Abstract
Surface architecture of pharmaceutical nanocarriers (using polymeric micelles as an example) and the length of the spacer group through which specific ligand is attached to the carrier surface determine the interaction of ligand-bearing nanocarrier with cells. We have prepared surface-modified polyethyleneglycol–phosphatidylethanolamine (PEG–PE) micelles containing TATp attached to PEG–PE with a PEG block longer or shorter (TATp–PEG1000–PE or TATp–PEG3400–PE) than the PEG block in the main micelle-forming material (PEG750–PE and/or PEG2000–PE). The length of the PEG spacer in TATp–PEG–PE should allow for a non-hindered interaction of TATp with the cell surface, but it should not be too long to allow for the conformational “folding in” of TATp moiety inside the PEG globule making it unable to interact with the cells. The “folding in” of the ligand attached to an unnecessary long PEG spacer was further supported by the fluorescence resonance energy transfer (FRET) study between fluorescently labeled lipid 1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine-N-(7-nitro-2-1,3-benzoxadiazol-4-yl) (NBD-PE) inserted into the core of PEG750–PE micelles and micelle-incorporated rhodamine-labeled TATp–PEG–PE. Micelles containing rhodamine-labeled TATp–PEG–PE with the longest PEG spacer (3400 Da) demonstrated strongly enhanced quenching of NBD-PE fluorescence with rhodamine-TATp confirming the “folding in” of TATp moiety into PEG globule bringing it closer to the micelle core-incorporated NBD.
Keywords: Polymeric micelles, polyethyleneglycol–phosphatidylethanolamine, cell-penetrating peptide, TAT peptide, intracellular delivery
Introduction
Various in vitro and in vivo studies have proved the potential of cell-penetrating peptides (CPPs), including TAT peptide (TATp) and oligoarginines, for the intracellular delivery of different cargoes. TATp-mediated cytoplasmic uptake of polymers (Nori et al. 2003; Hyndman et al. 2004), bacteriophages (Paschke et al. 2005), plasmid DNA (Torchilin et al. 2001, 2003; Kleemann et al. 2005), magnetic nanoparticles (Dodd et al. 2001; Zhao et al. 2002; Nitin et al. 2004), liposomes (Cryan et al. 2006; Sawant et al. 2006; Gupta et al. 2007), and micelles (Sawant et al. 2006; Sethuraman et al. 2007, 2008) has been reported. Successful intracellular delivery requires direct contact between the surface-attached CPPs on the pharmaceutical nanocarrier and the cell surface (Torchilin et al. 2001; Levchenko et al. 2003). Actually, this is true and generally applicable to any ligand-bearing carrier for targeted delivery. Reasonable assumption is usually made that such ligand should be sufficiently distant from the carrier surface or the surface of any coating applied to the carrier (such as sterically protecting PEG coating) to allow for the required unhindered interaction with the cell. A natural practical consequence from such assumption is using long spacer groups to attach ligands to nanocarriers. To which extent this assumption is correct and how long the spacer arm should really be?
We prepared and studied the in vitro PEG–PE polymeric micelles modified with TATp moieties. TATp–PEG–PE conjugates with different PEG-spacer lengths were used allowing or not allowing for direct TATp-to-cell interaction. The specific objective of the present work was to study the effect of PEG-spacer length in TATp–PEG–PE on the TATp-mediated uptake of the nanocarriers (micelles) by cells.
Materials and methods
Materials
Cell line, BT20 (human breast adenocarcinoma), was purchased from the ATCC (Manassas, VA, USA). Cell culture media, Dulbecco’s modified Eagle’s medium (DMEM), heat-inactivated fetal bovine serum (FBS), and concentrated solutions of sodium pyruvate and penicillin/streptomycin stock solutions were purchased from Cellgro (Herndon, VA, USA). TAT-peptide (TATp-NH2) (11-mer: TyrGlyArgLysLysArgArgGlnArgArgArg) and TAT-cysteine peptide (TATp-Cys) (12-mer: CysTyrGlyArgLysLysArgArgGlnArgArgArg) were synthesized by Tufts University Core Facility (Boston,MA,USA). 1,2-Distearoyl-sn-glycero-3-phos-phoethanolamine-N-[methoxy (poly(ethylene glycol))-750 or -2000] (PEG750–PE or PEG2000–PE), 1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine-N-(lissamine rhodamine B sulfonyl) (Rh-PE), 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine (DOPE), and 1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine-N-(7-nitro-2-1,3-benzoxadiazol-4-yl) (NBD-PE) were from Avanti Polar Lipids (Alabaster, AL, USA). Carboxytetramethyl rhodamine succinimidyl ester (Rh-NHS) was from Pierce (Rockford, IL, USA). All other chemicals were analytical grade preparations from Sigma (St Louis, MO, USA).
Synthesis of TATp–PEG–PE conjugates
TATp–PEG1000–PE was synthesized using a heterobifunctional NHS–PEG1000–Mal derivative, DOPE, and TATp-cysteine as in Kale et al. (2007a, 2007b). TATp–PEG3400–PE was synthesized by conjugating TATp to p-nitrophenyl-PEG3400–PE (pNP–PEG3400–PE; Torchilin et al. 2001). In brief, a solution of 5mg of TATp-NH2 in 1ml chloroform supplemented with 10 µl of triethylamine (TEA) was incubated with 12 mg of pNP–PEG3400–PE in 0.6 ml chloroform overnight with stirring at room temperature (RT). Organic solvents were removed by rotary evaporation and freeze-drying. To the resultant film, 1ml of deionized water was added and vortexed to remove the film from the flask. To purify the TATp–PEG3400–PE from unconjugated TATp–NH2 and pNP, the solution was dialyzed using cellulose ester dialysis membrane MWCO 2000 (Spectrum Medical Industries, Rancho Dominguez, CA, USA) at RT against water.
Synthesis of rhodamine-labeled TATp–PEG–PE for fluorescence resonance energy transfer (FRET) studies
For labeling TATp–PEG1000–PE or TATp–PEG3400–PE with rhodamine, 0.34µmol of TATp–PEG–PE in 2ml dry chloroform was mixed with 5 molar excess of Rh-NHS and 5µl of TEA. The mixture was stirred for 24 h at RT under argon. Organic solvent was removed using rotary evaporator and freeze-drying to forma thin film, which was rehydrated and vortexed to ensure the formation of Rh-TATp–PEG–PE suspension. The Rh-TATp–PEG–PE conjugate was purified by dialysis against water using MWCO 1000 dialysis bag followed by freeze-drying to a constant weight. The product was dissolved in dry chloroformat 2mg/ml and stored under argon at −80°C.
Preparation of micelles
For micelle preparation, a mixture of PEG–PE (750 and/or 2000 Da PEG blocks) and TATp–PEG–PE (1000 or 3400 Da PEG blocks) was prepared in chloroform at a molar ratio of 97.5:2.5. Chloroform was removed by rotary evaporation and freeze-drying. The film was hydrated with HEPES-buffered saline (HBS, pH 7.4) at room temperature and vortexed for 5 min. For cell interaction study, 0.5 mol% of the fluorescent probe, Rh-PE, was added to the micelle composition. For FRET experiments, micelles were prepared with varying quantities of Rh-TATp–PEG–PE and 1 mol% of the micelle core-incorporated fluorescent probe, NBD-PE (for compositions see Table I). Micelle size was measured using a Coulter N4 Plus Submicron Particle Analyzer (Coulter Corporation, Miami, FL, USA).
Table I.
Compositions of micelles used in the study.
| Micelle composition (mol%) | ||||
|---|---|---|---|---|
| Sr. No. | PEG–PE main component | Fluorescent label | PEG–PE second component | TAT-component |
| 1. | PEG750–PE (99.5) | Rh-PE (0.5) | – | – |
| 2. | PEG750–PE (97.0) | Rh-PE (0.5) | – | TATp–PEG1000–PE (2.5) |
| 3. | PEG750–PE (97.0) | Rh-PE (0.5) | – | TATp–PEG3400–PE (2.5) |
| 4. | PEG750–PE (99.0–95.0) | NBD-PE (1.0) | – | Rh-TATp–PEG1000–PE (0–4) |
| 5. | PEG750–PE (99.0–97.0) | NBD-PE (1.0) | – | Rh-TATp–PEG3400–PE (0–2) |
| 6. | PEG750–PE (67.0) | Rh-PE (0.5) | PEG2000–PE (30) | TATp–PEG1000–PE (2.5) |
| 7. | PEG750–PE (47.0) | Rh-PE (0.5) | PEG2000–PE (50) | TATp–PEG1000–PE (2.5) |
| 8. | PEG2000–PE (99.5) | Rh-PE (0.5) | – | – |
| 9. | PEG2000–PE (97.0) | Rh-PE (0.5) | – | TATp–PEG1000–PE (2.5) |
| 10. | PEG2000–PE (97.0) | Rh-PE (0.5) | – | TATp–PEG3400–PE (2.5) |
Interaction of TATp-nanocarriers with cells
BT20 cells were grown on cover slips placed in six-well tissue culture plates. After the cells reached a confluence of 60–70%, the plates were washed with DMEM and the cells were treated with various Rh-PE-labeled micelle (0.2 mg/ml of the micelle-forming material) samples (with and without TATp) in DMEM. After 1 h, the medium was removed and the plates were washed with sterile PBS (pH 7.4). Individual cover slips were mounted cell-side down onto fresh glass slides with PBS. Cells were viewed with a Nikon Eclipse E400 microscope under bright light or under epifluorescence with rhodamine/-TRITC filter.
FRET studies of TATp behavior on the surface
FRET between two fluorescent probes was used to follow the spatial behavior of TATp attached to a spacer arm. FRET results in the quenching of the donor fluorescence. Since the extent of FRET depends on the distance between the donor and the acceptor molecules, the interaction between the “outer” peptide-attached and core lipid-anchored probes can be monitored following the changes in fluorescence intensities. The fluorescent lipid NBD-PE (1 mol%) and Rh-TATp–PEG–PE (varying quantities) were incorporated into the micelles. The NBD-PE fluorescence intensity at the excitation wavelength of 460nm and the emission wavelength of 534nm was monitored at the fixed lipid concentration of 1mg/ml using F-2000 fluorescence spectrometer (Hitachi, Japan).
Results and discussion
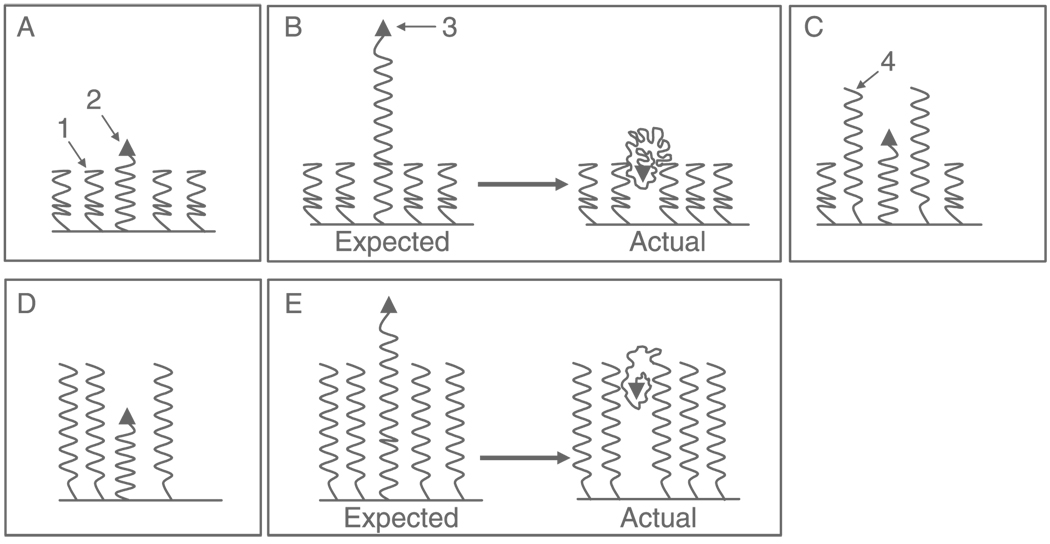
It was assumed that for efficient interaction of the nanocarrier-attached TATp with the cell, TATp moiety should be located “above” the carrier surface. Thus, in case of PEG–PE micelles, TATp was attached to the distal PEG tip of some PEG–PE moieties, and these moieties contained longer PEG block than the main body of micelle-forming PEG–PE unimers. Micelles were prepared using PEG750–PE or PEG2000–PE or the mixture of PEG750–PE and PEG2000–PE as main micelle-forming components with the addition (depending on the micelle composition) of either TATp–PEG1000–PE or TATp–PEG3400–PE. In some samples, micelles have also been prepared with TATp attached to the micelle core via a shorter PEG spacer (1000 Da) than PEG moiety in micelle-forming PEG–PE (2000 Da); see Table I and Figure 1 for micelle compositions and hypothetical architecture. All micelles had the size within the 10–20 nm range.
Figure 1.
Schematic representation of the TATp-containing micelles: (a) PEG750–PE: TATp–PEG1000–PE micelles, (b) PEG750–PE: TATp–PEG3400–PE micelles, (c) PEG750–PE: PEG2000–PE: TATp–PEG1000–PE micelles, (d) PEG2000–PE: TATp–PEG1000–PE micelles, and (e) PEG2000–PE: TATp–PEG3400–PE micelles. 1, PEG750–PE; 2, TATp–PEG1000–PE; 3, TATp–PEG3400–PE; 4, PEG2000–PE.
Preparations with TATp located “above” PEG blocks of the micelle-forming PEG–PE were expected to demonstrate an increased cell uptake due to assumed unhindered contact of TATp with cells.
TATp-mediated interaction of nanocarriers with cells in vitro
The importance of unhindered contact of TATp with cells for successful intracellular delivery of TATp-modified nanocarriers has already been demonstrated by us with the “smart” pH-sensitive micelles and liposomes (Sawant et al. 2006). Such micelles were composed of PEG750–PE, pH-sensitive detachable PEG2000–Hz–PE, and TATp–PE. These micelles, when intact (at pH 8.0), exhibited minimal cell uptake due to the shielding of the TATp function. Their incubation at a lowered pH (5.0) facilitating the removal of PEG2000 blocks due to breaking of the pH-sensitive hydrazone bond resulted in increasingly strong interaction of the micelles with the cells because of deshielding of the TATp function. However, the role of the length of the spacer group via which TATp was attached to micelles was not specifically investigated.
The interaction of different micelle preparations with BT20 cells is shown in Figure 2. Micelles composed of PEG750–PE or PEG2000–PE demonstrated limited uptake by the cells (Figure 2a,b); however, the addition of the TATp–PEG1000–PE component to PEG750–PE micelles resulted in the expected strong interaction with the cells compared with plain micelles (Figure 2c). Gradual replacement of PEG750–PE in the micelle composition with PEG2000–PE component (at the same molar fraction of TATp–PEG1000–PE) resulted in gradual decrease in TATp-mediated micelle interaction with cells because of eventual shielding of TATp function by the longer PEG chains (Figure 2d–f). This interaction remains significant for the micelles containing 30 mol% of PEG2000–PE (Figure 2d), but decreases dramatically for the micelles containing 50% or composed of only PEG2000–PE (Figure 2e,f).
Figure 2.
In each panel, the left figure shows the bright field and the right figure shows the fluorescent microscopy of BT20 cells treated with (a) Rh-PE:PEG750–PE micelles, (b) Rh-PE: PEG2000–PE micelles, (c) Rh-PE: PEG750–PE: TATp–PEG1000–PE micelles, (d) Rh-PE: PEG750–PE: PEG2000–PE (30 mol%): TATp–PEG1000–PE micelles, (e) Rh-PE: PEG750–PE: PEG2000–PE (50 mol%): TATp–PEG1000–PE micelles, (f) Rh-PE: PEG2000–PE: TATp–PEG1000–PE micelles, (g) Rh-PE: PEG750–PE: TATp–PEG3400–PE micelles, and (h) Rh-PE: PEG2000–PE: TATp–PEG3400–PE micelles. Magnification 40 × objective.
Somewhat different pattern was observed for the micelles with TATp–PEG3400–PE component (same molar fraction as TATp–PEG1000–PE). Such micelles (built of PEG750–PE or PEG2000–PE main component) demonstrated minimal cell interaction similar to that for the plain micelles (Figure 2g,h). This result looks quite surprising since we assumed that the TATp function attached to a long PEG spacer should be unhindered and provide easy contact of TATp moiety with the cell surface.
The most probable explanation of this phenomenon comes from the behavior of soluble polymers in aqueous solutions. Soluble polymers with a flexible main chain (and PEG is a very good example of such polymers) in aqueous solutions have a strong tendency to fold into a statistical globule rather than maintain a stretched conformation (Jannink et al. 1990). In this case, TATp moiety attached to the relatively long PEG3400 chain has a good chance to be “folded into” the PEG globule thus being removed from the “above-the-surface” position closer to the inner part of the nanocarrier (micelle core in this case). As a result of such “self-shielding”, TATp moiety loses its ability to directly interact with the cell surface. At the same time, shorter PEG1000 spacer does not have the length sufficient for the folding into the statistical globule, and TATp moiety always remain protruded from the nanocarrier surface and available for the interaction with the cell surface (unless it is “shielded” by surrounding longer PEG chains). To check this hypothesis, we have performed fluorescence quenching experiments.
FRET experiments
To gain information on the “folding” of TATp from the “above-the-surface” position closer to the micelle core when attached to a longer PEG spacer using FRET approach, we have utilized the NBD-PE probe (with no spacer between PE and NBD) as the micelle core-incorporated donor probe (it should be incorporated into the core via its lipid anchor with the fluorescent part of the molecule located also in the core or very close to it). Rhodamine attached to the TATp moiety of TATp–PEG–PE conjugate served as acceptor probe. One can expect that with no PEG-spacer folding, Rh-labeled TATp should remain distant from the core-incorporated NBD, while if the PEG spacer is long enough to allow for the chain folding, TATp moiety could get inserted into the micelle corona, and TATp-attached Rh group should come closer to the NBD group and cause the higher quenching of NBD fluorescence.
NBD–PE–labeled (fixed 1 mol%) PEG750–PE micelles containing varying quantities of Rh-TATp– PEG1000–PE or Rh-TATp–PEG3400–PE were used. The NBD–PE fluorescence intensity was measured at the fixed concentration of the micelle-forming components of 1 mg/ml. The data in Figure 3 clearly show that with the increase in TATp–PEG–PE content, micelles containing Rh-TATp–PEG3400–PE demonstrate significant quenching of NBD-PE fluorescence compared with that in case of Rh-TATp–PEG1000–PE-containing micelles.
Figure 3.
Fluorescence intensity of NBD-PE containing micelles with rhodamine-labeled TATp–PEG3400–PE or TATp–PEG1000–PE, n = 3, ★p < 0.05.
As was already mentioned, the extent of FRET strongly depends on the distance between the donor and the acceptor molecules, and theoretically, assuming a fully extended conformation of PEG spacer at any length, the NBD-PE fluorescence should be less affected in case of Rh-labeled TATp attached to longer PEG3400–PE than to shorter PEG1000–PE. The fact that we observe quite opposite result clearly confirms the folding phenomenon of longer PEG spacer bringing TATp moiety (and fluorescent tag attached to it) into the inner part of the micelle corona more close to the micelle core compartment where the NBD-PE is located. This namely phenomenon affects also the interaction of such micelles with the cells.
Our result shows the importance of the proper choice of the length of spacer group for the attachment of various specific ligands to the surface of pharmaceutical nanocarriers to allow for the optimal surface architecture and thus the effective interaction with target cells.
Acknowledgment
This work was supported by the NIH grant RO1 CA121838 to Vladimir P. Torchilin.
Abbreviations
- CPP
cell-penetrating peptide
- FRET
fluorescence resonance energy transfer
- PEG–PE
polyethyleneglycol–phosphatidylethanolamine
- PEG750–PE or PEG2000–PE
1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy (poly(ethylene glycol))-750 or -2000]
- pNP–PEG3400–PE
p-nitrophenyl–PEG3400–PE
- DOPE
1,2-dioleoyl-sn-glycero-3-phosphoethanolamine
- TATp–PEG1000–PE or TATp–PEG3400–PE
TAT peptide conjugated to 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy (poly(ethylene glycol))-1000 or -3400]
- Rh-TATp–PEG1000–PE or Rh-TATp–PEG3400–PE
rhodamine-labeled TAT peptide conjugated to 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy (poly(ethylene glycol))-1000 or -3400]
- NBD-PE
1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine-N-(7-nitro-2-1,3-benzoxadiazol-4-yl)
- Rh-PE
1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine-N-(lissamine rhodamine B sulfonyl)
- Rh-NHS
carboxytetramethyl rhodamine succinimidyl ester
Footnotes
Declaration of interest: The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
References
- Cryan SA, Devocelle M, Moran PJ, Hickey AJ, Kelly JG. Increased intracellular targeting to airway cells using octaargi-nine-coated liposomes: In vitro assessment of their suitability for inhalation. Mol Pharm. 2006;3(2):104–112. doi: 10.1021/mp050070i. [DOI] [PubMed] [Google Scholar]
- Dodd CH, Hsu HC, Chu WJ, Yang P, Zhang HG, Mountz JD, Jr, Zinn K, Forder J, Josephson L, Weissleder R, Mountz JM, Mountz JD. Normal T-cell response and in vivo magnetic resonance imaging of T cells loaded with HIV transactivator-peptide-derived superparamagnetic nanoparticles. J Immunol Methods. 2001;256(1–2):89–105. doi: 10.1016/s0022-1759(01)00433-1. [DOI] [PubMed] [Google Scholar]
- Gupta B, Levchenko TS, Torchilin VP. TAT peptide-modified liposomes provide enhanced gene delivery to intracranial human brain tumor xenografts in nude mice. Oncol Res. 2007;16(8):351–359. doi: 10.3727/000000006783980946. [DOI] [PubMed] [Google Scholar]
- Hyndman L, Lemoine JL, Huang L, Porteous DJ, Boyd AC, Nan X. HIV-1 Tat protein transduction domain peptide facilitates gene transfer in combination with cationic liposomes. J Control Release. 2004;99(3):435–444. doi: 10.1016/j.jconrel.2004.07.023. [DOI] [PubMed] [Google Scholar]
- Jannink G, Cloizeaux Jd. Polymers in solution. J Phys: Condens Matter. 1990;2:1–24. [Google Scholar]
- Kale AA, Torchilin VP. Design, synthesis, and characterization of pH-sensitive PEG-PE conjugates for stimuli-sensitive pharmaceutical nanocarriers: The effect of substitutes at the hydrazone linkage on the pH stability of PEG-PE conjugates. Bioconjug Chem. 2007a;18(2):363–370. doi: 10.1021/bc060228x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kale AA, Torchilin VP. Enhanced transfection of tumor cells in vivo using “Smart” pH-sensitive TAT-modified pegylated liposomes. J Drug Targeting. 2007b;15(7–8):538–545. doi: 10.1080/10611860701498203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleemann E, Neu M, Jekel N, Fink L, Schmehl T, Gessler T, Seeger W, Kissel T. Nano-carriers for DNA delivery to the lung based upon a TAT-derived peptide covalently coupled to PEG–PEI. J Control Release. 2005;109(1–3):299–316. doi: 10.1016/j.jconrel.2005.09.036. [DOI] [PubMed] [Google Scholar]
- Levchenko TS, Rammohan R, Volodina N, Torchilin VP. Tat peptide-mediated intracellular delivery of liposomes. Methods Enzymol. 2003;372:339–349. doi: 10.1016/s0076-6879(03)72019-9. [DOI] [PubMed] [Google Scholar]
- Nitin N, LaConte LE, Zurkiya O, Hu X, Bao G. Functionalization and peptide-based delivery of magnetic nanoparticles as an intracellular MRI contrast agent. J Biol Inorg Chem. 2004;9(6):706–712. doi: 10.1007/s00775-004-0560-1. [DOI] [PubMed] [Google Scholar]
- Nori A, Jensen KD, Tijerina M, Kopeckova P, Kopecek J. Subcellular trafficking of HPMA copolymer–Tat conjugates in human ovarian carcinoma cells. J Control Release. 2003;91(1–2):53–59. doi: 10.1016/s0168-3659(03)00213-x. [DOI] [PubMed] [Google Scholar]
- Paschke M, Hohne W. A twin-arginine translocation (Tat)-mediated phage display system. Gene. 2005;350(1):79–88. doi: 10.1016/j.gene.2005.02.005. [DOI] [PubMed] [Google Scholar]
- Sawant RM, Hurley JP, Salmaso S, Kale A, Tolcheva E, Levchenko TS, Torchilin VP. “SMART” drug delivery systems: Double-targeted pH-responsive pharmaceutical nanocarriers. Bioconjug Chem. 2006;17(4):943–949. doi: 10.1021/bc060080h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sethuraman VA, Bae YH. TAT peptide-based micelle system for potential active targeting of anti-cancer agents to acidic solid tumors. J Control Release. 2007;118(2):216–224. doi: 10.1016/j.jconrel.2006.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sethuraman VA, Lee MC, Bae YH. A biodegradable pH-sensitive Micelle system for targeting acidic solid tumors. Pharm Res. 2008;25(3):657–666. doi: 10.1007/s11095-007-9480-4. [DOI] [PubMed] [Google Scholar]
- Torchilin VP, Levchenko TS, Lukyanov AN, Khaw BA, Klibanov AL, Rammohan R, Samokhin GP, Whiteman KR. p-Nitrophenylcarbonyl-PEG–PE–liposomes: Fast and simple attachment of specific ligands, including monoclonal antibodies, to distal ends of PEG chains via p-nitrophenylcarbonyl groups. Biochim Biophys Acta Biomembr. 2001;1511(2):397–411. doi: 10.1016/s0005-2728(01)00165-7. [DOI] [PubMed] [Google Scholar]
- Torchilin VP, Levchenko TS, Rammohan R, Volodina N, Papahadjopoulos-Sternberg B, D’Souza GG. Cell transfection in vitro and in vivo with nontoxic TAT peptide–liposome–DNA complexes. Proc Natl Acad Sci U S A. 2003;100(4):1972–1977. doi: 10.1073/pnas.0435906100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torchilin VP, Rammohan R, Weissig V, Levchenko TS. TAT peptide on the surface of liposomes affords their efficient intracellular delivery even at low temperature and in the presence of metabolic inhibitors. Proc Natl Acad Sci U S A. 2001;98(15):8786–8791. doi: 10.1073/pnas.151247498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao M, Kircher MF, Josephson L, Weissleder R. Differential conjugation of tat peptide to superparamagnetic nanoparticles and its effect on cellular uptake. Bioconjug Chem. 2002;13(4):840–844. doi: 10.1021/bc0255236. [DOI] [PubMed] [Google Scholar]