Abstract
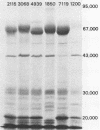
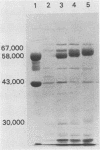
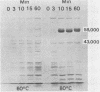
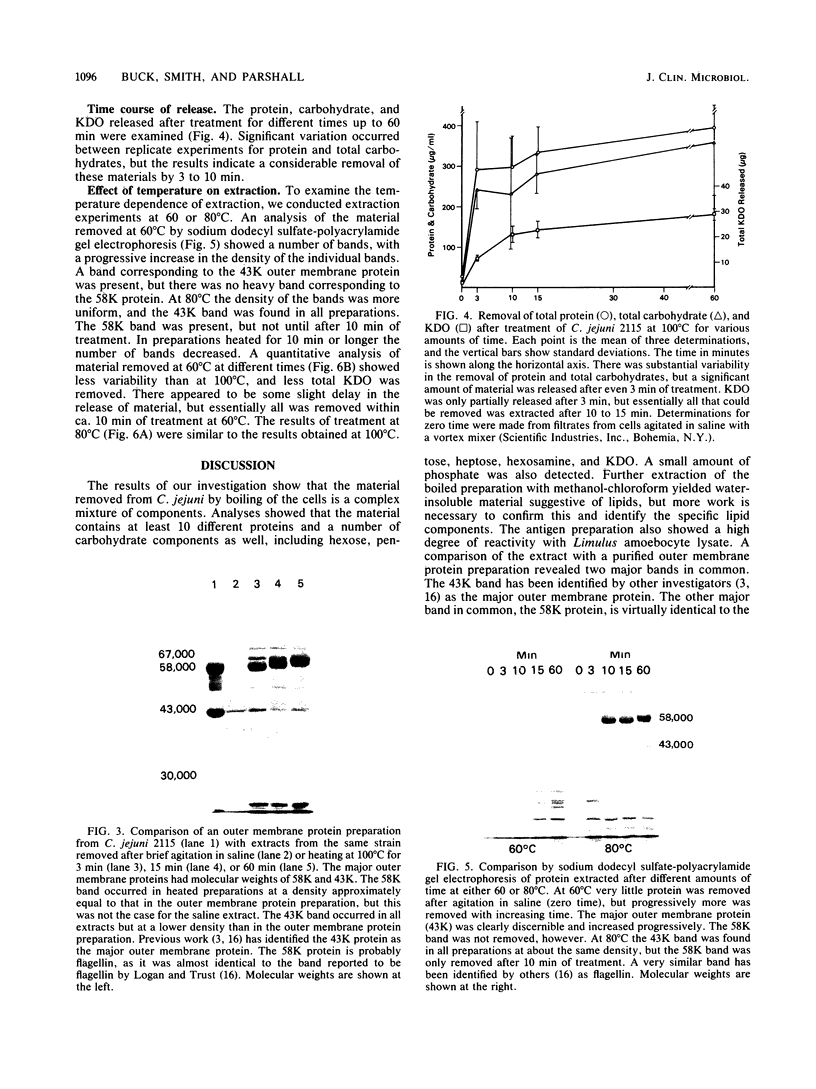
The antigenic material removed form Campylobacter jejuni by the boiling of whole cells in saline was examined biochemically. Analyses showed that the extracted material contained 3 micrograms of protein per ml per mg of wet cells and ca. 2.6 micrograms of carbohydrate per ml per mg of wet cells. Further extraction of the material with chloroform-methanol produced about 0.5 microgram of water-insoluble residue per ml per mg of wet cells, suggesting the presence of lipid as well. Additional analyses revealed the presence of hexose, pentose, heptose, hexosamine, and 2-keto-3-deoxyoctonic acid, and the extract was also positive by the Limulus amoebocyte lysate assay for lipopolysaccharide. An examination by sodium dodecyl sulfate-polyacrylamide gel electrophoresis showed that at least 10 different protein bands could be detected. One of the major bands corresponded to the major outer membrane protein, as determined by comparison with an outer membrane protein preparation by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Another major protein in the heated extract corresponded to a band previously shown to be flagellin. An analysis of the time course of the release of material showed that a significant amount was removed after 3 to 10 min at 100 degrees C, but the release of material seemed to be delayed at lower temperatures. These results show that the treatment of C. jejuni with heat produces a complex mixture of components, including cell wall lipopolysaccharide, the major outer membrane protein, and flagellin. It is likely that some cytoplasmic components are present as well. Blebs of outer membrane have been observed with this organism by electron microscopy. Our results confirm this and suggest that the heating of cells accelerates this blebbing process.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abbott J. D., Dale B., Eldridge J., Jones D. M., Sutcliffe E. M. Serotyping of Campylobacter jejuni/coli. J Clin Pathol. 1980 Aug;33(8):762–766. doi: 10.1136/jcp.33.8.762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berg R. L., Jutila J. W., Firehammer B. D. A revised classification of Vibrio fetus. Am J Vet Res. 1971 Jan;32(1):11–22. [PubMed] [Google Scholar]
- Blaser M. J., Hopkins J. A., Berka R. M., Vasil M. L., Wang W. L. Identification and characterization of Campylobacter jejuni outer membrane proteins. Infect Immun. 1983 Oct;42(1):276–284. doi: 10.1128/iai.42.1.276-284.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- DISCHE Z., SHETTLES L. B. A new spectrophotometric test for the detection of methylpentose. J Biol Chem. 1951 Oct;192(2):579–582. [PubMed] [Google Scholar]
- DRYER R. L., TAMMES A. R., ROUTH J. I. The determination of phosphorus and phosphatase with N-phenyl-p-phenylenediamine. J Biol Chem. 1957 Mar;225(1):177–183. [PubMed] [Google Scholar]
- Hébert G. A., Hollis D. G., Weaver R. E., Lambert M. A., Blaser M. J., Moss C. W. 30 years of campylobacters: biochemical characteristics and a biotyping proposal for Campylobacter jejuni. J Clin Microbiol. 1982 Jun;15(6):1065–1073. doi: 10.1128/jcm.15.6.1065-1073.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karkhanis Y. D., Zeltner J. Y., Jackson J. J., Carlo D. J. A new and improved microassay to determine 2-keto-3-deoxyoctonate in lipopolysaccharide of Gram-negative bacteria. Anal Biochem. 1978 Apr;85(2):595–601. doi: 10.1016/0003-2697(78)90260-9. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leive L., Shovlin V. K., Mergenhagen S. E. Physical, chemical, and immunological properties of lipopolysaccharide released from Escherichia coli by ethylenediaminetetraacetate. J Biol Chem. 1968 Dec 25;243(24):6384–6391. [PubMed] [Google Scholar]
- Lior H., Woodward D. L., Edgar J. A., Laroche L. J., Gill P. Serotyping of Campylobacter jejuni by slide agglutination based on heat-labile antigenic factors. J Clin Microbiol. 1982 May;15(5):761–768. doi: 10.1128/jcm.15.5.761-768.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logan S. M., Trust T. J. Molecular identification of surface protein antigens of Campylobacter jejuni. Infect Immun. 1983 Nov;42(2):675–682. doi: 10.1128/iai.42.2.675-682.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKelvy J. F., Lee Y. C. Microheterogeneity of the carbohydrate group of aspergillus oryzae alpha-amylase. Arch Biochem Biophys. 1969 Jun;132(1):99–110. doi: 10.1016/0003-9861(69)90341-5. [DOI] [PubMed] [Google Scholar]
- NOWOTNY A. M., THOMAS S., DURON O. S., NOWOTNY A. Relation of structure to function in bacterial O antigens. I. Isolation methods. J Bacteriol. 1963 Feb;85:418–426. doi: 10.1128/jb.85.2.418-426.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pead P. J. Electron microscopy of Campylobacter jejuni. J Med Microbiol. 1979 Aug;12(3):383–385. doi: 10.1099/00222615-12-3-383. [DOI] [PubMed] [Google Scholar]
- Penner J. L., Hennessy J. N. Passive hemagglutination technique for serotyping Campylobacter fetus subsp. jejuni on the basis of soluble heat-stable antigens. J Clin Microbiol. 1980 Dec;12(6):732–737. doi: 10.1128/jcm.12.6.732-737.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RISTIC M., WHITE F. H., DOTY R. B., HERZBERG M., SANDERS D. A. The characteristics of agglutinating antigens of Vibrio fetus variants. I. Effects of heat and formalin on serological activity. Am J Vet Res. 1957 Oct;18(69):764–770. [PubMed] [Google Scholar]
- Rogol M., Sechter I., Braunstein I., Gerichter C. B. Extended scheme for serotyping Campylobacter jejuni: results obtained in Israel from 1980 to 1981. J Clin Microbiol. 1983 Aug;18(2):283–286. doi: 10.1128/jcm.18.2.283-286.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STROMINGER J. L., PARK J. T., THOMPSON R. E. Composition of the cell wall of Staphylococcus aureus: its relation to the mechanism of action of penicillin. J Biol Chem. 1959 Dec;234:3263–3268. [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- Wright B. G., Rebers P. A. Procedure for determining heptose and hexose in lipopolysaccharides. Modification of the cysteine-sulfuric acid method. Anal Biochem. 1972 Oct;49(2):307–319. doi: 10.1016/0003-2697(72)90433-2. [DOI] [PubMed] [Google Scholar]