Abstract
Objective
The aim was to evaluate the therapeutic potential of human skeletal muscle-derived myoendothelial cells for myocardial infarct repair.
Background
We have recently identified and purified a novel population of myoendothelial cells from human skeletal muscle. These cells co-express myogenic and endothelial cell markers and produce robust muscle regeneration when injected into cardiotoxin-injured skeletal muscle.
Methods
Myoendothelial cells were isolated from biopsies of human skeletal muscle using a fluorescence-activated cell sorter along with populations of regular myoblasts and endothelial cells. Acute myocardial infarction was induced in male immune-deficient mice, and cells were directly injected into the ischemic area. Cardiac function was assessed by echocardiography, and donor cell engraftment, angiogenesis, scar tissue, endogenous cardiomyocyte proliferation and apoptosis were all evaluated by immunohistochemistry.
Results
A greater improvement in left ventricular function was observed after intramyocardial injection of myoendothelial cells when compared to hearts injected with myoblast or endothelial cells. Transplanted myoendothelial cells generated robust engraftments within the infarcted myocardium, and also stimulated angiogenesis, attenuation of scar tissue and proliferation and survival of endogenous cardiomyocytes more effectively than transplanted myoblasts or endothelial cells.
Conclusions
Our findings suggest that myoendothelial cells represent a novel cell population from human skeletal muscle that may hold promise for cardiac repair.
Keywords: Human Stem Cells, Myocardial Infarction, Paracrine factors, Skeletal muscle, VEGF
Introduction
The number of patients suffering from severe heart failure after myocardial infarction is increasing in Western countries. As an alternative to heart transplantation, cellular cardiomyoplasty is a promising new approach for preventing advanced heart failure and restoring cardiac function in injured hearts. Researchers have investigated a variety of cell sources for cardiac repair (1-6). Initial studies in cell therapy for cardiac repair used committed skeletal myoblasts (SkMs), largely because they can be readily harvested and easily expanded in vitro prior to autologous transplantation. However, high cell-death rates were reported shortly after SkMs injection in both cardiac and skeletal muscles (7-11), which has hindered the therapeutic usefulness of SkMs. Many subsequent attempts have been made to identify and isolate alternative stem cells or progenitors from skeletal muscle that could have a greater capacity for post-transplantation cell survival and muscle regeneration (12).
We have previously isolated a population of skeletal muscle-derived stem cells (MDSCs) that adhere much slower to culture flasks than committed SkMs following enzymatic dissociation of murine skeletal muscle (13). When injected into the myocardium of infarcted hearts, MDSCs displayed excellent survival and engraftment, induced angiogenesis, and improved left ventricular function in a more effective manner when compared to the transplantation of SkMs (14). This distinct difference between mouse SkMs and MDSCs for MI repair indicated that skeletal muscle-derived progenitor cells are heterogeneous in therapeutic efficacy.
Although murine MDSCs display many characteristics associated with the myogenic lineage, these cells also have attributes typically associated with the endothelial lineage. Upon isolation, MDSCs expressed CD34, a marker characteristic of endothelial progenitor cells. In addition, these cells differentiated into endothelial cells and promoted angiogenesis either upon stimulation with vascular endothelial growth factor (VEGF) in culture or after transplantation into skeletal and cardiac muscles (13-15). Studies published by other investigators have documented the presence of myogenic progenitor cells that coexpress markers associated with the myogenic and endothelial lineages in the interstitial spaces of adult mouse skeletal muscle and in the dorsal aorta of mouse embryos (16,17). Together, these findings led us to the recent identification and isolation of progenitor cells residing within adult human skeletal muscle that also coexpress myogenic and endothelial cell markers (18). Transplantation of these myoendothelial cells into the injured skeletal muscles of immunodeficient mice resulted in considerably more de novo myofibers when compared to the injection of conventional myogenic and endothelial cells (18).
Here, we investigated the therapeutic potential of myoendothelial cells for cardiac repair using an acute myocardial infarction model created in immunodeficient mice. We show that myoendothelial cells provided greater therapeutic benefit after myocardial infarction than did endothelial cells, myogenic cells, and unsorted myoblasts. This therapeutic advantage may be collectively attributed to the high regenerative, angiogenic, anti-fibrotic, and cardio-protective and -stimulatory effects of myoendothelial cells.
Methods
Human Skeletal Muscle Cell Isolation
The procurement of human skeletal muscle biopsies (male, 21 and 23 yrs old, female, 26 yrs old) from The National Disease Research Interchange was approved by the Institutional Review Board at the University of Pittsburgh Medical Center. The human skeletal muscle biopsies were placed in solution with Hank's balanced salt solution (HBSS) and were transported to the laboratory on ice.
The human skeletal muscle biopsy was finely minced, and then digested for 60 minutes at 37°C with type I and type IV collagenase (100 u/mL) and dispase (1.2 u/mL; all from GIBCO, Invitrogen Corporation, Carlsbad, CA). The digested tissue was pelleted and resuspended in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin (P/S). The pellet was then enzymatically dissociated, and passed through a 40-μm filter to obtain a single- cell suspension. Approximately 7×105 cells were recovered per gram of tissue.
We used fluorescence-activated cell sorting (FACS) to isolate various cell fractions based on their expression of the cell surface markers CD56, CD34, and CD144, as previously described (18). We also used an unpurified (unsorted) cell population as a control. Briefly, the cell pellet was suspended in DMEM that was supplemented with 2% FBS. The cell solution was then incubated on ice with APC-Cy7-conjugated mouse anti-human CD45, APC-conjugated mouse anti-human CD34, PE-Cy7-conjugated mouse anti-human CD56 (all from BD Biosciences, San Jose, CA), and PE-conjugated mouse anti-human CD144 (Beckman Coulter, Fullerton, CA). Isotype control antibodies were APC-Cy7-, APC-, PE-Cy7- and PE-conjugated mouse IgG1 (all from BD Biosciences, San Jose, CA). 7-amino-actinomycin D (7-AAD, ViaProbe; BD Pharmingen, San Jose, CA) was added to each tube for dead cell exclusion. Background staining was evaluated with isotype-matched control antibodies and a CompBeads set (Becton-Dickinson, San Jose, CA) was used to optimize fluorescence compensation settings for multi-color analyses and sorts. A minimum of 1×105 live cell events were analyzed using a FACSCalibur flow-cytometer (Becton-Dickinson, San Jose, CA) and the CellQuest™ software (Becton-Dickinson). Cell sorting was performed on a FACSAria™ dual-laser fluorescence cell sorter (Becton-Dickinson, San Jose, CA). Sorted cells were re-analyzed in all experiments. For this study, we collected the following populations: CD56+CD34-CD144-, CD56-CD34+CD144+, and CD56+CD34+CD144+. The 3 sorted cell populations were plated in collagen-coated 96-well plates at a density of 500 cells per well in proliferation medium consisting of DMEM, 10% (v/v) FBS, 10% (v/v) horse serum, 1% (v/v) penicillin/streptomycin, 1% (v/v) chick embryo extract (GIBCO-BRL, Carlsbad, CA) and incubated at 37°C in a 5% CO2 atmosphere. At 70% confluence, cells were passage dissociated with trypsin/EDTA (GIBCO-BRL, Carlsbad, CA), replated at a cell density of 1.0-2.5×103/cm2 cells and cultured for 3 to 4 weeks prior to intracardiac transplantation.
To track donor cell fate after injection, we transduced myoendothelial cells with a retrovirus encoding for a nuclear LacZ (nLacZ) reporter gene as previously described (15,18).
Intramyocardial Cell Transplantation into an Acute MI
The Institutional Animal Care and Use Committee, Children's Hospital of Pittsburgh, approved the animal and surgical procedures performed in this study (Protocol 37/04). A total of 74 male NOD/SCID mice (The Jackson Laboratory, Bar Harbor, ME, USA) were used for this study. Overall, 5 animals died during surgery, resulting in a 6.8% operative mortality. One animal was excluded from this study due to the presence of a thymic lymphoma, which is known to naturally occur on occasion in this mouse strain (19). Infarcted mice were randomly allocated between the types of treatment (saline, CD56+CD34-CD144-, CD56-CD34+CD144+, and CD56+CD34+CD144+ cells) for each donor. The 3 sorted cell populations and unsorted cells were injected (3×105 cells in solution of 30 μL of PBS per heart) into the hearts of mice immediately after inducing myocardial infarction as previously described (14). In control mice, 30 μL of phosphate buffered saline (PBS) alone was injected. The investigator creating the infarction injury and injecting the cells was blinded to the contents of the injectant. Histological and functional studies were performed at 5 days and 2 and 6 weeks post-transplantation (N=14 animals sacrificed at 5 days, 25 animals sacrificed at 2 weeks, and 29 animals sacrificed at 6 weeks). Echocardiography was performed by a blinded investigator to assess the heart function of anesthetized mice, as previously described (14).
Histological analysis
The mice were sacrificed at 5 days, 2 weeks and 6 weeks after cell transplantation for histological evaluation of the heart tissue. Hearts were flash frozen in 2-methylbutane (Sigma) that was pre-cooled in liquid nitrogen. For immunohistochemical assays, some excised hearts were fixed in 10% formalin by perfusion from the ascending aorta and embedded in paraffin.
To detect proliferating human cells, cryosections were incubated with human anti-human proliferating cell nuclei antigen (1:400, PCNA, US Biological, Swampscott, MA), at room temperature for 2 hrs followed by the addition of goat anti-human IgG 555 (1:800, Molecular Probes, Eugene, OR). Then, the sections were co-stained for fskMyHC as described previously (14,15). Sections containing the highest number of fibers expressing fskMyHC were used, for each group, to determine a regeneration index, defined as the number of fskMyHC-positive fibers per section (14,15).
For immunofluorescent human VEGF staining, cryosections were incubated with Xgal substrate for 2 hours and incubated overnight at 4°C with a goat anti-human VEGF (1:100, R&D systems, Minneapolis, MN), followed by incubation with a donkey anti-goat IgG 555 (1:200, Molecular Probes, Eugene, OR).
To determine whether donor cells expressed cardiac cell markers, tissue samples were incubated with Xgal substrate for 2 hours. Then the sections were incubated with mouse anti-human cardiac troponin T (1:50, Abcam, Cambridge, MA), followed by donkey-anti-mouse IgG 488 (1:400, Molecular Probes, Eugene, OR). Alternatively, Xgal-stained slides were incubated with anti-cardiac troponin I at room temperature for 2 hrs followed by the addition of donkey anti-goat IgG 555 (1:200, Molecular Probes, Eugene, OR).
To determine capillary density within the infarct zone, we performed immunostaining of CD31 on cryosections and measured capillary density within the infarct region of each heart, as previously described (14).
For Ki-67 immunohistochemical staining, deparaffinized sections were immersed in preheated sodium citrate buffer and were incubated overnight at 4°C with a rat monoclonal anti-mouse Ki-67 antibody (1:50, DAKO, Carpinteria, CA) that does not cross-react with human Ki-67. Positive reactivity was determined with Vectastain® Elite ABC Kit (Vector Laboratories, Burlingame, CA) and visualized as purple with the VIP (violet-colored precipitate) substrate kit (Vector Laboratories). To determine the number of proliferating myocytes, tissues were counterstained with anti-cardiac troponin I (cTnI, 1:20,000, Scripps, San Diego, CA), processed with Vectastain® Elite ABC Kit (Vector), and then visualized as brown through the DAB (3,3′- diaminobenzidine) substrate kit (Vector). The number of mitotically active endogenous cardiomyocytes within the peri-infarct region was measured in 8 high power fields (HPF, 400× magnification).
For TUNEL staining, the deparaffinized sections were digested with proteinase K following the protocols provided by manufacturer (ApopTag® Plus Peroxidase In Situ Apoptosis Detection Kit; Chemicon, Temecula, CA). The TUNEL stain was visualized with a substrate system that stained purple (VIP substrate kit; Vector Laboratories, Burlingame, CA). The samples were also lightly counterstained with anti-cardiac troponin I as described above with the Ki-67 stain. The number of apoptotic endogenous cardiomyocytes within the peri-infarct region was measured in 4 HPF (400× magnification) (20).
For human specific VEGF immunohistochemical staining, deparaffinzed sections were immersed in preheated sodium citrate buffer and were incubated overnight at 4°C with a goat anti-human VEGF (1:50, R&D systems, Minneapolis, MN). The slides were then processed with Vectastain® Elite ABC Kit (Vector). A positive stain was visualized as brown using the DAB substrate kit (Vector). These sections were counterstained with eosin.
We used Masson's trichrome staining kit (San Marcos, CA) to stain collagen deposition on frozen cryosections of the heart. The area of infarct scar and the area of the entire cardiac muscle section were measured using a digital image analyzer (Image J). Scar area fraction was calculated as the ratio of scar area to the entire cardiac muscle area and was averaged from 7 sections per heart.
Hypoxia assay
We cultured all three cell populations under hypoxic conditions (2.5% O2) in vitro and measured the amount of VEGF that was secreted into the cell culture supernatant by ELISA (R&D Systems, Minneapolis, MN), as previously described (21). Total RNA (n=3) was extracted from cell lysates using an RNeasy kit with DNase I digestion (Qiagen, Valencia, CA). After RNA extraction, quantitative real-time PCR (qPCR) analysis was carried out as 10 μL reactions as described previously (22,23) using TaqMan® One-Step RT-PCR Master Mix (Applied Biosystems Inc (ABI), Foster City, CA) and 1 μL total RNA as the template. Sequences for target gene primers and probes (IDT) were as follows:
Human Vegf165 (NM 001033756) – forward: 5′ CATGCAGATTATGCGGATCAA 3′;
Reverse: 5′ TTTGTTGTGCTGTAGGAAGCTCAT 3′ and TaqMan® probe: 5′ CCTCACCAAGGCCAGCACATAGGAGA 3′;
Human Igf-I (X57025): forward-5′TGTATTGCGCACCCCTCAA3′;
Reverse: 5′TGCGTTCTTCAAATGTACTTCCTT3′; and TaqMan® probe: 5′ CAGCTCGCTCTGTCCGTGCCC 3′.
Human hgf - forward AGGCCATGGTGCTATACTCTTG
Reverse: TCAGCGCATGTTTTAATTGCA and TaqMan® probe: CCCTCACACCCGCTGGGAGTA (5′ 6FAM and 3′ TAMRA-Sp)
Human b-fgf - forward: GGCGTGTACATGTGGTCTCAGA
Reverse: TTATGGCTCACTGCAACCTTGA and TaqMan® probe: AAGTGATCAACCCTCCCACCTCAGCCT (5′ 6FAM 3′ TAMRA-Sp).
All target genes were normalized to the reference housekeeping gene 18S (ABI). Gene expression levels were analyzed using SDS 2.2 Software (ABI) and were calculated as total amount RNA (in arbitrary fluorescence units) compared to negative controls (normal O2) for each time point based on the comparative ΔCT method (separate tubes)(24).
Statistical analysis
All measured data are presented as mean ± standard error (SE). Kaplan-Meier survival curve estimation with log-rank test was performed to compare the survival rate among experimental groups. For the analysis of the echocardiographic data, the scar tissue area data, the endogenous cardiomyocyte proliferation data, and the VEGF qPCR and ELISA data, we applied the two-way analysis of variance (ANOVA) test and the Tukey multiple comparison test. The one-way ANOVA and the Tukey post-hoc tests were performed to analyze the number of fskMyHC expressing myocytes, capillary density data in all groups. For the endogenous cardiomyocyte apoptosis data, we applied the Kruskal-Wallis and the Dunn's multiple comparison tests at each time point. The Student's t-test was performed for the VEGF and HGF gene expression data in myoendothelial cells cultured under normoxic and hypoxic culture conditions. Statistical significance was set at P<0.05. All statistical tests were performed using SigmaStat (Systat Software Inc, Point Richmond, CA).
Results
Cell Isolation by FACS
Cells were isolated from 3 samples of adult human skeletal muscle [(1) male, age 21 years; (2) male, age 23 years; (3) female, age 26 years] by FACS on the basis of their differential expression of the myogenic (CD56) and endothelial cell (CD34 and CD144) surface markers (18). CD45 was used to exclude hematopoietic cells (18). The fraction of myoendothelial cells (CD56+CD34+CD144+CD45-) represented 1.8% of the total cell population, while endothelial cells (CD56-CD34+CD144+CD45-) and myogenic cells (CD56+CD34-CD144-CD45-) accounted for 9.0% and 2.6%, respectively. The mean numbers of viable sorted cells which were recovered per experiment were 9.6×104 CD56+CD34-CD144-CD45- cells, 45.8×104 CD56-CD34+CD144+CD45- cells, and 8.8×104 CD56+CD34+CD144+CD45- cells. Purities of these 3 sorted cell populations were, respectively, 89.1%, 92.8% and 81.1%, as confirmed by flow re-analysis. The myogenic, endothelial and myoendothelial cell isolates were expanded in culture prior to intramyocardial transplantation, using a protocol previously described (18).
We have previously evaluated the myogenic and endothelial cell phenotype of sorted cells after cultivation using the myogenic marker CD56 and the endothelial cell markers CD146 and Ulex europeaus agglutinin 1 receptor (UEA-1-R)(18). The phenotype of the sorted endothelial cells (CD56-CD34+CD144+) and myoendothelial cells (CD56+CD34+CD144+) changed after expansion in tissue culture flasks. Myogenic cells (CD56+CD34-CD144-) retained a high level of CD56 expression but a portion of cells also expressed CD146 after cultivation. Interestingly, 65% of cultured endothelial cells expressed CD56 even though this population was 92.8% pure (CD56-CD34+CD144+) after FACS isolation. A large portion of cultured myoendothelial cells retained a myogenic and endothelial cell phenotype. Expression of UEA-1-R was low in all cell types.
Cardiac Repair
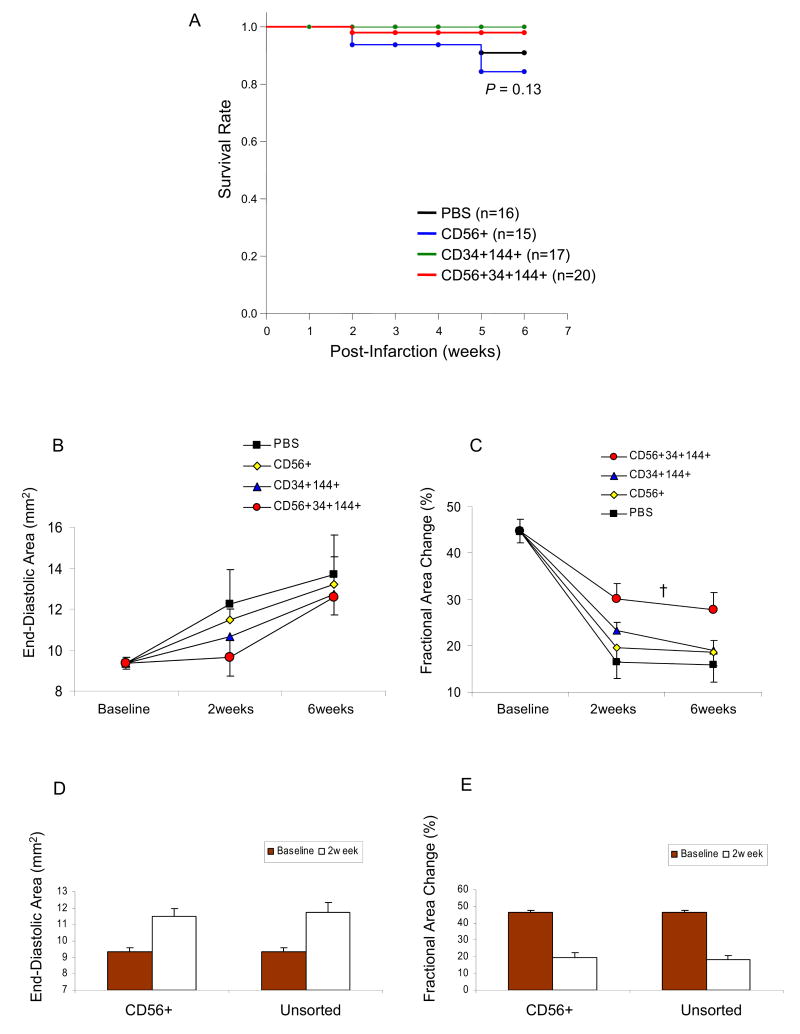
The therapeutic effect of myogenic, endothelial, and myoendothelial cells for cardiac repair was assessed in an immunodeficient mouse model of an acute myocardial infarction (MI). Over the course of this study, we did not observe a significant difference in survival rates among the groups (P=0.13; Kaplan-Meier Survival Curve, Figure1A). In uninjured NOD/SCID mice (age 12 weeks, weight 27.2 ± 1.4 g), we have measured baseline LV dimensions and function by echocardiography. Baseline end-diastolic area (EDA) of uninjured mice (n=5 mice) was 9.3 ± 0.3 mm2 (Figure 1B), and baseline fractional area change (FAC) was 46.4 ± 1.5 % (n=5, Figure 1C). The hearts in all 3 cell groups had smaller left ventricular (LV) dimensions, as assessed by end diastolic area (EDA, Figure 1B) and end systolic area (ESA, data not shown), when compared to the control group, which consisted of phosphate-buffered saline (PBS) injections into the heart. Compared to baseline uninjured hearts and control PBS-injected hearts (12.2 ± 1.5 mm2), intracardiac injections of myoendothelial cells (9.6 ± 0.8 mm2) tended to preserve LV dimensions at 2 weeks after cell transplantation. However, by 6 weeks, the LV expanded in hearts injected with myoendothelial cells (12.6 ± 0.8 mm2), and was only slightly less than PBS-injected hearts (13.8 ± 1.7 mm2; Figure 1B).
Figure 1. Survival rates and echocardiographic assessment.
(A) Survival rates over 6weeks after cell transplantation in the animals injected with human muscle derived cells or control-PBS (Kaplan-Meier Survival Curve, P=0.13). (B) Echocardiography performed 2 and 6 weeks after cell transplantation demonstrated smaller left ventricular (LV) areas in end-diastole of hearts injected with CD56+CD34+CD144+ cells when compared to the other groups. (Baseline: data from NOD/SCID mice without infarction and cellular injection.) (C) The CD56+CD34+CD144+ myoendothelial cell group also displayed greater LV contractility, as measured by fractional area change (FAC), when compared to the other groups at both the 2 and 6 weeks time points († P<0.05, CD56+CD34+CD144+ cells versus CD56+CD34-CD144- cells and PBS). (D) No difference in LV end-diastolic areas was observed between hearts injected with unsorted myoblasts and hearts injected with myogenic CD56+ sorted cells 2 weeks after cell transplantation. (E) The unsorted group also displayed a similar level of LV contractility, as measured by fractional area change (FAC), as the CD56+ cell group.
LV contractility, as determined by fractional area change (FAC) values, improved 20% and 17% at 2 and 6 weeks after MI, respectively, in the myogenic group (2 weeks: FAC 19.6 ± 2.9%, 6 weeks: 18.5 ± 2.4%) compared to the control PBS group (2 weeks: 16.4 ± 2.6%, 6 weeks: 15.9 ± 3.1%) (Figure 1C). Transplanted endothelial cells (2 weeks: 23.4 ± 1.4%, 6 weeks: 19.0 ± 1.9%) improved systolic function by 47% and 25% at 2 and 6 weeks, respectively, compared to control injections of PBS (Figure 1C). In comparison to the other 2 cell groups, LV contractility was the strongest in the myoendothelial group (2 weeks: 30.0 ± 3.0%, 6 weeks: 27.8 ± 3.3%) resulting in FAC values that were 84% and 75% greater than the control PBS group at the 2 and 6 week time points, respectively (Figure 1C; P<0.05, myoendothelial cells versus myogenic cells and PBS).
We performed injections using unsorted human myoblasts isolated from the same donor (Figure 1D and E). No difference in LV dimensions was observed between the unsorted myoblasts (EDA: 11.7 ± 0.6 mm2) and sorted CD56+ myogenic cells (11.5 ± 0.5 mm2; Figure 1D). Hearts injected with unsorted human myoblasts displayed poor LV contractility (FAC: 18.3 ± 2.1%) at 2 weeks after MI, which was comparable to hearts injected with the sorted CD56+ myogenic cell population (CD56+CD34-CD144-, FAC 19.6 ± 2.9%, Figure 1E).
Injection of all 3 sorted cell populations was repeated using cell isolates from another donor (male, 21 years old). Again, hearts injected with myoendothelial cells (FAC: 37.4 ± 1.9%) displayed the strongest LV contraction compared to the other groups at 2 weeks after MI (P<0.05, myoendothelial cells vs. myogenic cells, endothelial cells, and control PBS). In comparison to the injection of PBS only (23.0 ± 1.9%), cardiac contractility improved 62% with the injection of myoendothelial cells, 28% with the injection of endothelial cells (28.7 ± 2.8%), and 7% with the injection of myogenic cells (24.6 ± 1.3%).
Scar Tissue Formation
The extent of scar tissue formation within the infarcted area was evaluated using Masson's trichrome stain (Figure 2A). At both time points (2 weeks and 6 weeks), LV infarct scar area was smaller, as determined by scar area fraction values, in all 3 cell groups when compared to the control PBS group, with the most notable improvement occurring at the 6 week time point (Figure 2B). Hearts injected with myoendothelial cells exhibited less scar tissue than those injected with PBS 2 and 6 weeks after infarction (P<0.05, myoendothelial cells versus PBS, Figure 2B).
Figure 2. Injection of myoendothelial cells reduces scar tissue formation after myocardial infarction.
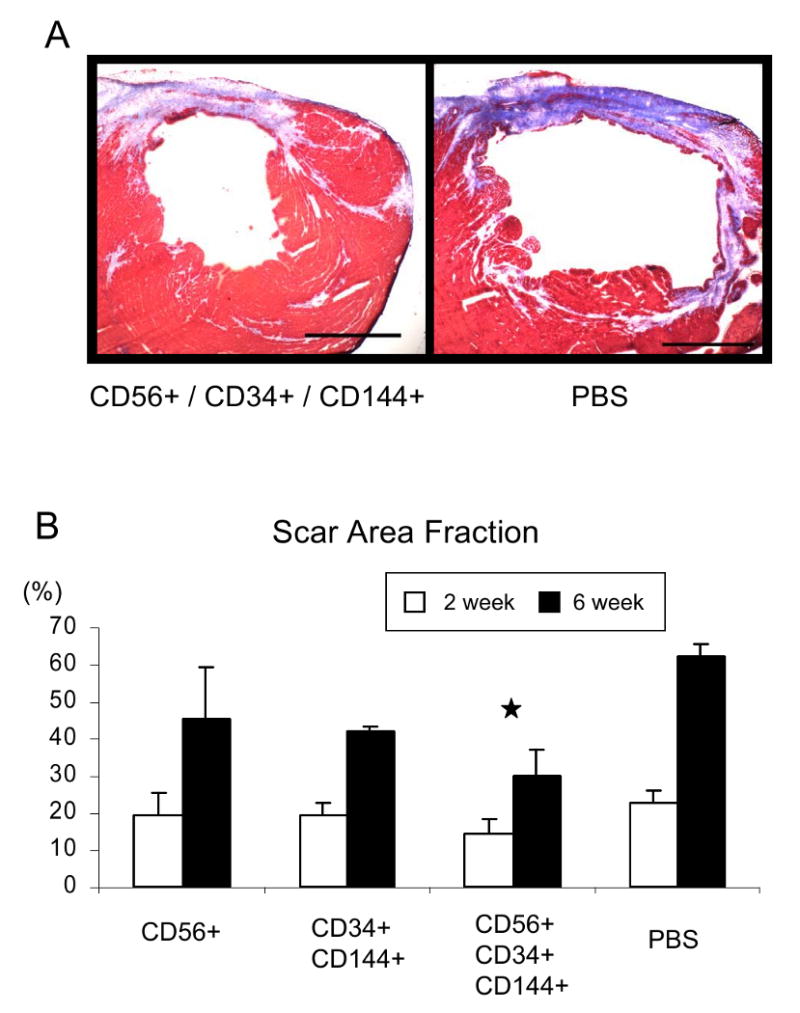
(A) Representative images taken from transverse sections of the left ventricle stained by Masson's trichrome (muscle is stained red, collagen blue, nuclei black). The hearts injected with CD56+CD34+CD144+ cells displayed smaller infarct scar areas than did the control hearts injected with PBS only. Scale bars equal 1 mm. (B) Compared to all groups, the CD56+CD34+CD144+ cell-injected hearts had the smallest scar tissue area 2 and 6 weeks after infarction (* P<0.05 CD56+CD34+CD144+ cells versus PBS).
Engraftment
Cell engraftment was assessed by immunohistochemical analysis of the skeletal muscle-specific marker fast skeletal myosin heavy chain (fskMyHC, green, Figure 3B). The majority of engrafted human cell-derived myocytes had aligned with the host myocardial fibers and along the ischemic border zone and scar area (Figure 3A, B). Interestingly, the CD56+ myogenic cells regenerated less fskMyHC+ myofibers when compared to the CD34+CD144+ endothelial cells (Figure 3C). Compared to both of these groups, the CD56+CD34+CD144+ myoendothelial cells regenerated significantly more skeletal myofibers (P<0.05, myoendothelial cells versus myogenic cells, endothelial cells; Figure 3C), indicating that these cells have an efficient transplantation capacity, as observed in skeletal muscle (18). Heart sections were also co-stained with antibodies directed against human-specific proliferating cell nuclear antigen (PCNA) and fskMyHC (Figure 3D). A few nuclei (blue) in the fskMyHC engraftment region (green) expressed PCNA (red). This finding confirms the presence of human cells within the fskMyHC+ engraftment region, and suggests that a fraction of myoendothelial cells are proliferative for up to 2 weeks following implantation.
Figure 3. Engraftment of human muscle-derived cells in the infracted heart.
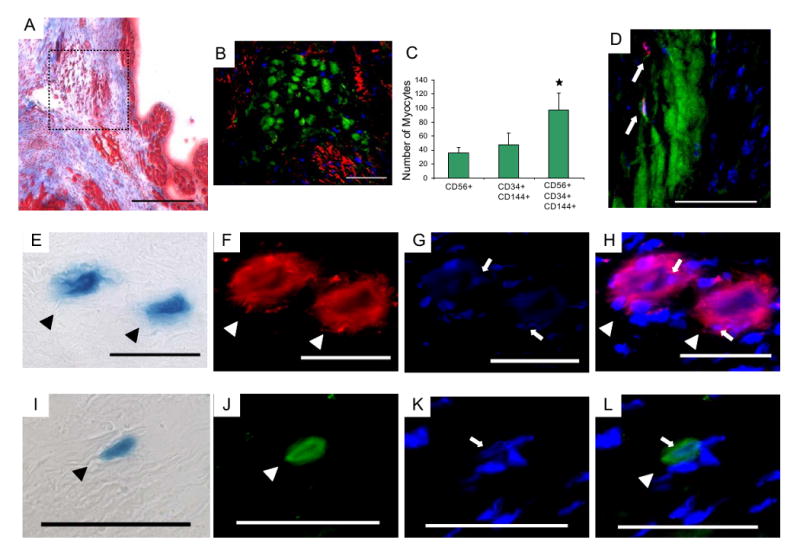
(A) Masson's trichrome staining (muscle is stained red, collagen blue, nuclei black) at the peri-infarcted area of CD56+CD34+CD144+ cell-injected heart 2 weeks after cell transplantation. Scale bar equals 500 μm. The dotted line shows the engraftment area of human muscle-derived cells, (B) which corresponds to fast skeletal myosin heavy chain (fskMyHC)-positive (green) immunostaining. In the image, the fskMyHC-postive myofibers are stained green, nuclei blue, and cTnI-positive cardiomyocytes red. Only the engraftment regions expressed fskMyHC; normal myocardium was negative for fskMyHC. Scale bar equals 50 μm. (C) CD56+CD34+CD144+ myoendothelial cells regenerated more fskMyHC expressing myocytes than did CD56+CD34-CD144- myogenic and CD56-CD34+CD144+ endothelial cells (* P<0.05). (D) A few nuclei (blue) found in the fskMyHC-positive (green) engraftment area stained positive for human-specific proliferating cell nuclear antigen (PCNA) (red, arrows). Scale bar equals 33.3 μm. (E)-(L) Expression of a cardiac cell markers by myoendothelial cells in vivo. (E) Engrafted myoendothelial cells expressing the nLacZ reporter gene (blue, arrowheads) colocalized with (F) cardiac troponin I (red, arrowheads) staining. DAPI-only (G, blue, arrow) and colocalized with troponin I (H, red and blue). (I) A nLacZ-expressing donor cell (blue, arrowhead) colocalized with the (J) cardiac troponin T marker (green, arrowhead). DAPI-only (K, blue, arrow) and colocalized with troponin T (L, green and blue). Scale bars equal 50 μm.
Evaluation of Cardiac Differentiation
In hearts injected with nLacZ-transduced myoendothelial cells, we evaluated whether any donor cells colocalized with the cardiac-specific isoforms of troponin I (cTnI) and troponin T (cTnT) and the gap junction protein connexin43, a protein essential for intercellular electrical connections between cardiomyocytes (14,15).
We observed only a few cells containing nLacZ+ nuclei that colocalized with cTnI (Figure 3E-H) and cTnT (Figure 3I-L). However, the vast majority of nLacZ+ cells did not colocalize with these cardiac markers. In addition, we did not observe expression of connexin43 at the interface of the graft and host myocardium, suggesting that engrafted cells were electrically isolated from host cardiomyocytes. Connexin43 gap junction proteins were not expressed by donor cells within the graft.
Neoangiogenesis
Capillary density within the infarct tissue was assessed by CD31 [PECAM] immunostaining (red, Figure 4A). Intramyocardial transplantation of all 3 cell populations resulted in an increase in the number of CD31+ capillaries when compared to the transplantation of PBS only (P<0.05, myoendothelial cells, myogenic cells, and endothelial cells versus PBS; Figure 4B). Of the 3 cell types, the myoendothelial cells induced the greatest increase of capillaries in the infarct area (P<0.05, myoendothelial cells versus myogenic cells and endothelial cells; Figure 4B), which was followed by endothelial cells and myogenic cells, respectively. These findings suggest that each transplanted cell population had an angiogenic effect; however, myoendothelial cells were the most effective at stimulating angiogenesis.
Figure 4. Transplanted human cells induce neoangiogenesis in the infracted heart.
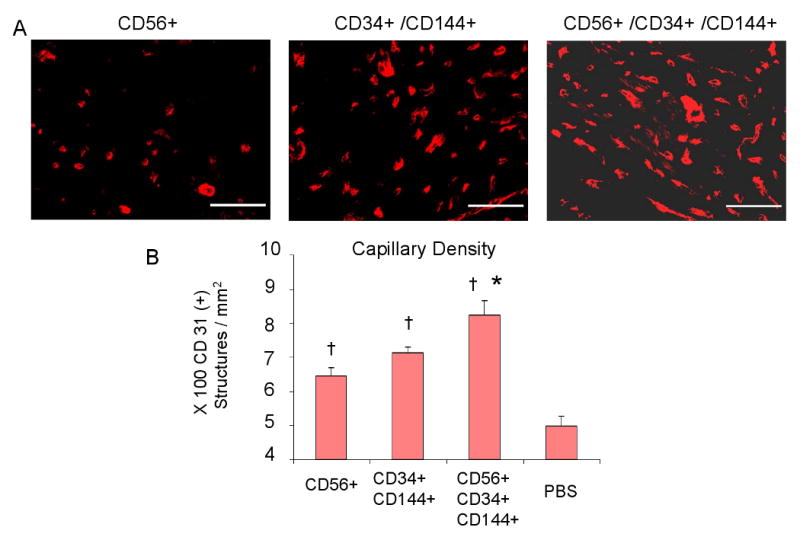
(A) Representative images of CD31 immunostaining in the peri-infarct and the infarct regions of hearts transplanted with CD56+CD34-CD144- cells, CD56-CD34+CD144+ cells, and CD56+CD34+CD144+ cells. Scale bars equal 100 μm. (B) Hearts transplanted with CD56+CD34+CD144+ cells display a higher capillary density within the infarct when compared to the other groups (*P<0.05, CD56+CD34+CD144+ versus CD56+CD34-CD144- and CD56-CD34+CD144+; †P<0.05, all cell groups versus PBS).
Endogenous Cardiomyocyte Proliferation and Apoptosis
We have assessed host cardiomyocyte proliferation and apoptosis at 5 days and 6 weeks after MI. Five days after MI, we observed numerous proliferating endogenous cells located at the infarct border zone. We observed no difference among the groups for the total number of ki-67–positive cells at the peri-infarct zone (myoendothelial cells 68 ± 10 ki-67+ cells, n=3 hearts; endothelial cells 63 ± 16, n=4; myogenic cells 63 ± 12, n=3; saline control 60 ± 5, n=4; P=0.859). We also measured the number of cells that co-expressed both ki-67 and cardiac troponin I (cTnI) within the peri-infarct region (Figure 5A, B, and C). Five days after cell transplantation, hearts injected with myoendothelial cells had more proliferating ki-67(+)/cTnI(+) cardiomyocytes (13 ± 4 cardiomyocytes) within the infarct border zone than hearts injected with either endothelial cells (3 ± 1), myogenic cells (2 ± 1), or saline (3 ± 1) (P<0.05, myoendothelial cells versus myogenic cells, endothelial cells, and PBS; Figure 5C). Six weeks after cell transplantation, in hearts injected with CD56+ myogenic cells (1 ± 1) and CD34+CD144+ endothelial cells (2 ± 1), the number of proliferating endogenous cTnI+ cardiomyocytes was comparable to control hearts injected with PBS (1 ± 1) 6 weeks after MI (Figure 5C). However, endogenous cardiomyocyte proliferation was the highest in hearts injected with myoendothelial cells (6 ± 1) (P<0.05, myoendothelial cells versus myogenic cells, endothelial cells, and PBS; Figure 5C).
Figure 5. Endogenous cardiomyocyte proliferation and apoptotis.

(A), (B) Proliferating cardiomyocytes within the peri-infarct region were identified by colocalization of both ki-67 (purple nuclear stain, arrow) and cardiac troponin I (cTnI, brown cytoplasmic stain) within the peri-infarct region (arrow). Some of the proliferating cells stained by Ki-67 exist within the infarct zone, but do not colocalize with cTnI (arrow head). Scale bars equal 50 μm. (C) The number of endogenous proliferating cardiomyocytes was higher in the peri-infarct region of the heart injected with CD56+CD34+CD144+ cells compared with other groups at both 5 days and 6 weeks after cell transplantation (*P<0.05, CD56+CD34+CD144+ cells versus CD56+CD34-CD144- cells, CD56- CD34+CD144+ cells, and PBS). (D) The number of apoptotic cardiomyocytes was measured at both 5 days and 6 weeks after MI (†P< 0.05, CD56+CD34+CD144+ cells versus PBS).
We performed multi-label staining for DNA end-labeling by terminal dUPT nick end-labeling (TUNEL) and cTnI to determine the effect of cell therapy on endogenous cardiomyocyte apoptosis in the peri-infarct regions. At 5 days after MI, the occurrence of apoptotic cardiomyocytes within the peri-infarct regions was less frequent in hearts injected with myoendothelial cells (7 ± 1 TUNEL+ cardiomyocytes) when compared to the transplantation of PBS (27 ± 6), myogenic cells (20 ± 3) and endothelial cells (14 ± 2) (P<0.05, myoendothelial cells versus PBS, Figure 5D). By six weeks after cell transplantation, the number of apoptotic cardiomyocytes decreased considerably. Hearts injected with all 3 cell types had slightly fewer apoptotic cardiomyocytes in the peri-infarct regions when compared to the transplantation of PBS only (P<0.05 myoendothelial cells versus PBS, Figure 5D). Taken together, these results suggest that the transplantation of myoendothelial cells had a beneficial effect on endogenous cardiomyocyte proliferation and apoptosis, with the most marked effect occurring shortly after implantation.
Analysis of Paracrine Factors
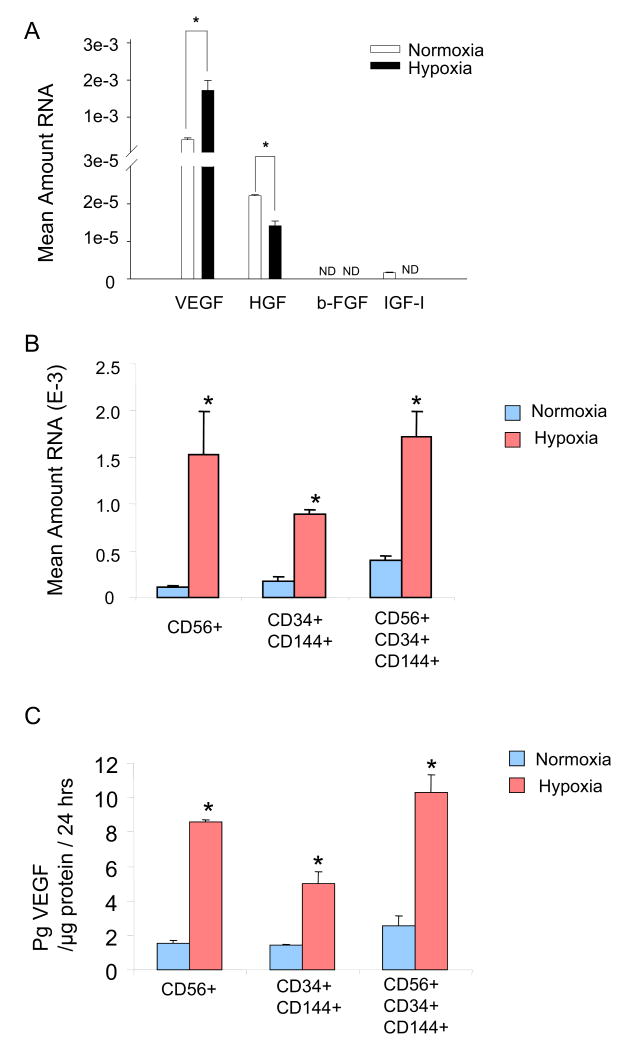
For all 3 populations, we used quantitative real-time PCR (qPCR) to measure the expression of secretable factor genes, including vascular endothelial growth factor (Vegf), hepatocyte growth factor (Hgf) (25-27), insulin-like growth factor-I (Igf-I), and basic fibroblast growth factor (b-Fgf). All of these factors are known to act as therapeutic agents in the heart (28,29). Under normal culture conditions, we observed expression of Vegf, Hgf, and Igf-I genes from myoendothelial cells of 1st donor sample. There was no detectable expression of the b-Fgf gene (Figure 6A). Interestingly, while the Hgf gene was expressed in CD56+CD34+CD144+ cells (myoendothelial), we did not observe measurable expression in CD56+CD34-CD144- (myogenic) and CD56-CD34+CD144+ (endothelial) cells (data not shown). Hypoxic conditions stimulated a 4.4-fold increase in Vegf gene expression by myoendothelial cells. In contrast, the expression of Hgf and Igf-1 genes decreased upon exposure to hypoxia, and b-Fgf gene expression remained undetectable (Figure 6A). Of note, Vegf gene expression was considerably higher when compared to other analyzed factors (Figure 6A), suggesting that VEGF may represent a major factor secreted by myoendothelial cells, as previously observed with murine MDSCs (21). All 3 populations increased Vegf gene expression under hypoxic conditions relative to normoxia (P<0.01, normoxia versus hypoxia, Figure 6B). The VEGF mRNA results were further confirmed by ELISA where all tested cell types had at least a 4-fold increase in VEGF protein secretion under hypoxic conditions when compared to control normoxic conditions (P<0.05, normoxia versus hypoxia, Figure 6C). Although only a few factors were analyzed here, our findings suggest that myoendothelial cells secrete factors, especially when subjected to ischemic conditions, which may have therapeutic effect in vivo.
Figure 6. Growth factor expression in vitro.
(A) An increase in Vegf mRNA production by CD56+CD34+CD144+ cells was observed when cultured under hypoxia for 24 hours in comparison to control normoxic conditions. In contrast, Hgf, b-Fgf, and Igf-I mRNA expression by human CD56+CD34+CD144+ cells either significantly decreased or was not detectable after 24 hours of hypoxia. (B) All cell types greatly increased their secretion of Vegf mRNA after exposure to hypoxic culture conditions for 24 hrs when compared to control normoxia culture conditions by quantitative PCR analysis of Vegf mRNA expression (*P<0.01, normoxia versus hypoxia). (C) These results were further confirmed by VEGF ELISA analysis (*P<0.05, normoxia versus hypoxia).
In Vivo VEGF Expression
We evaluated whether human VEGF was expressed in vivo. Using an antibody that is specific for human VEGF, we observed human VEGF expression within cell-injected hearts 5 days after MI (data not shown). Engrafted nLacZ-tranduced donor cells were found to colocalize with human VEGF (data not shown).
Discussion
Satellite cells, which are myogenic progenitor cells located beneath the basal lamina of skeletal myofibers, are known to reside in close proximity to capillaries (30). A developmental relationship between satellite cells and endothelial cells, besides their spatially close proximity in skeletal muscle, has been suggested by Tamaki and colleagues, who have documented the presence of myogenic progenitor cells that coexpress markers associated with the myogenic and endothelial lineages within the interstitial spaces of murine skeletal muscle (16). In addition, the identification of skeletal muscle progenitors originating from the embryonic dorsal aorta has led to the notion that a subset of satellite cells may have a vascular origin (17). Our research has resulted in the identification and isolation of progenitor cells residing within adult human skeletal muscle that coexpress myogenic and endothelial cell markers (18). These myoendothelial cells also demonstrated a potent regenerative capacity when transplanted into injured skeletal muscles (18), indicating that they may yield a better therapeutic promise than myoblasts.
The main finding in this study is that transplantation of human myoendothelial cells attenuated LV dysfunction more effectively than either myogenic or endothelial cells in an acute MI. At the 6 week time point, cardiac contractility in the myoendothelial cell-injected hearts was 40% and 50% greater compared to hearts injected with either endothelial cells or myogenic cells, respectively. The improved therapeutic benefit elicited by the injection of myoendothelial cells could be at least partially explained by the reduced levels of scar tissue and apoptotic cardiomyocytes and the increased levels of engraftment, angiogenesis, and endogenous cardiomyocyte proliferation when compared to that observed with myogenic and endothelial cells.
A key goal of cardiac cell transplantation for myocardial infarct repair is to modify LV remodeling in order to increase functional repair. Despite the fact that all 3 cell types did not prevent LV dilation compared to PBS controls, myoendothelial cells significantly reduced LV infarct size and sustained LV contractile function better than the other 2 cell types and PBS. It remains to be investigated if the beneficial effects of myoendothelial cells will reduce LV dilation at longer time points.
Our histological analysis demonstrated a higher level of engraftment based on the number of regenerated myofibers in myoendothelial cell-injected hearts when compared to the other cell-injected hearts. Because there is a correlation between the magnitude of functional improvement and donor cell engraftment (31-33), the large degree of engraftment of myoendothelial (CD56+CD34+CD144+) cells is a good indicator of their enhanced ability to improve cardiac function. This superior engraftment is likely related to the superior survival characteristics of myoendothelial cells, as observed in skeletal muscle (18) which could be due to their enhanced ability to resist oxidative stress compared to myogenic and endothelial cells. This same enhanced survival characteristic was also observed with murine MDSCs, which were found to be more resistant to oxidative stress than skeletal myoblasts (14).
A few of the injected myoendothelial cells were observed to express cardiac cell markers within the infarcted heart. The frequency of this event was quite rare as the vast majority of engrafted donor cells were negative for cTnI and cTnT. It is undetermined from these experiments whether these cardiac marker-expressing donor cells originated from differentiation or fusion with host cardiomyocyte (14,15) (34).
All 3 cell populations had an angiogenic effect upon transplantation, a finding that is consistent with many reports in the field (1,14,20,35-37). Neovascularization of the infarct may help to salvage at-risk myocardium by promoting endogenous cell survival and proliferation, stimulating hibernating myocardium, enhancing scar viability, and attenuating adverse remodeling (20). When we previously reported on the intramyocardial injection of MDSCs, we observed that most of the new vasculature that formed within the infarct was of host origin. The occurrence of transplanted murine MDSCs differentiating into blood vessels was rare (14,15), indicating that the presence of new vasculature within the infarct may be due to paracrine factors. Likewise, in this study, we found that only a few donor cells had incorporated into the neovasculature, as evidenced by our use of a human-specific CD31 antibody (data not shown), suggesting that the new capillaries are of host-origin and may be induced by paracrine factor released by the donor cells. In a recent study, we reported that one of the most potent angiogenic factors, VEGF, may be critical for the occurrence of angiogenesis in murine MDSCs-treated hearts (21). Overall, the ability of myoendothelial cells to induce the greatest level of angiogenesis within the infarct may be a major factor underlying their strong therapeutic effect.
Recent reports have demonstrated that a population of myocytes within the myocardium is capable of proliferating after infarction (38,39). Schuster et al. report that myocardial neovascularization results in regeneration and cell cycling of endogenous cardiomyocytes, suggesting that agents that increase myocardial homing of bone marrow angioblasts could induce endogenous cardiomyocytes to enter the cell cycle and improve functional cardiac recovery (40). We observed that mouse cardiomyocytes located within the peri-infarct region in animals receiving human myoendothelial cells demonstrated much higher mitotic activity than in those receiving PBS at 5days after cell transplantation. The origin of these proliferating cells remains unknown, but they could originate from resident cardiomyocytes, differentiating cardiac stem cells, or from circulating stem cells that home to the myocardium after infarction (39).
Additionally, TUNEL staining showed reduced levels of apoptosis, suggesting that myoendothelial (CD56+CD34+CD144+) cells transplantation was effective at reducing endogenous cardiomyocyte apoptosis at the initial phase after myocardial infarction.
Transplanted cells may have a beneficial paracrine effect in the heart by releasing growth factors that promote angiogenesis, cell proliferation, anti-apoptosis, anti-fibrotic effects, and cardioprotective effect (40,41). Although we only evaluated the expression of a few secretable factors, quantitative real-time PCR analysis showed that VEGF and HGF are two factors secreted by transplanted human muscle cells in response to the ischemic microenvironment of the infarcted hearts. We observed elevated HGF expression in myoendothelial (CD56+CD34+CD144+) cells but not in the other tested cell populations. It has been previously demonstrated that HGF can greatly increase myogenic regeneration and vascularity while reducing fibrosis inside the graft, which enhances the efficacy of SkM transplantation to infarcted hearts (25). Out of all the factors evaluated, VEGF was the only factor to significantly increase upon exposure to hypoxic culture conditions. Since the cells are transplanted into an ischemic environment, VEGF may be highly secreted by the donor cells and be a major factor in the induction of angiogenesis and promotion of endogenous cell survival (1,21,37). In addition, VEGF and HGF may have a synergistic effect resulting in a more robust proliferative, chemotactic, and angiogenic response than either growth factor alone (42,43).
Here, we have shown that human skeletal muscle-derived myoendothelial (CD56+CD34+CD144+) cells represent a unique cell population with a superior ability for myocardial infarct repair when compared to conventional myogenic or endothelial cells, as recently observed in skeletal muscle (18). Based on the results presented here, the beneficial therapeutic outcomes of myoendothelial cell transplantation, including the improvement of cardiac function, attenuation of adverse cardiac remodeling, cardioprotection of at-risk endogenous cardiac cells, and the stimulation of both infarct neovascularization and endogenous cell proliferation, may emanate from the robust engraftment of myoendothelial cells. In conclusion, myoendothelial cells represent a unique population of progenitor cells from human skeletal muscle that may be similar to murine MDSCs (14) in regards to the therapeutic repair of skeletal (18) and cardiac muscles.
Footnotes
After working with the University of Pittsburgh's Office of Technology Management and the Conflict of Interest Office it was determined that Dr. Johnny Huard (senior corresponding author on this paper) should declare the existence of a potential financial conflict of interest with the current study. It was also recommended that Dr. Thomas Payne should list his current affiliation as an employee of Cook Myosite, Inc., 105 Delta Drive, Pittsburgh, PA 15238. It should be noted that Dr. Morey Moreland acted as an independent data steward for this manuscript and concluded that “…there was no evidence that the study was objectively changed or influenced by him” [Dr. Huard].
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Suzuki K, Murtuza B, Smolenski RT, et al. Cell transplantation for the treatment of acute myocardial infarction using vascular endothelial growth factor-expressing skeletal myoblasts. Circulation. 2001;104:I207–12. doi: 10.1161/hc37t1.094524. [DOI] [PubMed] [Google Scholar]
- 2.Noiseux N, Gnecchi M, Lopez-Ilasaca M, et al. Mesenchymal stem cells overexpressing Akt dramatically repair infarcted myocardium and improve cardiac function despite infrequent cellular fusion or differentiation. Mol Ther. 2006;14:840–50. doi: 10.1016/j.ymthe.2006.05.016. [DOI] [PubMed] [Google Scholar]
- 3.Orlic D, Kajstura J, Chimenti S, et al. Bone marrow cells regenerate infarcted myocardium. Nature. 2001;410:701–5. doi: 10.1038/35070587. [DOI] [PubMed] [Google Scholar]
- 4.Yuasa S, Itabashi Y, Koshimizu U, et al. Transient inhibition of BMP signaling by Noggin induces cardiomyocyte differentiation of mouse embryonic stem cells. Nat Biotechnol. 2005;23:607–11. doi: 10.1038/nbt1093. [DOI] [PubMed] [Google Scholar]
- 5.Askari AT, Unzek S, Popovic ZB, et al. Effect of stromal-cell-derived factor 1 on stem-cell homing and tissue regeneration in ischaemic cardiomyopathy. Lancet. 2003;362:697–703. doi: 10.1016/S0140-6736(03)14232-8. [DOI] [PubMed] [Google Scholar]
- 6.Beltrami AP, Barlucchi L, Torella D, et al. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell. 2003;114:763–76. doi: 10.1016/s0092-8674(03)00687-1. [DOI] [PubMed] [Google Scholar]
- 7.Fan Y, Maley M, Beilharz M, Grounds M. Rapid death of injected myoblasts in myoblast transfer therapy. Muscle Nerve. 1996;19:853–60. doi: 10.1002/(SICI)1097-4598(199607)19:7<853::AID-MUS7>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- 8.Qu Z, Balkir L, van Deutekom JC, Robbins PD, Pruchnic R, Huard J. Development of approaches to improve cell survival in myoblast transfer therapy. J Cell Biol. 1998;142:1257–67. doi: 10.1083/jcb.142.5.1257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huard J, Verreault S, Roy R, Tremblay M, Tremblay JP. High efficiency of muscle regeneration after human myoblast clone transplantation in SCID mice. J Clin Invest. 1994;93:586–99. doi: 10.1172/JCI117011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Menasche P. Cellular transplantation: hurdles remaining before widespread clinical use. Curr Opin Cardiol. 2004;19:154–61. doi: 10.1097/00001573-200403000-00016. [DOI] [PubMed] [Google Scholar]
- 11.Tremblay JP, Malouin F, Roy R, et al. Results of a triple blind clinical study of myoblast transplantations without immunosuppressive treatment in young boys with Duchenne muscular dystrophy. Cell Transplant. 1993;2:99–112. doi: 10.1177/096368979300200203. [DOI] [PubMed] [Google Scholar]
- 12.Beauchamp JR, Morgan JE, Pagel CN, Partridge TA. Dynamics of myoblast transplantation reveal a discrete minority of precursors with stem cell-like properties as the myogenic source. J Cell Biol. 1999;144:1113–22. doi: 10.1083/jcb.144.6.1113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Qu-Petersen Z, Deasy B, Jankowski R, et al. Identification of a novel population of muscle stem cells in mice: potential for muscle regeneration. J Cell Biol. 2002;157:851–64. doi: 10.1083/jcb.200108150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Oshima H, Payne TR, Urish KL, et al. Differential myocardial infarct repair with muscle stem cells compared to myoblasts. Mol Ther. 2005;12:1130–41. doi: 10.1016/j.ymthe.2005.07.686. [DOI] [PubMed] [Google Scholar]
- 15.Payne TR, Oshima H, Sakai T, et al. Regeneration of dystrophin-expressing myocytes in the mdx heart by skeletal muscle stem cells. Gene Ther. 2005;12:1264–74. doi: 10.1038/sj.gt.3302521. [DOI] [PubMed] [Google Scholar]
- 16.Tamaki T, Akatsuka A, Ando K, et al. Identification of myogenic-endothelial progenitor cells in the interstitial spaces of skeletal muscle. J Cell Biol. 2002;157:571–7. doi: 10.1083/jcb.200112106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.De Angelis L, Berghella L, Coletta M, et al. Skeletal myogenic progenitors originating from embryonic dorsal aorta coexpress endothelial and myogenic markers and contribute to postnatal muscle growth and regeneration. J Cell Biol. 1999;147:869–78. doi: 10.1083/jcb.147.4.869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zheng B, Cao B, Crisan M, et al. Prospective identification of myogenic endothelial cells in human skeletal muscle. Nat Biotechnol. 2007;25:1025–34. doi: 10.1038/nbt1334. [DOI] [PubMed] [Google Scholar]
- 19.Prochazka M, Gaskins HR, Shultz LD, Leiter EH. The nonobese diabetic scid mouse: model for spontaneous thymomagenesis associated with immunodeficiency. Proc Natl Acad Sci U S A. 1992;89:3290–4. doi: 10.1073/pnas.89.8.3290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kocher AA, Schuster MD, Szabolcs MJ, et al. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med. 2001;7:430–6. doi: 10.1038/86498. [DOI] [PubMed] [Google Scholar]
- 21.Payne TR, Oshima H, Okada M, et al. A relationship between vascular endothelial growth factor, angiogenesis, and cardiac repair after muscle stem cell transplantation into ischemic hearts. J Am Coll Cardiol. 2007;50:1677–84. doi: 10.1016/j.jacc.2007.04.100. [DOI] [PubMed] [Google Scholar]
- 22.Jadlowiec JA, Zhang X, Li J, Campbell PG, Sfeir C. Extracellular Matrix-mediated Signaling by Dentin Phosphophoryn Involves Activation of the Smad Pathway Independent of Bone Morphogenetic Protein. J Biol Chem. 2006;281:5341–7. doi: 10.1074/jbc.M506158200. [DOI] [PubMed] [Google Scholar]
- 23.Jadlowiec J, Koch H, Zhang X, Campbell PG, Seyedain M, Sfeir C. Phosphophoryn regulates the gene expression and differentiation of NIH3T3, MC3T3-E1, and human mesenchymal stem cells via the integrin/MAPK signaling pathway. J Biol Chem. 2004;279:53323–30. doi: 10.1074/jbc.M404934200. [DOI] [PubMed] [Google Scholar]
- 24.Biosystems. User Bulletin #2 ABI Prism 7700 Sequence Detection System. Applied Biosystems; 2001. [Google Scholar]
- 25.Tambara K, Premaratne GU, Sakaguchi G, et al. Administration of control-released hepatocyte growth factor enhances the efficacy of skeletal myoblast transplantation in rat infarcted hearts by greatly increasing both quantity and quality of the graft. Circulation. 2005;112:I129–34. doi: 10.1161/CIRCULATIONAHA.104.526293. [DOI] [PubMed] [Google Scholar]
- 26.Ueda H, Nakamura T, Matsumoto K, Sawa Y, Matsuda H, Nakamura T. A potential cardioprotective role of hepatocyte growth factor in myocardial infarction in rats. Cardiovasc Res. 2001;51:41–50. doi: 10.1016/s0008-6363(01)00272-3. [DOI] [PubMed] [Google Scholar]
- 27.Miyagawa S, Sawa Y, Taketani S, et al. Myocardial regeneration therapy for heart failure: hepatocyte growth factor enhances the effect of cellular cardiomyoplasty. Circulation. 2002;105:2556–61. doi: 10.1161/01.cir.0000016722.37138.f2. [DOI] [PubMed] [Google Scholar]
- 28.Buerke M, Murohara T, Skurk C, Nuss C, Tomaselli K, Lefer AM. Cardioprotective effect of insulin-like growth factor I in myocardial ischemia followed by reperfusion. Proc Natl Acad Sci U S A. 1995;92:8031–5. doi: 10.1073/pnas.92.17.8031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Detillieux KA, Sheikh F, Kardami E, Cattini PA. Biological activities of fibroblast growth factor-2 in the adult myocardium. Cardiovasc Res. 2003;57:8–19. doi: 10.1016/s0008-6363(02)00708-3. [DOI] [PubMed] [Google Scholar]
- 30.Christov C, Chretien F, Abou-Khalil R, et al. Muscle satellite cells and endothelial cells: close neighbors and privileged partners. Mol Biol Cell. 2007;18:1397–409. doi: 10.1091/mbc.E06-08-0693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pouzet B, Vilquin JT, Hagege AA, et al. Factors affecting functional outcome after autologous skeletal myoblast transplantation. Ann Thorac Surg. 2001;71:844–50. doi: 10.1016/s0003-4975(00)01785-9. discussion 850-1. [DOI] [PubMed] [Google Scholar]
- 32.Tambara K, Sakakibara Y, Sakaguchi G, et al. Transplanted skeletal myoblasts can fully replace the infarcted myocardium when they survive in the host in large numbers. Circulation. 2003;108 1:II259–63. doi: 10.1161/01.cir.0000087430.17543.b8. [DOI] [PubMed] [Google Scholar]
- 33.McConnell PI, del Rio CL, Jacoby DB, et al. Correlation of autologous skeletal myoblast survival with changes in left ventricular remodeling in dilated ischemic heart failure. J Thorac Cardiovasc Surg. 2005;130:1001. doi: 10.1016/j.jtcvs.2005.02.030. [DOI] [PubMed] [Google Scholar]
- 34.Reinecke H, Minami E, Poppa V, Murry CE. Evidence for fusion between cardiac and skeletal muscle cells. Circ Res. 2004;94:e56–60. doi: 10.1161/01.RES.0000125294.04612.81. [DOI] [PubMed] [Google Scholar]
- 35.Gnecchi M, He H, Noiseux N, et al. Evidence supporting paracrine hypothesis for Akt-modified mesenchymal stem cell-mediated cardiac protection and functional improvement. Faseb J. 2006;20:661–9. doi: 10.1096/fj.05-5211com. [DOI] [PubMed] [Google Scholar]
- 36.Jiang S, Haider H, Idris NM, Salim A, Ashraf M. Supportive interaction between cell survival signaling and angiocompetent factors enhances donor cell survival and promotes angiomyogenesis for cardiac repair. Circ Res. 2006;99:776–84. doi: 10.1161/01.RES.0000244687.97719.4f. [DOI] [PubMed] [Google Scholar]
- 37.Kim C, Li RK, Li G, Zhang Y, Weisel RD, Yau TM. Effects of cell-based angiogenic gene therapy at 6 months: persistent angiogenesis and absence of oncogenicity. Ann Thorac Surg. 2007;83:640–6. doi: 10.1016/j.athoracsur.2006.09.044. [DOI] [PubMed] [Google Scholar]
- 38.Kajstura J, Leri A, Finato N, Di Loreto C, Beltrami CA, Anversa P. Myocyte proliferation in end-stage cardiac failure in humans. Proc Natl Acad Sci U S A. 1998;95:8801–5. doi: 10.1073/pnas.95.15.8801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Beltrami AP, Urbanek K, Kajstura J, et al. Evidence that human cardiac myocytes divide after myocardial infarction. N Engl J Med. 2001;344:1750–7. doi: 10.1056/NEJM200106073442303. [DOI] [PubMed] [Google Scholar]
- 40.Schuster MD, Kocher AA, Seki T, et al. Myocardial neovascularization by bone marrow angioblasts results in cardiomyocyte regeneration. Am J Physiol Heart Circ Physiol. 2004;287:H525–32. doi: 10.1152/ajpheart.00058.2004. [DOI] [PubMed] [Google Scholar]
- 41.Dai Y, Xu M, Wang Y, Pasha Z, Li T, Ashraf M. HIF-1alpha induced-VEGF overexpression in bone marrow stem cells protects cardiomyocytes against ischemia. J Mol Cell Cardiol. 2007;42:1036–44. doi: 10.1016/j.yjmcc.2007.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Van Belle E, Witzenbichler B, Chen D, et al. Potentiated angiogenic effect of scatter factor/hepatocyte growth factor via induction of vascular endothelial growth factor: the case for paracrine amplification of angiogenesis. Circulation. 1998;97:381–90. doi: 10.1161/01.cir.97.4.381. [DOI] [PubMed] [Google Scholar]
- 43.Xin X, Yang S, Ingle G, et al. Hepatocyte growth factor enhances vascular endothelial growth factor-induced angiogenesis in vitro and in vivo. Am J Pathol. 2001;158:1111–20. doi: 10.1016/S0002-9440(10)64058-8. [DOI] [PMC free article] [PubMed] [Google Scholar]