Abstract
Prostate cancer (PCa) is one of the most common cancers in the world. Inflammation has been described as a risk factor for PCa and depends on the production of cytokines in response to tissue damage or the presence of stimuli that induces cellular stress. Interindividual variation in cytokine production is partially controlled by single-nucleotide polymorphisms (SNPs) that have been associated with differential production of cytokines. We have recently showed that SNP–SNP interactions of cytokine genes are associated with PCa risk. However, little is known about the association of cytokine SNPs and PCa aggressiveness. In this study, we evaluated the association of 15 SNPs in five cytokine genes and aggressiveness of PCa in African- and Caucasian-American individuals. Caucasian Americans with the genotypes IL10−1082GG or IL1B+3954TT had 2.31-fold [95% confidence interval (CI) = 1.13–4.72] and 3.11 (95% CI = 1.20–8.06)-fold risk, respectively, of developing aggressive PCa, as compared with individuals without those genotypes. We did not find any associations in the African-American group. Using Multivariate Adaptive Regression Splines modeling for exploratory SNP–SNP interactions, our results showed that more aggressive PCa in Caucasians Americans is associated with the CT genotype at IL8−47 [odds ratios (OR) = 3.50; 95% CI = 1.13–10.88] or combined genotypes of IL1B−511CC and IL10−1082GG (OR = 3.38; 95% CI = 1.70–6.71). Unfortunately, the same analysis could not be performed in the African-Americans due to limited number of individuals. With limited sample size, the results from this study suggest that SNPs in cytokine genes may be associated with PCa aggressiveness. More extensive studies are warranted to validate our findings.
Introduction
Prostate cancer (PCa) is the fifth most common cancer in the world and the second most common cancer in men (1). The incidence of the disease varies according to geographical location. Regions in Western Asia and China, for example, have reported incidence rates of 11 and 2 per 100 000 individuals, respectively, whereas in North America, the average annual incidence rate is 120 per 100 000 individuals (1). In the USA alone, it is estimated that ∼186 320 new cases of PCa will be diagnosed in 2008 and that close to 28 660 men will die due to the disease (2).
Studies have identified both genetic and non-genetic factors associated with the risk of PCa. However, it can be difficult to separate the effects of both types of factors (3). The most recognized factors associated with PCa risk are ethnicity, age and family history of the disease. African-American men have approximately a 1.6-fold increased risk of developing PCa and almost 2.5 higher risk of dying from the disease than Caucasian American (4,5). Older individuals have a higher risk of PCa than younger individuals, although individuals with a positive family history are more probably to have earlier onset of the disease (6). The probability of PCa increases dramatically from 0.01% in men <39 years old to almost 14% in men ≥70 years of age (4). Individuals with at least one first-degree relative with PCa have between 3 and 5 times higher chance of developing the disease than those with a negative family history (6). In addition, individuals with both first- and second-degree relatives affected by PCa had a 11-fold increased risk of PCa (6). Additionally, those with a positive family history also had a higher probability of having an earlier onset of the disease (7). Brothers of an affected individual have a higher probability of being diagnosed with PCa than the sons of affected fathers [pooled estimated relative risk is 3.4, 95% confidence interval (CI) 2.9–4.1 for brother-to-brother risk compared with 2.5, 95% CI = 2.1–3.1 for father-to-son risk] (7,8). It was recently suggested that ∼42% of the risk for PCa may be explained by heritable factors (9). Studies with twins have shown that the relative risk of a monozygotic twin developing PCa if his brother is diagnosed with PCa is more than five times higher than the RR of a dizygotic twin to develop the disease if his brother is diagnosed (4.9, 95% CI = 2.8–8.6 versus 0.8, 95% CI = 0.3–1.8, respectively) (10). Many genetic abnormalities have been associated with risk of PCa, including allelic losses observed at chromosomes 7q, 8q, 10q, 13q, 16q, 17p and 18q (11–16).
In recent years, chronic inflammation has emerged as a risk factor for in many types of cancer, including PCa (17,18). The importance of inflammation in PCa development is highlighted by the finding that ∼20% of patients presenting with chronic inflammation in the prostate developed PCa after a 5 year follow-up, when compared with patients with no chronic inflammatory changes (19). Interestingly, many tumors induce the production of cytokines to promote their own growth and to limit immune responses (20–23). The activation of inflammatory cells, like monocytes/macrophages, lead to the production of cytokines that initiate, maintain and promote inflammation, including interleukin (IL) 1-beta, tumor necrosis factor alpha, IL6, IL8 and IL10. Modulation of cytokine production is pivotal for the control of inflammation and tumor progression. Single-nucleotide polymorphisms (SNPs) in cytokine genes, including some in this report, have been associated with modulation of cytokine levels and with the development of cancer (24–29). We have recently shown that the risk of PCa is differentially associated with cytokine SNPs in African and Caucasians Americans (30). However, very little information about the association of cytokine SNPs and PCa aggressiveness is known. Our goal in this study was to evaluate the association between 15 SNPs in five cytokine genes and PCa aggressiveness in African and Caucasians Americans.
Materials and methods
Patient samples
The samples were obtained by a clinic-based all-patients approach for which the DNA samples from PCa patients from the Departments of Urology and Internal Medicine of the Wake Forest University School of Medicine were recruited with sequential patient population, as described previously (31,32). Briefly, a blood sample was taken from participants who had received a description of the study and signed an informed consent. In order to be included in the study, the participants should not have been diagnosed with immune diseases, chronic inflammatory conditions or infections in the past 6 weeks before the sample was taken. Patients were either incident PCa cases (newly diagnosed) or prevalent cases (diagnosed with PCa within 5 years but without PCa and treatment for at least 6 months before the study). All the procedures were approved by the Medical Center's Institutional Review Board.
SNP analysis
SNP genotyping was done using 4 ng of DNA as described previously (30). For this study, we genotyped three SNPs in the IL1B gene (IL1B−511C>T, IL1B−31T>C and IL1B+3954C>T), three SNPs in the IL10 gene (IL10−1082G>A, IL10−819C>T and IL10−592C>A), three SNPs in the TNFA gene (TNF−857C>T, TNF−308G>A and TNF−238G>A), three SNPs in the IL6 gene (IL6−174C>G, IL6−598G>A and IL6IVS2+180G>A) and three SNPs in the IL8 gene (IL8−251A>T, IL8EX1−47C>T and IL8IVS1+230G>T). Controls of known genotype for each polymorphic locus were always run in parallel with each experiment per SNP analysis. In addition, four internal controls were included in each 96-well plate. The concordance of the genotypes for both types of controls was always >98.5%. Individuals with missing information were not included in the analysis.
Statistical analysis
The goal of this study was to evaluate the association between 15 cytokine SNPs and PCa aggressiveness; therefore, only PCa cases were included. PCa cases were classified based on Gleason's grade, clinical stage and prostate-specific antigen (PSA) at diagnosis as follows: high aggressive (Gleason's sum ≥8 or PSA >20 ng/ml or Gleason's sum ≥7 and clinical stages T3–T4), low aggressive (Gleason's sum <7 and clinical stage T1–T2 and PSA <10 ng/ml) or intermediate aggressive (all other cases) (33). Because of insufficient information on Gleason's grade and/or PSA to determine PCa aggressiveness, 71 Caucasian-American patients (12.1%) and 7 African-American patients (8.5%) were excluded from the analysis. Additionally, three subjects of other ethnic groups were excluded. With differential distributions of aggressiveness in the two racial groups, all the statistical analyses were stratified by race.
The genotype frequencies of the cytokine SNPs in low and intermediate PCa cases were very similar; therefore, we joined them in a single group to compare with the high PCa aggressiveness. Chi-square and Fisher's exact tests were used to compare the demographic, clinical characteristics and individual SNPs between high and low/intermediate aggressiveness. Multivariate logistic regression models were used for testing the one-to-one association between PCa aggressiveness and each of the SNPs. The potential confounding factors included age, smoking history as risk for aggressive disease (34) and history of benign prostatic hyperplasia diagnosis. The smoking history was evaluated by whether the individual has ever smoked at least 100 cigarettes in his lifetime. The crude and adjusted odds ratios (ORs) and 95% confidence interval (CI) are presented. Finally, the association between cytokine gene haplotype and PCa aggressiveness was examined using Haplostat (35). Multivariate logistic regression adjusting for age, smoking history and history of benign prostatic hyperplasia diagnosis was examined for each SNP.
SNP–SNP interactions were only performed in Caucasian Americans because of the small sample size for African-Americans in this study. Three SNP pairs were observed to be highly correlated (r2 > 0.8) and in strong linkage (D′ > 0.8): (i) IL1B−511C>T and IL1B−31T>C; (ii) IL10−819C>T and IL10−592C>A and (iii) IL6−598G>A and IL6IVS2+180G>A. Thus, three SNPs (IL1B−31T>C, IL10−592C>A and IL6IVS2+180G>A) were excluded from the analyses of SNP interactions. Multivariate Adaptive Regression Splines (MARS) were applied to explore SNP–SNP interactions associated with PCa aggressiveness. MARS with the features of flexible group selection, automatic genotype combination and automatic interaction pattern detection is shown to be a powerful method for analyzing SNP–SNP interactions for complex diseases (36). Statistical analyses were performed using SAS 9.1 and MARS 2.0.
Results
The racial sociodemographic characteristics of our population are shown in Table I, as a function of PCa aggressiveness. As shown, none of the factors studied, including age, smoking history and benign prostatic hyperplasia history, presented any differential distribution between both African- and Caucasian-American individuals with either low/intermediate or high aggressive disease.
Table I.
Demographic and clinical characteristics of study subjects
| Prostate cancer aggressiveness | P-value* | ||
| High, n (%) | Low/intermediate, n (%) | ||
| African-Americans | |||
| Age | |||
| <60 | 5 (33.3) | 31 (50.0) | 0.18 |
| 60–69 | 6 (40.0) | 25 (40.3) | |
| ≥70 | 4 (26.7) | 6 (9.7) | |
| Smoking history | |||
| Yes | 12 (80.0) | 38 (61.3) | 0.20 |
| No | 1 (6.7) | 18 (29.0) | |
| Missing | 2 | 6 | |
| History of BPH | |||
| Yes | 8 (53.3) | 18 (29.0) | 0.09 |
| No | 4 (26.7) | 36 (58.1) | |
| Missing | 3 (20) | 8 (12.9) | |
| Caucasians Americans | |||
| Age | |||
| <60 | 17 (20.5) | 143 (31.9) | 0.09 |
| 60–69 | 42 (50.6) | 207 (46.2) | |
| ≥70 | 24 (28.9) | 98 (21.9) | |
| Smoking history | |||
| Yes | 56 (67.5) | 285 (63.6) | 0.67 |
| No | 26 (31.3) | 160 (35.7) | |
| Missing | 1 | ||
| History of BPH | |||
| Yes | 40 (48.2) | 199 (44.4) | 0.12 |
| No | 41 (49.4) | 247 (55.1) | |
| Missing | 2 (2.4) | 2 (0.5) | |
*P-value of chi-square or Fisher's exact test, not including missing values in all variables.
With the knowledge that there is a differential distribution of cytokine gene SNPs in African- and Caucasian-American individuals with PCa, we designed the study to test whether cytokine SNPs distributions contribute to PCa aggressiveness. The results in Table II show the adjusted models determining the associated risk (OR and 95% CI) of diagnosis of PCa aggressive disease in African- and Caucasian-American PCa patients based on different cytokine genotypes, as compared with the corresponding reference genotypes. As shown, none of the genotypes studied alone was associated with aggressive disease in African-Americans, a finding that may be due to limited sample size (Table II). In contrast, Caucasian-American individuals carrying the genotype IL1B+3954TT had >3-fold risk of being diagnosed with aggressive PCa (OR = 3.11; 95% CI = 1.20–8.06). Similarly, Caucasian Americans with the genotype IL10−1082GG had increased risk of aggressive PCa when compared with individuals who do not carry such a genotype (OR = 2.31; 95% CI = 1.13–4.72). In addition, Caucasian-American patients with the genotype IL8−47CT or carrying the allele T at this position (IL8−47CT/TT) had increased risk of aggressive PCa (OR = 3.81; 95% CI = 1.35–10.75 and OR = 3.01; 95% CI = 1.20–7.56, respectively). None of the associations between cytokine gene haplotypes and PCa aggressiveness were statistically significant in either African- or Caucasian-American individuals.
Table II.
Cytokine genetic polymorphisms and risk of aggressive PCa stratified by race
| Gene-SNP location | Genotype | Caucasian Americans | African-Americans |
||
| High/low-intermediate aggressivenessa | Adjusted ORb (95% CI) | High/low-intermediate aggressiveness | Adjusted OR (95% CI) | ||
| IL1B−511 | CC | 34/180 | 1.00 | 1/12 | 1.00 |
| CT | 31/196 | 0.94 (0.55–1.61) | 6/25 | 9.48 (0.73–122.86) | |
| TT | 9/42 | 0.99 (0.42–2.31) | 6/21 | 6.55 (0.59–72.35) | |
| CT/TT | 40/238 | 0.95 (0.57–1.58) | 12/46 | 7.40 (0.72–76.48) | |
| IL1B−31 | TT | 32/179 | 1.00 | 1/12 | 1.00 |
| CC | 32/197 | 1.25 (0.55–2.86) | 6/23 | 6.64 (0.60–73.78) | |
| TC | 10/41 | 1.02 (0.59–1.75) | 6/22 | 11.33 (0.84–152.86) | |
| TC/CC | 42/238 | 1.06 (0.63–1.78) | 12/45 | 7.83 (0.75–81.22) | |
| ILB+3954 | CC | 45/244 | 1.00 | 8/46 | 1.00 |
| CT | 22/155 | 0.80 (0.46–1.40) | 4/9 | 3.62 (0.72–18.19) | |
| TT | 8/14 | 3.11 (1.20–8.06) | 1/3 | 3.07 (0.22–43.40) | |
| CT/TT | 30/169 | 1.00 (0.60–1.66) | 5/12 | 3.48 (0.80–15.11) | |
| IL10−1082 | AA | 14/110 | 1.00 | 5/16 | 1.00 |
| GA | 32/203 | 1.21 (0.60–2.43) | 7/35 | 0.70 (0.17–2.90) | |
| GG | 29/105 | 2.31 (1.13–4.72) | 1/7 | 0.68 (0.05–8.52) | |
| GG/GA | 61/308 | 1.58 (0.83–3.02) | 8/42 | 0.70 (0.18–2.78) | |
| IL10−819 | CC | 47/243 | 1.00 | 6/22 | 1.00 |
| CT | 18/131 | 0.69 (0.38–1.26) | 6/26 | 0.63 (0.15–2.70) | |
| TT | 5/30 | 0.95 (0.35–2.61) | 0/8 | NCc | |
| TT/CT | 23/161 | 0.74 (0.42–1.27) | 6/34 | 0.50 (0.12–2.10) | |
| IL10−592 | CC | 48/251 | 1.00 | 6/20 | 1.00 |
| AA | 6/31 | 1.13 (0.44–2.91) | 1/7 | 0.46 (0.04–5.29) | |
| CA | 20/140 | 0.73 (0.41–1.29) | 6/30 | 0.48 (0.12–2.01) | |
| CA/AA | 26/141 | 0.80 (0.47–1.35) | 7/37 | 0.48 (0.12–1.87) | |
| TNF−857 | CC | 64/349 | 1.00 | 12/52 | 1.00 |
| CT | 7/52 | 0.75 (0.32–1.74) | 0/4 | NC | |
| TT | 0/6 | NC | 0/0 | NC | |
| CT/TT | 7/58 | 0.68 (0.30–1.57) | 0/4 | NC | |
| TNF−238 | GG | 69/368 | 1.00 | 11/52 | 1.00 |
| AA | 0/35 | NC | 0/0 | NC | |
| GA | 6/35 | 0.90 (0.36–2.25) | 1/4 | 1.40 (0.13–15.75) | |
| GA/AA | 6/70 | 0.78 (0.32–1.93) | 1/4 | 1.40 (0.13–15.75) | |
| TNF−308 | GG | 47/291 | 1.00 | 11/50 | 1.00 |
| AA | 3/10 | 1.70 (0.44–6.53) | 0/1 | NC | |
| GA | 24/121 | 1.18 (0.69–2.05) | 2/7 | 0.87 (0.14–5.6) | |
| GA/AA | 27/131 | 1.22 (0.72–2.08) | 2/8 | 0.83 (0.13–5.32) | |
| IL6−598 | GG | 23/128 | 1.00 | 9/43 | 1.00 |
| AA | 9/79 | 0.68 (0.30–1.57) | 0/1 | NC | |
| GA | 38/176 | 1.19 (0.66–2.14) | 2/10 | 1.19 (0.18–7.70) | |
| GA/AA | 47/255 | 1.04 (0.59–1.81) | 2/11 | 1.19 (0.18–7.70) | |
| IL6+180 | GG | 22/123 | 1.00 | 10/43 | 1.00 |
| AA | 10/19 | 0.73 (0.32–1.65) | 0/2 | NC | |
| GA | 34/34 | 1.08 (0.59–1.97) | 2/34 | 1.71 (0.25–11.57) | |
| GA/AA | 44/53 | 0.97 (0.54–1.72) | 2/36 | 1.68 (0.25–11.32) | |
| IL6−174 | CC | 21/112 | 1.00 | 3/6 | 1.00 |
| CG | 34/163 | 1.08 (0.59–2.00) | 2/10 | 0.66 (0.05–8.92) | |
| GG | 19/126 | 0.77 (0.39–1.52) | 10/41 | 0.38 (0.05–3.13) | |
| CG/GG | 53/289 | 0.94 (0.54–1.64) | 12/51 | 0.41 (0.01–3.29) | |
| IL8+230 | GG | 19/98 | 1.00 | 3/15 | 1.00 |
| GT | 25/176 | 0.76 (0.40–1.46) | 7/23 | 1.13 (0.22–5.82) | |
| TT | 27/119 | 1.10 (0.57–2.13) | 2/17 | 0.43 (0.05–3.65) | |
| GT/TT | 52/295 | 0.90 (0.50–1.61) | 9/40 | 0.84 (0.18–3.97) | |
| IL8−47 | CC | 63/372 | 1.00 | 11/49 | 1.00 |
| CT | 7/10 | 3.81 (1.35–10.76) | 1/4 | 1.35 (0.12–14.97) | |
| TT | 1/5 | 1.29 (0.14–11.67) | 1/3 | 5.10 (0.32–81.48) | |
| CT/TT | 8/15 | 3.01 (1.20–7.56) | 2/7 | 2.19 (0.33–14.51) | |
| IL8−251TT | TT | 27/112 | 1.00 | 1/4 | 1.00 |
| AA | 16/87 | 0.78 (0.39–1.56) | 6/31 | 1.44 (0.10–19.95) | |
| TA | 22/154 | 0.65 (0.34–1.23) | 4/13 | 1.78 (0.13–25.12) | |
| TA/AA | 38/241 | 0.70 (0.40–1.23) | 10/44 | 1.59 (0.13–19.79) | |
NC, non-convergent.
Number of subjects with high aggressive PCa compared with number of subjects with low or intermediate aggressive PCa.
Adjusted for age, smoking history and history of BPH.
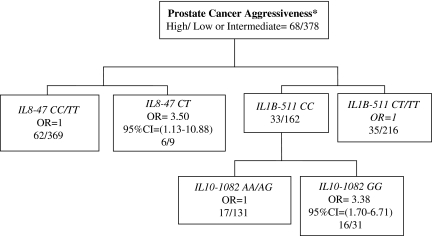
We investigated whether interactions of SNPs modify the risk of PCa aggressiveness. Due to the limited number of African-American patients, we did the analysis in the Caucasian-American group only. As shown in Figure 1, the MARS analysis revealed one main effect and a two-way SNP interaction associated with PCa aggressiveness. Caucasian-American individuals carrying the IL8−47CT genotype had a 3.5-fold risk of having aggressive PCa as compared with those individuals who do not carry that genotype at the IL8 gene locus (OR = 3.5; 95% CI = 1.13–10.88) (Figure 1). In addition, Caucasian-American PCa patients simultaneously carrying the genotypes IL1B−511CC and IL10−1082GG had almost 3.4-fold risk of presenting an aggressive disease when compared with individuals who do not carry that genotype combination (OR = 3.38; 95% CI = 1.70–6.71) (Figure 1).
Fig. 1.
MARS model of PCa aggressiveness in Caucasians. MARS analysis to determine the association of SNP–SNP interactions and PCa aggressiveness in Caucasian individuals was done as described in Materials and Methods. Only one of the SNPs with strong linkage disequilibrium or highly correlated were considered for the MARS analysis. * Frequency of high/low or intermediate for complete data in the model. OR adjusted for age, smoking history and BPH.
Discussion
Cancer is an actively progressive disease. Therefore, we believe that it is imperative to establish mechanisms to identify people at risk of developing more aggressive diseases and those who would get more benefit of the current therapies. Previous work of our group studying patients with gastric premalignant lesions showed that the presence of IL1B+3954T allele was associated with the development of multifocal atrophic gastritis without regard to race (37). Since only a minor fraction of patients with multifocal atrophic gastritis progress to dysplasia and to gastric cancer (38), we proposed that allele IL1B+3954T is a marker for those individuals who do not advance to more aggressive stages of the disease. In PCa, however, the definition of these molecular markers has not been established, even though genetic factors are considered to play major role in PCa risk (9). A recent case–control study by Michaud et al. (39) showed that seven SNPs in cytokine genes, including some of the ones reported here, did not have any effect on either risk of PCa or risk of advanced disease in African- and Caucasian-American individuals. However, we have shown that in PCa, SNPs in cytokine genes are differentially distributed between African and Caucasians Americans and they interact differently in both ethnic groups to modify PCa risk (30). One possible explanation for these different results is the way the analyses were done in both studies. In our case, all the analyses were done separately in African and Caucasian Americans, whereas in the study by Michaud et al. (39), all cases were mixed for the analysis of the association of individual SNPs and PCa risk. Xu et al. (40) also analyzed the effect of the interaction of 57 SNPs in several genes in Caucasian-American individuals. They found that out of all the possible combinations of four SNPs, those in the IL10, IL1RN, TIRAP and TLR5 genes increase the probability to predict PCa status (P = 0.019). In the present all-cases study, we determined the association of SNPs in genes involved in the inflammatory cascade and the risk of aggressive PCa. Interestingly, even though African- and Caucasian-American patients with either high or low/intermediate aggressive disease had specific and differential arrays of SNPs, African-American patients with low/intermediate aggressive PCa presented the higher frequencies of IL10−819T, IL10−592A and IL8−47T. In contrast, the presence of IL6−598A and IL6+180A was more frequently associated with Caucasian-American patients with low/intermediate aggressive PCa. However, after comparing low/intermediate PCa grades to high-grade PCa, we found that the risk of developing aggressive PCa was increased in Caucasian-American patients with inflammatory alleles at positions IL1B+3954, IL10−1802 and IL8−47. One of these genotypes, IL10−1082GG, is not only associated with risk of PCa (30) but also with aggressive PCa disease (this study). None of the studied SNPs were associated with aggressive disease in African-Americans and this may be due to the low number of patients from this ethnic group included in the study.
Another way to modulate the risk of PCa would be through the interaction of genes. In this way, one gene that initially might not be associated with risk becomes associated when interacts with another gene. In order to explore the role that cytokine gene SNP interactions might have in PCa aggressiveness, MARS analysis, a flexible and powerful method to detect gene–gene interactions, was applied (36). We found that Caucasian Americans carrying the IL8−47CT genotype had more risk of having aggressive PCa (OR = 3.50; 95% CI = 1.13–10.38). Similarly, Caucasian-American individuals carrying simultaneously the genotypes IL1B−511CC and IL10−1082GG had almost 3.4-fold increased aggressive PCa risk, as compared with those individuals who do not carry that genotype combination. In our previous report (30), we found the same gene–gene interaction, although involving different SNPs, to be associated with the presence of PCa in Caucasian Americans. This is important because several cytokine gene SNPs have been associated with modulation of cytokine levels. For example, it has been shown that SNPs in the IL1B gene are associated with modulation of IL1-beta production (28,41) and the same is true for IL10 and IL8 gene SNPs (29,42). The association of these SNPs with PCa aggressiveness in Caucasian-American individuals may then be linked to a highly inflammatory environment characterized by increased production of IL1-beta and IL8 and reduced levels of IL10, but this is still to be proven.
Even though the genotype profiling of a person with cancer may be associated with risk of the disease, these analyses should be coupled to complementary studies aiming to set a complete molecular signature of individuals at risk, including gene expression profiles and microRNA expression. Recent studies using gene expression assays have been used to predict the survival of patients with lung and colorectal carcinoma (43,44) and have proven to be more precise in the classification of the disease stage than the current systems. Our analysis, even though it has the limitation given by a small sample size and limited number of SNPs and genes, is encouraging because it warrants future research on PCa genomics.
Funding
American Cancer Society (CNE-101119) to J.J.H.; National Research Foundation to the Wake Forest University's General Clinical Research Center (M01-RR07122); National Cancer Institute; National Institutes of Health (CA82689 and CA107974 to A.C.O.) and (K08 CA 113452 to P.E.C.).
Acknowledgments
The authors are grateful to study participants. We also want to acknowledge the contributions of Frank M.Torti, Robert Lee, Charles J.Rosser, Dean G.Assimos, Elizabeth Albertson, Dominick J.Carbone, William Rice, Francis O'Brien, Ray Morrow, Franklyn Millman, Nadine Shelton, Joel Anderson, Shirley Cothren and Eunkyung Chang, the General Clinical Research Center, the Urology Clinic and the Internal Medicine Clinic at Wake Forest University School of Medicine.
Conflict of Interest Statement: None declared.
Glossary
Abbreviations
- CI
confidence interval
- IL
interleukin
- MARS
Multivariate Adaptive Regression Splines
- OR
odds ratio
- PCa
prostate cancer
- PSA
prostate-specific antigen
- SNP
single-nucleotide polymorphism
References
- 1.Parkin DM, et al. Global cancer statistics, 2002. CA Cancer J. Clin. 2005;55:74–108. doi: 10.3322/canjclin.55.2.74. [DOI] [PubMed] [Google Scholar]
- 2.American Cancer Society. Cancer Facts and Figures. 2007. www.cancer.org. (Nov 2008, date last accessed) [Google Scholar]
- 3.Schaid DJ. The complex genetic epidemiology of prostate cancer. Hum. Mol. Genet. 2004 doi: 10.1093/hmg/ddh072. 13 Spec No. 1, R103–R121. [DOI] [PubMed] [Google Scholar]
- 4.Jemal A, et al. Cancer statistics, 2007. CA Cancer J. Clin. 2007;57:43–66. doi: 10.3322/canjclin.57.1.43. [DOI] [PubMed] [Google Scholar]
- 5.National Cancer Institute. Surveillance, Epidemiology and End Results. 2007. http://seer.cancer.gov/. (Nov 2008, date last accessed) [Google Scholar]
- 6.Carter BS, et al. Hereditary prostate cancer: epidemiologic and clinical features. J. Urol. 1993;150:797–802. doi: 10.1016/s0022-5347(17)35617-3. [DOI] [PubMed] [Google Scholar]
- 7.Lesko SM, et al. Family history and prostate cancer risk. Am. J. Epidemiol. 1996;144:1041–1047. doi: 10.1093/oxfordjournals.aje.a008876. [DOI] [PubMed] [Google Scholar]
- 8.Johns LE, et al. A systematic review and meta-analysis of familial prostate cancer risk. BJU Int. 2003;91:789–794. doi: 10.1046/j.1464-410x.2003.04232.x. [DOI] [PubMed] [Google Scholar]
- 9.Lichtenstein P, et al. Environmental and heritable factors in the causation of cancer–analyses of cohorts of twins from Sweden, Denmark, and Finland. N. Engl. J. Med. 2000;343:78–85. doi: 10.1056/NEJM200007133430201. [DOI] [PubMed] [Google Scholar]
- 10.Ahlbom A, et al. Cancer in twins: genetic and nongenetic familial risk factors. J. Natl. Cancer Inst. 1997;89:287–293. doi: 10.1093/jnci/89.4.287. [DOI] [PubMed] [Google Scholar]
- 11.Aprikian AG, et al. Immunohistochemical determination of p53 protein nuclear accumulation in prostatic adenocarcinoma. J. Urol. 1994;151:1276–1280. doi: 10.1016/s0022-5347(17)35231-x. [DOI] [PubMed] [Google Scholar]
- 12.Bookstein R, et al. Promoter deletion and loss of retinoblastoma gene expression in human prostate carcinoma. Proc. Natl Acad. Sci. USA. 1990;87:7762–7766. doi: 10.1073/pnas.87.19.7762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bookstein R, et al. p53 is mutated in a subset of advanced-stage prostate cancers. Cancer Res. 1993;53:3369–3373. [PubMed] [Google Scholar]
- 14.Carter BS, et al. Allelic loss of chromosomes 16q and 10q in human prostate cancer. Proc. Natl Acad. Sci. USA. 1990;87:8751–8755. doi: 10.1073/pnas.87.22.8751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Latil A, et al. Genetic alterations in localized prostate cancer: identification of a common region of deletion on chromosome arm 18q. Genes Chromosomes Cancer. 1994;11:119–125. doi: 10.1002/gcc.2870110208. [DOI] [PubMed] [Google Scholar]
- 16.Richmond PJ, et al. Aberrant E-cadherin and alpha-catenin expression in prostate cancer: correlation with patient survival. Cancer Res. 1997;57:3189–3193. [PubMed] [Google Scholar]
- 17.Coussens LM, et al. Inflammatory cells and cancer: think different! J. Exp. Med. 2001;193:F23–F26. doi: 10.1084/jem.193.6.f23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Coussens LM, et al. Inflammation and cancer. Nature. 2002;420:860–867. doi: 10.1038/nature01322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.MacLennan GT, et al. The influence of chronic inflammation in prostatic carcinogenesis: a 5-year followup study. J. Urol. 2006;176:1012–1016. doi: 10.1016/j.juro.2006.04.033. [DOI] [PubMed] [Google Scholar]
- 20.Allavena P, et al. The chemokine receptor switch paradigm and dendritic cell migration: its significance in tumor tissues. Immunol. Rev. 2000;177:141–149. doi: 10.1034/j.1600-065x.2000.17714.x. [DOI] [PubMed] [Google Scholar]
- 21.Jonjic N, et al. Expression of adhesion molecules and chemotactic cytokines in cultured human mesothelial cells. J. Exp. Med. 1992;176:1165–1174. doi: 10.1084/jem.176.4.1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schoppmann SF, et al. Tumor-associated macrophages express lymphatic endothelial growth factors and are related to peritumoral lymphangiogenesis. Am. J. Pathol. 2002;161:947–956. doi: 10.1016/S0002-9440(10)64255-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Torisu H, et al. Macrophage infiltration correlates with tumor stage and angiogenesis in human malignant melanoma: possible involvement of TNFalpha and IL-1alpha. Int. J. Cancer. 2000;85:182–188. [PubMed] [Google Scholar]
- 24.Alpizar-Alpizar W, et al. Association of interleukin-1B and interleukin-1RN polymorphisms with gastric cancer in a high-risk population of Costa Rica. Clin. Exp. Med. 2005;5:169–176. doi: 10.1007/s10238-005-0082-3. [DOI] [PubMed] [Google Scholar]
- 25.El-Omar EM, et al. Interleukin-1 polymorphisms associated with increased risk of gastric cancer. Nature. 2000;404:398–402. doi: 10.1038/35006081. [DOI] [PubMed] [Google Scholar]
- 26.Machado JC, et al. Interleukin 1B and interleukin 1RN polymorphisms are associated with increased risk of gastric carcinoma. Gastroenterology. 2001;121:823–829. doi: 10.1053/gast.2001.28000. [DOI] [PubMed] [Google Scholar]
- 27.Machado JC, et al. A proinflammatory genetic profile increases the risk for chronic atrophic gastritis and gastric carcinoma. Gastroenterology. 2003;125:364–371. doi: 10.1016/s0016-5085(03)00899-0. [DOI] [PubMed] [Google Scholar]
- 28.Rad R, et al. Cytokine gene polymorphisms influence mucosal cytokine expression, gastric inflammation, and host specific colonisation during Helicobacter pylori infection. Gut. 2004;53:1082–1089. doi: 10.1136/gut.2003.029736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Turner DM, et al. An investigation of polymorphism in the interleukin-10 gene promoter. Eur. J. Immunogenet. 1997;24:1–8. doi: 10.1111/j.1365-2370.1997.tb00001.x. [DOI] [PubMed] [Google Scholar]
- 30.Zabaleta J, et al. Interactions of cytokine gene polymorphisms in prostate cancer risk. Carcinogenesis. 2008;29:573–578. doi: 10.1093/carcin/bgm277. [DOI] [PubMed] [Google Scholar]
- 31.Hu JJ, et al. Deficient nucleotide excision repair capacity enhances human prostate cancer risk. Cancer Res. 2004;64:1197–1201. doi: 10.1158/0008-5472.can-03-2670. [DOI] [PubMed] [Google Scholar]
- 32.Lockett KL, et al. DNA damage levels in prostate cancer cases and controls. Carcinogenesis. 2006;27:1187–1193. doi: 10.1093/carcin/bgi288. [DOI] [PubMed] [Google Scholar]
- 33.Schroeder JC, et al. The North Carolina-Louisiana Prostate Cancer Project (PCaP): methods and design of a multidisciplinary population-based cohort study of racial differences in prostate cancer outcomes. Prostate. 2006;66:1162–1176. doi: 10.1002/pros.20449. [DOI] [PubMed] [Google Scholar]
- 34.Plaskon LA, et al. Cigarette smoking and risk of prostate cancer in middle-aged men. Cancer Epidemiol. Biomarkers Prev. 2003;12:604–609. [PubMed] [Google Scholar]
- 35.Lin DY, et al. Simple and efficient analysis of disease association with missing genotype data. Am. J. Hum. Genet. 2008;82:444–452. doi: 10.1016/j.ajhg.2007.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lin HY, et al. Comparison of multivariate adaptive regression splines and logistic regression in detecting SNP-SNP interactions and their application in prostate cancer. J. Hum. Genet. 2008;53:802–811. doi: 10.1007/s10038-008-0313-z. [DOI] [PubMed] [Google Scholar]
- 37.Zabaleta J, et al. Association of interleukin-1beta gene polymorphisms with precancerous gastric lesions in African Americans and Caucasians. Am. J. Gastroenterol. 2006;101:163–171. doi: 10.1111/j.1572-0241.2006.00387.x. [DOI] [PubMed] [Google Scholar]
- 38.Correa P, et al. Gastric precancerous process in a high risk population: cohort follow-up. Cancer Res. 1990;50:4737–4740. [PubMed] [Google Scholar]
- 39.Michaud DS, et al. Genetic polymorphisms of interleukin-1B (IL-1B), IL-6, IL-8, and IL-10 and risk of prostate cancer. Cancer Res. 2006;66:4525–4530. doi: 10.1158/0008-5472.CAN-05-3987. [DOI] [PubMed] [Google Scholar]
- 40.Xu J, et al. The interaction of four genes in the inflammation pathway significantly predicts prostate cancer risk. Cancer Epidemiol. Biomarkers Prev. 2005;14:2563–2568. doi: 10.1158/1055-9965.EPI-05-0356. [DOI] [PubMed] [Google Scholar]
- 41.Pociot F, et al. A TaqI polymorphism in the human interleukin-1 beta (IL-1 beta) gene correlates with IL-1 beta secretion in vitro. Eur. J. Clin. Invest. 1992;22:396–402. doi: 10.1111/j.1365-2362.1992.tb01480.x. [DOI] [PubMed] [Google Scholar]
- 42.Canedo P, et al. The interleukin-8-251*T/*A polymorphism is not associated with risk for gastric carcinoma development in a Portuguese population. Eur. J. Cancer Prev. 2008;17:28–32. doi: 10.1097/CEJ.0b013e32809b4d0f. [DOI] [PubMed] [Google Scholar]
- 43.Eschrich S, et al. Molecular staging for survival prediction of colorectal cancer patients. J. Clin. Oncol. 2005;23:3526–3535. doi: 10.1200/JCO.2005.00.695. [DOI] [PubMed] [Google Scholar]
- 44.Shedden K, et al. Gene expression-based survival prediction in lung adenocarcinoma: a multi-site, blinded validation study. Nat. Med. 2008;14:822–827. doi: 10.1038/nm.1790. [DOI] [PMC free article] [PubMed] [Google Scholar]