Abstract
The development of a biosensor system capable of continuous, real-time measurement of small-molecule analytes directly in complex, unprocessed aqueous samples has been a significant challenge, and successful implementation has been achieved for only a limited number of targets. Towards a general solution to this problem, we report here the Microfluidic Electrochemical Aptamer-based Sensor (MECAS) chip wherein we integrate target-specific DNA aptamers that fold, and thus generate an electrochemical signal, in response to the analyte with a microfluidic detection system. As a model, we demonstrate the continuous, real-time (~1 minute time resolution) detection of the small molecule drug cocaine at near physiological, low micromolar concentrations directly in undiluted, otherwise unmodified blood serum. We believe our approach of integrating folding-based electrochemical sensors with miniaturized detection systems may lay the ground work for the real-time, point-of-care detection of a wide variety of molecular targets.
The capability to perform in situ, continuous, real-time monitoring of specific small molecules in complex, unprocessed aqueous samples is important for a broad spectrum of applications ranging from medical diagnostics to environmental monitoring.1 However, successful implementation of such strategies has proven elusive, and thus far has been achieved for only a few, specialized targets (e.g., neurotransmitters, such as 5-hydroxytryptamine,2 hydrogen ions3 and glucose4). The challenge of real-time sensing arises from the fact that, to meet this demanding application, a sensor needs not only be sensitive, stable and selective enough to deploy directly in complex sample matrices, but it also needs to be reagentless, regenerable and able to respond rapidly relative to the timescale with which the target concentration fluctuates.
Previously-developed strategies for the real-time detection of small-molecule analytes have typically made use of sensors that measure changes in mass,5 index of refraction6 or charge7 that occur when the target molecules bind to the sensor surface. Such measurements, however, suffer from often severe false positives; for example, it has proven difficult to discriminate between changes in mass arising from the binding of authentic target molecules and those arising due to the non-specific adsorption of contaminants, which has significantly hindered the application of these adsorption-based sensors in complex samples, such as unprocessed clinical or environmental materials.5–7 In contrast, sensors based on binding-induced conformational changes in a bimolecular probe8–9 have proven more effective in rejecting such false positives. An example is the electrochemical, aptamer-based (E-AB) sensor platform, which operates via the target binding-induced folding of DNA and RNA aptamers and has been shown to work directly in blood serum,10–12 crude cellular extracts,13 soil extracts14 and foodstuffs.15 The high selectivity of E-AB sensors is attributed to two effects. First, in this architecture, the non-specific binding of interferants does not trigger a conformational change in the aptamer, due to lack of molecular recognition. Second, the target binding-induced conformational change is signaled electrochemically, and electroactive contaminants within the potential range of the employed redox tags are rare. These advantages, in combination with the stability of DNA probes and reagentless, regenerable characteristics of the platform, suggest that it is well suited for real-time operation.
In addition to requiring a rapidly equilibrating sensing element, the continuous, real-time detection of aqueous analytes also requires that we overcome the limitations imposed by diffusion and mass transport in bringing the target molecules to the sensor surface. Here, due to the fact that microfabrication allows precise patterning of multiple electrodes in miniaturized flow cells, microfluidics technology offers important advantages on mass transport and diffusion over conventional macro-scale devices. In this work, we report the development of the Microfluidic Electrochemical Aptamer-based Sensor (MECAS) chip, wherein we integrate an E-AB sensor within a miniaturized electrochemical cell, to achieve continuous, real-time monitoring of cocaine in undiluted, unmodified blood serum at physiologically-relevant concentrations and with physiologically-relevant time resolution.
EXPERIMENTAL SECTION
Materials
Tris-(2-carboxyethyl) phosphine hydrochloride (TCEP), cocaine, 6-mercaptohexanol, Saline-sodium citrate (SSC) buffer (20× concentrate, 0.3 M sodium citrate, pH 7.0, containing 3 M NaCl) and fetal calf serum (from formula-fed bovine calves, sterile-filtered, cell culture tested, iron-supplemented) were purchased from Sigma-Aldrich, Inc. (St. Louis, MO) and used as received without further purification. Our thiolated, methylene blue (MB)-labeled DNA aptamer probe was synthesized and purified by Biosearch Technologies, Inc. (Novato, CA). The sequence of the modified cocaine binding aptamer is: 5’-HS-(CH2)11-AGACAAGGAAAATCCTTCAATGAAGTGGGTCG-(CH2)7-MB-3’. As reported, the selectivity of the cocaine aptamer for cocaine over its metabolites (for example, benzoyl ecgonine and ecgonine methyl ester) is high.16
MECAS chip Fabrication
Four-inch-diameter Borofloat glass wafers with a thickness of 500 µm (Precision Glass & Optics, Santa Ana, CA) were used as the substrate for the MECAS chip. The chip incorporates a 750-nl detection chamber containing three gold working electrodes, a platinum reference electrode and a platinum counter electrode patterned using standard microfabrication processes.
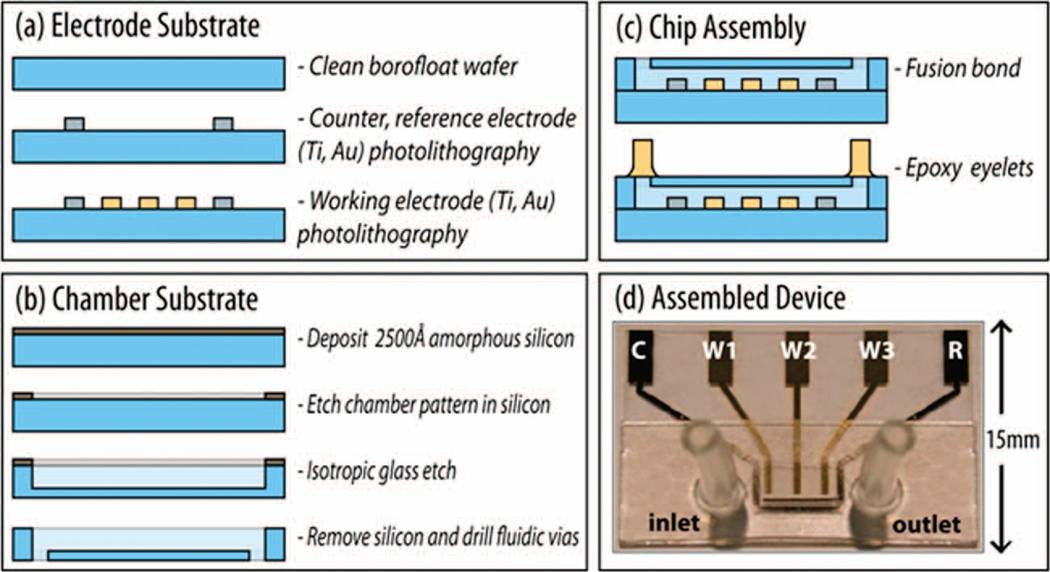
The MECAS fabrication architecture is modular in design (Fig. 1) consisting of three processes: (a) fabrication of the electrode substrate, (b) fabrication of the chamber substrate, and (c) assembly of the two substrates to yield a completed device (d). The electrode substrate (Fig. 1a) incorporated platinum counter and electrodes (size: 3200 µm2, 1300 µm2) which were patterned via optical photolithography. An electron beam evaporator was used to deposit a 20 Å Ti glass-adhesion layer and then 2500 Å of platinum. Gold working electrodes (three electrodes each at 800 µm2) were aligned and patterned with respect to the existing platinum electrodes and 2500 Å Au on 20 Å Ti layers were deposited using the same deposition method. We fabricated three working gold electrodes on the MECAS chip because this design allows the simultaneous collection of three sets of data at the same concentrations of cocaine in order to calculate the standard derivation of sensor response.
Figure 1.
The microfabrication process flow of the MECAS chip is modular, allowing independent processing of (a) the electrode and (b) the chamber substrates. The two substrates are then fusion bonded to form a monolithic device (c). The electrode substrate contains microfabricated platinum reference (R) and counter (C) electrodes in additions to three independent gold working (sensing) electrodes (W1,W2,W3). (d) A photo of the completed MECAS chip.
The chamber substrate (Fig. 1b) was processed in a second glass wafer. An amorphous silicon (α-Si) sacrificial etch mask was fist sputtered onto the glass wafer (2500 Å). In order to define the chamber area, vias were patterned and developed in the photoresist, exposing the underlying amorphous silicon layer on the surface of the glass wafer. The exposed amorphous silicon was completely etched down to the underlying glass surface using a reactive ion CF4 plasma etch. Borofloat glass was exposed only where the chamber is to be etched into the wafer. Next, the chamber was isotropically etched for 7 minutes in 45% hydrofluoric acid to ~35 µm depth. Any residual photoresist was washed in acetone, isopropyl alcohol and distilled, deionized water (DI water). The remaining α-Si was etched away in CF4 plasma again, leaving only glass. Finally, 0.75 mm inlet and outlet holes were drilled through the chamber wafer with a computerized mill for precision placement.
The MECAS chip was assembled as follows (Fig. 1c). The electrode wafer and the chamber wafer were diced to the desired dimensions to create glass chips which were then cleaned by sonication in acetone and isopropanol and rinsing in DI water. Following this step the chips were soaked face-up in a piranha cleaning solution for 10 minutes before being rinsed thoroughly in DI water and dried under nitrogen flow. The cleaned chips were then brought together, aligned, and sandwiched between two graphite blocks for glass fusion bonding in a furnace. A 2 kg mass was rested on the top graphite block, applying a constant pressure of approximately 3 kPa to the aligned chip. Under constant nitrogen flow, the furnace temperature was raised from room temperature to 590 °C at a rate of 10 °C/minute and held for 3 hours. The furnace was then allowed to cool passively back to room temperature. Upon removal from the furnace, the two chips were fused into a completed MECAS chip with a microfluidic channel defining fluid delivery to the patterned gold and platinum electrodes. Eyelets made of Tygon tubing were aligned and glued to the inlet and outlet holes of the MECAS chip to allow for fluid delivery into the chip (Fig. 1d).
Immobilization of the Aptamer probe
In order to clean the gold working electrodes prior to immobilization of the thiolated, MB-modified, cocaine-binding aptamer we performed a series of oxidation and reduction cycles in 0.5 M H2SO4, 0.01 M KCl/0.1 M H2SO4, and 0.05 M H2SO4 (Fig. 2a).17 The chamber was then immediately filled with 1.0 µM aptamer probe solution. To prepare this solution, a 0.2 mM aptamer in 10 mM Tris solution was first reduced with 5 mM TCEP for 1 hr to cleave disulfide bonds and then diluted to 1.0 µM with phosphate buffer (0.1 M sodium phosphate, 1.5 M NaCl, 1 mM MgCl2, pH 7.4) for 1 hr, all at room temperature. After probe immobilization, the chamber was rinsed with DI water and then filled with a freshly-prepared solution of 2 mM 6-mercaptohexanol for 4 hr to passivate the remaining bare electrode area and displace physically adsorbed DNA.18 The chamber was then rinsed again with DI water and filled with 1× SSC buffer (15 mM sodium citrate, 150 mM NaCl, pH 7.0) prior to measurements. This process yielded highly robust and stable sensors that can operate in blood serum for > 1 hour (Supporting information, Fig S1). Previous work19 suggests that that the stability of C11-based sensors is significantly improved relative to that of C6-based sensors, suggestion that the use of C11-chain aptamer would result in more stable SAMs and longer sensor shelf life in serum.
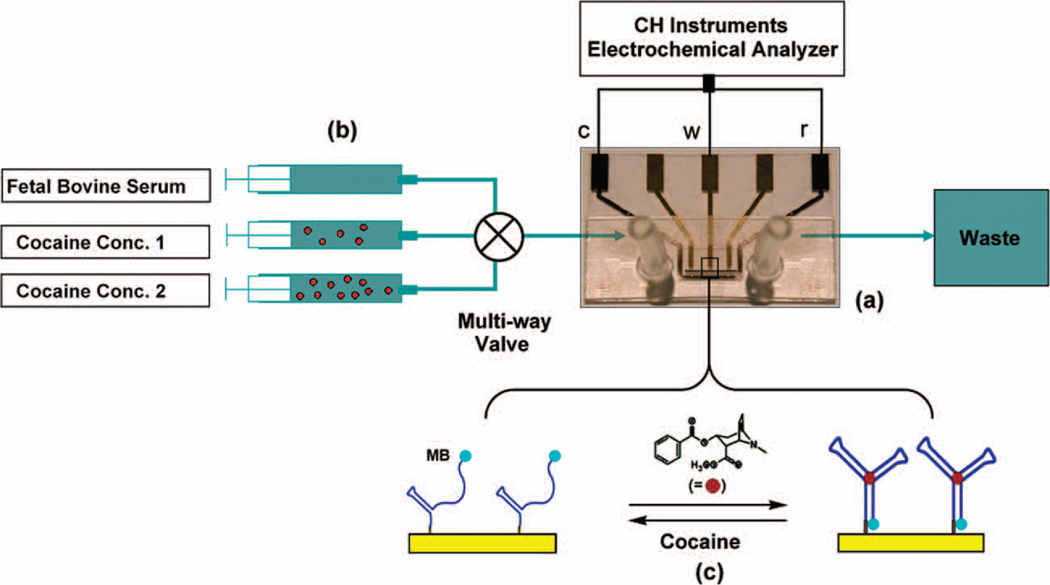
Figure 2.
Shown schematically is experimental setup for real time cocaine detection in continuously-flowing, undiluted blood serum. (a): The microfabricated MECAS chip contains a 750-nl detection chamber with integrated electrodes. (b): The syringe pumps are connected to a four-input, single-output multiplexed valve via Tygon tubing such that only one sample enters the chip at a time. (c): The detection mechanism of the folding-based E-AB sensor.
Characterization of the aptamer probe on the sensor chip
The immobilization of the aptamer probe was verified in 1× SSC buffer solution with a platinum counter electrode and a platinum reference electrode in the detection chamber via reduction scans on a CHI 730B potentiostat (CH Instruments, Austin, TX). Alternating current voltammograms (ACV) were acquired in the buffer using a 25 mV amplitude signal at 10 Hz over a potential window of −0.25 V to −0.65 V. The aptamer probe density was measured using ACV;20 optimal probe densities (which, as reported by this measure, are in the range of 20 ± 3 pmol·cm−2 -data not shown) were employed throughout this study. Some chip-to-chip variations in the baseline currents were found. We attribute this phenomenon to differences in the surface area of the electrodes caused by inaccuracies in device fabrication, deviations in the surface coverage of the modified aptamers on the electrodes, and minor changes in the resistances of the leads and connections. In particular, we found that the surface coverage of the aptamers was highly dependent on the surface roughness of the deposited gold film. Thus, relative sensor response (%), rather than the absolute values of the current, is employed in the calibration curve.
Real-time detection of cocaine in continuously flowing serum
For real-time cocaine detection, four samples of undiluted, otherwise unmodified fetal bovine serum were prepared in separate syringes, each doped with different concentrations of cocaine (0 µM, 10 µM, 50 µM, and 100 µM). The syringes were connected to a four-input, single-output multiplexed valve (Cole-Parmer, Vernon Hills, IL) via Tygon tubing such that only one sample can be selected to enter the MECAS chip at a time (Fig. 2 a,b). Each selected sample was pumped through the chip at a constant flow rate of 600 µl/hr. Trial studies using a fluorescent dye indicate a sample chamber can be filled in 4.5 seconds (166 nl/s) (data not shown). Using this setup, the concentration of cocaine in the serum within the chip was modulated from 0 µM to 10 µM to 50 µM to 0 µM to 100 µM to 0 µM in a continuous manner. At each concentration, ACV measurements were performed every 27 seconds scanning from −0.25 V to −0.65 V, and the peak reduction current is recorded using a computer controlled data acquisition system.
RESULTS AND DISCUSSION
The MECAS chip incorporates three gold working electrodes, a platinum reference electrode and a platinum counter electrode within a 750-nl detection chamber. The working electrodes are functionalized with a 32-base cocaine-binding aptamer12 that is partially unfolded in the absence of its target molecule (Fig. 2c, left). The aptamer probe is modified with the redoxable methylene blue (MB) moiety at its 3’ terminus, and is attached to the gold working electrode at its 5’ terminus via gold-thiol linkage.21 Upon target binding, the aptamer folds to its folded, cocaine-binding conformation, increasing the efficiency with which electrons are transferred from the MB tag to the electrode, thereby producing a readily measurable increase in faradaic current (Fig. 2c, right).
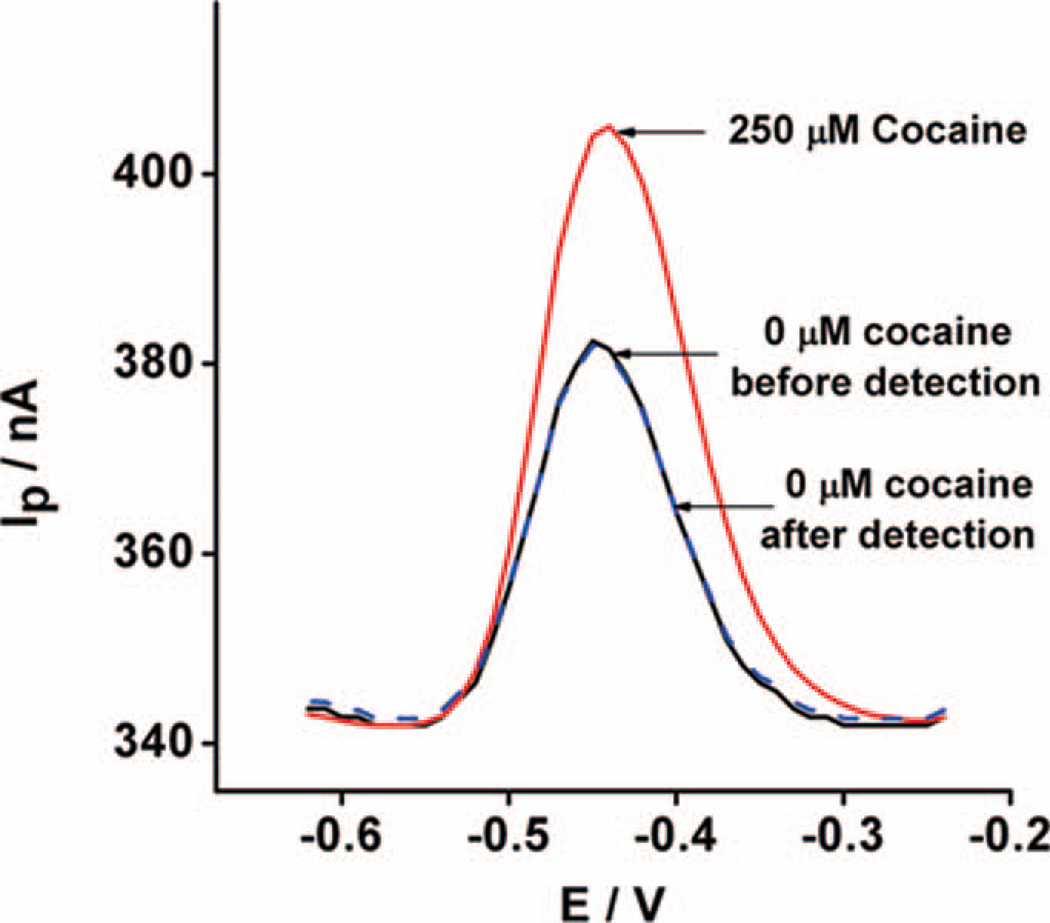
The MECAS chip responds sensitively and selectively to its target analyte. As an initial test of this we performed static measurements of the response of the aptamer-based sensor contained within. In doing so, we found that the chip rapidly responds to micromolar concentrations of cocaine in undiluted, unmodified blood serum: when challenged with 250 µM cocaine doped in serum the faradaic current increases by ~38% relative to the current observed in the absence of target (Fig. 3). Upon a brief wash with serum lacking cocaine, the sensor is readily regenerated, with the signal returning to 97% of its initial value (Fig. 3). Sensor regeneration allows for the MECAS sensor reuse at 10 µM cocaine for more than three times in undiluted blood serum with a mean recovery of > 96% of the original signal before significant degradation is observed (Supporting information, Fig. S1).
Figure 3.
The MECAS chip readily detects cocaine in undiluted, unmodified blood serum. Shown are alternating current (AC) voltammograms collected using the MECAS chip filled with 250 µM of cocaine in static, undiluted serum. Regeneration of 97% was obtained after challenging with a target-free serum.
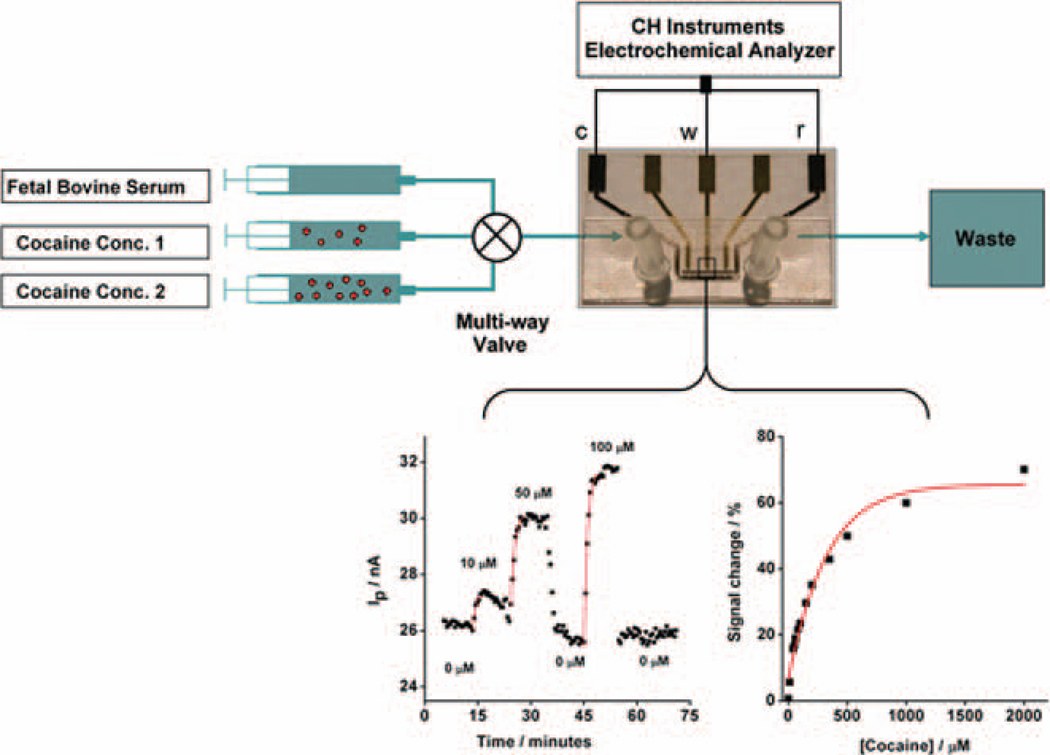
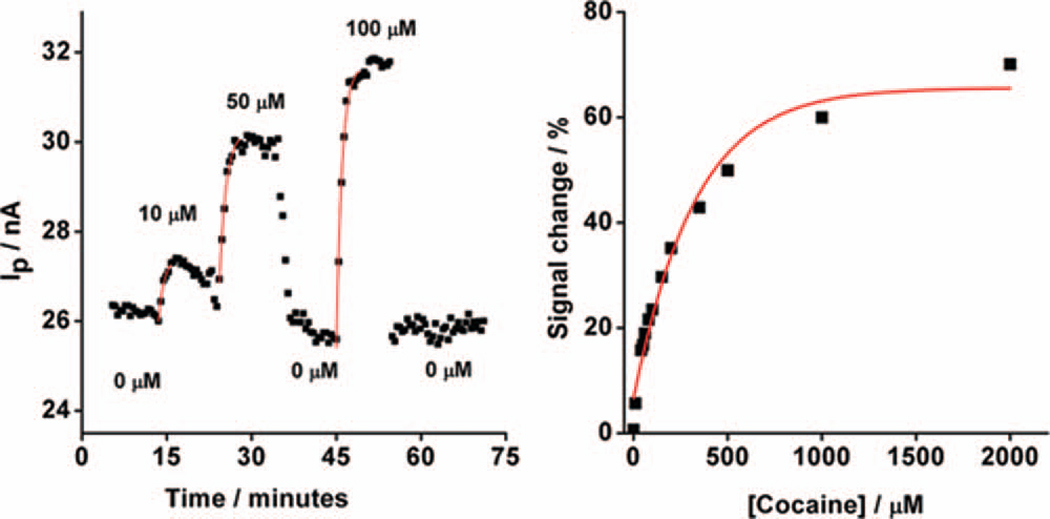
In order to measure the sensor’s response to continuously flowing serum containing cocaine at physiological concentrations,22 we employed a multi-port valve to deliver four different serum samples doped with varying levels of cocaine and measured the associated faradaic currents from the MECAS chip. We find that the chip responds selectively and rapidly to varying target cocaine concentrations with a signal effectively indistinguishable from that observed with static samples (Fig. 4, left). For example, a stream of undiluted, cocaine-free serum produced a relatively flat, low-current baseline, as would be expected for the sensor in the absence of target. When sample containing 10 µM cocaine is introduced into the chip the faradaic current increases by 6%. Further increases in the concentration of cocaine produce progressively larger increases in faradaic current, with a gain of 26% observed at the highest concentration used in this experiment (100 µM). The observed signal exhibits the hyperbolic “dose-response” curve expected for single-site binding, with an estimated dissociation constant of 100±9 µM (Fig. 4, right). This dissociation constant is effectively identical to that reported for the sensing aptamer in solution,16 suggesting that the binding affinity of the aptamer is not affected by its immobilization within the microfluidic device or by the collection of measurements in a continuously-flowing sample stream. Furthermore, we also performed titration experiments in which the direction of the cocaine concentration gradient was reversed (i.e. from high to low concentrations) (Supporting information, Fig. S2). The performance of the sensor did not depend on the direction of the gradient, and the observed sensor signals were almost identical to those reported in Figure 4.
Figure 4.
The MECAS device supports the real-time monitoring of cocaine at micromolar concentrations in continuously flowing, undiluted, unmodified blood serum. (Left) Real-time detection of cocaine in flowing serum, with the concentration modulated over the course of the experiment as denoted. (Right) A calibration curve derived for various concentrations of cocaine in undiluted serum (monitored in real-time in continuously-flowing serum) indicates that the cocaine dissociation constant measured in the chip is effectively indistinguishable from reported values in solution.21
The MECAS chip exhibits excellent baseline stability in continuously-flowing serum: the signaling current of the device rapidly returns to baseline values when the flow of cocaine-doped serum is replaced with cocaine-free serum (Fig. 4, left). This occurs even after 60 minutes of exposure to flowing serum, which is equivalent to approximately 800 flow-cell volumes, and occurs despite the numerous nucleases contained in serum that might be expected to degrade the sensing aptamer. The apparent stability of the sensing aptamer may be a result of its surface immobilization and MB modification;23 we have previously shown that a similar electrochemical DNA-based sensor can be stored in blood serum for more than a week at room temperature without exhibiting substantial degradation.19 It also appears that physical adsorption of background serum protein to the hydroxyl-coated SAM passivation layer does not affect target access to or folding of the aptamer probe during the 60 minutes of exposure. We note, however, that longer exposure times (3+ hours) under continuously flowing serum lead to some signal loss (data not shown), presumably due to the non-specific protein deposition from serum onto the sensor surface. Consistent with this, under these conditions we find that flushing the chamber with a protein denaturing solution (6M Guanadine HCl) recovers nearly the entire original peak signal (9.7% signal loss; data not shown). For continuous, in situ, real-time measurements in aqueous analyte, it is important to overcome the limitations imposed by mass transport of the target to the sensor surface. Here microfluidic technology provides an important advantage; because the sample stream is flowing at a rate that fills the entire chamber in just 4.5 seconds (166 nl/s), in comparison to macro-scale counterparts, the miniaturized detection chamber readily allows continuous delivery of the analyte to the sensor surface. Consistent with this feature, the sensor exhibits concentration-dependent rise-times of 1 to 2 minutes (and, because we are operating near the dissociation constant of the aptamer, similarly rapid fall times), which compares favorably with the minutes-to-hours physiological timescale of the uptake, action and metabolism of the drug.24 For example, with insufflation, the onset of cocaine’s action occurs 1–3 minutes after use, peaks 20–30 minutes later and lasts 45–90 minutes overall. Likewise the serum half-life of the drug is typically 60 to 90 minutes.25
In summary, we report here an analytical strategy that combines electrochemical, aptamer-based sensing with miniaturized, microfluidic sample handling technology to perform continuous, real-time detection of a small molecule analyte directly in a complex, flowing sample matrix. To apply our approach to other molecular targets, aptamers that undergo target binding-induced folding are necessary. Toward this end, a number of such aptamers has already been reported against a wide range of molecular targets,26 and innovative screening methods have been developed to directly isolate such structure-changing aptamer molecules.27–28 Importantly, general methodologies to incorporate the binding-induced folding feature into existing aptamers have also been described.29 Given this, it may be possible to extend our approach beyond small-molecule drugs to a range of other analytes for convenient, quantitative, real-time detection at the point-of-care.
Supplementary Material
Figures showing the specificity and stability of the MECAS device in undiluted blood serum and the real-time monitoring of cocaine concentration gradients from high to low on MECAS device in continuously flowing, undiluted, unmodified blood serum. SI file 2 has the complete reference 24. This material is available free of charge via the Internet at http://pubs.acs.org.
Acknowledgements
This work was supported by Office of Naval Research (N00014-08-1-0469), and National Institutes of Health (R21 EB008215) and by the Institute for Collaborative Biotechnologies through grant DAAD19-03-D-0004 from the U.S. Army Research Office. Microfabrication was carried out in the Nanofabrication Facility at UC Santa Barbara.
References
- 1.Roisin MP, Brassart JL, Charton G, Crepel V, Ben Ari Y. J. Neurosci. Methods. 1991;37:183–189. doi: 10.1016/0165-0270(91)90129-n. [DOI] [PubMed] [Google Scholar]
- 2.Schulze HG, Greek LS, Barbosa CJ, Blades MW, Gorzalka BB, Turner RFB. J. Neurosci. Methods. 1999;92:15–24. doi: 10.1016/s0165-0270(99)00081-3. [DOI] [PubMed] [Google Scholar]
- 3.Ordeig O, Mas R, Gonzalo J, Del Campo FJ, Muñoz FJ, de Haro C. Electroanalysis. 2005;17:1641–1648. [Google Scholar]
- 4.Kerr D. Int. J Clin. Pract. Suppl. 2001;123:43–46. [PubMed] [Google Scholar]
- 5.Janshoff A, Galla HJ, Steinem C. Angew. Chem. Int. Ed. 2000;39:4004–4032. doi: 10.1002/1521-3773(20001117)39:22<4004::aid-anie4004>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 6.Homola J, Yee SS, Gauglitz G. Sens. Actuators. B. 1999;54:3–15. [Google Scholar]
- 7.Zurn A, Rabolt B, Grafe M, Muller H. Fresenius J. Anal. Chem. 1994;349:666–669. [Google Scholar]
- 8.Ellington AD, Szostak JW. Nature. 1990;346:818–822. doi: 10.1038/346818a0. [DOI] [PubMed] [Google Scholar]
- 9.Tuerk C, Gold L. Science. 1990;249:505–510. doi: 10.1126/science.2200121. [DOI] [PubMed] [Google Scholar]
- 10.Xiao Y, Lubin AA, Heeger AJ, Plaxco KW. Angew. Chem. Int. Ed. 2005;44:5456–5459. doi: 10.1002/anie.200500989. [DOI] [PubMed] [Google Scholar]
- 11.Lai RY, Plaxco KW, Heeger AJ. Anal. Chem. 2006;79:229–233. doi: 10.1021/ac061592s. [DOI] [PubMed] [Google Scholar]
- 12.Baker BR, Lai RY, Wood MS, Doctor EH, Heeger AJ, Plaxco KW. J. Am. Chem. Soc. 2006;128:3138–3139. doi: 10.1021/ja056957p. [DOI] [PubMed] [Google Scholar]
- 13.Zuo X, Song S, Zhang J, Pan D, Wang L, Fan C. J. Am. Chem. Soc. 2007;129:1042–1043. doi: 10.1021/ja067024b. [DOI] [PubMed] [Google Scholar]
- 14.Xiao Y, Rowe AA, Plaxco KW. J. Am. Chem. Soc. 2007;129:262–263. doi: 10.1021/ja067278x. [DOI] [PubMed] [Google Scholar]
- 15.Lubin AA, Lai RY, Baker BR, Heeger AJ, Plaxco KW. Anal. Chem. 2006;78:5671–5677. doi: 10.1021/ac0601819. [DOI] [PubMed] [Google Scholar]
- 16.Stojanovic MN, de Prada P, Landry DW. J. Am. Chem. Soc. 2001;123:4928–4931. doi: 10.1021/ja0038171. [DOI] [PubMed] [Google Scholar]
- 17.Xiao Y, Lai RY, Plaxco KW. Nat. Protocols. 2007;2:2875–2880. doi: 10.1038/nprot.2007.413. [DOI] [PubMed] [Google Scholar]
- 18.Herne TM, Tarlov MJ. J. Am. Chem. Soc. 1997;119:8916–8920. [Google Scholar]
- 19.Lai RY, Seferos DS, Heeger AJ, Bazan G, Plaxco KW. Langmuir. 2006;22:10796–10800. doi: 10.1021/la0611817. [DOI] [PubMed] [Google Scholar]
- 20.O'Connor SD, Olsen GT, Creager SE. J. Electroanal. Chem. 1999;466:197–202. [Google Scholar]
- 21.Fan CH, Plaxco KW, Heeger AJ. Proc. Natl. Acad. Sci. U.S.A. 2003;100:9134–9137. doi: 10.1073/pnas.1633515100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Peat M, Davis AE. Drug Abuse Handbook. Boca Raton, Florida: CRC Press; 1998. [Google Scholar]
- 23.Truong-Le VL, Walsh SM, Schweibert E, Mao HQ, Guggino WB, August JT, Leong KW. Arch. Biochem. Biophys. 1999;361:47–56. doi: 10.1006/abbi.1998.0975. [DOI] [PubMed] [Google Scholar]
- 24.Benveniste H, et al. Journal of Nuclear Medicine. 2005;46:312–320. [PubMed] [Google Scholar]
- 25.Devane CL, Simpkins JW, Miller RL, Braun SB. Life Sci. 1989;45:1271–1276. doi: 10.1016/0024-3205(89)90129-x. [DOI] [PubMed] [Google Scholar]
- 26.Tombelli S, Minunni M, Mascini M. Biomol. Eng. 2007;24:191–200. doi: 10.1016/j.bioeng.2007.03.003. [DOI] [PubMed] [Google Scholar]
- 27.Hamaguchi N, Ellington AD, Stanton M. Anal. Biochem. 2001;294:126–131. doi: 10.1006/abio.2001.5169. [DOI] [PubMed] [Google Scholar]
- 28.Nutiu R, Li YF. Angew. Chem. Int. Ed. 2005;44:1061–1065. doi: 10.1002/anie.200461848. [DOI] [PubMed] [Google Scholar]
- 29.a) Hamaguchi N, Ellington AD, Stanton M. Anal. Biochem. 2001;294:126–131. doi: 10.1006/abio.2001.5169. [DOI] [PubMed] [Google Scholar]; b) Tang ZW, Mallikaratchy P, Yang RH, Kim Y, Zhu Z, Wang H, Tan WH. J. Am. Chem. Soc. 2008;130:11268–11269. doi: 10.1021/ja804119s. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figures showing the specificity and stability of the MECAS device in undiluted blood serum and the real-time monitoring of cocaine concentration gradients from high to low on MECAS device in continuously flowing, undiluted, unmodified blood serum. SI file 2 has the complete reference 24. This material is available free of charge via the Internet at http://pubs.acs.org.