Abstract
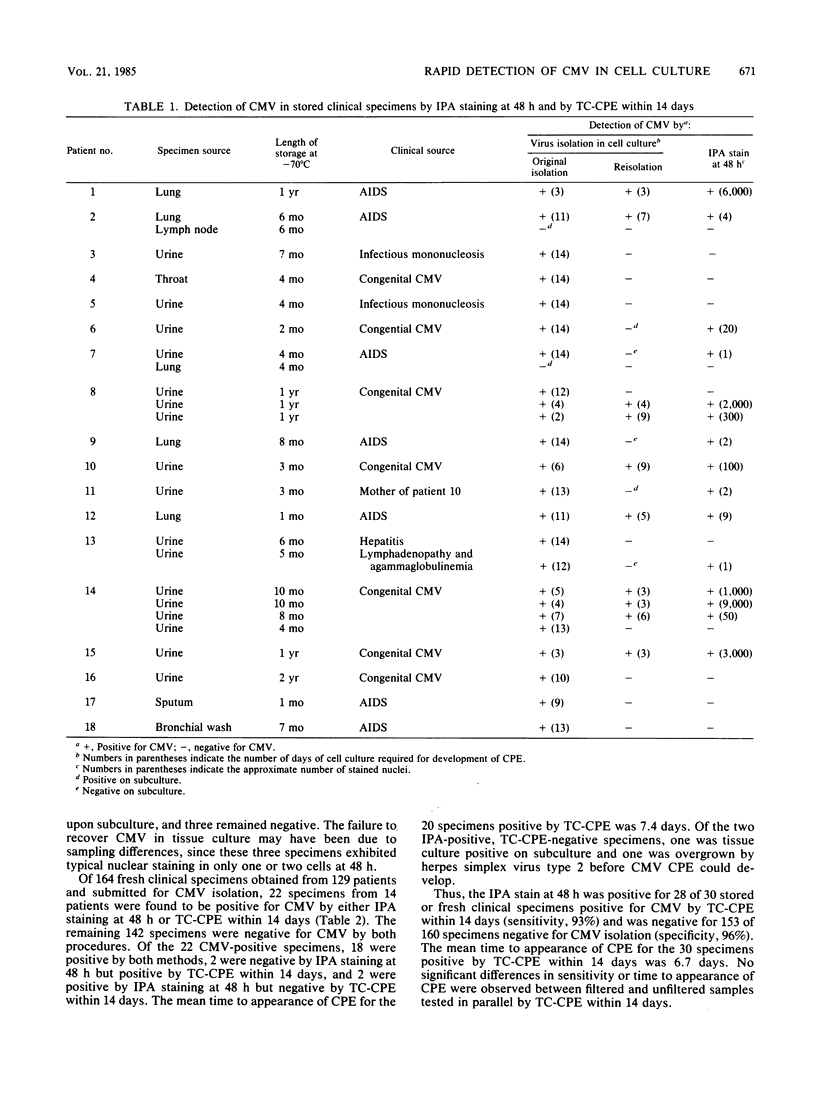
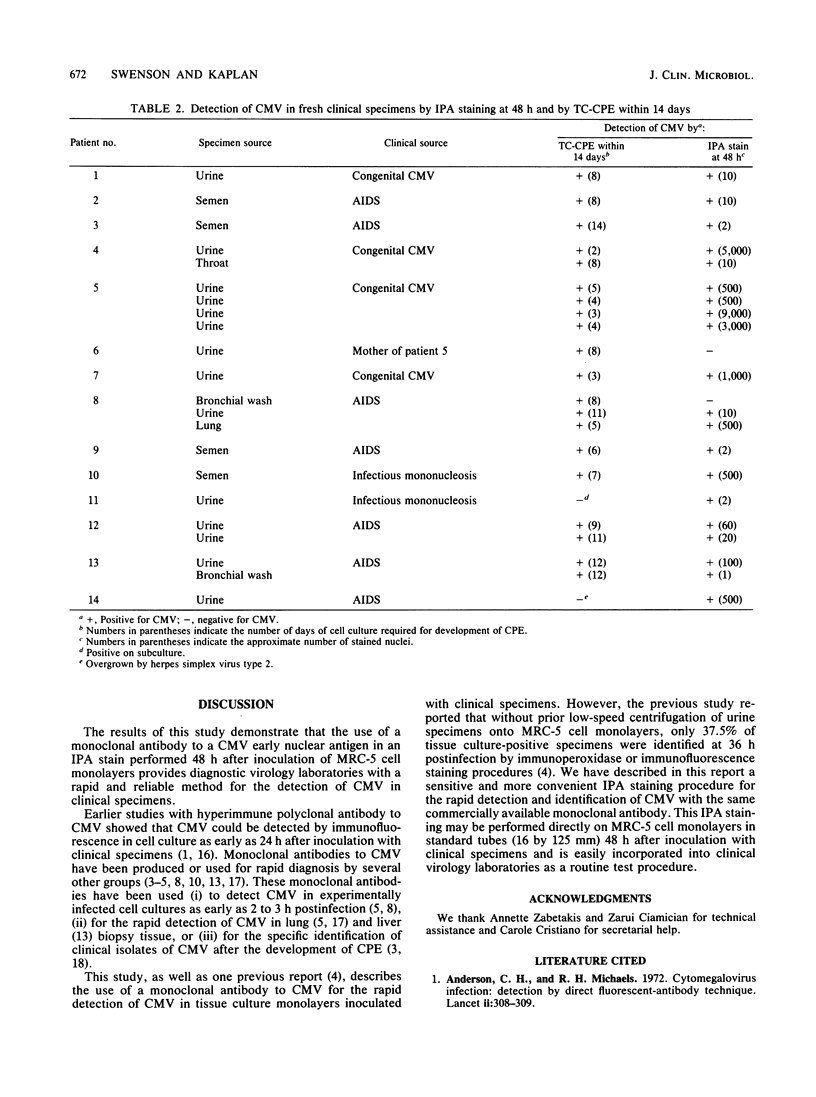
A method for the rapid detection of cytomegalovirus (CMV) in MRC-5 cells 48 h after inoculation with clinical specimens was developed. A commercially available monoclonal antibody to a CMV early nuclear antigen was used in an indirect immunoperoxidase (IPA) staining procedure performed directly on acetone-fixed cell monolayers in standard tubes (16 by 125 mm). Of 190 clinical specimens tested, 30 specimens produced CMV cytopathic effect in tissue culture (TC-CPE) within 14 days after inoculation and, of these 30, 28 were positive for CMV after 48 h by the IPA staining procedure (sensitivity, 93%). Of the remaining 160 clinical specimens negative by TC-CPE within 14 days, 7 were positive by the IPA stain (specificity, 96%). However, three of these seven specimens were positive by TC-CPE upon subculture after the initial 14-day incubation period, and one specimen was overgrown by herpes simplex virus type 2 before CMV cytopathic effect could develop. The mean time to appearance of cytopathic effect for the 30 specimens positive by TC-CPE within 14 days was 6.7 days. These findings indicate that this IPA staining is a useful method for the rapid detection of CMV in cell monolayers inoculated with clinical specimens.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson C. H., Michaels R. H. Cytomegalovirus infection: detction by direct fluorescent-antibody technique. Lancet. 1972 Aug 12;2(7772):308–309. doi: 10.1016/s0140-6736(72)92910-8. [DOI] [PubMed] [Google Scholar]
- Chou S., Merigan T. C. Rapid detection and quantitation of human cytomegalovirus in urine through DNA hybridization. N Engl J Med. 1983 Apr 21;308(16):921–925. doi: 10.1056/NEJM198304213081603. [DOI] [PubMed] [Google Scholar]
- Fiacco V., Bryson Y. J., Bruckner D. A. Comparison of monoclonal and polyclonal antibody for confirmation of cytomegalovirus isolates by fluorescent staining. J Clin Microbiol. 1984 Jun;19(6):928–930. doi: 10.1128/jcm.19.6.928-930.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gleaves C. A., Smith T. F., Shuster E. A., Pearson G. R. Rapid detection of cytomegalovirus in MRC-5 cells inoculated with urine specimens by using low-speed centrifugation and monoclonal antibody to an early antigen. J Clin Microbiol. 1984 Jun;19(6):917–919. doi: 10.1128/jcm.19.6.917-919.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein L. C., McDougall J., Hackman R., Meyers J. D., Thomas E. D., Nowinski R. C. Monoclonal antibodies to cytomegalovirus: rapid identification of clinical isolates and preliminary use in diagnosis of cytomegalovirus pneumonia. Infect Immun. 1982 Oct;38(1):273–281. doi: 10.1128/iai.38.1.273-281.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregory W. W., Menegus M. A. Practical protocol for cytomegalovirus isolation: use of MRC-5 cell monolayers incubated for 2 weeks. J Clin Microbiol. 1983 Apr;17(4):605–609. doi: 10.1128/jcm.17.4.605-609.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths P. D., Stagno S., Pass R. F., Smith R. J., Alford C. A., Jr Infection with cytomegalovirus during pregnancy: specific IgM antibodies as a marker of recent primary infection. J Infect Dis. 1982 May;145(5):647–653. doi: 10.1093/infdis/145.2.647. [DOI] [PubMed] [Google Scholar]
- Kim K. S., Sapienza V. J., Chen C. M., Wisniewski K. Production and characterization of monoclonal antibodies specific for a glycosylated polypeptide of human cytomegalovirus. J Clin Microbiol. 1983 Aug;18(2):331–343. doi: 10.1128/jcm.18.2.331-343.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee F. K., Nahmias A. J., Stagno S. Rapid diagnosis of cytomegalovirus infection in infants by electron microscopy. N Engl J Med. 1978 Dec 7;299(23):1266–1270. doi: 10.1056/NEJM197812072992302. [DOI] [PubMed] [Google Scholar]
- Pereira L., Hoffman M., Gallo D., Cremer N. Monoclonal antibodies to human cytomegalovirus: three surface membrane proteins with unique immunological and electrophoretic properties specify cross-reactive determinants. Infect Immun. 1982 Jun;36(3):924–932. doi: 10.1128/iai.36.3.924-932.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pronovost A. D., Baumgarten A., Andiman W. A. Chemiluminescent immunoenzymatic assay for rapid diagnosis of viral infections. J Clin Microbiol. 1982 Aug;16(2):345–349. doi: 10.1128/jcm.16.2.345-349.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sacks S. L., Freeman H. J. Cytomegalovirus hepatitis: evidence for direct hepatic viral infection using monoclonal antibodies. Gastroenterology. 1984 Feb;86(2):346–350. [PubMed] [Google Scholar]
- Shulman H. M., Hackman R. C., Sale G. E., Meyers J. D. Rapid cytologic diagnosis of cytomegalovirus interstitial pneumonia on touch imprints from open-lung biopsy. Am J Clin Pathol. 1982 Jan;77(1):90–94. doi: 10.1093/ajcp/77.1.90. [DOI] [PubMed] [Google Scholar]
- Spector S. A., Rua J. A., Spector D. H., McMillan R. Detection of human cytomegalovirus in clinical specimens by DNA-DNA hybridization. J Infect Dis. 1984 Jul;150(1):121–126. doi: 10.1093/infdis/150.1.121. [DOI] [PubMed] [Google Scholar]
- Stagno S., Pass R. F., Reynolds D. W., Moore M. A., Nahmias A. J., Alford C. A. Comparative study of diagnostic procedures for congenital cytomegalovirus infection. Pediatrics. 1980 Feb;65(2):251–257. [PubMed] [Google Scholar]
- Volpi A., Whitley R. J., Ceballos R., Stagno S., Pereira L. Rapid diagnosis of pneumonia due to cytomegalovirus with specific monoclonal antibodies. J Infect Dis. 1983 Jun;147(6):1119–1120. doi: 10.1093/infdis/147.6.1119. [DOI] [PubMed] [Google Scholar]
- Warford A. L., Eveland W. G., Strong C. A., Levy R. A., Rekrut K. A. Enhanced virus isolation by use of the transporter for a regional laboratory. J Clin Microbiol. 1984 Apr;19(4):561–562. doi: 10.1128/jcm.19.4.561-562.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]



