. For BAL and lung tissue digest, data are expressed as mean T1ST2+/CD4+ cell numbers ± SEM by multiplying the percentage expression on live cells by the total cell counts. For lymph node cells, data shown are the percentage of expression within the CD4+ population. T1ST2 expression on CD4+ cells of unsensitized mice (white bars), sensitized/HA(306–318)-treated (black bars), and sensitized/Feld1(29–45)-treated mice (gray bars). Data are mean ± SEM. n = 5 mice/group and data are representative of 4 independent experiments. *, P < 0.05 and **, P < 0.01 for sensitized/HA(306–318) versus sensitized/Feld1(29–45) treated groups, Mann-Whitney U test.
Figure 8.
Peptide immunotherapy reduces proportions of lung tissue IL-4+ and IL-5+ CD4+ cells after antigen restimulation in vitro. Mice were sacrificed at day 61 and cells from collagenase digests of lung tissue were restimulated in vitro with 10 µg/ml rFel d 1 for 6 d. Cells were further stimulated with PMA/ionomycin in the presence of brefeldin A for 4 h. (a) Percentage of IL-4+ cells CD4+ cells. (b) Percentage of IL-5+ cells CD4+ cells are shown with isotype control staining subtracted. Individual data points are shown with medians indicated. n = 6–10/group from 2 independent experiments. P values were determined using Mann-Whitney U test.
Figure 9.
Peptide immunotherapy inhibits levels of Th2 cytokines, Th2 chemokines, and serum IgE levels. Mice were sacrificed at day 61. Serum was obtained by cardiac puncture, BAL was performed to obtain BAL fluid, and single lung lobes were harvested and enzymatically digested. (a) BAL fluid and (b) lung digest homogenate cytokines IL-4, IL-5, IL-13, and IFN-γ were measured by ELISA according to manufacturer’s protocols. Th2 chemokines CCL11/eotaxin, CCL17/TARC, and CCL22/MDC in BAL fluid (c) and lung digest homogenates (d) were measured by ELISA according to the manufacturer’s protocols. Total IgE in serum samples (e) was quantified by ELISA using paired antibodies according to the manufacturer’s protocol. Levels of Fel d 1–specific IgE (f) were measured in serum by coating plates with recombinant Fel d 1 (5 µg/ml), incubating with serum samples, and detecting bound IgE with chromogen-labeled anti-IgE. Data are expressed as mean ± SEM. n = 5 mice/group, and are representative of three independent experiments. *, P < 0.05 and **, P < 0.01 for sensitized/HA(306–318) compared with sensitized/DR1Feld1-treated mice.
Neutralization of IL-10 activity during immunotherapy with Feld1(29–45) reverses peptide-induced tolerance
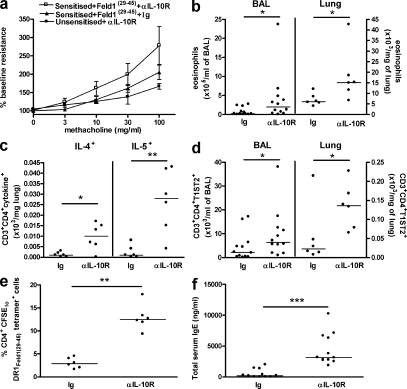
Treatment with Feld1(29–45) reduced cat allergen–stimulated Th2 responses systemically and in the lung. Treatment was associated with elevated IL-10+ CD4+ T cells in lung tissue. To determine whether peptide immunotherapy in this model was IL-10 dependent, we neutralized IL-10 activity in vivo beginning at the time of peptide-treatment and until cat allergen rechallenge. In vivo administration of anti-IL-10 receptor (R) antibody effectively reversed peptide-induced tolerance. Anti–IL-10R treatment increased airway resistance, abrogating the therapeutic effect of Feld1(29–45) on lung function, but was without effect on airway resistance in control mice, which remained at basal levels (Fig. 10 a). In addition to reversal of peptide therapy effects on lung function, anti–IL-10R treatment was associated with exacerbation of Th2 lung inflammation. Significant increases in BAL and lung eosinophils (Fig. 10 b), together with increased numbers of T cells expressing the Th2 cytokines IL-4 or IL-5 (Fig. 10 c) and the Th2 cell marker T1ST2 (Fig. 10 d), were observed. Feld1(29–45) therapy was shown to reduce the numbers of proliferating Fel d 1–specific T cells, including CD4+DR1Feld1(29–45)tetramer+ cells (Fig. 4). Anti–IL-10R treatment reversed this effect and restored Fel d 1–stimulated CD4+DR1Feld1(29–45)tetramer+ cell numbers (Fig. 10 e). Finally, neutralization of IL-10R in peptide-treated mice also resulted in an elevated systemic Th2 response, as shown by significantly higher total IgE levels in anti–IL-10R–treated mice (Fig. 10 f). Collectively, these data indicate that the mechanism of low-dose peptide immunotherapy is IL-10–dependent in a wide range of physiological, cellular, and humoral parameters.
Figure 10.
Feld(29–45) peptide immunotherapy is dependent on IL-10. Mice were sensitized, allergen-challenged, and treated with Feld1(29–45) peptide as described in Fig. 2. Mice were then treated with neutralizing antibody to the IL-10 receptor (αIL-10R) or an isotype control antibody (Ig) weekly from day 25. Mice were rechallenged with cat allergen extract (days 59 and 60), lung function was analyzed, and the mice were sacrificed (day 61) for tissue harvest. Unsensitized control mice received PBS in alum and were challenged in the same manner as sensitized animals. Airway resistance (a) was measured in anesthetized and tracheotomized mice. BALF and lung tissue cells were isolated as described in the Materials and methods. Eosinophil numbers in BAL and lung tissue (b) were determined after differential counting of Wright-Giemsa–stained cytospins. CD3+CD4+ T cells in lung digests expressing IL-4 and IL-5 (c) were evaluated by flow cytometry of light-scatter lymphocyte-gated cells. Numbers of Th2 cells (CD3+CD4+T1ST2+; d) in the BAL and lung tissue were determined by flow cytometry. Numbers of proliferating CD4+ CFSElo DR1Feld1(29–45) tetramer+ cells/mouse (e) were evaluated as described in Fig. 4. Total serum IgE (f) was quantified as described in the Materials and methods. Data are expressed as mean ± SEM or as individual data points with medians indicated. n = 5–12 mice/group. *, P < 0.05; **, P < 0.01; and ***, P < 0.001 for Ig-treated compared with αIL-10R–treated Feld1(29–45)-tolerized mice. Two independent experiments were performed, with some outcomes performed in one or other of the experiments. Data in a, c, and e represent a single experiment; those in panels b, d, and f represent two experiments.
DISCUSSION
Peptide immunotherapy with CD4 T cell epitopes has been used extensively in mouse models to prevent and ameliorate antigen-specific inflammatory responses and is the subject of current clinical trials in allergy and autoimmunity (Larche and Wraith, 2005). Improved understanding of mechanisms is likely to significantly improve efficacy. In our clinical studies, Fel d 1 peptides were delivered i.d., at low dose, to redirect allergen-specific responses, decrease symptoms, and reduce inflammation. Systemic doses of peptide as low as 5 µg reduced skin allergen sensitivity and proinflammatory PBMC responses to allergen (Oldfield et al., 2001; Oldfield et al., 2002; Alexander et al., 2005a; Alexander et al., 2005b). Reduced proliferative responses to allergen were associated with increased IL-10 production from PBMC and induction of functional CD4+ regulatory T cells (Verhoef et al., 2005).
To define the immunological mechanisms underlying the effects of peptide immunotherapy, we performed a detailed analysis of peptide-specific responses in allergic asthmatic subjects treated with a therapeutic peptide vaccine or with placebo. Peptide treatment reduced proliferative and cytokine responses to both treatment and nontreatment peptides, which is indicative of linked epitope suppression, a process through which cells rendered tolerant to one epitope suppress the function of cells specific for other epitopes within the same molecule (Holan and Mitchison, 1983). To determine the functional effects of peptide immunotherapy on T cells from the lung parenchyma and airways, which are more relevant to clinical asthma but cannot be studied in human volunteers, we generated a novel mouse model of peptide immunotherapy. The model was specifically designed to closely mimic our human experience. Mice were treated with a single Fel d 1 peptide, which forms part of the therapeutic clinical vaccine used in human studies. Presentation of the peptide to T cells was restricted by the only MHC class II molecule expressed by the mice, the product of a human HLA-DR1 transgene. Using this novel model we were able for the first time to directly track T cell responses to the treatment peptide using HLA-DR1 tetramer reagents. Tetramer analysis revealed reduced antigen-specific proliferative responses of both DR1Feld1(29–45)tetramer+ cells and tetramerneg cells supporting the observation of linked epitope suppression in the clinical study. Given the marked reduction in lung inflammation and decreases in Th2 responses after peptide treatment in the mouse model, it is clear that the peptide-specific T cell population was capable of down-regulating an established inflammatory response driven by multiple T cell epitopes. Indeed, in this model, targeting of a relatively rare population of peptide-specific T cells was associated with the production of IL-10 by a much larger proportion of the T cell pool. Similar induction of IL-10–producing “bystander” T cells was recently reported in a related model after transfer of antigen-specific CD4+CD25+ regulatory T cells (Kearley et al., 2005). “Infectious” expansion of IL-10–producing T cell populations has been described previously in other disease scenarios (Qin et al., 1993).
Peptide therapy markedly increased BAL levels of IL-10 and numbers of IL-10+ T cells in lung tissue, the latter increasing threefold. Lung digest T cells cultured for 7 d with recombinant allergen showed lower levels of proliferation after Fel d 1 peptide treatment compared with control peptide. This may have been caused by suppressive effects of increased numbers of IL-10+ T cells, and/or caused by a reduction in the numbers of allergen-specific T cells through clonal deletion.
PBMCs from allergen peptide-treated subjects, and heat shock protein peptide-treated subjects with type I diabetes and rheumatoid arthritis, demonstrated enhanced levels of antigen-stimulated IL-10 production in vitro (Akdis and Blaser, 1999; Raz et al., 2001; Oldfield et al., 2002; Prakken et al., 2004). IL-10+ cells have been found to increase in number in blood and tissues taken in clinical studies of grass pollen and insect venom immunotherapy (Bellinghausen et al., 1997; Nasser et al., 2001; Nouri-Aria et al., 2004) and a higher frequency of IL-10–secreting T cells is found in peripheral blood of nonatopic versus atopic individuals (Akdis et al., 2004). Adoptive transfer of IL-10–secreting cells has been shown to ameliorate allergic airway inflammation. We have shown that transfer of CD4+CD25+ regulatory T cells suppresses allergic lung inflammation by an IL-10–dependent mechanism (Kearley et al., 2005). Furthermore, IL-10–transduced dendritic cells down-regulate allergic airway inflammation in mice by induction of IL-10–expressing T cells (Henry et al., 2008), and OVA-specific T cells engineered to express IL-10 also inhibit Th2-induced AHR and inflammation (Oh et al., 2002). In this study, neutralization of IL-10 activity via blockade of IL-10R reversed peptide-induced tolerance, as shown by lung function analysis, elevation of lung and systemic Th2 responses, and reversal of peptide treatment effects on proliferation of Feld1(29–45)-specific T cells. These results specifically demonstrate the IL-10 dependence of peptide therapy in this model, which does not rely on the transfer of manipulated cells.
We did not observe an increase in the number of DR1Feld1(29–45)tetramer+ nondividing cells which would have been expected had they been anergized. Thus, we further conclude that peptide therapy does not result in the induction of anergy in the target T cell population, a possibility we have been thus far unable to exclude in clinical studies.
Resolution of allergic airway disease in our mouse model was achieved with administration of only a single peptide. Chai et al. (2004) prevented graft rejection by prophylactic administration of 9 µg (3 administrations) of peptide intranasally (i.n.). Apostolou and Von Boehmer (2004) induced de novo conversion of naive T cells to CD4+CD25+ antigen-specific regulatory cells through chronic exposure (10 µg/d for 14 d) to peptide. By directly targeting dendritic cells with an influenza peptide integrated into an antibody to DEC-205, Kretschmer et al. generated antigen-specific regulatory T cells with the equivalent of 500 pg of epitope (Kretschmer et al., 2005). Here we report the induction of antigen-specific tolerance and resolution of inflammation after the single administration of 1 µg of peptide without adjuvant or cell-targeting strategy. Thus, ultra low-dose delivery of CD4 T cell epitopes can induce T cells with regulatory function that are capable of reversing existing pathology.
Using lavage and whole lung tissue, we demonstrated a marked reduction in airway and tissue eosinophilia and reduced in situ Th2 inflammation. BAL and lung tissue IL-4, IL-5, and IL-13 cytokine production was reduced. Local IFN-γ production was not elevated after Feld1(29–45) treatment, suggesting that peptide therapy did not result in deviation from a Th2 to a Th1 response. Levels of the Th2-associated chemokines CCL11, CCL17, and CCL22 were also significantly decreased. Possibly as a direct result of this, we observed fewer CD4+ T cells expressing IL-4 and IL-5 and reduced recruitment of Th2 cells to lung tissue and BAL after peptide treatment.
TGF-β has been implicated in T cell regulation of immune responses through conversion of naive CD25− T cells to regulatory CD25+ cells through induction of Foxp3 expression and reduction of T cell proliferation (Chen et al., 2003; Apostolou and Von Boehmer, 2004). However, in this study, we found no change in levels of active TGF-β1 in BAL or lung tissue homogenates after peptide challenge. This implies that TGF-β does not have a significant role in suppression of pulmonary pathophysiology in this model. We have described similar findings in another lung model (Kearley et al., 2005). Because Foxp3 expression is thought to be a marker for CD4+CD25+ regulatory T cells (Hori et al., 2003) we measured intracellular Foxp3 expression in CD4+ T cells isolated from BAL, lung tissue, and peribronchial lymph nodes. Peptide treatment did not result in increased numbers of Foxp3+ cells in any of these tissues. These data may discount a role for CD4+CD25+ regulatory T cells in peptide-directed resolution of pathophysiology, in agreement with our published clinical findings (Smith et al., 2004), but importantly they highlight a role for IL-10–secreting regulatory cells.
In summary, our data indicate that peptide immunotherapy ameliorates allergic inflammation in a mouse model via an IL-10–dependent mechanism and substantially reduces clinical surrogate markers of allergy in human cat allergic asthmatics through a process involving linked epitope suppression associated with induction of IL-10. No evidence for the induction of clonal T cell anergy was obtained. Treatment of mice was associated with reduced eosinophilia and mucus production, improved lung function, reduced Th2 cytokine and chemokine levels, lower total and specific IgE, and reduced numbers of Th2 cells infiltrating the lung tissue and BAL. Moreover, we demonstrate, for the first time, the direct effect of peptide administration on functional responses of lung parenchymal T cells specific for the treatment peptide using MHC class II tetramers. The percentage of IL-10–producing T cells was markedly enhanced in the lung after peptide therapy and was substantially greater than the percentage of cells specific for the treatment peptide, an observation that is reminiscent of infectious tolerance. Finally, tolerance induction in the mouse model appeared to be independent of TGF-β and Foxp3 expression. These studies further our understanding of mechanisms of peptide-induced tolerance in allergic asthma. These results will inform the design and evaluation of peptide interventions to ameliorate chronic allergic and autoimmune diseases.
MATERIALS AND METHODS
Randomized, double-blind, placebo-controlled trial of peptide immunotherapy in allergic asthma
The protocol and clinical outcomes of this trial have been reported previously (Oldfield et al., 2002). The study was approved by the Royal Brompton and Harefield National Health Service Trust Ethics Committee. All subjects gave written informed consent. For this study, peripheral blood samples were obtained before and after therapy. PBMCs were isolated and cryopreserved. PBMCs were cultured in 96-well plates for 6 d at a concentration of 106 cells/ml in the presence of 100 µg/ml of each peptide, with medium alone and cat dander allergen extract acting as controls. 12 replicate wells were cultured for each condition. Proliferative responses were evaluated by [3H]thymidine incorporation. For cytokine analysis, cells were cultured with medium, individual peptides or allergen at a concentration of 5 × 106 cells/ml. Supernatants were harvested at 6 d and frozen. Cytokine production was measured by ELISA (Pelikine).
Transgenic mice
HLA-DR1 (DRA*0101/DRB1*0101) transgenic mice on a FVB/N background were described previously (Altmann et al., 1995). DR1 expression in these animals mirrors that of mouse MHC II, and antigen presentation to mouse T cells occurs. DR1 transgenic mice were crossed for eight generations with C57BL/6 Aβo mice to yield animals on a C57BL/6 background with no endogenous mouse MHC II (DR1-tg/Aβo). Polymerase chain reaction was routinely used to confirm the presence of HLA-DRA1*0101 and HLA-DRB1*0101 and the absence of endogenous H2-Aβ in mice. 8–12-wk-old age, sex, and weight-matched animals were used in all experiments. All animal procedures were approved by the UK Home Office. UK Home Office regulations for animal welfare, based on the Animals (Scientific Procedures) Act 1986, were strictly observed.
Allergens and peptides
Recombinant Fel d 1 was prepared as described previously (Gronlund et al., 2003). Cat allergen extract (predominantly Fel d 1, but also containing other cat proteins) was obtained from Laboratorios Leti. Peptide sequences for the clinical study were: Fel d 1 chain 1 (1–17) EICPAVKRDVDLFLTGT, (12–28) FLTGTPDEYVEQVAQY, (23–38) EQVAQYKALPVVLENA, (29–45) KALPVVLENARILKNCV, (39–55) RILKNCVDAKMTEEDKE, (48–63) KMTEEDKENALSLLDK, (54–69) KENALSVLDKIYTSPL, Fel d 1 chain 2 (29–44) LTKVNATEPERTAMKK, (40–55) TAMKKIQDCYVENGLI, (56–71) SRVLDGLVMTTISSSK, (67–82) ISSSKDCMGEAVQNTV, and (77–92) AVQNTVEDLKLNTLGR. The treatment peptide for the mouse model Feld1(29–45) (residues 29–45 of Fel d 1 chain 1; KALPVVLENARILKNCV) was previously shown to be HLA-DR1 restricted (Haselden et al., 1999). Peptide HA(306–318) (residues 306–318 of influenza virus hemagglutinin; PKYVKQNTLKLAT) was used as a DR1-binding peptide control (Stern et al., 1994). Peptides were synthesized by F-moc chemistry at the Peptide Synthesis Unit, Medical Research Council Clinical Sciences Centre, Imperial College, London, UK.
Allergen-induced airway inflammation and peptide-induced tolerance
Before therapeutic treatment with Feld1(29–45) allergic airway disease was induced in DR1-tg/Aβo mice by sensitization to Fel d 1, followed by inhaled challenge with cat allergen extract to localize inflammation to the lung tissues (experiment flow chart given in Fig. 2). Mice were sensitized by injection of 10 µg Fel d 1/mouse in 0.2 ml of alum (Au-Gel-S; Serva Electrophoresis) i.p. on days 0 and 14. Unsensitized (control) mice received the same volume of PBS in alum. Mice were challenged i.n. under halothane anesthesia, with 25 µl PBS containing 250 µg cat allergen extract or 25 µl PBS alone in the case of unsensitized controls on days 22, 23, and 24. Some mice were sacrificed on day 25 to confirm the presence of eosinophilic lung inflammation. To induce tolerance in mice with established airways disease, a single i.d. injection of 1 µg Feld1(29–45) (or peptide control, HA(306–318)) was administered on day 25. In select experiments, mice were treated weekly with 250 µg of anti–IL-10R antibody (1B1.3), or isotype control antibody on days 32, 39, 46, 53, and 58. All mice were challenged again i.n. with cat allergen extract on day 59 and 60. Immediately before the second of these challenges, AHR was assessed. 24 h after the last challenge, airway resistance and dynamic compliance were analyzed and mice were killed by exsanguination under terminal anesthesia. Blood, BAL, lung tissue, and draining lymph nodes were taken for analysis.
Measurement of airway function
AHR was measured at day 60 by recording respiratory pressure curves by whole-body plethysmography (Buxco Technologies) in response to inhaled methacholine (Sigma-Aldrich) at concentrations of 3–100 mg/ml for 1 min, as described previously (Hamelmann et al., 1997). In select experiments, AHR was also measured at day 25, 24 h after the i.n. challenge on day 24. Penh data were confirmed by direct measurement of lung resistance and dynamic compliance in anesthetized and tracheotomized mice in response to inhaled methacholine at concentrations of 3–100 mg/ml using a Buxco Technologies system in a modified version of previously described methods (Martin et al., 1988).
Cell recovery
Airway lumen.
BAL was performed as previously described (Gonzalo et al., 1996). Airways were lavaged three times with 0.4 ml PBS via a tracheal cannula. BAL fluid was centrifuged (700 g, 5 min, 4°C), and cells were transferred to slides by cytocentrifugation (5 × 104 cells/slide). Differential cell counts were performed on Wright-Giemsa–stained (Shandon) cytospins. Differential cell counts were determined by counting 8 high power fields (40× magnification; total area 0.5 mm2 per area randomly selected), and dividing this number by the total number of cells per high power field. To obtain absolute numbers of each leukocyte subtype, percentages were multiplied by the total number of cells obtained in the lavage fluid. All differential counts were performed blind and in a randomized order at the end of the study by the same observer.
Lung parenchyma.
To disaggregate cells from the lung tissue, one lobe (∼100 mg) was incubated (37°C) for 1 h in digest reagent: 30 mg/ml collagenase (type D; Boehringer Mannheim), 5 mg/ml DNase (type 1; Boehringer Mannheim), 100 U/ml penicillin, and 100 µg/ml streptomycin (Invitrogen) in RPMI 1640(Sigma-Aldrich)/10% FCS. The recovered cells were filtered through a 70-μm nylon sieve (Falcon; Marathon Laboratory Supplies), washed twice, resuspended in RPMI 1640/10% FCS, and counted in a hemocytometer. Cytocentrifuge preparations were made and Wright-Giemsa stained, and differential counts were performed as for BAL.
Peribronchial lymph nodes.
Draining lymph nodes were dissected out, teased apart, and passed through a 70-μm nylon sieve to generate single-cell suspensions. Cells were resuspended in complete culture medium: RPMI 1640/10% FCS, supplemented with 100 U/ml penicillin, 100 µg/ml streptomycin, and 2 mMl-glutamine (Invitrogen).
Flow cytometry
Surface marker staining.
Suspensions of cells from lung tissue, lymph nodes, and BAL were stained with fluorescently labeled antibodies in PBS containing 1% FCS and 0.1% sodium azide. To reduce nonspecific binding, cells were incubated with rabbit serum (Sigma-Aldrich) for 15 min before staining. For cell surface staining, anti–mouse CD4-APC (BD) and anti–mouse T1ST2-FITC (Morwell Diagnostics) were used.
Foxp3 expression.
Intracellular Foxp3 expression in CD4+ cells was detected using an anti–mouse Foxp3 antibody kit (eBioscience) according to the manufacturer’s instructions.
Intracellular cytokines.
Cells from lung tissue or lymph nodes were first restimulated with 10 µg/ml recombinant Fel d 1 in vitro at 5 × 105 cells/well in complete RPMI 1640 in round-bottom 96-well plates at 37°C for 6 d to preferentially expand cat allergen–specific T cells. Cells were then treated with PMA/ionomycin (10 ng/ml and 1 µg/ml; Merck Biosciences) in the presence of brefeldin A (Sigma-Aldrich) for 4 h. Cells were stained with anti–CD4-APC fixed and permeabilized in 0.5% saponin (Sigma-Aldrich) before intracellular staining with phycoerythrin (PE)-conjugated anti-cytokine antibodies. Cells were stained with anti–mouse IL-4, IL-5, IL-10 and appropriate isotype controls (all from BD). PE-labeled anti–human TGF-β (IQ Products), which cross reacts with mouse TGF-β (Ostroukhova et al., 2004), was used to detect surface TGF-β and, after permeabilization, intracellular TGF-β.
CFSE and MHC II tetramer staining.
For the detection of antigen-specific cell proliferation, 2.5 × 106 cells of each sample (pooled/treatment group) were labeled with CFSE (Invitrogen), as previously described (Verhoef et al., 2005). CFSE-treated cells were cultured with 10 µg/ml recombinant Fel d 1, as described in the previosu paragrph, for 6 d before staining with MHC class II (HLA-DR1) tetramers specific for either the treatment peptide (DR1Feld1[29–45]tetramer; peptide KALPVVLENARILKNCV) or an irrelevant control peptide from HLA-A2 (DR1A2tetramer; peptide VGSDWRFLRGYHQYA). Biotinylated DR1 complexes were prepared as described previously (Cameron et al., 2002). Tetramers were formed by stepwise addition of fluorescently labeled streptavidin-PE (Biosource International). Cells (5 × 105) were mixed with DR1 tetramers for 4 h at 37°C, chilled for 5 min, stained with anti–mouse CD4-APC for 30 min, washed, and fixed. Flow cytometric analysis was performed using a FACSCalibur cytometer and CellQuest software (BD). A primary gate was set on lymphocytes, as defined by light scatter, and a secondary gate was set on the CD4+ population.
Cytokine analysis
Cytokines were analyzed in BAL samples and lung tissue homogenate supernatants. Lung tissue was homogenized at 100 mg/ml in HBSS (Invitrogen) in the presence of protease inhibitors (Complete mini protease inhibitor cocktail; Roche). Homogenates were centrifuged at 500 g for 10 min, and supernatants were collected. Paired antibodies for mouse IFN-γ, IL-4, TGF-β (BD), IL-5 (Endogen), eotaxin/CCL11, and TARC/CCL17 (R&D Systems) were used in standardized ELISAs according to the manufacturers’ protocols. CCL22 levels were measured using a sandwich ELISA generated by coating ELISA plates with anti–mouse MDC (a gift from ICOS Corp.) and detecting bound antibody with biotinylated anti–mouse MDC (R&D Systems) against a standard curve generated using recombinant MDC (R&D Systems) as previously described (McMillan et al., 2004). Kits to measure IL-10 and IL-13 (eBioscience and R&D Systems) were used according to the manufacturers’ protocols.
IgE measurements
Total IgE in serum samples was quantified by ELISA using paired antibodies according to the manufacturer’s protocol (BD). Levels of Fel d 1–specific IgE were measured in serum by coating plates with recombinant Fel d 1 (5 µg/ml), incubating with serum samples, and detecting bound IgE as per the total IgE ELISA.
Statistical analysis
For peripheral blood studies, data were tested for normality with a modified Shapiro-Wilks test. Significance of changes within groups was tested with a Wilcoxon’s rank sum test. Data are expressed as mean ± SEM for bar graphs and medians for scatter plots. Statistical significance was accepted when P ≤ 0.05 using the Mann-Whitney U test.
Acknowledgments
We thank Liying Lu for preparation of DR1-peptide complexes, Lorraine Lawrence for histology, and Enrique Fernandez-Caldas for the gift of cat allergen extract.
This study was supported by the Medical Research Council (MRC G0100509), National Institutes of Health (R01-AI48833), the Canada Research Chairs program and the Canadian Foundation for Innovation, The Wellcome Trust (057704), the Swedish Research Council, and Asthma UK (05/045). M. Larché was an Asthma UK Senior Research Fellow (2001-2006), C.M. Lloyd is a Wellcome Trust Senior Fellow (057704), J.D. Campbell and K.F. Buckland were supported by the MRC, R.J. Boyton is an MRC Clinician Scientist, J. Kearley is the recipient of a BBSRC CASE studentship, and C.J. Reynolds is funded by the MRC and Asthma UK.
M. Larché and A.B. Kay are founders, shareholders, and consultants to Circassia Ltd. The authors have no other competing financial interests.
Footnotes
Abbreviations used: AHR, airway hyperreactivity; BAL, bronchoalveolar lavage; i.d., intradermal(ly); i.n., intranasal(ly); PE, phycoerythrin.
References
- Akdis C.A., Blaser K. 1999. IL-10-induced anergy in peripheral T cell and reactivation by microenvironmental cytokines: two key steps in specific immunotherapy.FASEB J. 13:603–609 [DOI] [PubMed] [Google Scholar]
- Akdis M., Verhagen J., Taylor A., Karamloo F., Karagiannidis C., Crameri R., Thunberg S., Deniz G., Valenta R., Fiebig H., et al. 2004. Immune responses in healthy and allergic individuals are characterized by a fine balance between allergen-specific T regulatory 1 and T helper 2 cells.J. Exp. Med. 199:1567–1575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander C., Tarzi M., Larche M., Kay A.B. 2005a. The effect of Fel d 1-derived T-cell peptides on upper and lower airway outcome measurements in cat-allergic subjects.Allergy. 60:1269–1274 [DOI] [PubMed] [Google Scholar]
- Alexander C., Ying S., Kay B., Larche M. 2005b. Fel d 1-derived T cell peptide therapy induces recruitment of CD4CD25; CD4 interferon-gamma T helper type 1 cells to sites of allergen-induced late-phase skin reactions in cat-allergic subjects.Clin. Exp. Allergy. 35:52–58 [DOI] [PubMed] [Google Scholar]
- Altmann D.M., Douek D.C., Frater A.J., Hetherington C.M., Inoko H., Elliott J.I. 1995. The T cell response of HLA-DR transgenic mice to human myelin basic protein and other antigens in the presence and absence of human CD4.J. Exp. Med. 181:867–875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apostolou I., Von Boehmer H. 2004. In vivo instruction of suppressor commitment in naive T cells.J. Exp. Med. 199:1401–1408 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellinghausen I., Metz G., Enk A.H., Christmann S., Knop J., Saloga J. 1997. Insect venom immunotherapy induces interleukin-10 production and a Th2-to-Th1 shift, and changes surface marker expression in venom-allergic subjects.Eur. J. Immunol. 27:1131–1139 [DOI] [PubMed] [Google Scholar]
- Cameron T.O., Norris P.J., Patel A., Moulon C., Rosenberg E.S., Mellins E.D., Wedderburn L.R., Stern L.J. 2002. Labeling antigen-specific CD4(+) T cells with class II MHC oligomers.J. Immunol. Methods. 268:51–69 [DOI] [PubMed] [Google Scholar]
- Chai J.G., James E., Dewchand H., Simpson E., Scott D. 2004. Transplantation tolerance induced by intranasal administration of HY peptides.Blood. 103:3951–3959 [DOI] [PubMed] [Google Scholar]
- Chen W., Jin W., Hardegen N., Lei K.J., Li L., Marinos N., McGrady G., Wahl S.M. 2003. Conversion of peripheral CD4+CD25− naive T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3.J. Exp. Med. 198:1875–1886 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coyle A.J., Lloyd C., Tian J., Nguyen T., Erikkson C., Wang L., Ottoson P., Persson P., Delaney T., Lehar S., et al. 1999. Crucial role of the interleukin 1 receptor family member T1/ST2 in T helper cell type 2-mediated lung mucosal immune responses.J. Exp. Med. 190:895–902 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daugherty B.L., Siciliano S.J., DeMartino J.A., Malkowitz L., Sirotina A., Springer M.S. 1996. Cloning, expression, and characterization of the human eosinophil eotaxin receptor.J. Exp. Med. 183:2349–2354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalo J.A., Lloyd C.M., Kremer L., Finger E., Martinez A., Siegelman M.H., Cybulsky M., Gutierrez-Ramos J.C. 1996. Eosinophil recruitment to the lung in a murine model of allergic inflammation. The role of T cells, chemokines, and adhesion receptors.J. Clin. Invest. 98:2332–2345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gronlund H., Bergman T., Sandstrom K., Alvelius G., Reininger R., Verdino P., Hauswirth A., Liderot K., Valent P., Spitzauer S., et al. 2003. Formation of disulfide bonds and homodimers of the major cat allergen Fel d 1 equivalent to the natural allergen by expression in Escherichia coli.J. Biol. Chem. 278:40144–40151 [DOI] [PubMed] [Google Scholar]
- Hamelmann E., Schwarze J., Takeda K., Oshiba A., Larsen G.L., Irvin C.G., Gelfand E.W. 1997. Noninvasive measurement of airway responsiveness in allergic mice using barometric plethysmography.Am. J. Respir. Crit Care Med. 156:766–775 [DOI] [PubMed] [Google Scholar]
- Haselden B.M., Kay A.B., Larche M. 1999. Immunoglobulin E-independent major histocompatibility complex–restricted T cell peptide epitope-induced late asthmatic reactions.J. Exp. Med. 189:1885–1894 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry E., Desmet C.J., Garze V., Fievez L., Bedoret D., Heirman C., Faisca P., Jaspar F.J., Gosset P., Jacquet A.P., et al. 2008. Dendritic cells genetically engineered to express IL-10 induce long-lasting antigen-specific tolerance in experimental asthma.J. Immunol. 181:7230–7242 [DOI] [PubMed] [Google Scholar]
- Holan V., Mitchison N.A. 1983. Haplotype-specific suppressor T cells mediating linked suppression of immune responses elicited by third-party H-2 alloantigens.Eur. J. Immunol. 13:652–657 [DOI] [PubMed] [Google Scholar]
- Hori S., Nomura T., Sakaguchi S. 2003. Control of regulatory T cell development by the transcription factor Foxp3.Science. 299(5609):1057–1061 [DOI] [PubMed] [Google Scholar]
- Kearley J., Barker J.E., Robinson D.S., Lloyd C.M. 2005. Resolution of airway inflammation and hyperreactivity after in vivo transfer of CD4+CD25+ regulatory T cells is interleukin 10 dependent.J. Exp. Med. 202:1539–1547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kretschmer K., Apostolou I., Hawiger D., Khazaie K., Nussenzweig M.C., von Boehmer H. 2005. Inducing and expanding regulatory T cell populations by foreign antigen.Nat. Immunol. 6:1219–1227 [DOI] [PubMed] [Google Scholar]
- Larche M., Wraith D.C. 2005. Peptide-based therapeutic vaccines for allergic and autoimmune diseases.Nat. Med. 11:S69–S76 [DOI] [PubMed] [Google Scholar]
- Lloyd C.M., Delaney T., Nguyen T., Tian J., Martinez A., Coyle A.J., Gutierrez-Ramos J.C. 2000. CC chemokine receptor (CCR)3/eotaxin is followed by CCR4/monocyte-derived chemokine in mediating pulmonary T helper lymphocyte type 2 recruitment after serial antigen challenge in vivo.J. Exp. Med. 191:265–274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin T.R., Gerard N.P., Galli S.J., Drazen J.M. 1988. Pulmonary responses to bronchoconstrictor agonists in the mouse.J. Appl. Physiol. 64:2318–2323 [DOI] [PubMed] [Google Scholar]
- McMillan S.J., Kearley J., Campbell J.D., Zhu X.W., Larbi K.Y., Shipley J.M., Senior R.M., Nourshargh S., Lloyd C.M. 2004. Matrix metalloproteinase-9 deficiency results in enhanced allergen-induced airway inflammation.J. Immunol. 172:2586–2594 [DOI] [PubMed] [Google Scholar]
- Nasser S.M., Ying S., Meng Q., Kay A.B., Ewan P.W. 2001. Interleukin-10 levels increase in cutaneous biopsies of patients undergoing wasp venom immunotherapy.Eur. J. Immunol. 31:3704–3713 [DOI] [PubMed] [Google Scholar]
- Nouri-Aria K.T., Wachholz P.A., Francis J.N., Jacobson M.R., Walker S.M., Wilcock L.K., Staple S.Q., Aalberse R.C., Till S.J., Durham S.R. 2004. Grass pollen immunotherapy induces mucosal and peripheral IL-10 responses and blocking IgG activity.J. Immunol. 172:3252–3259 [DOI] [PubMed] [Google Scholar]
- Oh J.W., Seroogy C.M., Meyer E.H., Akbari O., Berry G., Fathman C.G., DeKruyff R.H., Umetsu D.T. 2002. CD4 T-helper cells engineered to produce IL-10 prevent allergen-induced airway hyperreactivity and inflammation.J. Allergy Clin. Immunol. 110:460–468 [DOI] [PubMed] [Google Scholar]
- Oldfield W.L., Kay A.B., Larche M. 2001. Allergen-derived T cell peptide-induced late asthmatic reactions precede the induction of antigen-specific hyporesponsiveness in atopic allergic asthmatic subjects.J. Immunol. 167:1734–1739 [DOI] [PubMed] [Google Scholar]
- Oldfield W.L., Larche M., Kay A.B. 2002. Effect of T-cell peptides derived from Fel d 1 on allergic reactions and cytokine production in patients sensitive to cats: a randomised controlled trial.Lancet. 360:47–53 [DOI] [PubMed] [Google Scholar]
- Ostroukhova M., Seguin-Devaux C., Oriss T.B., Dixon-McCarthy B., Yang L., Ameredes B.T., Corcoran T.E., Ray A. 2004. Tolerance induced by inhaled antigen involves CD4(+) T cells expressing membrane-bound TGF-beta and FOXP3.J. Clin. Invest. 114:28–38 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prakken B.J., Samodal R., Le T.D., Giannoni F., Yung G.P., Scavulli J., Amox D., Roord S., de Kleer I., Bonnin D., Lanza P., Berry C., Massa M., Billetta R., Albani S. 2004. Epitope-specific immunotherapy induces immune deviation of proinflammatory T cells in rheumatoid arthritis.Proc. Natl. Acad. Sci. USA. 101:4228–4233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin S., Cobbold S.P., Pope H., Elliott J., Kioussis D., Davies J., Waldmann H. 1993. “Infectious” transplantation tolerance.Science. 259:974–977 [DOI] [PubMed] [Google Scholar]
- Raz I., Elias D., Avron A., Tamir M., Metzger M., Cohen I.R. 2001. Beta-cell function in new-onset type 1 diabetes and immunomodulation with a heat-shock protein peptide (DiaPep277): a randomised, double-blind, phase II trial.Lancet. 358:1749–1753 [DOI] [PubMed] [Google Scholar]
- Sallusto F., Mackay C.R., Lanzavecchia A. 1997. Selective expression of the eotaxin receptor CCR3 by human T helper 2 cells.Science. 277:2005–2007 [DOI] [PubMed] [Google Scholar]
- Schwartz R.H. 2003. T cell anergy.Annu. Rev. Immunol. 21:305–334 [DOI] [PubMed] [Google Scholar]
- Smith T.R., Alexander C., Kay A.B., Larche M., Robinson D.S. 2004. Cat allergen peptide immunotherapy reduces CD4 T cell responses to cat allergen but does not alter suppression by CD4 CD25 T cells: a double-blind placebo-controlled study.Allergy. 59:1097–1101 [DOI] [PubMed] [Google Scholar]
- Stern L.J., Brown J.H., Jardetzky T.S., Gorga J.C., Urban R.G., Strominger J.L., Wiley D.C. 1994. Crystal structure of the human class II MHC protein HLA-DR1 complexed with an influenza virus peptide.Nature. 368:215–221 [DOI] [PubMed] [Google Scholar]
- Sundstedt A., O’Neill E.J., Nicolson K.S., Wraith D.C. 2003. Role for IL-10 in suppression mediated by peptide-induced regulatory T cells in vivo.J. Immunol. 170:1240–1248 [DOI] [PubMed] [Google Scholar]
- Tarzi M., Klunker S., Texier C., Verhoef A., Stapel S.O., Akdis C.A., Maillere B., Kay A.B., Larche M. 2006. Induction of interleukin-10 and suppressor of cytokine signalling-3 gene expression following peptide immunotherapy.Clin. Exp. Allergy. 36:465–474 [DOI] [PubMed] [Google Scholar]
- Verhoef A., Alexander C., Kay A.B., Larche M. 2005. T cell epitope immunotherapy induces a CD4(+) T cell population with regulatory activity.PLoS Med. 2:e78. [DOI] [PMC free article] [PubMed] [Google Scholar]