Abstract
We compared the DNA sequences of the genus Metagonimus: M. yokogawai, M. takahashii, and M. miyatai. We obtained 28S D1 ribosomal DNA (rDNA) and mitochondrial cytochrome c oxidase subunit I (mtCOI) fragments from the adult worms by PCR, that were cloned and sequenced. Phylogenetic relationships inferred from the nucleotide sequences of the 28S D1 rDNA and mtCOI gene. M. takahashii and M. yokogawai are placed in the same clade supported by DNA sequence and phylogenic tree analysis in 28S D1 rDNA and mtCOI gene region. The above findings tell us that M. takahashii is closer to M. yokogawai than to M. miyatai genetically. This phylogenetic data also support the nomination of M. miyatai as a separate species.
Keywords: Metagonimus, Heterophyidae, polymerase chain reaction, sequence analysis, DNA, classification
INTRODUCTION
Metagonimiasis is an endemic intestinal trematode infection in Korea with 0.3% egg positives in the general population, i.e. 130,000 infected people (Ministry of Health and Social Welfare, and Korea Association of Health, 1997). Human infection with the three Metagonimus (Digenea: Heterophyidae) species is due to eating of raw fishes (intermediate hosts), and is regarded as a parasitic disease of importance to public health in the riverside areas of the southern and eastern coasts of Korea (Yu et al., 1994; Chai et al., 2000b; Lee et al., 2002). Three species of the genus Metagonimus are known in Korea (Chai et al., 1991): M. yokogawai (Katsurada, 1912), M. takahashii (Suzuki, 1930), and M. miyatai (Miyata, 1941, 1944; Saito et al., 1984, 1997). These three species are differentiated by their morphology (Chai et al., 1991, 1998, 2000a), as they have different fish species as hosts (Rim et al., 1996), different geographical distributions (Kim et al., 1987; Chai et al., 1993; Yu et al., 1994; Lee et al., 2002). In addition, they have different polymerase chain reaction-based restriction fragment length polymorphism (PCR-RFLP) patterns and random amplified polymorphic DNA (RAPD) patterns (Yu et al., 1997a, 1997b), and simple sequence repeat anchored polymerase chain reaction (SSR-PCR) patterns of genomic DNA of ribosomal RNA and mitochondrial cytochrome c oxidase 1 (mtCOI) gene (Yang et al., 2000). However, few studies have been conducted on the DNA sequences of Metagonimus spp. In the present study, we compared the 28S D1 rDNA and mtCOI gene sequences of the three species of Metagonimus distributed in the Republic of Korea, to determine their molecular phylogenies.
MATERIALS AND METHODS
Genomic DNA purification from worms
The adult specimens of M. takahashii, M. miyatai and M. yokogawai species were obtained 7 days post-infection from rats (Sprague-Dawley, 4 to 6-wk-old). Metacercariae of each species were collected from Korean freshwater fishes, Carassius auratus, Zacco platypus, and Plecoglossus altivelis, respectively (Saito et al., 1997; Yu et al., 1997b). Two adult trematodes of family Heterophyidae, Pygidiopsis summa and Stellantchasmus falcatus obtained from experimentally infected animals were included and S. falcatus was used as an outgroup to infer phylogeny. Adult worms were stored at -70℃ until used. DNA was extracted using the phenol/chloroform method and precipitated in ethanol, as reported by Sambrook and Russell (2001).
28S D1 rDNA and mtCOI PCR
PCR was conducted using a mixed solution of extracted DNA as a template (0.01 µg/µl), primer, and ExTaq enzyme (TaKaRa Ex Taq Kit, TAKARA Shuzo Co., LTD. Japan) in a GeneAmp PCR System 9600 (Perkin Elmer, USA). The PCR reaction cycle consisted of 40 cycles of denaturation at 95℃ for 20 seconds, annealing at 55℃ for 30 seconds and extension at 72℃ for 30 seconds followed by a final extension of 6 minutes. The forward primer (JB10, 5' GATTACCCGCTGAACTTAAGCATA 3') consisted of the conserved region from the 21st-45th base pairs of 28S rDNA sequence of mouse. The reverse primer (JB9, 5' GCTGCATTCACAAACACCCCGACTC 3') was obtained from the 278th-302nd base pairs of the same gene (Hassounna et al., 1984; Qu et al., 1988; Bowles and McManus, 1994). The PCR of mtCOI were performed using the above described method, but with different primers. The forward primer was JB3 (5' TTTTTTGGGCATCCTGAGGTTTAT 3') (2575), and the reverse primer was JB4.5 (5' TAAAGAAAGAACATAATGAAAATG 3') (3021). The numbers in brackets refer to the position of the 5' end of the primer from the nucleotide sequences of Fasciola hepatica (Bowles and McManus, 1994). The PCR condition used was the same as that described above, except for that annealing was done at 48℃ for 30 seconds.
Cloning and sequencing of 28S D1 rDNA and mtCOI
The PCR products amplified with the primer sets were purified by gel extraction (QIAEX DNA Gel extraction Kit, QIAGEN Co., Germany) and were subcloned into the EcoRV site of a pT7Blue T-vector Kit plus ligase (Novagen Co., USA), according to the protocol from the supplier. For transformation, NovaBlue competent cells were used as a host cell (Novagen Co., WI, USA). The recombinant plasmid was screened using isopropyl-β-thiogalactoside (IPTG) and 5-bromo-4 chloro-3-indolyl-β-D-galactoside (X-gal). The cloned fragments in the recombinant plasmids were digested with BamHI and HindIII enzyme and purified from agarose gel using a QIAprep spin plasmid kit (QIAGEN Co.). DNA sequencing was performed by dideoxy chain termination method (Sanger et al., 1977) using a Sequenase kit (ABI prism dye terminator cycle sequencing core kit, Perkin Elmer) and an automated DNA sequencer (Applied Biosystems model 373A, Perkin Elmer) on both strands with T3 and T7 primers. At least three clones were sequenced per sample with additional clones sequenced as necessary to resolve ambiguous sites.
Sequence analysis and alignment
For the analysis of DNA sequences, NCBI (National Center for Biotechnology Information) databases were used for homology analysis (BLAST2). We also calculated the fractional GC content of nucleic acid sequences using an EMBOSS GEECEE program in Sanger Institute, Cambridge, U.K. (http://analysis.molbiol.ox.ac.uk/pise_html/geecee.html). All obtained sequences were aligned automatically using CLUSTAL W program (version 1.82, CLUSTAL W WWW Service at the European Bioinformatics Institute, http://www.ebi.ac.uk/clustalw) for multiple sequence alignments while alignment gaps were treated as missing data (Higgins et al., 1994, European Bioinformatics Institute, http://www.ebi.ac.uk/clustalw/). Sequence format was Pearson (Fasta) (Pearson and Lipman, 1988); sequence type was nucleotide (nt). The sequences were submitted to GenBank data base to get the accession numbers.
Phylogenetic tree
Phylogenetic trees were generated by TREEVIEW tree drawing software for windows (version 1.6.6, Page, 1996) from aligned nucleotide sequence by CLUSTAL W program. To observe the tree type, we used Neighbor-Joining (NJ) as distance method (Saitou and Nei, 1987), phylogeny inference package (PHYLIP) as parsimony method (version 3.4., Felsenstein, 1989, 1993). Alignment gaps were treated as missing data (Hendy and Penny, 1982, Hillis et al., 1996). The correction for nucleotide distance was done by the Kimura's 2 parameter method (Kimura, 1980). The phylogenetic trees were outgroup rooted using the 28S D1 rDNA and mtCOI gene nucleotide sequences of S. falcatus, because it represented a sister taxon.
RESULTS
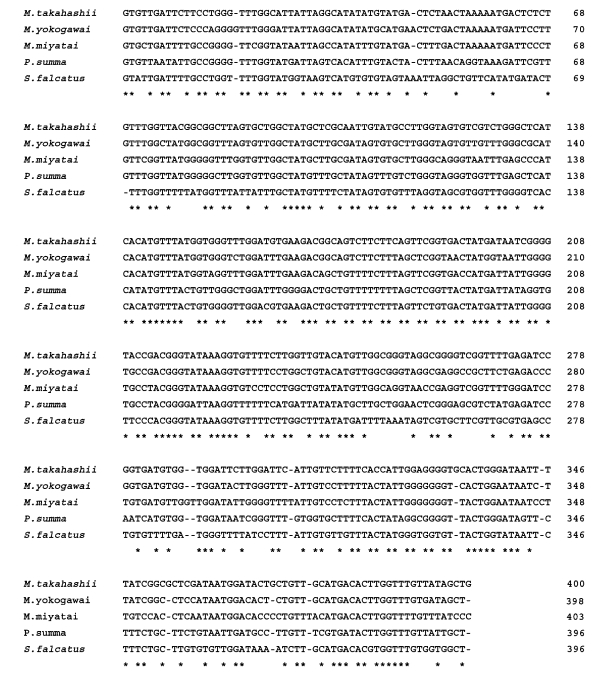
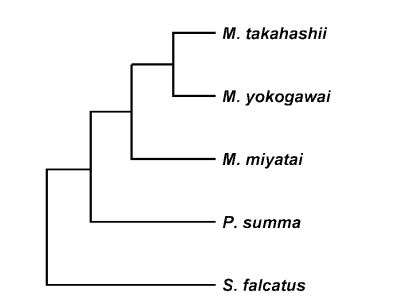
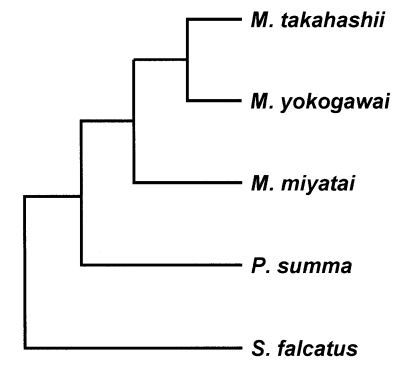
The length of the 28S D1 region rDNA sequence of the three Metagonimus species was 248 bp (adjusted for missing data) and its G+C content was 52% (M. takahashii, M. yokogawai and M. miyatai). Nucleotide sequence differences among Metagonimus species were less than 0.8% (2/248 bp), and that between the species, 0.4% (1/248 bp). There was no sequence gap in 28S D1 rDNA region (Fig. 1). The length of the mtCOI sequence averaged 400 bp (398-403 bp, adjusted for missing data) with a G+C content ranged from 44% (M. miyatai), 46% (M. takahashii) to 47% (M. yokogawai) (data not shown). Nucleotide sequence differences between species were 23.0% (92/400 bp) between M. miyatai and M. takahashii, 16.2% (65/400 bp) between M. miyatai and M. yokogawai, 13.2% (53/400 bp) between M. takahashii and M. yokogawai adjusted for missing data (Fig. 2). Nucleotide gaps sequence differences were 2.5% (10/400 bp) between M. miyatai and M. takahashii, 2.7% (11/400 bp) between M. miyatai and M. yokogawai, and 2.4% (11/400 bp) between M. takahashii and M. yokogawai (Fig. 2). The aligned sequences of three Metagonimus species showed high similarities with other comparative human intestinal trematodes (P. summa and S. falcatus) for 28S D1 rDNA (95.0%, 234/248 bp and 94%, 233/248 bp) and the mtCOI gene (68.5%: 274/400 bp and 78.0%: 312/400 bp) from nucleotide BLAST database program in NCBI (Figs. 1, 2). M. takahashii and M. yokogawai placed in the same clade supported by DNA sequence and phylogenetic tree analysis for 28S D1 rDNA and mtCOI gene by neighbor-joining and parsimony method. M. miyatai placed in different clade from two other Metagonimus species (Figs. 3, 4).
Fig. 1.
Sequences alignment of 28S D1 rDNA region of Metagonimus species. An asterisk (*) denotes an identical nucleotide position, and alignment gaps are indicated by a hyphen. Sequences for each species have been deposited in the GenBank databases (GenBank accession number: M. miyatai: AF095333, M. takahashii: AF095332, M. yokogawai: AF095331, P. summa: AF181885, S. falcatus: AF181886).
Fig. 2.
Sequences alignment of mtCOI region of Metagonimus species. An asterisk (*) denotes an identical nucleotide position, and alignment gaps are indicated by a hyphen. Sequences for each species have been deposited in the GenBank databases (GenBank accession number: M. miyatai: AF096232, M. takahashii: AF096231, M. yokogawai: AF096230, P. summa: AF181884, S. falcatus: AF181887).
Fig. 3.
Phylogenetic trees of Metagonimus species inferred from the 28S rDNA D1 gene nucleotide sequences by TREEVIEW program with the neighbor-joining and parsimony methods. The phylogenetic trees were outgroup rooted using the 28S D1 gene nucleotide sequence of Stellantchasmus falcatus.
Fig. 4.
Phylogenetic trees of Metagonimus species inferred from the mtCOI gene nucleotide sequences by TREEVIEW program with the neighbor-joining and parsimony methods. The phylogenetic trees were outgroup rooted using the mtCOI gene nucleotide sequence of Stellantchasmus falcatus.
DISCUSSION
Our data coincided with the previously reported data, i.e., general morphology, PCR-RFLP, PCRRAPD and SSR-PCR patterns of the three Metagonimus species. Rim et al. (1996) reported that the general morphology of the three species of Metagonimus was distinct and M. miyatai type was distinctive from other species of Metagonimus. Yu et al (1997a & 1997b) suggested that the M. miyata type had different DNA sequences from M. yakogawai by RAPD and RFLP patterns of ITS1 and mtCOI gene. Yang et al. (2000) showed that the three species of Metagonimus had different genotypes. Our data supported the suggestions of previous genetic studies. However, sequence data did not coincide with the results of the chromosome analyses of these three Metagonimus species. Lee et al. (1999) also suggested that the karyology of Metagonimus species had closer relationship between M. miyatai and M. takahashii (both 2n = 18) rather than between M. miyatai and M. yokogawai (2n = 32). This kind of discrepancy may be seen between karyologic and sequencing studies. We add a genetic evidence on the distinctness of M. miyatai from M. yokogawai and M. takahashii.
Footnotes
This study was supported by the Academic Research Fund of Ministry of Education, Republic of Korea, 1997-1998 (Grant No. BM 97-204).
References
- 1.Bowles J, McManus DP. Genetic characterization of the Asian Taenia, a newly described Taenid cestode of humans. Am J Trop Med Hyg. 1994;50:33–44. [PubMed] [Google Scholar]
- 2.Chai JY, Guk SM, Han ET, et al. Surface ultrastructure of Metagonimus takahashii metacercariae and adults. Korean J Parasitol. 2000a;38:9–15. doi: 10.3347/kjp.2000.38.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chai JY, Han ET, Park YK, Guk SM, Kim JL, Lee SH. High endemicity of Metagonimus yokogawai infection among residents of Samchok-shi, Kangwon-do. Korean J Parasitol. 2000b;38:33–36. doi: 10.3347/kjp.2000.38.1.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chai JY, Huh S, Yu JR, Kook JN, Jung KC. An epidemiological study of metagonimiasis along the upperreaches of the Namhan River. Korean J Parasitol. 1993;31:99–108. doi: 10.3347/kjp.1993.31.2.99. [DOI] [PubMed] [Google Scholar]
- 5.Chai JY, Kang YJ, Choi SY, Guk SM, Yu JR, Lee SH. Surface ultrastructure of Metagonimus miyatai metacercariae and adults. Korean J Parasitol. 1998;36:217–225. doi: 10.3347/kjp.1998.36.4.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chai JY, Sohn WM, Kim MH, Hong ST, Lee SH. Three morphological types of the genus Metagonimus encysted in the dace, Tribolodon taczanowsjii, caught from the Sumjin river. Korean J Parasitol. 1991;29:217–225. doi: 10.3347/kjp.1991.29.3.217. [DOI] [PubMed] [Google Scholar]
- 7.Felsenstein J. PHYLIP-Phylogeny inference package (version 3.2) Cladistics. 1989;5:164–166. [Google Scholar]
- 8.Felsenstein J. PHYLIP (Phylogeny Inference Package), version 3.5c. Seattle: Department of Genetics, University of Washington; 1993. [Google Scholar]
- 9.Hassounna N, Michot B, Bachellerie JP. The complete nucleotide sequence of mouse 28S rRNA gene. Implications for the process of size increase of the large subunit rRNA in higher eukaryotes. Nucleic Acids Res. 1984;12:3563–3583. doi: 10.1093/nar/12.8.3563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hendy MD, Penny D. Branch and bound algorithms to determine minimal evolutionary trees. Math Biosc. 1982;59:277–290. [Google Scholar]
- 11.Higgins D, Thompson J, Gibson T, Thompson JD, Higgins DG, Gibson TJ. CLUSTA W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994;22:4673–4680. doi: 10.1093/nar/22.22.4673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hillis DM, Moritz C, Mable BK. Chapter 11, Phylogenetic Inference. In: Swofford Davis L., Olsen Gary J., Waddell Peter J., Hillis Davis M., editors. Molecular Systematics. 2nd ed. Sunderland, Massachusetts, USA: Sinauer Associates, Inc. Publishers; 1996. pp. 407–514. [Google Scholar]
- 13.Katsurada F. Heterophyes in Japan, II. Creation of a new genus Metagonimus. Okayama Igakkai Zasshi. 1912;273:768–778. (in Japanese) [Google Scholar]
- 14.Kim CH, Kim NM, Lee CH, Park JS. Studies on the Metagonimus fluke in the Daechung reservoir and the upper stream of Geum river. Korean J Parasitol. 1987;25:69–82. doi: 10.3347/kjp.1987.25.1.69. [DOI] [PubMed] [Google Scholar]
- 15.Kimura M. A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J Mol Evol. 1980;16:111–120. doi: 10.1007/BF01731581. [DOI] [PubMed] [Google Scholar]
- 16.Lee GS, Cho IS, Lee YH, et al. Epidemiological study of clonorchiasis and metagonimiasis along the Geumgang (river) in Okcheon-gun (County), Korea. Korean J Parasitol. 2002;40:9–16. doi: 10.3347/kjp.2002.40.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee SU, Huh S, Park GM, Chai JY. A cytogenetic study of human intestinal parasites of the Genus Metagonimus (Digenea: Heterophyidae) in Korea. Korean J Parasitol. 1999;37:237–241. doi: 10.3347/kjp.1999.37.4.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ministry of Health and Social Welfare; Korea Association of Health. Prevalence of intestinal parasitic infections in Korea. The sixth report (Monographic series) Seoul, Korea: 1997. pp. 1–71. [Google Scholar]
- 19.Miyata I. Supplementary discussion on the classification of the genus Metagonimus. Shokubutsu Oyobi Cobutsu. 1941;9:533–534. (in Japanese) [Google Scholar]
- 20.Miyata I. Some discussions on the classification of the genus Metagonimus in Japan. Dobutsugaku Zasshi. 1944;56(1-3):6–19. (in Japanese) [Google Scholar]
- 21.Page RDM. TREEVIEW: an application to display phylogenetic trees on personal computer. Comput Appl Biosci. 1996;12:357–358. doi: 10.1093/bioinformatics/12.4.357. [DOI] [PubMed] [Google Scholar]
- 22.Pearson WR, Lipman DJ. Improved tools for biological sequence comparison. Proc Natl Acad Sci USA. 1988;85:2444–2448. doi: 10.1073/pnas.85.8.2444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Qu LH, Nioloso M, Bachellerie JP. Phylogenetic calibration of the 5' terminal domain of large rRNA achieved by determining twenty eukaryotic sequences. J Mol Evol. 1988;28:113–124. doi: 10.1007/BF02143502. [DOI] [PubMed] [Google Scholar]
- 24.Rim HJ, Kim KH, Joo KH. Classification and host specificity of Metagonimus spp. from Korean freshwater fish. Korean J Parasitol. 1996;34:7–14. doi: 10.3347/kjp.1996.34.1.7. [DOI] [PubMed] [Google Scholar]
- 25.Saito S. Species differentiation of genus Metagonimus. Reports on Meeting for Parasite Taxonomy and Morphology, Japan. 1984;2:1–4. [Google Scholar]
- 26.Saito S, Chai JY, Kim KH, Lee SH, Rim HJ. Metagonimus miyatai sp. nov. (Digenea: Heterophyidae), a new intestinal trematode transmitted by fresh water fishes in Japan and Korea. Korean J Parasitol. 1997;35:223–232. doi: 10.3347/kjp.1997.35.4.223. [DOI] [PubMed] [Google Scholar]
- 27.Saitou N, Nei M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol. 1987;4:406–425. doi: 10.1093/oxfordjournals.molbev.a040454. [DOI] [PubMed] [Google Scholar]
- 28.Sambrook J, Russell DW. Molecular cloning: a laboratory manual. 3rd ed. New York, USA: Cold Spring Harbor Laboratory Press; 2001. Chapter 6; pp. 6.1–6.30. [Google Scholar]
- 29.Sanger F, Nicklen S, Coulson AR. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA. 1977;74:5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Suzuki M. Collected papers on studies of special animals in Okayama Prefecture. 1930. On Metagonimus yakogawai; pp. 146–168.pp. XXII–XXIII. Pls.(in Japanese) [Google Scholar]
- 31.Yang HJ, Guk SM, Han ET, Chai JY. Molecular differentiation of three species of Metagonimus by simple sequence repeat anchored polymerase chain reaction (SSR-PCR) amplification. J Parasitol. 2000;86:1170–1172. doi: 10.1645/0022-3395(2000)086[1170:MDOTSO]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 32.Yu JR, Chung JS, Chai JY. Differential RAPD patterns between Metagonimus yokogawai and Metagonimus Miyata type. Korean J Parasitol. 1997a;35:295–298. doi: 10.3347/kjp.1997.35.4.295. [DOI] [PubMed] [Google Scholar]
- 33.Yu JR, Chung JS, Huh S, Lee SH, Chai JY. PCR-RFLP pattern of three kinds of Metagonimus in Korea. Korean J Parasitol. 1997b;35:271–276. doi: 10.3347/kjp.1997.35.4.271. [DOI] [PubMed] [Google Scholar]
- 34.Yu JR, Kwon SO, Lee SH. Clonorchiasis and metagonimiasis in the inhabitants along Talchongang (River), Chungwon-gun. Korean J Parasitol. 1994;32:267–269. doi: 10.3347/kjp.1994.32.4.267. [DOI] [PubMed] [Google Scholar]